Selective Laser Sintering of Solid Oral Dosage Forms with Copovidone and Paracetamol Using a CO2 Laser
Abstract
1. Introduction
2. Materials and Methods
2.1. Physicochemical Characterization of Powders
2.1.1. Scanning Electron Microscopy (SEM)
2.1.2. Preparation of Mixtures
2.1.3. Laser Granulometry
2.1.4. Study of Flowability and Compactness
2.1.5. Fourier-Transform Infrared Spectroscopy (FTIR)
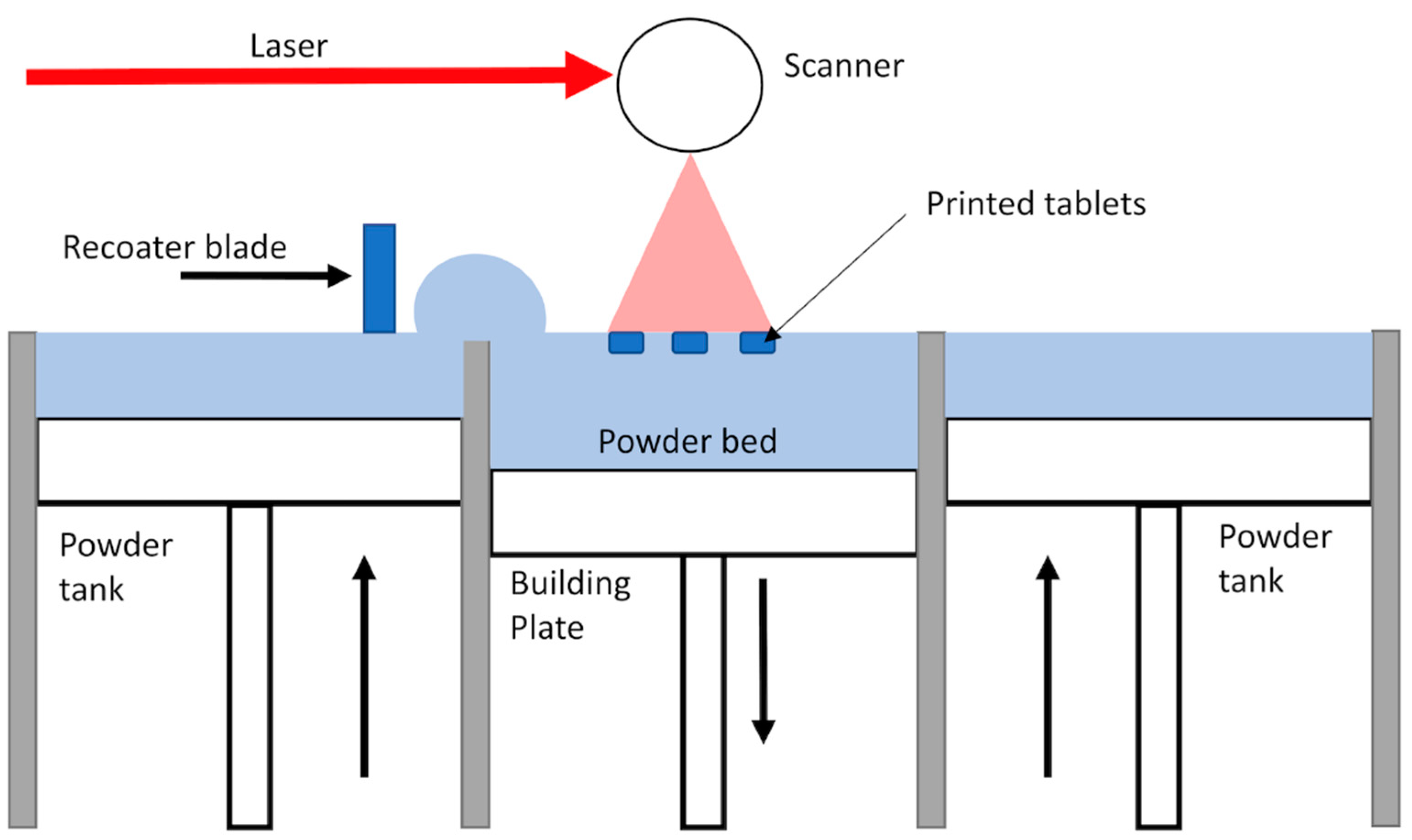
2.2. Printing of SODFs:
2.3. Characterization of the Printed SODFs
2.3.1. Differential Scanning Calorimetry (DSC)
2.3.2. X-ray Powder Diffraction (XRPD)
2.3.3. Weight, Dimensions and Mechanical Strength of the Printed SODFs
2.3.4. Disintegration Time of the Printed SODFs
2.3.5. Drug Content of the Printed SODFs
2.3.6. Size Exclusion Chromatography—Multi Angle Light Scattering (SEC-MALS)
2.3.7. Drug Release of the Printed SODFs
3. Results & Discussion
3.1. Physicochemical Characterization of Powders
3.1.1. Particle Morphology and Size Distribution
3.1.2. Study of Flowability and Compactness
3.1.3. Infrared Absorbance
3.2. Printing of SODFs
3.3. Characterization of the Printed SODFs:
3.3.1. Differential Scanning Calorimetry
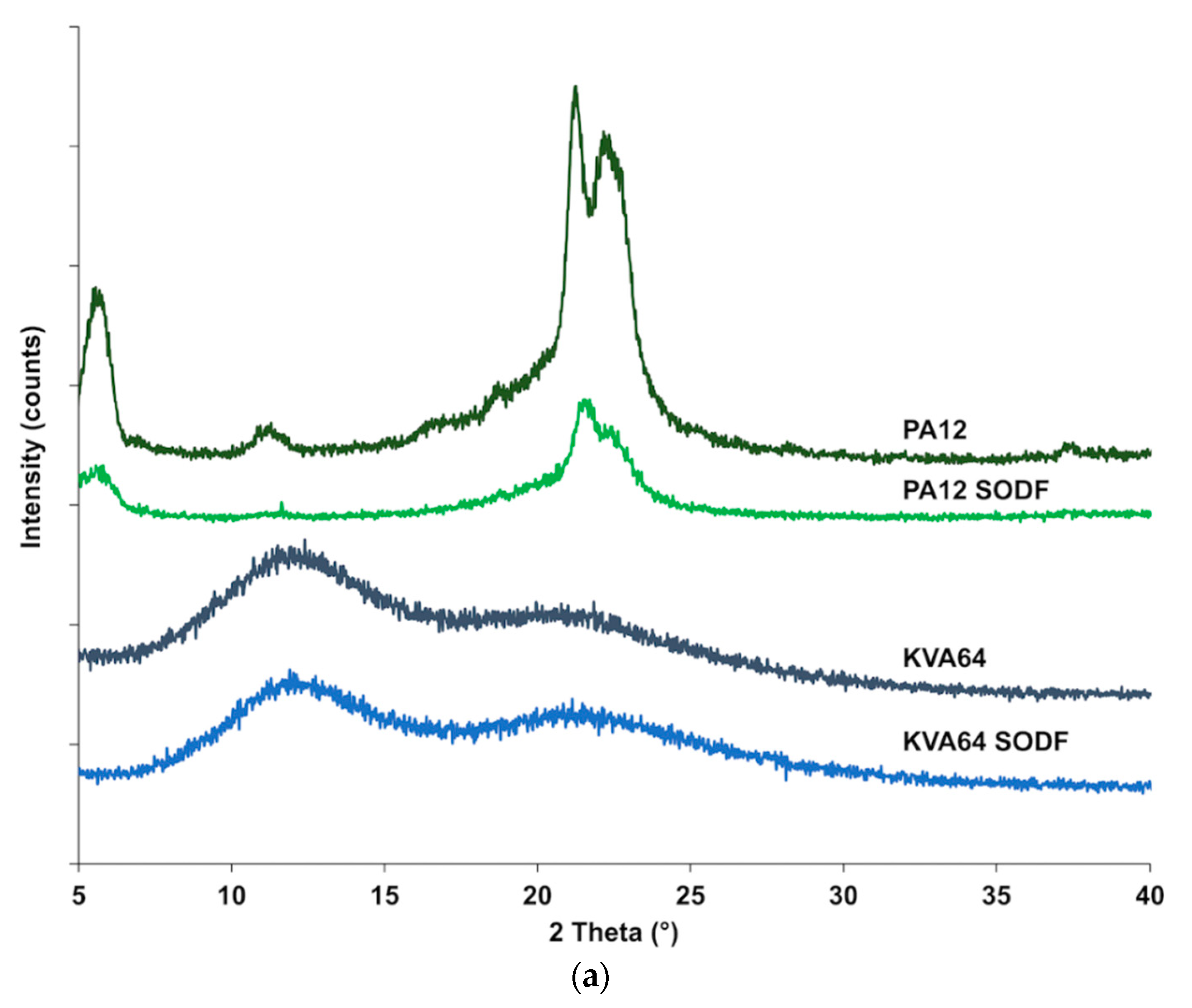
3.3.2. XRPD
3.3.3. Properties of the Printed SODFs
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Apprecia Pharmaceuticals What is SPRITAM? Available online: https://www.spritam.com/#/patient/about-spritam/what-is-spritam (accessed on 7 October 2019).
- Cailleaux, S.; Sanchez-Ballester, N.M.; Gueche, Y.A.; Bataille, B.; Soulairol, I. Fused Deposition Modeling (FDM), the new asset for the production of tailored medicines. J. Control. Release 2020. [Google Scholar] [CrossRef] [PubMed]
- Charoo, N.A.; Barakh Ali, S.F.; Mohamed, E.M.; Kuttolamadom, M.A.; Ozkan, T.; Khan, M.A.; Rahman, Z. Selective laser sintering 3D printing—An overview of the technology and pharmaceutical applications. Drug Dev. Ind. Pharm. 2020, 46, 869–877. [Google Scholar] [CrossRef] [PubMed]
- Awad, A.; Fina, F.; Goyanes, A.; Gaisford, S.; Basit, A.W. 3D printing: Principles and pharmaceutical applications of selective laser sintering. Int. J. Pharm. 2020, 586, 119594. [Google Scholar] [CrossRef] [PubMed]
- Goodridge, R.D.; Tuck, C.J.; Hague, R.J.M. Laser sintering of polyamides and other polymers. Prog. Mater. Sci. 2012, 57, 229–267. [Google Scholar] [CrossRef]
- Goyanes, A.; Fina, F.; Martorana, A.; Sedough, D.; Gaisford, S.; Basit, A.W. Development of modified release 3D printed tablets (printlets) with pharmaceutical excipients using additive manufacturing. Int. J. Pharm. 2017, 527, 21–30. [Google Scholar] [CrossRef]
- Fina, F.; Goyanes, A.; Gaisford, S.; Basit, A.W. Selective laser sintering (SLS) 3D printing of medicines. Int. J. Pharm. 2017, 529, 285–293. [Google Scholar] [CrossRef]
- Fina, F.; Madla, C.M.; Goyanes, A.; Zhang, J.; Gaisford, S.; Basit, A.W. Fabricating 3D printed orally disintegrating printlets using selective laser sintering. Int. J. Pharm. 2018, 541, 101–107. [Google Scholar] [CrossRef]
- Fina, F.; Goyanes, A.; Madla, C.M.; Awad, A.; Trenfield, S.J.; Kuek, J.M.; Patel, P.; Gaisford, S.; Basit, A.W. 3D printing of drug-loaded gyroid lattices using selective laser sintering. Int. J. Pharm. 2018, 547, 44–52. [Google Scholar] [CrossRef]
- Trenfield, S.J.; Goyanes, A.; Telford, R.; Wilsdon, D.; Rowland, M.; Gaisford, S.; Basit, A.W. 3D printed drug products: Non-destructive dose verification using a rapid point-and-shoot approach. Int. J. Pharm. 2018, 549, 283–292. [Google Scholar] [CrossRef]
- Awad, A.; Fina, F.; Trenfield, S.; Patel, P.; Goyanes, A.; Gaisford, S.; Basit, A. 3D Printed Pellets (Miniprintlets): A Novel, Multi-Drug, Controlled Release Platform Technology. Pharmaceutics 2019, 11, 148. [Google Scholar] [CrossRef]
- Barakh Ali, S.F.; Mohamed, E.M.; Ozkan, T.; Kuttolamadom, M.A.; Khan, M.A.; Asadi, A.; Rahman, Z. Understanding the effects of formulation and process variables on the printlets quality manufactured by selective laser sintering 3D printing. Int. J. Pharm. 2019, 570, 118651. [Google Scholar] [CrossRef] [PubMed]
- Trenfield, S.J.; Tan, H.X.; Goyanes, A.; Wilsdon, D.; Rowland, M.; Gaisford, S.; Basit, A.W. Non-destructive dose verification of two drugs within 3D printed polyprintlets. Int. J. Pharm. 2020, 577, 119066. [Google Scholar] [CrossRef] [PubMed]
- Allahham, N.; Fina, F.; Marcuta, C.; Kraschew, L.; Mohr, W.; Gaisford, S.; Basit, A.W.; Goyanes, A. Selective Laser Sintering 3D Printing of Orally Disintegrating Printlets Containing Ondansetron. Pharmaceutics 2020, 12, 110. [Google Scholar] [CrossRef] [PubMed]
- Awad, A.; Yao, A.; Trenfield, S.J.; Goyanes, A.; Gaisford, S.; Basit, A.W. 3D Printed Tablets (Printlets) with Braille and Moon Patterns for Visually Impaired Patients. Pharmaceutics 2020, 12, 172. [Google Scholar] [CrossRef]
- Mohamed, E.M.; Barakh Ali, S.F.; Rahman, Z.; Dharani, S.; Ozkan, T.; Kuttolamadom, M.A.; Khan, M.A. Formulation Optimization of Selective Laser Sintering 3D-Printed Tablets of Clindamycin Palmitate Hydrochloride by Response Surface Methodology. Aaps Pharmscitech 2020, 21, 232. [Google Scholar] [CrossRef]
- Davis, D.A.; Thakkar, R.; Su, Y.; Williams, R.O.; Maniruzzaman, M. Selective Laser Sintering 3-Dimensional Printing as a Single Step Process to Prepare Amorphous Solid Dispersion Dosage Forms for Improved Solubility and Dissolution rate. J. Pharm. Sci. 2020. [Google Scholar] [CrossRef]
- Hamed, R.; Mohamed, E.M.; Rahman, Z.; Khan, M.A. 3D-printing of lopinavir printlets by selective laser sintering and quantification of crystalline fraction by XRPD-chemometric models. Int. J. Pharm. 2020, 120059. [Google Scholar] [CrossRef]
- Januskaite, P.; Xu, X.; Ranmal, S.R.; Gaisford, S.; Basit, A.W.; Tuleu, C.; Goyanes, A. I Spy with My Little Eye: A Paediatric Visual Preferences Survey of 3D Printed Tablets. Pharmaceutics 2020, 12, 1100. [Google Scholar] [CrossRef]
- Yang, Y.; Xu, Y.; Wei, S.; Shan, W. Oral preparations with tunable dissolution behavior based on selective laser sintering technique. Int. J. Pharm. 2020, 120127. [Google Scholar] [CrossRef]
- Salmoria, G.V.; Klauss, P.; Zepon, K.; Kanis, L.A.; Roesler, C.R.M.; Vieira, L.F. Development of functionally-graded reservoir of PCL/PG by selective laser sintering for drug delivery devices: This paper presents a selective laser sintering-fabricated drug delivery system that contains graded progesterone content. Virtual Phys. Prototyp. 2012, 7, 107–115. [Google Scholar] [CrossRef]
- Salmoria, G.V.; Klauss, P.; Zepon, K.M.; Kanis, L.A. The effects of laser energy density and particle size in the selective laser sintering of polycaprolactone/progesterone specimens: Morphology and drug release. Int. J. Adv. Manuf. Technol. 2013, 66, 1113–1118. [Google Scholar] [CrossRef]
- Salmoria, G.V.; Vieira, F.E.; Ghizoni, G.B.; Gindri, I.M.; Kanis, L.A. Additive Manufacturing of PE/Fluorouracil Waffles for Implantable Drug Delivery in Bone Cancer Treatment. Eng. J. 2017, 3, 62–70. [Google Scholar] [CrossRef]
- Aho, J.; Bøtker, J.P.; Genina, N.; Edinger, M.; Arnfast, L.; Rantanen, J. Roadmap to 3D-Printed Oral Pharmaceutical Dosage Forms: Feedstock Filament Properties and Characterization for Fused Deposition Modeling. J. Pharm. Sci. 2019, 108, 26–35. [Google Scholar] [CrossRef] [PubMed]
- Fuenmayor, E.; Forde, M.; Healy, A.; Devine, D.; Lyons, J.; McConville, C.; Major, I. Material Considerations for Fused-Filament Fabrication of Solid Dosage Forms. Pharmaceutics 2018, 10, 44. [Google Scholar] [CrossRef] [PubMed]
- Tan, J.H.; Wong, W.L.E.; Dalgarno, K.W. An overview of powder granulometry on feedstock and part performance in the selective laser melting process. Addit. Manuf. 2017, 18, 228–255. [Google Scholar] [CrossRef]
- Chatham, C.A.; Long, T.E.; Williams, C.B. A review of the process physics and material screening methods for polymer powder bed fusion additive manufacturing. Prog. Polym. Sci. 2019, 93, 68–95. [Google Scholar] [CrossRef]
- Kolter, L.; Karl, M.; Gryczke, A. Hot-Melt Extrusion with BASF Pharma Polymers. Extrusion Compendium, 2nd ed.; BASF: Ludwigshafen, Germany, 2012. [Google Scholar]
- Kollamaram, G.; Croker, D.M.; Walker, G.M.; Goyanes, A.; Basit, A.W.; Gaisford, S. Low temperature fused deposition modeling (FDM) 3D printing of thermolabile drugs. Int. J. Pharm. 2018, 545, 144–152. [Google Scholar] [CrossRef]
- European Directorate for the Quality of Medicines & Healthcare. Council of Europe Bulk density and tapped density of powders (monograph 2.9.34). In European Pharmacopeia; Council of Europe: Strasbourg, France, 2019; pp. 384–387. [Google Scholar]
- European Directorate for the Quality of Medicines & Healthcare. Council of Europe Powder flow (monograph 2.9.36). In European Pharmacopeia; Council of Europe: Strasbourg, France, 2019; pp. 387–390. [Google Scholar]
- European Directorate for the Quality of Medicines & Healthcare. Council of Europe Disintegration of tablets and capsules (monograph 2.9.1). In European Pharmacopeia; Council of Europe: Strasbourg, France, 2019; pp. 323–325. [Google Scholar]
- Frank, K.J.; Westedt, U.; Rosenblatt, K.M.; Hölig, P.; Rosenberg, J.; Mägerlein, M.; Fricker, G.; Brandl, M. The amorphous solid dispersion of the poorly soluble ABT-102 forms nano/microparticulate structures in aqueous medium: Impact on solubility. Int. J. Nanomed. 2012, 7, 5757–5768. [Google Scholar]
- European Directorate for the Quality of Medicines & Healthcare. Council of Europe Dissolution test for solid dosage forms (monograph 2.9.3). In European Pharmacopeia; Council of Europe: Strasbourg, France, 2019; pp. 326–333. [Google Scholar]
- Gu, D.D.; Meiners, W.; Wissenbach, K.; Poprawe, R. Laser additive manufacturing of metallic components: Materials, processes and mechanisms. Int. Mater. Rev. 2012, 57, 133–164. [Google Scholar] [CrossRef]
- Tolochko, N.K.; Khlopkov, Y.V.; Mozzharov, S.E.; Ignatiev, M.B.; Laoui, T.; Titov, V.I. Absorptance of powder materials suitable for laser sintering. Rapid Prototyp. J. 2000, 6, 155–161. [Google Scholar] [CrossRef]
- Wohlers, T.T. Wohlers Report 2009: State of the Industry Annual Worldwide Progress Report; Wohlers Associates: Fort Collins, CO, USA, 2009; ISBN 978-0-9754429-5-1. [Google Scholar]
- Nasereddin, J.M.; Wellner, N.; Alhijjaj, M.; Belton, P.; Qi, S. Development of a Simple Mechanical Screening Method for Predicting the Feedability of a Pharmaceutical FDM 3D Printing Filament. Pharm. Res. 2018, 35, 151. [Google Scholar] [CrossRef] [PubMed]
- Cleary, P.W.; Sawley, M.L. DEM modelling of industrial granular flows: 3D case studies and the effect of particle shape on hopper discharge. Appl. Math. Model. 2002, 26, 89–111. [Google Scholar] [CrossRef]
- Olakanmi, E.O. Effect of mixing time on the bed density, and microstructure of selective laser sintered (sls) aluminium powders. Mat. Res. 2012, 15, 167–176. [Google Scholar] [CrossRef]
- Tarlier, N.; Soulairol, I.; Sanchez-Ballester, N.; Baylac, G.; Aubert, A.; Lefevre, P.; Bataille, B.; Sharkawi, T. Deformation behavior of crystallized mannitol during compression using a rotary tablet press simulator. Int. J. Pharm. 2018, 547, 142–149. [Google Scholar] [CrossRef] [PubMed]
- Jain, P.K.; Pandey, P.M.; Rao, P.V.M. Selective laser sintering of clay-reinforced polyamide. Polym. Compos. 2009, 732–743. [Google Scholar] [CrossRef]
- Spierings, A.B.; Voegtlin, M.; Bauer, T.; Wegener, K. Powder flowability characterisation methodology for powder-bed-based metal additive manufacturing. Prog. Addit. Manuf. 2016, 1, 9–20. [Google Scholar] [CrossRef]
- Liu, S.; Ha, Z. Prediction of random packing limit for multimodal particle mixtures. Powder Technol. 2002, 126, 283–296. [Google Scholar] [CrossRef]
- Furnas, C.C. Grading Aggregates-I-Mathematical Relations for Beds of Broken Solids of Maximum Density. Ind. Eng. Chem. 1931, 23, 1052–1058. [Google Scholar] [CrossRef]
- McGEARY, R.K. Mechanical Packing of Spherical Particles. J. Am. Ceram. Soc. 1961, 44, 513–522. [Google Scholar] [CrossRef]
- Li, R.; Shi, Y.; Wang, Z.; Wang, L.; Liu, J.; Jiang, W. Densification behavior of gas and water atomized 316L stainless steel powder during selective laser melting. Appl. Surf. Sci. 2010, 256, 4350–4356. [Google Scholar] [CrossRef]
- Salmoria, G.V.; Paggi, R.A.; Lago, A.; Beal, V.E. Microstructural and mechanical characterization of PA12/MWCNTs nanocomposite manufactured by selective laser sintering. Polym. Test. 2011, 30, 611–615. [Google Scholar] [CrossRef]
- Schmid, M.; Amado, F.; Levy, G.; Wegener, K. Flowability of Powders for Selective Laser Sintering (SLS) Investigated by Round Robin. In High Value Manufacturing: Proceedings of the 6th International Conference on Advanced Research in Virtual and Rapid Prototyping, Leiria, Portugal, 1–5 October, 2013; Taylor & Francis: Abingdon-on-Thames, UK, 2014; pp. 95–99. [Google Scholar]
- Drummer, D.; Drexler, M.; Wudy, K. Density of Laser Molten Polymer Parts as Function of Powder Coating Process during Additive Manufacturing. Procedia. Eng. 2015, 102, 1908–1917. [Google Scholar] [CrossRef]
- Velu, R.; Singamneni, S. Evaluation of the influences of process parameters while selective laser sintering PMMA powders. Proc. Inst. Mech. Eng. Part. C J. Mech. Eng. Sci. 2015, 229, 603–613. [Google Scholar] [CrossRef]
- Benedetti, L.; Brulé, B.; Decreamer, N.; Evans, K.E.; Ghita, O. Shrinkage behaviour of semi-crystalline polymers in laser sintering: PEKK and PA12. Mater. Des. 2019, 181, 107906. [Google Scholar] [CrossRef]
- Bikiaris, D.; Papageorgiou, G.Z.; Stergiou, A.; Pavlidou, E.; Karavas, E.; Kanaze, F.; Georgarakis, M. Physicochemical studies on solid dispersions of poorly water-soluble drugs. Thermochim. Acta 2005, 439, 58–67. [Google Scholar] [CrossRef]
- Rombouts, M.; Froyen, L.; Gusarov, A.V.; Bentefour, E.H.; Glorieux, C. Photopyroelectric measurement of thermal conductivity of metallic powders. J. Appl. Phys. 2005, 97, 024905. [Google Scholar] [CrossRef]
- Spierings, A.B.; Herres, N.; Levy, G. Influence of the particle size distribution on surface quality and mechanical properties in AM steel parts. Rapid Prototyp. J. 2011, 17, 195–202. [Google Scholar] [CrossRef]
- Ligon, S.C.; Liska, R.; Stampfl, J.; Gurr, M.; Mülhaupt, R. Polymers for 3D Printing and Customized Additive Manufacturing. Chem. Rev. 2017, 117, 10212–10290. [Google Scholar] [CrossRef]
- European Directorate for the Quality of Medicines & Healthcare. Council of Europe Tablets. In European Pharmacopeia; Council of Europe: Strasbourg, France, 2019; pp. 937–939. [Google Scholar]
- European Medicines Agency. Committee for medicinal products for human use. In Guideline on the Investigation of Bioequivalence; CPMP/EWP/QWP/1401/98 Rev.1/Corr**; European Medicines Agency: Amsterdam, Netherlands, 2010. [Google Scholar]
- Školáková, T.; Slámová, M.; Školáková, A.; Kadeřábková, A.; Patera, J.; Zámostný, P. Investigation of Dissolution Mechanism and Release Kinetics of Poorly Water-Soluble Tadalafil from Amorphous Solid Dispersions Prepared by Various Methods. Pharmaceutics 2019, 11, 383. [Google Scholar] [CrossRef]
- Goyanes, A.; Wang, J.; Buanz, A.; Martínez-Pacheco, R.; Telford, R.; Gaisford, S.; Basit, A.W. 3D Printing of Medicines: Engineering Novel Oral Devices with Unique Design and Drug Release Characteristics. Mol. Pharm. 2015, 12, 4077–4084. [Google Scholar] [CrossRef]
- Yang, Y.; Wang, H.; Li, H.; Ou, Z.; Yang, G. 3D printed tablets with internal scaffold structure using ethyl cellulose to achieve sustained ibuprofen release. Eur. J. Pharm. Sci. 2018, 115, 11–18. [Google Scholar] [CrossRef] [PubMed]








| Mixtures | KVA64 | KVA64F | PAR | PAR F |
|---|---|---|---|---|
| 90% KVA64/10% KVA64F | 90% | 10% | / | / |
| 80% KVA64/20% KVA64F | 80% | 20% | / | / |
| 70% KVA64/30% KVA64F | 70% | 30% | / | / |
| 90% KVA64/10% PAR | 90% | / | 10% | / |
| 80% KVA64/20% PAR | 80% | / | 20% | / |
| 70% KVA64/30% PAR | 70% | / | 30% | / |
| 90% KVA64/10% PAR F | 90% | / | / | 10% |
| 80% KVA64/20% PAR F | 80% | / | / | 20% |
| 70% KVA64/30% PAR F | 70% | / | / | 30% |
| Printing Parameters | Heating Temperature (°C) | Laser Power (%) | Scan Space (pps) | Layer Thickness (mm) |
|---|---|---|---|---|
| PA12 | 165 | 25 | 45,000 | 0.1 |
| KVA64 | 110 | 25,000 | ||
| 90% KVA64 10% KVA64F | ||||
| 90% KVA64 10% PAR | 100 | |||
| 80% KVA64 20% PAR | 95 | |||
| 70% KVA64 30% PAR | 90 |
| Powder | D (4,3) (µm) | Span |
|---|---|---|
| KVA64 | 71.49 ± 0.96 | 2.16 ± 0.03 |
| KVA64F | 26.00 ± 0.15 | 2.16 ± 0.02 |
| PA12 | 63.66 ± 0.29 | 0.93 ± 0.04 |
| PAR | 124.66 ± 4.49 | 3.34 ± 0.02 |
| PAR F | 15.40 ± 0.12 | 2.25 ± 0.02 |
| 90% KVA64/10% KVA64F | 64.20 ± 2.94 | 2.42 ± 0.06 |
| 80% KVA64/20% KVA64F | 56.84 ± 0.76 | 2.63 ± 0.03 |
| 70% KVA64/30% KVA64F | 51.44 ± 0.37 | 2.84 ± 0.02 |
| 90% KVA64/10% PAR | 81.91 ± 0.28 | 2.32 ± 0.01 |
| 80% KVA64/20% PAR | 88.87 ± 3.31 | 2.59 ± 0.05 |
| 70% KVA64/30% PAR | 92.46 ± 1.89 | 2.84 ± 0.04 |
| 90% KVA64/10% PAR F | 68.28 ± 1.65 | 2.28 ± 0.02 |
| 80% KVA64/20% PAR F | 63.20 ± 2.26 | 2.54 ± 0.06 |
| 70% KVA64/30% PAR F | 58.38 ± 0.70 | 2.77 ± 0.05 |
| Powder | BD (g/cm3) | TD (g/cm3) | HR | AOR (°) | Flow Property * |
|---|---|---|---|---|---|
| KVA64 | 0.38 ± 0.00 | 0.48 ± 0.01 | 1.25 ± 0.02 | 37.73 ± 0.93 | Fair |
| KVA64F | 0.12 ± 0.00 | 0.19 ± 0.01 | 1.57 ± 0.10 | / | Very poor |
| PA12 | 0.48 ± 0.01 | 0.58 ± 0.01 | 1.19 ± 0.04 | 34.00 ± 0.17 | Good |
| 90% KVA64/10% KVA64F | 0.35 ± 0.01 | 0.43 ± 0.00 | 1.24 ± 0.02 | 39.17 ± 1.62 | Fair |
| 80% KVA64/20% KVA64F | 0.30 ± 0.01 | 0.40 ± 0.00 | 1.33 ± 0.05 | / | Passable |
| 70% KVA64/30% KVA64F | 0.26 ± 0.01 | 0.35 ± 0.01 | 1.36 ± 0.02 | / | Poor |
| 90% KVA64/10% PAR | 0.34 ± 0.00 | 0.49 ± 0.01 | 1.43 ± 0.03 | 36.84 ± 0.06 | Fair |
| 80% KVA64/20% PAR | 0.34 ± 0.00 | 0.51 ± 0.01 | 1.50 ± 0.03 | 39.67 ± 0.55 | Fair |
| 70% KVA64/30% PAR | 0.34 ± 0.00 | 0.52 ± 0.01 | 1.53 ± 0.03 | 40.67 ± 0.97 | Fair |
| 90% KVA64/10% PAR F | 0.31 ± 0.01 | 0.48 ± 0.00 | 1.56 ± 0.04 | / | Very poor |
| 80% KVA64/20% PAR F | 0.28 ± 0.00 | 0.49 ± 0.01 | 1.79 ± 0.04 | / | Very, very poor |
| 70% KVA64/30% PAR F | 0.26 ± 0.00 | 0.49 ± 0.02 | 1.85 ± 0.06 | / | Very, very poor |
| Powder | Flow Property | Flat Layer at the 1st Attempt | Printability |
|---|---|---|---|
| KVA64 | Fair | YES | YES |
| KVA64F | Very poor | NO | NO |
| PA12 | Good | YES | YES |
| 90% KVA64/10% KVA64F | Fair | YES | YES |
| 80% KVA64/20% KVA64F | Passable | YES | NO |
| 70% KVA64/30% KVA64F | Poor | YES | NO |
| 90% KVA64/10% PAR | Fair | YES | YES |
| 80% KVA64/20% PAR | Fair | YES | YES |
| 70% KVA64/30% PAR | Fair | YES | YES |
| 90% KVA64/10% PAR F | Very poor | NO | NO |
| 80% KVA64/20% PAR F | Very, very poor | NO | NO |
| 70% KVA64/30% PAR F | Very, very poor | NO | NO |
| SODF | PA12 | KVA64 | 90% KVA64 10% KVA64F | 90% KVA64 10% PAR | 80% KVA64 20% PAR | 70% KVA64 30% PAR |
|---|---|---|---|---|---|---|
| T (mm) | 3.20 ± 0.02 | 4.15 ± 0.09 | 4.08 ± 0.20 | 3.99 ± 0.14 | 3.72 ± 0.06 | 3.76 ± 0.09 |
| D (mm) | 10.00 ± 0.12 | 10.51 ± 0.20 | 10.44 ± 0.08 | 10.60 ± 0.11 | 10.52 ± 0.13 | 10.50 ± 0.12 |
| W (mg) | 196.00 ± 6.33 | 200.80 ± 4.66 | 192.90 ± 9.05 | 188.40 ± 2.63 | 183.80 ± 3.65 | 184.80 ± 1.99 |
| H (N) | >500 | 89.32 ± 12.94 | 85.57 ± 9.48 | 55.59 ± 4.28 | 47.93 ± 7.19 | 47.18 ± 4.36 |
| DT (s) | / | 328.0 ± 67.0 | 262.0 ± 39.0 | 223.0 ± 8.1 | 134.0 ± 6.6 | 119.0 ± 5.0 |
| DC (%) | / | / | / | 10.03 ± 0.82 | 20.18 ± 0.75 | 29.81 ± 0.49 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gueche, Y.A.; Sanchez-Ballester, N.M.; Bataille, B.; Aubert, A.; Leclercq, L.; Rossi, J.-C.; Soulairol, I. Selective Laser Sintering of Solid Oral Dosage Forms with Copovidone and Paracetamol Using a CO2 Laser. Pharmaceutics 2021, 13, 160. https://doi.org/10.3390/pharmaceutics13020160
Gueche YA, Sanchez-Ballester NM, Bataille B, Aubert A, Leclercq L, Rossi J-C, Soulairol I. Selective Laser Sintering of Solid Oral Dosage Forms with Copovidone and Paracetamol Using a CO2 Laser. Pharmaceutics. 2021; 13(2):160. https://doi.org/10.3390/pharmaceutics13020160
Chicago/Turabian StyleGueche, Yanis A., Noelia M. Sanchez-Ballester, Bernard Bataille, Adrien Aubert, Laurent Leclercq, Jean-Christophe Rossi, and Ian Soulairol. 2021. "Selective Laser Sintering of Solid Oral Dosage Forms with Copovidone and Paracetamol Using a CO2 Laser" Pharmaceutics 13, no. 2: 160. https://doi.org/10.3390/pharmaceutics13020160
APA StyleGueche, Y. A., Sanchez-Ballester, N. M., Bataille, B., Aubert, A., Leclercq, L., Rossi, J.-C., & Soulairol, I. (2021). Selective Laser Sintering of Solid Oral Dosage Forms with Copovidone and Paracetamol Using a CO2 Laser. Pharmaceutics, 13(2), 160. https://doi.org/10.3390/pharmaceutics13020160





