The Fate of Sialic Acid and PEG Modified Epirubicin Liposomes in Aged versus Young Cells and Tumor Mice Models
Abstract
:1. Introduction
2. Materials, Animals, and Methods
2.1. Materials and Animals
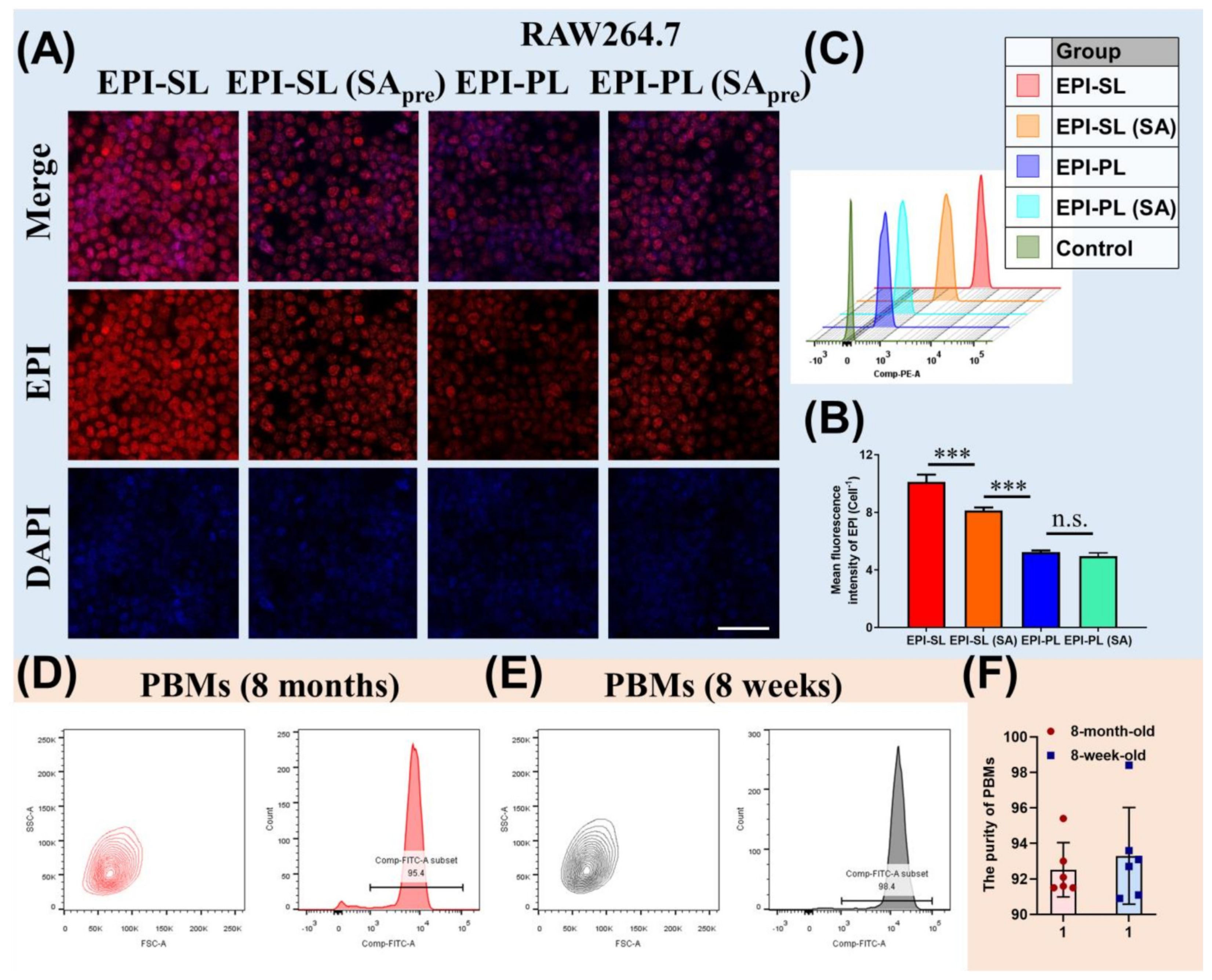
2.2. Peripheral Blood Monocytes in Aged and Young Mice
2.2.1. Isolation of Peripheral Blood Monocytes
2.2.2. Volume and Density of Peripheral Circulating Blood
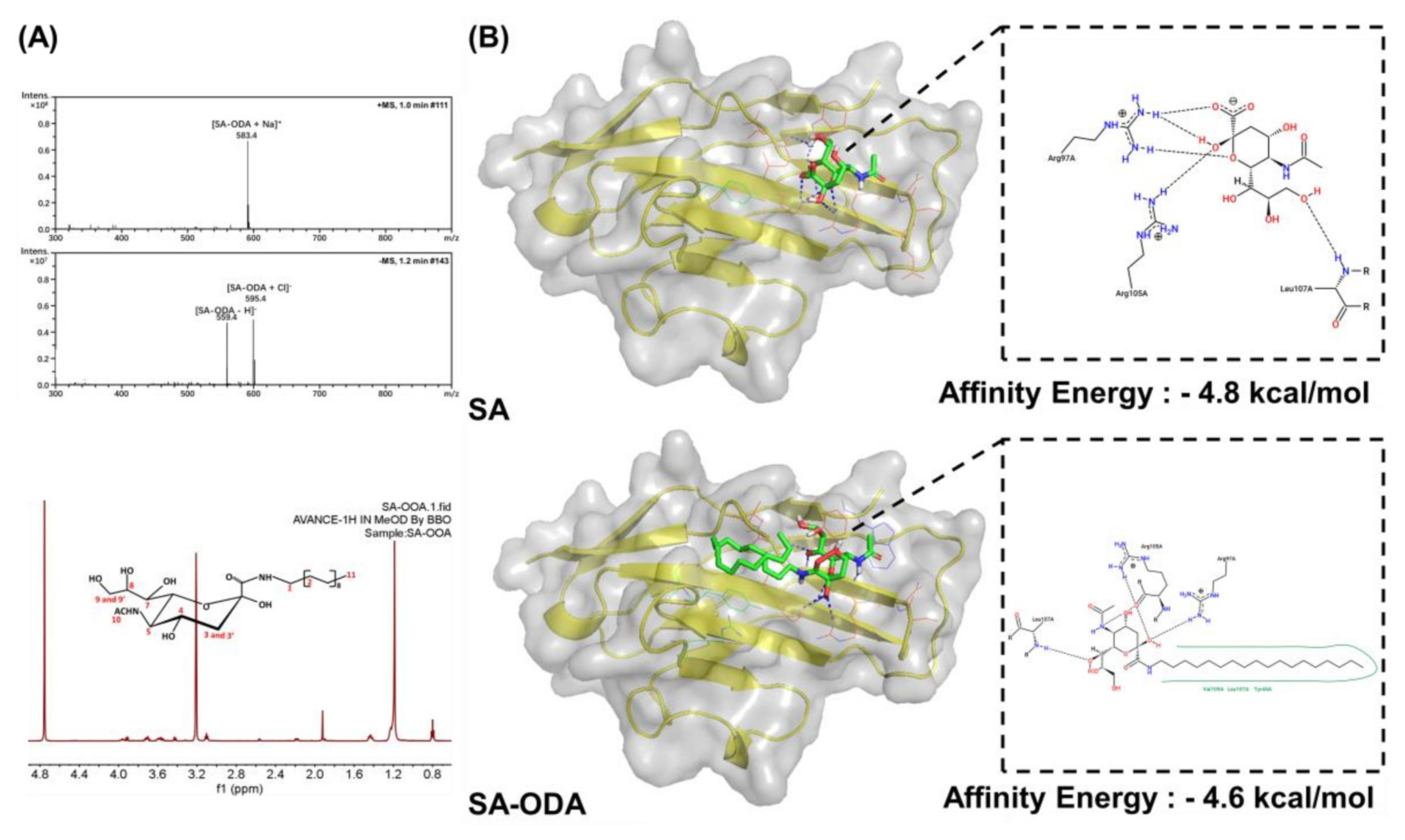
2.3. Chemical Synthesis of SA-ODA
2.4. Molecular Docking of Siglec-1 Receptor and SA-ODA
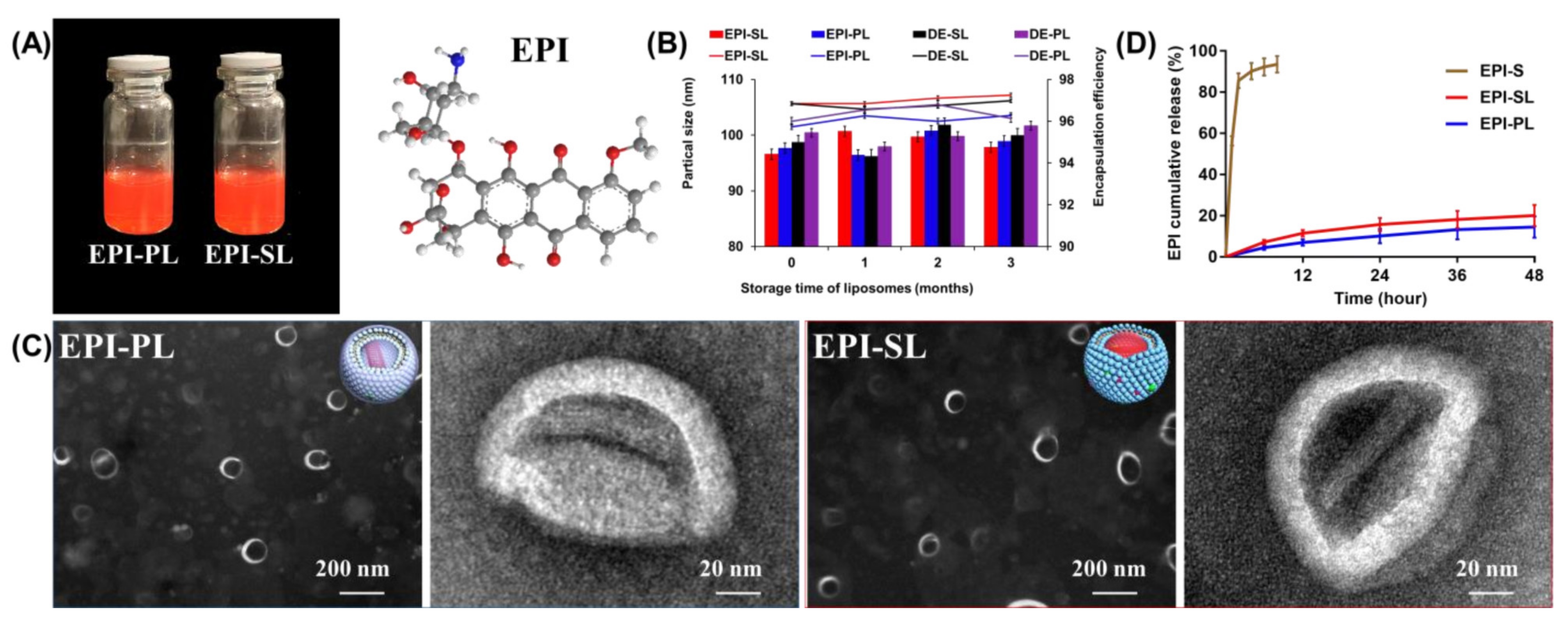
2.5. Preparation of EPI-SL and EPI-PL
2.6. Characterization of EPI-SL and EPI-PL
2.7. Drug Release of EPI-SL, EPI-PL, and EPI-Solution
2.8. Cytotoxicity of EPI-SL, EPI-PL, and EPI-Solution
2.9. Uptake Ability of PBMs and RAW264.7 Cells
2.10. Pharmacokinetics of EPI-SL and EPI-PL in Aged and Young Mice
2.11. Tissue Distribution of DE-SL and DE-PL in Aged and Young Mice
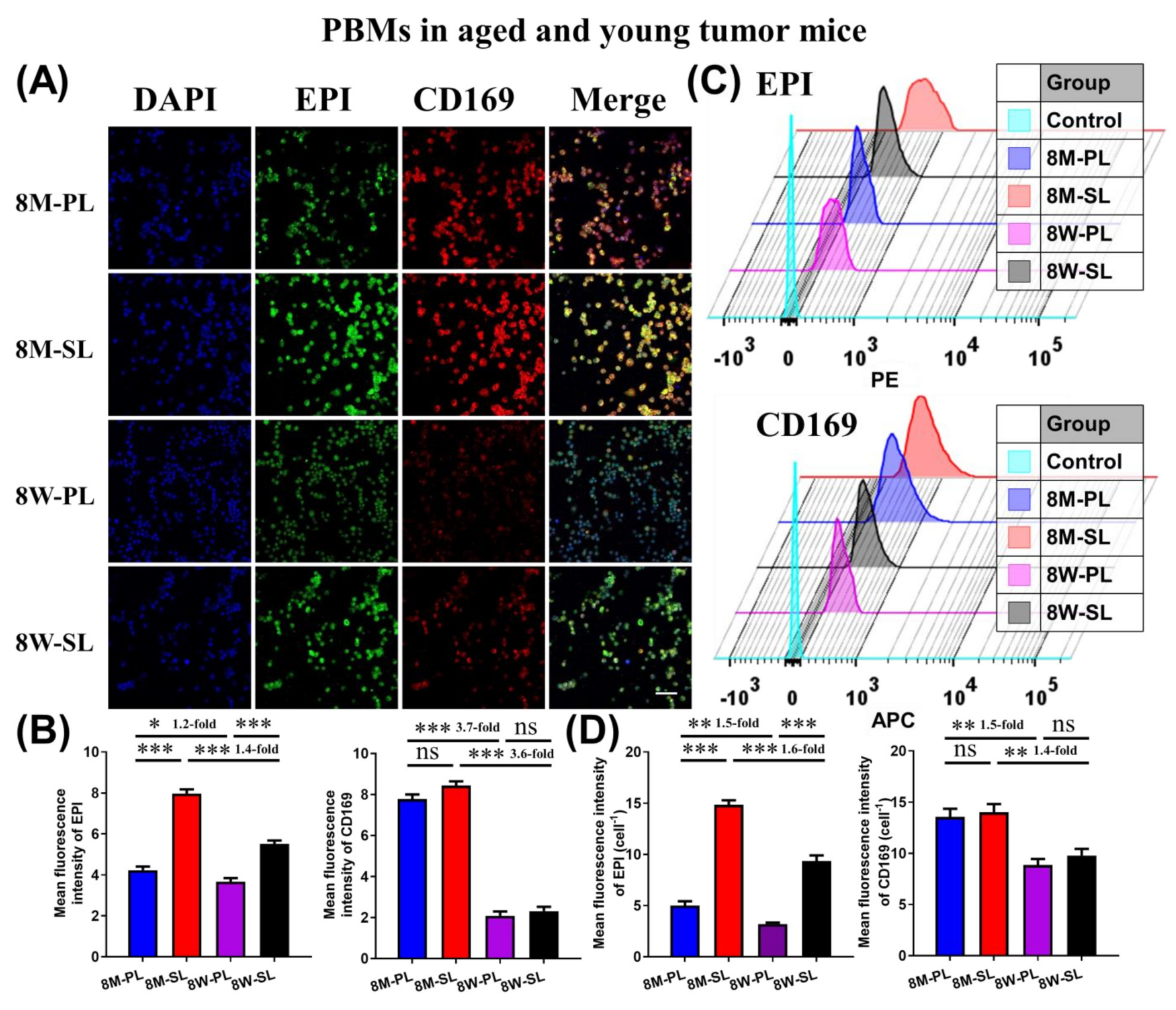
2.12. Pharmacodynamics of EPI-SL and EPI-PL in Aged and Young Mice
2.13. Statistical Analysis
3. Results and Discussion
3.1. Characterization and Molecular Docking of SA-ODA
3.2. Characterization of EPI-SL and EPI-PL
3.3. Uptake and Cytotoxicity In Vitro
3.4. Tissue Distribution of EPI-SL and EPI-PL in Aged and Young Mice
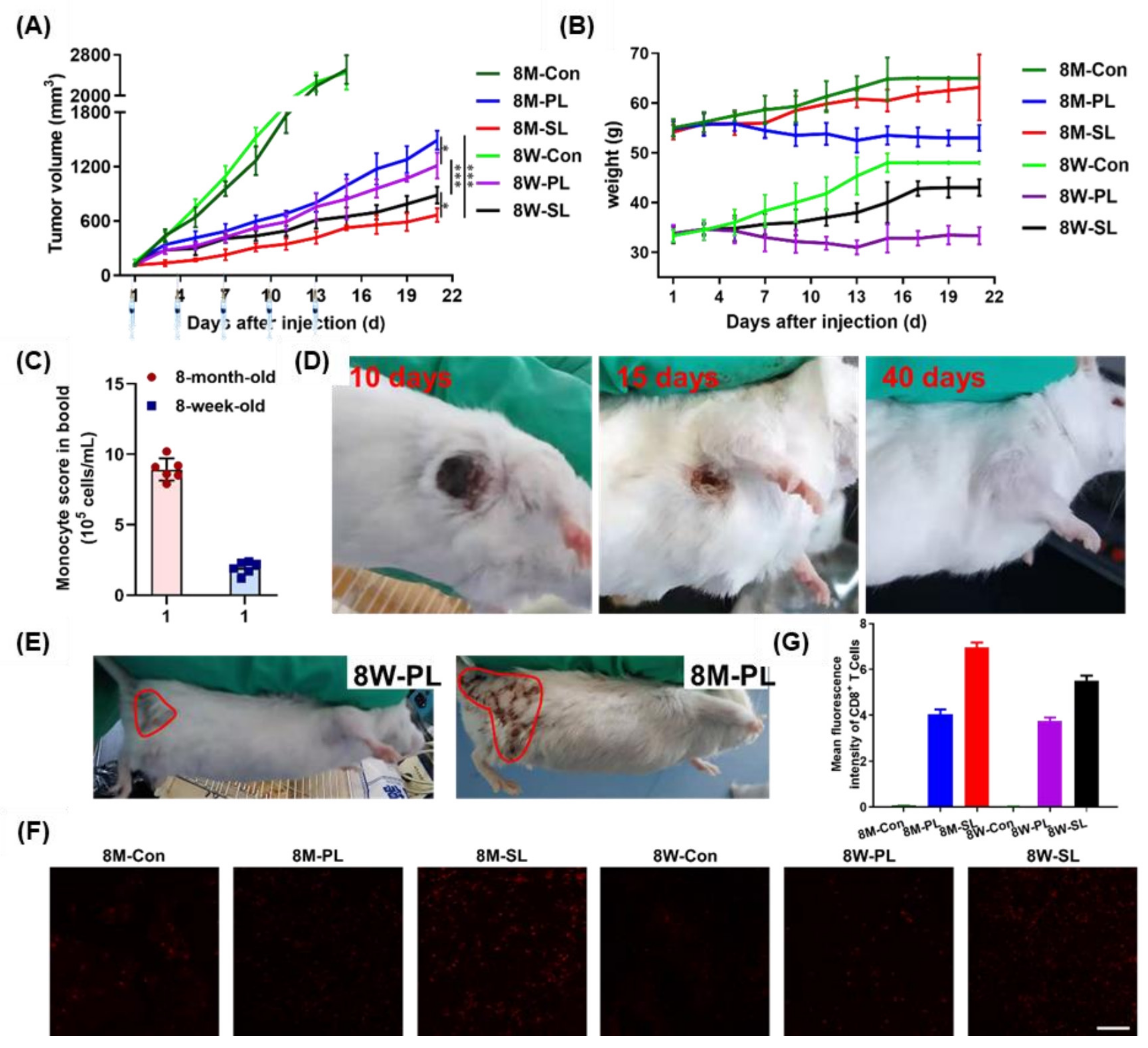
3.5. Pharmacodynamics of EPI-SL and EPI-PL in Aged and Young Mice
3.6. Tissue Section Staining of EPI-SL and EPI-PL in Aged and Young Mice
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| Allophycocyanin | APC |
| Cholesterol | CH |
| Drug delivery system | DDS |
| SA-modified DiR and EPI liposomes | DiR-SL |
| PEG-modified DiR and EPI liposomes | DiR-PL |
| 1,1′-dioctadecyl-3,3,3′,3′-tetramethylindotricarbocyanine iodide | DiR |
| Encapsulation efficiency | EE |
| Epirubicin | EPI |
| EPI Solution | EPI-S |
| PEG-modified epirubicin liposomes | EPI-PL |
| SA-modified epirubicin liposomes | EPI-SL |
| Enhanced permeability and retention | EPR |
| Hydrogenated Soy Phosphatidylcholine | HSPC |
| Sialic acid | SA |
| Sialic acid-octadecylamine derivatives | SA-ODA |
| Octadecylamine | ODA |
| Peripheral Blood Monocytes | PBMs |
| Polydispersity index | PDI |
| Polyethylene glycol | PEG |
| Particle size | PS |
| Zeta potential | ZP |
References
- Ciabattini, A.; Nardini, C.; Santoro, F.; Garagnani, P.; Franceschi, C.; Medaglini, D. Vaccination in the elderly: The challenge of immune changes with aging. Semin. Immunol. 2018, 40, 83–94. [Google Scholar] [CrossRef] [PubMed]
- Andrew, M.K.; McElhaney, J.E. Age and frailty in COVID-19 vaccine development. Lancet 2020, 396, 1942–1944. [Google Scholar] [CrossRef]
- Cox, L.S.; Bellantuono, I.; Lord, J.M.; Sapey, E.; Mannick, J.B.; Partridge, L.; Gordon, A.L.; Steves, C.J.; Witham, M.D. Tackling immunosenescence to improve COVID-19 outcomes and vaccine response in older adults. Lancet Healthy Longev. 2020, 1, e55–e57. [Google Scholar] [CrossRef]
- Narita, M.; Lowe, S.W. Senescence comes of age. Nat. Med. 2005, 11, 920–922. [Google Scholar] [CrossRef] [PubMed]
- Hernandez-Segura, A.; Nehme, J.; Demaria, M. Hallmarks of Cellular Senescence. Trends Cell Biol. 2018, 28, 436–453. [Google Scholar] [CrossRef]
- Papamichael, D.; Audisio, R.A.; Glimelius, B.; Gramont, A.D.; Glynne-Jones, R.; Haller, D.; Köhne, C.-H.; Rostoft, S.; Lemmens, V.; Mitry, E.; et al. Treatment of colorectal cancer in older patients: International Society of Geriatric Oncology (SIOG) consensus recommendations 2013. Ann. Oncol. 2015, 26, 463–476. [Google Scholar] [CrossRef] [PubMed]
- Prieto, L.I.; Graves, S.I.; Baker, D.J. Insights from In Vivo Studies of Cellular Senescence. Cells 2020, 9, 954. [Google Scholar] [CrossRef]
- Perrie, Y.; Badhan, R.K.S.; Kirby, D.J.; Lowry, D.; Mohammed, A.R.; Ouyang, D. The impact of ageing on the barriers to drug delivery. J. Control. Release 2012, 161, 389–398. [Google Scholar] [CrossRef]
- Toita, R.; Shimizu, E.; Murata, M.; Kang, J.H. Protective and healing effects of apoptotic mimic-induced M2-like macrophage polarization on pressure ulcers in young and middle-aged mice. J. Control. Release Off. J. Control. Release Soc. 2021, 330, 705–714. [Google Scholar] [CrossRef] [PubMed]
- Di Micco, R.; Krizhanovsky, V.; Baker, D.; d’Adda di Fagagna, F. Cellular senescence in ageing: From mechanisms to therapeutic opportunities. Nat. Rev. Mol. Cell Biol. 2020, 22, 75–95. [Google Scholar] [CrossRef] [PubMed]
- Cote, B.; Rao, D.; Alani, A.W. Nanomedicine for Drug Delivery throughout the Alimentary Canal. Mol. Pharm. 2021, 17, 1–22. [Google Scholar] [CrossRef] [PubMed]
- Ha, C.Y.; Katz, S. Clinical implications of ageing for the management of IBD. Nat. Rev. Gastroenterol. Hepatol. 2014, 11, 128–138. [Google Scholar] [CrossRef] [PubMed]
- Nunes, M.M.; Menezes, P.F.; Alves, R.M.; Gonçalves, R.J.; Alexander, J.; Formentin, M.W.; Mendonça, D.F.; Angélica, L.R.; Bueno, M.F.; Patrícia, S.; et al. Chitosan and chitosan/PEG nanoparticles loaded with indole-3-carbinol: Characterization, computational study and potential effect on human bladder cancer cells. Mater. Sci. Eng. C Mater. Biol. Appl. 2021, 124, 12089. [Google Scholar]
- Jain, S.; Spandana, G.; Agrawal, A.K.; Kushwah, V.; Thanki, K. Enhanced Antitumor Efficacy and Reduced Toxicity of Docetaxel Loaded Estradiol Functionalized Stealth Polymeric Nanoparticles. Mol. Pharm. 2015, 12, 3871–3884. [Google Scholar] [CrossRef] [PubMed]
- Barenholz, Y. Doxil®—The first FDA-approved nano-drug: Lessons learned. J. Control. Release 2012, 160, 117–134. [Google Scholar] [CrossRef]
- Andriyanov, A.V.; Portnoy, E.; Koren, E.; Inesa, S.; Eyal, S.; Goldberg, S.N.; Barenholz, Y. Therapeutic efficacy of combined PEGylated liposomal doxorubicin and radiofrequency ablation: Comparing single and combined therapy in young and old mice. J. Control. Release 2017, 257, 2–9. [Google Scholar] [CrossRef] [PubMed]
- Dobrovolskaia, M.A.; Afonin, K.A. Use of human peripheral blood mononuclear cells to define immunological properties of nucleic acid nanoparticles. Nat. Protoc. 2020, 15, 1091–1136. [Google Scholar] [CrossRef]
- Nattayaporn, A.; Sirawit, S.; Arintaya, P.; Thidarat, J.; Sasiwan, K.; Sirintorn, C.; Nisakron, T.; Usanee, M.; Prin, V.; Chagriya, K.; et al. Cognitive impairment is associated with mitochondrial dysfunction in peripheral blood mononuclear cells of elderly population. Sci. Rep. 2020, 10, 21400. [Google Scholar]
- Christopher, D.; Danielle, S. Insights into the role of sialylation in cancer progression and metastasis. Br. J. Cancer 2020, 124, 76–90. [Google Scholar]
- Collado, M.; Serrano, M. Senescence in tumours: Evidence from mice and humans. Nat. Cancer 2010, 10, 51–57. [Google Scholar] [CrossRef] [Green Version]
- Cong, L.; Qiujun, Q.; Min, L.; Xinrong, L.; Ling, H.; Xiang, L.; Chaoyang, L.; Dan, Z.; Hongxia, Z.; Xin, G.; et al. Sialic acid-conjugate modified liposomes targeting neutrophils for improved tumour therapy. Biomater. Sci. 2020, 8, 2189–2201. [Google Scholar]
- Chakradhar, S. A tale of two diseases: Aging HIV patients inspire a closer look at Alzheimer’s disease. Nat. Med. 2018, 24, 376–378. [Google Scholar] [CrossRef]
- Wang, S.; Lai, X.; Deng, Y.; Song, Y. Correlation between mouse age and human age in anti-tumor research: Significance and method establishment. Life Sci. 2019, 242, 117242. [Google Scholar] [CrossRef] [PubMed]
- Ajit, V. Colloquium paper: Uniquely human evolution of sialic acid genetics and biology. Proc. Natl. Acad. Sci. USA 2010, 107 (Suppl. S2), 8939–8946. [Google Scholar]
- Brinkman-Van der Linden, E.C.; Sjoberg, E.R.; Juneja, L.R.; Crocker, P.R.; Varki, N.; Varki, A. Loss of N-glycolylneuraminic acid in human evolution. Implications for sialic acid recognition by siglecs. J. Biol. Chem. 2000, 275, 8633–8640. [Google Scholar] [CrossRef] [Green Version]
- Brinkman-Van der Linden, E.C.; Varki, A. New Aspects of Siglec Binding Specificities, Including the Significance of Fucosylation and of the Sialyl-Tn Epitope. J. Biol. Chem. 2000, 275, 8625–8632. [Google Scholar] [CrossRef] [Green Version]
- Zhou, S.; Zhang, T.; Peng, B.; Luo, X.; Liu, X.; Hu, L.; Liu, Y.; Di, D.; Song, Y.; Deng, Y. Targeted delivery of epirubicin to tumor-associated macrophages by sialic acid-cholesterol conjugate modified liposomes with improved antitumor activity. Int. J. Pharm. 2017, 523, 203–216. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.Y.; Nam, S.; Koo, J.S.; Kim, S.; Yang, M.; Jeong, D.I.; Hwang, C.; Park, J.; Cho, H.J. Possible contribution of sialic acid to the enhanced tumor targeting efficiency of nanoparticles engineered with doxorubicin. Sci. Rep. 2020, 10, 19738. [Google Scholar] [CrossRef] [PubMed]
- Childs, B.G.; Durik, M.; Baker, D.J.; Deursen, J.M.V. Cellular senescence in aging and age-related disease: From mechanisms to therapy. Nat. Med. 2015, 21, 1424–1435. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kawasaki, N.; Vela, J.L.; Nycholat, C.M.; Rademacher, C.; Khurana, A.; Rooijen, N.V.; Crocker, P.R.; Kronenberg, M.; Paulson, J.C. Targeted delivery of lipid antigen to macrophages via the CD169/sialoadhesin endocytic pathway induces robust invariant natural killer T cell activation. Proc. Natl. Acad. Sci. USA 2013, 110, 7826–7831. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hachim, D.; Iftikhar, A.; LoPresti, S.T.; Nolfi, A.L.; Ravichandar, S.; Skillen, C.D.; Brown, B.N. Distinct release strategies are required to modulate macrophage phenotype in young versus aged animals. J. Control. Release 2019, 305, 65–74. [Google Scholar] [CrossRef] [PubMed]
- Kathleen, R.; Sujata, S.; Jessica, A.; Ross, D.; Jonathan, G.; Matthew, J.; Metin, U.; Baoqing, G.; Kyoung-Jin, Y.; David, V.; et al. Single dose combination nanovaccine provides protection against influenza A virus in young and aged mice. Biomater. Sci. 2019, 7, 809–821. [Google Scholar]
- Das, S.; Devarajan, P.V. Enhancing Safety and Efficacy by Altering the Toxic Aggregated State of Amphotericin B in Lipidic Nanoformulations. Mol. Pharm. 2020, 17, 2186–2195. [Google Scholar] [CrossRef]
- Lee, S.; Schmitt, C.A. The dynamic nature of senescence in cancer. Nat. Cell Biol. 2019, 21, 94–101. [Google Scholar] [CrossRef] [PubMed]
- Tsai, S.; Balch, C.; Lange, J. Epidemiology and treatment of melanoma in elderly patients. Nat. Rev. Clin. Oncol. 2010, 7 (Suppl. S1), 148–152. [Google Scholar] [CrossRef]
- Kalyane, D.; Raval, N.; Maheshwari, R.; Tambe, V.; Kalia, K.; Tekade, R.K. Employment of enhanced permeability and retention effect (EPR): Nanoparticle-based precision tools for targeting of therapeutic and diagnostic agent in cancer. Mater. Sci. Eng. C Mater. Biol. Appl. 2019, 98, 1252–1276. [Google Scholar] [CrossRef]


| Drug/Lipid (mg/mg) | Composition (n/n/n) | Particle Size (nm) | Zeta Potential (mV) | PDI | EE (%) | |
|---|---|---|---|---|---|---|
| EPI-SL | 1:10 | SA-ODA/HSPC/CH = 5/50/45 | 110 ± 1 | −18 ± 1 | 0.12 ± 0.01 | 96 ± 2 |
| EPI-PL | 1:10 | PEG2000-DSPE/HSPC/CH = 5/50/45 | 108 ± 1 | −31 ± 2 | 0.11 ± 0.02 | 96 ± 2 |
| DE-SL | 1:50 | SA-ODA/HSPC/CH = 5/50/45 | 114 ± 1 | −17 ± 1 | 0.11 ± 0.02 | 98 ± 3 |
| DE-PL | 1:50 | PEG2000-DSPE/HSPC/CH = 5/50/45 | 109 ± 1 | −31 ± 2 | 0.11 ± 0.01 | 98 ± 2 |
| EPI-S (mg/L) | EPI-SL (mg/L) | EPI-PL (mg/L) | |
|---|---|---|---|
| RAW 264.7 | 1.0 ± 0.1 | 4.2 ± 0.3 | 25 ± 1 |
| S180 | 1.1 ± 0.1 | 7.5 ± 0.7 | 41 ± 3 |
| Kunming Mice | AUC(0–24) (mg/L × h) | V1 (L/kg) | Weight of Mice | Volume of Peripheral Circulating Blood | Density of Peripheral Circulating Blood |
|---|---|---|---|---|---|
| 8M-PL | 1011 ± 28 | 0.057 ± 0.001 | 55 ± 1 g | 1.89 ± 0.03 mL | 1.03 ± 0.04 mg/mL |
| 8M-SL | 632 ± 23 | 0.059 ± 0.002 | 53 ± 1 g | 1.86 ± 0.06 mL | 1.05 ± 0.06 mg/mL |
| 8W-PL | 1092 ± 31 | 0.039 ± 0.001 | 30 ± 2 g | 1.04 ± 0.04 mL | 1.06 ± 0.05 mg/mL |
| 8W-SL | 639 ± 22 | 0.041 ± 0.001 | 33 ± 1 g | 1.01 ± 0.02 mL | 1.09 ± 0.06 mg/mL |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sui, D.; Meng, X.; Li, C.; Tang, X.; Qin, Y.; Zhang, N.; Ding, J.; Liu, X.; Deng, Y.; Song, Y. The Fate of Sialic Acid and PEG Modified Epirubicin Liposomes in Aged versus Young Cells and Tumor Mice Models. Pharmaceutics 2022, 14, 545. https://doi.org/10.3390/pharmaceutics14030545
Sui D, Meng X, Li C, Tang X, Qin Y, Zhang N, Ding J, Liu X, Deng Y, Song Y. The Fate of Sialic Acid and PEG Modified Epirubicin Liposomes in Aged versus Young Cells and Tumor Mice Models. Pharmaceutics. 2022; 14(3):545. https://doi.org/10.3390/pharmaceutics14030545
Chicago/Turabian StyleSui, Dezhi, Xianmin Meng, Changzhi Li, Xueying Tang, Ying Qin, Ning Zhang, Junqiang Ding, Xinrong Liu, Yihui Deng, and Yanzhi Song. 2022. "The Fate of Sialic Acid and PEG Modified Epirubicin Liposomes in Aged versus Young Cells and Tumor Mice Models" Pharmaceutics 14, no. 3: 545. https://doi.org/10.3390/pharmaceutics14030545
APA StyleSui, D., Meng, X., Li, C., Tang, X., Qin, Y., Zhang, N., Ding, J., Liu, X., Deng, Y., & Song, Y. (2022). The Fate of Sialic Acid and PEG Modified Epirubicin Liposomes in Aged versus Young Cells and Tumor Mice Models. Pharmaceutics, 14(3), 545. https://doi.org/10.3390/pharmaceutics14030545






