1. Introduction
Depressive disorders are some of the most prevalent, impairing and costly illnesses, having recently been estimated to affect more than 246 million people worldwide [
1,
2,
3]. Although they can be divided according to subtype and level of severity, these disorders are generally characterized by a depressed mood or general loss of pleasure or interest, usually accompanied by symptoms such as feelings of guilt, worthlessness or hopelessness; low self-esteem; indecisiveness or difficulty in concentrating or thinking; fatigue; psychomotor agitation or retardation; change in appetite; insomnia or hypersomnia; mood swings; and, in most severe cases, recurrent thoughts of death or suicidal ideation (
Figure 1). The depressed person usually has a loss in work productivity and difficulty in managing social situations, with a decrease in general quality of life, and increased risk of suicide. Moreover, coexisting with other diseases can exacerbate their symptoms, being associated, for example, with a higher risk of severe complications in diabetic patients (amputation, blindness, dementia), and increasing the relative risk of developing heart disease by 200% [
1,
4,
5]. Pharmacological treatment of depressive disorders can be quite variable, but selective serotonin reuptake inhibitors (fluoxetine, paroxetine, sertraline) are usually considered as first-line options. Other alternatives include serotonin and norepinephrine reuptake inhibitors (duloxetine, venlafaxine), tricyclic antidepressants (amitriptyline, imipramine, nortriptyline), tetracyclic antidepressants (mirtazapine), and atypical antidepressants (trazodone, nefazodone, agomelatine). Adjuvant hormonal and psychological treatments are also recommended in some cases, as well as lifestyle changes [
1,
4,
6,
7,
8]. With so many available treatments to choose from, the choice should be made carefully, in a case-by-case trial-and-error approach, being individualized according to the patient’s characteristics (ethnicity, gender, age, socioeconomic status, comorbidities, etc.) and symptomatology [
1,
4].
Depression frequently coexists with other mental health disorders. There is a significantly increased risk of developing a comorbid depressive disorder when someone already has an anxiety disorder [
1,
5,
9,
10,
11]. Anxiety disorders are also among the most common mental disorders, having been recently estimated to globally affect more than 265 million people. They are associated with substantial functional impairment, which leads to decreased work productivity and quality of life [
2,
9,
12]. Aside from generalized anxiety disorder, there is a wide spectrum of related disorders (such as obsessive-compulsive, posttraumatic stress, panic and social anxiety disorders), but in general symptoms can include feeling exceptionally or chronically nervous, anxious or on edge; having excessive or uncontrollable fear and worry; sleep disturbances and hypervigilance; and constant avoidance of situations that relate to the previously mentioned symptoms (
Figure 1) [
9,
11,
13,
14]. Patients with anxiety disorders also have a higher prevalence of other diseases, such as cardiovascular, respiratory and gastrointestinal conditions [
9,
12]. Treatment of anxiety and related disorders includes psychological and pharmacological options, and the choice again depends on patient related factors, such as severity of illness, prior treatment, comorbid disorders, patient preference and motivation, etc. [
9]. The first-line pharmacological options are similar to those prescribed for depressive disorders: either selective serotonin reuptake inhibitors (escitalopram, fluoxetine, fluvoxamine, paroxetine, sertraline) or serotonin and norepinephrine reuptake inhibitors (duloxetine, venlafaxine), for being generally better tolerated and safer than other treatments [
9,
12]. Other options include noradrenergic and specific serotonergic antidepressants, tricyclic antidepressants, monoamine oxidase inhibitors, and reversible inhibitors of monoamine oxidase A [
9,
11]. Benzodiazepines can also be used, but as adjunctive short-term therapy, since they can cause dependency, sedation and cognitive impairment (especially with prolonged use) [
9,
12]. Some anticonvulsants and atypical antipsychotics have also demonstrated efficacy, but are generally recommended as second-line, third-line, or adjunctive therapies (due to side effects). Given the variety in treatment options, again it should be a case-by-case approach, taking into consideration efficacy versus safety, the specific characteristics of the anxiety disorder, comorbid conditions and treatment duration [
9].
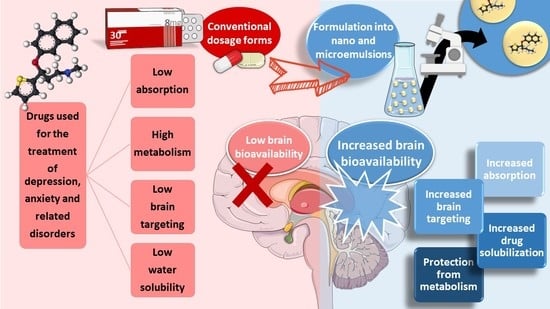
Yet, despite pharmacological treatment options for depressive and anxiety disorders being many, a great number of these drugs (including the grand majority of new drug candidates) have low water solubility, which can make it hard to formulate them at high strengths in liquid preparations [
15]. This problem can be tackled by formulating these molecules into solid forms, with oral tablets being the most common option, but dose adjustment can sometimes be difficult, and inappropriate tablet splitting can lead to dose intake variation, which in turn can result in a reduction in treatment efficacy or exacerbation of adverse effects. Moreover, swallowing these formulations can be challenging, especially in the younger population (children and adolescents) or older individuals (particularly if having diseases linked to dysphagia, such as stroke, Parkinson’s, Alzheimer’s or cancer) [
16,
17]. Intravenous treatments require liquid solutions, but drug solubilization is usually achieved either by pH adjustments in the formulation, which if very low or very high could be potentially harmful, or using great amounts of organic cosolvents or surfactants, which are potentially toxic excipients, having been reported to cause hemotoxicity and hypersensitivity reactions (pruritus, erythema, rash or urticaria) [
15]. Moreover, in these types of formulations, drugs are highly susceptible to metabolism, which can occur in all administration routes, but especially systemic ones, due to hepatic first-pass metabolism, being aggravated in oral administration, due to additional gastrointestinal degradation [
18]. Additionally, in general, the treatment of central nervous systems disorders can be compromised by the very low permeability of the blood-brain barrier, which restricts the transport of most drug molecules, and this is especially relevant for the most common administration routes, in which the drug is transported to the brain from the bloodstream (oral and intravenous) [
18,
19].
Incorporating drugs into a nanosystem can be an effective strategy to tackle these issues. Nanosystems (also known as nanocarriers) are colloidal structures with a mean diameter of less than 500 nm. Among their many advantages, they allow: the enhancement of drug solubilization; metabolic and chemical degradation drug protection; the reduction of high plasma protein binding; increased permeability through biological membranes; and the promotion of brain bioavailability, which is especially useful for diseases with a brain etiology [
19,
20,
21,
22,
23] (
Figure 2). The many types of nanosystems can be divided into four main categories: polymeric nanocarriers, such as polymeric nanoparticles and micelles; lipid nanoparticles, such as solid lipid nanoparticles or nanostructured lipid carriers; liposomes and their derived counterparts, such as niosomes, ethosomes, transfersomes, cubosomes and phytosomes; and nanometric emulsions, such as nanoemulsions and microemulsions [
18,
24,
25,
26]. Yet, despite all mentioned advantages, most of these nanosystems have several drawbacks, such as a low encapsulation efficiency; reduced physical stability; requiring the use of organic solvents during preparation; requiring complex and time-consuming preparation methods; and having non-biocompatible components [
27,
28,
29] (
Figure 2).
However, nanometric emulsions can surpasses all of these drawbacks. Being colloidal liquid-in-liquid dispersions, they are usually made of a water phase, an oil, a surfactant, a cosurfactant and/or a cosolvent. They can be classified according to droplet size, between nanoemulsions (20,200 nm) or microemulsions (10–100 nm), although the size range can differ between authors. Regarding what concerns the differences in their characteristics, while microemulsions have thermodynamic stability, nanoemulsions have a relatively high kinetic stability, and both have higher surface area and free energy than macroemulsions, which makes them more physically stable in comparison. They can also be classified according to the nature of their internal and external phases, as: oil-in-water (
o/
w) or water-in-oil (
w/
o), if they are biphasic (most common); or oil-in-water-in-oil or water-in-oil-in-water, if they are triphasic (
Figure 3) [
18,
30,
31].
Nanometric emulsions are lipophilic, biocompatible, have a solubilizing effect and high encapsulation efficiency, which makes them promising options for encapsulating lipophilic drugs. Furthermore, formulas with the right composition (excipients and the proportion between them) can be very stable, and components such as surfactants and cosolvents allow them enhance drug permeation. In addition, unlike many other nanosystems, nano and microemulsions do not require the use of organic solvents during production, and aside from preparation methods that require high energy inputs (such as sonication, high pressure homogenization, extrusion through a small pore membrane), they can form spontaneously just by adding their components in specific proportions, which makes their preparation simpler, cheaper and less time-consuming than other nanocarriers, making them ideal for industrial application, where these are key factors (
Figure 2) [
18,
30,
31].
Therefore, this review aimed to collect detailed information and conduct a critical analysis on nano and microemulsions for the treatment of depressive and/or anxiety disorders, including formulation composition and characterization (droplet size, polydispersity index—PDI, zeta potential, viscosity, osmolality and pH), in vitro drug release, ex vivo drug permeation, in vivo pharmacokinetics, in vivo pharmacodynamics and/or safety studies (depending on the available data). The final goal was to summarize and analyze what has been conducted so far in this specific field, providing a source of information for future studies.
3. Conclusions
The development of nanometric emulsions to encapsulate antidepressant and anxiolytic drugs has proven to be effective in increasing both drug strength and delivery, especially for lipophilic molecules. This happens not only due to small droplet size and the possibility of encapsulation of said molecules, but also due to the use of excipients with solubilizing capacity and permeation enhancing properties, such as surfactants, cosolvents and cyclodextrins. Furthermore, formulation characterization is not complete without determining and reporting droplet size, PDI, zeta potential, viscosity, osmolality and pH, which are all factors that could influence their in vivo performance and/or safety. Formulation stability studies are also recommended in order to know the time during which a selected formula will keep its properties. In vitro drug release, ex vivo drug permeation and specific biochemical estimations are not as indispensable, but might provide useful information that could help explain, deepen the knowledge or predict the outcomes of in vivo studies. On the other hand, in vivo animal pharmacokinetic and/or pharmacodynamic experiments are essential in order to assess the full potential of a developed formulation, and without them that assessment is left incomplete. Safety studies should also be more frequently performed, since even if a certain formulation is therapeutically effective, it is not promising unless it has a reasonably favorable efficacy/safety ratio. Hence, although the number of studies that have been performed so far is still small, which presents a limitation for drawing generalized conclusions, overall, nano and microemulsions have shown to be promising strategies to improve the solubilization and increase the bioavailability of antidepressant and/or anxiolytic drugs, being potential strategies to replace current therapies. More experimental studies should be conducted in the future, including clinical trials, in order to address these formulations’ true medical applicability.