1. Introduction
Infected wounds represent a complex, non-trivial, problem which seriously jeopardizes the health and life of patients. One of the most frequent problems is a chronic ulcer with deep tissue damage in patients with diabetes, connected with abnormal fibroblast and keratinocyte proliferation, reduced cell migration and decreased angiogenesis, which can lead to impairment of vascularization, delayed wound contraction and subsequent formation of non-healing diabetic wounds [
1]. Not only diabetic wounds but also leg ulcers, arterial insufficiency, pressure ulcers and burns impose substantial morbidity and mortality, deeply affecting quality of life, with high economic burden [
2]. Generally, wounds can be divided into acute and chronic [
3]. When chronic wounds were analyzed in a mouse model, a huge diversity of microorganisms formatting biofilm (for example,
Enterobacter cloacae,
Streptococcus thermophilus,
Propionibacterium acnes) and without biofilm formation (
Achromobacter xylosoxidans) were observed. Biofilms as sessile microbial consortia established in a three-dimensional structure are an important strategy implemented by microorganisms to survive in sometimes harsh environmental conditions [
4]. Biofilms within the in vitro environment only are often referred to as immature when they have been growing for less than 24 h and mature when they have been growing for more than 24 h [
5]. The ratio between different biofilm and non-biofilm creating bacterial types in a wound is dependent on type and time of treatment [
6]. Thus, because a variety of different bacterial species live in the wounds, there is a widely acknowledged need for new antibacterial agents to address the global increase in resistance [
7]. The level of resistance, especially against traditional antibiotics (for example, methicillin-resistant
Staphylococcus aureus [
8], penicillin-resistant
Enterococcus faecalis [
9] or multiresistant
Mycobacterium tuberculosis [
10]) is still increasing, and the rising incidence of antimicrobial resistance among pathogenic bacteria is one of the greatest healthcare challenges facing humanity today [
11]. Thus, new approaches to overcome bacterial resistance are being tried, comprising by using plant extracts [
12] or a combination of antibiotics and other antibacterial materials [
13], especially nanomaterials, which comprise unary [
14], binary (AgAu, AgPt NPs) [
15,
16] or multicomponent materials (CdZnSe-CdZnS nanoalloy) [
17]. Nanotechnology is nowadays a flourishing scientific field, which associates nanoparticles with extraordinary functions and size-dependent physicochemical properties, differing significantly from the macroscopic forms of these elements. Nanomaterials comprise nanoparticles (NPs), which can be divided into inorganic and organic nanoparticles and nanocomposites, which can be grouped into the porous material, colloids, copolymers and gels. Finally, the applications of nanoparticles and nanocomposites in scaffolds and coatings are via hydrogels, nanofibers and films (
Figure 1). Over the years, inorganic nanoparticles have been explored extensively in nanomedicine [
18], electronics [
19], drug delivery [
20], biomedical devices [
21], food sectors as package material [
22,
23], and dentistry such as: prosthetic, restoration, endodontic, orthodontic, periodontal and dental implant treatment. In addition, an improvement of mechanical properties of dental materials; inhibition of adhesion and bacteria growth of
Escherichia coli,
Streptococcus mutans,
Enterococcus faecalis,
Pseudomonas aeruginosa,
S. aureus and the fungus
Candida albicanshave been observed [
24], etc.
Among the several inorganic NPs, silver NPs (AgNPs), due to their novel chemical, biological and physical properties as compared to their bulk form, have attracted the interest of researchers from the whole academic sphere. AgNPs have typical physical and chemical properties—for example, high thermal and electrical conductivity [
26], surface-enhanced Raman scattering [
27], chemical stability [
28], catalytic activity [
29] and non-linear optical behavior [
30]. These properties take AgNPs to the top of the priority list, and they can be used in inks, in electronics and for medical purposes. Additionally, AgNPs are commonly known for their antibacterial, antifungal and antiviral properties. Mechanisms of effectivity comprise the disrupting of cell membranes, interaction with sulfhydryl groups of proteins, inactivation of respiratory enzymes, DNA modification, etc. (
Figure 2). Microorganisms, however, have also mechanisms to adapt and to resist to metals (for example, reduced uptake, through efflux, chemical modification of metals, etc.) [
31].
Thus, there is a need for a better understanding of these parameters on the efficacy and toxicity of AgNPs. Wide variety in size, concentration, shape, materials, polymers and surfaces of AgNPs are commonly being used [
32].
The antimicrobial properties of AgNPs have been described and proven in various in vitro and in vivo studies, and their applications in food packaging, soaps, cosmetics, fabrics and wound dressing materials have also been reported [
33]. There are a number of methods for antimicrobial susceptibility testing (AST) of bacteria, which can be divided into diffusion methods and dilution methods [
34,
35]. The most used diffusion methods are the agar disk-diffusion method and antimicrobial gradient method (Etest). The most used dilution methods are the broth dilution method and agar dilution method [
36]. The broth dilution method can be further divided into the broth macrodilution method and broth microdilution method [
36]. The broth macrodilution method has been standardized by the Clinical and Laboratory Standards Institute (CLSI) for testing bacteria that grow aerobically [
37]. The broth microdilution method is standardized by CLSI for both anaerobic [
38] and aerobic bacteria [
39]. As a representative example of the agar plate disk diffusion test [
40] and the broth microdilution test (a measurement of growth curves) [
41], our own experimental results for green-synthesized nanoparticles are shown in
Figure 3.
AgNPs are also used in water filter membranes for point-of-care water disinfection, utilizing their antimicrobial properties [
42]. The AgNPs can be used in all physical forms—for example, in colloidal form (in enamel coating and paints), liquid form (cosmetics) and solid form (blended with polymer scaffolds). For wounds, different types of treatment with silver and AgNPs were proposed and they are shown in
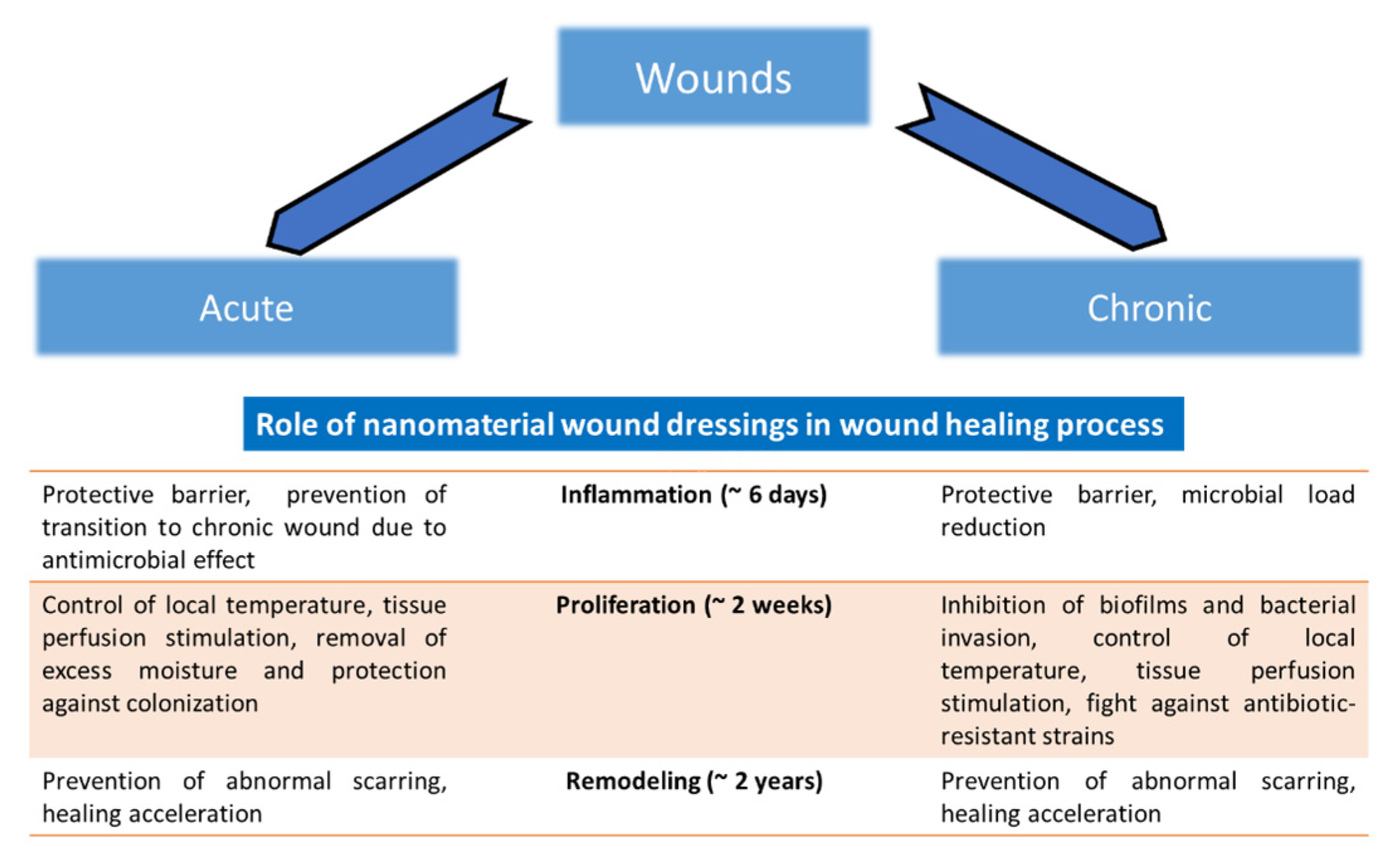
Figure 4. Application of nanomaterials on the wound can be direct, in the form of powdered AgNPs or their solutions. The second possibility is the use of non-compact materials for wound treatment as water-soluble polymers and hydrogels. The third possibility is the use of solid polymers (fibers, membranes and films). The role of nanomaterial wound dressings in the wound healing process, in general, is shown in
Figure 5. One problematic aspect of AgNPs wound dressings is that there are reported cases of bacterial resistance to AgNPs. For example, resistance to AgNPs attributed to
sil genes has been reported in clinical isolates of
Klebsiella pneumoniae and
Enterobacter cloacae from burn cases [
43]. Thus, the continuous development of a new combination of nanomaterials, antibiotics, polymers, methods of incorporation and types of reduction of silver to obtain various shapes of NPs is essentially necessary.
3. Synthesis of AgNPs
This review aims to conclude the newest findings of the antibacterial effect of AgNPs connected with covering materials for wound dressing and healing. As mentioned above, four types of AgNPs synthesis are described, namely chemical, green, irradiation and thermal. In this section, the basic characteristics of each type of synthesis are briefly described. In chemical synthesis, the Turkevich method and Brust–Schiffrin synthesis are mainly used [
43]. The Turkevich method is based on the reduction of the boiling solution of silver salt with a solution of citrate salt. The Brust–Schiffrin method is mainly used for golden nanoparticles, but applications for other metals were also described [
44]. Stabilization of AgNPs synthesis is ensured by using surfactants (thiols, amines, acids and alcohols) or polymers (poly(vinyl alcohol), poly(vinylpyrrolidone), poly(ethylene glycol), poly(methacrylic acid) and polymethyl methacrylate) [
45]. Green synthesis represents a low-cost, environmentally friendly way of producing AgNPs without using high temperatures and pressures [
46]. Typically, the plant [
46,
47,
48,
49,
50], fungus [
51,
52,
53] or bacteria extract is mixed with silver ions containing salt (silver nitrate is mainly used [
46,
47,
48,
49,
50,
51,
52,
53]) and bioactive molecules of extracts reduce silver ions to elementary silver. A huge advantage of using plant extracts is the fact that many plants contain health-beneficial compounds, also known from their use in conventional medicine (for example, paclitaxel, vincristine or artemisinin) [
54]. After nucleation, the AgNPs can be precipitated with alcohol [
55] (for example, methanol, ethanol or isopropyl alcohol). Model synthesis is shown in
Figure 7.
The process of reduction of Ag
+ to Ag
0 with biomolecules relates to the high reduction power of the biomolecule cocktail. Depending on the extract composition, the velocity of stirring and time of nucleation of AgNPs, AgNPs with different shapes can be obtained (
Figure 8).
Irradiation synthesis [
56,
57,
58] is connected with radiation of precursors or intermediate products of reaction with electromagnetic radiation with different wavelengths. Only specialized laboratories with a suitable radiation source and in respect to the difficult legislature can achieve this type of synthesis. Jia et al. [
56] prepared gold nanoclusters using triple helix glucan Lentiman via microwave-assisted synthesis and subsequently employed them as seeds for Ag-Au nanoparticle alloy; these nanoparticles increase in size with higher Ag amounts [
56]. Mixed cellulose nanofibrils and AgNPs composites were prepared by UV radiation [
57]. Graphene oxide (GO)-supported AgNPs nanocomposite was prepared via polyvinyl-pyrrolidone (PVP) and isopropanol (IPA)-assisted gamma irradiation [
58].
Thermal synthesis is based on a thermal reduction of silver salt. A representative example is the thermal reduction of AgNO
3 [
59] in an environment of poly(
N-isopropylacryamide-
co-2-acrylamido-2-methylpropane sulfonic acid) (NIPAMSA) to form the hybrid hydrogels with incorporated AgNPs.
4. AgNPs Immobilization into Membrane and Composite Material
Membrane and composite material-immobilized nanoparticles can have a plethora of different functions (e.g., disinfection, catalytic activity). An example which can be mentioned as a composite material created by Liang et al. is a material based on a pre-treated egg-shell membrane with procyanidin to reduce silver ions into nanoparticles incorporated into the membrane structure. This membrane had catalytic activity consisting of the conversion of 4-nitrophenol to 4-aminophenol [
60]. Silver ions reduced with ascorbic acid formed nanoparticles incorporated into polyethersulfone membranes and showed antibacterial activity against
S. aureus,
S. albus and
E. coli when tested on bacterial plates [
61]. Hanif et al. [
62] used a non-toxic and environmentally friendly method based on tannic acid-mediated silver salt layer-by-layer in-situ reductions. This method was used for the preparation of a cellulose-AgNPs composite with uniform and controlled size and distribution of AgNPs for point-of-use water disinfection, which was tested successfully on
E. coli bacteria. In the study published by Dong et al. [
63], casein-coated AgNPs were embedded into acetate-cellulose membrane for control of biofouling. AgNPs effectively suppressed the growth of
Serratia marcescens, and specifically, membranes with AgNPs displayed a decrease in microbial growth by 59–99% after this concentration was used [
63]. Silver nanoparticles incorporated into the reverse osmosis membrane with a diameter of approximately 30 nm significantly reduced (by 64.6%)
Pseudomonas sp. biofilm formation after 14 days of continuous cultivation [
64]. Another study which was proposed by Saraswathi et al. [
65] was based on polydopamine-coated poly(ether imide) membranes with incorporated AgNPs. Membranes showed antibacterial activity against both Gram-negative and Gram-positive bacteria and facilitated the separation of toxic contaminants. In general, it can be concluded that AgNPs can be used in drinking water treatment and as a pseudo-enzymatic catalyst in the reaction. Additionally, AgNPs-incorporated membranes are used for wound treatment; representative examples are shown in
Table 1.
4.1. Chemical-Synthesized AgNPs Incorporated into Membranes and Composite Materials
The bacterial cellulose (BC) membrane has been utilized as a template for the in-situ preparation of AgNPs through the chemical reduction method. The lyophilized cellulose membrane with AgNPs was placed on the
E. coli or
S. aureus grown on an agar plate and incubated at 37 °C for 24 h. The sample derived from maltose with a silver content of 1.06% realized >99.99% killing ratio in the number of viable
E. coli, when cultivated on maltose after a contact period of 18 h. Therefore, 1.2 mg of the silver-containing composite was sufficient to ensure no detectable growth of
E. coli in 0.1 mL of medium with a bacteria concentration of around 108 CFU (colony forming units)/mL [
66].
Levi-Polyachenko et al. [
67] prepared chitosan membrane by the solvent casting method and loaded it with a different concentration of hexagonal AgNPs synthesized through the chemical route to demonstrate the synergistic wound healing activity. The developed hexagonal AgNPs represented a new addition to the chitosan wound dressing materials, as they could facilitate cell proliferation, mitigate bacterial infection and generate mild hyperthermia for the delivery of small drug molecules [
67].
Another chemical method for the synthesis of AgNPs using AgNO
3 as a precursor was reported. The generated nanoparticles were loaded into casted poly(vinylpyrrolidone)-chitosan (PVP-chitosan) membrane. The PVP-chitosan-silver composite film found higher antibacterial activity than both chitosan and AgNO
3 alone. The L929 cell lines were used for a cytotoxicity study and adult male albino rats (140–180 g) were selected for the in vivo wound healing study. The prepared film exhibited higher wound healing efficiency than the cotton gauge, 100% chitosan and other reported chitosan-based dressings [
68].
The chemical reduction of AgNO
3 resulted in the formation of AgNPs, which were loaded into the chitin hydrogel. These hydrogels were lyophilized to obtain the AgNPs-impregnated chitin membrane. These chitin-nanosilver composite scaffolds exhibited good blood clotting ability and were also found to be bactericidal against
S. aureus and
E. coli [
69].
Maneerung et al. isolated BC [
70] produced by
Acetobacter xylinum. It was used as a template for the synthesis of AgNPs through the chemical method. The freeze-dried silver nanoparticle-impregnated BC exhibited strong antimicrobial activity against
E. coli (Gram-negative) and
S. aureus (Gram-positive) bacteria [
70].
4.2. Green-Synthesized AgNPs Incorporated into Membranes and Composite Materials
A composite sponge was made using konjac glucomannan (KGM) as the polymer and loaded with AgNPs that were prepared through a green synthesis method using egg white as a reducing agent. Animal models showed that the KGM-AgNPs composite sponges had effectively accelerated the wound healing process. The histological findings showed that they promoted fibroblast growth and accelerated epithelialization [
71].
The regenerated cellulose and chitosan were blended and made into a solution for film casting in the presence of AgNO
3 as a precursor for AgNPs production through the green reduction method. The tea grains (
Camellia sinensis) were used as a reducing as well as a capping agent for the NPs. Evaluation of the optimized composite film for temporary biological wound dressing materials on the experimental wounds of rats had revealed the significant wound healing of the experimentally treated wounds faster than the control wounds [
72].
The solvent casting method was performed for the preparation of chitosan-polyvinyl alcohol (PVA)-curcumin (CU) composite membrane loaded with AgNPs. The AgNO
3 solution precursor was added to the composite mixture, which led to the formation of AgNPs through the reduction of the precursor by chitosan and PVA. The antimicrobial and antifungal activity of the chitosan-PVA-AgNPs films demonstrated significant effects against
E. coli, Pseudomonas, Staphylococcus, Micrococcus, C. albicans and
P. aeruginosa. To improve the therapeutic efficiency further, CU was encapsulated along with chitosan-PVA-AgNPs nanocomposite films, which showed enormous growth inhibition of
E. coli compared to plain CU and chitosan-PVA-AgNPs film alone [
73].
The BC produced by
Acetobacter xylinum was isolated and dipped in AgNO
3 and AgCl solution to produce AgNPs. In-situ production of NPs was performed using the membrane as a template. The AgCl NPs-impregnated membrane exhibited high hydrophilic ability and strong antimicrobial activity against
E. coli and
S. aureus [
74].
4.3. Irradiation-Synthesized AgNPs Incorporated into Membranes and Composite Materials
Chitin membranes loaded with AgNPs were prepared by the solvent casting method in the presence of 5% LiCl and dimethylacetamide (DMA) solvent system. The NPs were prepared by the gamma irradiation method, using AgNO
3 as the precursor. The chitin membranes were loaded with ascending concentrations of AgNPs from 30 to 100 ppm. The chitin membranes with 100 ppm AgNPs showed promising antimicrobial activity against common wound pathogens (
P. aeruginosa and
S. aureus) [
75].
4.4. Thermal-Synthesized AgNPs Incorporated in Membranes and Composite Materials
In-situ preparation of AgNPs inside the BC membrane by thermal reduction at 80 °C was executed to obtain AgNPs-loaded BC scaffold. The results showed that AgNPs-BC exhibited significant antibacterial activity against
S. aureus. Moreover, AgNPs-BC allowed attachment and growth of rat fibroblasts with low cytotoxicity. The fresh epidermal and dermis thicknesses of the skin treated with AgNPs-BC samples were 111 and 855 μm, respectively, which was significantly higher than 74 and 619 μm for BC-treated groups and 57 and 473 μm for untreated control wounds [
76].
8. Safety of AgNPs in Wound Dressing Applications
The question of the potential toxicity of AgNPs is widely discussed. Nanomaterials can easily pass through cell membranes and adversely affect human health [
107]. In mouse models, they passed through the blood–brain and blood–testis barriers [
108]. AgNPs in higher concentrations (>44.0 μg/mL) are necrotic to cells, resulting in cell membrane rupture [
107]. After their passing through the blood–testis barrier, AgNPs are deposited in the testes and can have adverse effects on sperm cells [
109]. They have also been shown to damage other cells such as the brain [
110], liver [
111] and stem cells [
112]. However, the findings of many authors indicated that the use of AgNPs as a topical antibacterial agent is safe. The AgNPs-loaded hydrogel is considered an excellent wound dressing [
113]. If used in reasonable quantities, silver metal and silver dressings have no negative effects on human health [
114,
115]. For example, Kokura et al. [
116] studied the permeability of AgNPs in human skin and their cytotoxicity in human HaCaT (a spontaneously immortalized human keratinocyte line) keratinocytes under ultraviolet B (UVB) irradiation. Based on a transcutaneous passage assay using human skin, AgNPs did not penetrate healthy intact human skin. AgNPs showed no effect on keratinocytes and did not increase UVB-induced cell death. The efficacy of AgNPs increases at low concentrations. AgNPs do not easily penetrate the skin barrier and have no detrimental effects on keratinocytes [
116]. Thus, AgNPs possess the outstanding potential for use in wound dressing applications.
In addition, NPs are generally considered a promising replacement for antibiotics due to their bactericidal activity against a large number of pathogens and the non-induction of microbial resistance [
117]. AgNPs, in particular, have received a great deal of attention from scientists due to their inhibitory effect on around 650 microbe species and antibiotic-resistant bacteria [
118]. AgNPs inhibit bacterial reproduction by denaturing bacterial DNA, leading to the alteration and subsequently the death of the bacterial cell [
119]. Moreover, AgNPs can be an appropriate therapeutic agent or prophylactic because they prevent the colonization of the wound by antibiotic-resistant bacteria and other microbes that impede wound healing [
120]. Based on using human keratinocytes and dermal fibroblasts, AgNPs significantly reduced levels of inflammatory cytokines and promoted healing [
121]. In vitro cell culture tests exhibited no cytotoxicity in the hydrogel containing AgNPs and non-adherent character to dermal fibroblasts [
122]. Compared with conventional topical agents, AgNPs can both effectively prevent wound infections and improve the healing process of injured tissues. AgNPs-coated wound dressings possess considerable antibacterial activity and repair tissues faster and more efficiently [
123]. Application of silver modified nanoporous silica carriers loaded with sulfadiazine instead of silver sulfadiazine could overcome the poor aqueous solubility of silver sulfadiazine which limits its antibacterial effect [
124].
On the contrary, some authors are convinced of the risks of using AgNPs. Some cases where argyria appeared after treatment of burn wounds with dressings containing nanocrystalline silver were reported [
125,
126]. The use of these dressings caused the deposition of the silver particles into the mid and deep dermis [
125]. For silver itself, it was found that this metal had a concentration-dependent cytotoxic effect on human dermal fibroblast cells [
127]. However, thanks to the development of nanotechnology, its minimum inhibitory concentration, as well as toxicity to normal human cells, was reduced [
128] and a number of brands of wound dressings containing silver have been accepted by the U.S. Food and Drug Administration [
100]. On the other hand, Burd et al. [
129] demonstrated that some commercial silver-based dressings exhibited cytotoxic effects on cultured keratinocytes and fibroblasts.