Antimicrobial Peptides as an Alternative for the Eradication of Bacterial Biofilms of Multi-Drug Resistant Bacteria
Abstract
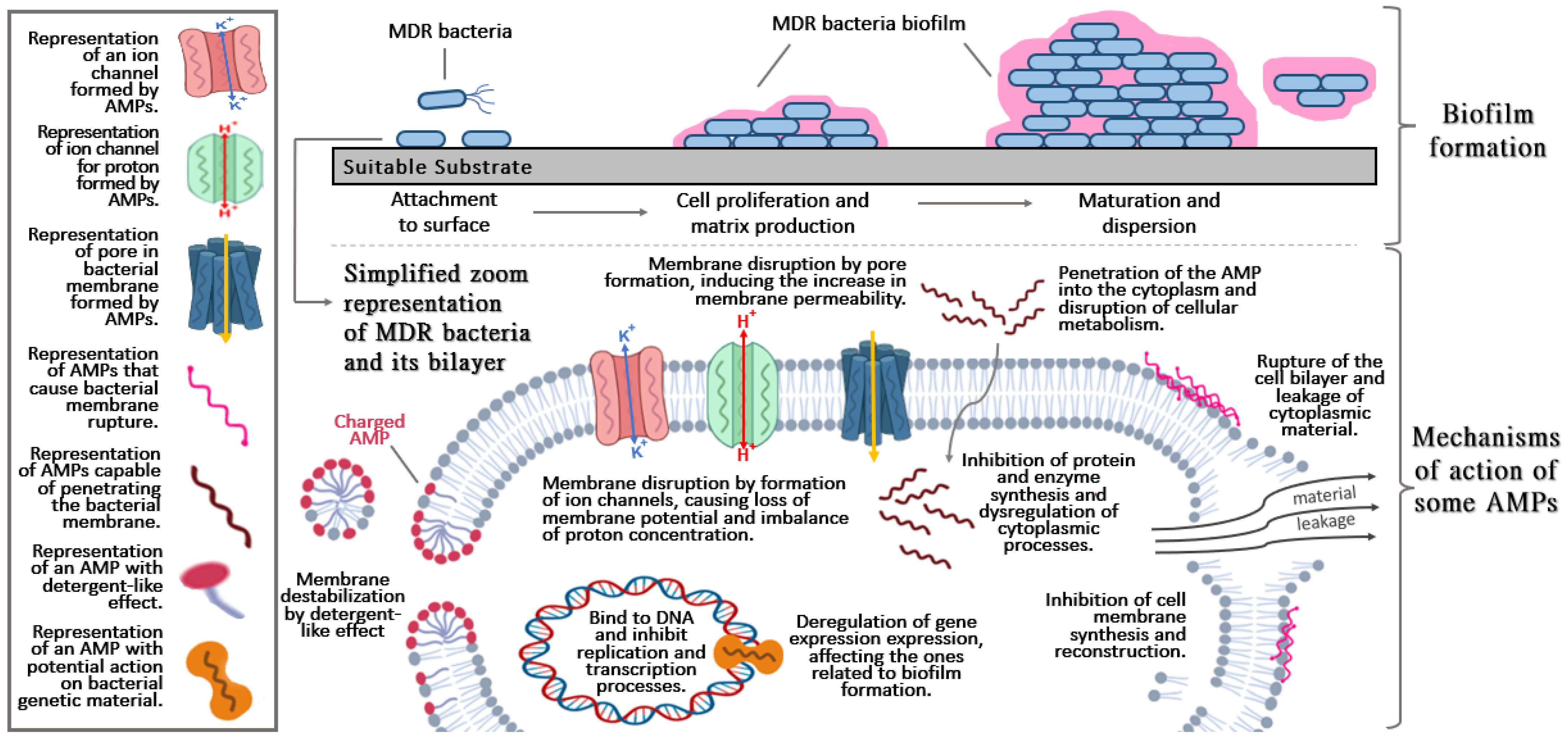
:1. Introduction
1.1. Antimicrobial Resistance
1.2. AMR Mechanisms
1.3. Biofilms
1.4. Control Mechanism
2. Antimicrobial Peptides and Applications
3. Perspectives and Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- WHO. Critically Important Antimicrobials for Human Medicine: Ranking of Antimicrobial Agents for Risk Management of Antimicrobial Resistance due to Non-Human Use, 5th ed.; World Health Organization: Geneva, Switzerland, 2017.
- Sultan, I.; Rahman, S.; Jan, A.T.; Siddiqui, M.T.; Mondal, A.H.; Haq, Q.M.R. Antibiotics, resistome and resistance mechanisms: A bacterial perspective. Front. Microbiol. 2018, 9, 2066. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gedefie, A.; Demsis, W.; Ashagrie, M.; Kassa, Y.; Tesfaye, M.; Tilahun, M.; Bisetegn, H.; Sahle, Z. Acinetobacter baumannii Biofilm Formation and Its Role in Disease Pathogenesis: A Review. Infect. Drug Resist. 2021, 14, 3711. [Google Scholar] [CrossRef] [PubMed]
- Magana, M.; Pushpanathan, M.; Santos, A.L.; Leanse, L.; Fernandez, M.; Ioannidis, A.; Giulianotti, M.A.; Apidianakis, Y.; Bradfute, S.; Ferguson, A.L.; et al. The value of antimicrobial peptides in the age of resistance. Lancet Infect. Dis. 2020, 20, e216–e230. [Google Scholar] [CrossRef]
- Vestby, L.K.; Grønseth, T.; Simm, R.; Nesse, L.L. Bacterial Biofilm and its Role in the Pathogenesis of Disease. Antibiotics 2020, 9, 59. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Jiménez Hernández, M.; Soriano, A.; Filella, X.; Calvo, M.; Coll, E.; Rebled, J.M.; Poch, E.; Graterol, F.; Compte, M.T.; Maduell, F.; et al. Impact of locking solutions on conditioning biofilm formation in tunnelled haemodialysis catheters and inflammatory response activation. J. Vasc. Access 2021, 22, 370–379. [Google Scholar] [CrossRef]
- Roque-Borda, C.A.; Silva, P.B.D.; Rodrigues, M.C.; Azevedo, R.B.; Filippo, L.; Di Duarte, J.L.; Chorilli, M.; Vicente, E.F.; Pavan, F.R. Challenge in the Discovery of New Drugs: Antimicrobial Peptides against WHO-List of Critical and High-Priority Bacteria. Pharmaceutics 2021, 13, 773. [Google Scholar] [CrossRef]
- WHO. Global Priority List of Antibiotic-Resistant Bacteria to Guide Research, Discovery, and Development of New Antibiotics; World Health Organization: Geneva, Switzerland, 2017.
- Tasneem, U.; Yasin, N.; Nisa, I.; Shah, F.; Rasheed, U.; Momin, F.; Zaman, S.; Qasim, M. Biofilm producing bacteria: A serious threat to public health in developing countries. J. Food Sci. Nutr. 2018, 1, 25–31. [Google Scholar] [CrossRef]
- New Report Calls for Urgent Action to Avert Antimicrobial Resistance Crisis. Available online: https://www.who.int/news/item/29-04-2019-new-report-calls-for-urgent-action-to-avert-antimicrobial-resistance-crisis (accessed on 11 July 2021).
- Grant, S.S.; Hung, D.T. Persistent bacterial infections, antibiotic tolerance, and the oxidative stress response. Virulence 2013, 4, 273–283. [Google Scholar] [CrossRef] [Green Version]
- Choi, S.K.; Myc, A.; Silpe, J.E.; Sumit, M.; Wong, P.T.; McCarthy, K.; Desai, A.M.; Thomas, T.P.; Kotlyar, A.; Holl, M.M.B.; et al. Dendrimer-based multivalent vancomycin nanoplatform for targeting the drug-resistant bacterial surface. ACS Nano 2013, 7, 214–228. [Google Scholar] [CrossRef]
- Capita, R.; Alonso-Calleja, C. Antibiotic-Resistant Bacteria: A Challenge for the Food Industry. Crit. Rev. Food Sci. Nutr. 2012, 53, 11–48. [Google Scholar] [CrossRef]
- Loree, J.; Lappin, S.L. Bacteriostatic Antibiotics. Treasure Island, FL: StatPearls Publishing. 2020. Available online: https://www.ncbi.nlm.nih.gov/books/NBK547678/ (accessed on 8 February 2022).
- Woodford, N.; Turton, J.F.; Livermore, D.M. Multiresistant Gram-negative bacteria: The role of high-risk clones in the dissemination of antibiotic resistance. FEMS Microbiol. Rev. 2011, 35, 736–755. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Huttner, A.; Harbarth, S.; Carlet, J.; Cosgrove, S.; Goossens, H.; Holmes, A.; Jarlier, V.; Voss, A.; Pittet, D. Antimicrobial resistance: A global view from the 2013 World Healthcare-Associated Infections Forum. Antimicrob. Resist. Infect. Control 2013, 2, 31. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Perović, S.; Veinović, G.; Stanković, J.A. A review on antibiotic resistance: Origin and mechanisms of bacterial resistance as biological phenomenon. Genetika 2018, 50, 1123–1135. [Google Scholar] [CrossRef] [Green Version]
- Moravej, H.; Moravej, Z.; Yazdanparast, M.; Heiat, M.; Mirhosseini, A.; Moosazadeh Moghaddam, M.; Mirnejad, R. Antimicrobial Peptides: Features, Action, and Their Resistance Mechanisms in Bacteria. Microb. Drug Resist. 2018, 24, 747–767. [Google Scholar] [CrossRef]
- Martinez, J.L. The role of natural environments in the evolution of resistance traits in pathogenic bacteria. Proc. R. Soc. B Biol. Sci. 2009, 276, 2521–2530. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Holmes, A.H.; Moore, L.S.P.; Sundsfjord, A.; Steinbakk, M.; Regmi, S.; Karkey, A.; Guerin, P.J.; Piddock, L.J.V. Understanding the mechanisms and drivers of antimicrobial resistance. Lancet 2016, 387, 176–187. [Google Scholar] [CrossRef]
- Martínez, J.L. Mechanisms of action and of resistance to quinolones. Antibiot. Drug Resist. 2019, 01805, 39–55. [Google Scholar] [CrossRef]
- Khondker, A.; Rheinstädter, M.C. How do bacterial membranes resist polymyxin antibiotics? Commun. Biol. 2020, 3, 77. [Google Scholar] [CrossRef]
- Santos, A.L.S.D.; Galdino, A.C.M.; Mello, T.P.D.; Ramos, L.D.S.; Branquinha, M.H.; Bolognese, A.M.; Columbano Neto, J.; Roudbary, M. What are the advantages of living in a community? A microbial biofilm perspective! Mem. Inst. Oswaldo Cruz 2018, 113, 1–7. [Google Scholar]
- Oliveira, D.; Borges, A.; Simões, M. Staphylococcus aureus Toxins and Their Molecular Activity in Infectious Diseases. Toxins 2018, 10, 252. [Google Scholar] [CrossRef] [Green Version]
- Roy, R.; Tiwari, M.; Donelli, G.; Tiwari, V. Strategies for combating bacterial biofilms: A focus on anti-biofilm agents and their mechanisms of action. Virulence 2018, 9, 522–554. [Google Scholar] [CrossRef] [PubMed]
- Wu, H.; Moser, C.; Wang, H.Z.; Høiby, N.; Song, Z.J. Strategies for combating bacterial biofilm infections. Int. J. Oral Sci. 2015, 7, 1–7. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Herrmann, G.; Yang, L.; Wu, H.; Song, Z.; Wang, H.; Høiby, N.; Ulrich, M.; Molin, S.; Riethmüller, J.; Döring, G. Colistin-tobramycin combinations are superior to monotherapy concerning the killing of biofilm Pseudomonas aeruginosa. J. Infect. Dis. 2010, 202, 1585–1592. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Donelli, G.; Francolini, I.; Piozzi, A.; Rosa, R.D.; Marconi, W. New Polymer-Antibiotic Systems to Inhibit Bacterial Biofilm Formation: A Suitable Approach to Prevent Central Venous Catheter-Associated Infections. J. Chemother. 2002, 14, 501–507. [Google Scholar] [CrossRef]
- Zhao, X.; Yu, Y.; Zhang, X.; Huang, B.; Bai, P.; Xu, C.; Li, D.; Zhang, B.; Liu, C. Decreased biofilm formation ability of Acinetobacter baumannii after spaceflight on China’s Shenzhou 11 spacecraft. Microbiologyopen 2019, 8, 763. [Google Scholar] [CrossRef] [Green Version]
- Oechslin, F. Resistance Development to Bacteriophages Occurring during Bacteriophage Therapy. Viruses 2018, 10, 351. [Google Scholar] [CrossRef] [Green Version]
- Łusiak-Szelachowska, M.; Weber-Dąbrowska, B.; Górski, A. Bacteriophages and Lysins in Biofilm Control. Virol. Sin. 2020, 35, 125–133. [Google Scholar] [CrossRef]
- Passador, L.; Cook, J.M.; Gambello, M.J.; Rust, L.; Iglewski, B.H. Expression of Pseudomonas aeruginosa virulence genes requires cell-to-cell communication. Science 1993, 260, 1127–1130. [Google Scholar] [CrossRef]
- Payne, D.E.; Martin, N.R.; Parzych, K.R.; Rickard, A.H.; Underwood, A.; Boles, B.R. Tannic acid inhibits Staphylococcus aureus surface colonization in an IsaA-dependent manner. Infect. Immun. 2013, 81, 496–504. [Google Scholar] [CrossRef] [Green Version]
- Maurice, N.M.; Bedi, B.; Sadikot, R.T. Pseudomonas aeruginosa biofilms: Host response and clinical implications in lung infections. Am. J. Respir. Cell Mol. Biol. 2018, 58, 428–439. [Google Scholar] [CrossRef]
- Mann, E.E.; Wozniak, D.J. Pseudomonas biofilm matrix composition and niche biology. FEMS Microbiol. Rev. 2012, 36, 893–916. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Guo, Y.; Wang, L.; Lei, J.; Xu, J.; Han, L. Antimicrobial and antibiofilm activity of human cationic antibacterial peptide (Ll-37) and its analogs against Pan-Drug-resistant Acinetobacter baumannii. Jundishapur J. Microbiol. 2017, 10, 4–10. [Google Scholar] [CrossRef]
- Gaddy, J.A.; Actis, L.A. Regulation of Acinetobacter baumannii biofilm formation. Future Microbiol. 2009, 4, 273–278. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Pakharukova, N.; Tuittila, M.; Paavilainen, S.; Malmi, H.; Parilova, O.; Teneberg, S.; Knight, S.D.; Zavialov, A.V. Structural basis for Acinetobacter baumannii biofilm formation. Proc. Natl. Acad. Sci. USA 2018, 115, 5558–5563. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Aslangul, E.; Massias, L.; Meulemans, A.; Chau, F.; Andremont, A.; Courvalin, P.; Fantin, B.; Ruimy, R. Acquired gentamicin resistance by permeability impairment in Enterococcus faecalis. Antimicrob. Agents Chemother. 2006, 50, 3615–3621. [Google Scholar] [CrossRef] [Green Version]
- Mielich-Süss, B.; Lopez, D. Molecular mechanisms involved in Bacillus subtilis biofilm formation. Environ. Microbiol. 2015, 17, 555–565. [Google Scholar] [CrossRef] [Green Version]
- Klausen, M.; Gjermansen, M.; Kreft, J.U.; Tolker-Nielsen, T. Dynamics of development and dispersal in sessile microbial communities: Examples from Pseudomonas aeruginosa and Pseudomonas putida model biofilms. FEMS Microbiol. Lett. 2006, 261, 1–11. [Google Scholar] [CrossRef] [Green Version]
- Ch’ng, J.H.; Chong, K.K.L.; Lam, L.N.; Wong, J.J.; Kline, K.A. Biofilm-associated infection by enterococci. Nat. Rev. Microbiol. 2018, 17, 82–94. [Google Scholar] [CrossRef]
- Dale, J.L.; Nilson, J.L.; Barnes, A.M.T.; Dunny, G.M. Restructuring of Enterococcus faecalis biofilm architecture in response to antibiotic-induced stress. NPJ Biofilms Microbiomes 2017, 3, 15. [Google Scholar] [CrossRef] [Green Version]
- Dale, J.L.; Cagnazzo, J.; Phan, C.Q.; Barnes, A.M.T.; Dunny, G.M. Multiple roles for Enterococcus faecalis glycosyltransferases in biofilm-associated antibiotic resistance, cell envelope integrity, and conjugative transfer. Antimicrob. Agents Chemother. 2015, 59, 4094–4105. [Google Scholar] [CrossRef] [Green Version]
- Fabretti, F.; Theilacker, C.; Baldassarri, L.; Kaczynski, Z.; Kropec, A.; Holst, O.; Huebner, J. Alanine esters of enterococcal lipoteichoic acid play a role in biofilm formation and resistance to antimicrobial peptides. Infect. Immun. 2006, 74, 4164–4171. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Paganelli, F.L.; Willems, R.J.L.W.; Jansen, P.; Hendrickx, A.; Zhang, X.; Bonten, M.J.M.B.; Leavis, H.L. Enterococcus faecium biofilm formation: Identification of major autolysin AtlAefm, associated acm surface localization, and AtlAefm-independent extracellular DNA release. MBio 2013, 4. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Popović, N.; Dinić, M.; Tolinački, M.; Mihajlović, S.; Terzić-Vidojević, A.; Bojić, S.; Djokić, J.; Golić, N.; Veljović, K. New Insight into Biofilm Formation Ability, the Presence of Virulence Genes and Probiotic Potential of Enterococcus sp. Dairy Isolates. Front. Microbiol. 2018, 9, 78. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Top, J.; Paganelli, F.L.; Zhang, X.; van Schaik, W.; Leavis, H.L.; van Luit-Asbroek, M.; van der Poll, T.; Leendertse, M.; Bonten, M.J.M.; Willems, R.J.L. The Enterococcus faecium Enterococcal Biofilm Regulator, EbrB, Regulates the esp Operon and Is Implicated in Biofilm Formation and Intestinal Colonization. PLoS ONE 2013, 8, e65224. [Google Scholar] [CrossRef] [Green Version]
- Heikens, E.; Leendertse, M.; Wijnands, L.M.; Van Luit-Asbroek, M.; Bonten, M.J.M.; Van Der Poll, T.; Willems, R.J.L. Enterococcal surface protein Esp is not essential for cell adhesion and intestinal colonization of Enterococcus faecium in mice. BMC Microbiol. 2009, 9, 19. [Google Scholar] [CrossRef] [Green Version]
- Haney, E.F.; Mansour, S.C.; Hilchie, A.L.; De La Fuente-Núñez, C.; Hancock, R.E.W. High throughput screening methods for assessing antibiofilm and immunomodulatory activities of synthetic peptides. Peptides 2015, 71, 276–285. [Google Scholar] [CrossRef] [Green Version]
- Magalhães, A.P.; Jorge, P.; Pereira, M.O. Pseudomonas aeruginosa and Staphylococcus aureus communication in biofilm infections: Insights through network and database construction. Crit. Rev. Microbiol. 2019, 45, 712–728. [Google Scholar] [CrossRef] [Green Version]
- Corrigan, R.M.; Bellows, L.E.; Wood, A.; Gründling, A. PpGpp negatively impacts ribosome assembly affecting growth and antimicrobial tolerance in Grampositive bacteria. Proc. Natl. Acad. Sci. USA 2016, 113, E1710–E1719. [Google Scholar] [CrossRef] [Green Version]
- Mosselhy, D.A.; Assad, M.; Sironen, T.; Elbahri, M. Nanotheranostics: A Possible Solution for Drug-Resistant Staphylococcus aureus and their Biofilms? Nanomaterials 2021, 11, 82. [Google Scholar] [CrossRef]
- Pamp, S.J.; Frees, D.; Engelmann, S.; Hecker, M.; Ingmer, H. Spx is a global effector impacting stress tolerance and biofilm formation in Staphylococcus aureus. J. Bacteriol. 2006, 188, 4861–4870. [Google Scholar] [CrossRef] [Green Version]
- Werner, G.; Freitas, A.R.; Coque, T.M.; Sollid, J.E.; Lester, C.; Hammerum, A.M.; Garcia-Migura, L.; Jensen, L.B.; Francia, M.V.; Witte, W.; et al. Host range of enterococcal vanA plasmids among Gram-positive intestinal bacteria. J. Antimicrob. Chemother. 2011, 66, 273–282. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Weigel, L.M.; Donlan, R.M.; Shin, D.H.; Jensen, B.; Clark, N.C.; McDougal, L.K.; Zhu, W.; Musser, K.A.; Thompson, J.; Kohlerschmidt, D.; et al. High-level vancomycin-resistant Staphylococcus aureus isolates associated with a polymicrobial biofilm. Antimicrob. Agents Chemother. 2007, 51, 231–238. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- González, S.; Fernández, L.; Campelo, A.B.; Gutiérrez, D.; Martínez, B.; Rodríguez, A.; García, P. The behavior of Staphylococcus aureus dual-species biofilms treated with bacteriophage phiIPLA-RODI depends on the accompanying microorganism. Appl. Environ. Microbiol. 2017, 83, 2821–2837. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Vuotto, C.; Longo, F.; Balice, M.P.; Donelli, G.; Varaldo, P.E. Antibiotic resistance related to biofilm formation in Klebsiella pneumoniae. Pathogens 2014, 3, 743–758. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chakraborty, P.; Bajeli, S.; Kaushal, D.; Radotra, B.D.; Kumar, A. Biofilm formation in the lung contributes to virulence and drug tolerance of Mycobacterium tuberculosis. Nat. Commun. 2021, 12, 1606. [Google Scholar] [CrossRef] [PubMed]
- AlMatar, M.; Makky, E.A.; Yakıcı, G.; Var, I.; Kayar, B.; Köksal, F. Antimicrobial peptides as an alternative to anti-tuberculosis drugs. Pharmacol. Res. 2018, 128, 288–305. [Google Scholar] [CrossRef]
- Silveira, R.F.; Roque Borda, C.A.; Vicente, E.F. Antimicrobial peptides as a feed additive alternative to animal production, food safety and public health implications: An overview. Anim. Nutr. 2021, 7, 896–904. [Google Scholar] [CrossRef]
- Roque-Borda, C.A.; Souza Saraiva, M.d.M.; Monte, D.F.M.; Rodrigues Alves, L.B.; de Almeida, A.M.; Ferreira, T.S.; de Lima, T.S.; Benevides, V.P.; Cabrera, J.M.; Claire, S.; et al. HPMCAS-Coated Alginate Microparticles Loaded with Ctx(Ile21)-Ha as a Promising Antimicrobial Agent against Salmonella Enteritidis in a Chicken Infection Model. ACS Infect. Dis. 2022, 8, 472–481. [Google Scholar] [CrossRef]
- Mahlapuu, M.; Håkansson, J.; Ringstad, L.; Björn, C. Antimicrobial Peptides: An Emerging Category of Therapeutic Agents. Front. Cell. Infect. Microbiol. 2016, 6, 194. [Google Scholar] [CrossRef] [Green Version]
- Roque-Borda, C.A.; Pereira, L.P.; Guastalli, E.A.L.; Soares, N.M.; Mac-Lean, P.A.B.; Salgado, D.D.; Meneguin, A.B.; Chorilli, M.; Vicente, E.F. Hpmcp-coated microcapsules containing the ctx(Ile21)-ha antimicrobial peptide reduce the mortality rate caused by resistant salmonella enteritidis in laying hens. Antibiotics 2021, 10, 616. [Google Scholar] [CrossRef]
- Morroni, G.; Simonetti, O.; Brenciani, A.; Brescini, L.; Kamysz, W.; Kamysz, E.; Neubauer, D.; Caffarini, M.; Orciani, M.; Giovanetti, E.; et al. In vitro activity of Protegrin-1, alone and in combination with clinically useful antibiotics, against Acinetobacter baumannii strains isolated from surgical wounds. Med. Microbiol. Immunol. 2019, 208, 877–883. [Google Scholar] [CrossRef] [PubMed]
- Martinez, M.; Gonçalves, S.; Felício, M.R.; Maturana, P.; Santos, N.C.; Semorile, L.; Hollmann, A.; Maffía, P.C. Synergistic and antibiofilm activity of the antimicrobial peptide P5 against carbapenem-resistant Pseudomonas aeruginosa. Biochim. Biophys. Acta-Biomembr. 2019, 1861, 1329–1337. [Google Scholar] [CrossRef] [PubMed]
- Pletzer, D.; Hancock, R.E.W. Antibiofilm peptides: Potential as broadspectrum agents. J. Bacteriol. 2016, 198, 2572–2578. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Raheem, N.; Straus, S.K. Mechanisms of Action for Antimicrobial Peptides With Antibacterial and Antibiofilm Functions. Front. Microbiol. 2019, 10, 2866. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wu, Q.; Patočka, J.; Kuča, K. Insect Antimicrobial Peptides, a Mini Review. Toxins 2018, 10, 461. [Google Scholar] [CrossRef]
- Seyfi, R.; Kahaki, F.A.; Ebrahimi, T.; Montazersaheb, S.; Eyvazi, S.; Babaeipour, V.; Tarhriz, V. Antimicrobial Peptides (AMPs): Roles, Functions and Mechanism of Action. Int. J. Pept. Res. Ther. 2020, 26, 1451–1463. [Google Scholar] [CrossRef]
- Galdiero, E.; Lombardi, L.; Falanga, A.; Libralato, G.; Guida, M.; Carotenuto, R. Biofilms: Novel Strategies Based on Antimicrobial Peptides. Pharmaceuticals 2019, 11, 322. [Google Scholar] [CrossRef] [Green Version]
- Pulido, D.; Nogús, M.V.; Boix, E.; Torrent, M. Lipopolysaccharide Neutralization by Antimicrobial Peptides: A Gambit in the Innate Host Defense Strategy. J. Innate Immun. 2012, 4, 327. [Google Scholar] [CrossRef]
- Vicente, E.F.; Basso, L.G.M.; Crusca Junior, E.; Roque-Borda, C.A.; Costa-Filho, A.J.; Cilli, E.M. Biophysical Studies of TOAC Analogs of the Ctx(Ile21)-Ha Antimicrobial Peptide Using Liposomes. Braz. J. Phys. 2022, 52, 71. [Google Scholar] [CrossRef]
- Yasir, M.; Willcox, M.D.P.; Dutta, D. Action of Antimicrobial Peptides against Bacterial Biofilms. Materials 2018, 11, 2468. [Google Scholar] [CrossRef] [Green Version]
- Peng, J.; Long, H.; Liu, W.; Wu, Z.; Wang, T.; Zeng, Z.; Guo, G.; Wu, J. Antibacterial mechanism of peptide Cec4 against Acinetobacter baumannii. Infect. Drug Resist. 2019, 12, 2417–2428. [Google Scholar] [CrossRef] [Green Version]
- Liu, L.; Liu, J.; Cui, Q.; Jia, B.Y.; Pei, Z.H.; Odah, K.A.; Wang, Y.M.; Dong, W.L.; Kong, L.C.; Ma, H.X. Design and Characterization of a Novel Hybrid Antimicrobial Peptide OM19R Based on Oncocin and MDAP-2. Int. J. Pept. Res. Ther. 2020, 26, 1839–1846. [Google Scholar] [CrossRef]
- Mwangi, J.; Yin, Y.; Wang, G.; Yang, M.; Li, Y.; Zhang, Z.; Lai, R. The antimicrobial peptide ZY4 combats multidrugresistant Pseudomonas aeruginosa and Acinetobacter baumannii infection. Proc. Natl. Acad. Sci. USA 2019, 116, 26516–26522. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Xiong, Y.Q.; Li, L.; Zhou, Y.; Kraus, C.N. Efficacy of ARV-1502, a Proline-Rich Antimicrobial Peptide, in a Murine Model of Bacteremia Caused by Multi-Drug Resistant (MDR) Acinetobacter baumannii. Molecules 2019, 24, 2820. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Mohan, N.M.; Zorgani, A.; Jalowicki, G.; Kerr, A.; Khaldi, N.; Martins, M. Unlocking NuriPep 1653 From Common Pea Protein: A Potent Antimicrobial Peptide to Tackle a Pan-Drug Resistant Acinetobacter baumannii. Front. Microbiol. 2019, 10, 2086. [Google Scholar] [CrossRef] [PubMed]
- Nagarajan, D.; Roy, N.; Kulkarni, O.; Nanajkar, N.; Datey, A.; Ravichandran, S.; Thakur, C.; Sandeep, T.; Aprameya, I.V.; Sarma, S.P.; et al. W76: A designed antimicrobial peptide to combat carbapenem-And tigecycline-resistant Acinetobacter baumannii. Sci. Adv. 2019, 5, 1946. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Riool, M.; de Breij, A.; Kwakman, P.H.S.; Schonkeren-Ravensbergen, E.; de Boer, L.; Cordfunke, R.A.; Malanovic, N.; Drijfhout, J.W.; Nibbering, P.H.; Zaat, S.A.J. Thrombocidin-1-derived antimicrobial peptide TC19 combats superficial multi-drug resistant bacterial wound infections. Biochim. Biophys. Acta-Biomembr. 2020, 1862, 183282. [Google Scholar] [CrossRef] [PubMed]
- Porto, W.F.; Irazazabal, L.N.; Humblot, V.; Haney, E.F.; Ribeiro, S.M.; Hancock, R.E.W.; Ladram, A.; Franco, O.L. EcDBS1R6: A novel cationic antimicrobial peptide derived from a signal peptide sequence. Biochim. Biophys. Acta-Gen. Subj. 2020, 1864, 129633. [Google Scholar] [CrossRef]
- Coyotl, E.A.P.; Palacios, J.B.; Muciño, G.; Moreno-Blas, D.; Costas, M.; Montes, T.M.; Diener, C.; Uribe-Carvajal, S.; Massieu, L.; Castro-Obregón, S.; et al. Antimicrobial peptide against mycobacterium tuberculosis that activates autophagy is an effective treatment for tuberculosis. Pharmaceutics 2020, 12, 1071. [Google Scholar] [CrossRef]
- Brunetti, J.; Carnicelli, V.; Ponzi, A.; Di Giulio, A.; Lizzi, A.R.; Cristiano, L.; Cresti, L.; Cappello, G.; Pollini, S.; Mosconi, L.; et al. Antibacterial and anti-inflammatory activity of an antimicrobial peptide synthesized with D amino acids. Antibiotics 2020, 9, 840. [Google Scholar] [CrossRef]
- Yin, Q.; Wu, S.; Wu, L.; Wang, Z.; Mu, Y.; Zhang, R.; Dong, C.; Zhou, B.; Zhao, B.; Zheng, J.; et al. A novel in silico antimicrobial peptide DP7 combats MDR Pseudomonas aeruginosa and related biofilm infections. J. Antimicrob. Chemother. 2020, 75, 3248–3259. [Google Scholar] [CrossRef] [PubMed]
- Parducho, K.R.; Beadell, B.; Ybarra, T.K.; Bush, M.; Escalera, E.; Trejos, A.T.; Chieng, A.; Mendez, M.; Anderson, C.; Park, H.; et al. The Antimicrobial Peptide Human Beta-Defensin 2 Inhibits Biofilm Production of Pseudomonas aeruginosa Without Compromising Metabolic Activity. Front. Immunol. 2020, 11, 805. [Google Scholar] [CrossRef] [PubMed]
- Hazam, P.K.; Chen, J.Y. Therapeutic utility of the antimicrobial peptide Tilapia Piscidin 4 (TP4). Aquac. Rep. 2020, 17, 100409. [Google Scholar] [CrossRef]
- Abraham, P.; Jose, L.; Maliekal, T.T.; Kumar, R.A.; Kumar, K.S. B1CTcu5: A frog-derived brevinin-1 peptide with anti-tuberculosis activity. Peptides 2020, 132, 170373. [Google Scholar] [CrossRef] [PubMed]
- Witherell, K.S.; Price, J.; Bandaranayake, A.D.; Olson, J.; Call, D.R. In vitro activity of antimicrobial peptide CDP-B11 alone and in combination with colistin against colistin-resistant and multidrug-resistant Escherichia coli. Sci. Rep. 2021, 11, 2151. [Google Scholar] [CrossRef]
- Roque-Borda, C.A.; Silva, H.R.L.; Crusca Junior, E.; Serafim, J.A.; Meneguin, A.B.; Chorilli, M.; Macedo, W.C.; Teixeira, S.R.; Guastalli, E.A.L.; Soares, N.M.; et al. Alginate-based microparticles coated with HPMCP/AS cellulose-derivatives enable the Ctx(Ile21)-Ha antimicrobial peptide application as a feed additive. Int. J. Biol. Macromol. 2021, 183, 1236–1247. [Google Scholar] [CrossRef]
- Tenland, E.; Pochert, A.; Krishnan, N.; Rao, K.U.; Kalsum, S.; Braun, K.; Glegola-Madejska, I.; Lerm, M.; Robertson, B.D.; Lindén, M.; et al. Effective delivery of the anti-mycobacterial peptide NZX in mesoporous silica nanoparticles. PLoS ONE 2019, 14, e0212858. [Google Scholar] [CrossRef] [Green Version]
- Mousavizadegan, M.; Mohabatkar, H. Computational prediction of antifungal peptides via Chou’s PseAAC and SVM. J. Bioinform. Comput. Biol. 2018, 16, 1850016. [Google Scholar] [CrossRef]
- De Aguiar, F.L.L.; Santos, N.C.; Cavalcante, C.S.d.P.; Andreu, D.; Baptista, G.R.; Gonçalves, S. Antibiofilm activity on candida albicans and mechanism of action on biomembrane models of the antimicrobial peptide Ctn[15–34]. Int. J. Mol. Sci. 2020, 21, 8339. [Google Scholar] [CrossRef]
- Do Nascimento Dias, J.; de Souza Silva, C.; de Araújo, A.R.; Souza, J.M.T.; de Holanda Veloso Júnior, P.H.; Cabral, W.F.; da Glória da Silva, M.; Eaton, P.; de Souza de Almeida Leite, J.R.; Nicola, A.M.; et al. Mechanisms of action of antimicrobial peptides ToAP2 and NDBP-5 7 against Candida albicans planktonic and biofilm cells. Sci. Rep. 2020, 10, 10327. [Google Scholar] [CrossRef]
- Seyedjavadi, S.S.; Khani, S.; Eslamifar, A.; Ajdary, S.; Goudarzi, M.; Halabian, R.; Akbari, R.; Zare-Zardini, H.; Imani Fooladi, A.A.; Amani, J.; et al. The Antifungal Peptide MCh-AMP1 Derived From Matricaria chamomilla Inhibits Candida albicans Growth via Inducing ROS Generation and Altering Fungal Cell Membrane Permeability. Front. Microbiol. 2020, 10, 3150. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ramamourthy, G.; Park, J.; Seo, C.; Vogel, H.J.; Park, Y. Antifungal and antibiofilm activities and the mechanism of action of repeating lysine-tryptophan peptides against candida albicans. Microorganisms 2020, 8, 758. [Google Scholar] [CrossRef] [PubMed]
- Zhao, H.; To, K.K.W.; Sze, K.H.; Yung, T.T.M.; Bian, M.; Lam, H.; Yeung, M.L.; Li, C.; Chu, H.; Yuen, K.Y. A broad-spectrum virus- and host-targeting peptide against respiratory viruses including influenza virus and SARS-CoV-2. Nat. Commun. 2020, 11, 4252. [Google Scholar] [CrossRef] [PubMed]
- Pen, G.; Yang, N.; Teng, D.; Mao, R.; Hao, Y.; Wang, J. A review on the use of antimicrobial peptides to combat porcine viruses. Antibiotics 2020, 9, 801. [Google Scholar] [CrossRef] [PubMed]
- Wang, H.Y.; Lin, L.; Tan, L.S.; Yu, H.Y.; Cheng, J.W.; Pan, Y.P. Molecular pathways underlying inhibitory effect of antimicrobial peptide Nal-P-113 on bacteria biofilms formation of Porphyromonas gingivalis W83 by DNA microarray. BMC Microbiol. 2017, 17, 37. [Google Scholar] [CrossRef] [Green Version]
- Yazici, H.; O’Neill, M.B.; Kacar, T.; Wilson, B.R.; Emre Oren, E.; Sarikaya, M.; Tamerler, C. Engineered Chimeric Peptides as Antimicrobial Surface Coating Agents toward Infection-Free Implants HHS Public Access. ACS Appl. Mater. Interfaces 2016, 8, 5070–5081. [Google Scholar] [CrossRef] [Green Version]
- Pletzer, D.; Mansour, S.C.; Hancock, R.E.W. Synergy between conventional antibiotics and anti-biofilm peptides in a murine, sub-cutaneous abscess model caused by recalcitrant ESKAPE pathogens. PLoS Pathog. 2018, 14, e1007084. [Google Scholar] [CrossRef]
- Otvos, L.; Ostorhazi, E.; Szabo, D.; Zumbrun, S.D.; Miller, L.L.; Halasohoris, S.A.; Desai, P.D.; Veldt, S.M.I.; Kraus, C.N. Synergy between proline-rich antimicrobial peptides and small molecule antibiotics against selected Gram-Negative pathogens in vitro and in vivo. Front. Chem. 2018, 6, 309. [Google Scholar] [CrossRef]
- Li, W.; Separovic, F.; O’Brien-Simpson, N.M.; Wade, J.D. Chemically modified and conjugated antimicrobial peptides against superbugs. Chem. Soc. Rev. 2021, 50, 4932–4973. [Google Scholar] [CrossRef]
- Dong, W.; Luo, X.; Sun, Y.; Li, Y.; Wang, C.; Guan, Y.; Shang, D. Binding Properties of DNA and Antimicrobial Peptide Chensinin-1b Containing Lipophilic Alkyl Tails. J. Fluoresc. 2020, 30, 131–142. [Google Scholar] [CrossRef]
- Li, W.; Lin, F.; Hung, A.; Barlow, A.; Sani, M.-A.; Paolini, R.; Singleton, W.; Holden, J.; Hossain, M.A.; Separovic, F.; et al. Enhancing proline-rich antimicrobial peptide action by homodimerization: Influence of bifunctional linker. Chem. Sci. 2022, 13, 2226–2237. [Google Scholar] [CrossRef]
- Han, H.M.; Gopal, R.; Park, Y. Design and membrane-disruption mechanism of charge-enriched AMPs exhibiting cell selectivity, high-salt resistance, and anti-biofilm properties. Amin. Acids 2015, 48, 505–522. [Google Scholar] [CrossRef] [PubMed]
- Dosler, S.; Karaaslan, E.; Gerceker, A.A. Antibacterial and anti-biofilm activities of melittin and colistin, alone and in combination with antibiotics against Gram-negative bacteria. J. Chemother. 2016, 28, 95–103. [Google Scholar] [CrossRef]
- Quilès, F.; Saadi, S.; Francius, G.; Bacharouche, J.; Humbert, F. In situ and real time investigation of the evolution of a Pseudomonas fluorescens nascent biofilm in the presence of an antimicrobial peptide. Biochim. Biophys. Acta-Biomembr. 2016, 1858, 75–84. [Google Scholar] [CrossRef]
- Maciejewska, M.; Bauer, M.; Neubauer, D.; Kamysz, W.; Dawgul, M. Influence of Amphibian Antimicrobial Peptides and Short Lipopeptides on Bacterial Biofilms Formed on Contact Lenses. Materials 2016, 9, 873. [Google Scholar] [CrossRef] [Green Version]
- Huang, Z.; Shi, X.; Mao, J.; Gong, S. Design of a hydroxyapatite-binding antimicrobial peptide with improved retention and antibacterial efficacy for oral pathogen control. Sci. Rep. 2016, 6, 38410. [Google Scholar] [CrossRef] [Green Version]
- De Gier, M.G.; Bauke Albada, H.; Josten, M.; Willems, R.; Leavis, H.; Van Mansveld, R.; Paganelli, F.L.; Dekker, B.; Lammers, J.W.J.; Sahl, H.G.; et al. Synergistic activity of a short lipidated antimicrobial peptide (lipoAMP) and colistin or tobramycin against Pseudomonas aeruginosa from cystic fibrosis patients. Medchemcomm 2016, 7, 148–156. [Google Scholar] [CrossRef] [Green Version]
- Zapotoczna, M.; Forde, É.; Hogan, S.; Humphreys, H.; O’Gara, J.P.; Fitzgerald-Hughes, D.; Devocelle, M.; O’Neill, E. Eradication of Staphylococcus aureus Biofilm Infections Using Synthetic Antimicrobial Peptides. J. Infect. Dis. 2017, 215, 975–983. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- LL-37-Derived Membrane-active FK-13 Analogs Possessing Cell Selectivity, Anti-Biofilm Activity and synergy with Chloramphenicol and Anti-Inflammatory Activity-ScienceDirect. Available online: https://www-sciencedirect.ez87.periodicos.capes.gov.br/science/article/pii/S0005273617300457?via%3Dihub (accessed on 10 October 2021).
- LL-37-Derived Short Antimicrobial Peptide KR-12-a5 and its D-Amino acid Substituted Analogs with Cell Selectivity, Anti-Biofilm Activity, Synergistic Effect with Conventional Antibiotics, and Anti-Inflammatory Activity-ScienceDirect. Available online: https://www-sciencedirect.ez87.periodicos.capes.gov.br/science/article/pii/S0223523417303902?via%3Dihub (accessed on 10 October 2021).
- Bormann, N.; Koliszak, A.; Kasper, S.; Schoen, L.; Hilpert, K.; Volkmer, R.; Kikhney, J.; Wildemann, B. A short artificial antimicrobial peptide shows potential to prevent or treat bone infections. Sci. Rep. 2017, 7, 1506. [Google Scholar] [CrossRef] [Green Version]
- Wang, Y.; Fan, Y.; Zhou, Z.; Tu, H.; Ren, Q.; Wang, X.; Ding, L.; Zhou, X.; Zhang, L. De novo synthetic short antimicrobial peptides against cariogenic bacteria. Arch. Oral Biol. 2017, 80, 41–50. [Google Scholar] [CrossRef] [PubMed]
- Chang, T.-W.; Wei, S.-Y.; Wang, S.-H.; Wei, H.-M.; Wang, Y.-J.; Wang, C.-F.; Chen, C.; Liao, Y.-D. Hydrophobic residues are critical for the helix-forming, hemolytic and bactericidal activities of amphipathic antimicrobial peptide TP4. PLoS ONE 2017, 12, e0186442. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Olivo, E.A.C.; Santos, D.; de Lima, M.E.; dos Santos, V.L.; Sinisterra, R.D.; Cortés, M.E. Antibacterial Effect of Synthetic Peptide LyeTxI and LyeTxI/β-Cyclodextrin Association Compound Against Planktonic and Multispecies Biofilms of Periodontal Pathogens. J. Periodontol. 2017, 88, e88–e96. [Google Scholar] [CrossRef] [PubMed]
- Loffredo, M.R.; Ghosh, A.; Harmouche, N.; Casciaro, B.; Luca, V.; Bortolotti, A.; Cappiello, F.; Stella, L.; Bhunia, A.; Bechinger, B.; et al. Membrane perturbing activities and structural properties of the frog-skin derived peptide Esculentin-1a(1-21)NH2 and its Diastereomer Esc(1-21)-1c: Correlation with their antipseudomonal and cytotoxic activity. Biochim. Biophys. Acta-Biomembr. 2017, 1859, 2327–2339. [Google Scholar] [CrossRef] [PubMed]
- Khara, J.S.; Obuobi, S.; Wang, Y.; Hamilton, M.S.; Robertson, B.D.; Newton, S.M.; Yang, Y.Y.; Langford, P.R.; Ee, P.L.R. Disruption of drug-resistant biofilms using de novo designed short α-helical antimicrobial peptides with idealized facial amphiphilicity. Acta Biomater. 2017, 57, 103–114. [Google Scholar] [CrossRef]
- Lin, Q.; Deslouches, B.; Montelaro, R.C.; Di, Y.P. Prevention of ESKAPE pathogen biofilm formation by antimicrobial peptides WLBU2 and LL37. Int. J. Antimicrob. Agents 2018, 52, 667–672. [Google Scholar] [CrossRef] [PubMed]
- Deslouches, B.; Gonzalez, I.A.; DeAlmeida, D.; Islam, K.; Steele, C.; Montelaro, R.C.; Mietzner, T.A. De novo-derived cationic antimicrobial peptide activity in a murine model of Pseudomonas aeruginosa bacteraemia. J. Antimicrob. Chemother. 2007, 60, 669–672. [Google Scholar] [CrossRef]
- Haisma, E.M.; De Breij, A.; Chan, H.; Van Dissel, J.T.; Drijfhout, J.W.; Hiemstra, P.S.; El Ghalbzouri, A.; Nibbering, P.H. LL-37-derived peptides eradicate multidrug-resistant Staphylococcus aureus from thermally wounded human skin equivalents. Antimicrob. Agents Chemother. 2014, 58, 4411–4419. [Google Scholar] [CrossRef] [Green Version]
- De Breij, A.; Riool, M.; Cordfunke, R.A.; Malanovic, N.; De Boer, L.; Koning, R.I.; Ravensbergen, E.; Franken, M.; Van Der Heijde, T.; Boekema, B.K.; et al. The antimicrobial peptide SAAP-148 combats drug-resistant bacteria and biofilms. Sci. Transl. Med. 2018, 10. [Google Scholar] [CrossRef] [Green Version]
- Spencer, J.J.; Pitts, R.E.; Pearson, R.A.; King, L.B. The effects of antimicrobial peptides WAM-1 and LL-37 on multidrug-resistant Acinetobacter baumannii. Pathog. Dis. 2018, 76, 7. [Google Scholar] [CrossRef]
- Almaaytah, A.; Qaoud, M.T.; Abualhaijaa, A.; Al-Balas, Q.; Alzoubi, K.H. Hybridization and antibiotic synergism as a tool for reducing the cytotoxicity of antimicrobial peptides. Infect. Drug Resist. 2018, 11, 835–847. [Google Scholar] [CrossRef] [Green Version]
- Mohanraj, G.; Mao, C.; Armine, A.; Kasher, R.; Arnusch, C.J. Ink-Jet Printing-Assisted Modification on Polyethersulfone Membranes Using a UV-Reactive Antimicrobial Peptide for Fouling-Resistant Surfaces. ACS Omega 2018, 3, 8752–8759. [Google Scholar] [CrossRef] [PubMed]
- Kang, H.K.; Seo, C.H.; Luchian, T.; Park, Y. Pse-T2, an antimicrobial peptide with high-level, broad-spectrum antimicrobial potency and skin biocompatibility against multidrug-resistant Pseudomonas aeruginosa infection. Antimicrob. Agents Chemother. 2018, 62, e01493-18. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kim, M.K.; Kang, N.H.; Ko, S.J.; Park, J.; Park, E.; Shin, D.W.; Kim, S.H.; Lee, S.A.; Lee, J.I.; Lee, S.H.; et al. Antibacterial and Antibiofilm Activity and Mode of Action of Magainin 2 against Drug-Resistant Acinetobacter baumannii. Int. J. Mol. Sci. 2018, 19, 3041. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Cardoso, M.H.; de Almeida, K.C.; Cândido, E.S.; Fernandes, G.d.R.; Dias, S.C.; de Alencar, S.A.; Franco, O.L. Comparative transcriptome analyses of magainin I-susceptible and -resistant Escherichia coli strains. Microbiology 2018, 164, 1383–1393. [Google Scholar] [CrossRef]
- Omardien, S.; Drijfhout, J.W.; Zaat, S.A.; Brul, S. Cationic Amphipathic Antimicrobial Peptides Perturb the Inner Membrane of Germinated Spores Thus Inhibiting Their Outgrowth. Front. Microbiol. 2018, 9, 2277. [Google Scholar] [CrossRef] [Green Version]
- Omardien, S.; Drijfhout, J.W.; van Veen, H.; Schachtschabel, S.; Riool, M.; Hamoen, L.W.; Brul, S.; Zaat, S.A.J. Synthetic antimicrobial peptides delocalize membrane bound proteins thereby inducing a cell envelope stress response. Biochim. Biophys. Acta-Biomembr. 2018, 1860, 2416–2427. [Google Scholar] [CrossRef]
- Kaletta, C.; Entian, K.D. Nisin, a peptide antibiotic: Cloning and sequencing of the nisA gene and posttranslational processing of its peptide product. J. Bacteriol. 1989, 171, 1597–1601. [Google Scholar] [CrossRef] [Green Version]
- Teixeira, M.C.; Carbone, C.; Sousa, M.C.; Espina, M.; Garcia, M.L.; Sanchez-Lopez, E.; Souto, E.B. Nanomedicines for the Delivery of Antimicrobial Peptides (AMPs). Nanomaterials 2020, 10, 560. [Google Scholar] [CrossRef] [Green Version]
| Bacteria | Mechanism of Biofilm Formation |
|---|---|
| Pseudomonas aeruginosa | Pseudomonas aeruginosa is one of the main opportunistic pathogens due to its high virulence, causing great concern in terms of public health. This microorganism is associated with several serious infections such as ventilator-associated pneumonia and chronic infections in cases of cystic fibrosis. P. aeruginosa has a pathogenic profile that makes it capable of developing resistance to several antibiotics. This is due to its complex genome, its various virulence factors, and its ability to form powerful biofilms [34]. P. aeruginosa forms a dense biofilm as their microcolonies start forming stalks that eventually develop into “mushroom” structures. This bacterial species requires polysaccharides in the extracellular matrix of its biofilm at most stages of maturation. Two classes of polysaccharides are important in biofilm formation: capsular polysaccharides, which maintain the outer structure of cells with dynamic protective polymers, and aggregative polysaccharides, which offer structural integrity and interaction with other matrix components, conferring an adhesive and aggregative capacity to the biofilm, mainly by the alginate produced by P. aeruginosa [35]. |
| Acinetobacter baumannii | Acinetobacter baumannii is a Gram-negative opportunistic nosocomial pathogen, known to have multi-resistance to various antibiotics due to, among other factors, its major virulence, which is its ability to produce biofilms [36]. This bacterium has the ability to survive and proliferate in favorable and unfavorable environments, such as hospital equipment. This ability to colonize abiotic environments (such as polystyrene and glass) is believed to be associated with its power to form characteristic biofilms [37]. An important organelle responsible for the initial fixation of this microorganism to surfaces is the pilus, which allows bacterial adhesion to different structures of substrates. This pilus has a hydrophobic tip, with which it probably bonds to hydrophobic surfaces of various substrates. Therefore, the use of hydrophilic materials instead of hydrophobic plastics, especially in medical devices, can help reduce cases of hospital infections with A. baumannii [38]. |
| Enterococcus sp. | Enterococci are Gram-positive lactic acid bacteria found in the intestinal microbiota, easily adapted to pH, temperature, and salt content conditions. Due to this versatility, its easy transmission is inevitable, even more so when bacteria such as S. faecalis and S. faecium predominate, since the ability of these enterococci to form biofilms increases their resistance to antibiotics. Enterococci have moderate resistance to aminoglycosides and intrinsic resistance to beta-lactams. When a bacterial infection is triggered (more common in patients with weakened immunity), they are capable of forming a niche of resistance genes that are easily transferred to other bacterial species, further aggravating the infection [39]. Unlike Pseudomonas and Bacillus biofilm formation, Enterococcus sp. biofilm formation is still difficult to understand [40,41]. In E. faecalis, several factors seem to be involved. Initially, the enterococci in their planktonic state adhere to the surface and, in this process, many adhesins are present, such as endocarditis and biofilm-associated pilus (Ebp), aggregating substance (Agg), enterococcal surface protein (Esp), E. faecalis collagen adhesin (Ace), proteases, and glycolipids [42]. Subsequently, the rhamnopolysaccharide matrix is formed, a process that has not yet been identified as to the mechanism by which enterococci form a thick or thin biofilm or why they could spread again [42]. However, it is known that the formation of microcolonies is characteristic of a mature stage of the biofilm, which is important for intestinal colonization in response to antibiotics [43,44]. Previous studies showed that the dltABCD operon had a strong relationship with biofilm formation and that its genetic deletion decreased biofilm formation; which allowed a better action of the antimicrobials, mainly of the antimicrobial peptides (AMPs) [45].Vancomycin-resistant Enterococcus faecium (VRE) is considered a priority on the WHO list, since it is also a great biofilm former and the third largest cause of nosocomial infection, especially in patients who require long-term invasive medical devices [46,47]. Several genes involved in in vitro biofilm development were reported, such as atlA, ebpABC, esp, fsrB, luxS, spx, acm, scm, sgrA, pilA, pilB, ecbA and asrR, but only some such as atlA, ebpABC, esp, acm and asrR would be directly associated with the development of a serious infection in vivo [42]. Due to controversial results of Esp expression, it cannot be clarified whether or not biofilm formation is mediated by enterococcal biofilm regulator B [48,49]. Although it is believed that virulence genes are related to the formation of biofilms, it was shown that it is more frequently associated with adhesion properties [47]. |
| Staphylococcus aureus | Vancomycin-resistant, vancomycin-intermediate, and methicillin-resistant Staphylococcus aureus (VRSA, VISA, and MRSA, respectively) are known as a major cause of bacteremia, infective endocarditis, osteoarticular infections, skin and soft tissue infections, pleuropulmonary infections, and invasive-device-related infections. These bacteria can express several resistance mechanisms and virulence factors, which gives them the ability to evade the host’s natural defenses and some drugs [50]. Biofilms are generally associated with chronic and serious acute infections, and the same can be said of the biofilm of S. aureus, which results in a complicated treatment because many of its clinical isolates are MDR bacteria [51]. S. aureus has the ability to express a diverse set of virulence factors that allow it to cause a broad range of pathologies and survive the host immune system. Additionally, heterogeneous biofilms that present S. aureus have a high rate of plasmid horizontal transfer, which increases their antibiotic resistance [24,51]. S. aureus has a multilayered biofilm with a matrix composed of about 80% teichoic acids, staphylococcal proteins, and host proteins [24]. It was previously reported that GTPases would be important biofilm production markers in S. aureus because their inhibition enabled these bacteria to reduce the size of the biofilm and the number of mature 70S ribosomes, allowing the action of conventional antibiotics [52]. In S. areus biofilm-forming, tolerant, resistant, and persistent bacterial cells have been detected; the last group of bacteria is believed to be tolerant to antibiotics, causing recurrent infections and producing antibiotic-susceptible offspring when they resume growth without antibiotics [53].Previous studies recognize that the regulation of Spx expression would be negatively related to biofilm formation, which means that the lower the expression, the greater the maturity of the biofilm matrix and its resistance to stress [54]. It was even reported that this microorganism obtained vancomycin resistance genes (VanA) through gene transfer through VRE biofilms (SA + VanA → VRSA) [55,56]. The scarcity of reports focused on the elimination of multispecies biofilms leads us to think that we are still far from finding an ideal mechanism and drug to eliminate these perfectly organized microcolonies [57]. |
| Klebsiella pneumoniae | Klebsiella pneumoniae (KP) is known to cause many nosocomial infections, particularly in patients using long-term medical devices. One of its virulence factors (also associated with antibiotic resistance) is biofilm formation. Growth of K. pneumoniae on biotic and abiotic surfaces is facilitated by type-3 fimbrial proteins (the MrkA type-3 fimbrial protein for abiotic and adhesin MrkD for biotic surfaces), which have regulated gene expression. Structures such as bacterial capsule and KP’s LPS also contribute to biofilm formation, with LPS being important in the initial adhesion to abiotic surfaces and capsule being important in the structure of a biofilm both in the early and mature stages [58]. |
| Mycobacterium tuberculosis | Tuberculosis, caused by Mycobacterium tuberculosis, is a chronic disease in which the microorganism evades the immune system (hiding in granulomas). There are treatment failures and high reinfection rates, characteristic of infections that exhibit biofilm formation. Cellulose is an important component of M. tuberculosis biofilms, and since humans do not produce this compound, its presence in the lungs of infected people points to biofilm formation. Biofilms play an essential role in establishing M. tuberculosis infection and protection of resident bacilli from the immune system responses and consequent antimycobacterial agents. Symptoms result from the activation of the immune system when some M. tuberculosis bacilli invade and reside in macrophages [59]. |
| AMP | Sequence | Microbial Strains | Highlights | Reference |
|---|---|---|---|---|
| Antibacterial Activity | ||||
| Cec4 | GWLKKIGKKIERVGQNTRD ATIQAIGVAQQAANVAATLKGK | A. baumannii | Mechanism of action based on bacterial membrane rupture. It has activity against standard A. baumannii and MDR strains. | [75] |
| OM19R (MDAP-2 + Oncocin) | VDKPPYLPRPR PIRRPGGR | E. coli, Salmonella, and Shigella | Peptide with great antibacterial activity and no cytotoxicity or hemolytic properties. | [76] |
| ZY4 | VCKRWKKWKRKWKKWCV In the sequence, disulfide bond (C-C) is formed by the Cystein. | P. aeruginosa and A. baumanni | Mechanism of action based on permeabilization of the bacterial membrane. | [77] |
| ARV-1502 | Chex-RPDKPRPTL PRPRPPRPVR | MDR A. baumanni | Promising peptide when combined with standard treatment antibiotics against multi-resistant bacterial infections. | [78] |
| Protegrin-1 | RGGRLCYCRRRFCVCVGR | A. baumannii (XDR andMDR strains fromsurgical wounds) | Highly active at high concentrations. Temporary effect at low concentrations. | [65] |
| NuriPep 1653 | VRGLAPKKSLWPF GGPFKSPFN | Pan-Drug Resistant Acinetobacter baumannii | Activity interrupted by salt sensitivity. Thermostability at 95 °C. | [79] |
| Ω76 peptide | FLKAIKKFGKEFKKIGAKLK | carbapenem- and tigecycline-resistantA. baumannii | Mechanism of action based on the formation of an a-helical structure in bacterial membranes, causing rapid disruption, leakage, and bacterial death. | [80] |
| TC19 | LRCMCIKWWSGKHPK | E. coli and S. aureus | High selectivity for bacterial membranes and low toxicity for human cells. | [81] |
| EcDBS1R6 | PMKKLFKLLARIAVKIPVW | E. coli, P. aeruginosa, K. pneumoniae, A. baumannii | Cationic AMPs derived from a signal peptide sequence. | [82] |
| Iztli peptide 1 (IP-1) | KFLNRFWHWLQLKPGQPMY | M. tuberculosis | Mechanism of action against M. tuberculosis (MTB) based on the induction of autophagy in infected macrophages, thus preventing the release of MTB to new cells and directly killing the microorganism. | [83] |
| SET-M33 (protease resistant) | (KKIRVRLSA)4K2KβA-OH | S. aureus (6 strains MDR/XDR), P. aeruginosa (7 strains MDR/XDR), A. baumannii (3 strains MDR/XDR), E. coli (8 strains MDR/XDR), K. pneumoniae (5 strains MDR/XDR) | The studies showed promising results in vitro and in vivo (5 and 2.5 mg/Kg) and showed anti-inflammatory power, decreasing the production of TNF-α, IL6, COX-2, KC, MIP-1, IP10, iNOS, NF-κB. | [84] |
| DP7 | VQWRIRVAVIRK | P. aeruginosa | In vitro reduction of biofilm formation of P. aeruginosa from 43% to 68%. | [85] |
| Human β-defensin 2 | HBD2/L-HBD2 | P. aeruginosa | Inhibition of biofilm production by A. baumannii without reducing metabolic activity at lower concentrations. | [86] |
| Tilapia Piscidin 4 (TP4) | FIHHIIGGLFSAGKAI HRLIRRRRR | P. aeruginosa, K. pneumoniae, E. coli, A. baumannii | Cancer cells usually have anionic membranes, and many cationic AMPs such as this one have anticancer properties. | [87] |
| B1CTcu5 | LIAGLAANFLPQILCKIARKC | M. tuberculosis | Mechanism of action based on the induction of morphological changes in the mycobacterial cell wall, such as cavitation and thinning of the cell wall. | [88] |
| CDP-B11 | VRNSQSCRRNKGICV PIRCPGSMRQIGTCL GAQVKCCRRK | A. baumannii, E. coli, P. aeruginosa, K. pneumoniae | Mechanism of action based on inhibition of bacteria by depolarization and damaging in bacterial membranes. | [89] |
| EcDBS1R6 | PMKKLFKLLARIAVKIPVW | A. baumannii, E. coli, P. aeruginosa, K. pneumoniae | Bactericidal mechanism based on induction of membrane permeabilization and loss of bacterial membrane potential. | [82] |
| SET-M33 protease- resistant | (KKIRVRLSA)4K2KβA-OH | S. aureus, A. baumannii, E. coli, P. aeruginosa and K. pneumoniae | Mechanism based on the strong neutralization of lipopolysaccharide (LPS) andlipoteichoic acid from bacteria. Strong anti-inflammatory effect, reducing the expression of cytokines, enzymes, and transcription factors involved in inflammatory processes. | [84] |
| Ctx(Ile21)-Ha | GWLDVAKKIGKAAFNVAKNFI | MDR P. aeruginosa, MDR A. baumannii, S. aureus, E. coli and S. enteritidis | Peptide originating from the frog. Promising antimicrobial activity, with physicochemical stability in different physiological conditions. Its application loaded within alginate microparticles greatly reduced hemolytic activity and even increased its bioavailability to prevent systemic infection. | [90] |
| NZX | GFGCNGPWSEDDIQCHNH CKSIKGYKGGYCARG GFVCKCY (Disulfide bonds at position C4–C30, C15–C37, C19–C39) | M. tuberculosis | Enhances the inhibition of intracellular mycobacteria in primary macrophages and preserves the ability to eliminate M. tuberculosis in vivo when carried into cells by nanoparticle systems such as mesoporous silica. | [91] |
| Antifungal Activity | ||||
| ARP788.14 | KRWIILGLNKIVRMYSPTSI | Candida utilis and in vitro antifungal activity against yeast | Study based on sequence prediction of antifungal peptides using computational algorithms specialized in biological studies and, after prediction, the sequences were tested in disk diffusion and broth microdilution methods, where some promising ones were obtained. | [92] |
| ARP788.13 | PPIPVGEIYKRWIILGLNK | in vitro antifungal activity against yeast | ||
| Ctn[15–34] (the C-terminal fragment of Crotalicidin peptide) | KKRLKKIFKKPMVIGVTIPF | Candida albicans biofilms | Its mechanism of action is based on its interaction with the fungal plasma membrane followed by its disruption, in addition to preventing biofilm formation or eradicating biofilm already present. Other Crotalicidin peptide fragments are also studied for having other properties (antimicrobial, antiparasitic and antiviral) and antiproliferative (antitumor) properties. | [93] |
| ToAP2 | FFGTLFKLGSKLIPGVMKLFSKKKER | Candida albicans in its planktonic form and in a biofilm colony | This peptide increases the permeability of the plasma membrane of C. albicans in its planktonic and biofilm form.In addition, it is effective at different stages of biofilm formation; that is, it acts on both newly formed and mature biofilms. | [94] |
| NDBP-5.7 | ILSAIWSGIKSLF-NH2 | This peptide also increases the permeability of the plasma membrane of C. albicans in its planktonic and biofilm form and, in addition, studies suggest that this molecule causes intracellular morphological changes in the fungus. | ||
| MCh-AMP1 | LSVKAFTGIQLRGVCGIEVKARG | Candida albicans | Peptide derived from the plant Matricaria chamomilla. Its mechanism of action is based on the interaction with the fungal membrane (the peptide is positively charged and contains hydrophobic residues, which causes it to interact with the negative components of the fungal membrane such as phosphatidylserine and phosphatidylinositol), making it more permeable. and causing loss of ions to the pathogenic cell. | [95] |
| KW2 | KWKW-NH2 | C. albican, C. catenulate, C. intermidia, C. rugosa, C. glabrata and C. melibiosica | According to the reference, the antifungal activity of these peptides increases as the peptide is extended, however, the extra amino acid residues of KW5 reduce its selectivity, despite having good antifungal activity, as does KW4. The authors also indicate that there must be a balance of cationicity and hydrophobicity for activity against C. albicans, including multidrug-resistant strains of C. albicans. | [96] |
| KW3 | KWKWKW-NH2 | |||
| KW4 | KWKWKWKW-NH2 | |||
| KW5 | KWKWKWKWKW-NH2 | |||
| Antiviral Activity | ||||
| P9R | NGAICWGPCPTAFRQIGNCGRFRVRCCRIR | Enveloped coronaviruses (SARS-CoV-2, SARS-CoV, and MERS-CoV), influenza virus, and non-enveloped rhinovirus. | The positive charge of this peptide is essential for its antiviral activity, as it targets the inhibition of the virus–host endosomal acidification process (a key step in the life cycle of many pH-dependent viruses). The positive charge inhibits this acidification. | [97] |
| Piscidin-1 | FFHHIFRGIVHVGKTIHRLVTG | PRV (pseudorabies virus), PEDV (porcine epidemic diarrhea virus), PRRSV (porcine reproductive and respiratory syndrome virus), TGEV (transmissible gastroenteritis virus), RV (rotavirus) | It is a polypeptide of natural origin, produced by fish.It has a potent effect on viruses such as catfish virus, frogvirus, and HIV-1. Furthermore, piscidin-1 has also been shown to have inhibitory effects on several common porcine pathogenic viruses. | [98] |
| Caerin 1.1 | GLLSVLGSVAKHVLPHVVPVIAEHL | It is a peptide derived from a granule from the skin glands of an Australian frog. Its activity against bacteria and viruses is based on the destruction of the pathogen’s integrity by forming pores in its membrane. | ||
| pBD-2 (Porcine β-Defensin-2) | DHYICAKKGGTCNFSPCPLFNRIEGTCYSGKAKCCIR | PRV (pseudorabies virus), PRRSV (porcine reproductive and respiratory syndrome virus) | This peptide belongs to the group of defensins, a group of cationic antibacterial peptides divided into α-, β- and θ-.The β-defensins family is mainly expressed in epithelial cells of animal skin, respiratory tract and gastrointestinal tract. Currently, more than 30 β-defensins are known in humans. | |
| Peptide | Sequence and Properties | Antimicrobial Activity | Highlights | Reference |
|---|---|---|---|---|
| Myxinidin2 Myxinidin3 | KIKWILKYWKWS RIRWILRYWRWS | P. aeruginosa, S. aureus, and L. monocytogenes | Effects against a wide range of bacteria, with its mechanism of action based on its ability to insert into bacterial membranes to produce an ion channel or pore that disrupts membrane function. | [106] |
| Colistin (colistin–imipenem and colistin–ciprofloxacin) | ALYKKLLKKLLKSAKKLG | Pseudomonas aeruginosa, Escherichia coli and Klebsiella pneumoniae | Bactericidal mechanism by a detergent-like effect. Recommended as a last choice in the treatment of infections caused by MDR Gram-negative bacteria because it rarely causes bacterial resistance. | [107] |
| S4(1–16)M4Ka | ALWKTLLKKVLKAAAK-NH2 | P. fluorescens | Greater antimicrobial effect and less toxicity than its parent peptide (dermaseptin S4) | [108] |
| Pexiganan | GIGKFLKKAKKFGKAFVKILKK-NH2 | S. aureus, S. epidermidis, S. pyogenes, S. pneumoniae, E. coli and P. aeruginosa | Weak anti-biofilm agent against structures formed on CL. | [109] |
| Citropin 1.1 | GLFDVIKKVASVIGGL-NH2 | Potent anti-biofilm agent against S. aureus strains. | ||
| Temporin A: | FLPLIGRVLSGIL-NH2 | Strong activity against vancomycin-resistant strains. | ||
| Palm-KK-NH2 | Palm-KK-NH2 (Palm–hexadecanoic acid residue) | Effective against most strains in the form of a biofilm. Activity potentiated when combined with standard antibiotics. | ||
| Palm-RR-NH2 | Palm-RR-NH2 (Palm–hexadecanoic acid residue) | Efficiency potentiated when combined with standard antibiotics. | ||
| HB AMP | KKVVFWVKFK + HAp-binding heptapeptide (HBP7) | S. mutans, L. acidophilus and A. viscosus | Adsorption capacity on the dental surface. | [110] |
| KSLW | KKVVFWVKFK | Promising peptide for oral use as it is resistant to the gastrointestinal tract and stable in human saliva. | ||
| TiBP1-GGG-AMP | RPRENRGRERGKGGGLKLLKKLLKLLKKL | S. mutans, S. epidermidis, and E. coli. | Bifunctional peptide capable of binding to titanium materials, enabling its use in biomaterials. Antibacterial functionality. | [100] |
| BA250-C10 | RWRWRWK(C10) | P. aeruginosa | Great activity when used in synergism with two conventional anti-pseudomonas antibiotics to inhibit the planktonic growth of four strains of P. aeruginosa. | [111] |
| D-HB43 | FAKLLAKLAKKLL | Methicillin-resistant S. aureus strains | High cytotoxic and hemolytic effect. | [112] |
| D-Ranalexin | FLGGLIKIVPAMICAVTKKC | Methicillin-resistant S. aureus strains | Effective in dose-dependent biofilm killing, but high cytotoxic and hemolytic effect. | |
| FK13-a1 | WKRIVRRIKRWLR-NH2 | Methicillin-resistant S. aureus, MDR P. aeruginosa and vancomycin-resistant E. faecium | Mechanism of action based on the induction of cytoplasmic membrane potential loss, permeabilization, and rupture. | [113] |
| FK13-a7 | WKRWVRRWKRWLR-NH2 | Methicillin-resistant S. aureus, MDR P. aeruginosa and vancomycin-resistant E. faecium | Mechanism of action based on the induction of cytoplasmic membrane potential loss, permeabilization, and rupture. | |
| KR-12-a5 | KRIVKLILKWLR-NH2 | E. coli, P. aeruginosa, S. typhimurium, S. aureus, B. subtilis, S. epidermidis | This peptide and its analogs kill microbial cells by inducing loss of cytoplasmic membrane potential, permeabilization, and disruption. | [114] |
| AMP2 | KRRWRIWLV | E. coli, P. aeruginosa, S. aureus, E. faecalis, S. epidermidis | 76% reduction of the biofilm area. | [115] |
| GH12 | GLLWHLLHHLLH-NH2 | S. mutans | Antimicrobial activity against cariogenic bacteria and its biofilms in vitro. | [116] |
| TP4 | FIHHIIGGLFSAGKAIHRLIRRRRR | P. aeruginosa, K. pneumoniae, S. aureus | Peptide driven into helix shape by an LPS-like surfactant before binding to the target. | [117] |
| LyeTxI | IWLTALKFLGKNLGKHLALKQQLAKL | F. nucleatum, P. gingivalis, A. actinomycetemcomitans | Active against periodontopathic bacteria. Rapid bactericidal effect, prevention of biofilm development. Can be used in the dental field. | [118] |
| Esc(1–21) | GIFSKLAGKKIKNLLISGLKG-NH2 | P. aeruginosa | Mechanism of action causes membrane thinning. | [119] |
| L12 | LKKLLKKLLKKL-NH2 | P. aeruginosa, K. pneumoniae, S. aureus, E. coli | Mechanism of action based on pore formation, inducing rapid permeabilization of bacterial membranes, inhibition of biofilm formation, disruption of drug-resistant biofilms, and suppression of LPS-induced pro-inflammatory mediators, even at low peptide concentrations. | [120] |
| W12 | WKKWWKKWWKKW-NH2 | Suppression of LPS-induced pro-inflammatory mediators, even at low peptide concentrations. | ||
| WLBU2 | RRWVRRVRRVWRRVVRVVRRWVRR | E. faecium, S. aureus, K. pneumoniae, A. baumannii, P. aeruginosa and Enterobacter species | Mechanism of action based on preventing bacterial adhesion and interfering with gene expression. | [121,122] |
| LL37 | LLGDFFRKSKEKIGKEFKRIVQRIKDFLRNLVPRTES | E. faecium, S. aureus, K. pneumoniae, A. baumannii, P. aeruginosa and Enterobacter species | One of the most important human AMPs that play roles in the defense against local and systemic infections. Bactericidal mechanism against Gram-positive and Gram-negative bacteria based on phospholipid-dependent bacterial membrane disruption. | [121,123] |
| SAAP-148 | LKRVWKRVFKLLKRYWRQLKKPVR | E. faecium, S. aureus, K. pneumoniae, A. baumannii, P. aeruginosa and Enterobacter species | Promising peptide fights difficult-to-treat infections due to its broad antimicrobial activity against MDR, biofilm, and persistent bacteria. | [124] |
| WAM-1 | KRGFGKKLRKRLKKFRNSIKKRLKNFNVVIPIPLPG | A. baumannii | This peptide originates from LL37 AMPs and is more effective in inhibiting biofilm dispersion than its parent peptide. | [125] |
| H4 | KFKKLFKKLSPVIGKEFKRIVERIKRFLR | S. aureus, S. epidermidis, S. pneumoniae, E. coli, E. faecium, K. pneumoniae, and P. aeruginosa | Insignificant rates of toxicity to eukaryotic cells. | [126] |
| RWRWRWA-(Bpa) | RWRWRWA-(4-benzophenylalanine) | P. aeruginosa | It targets the bacterial lipid membrane, but there is no specific receptor. It only affects a range of cellular processes. | [127] |
| Pse-T2 | LNALKKVFQKIHEAIKLI-NH2 | P. aeruginosa, S. aureus, E. coli | Mechanism of action based on the ability to disrupt the outer and inner membrane of Gram-negative bacteria and to bind DNA. | [128] |
| Magainin 2 | GIGKFLHSAKKFGKAFVGEIMNS-NH2 | A. baumannii strains | Strong antibacterial activity against A. baumannii, including MDR strains. Non-toxic to mammalian cells. | [129] |
| Magainin I | GIGKFLHSAGKFGKAFVGEIMKS | E. coli strains | Demands more energy metabolism, translational processes, and bacterial defense in E. coli strains when present. | [130] |
| TC19 | LRCMCIKWWSGKHPK | B. subtilis strains | Promising peptide against Gram-positive bacteria, as its activity on the membrane interferes with several essential cellular processes, leading to bacterial death. | [131] |
| TC84 | LRAMCIKWWSGKHPK | Promising peptide against Gram-positive bacteria, as its activity on the membrane interferes with several essential cellular processes, leading to bacterial death. | ||
| BP2 | GKWKLFKKAFKKFLKILAC | B. subtilis strains | Promising peptide against Gram-positive bacteria, as its activity by perturbation of the membrane interferes with several essential cellular processes, leading to bacterial death. | [132] |
| Nisin A | MSTKDFNLDLVSVSKKDSGASPRITSISLCTPGCKTGALMGCNMKTATCHCSIHVSK | B. subtilis spores | Application as an adjuvant to antibiotic peptides in providing a bactericidal coating for the spores. | [131,133] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Pontes, J.T.C.d.; Toledo Borges, A.B.; Roque-Borda, C.A.; Pavan, F.R. Antimicrobial Peptides as an Alternative for the Eradication of Bacterial Biofilms of Multi-Drug Resistant Bacteria. Pharmaceutics 2022, 14, 642. https://doi.org/10.3390/pharmaceutics14030642
Pontes JTCd, Toledo Borges AB, Roque-Borda CA, Pavan FR. Antimicrobial Peptides as an Alternative for the Eradication of Bacterial Biofilms of Multi-Drug Resistant Bacteria. Pharmaceutics. 2022; 14(3):642. https://doi.org/10.3390/pharmaceutics14030642
Chicago/Turabian StylePontes, Janaína Teixeira Costa de, Anna Beatriz Toledo Borges, Cesar Augusto Roque-Borda, and Fernando Rogério Pavan. 2022. "Antimicrobial Peptides as an Alternative for the Eradication of Bacterial Biofilms of Multi-Drug Resistant Bacteria" Pharmaceutics 14, no. 3: 642. https://doi.org/10.3390/pharmaceutics14030642








