Effectiveness and Nephrotoxicity of Loading Dose Colistin–Meropenem versus Loading Dose Colistin–Imipenem in the Treatment of Carbapenem-Resistant Acinetobacter baumannii Infection
Abstract
:1. Introduction
2. Materials and Methods
2.1. Study Setting and Participants
2.2. Data Collection
2.3. Evaluation of the Outcome
2.4. Antimicrobial Susceptibility Testing
2.5. Statistical Analysis
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Leu, H.S.; Ye, J.J.; Lee, M.H.; Su, L.H.; Huang, P.Y.; Wu, T.L.; Huang, C.T. Synergy of imipenem/colistin methanesulfonate combinations against imipenem-nonsusceptible multidrug-resistant Acinetobacter baumannii. J. Microbiol. Immunol. Infect. 2014, 47, 406–411. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bergen, P.J.; Li, J.; Nation, R.L. Dosing of colistin-back to basic PK/PD. Curr. Opin. Pharmacol. 2011, 11, 464–469. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Yahav, D.; Farbman, L.; Leibovici, L.; Paul, M. Colistin: New lessons on an old antibiotic. Clin. Microbiol. Infect. 2012, 18, 18–29. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Plachouras, D.; Karvanen, M.; Friberg, L.E.; Papadomichelakis, E.; Antoniadou, A.; Tsangaris, I.; Karaiskos, I.; Poulakou, G.; Kontopidou, F.; Armaganidis, A.; et al. Population pharmacokinetic analysis of colistin methanesulfonate and colistin after intravenous administration in critically ill patients with infections caused by Gram-negative bacteria. Antimicrob. Agents Chemother. 2009, 53, 3430–3436. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Imberti, R.; Cusato, M.; Villani, P.; Carnevale, L.; Iotti, G.A.; Langer, M.; Regazzi, M. Steady-state pharmacokinetics and BAL concentration of colistin in critically Ill patients after IV colistin methanesulfonate administration. Chest 2010, 138, 1333–1339. [Google Scholar] [CrossRef]
- Tripodi, M.F.; Durante-Mangoni, E.; Fortunato, R.; Utili, R.; Zarrilli, R. Comparative activities of colistin, rifampicin, imipenem and sulbactam/ampicillin alone or in combination against epidemic multidrug-resistant Acinetobacter baumannii isolates producing OXA-58 carbapenemases. Int. J. Antimicrob. Agents 2007, 30, 537–540. [Google Scholar] [CrossRef]
- Principe, L.; D’Arezzo, S.; Capone, A.; Petrosillo, N.; Visca, P. In vitro activity of tigecycline in combination with various antimicrobials against multidrug resistant Acinetobacter baumannii. Ann. Clin. Microbiol. Antimicrob. 2009, 8, 18. [Google Scholar] [CrossRef] [Green Version]
- Ni, W.; Shao, X.; Di, X.; Cui, J.; Wang, R.; Liu, Y. In vitro synergy of polymyxins with other antibiotics for Acinetobacter baumannii: A systematic review and meta-analysis. Int. J. Antimicrob. Agents 2015, 45, 8–18. [Google Scholar] [CrossRef]
- Li, J.; Rayner, C.R.; Nation, R.L.; Owen, R.J.; Spelman, D.; Tan, K.E.; Liolios, L. Heteroresistance to colistin in multidrug-resistant Acinetobacter baumannii. Antimicrob. Agents Chemother. 2006, 50, 2946–2950. [Google Scholar] [CrossRef] [Green Version]
- Gunderson, B.W.; Ibrahim, K.H.; Hovde, L.B.; Fromm, T.L.; Reed, M.D.; Rotschafer, J.C. Synergistic activity of colistin and ceftazidime against multiantibiotic-resistant Pseudomonas aeruginosa in an in vitro pharmacodynamic model. Antimicrob. Agents Chemother. 2003, 47, 905–909. [Google Scholar] [CrossRef] [Green Version]
- Tan, C.H.; Li, J.; Nation, R.L. Activity of colistin against heteroresistant Acinetobacter baumannii and emergence of resistance in an in vitro pharmacokinetic/pharmacodynamic model. Antimicrob. Agents Chemother. 2007, 51, 3413–3415. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Le Minh, V.; Thi Khanh Nhu, N.; Vinh Phat, V.; Thompson, C.; Huong Lan, N.P.; Thieu Nga, T.V.; Thanh Tam, P.T.; Tuyen, H.T.; Hoang Nhu, T.D.; Van Hao, N.; et al. In vitro activity of colistin in antimicrobial combination against carbapenem-resistant Acinetobacter baumannii isolated from patients with ventilator-associated pneumonia in Vietnam. J. Med. Microbiol. 2015, 64, 1162–1169. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Pongpech, P.; Amornnopparattanakul, S.; Panapakdee, S.; Fungwithaya, S.; Nannha, P.; Dhiraputra, C.; Leelarasamee, A. Antibacterial activity of carbapenem-based combinations againts multidrug-resistant Acinetobacter baumannii. J. Med. Assoc. Thail. 2010, 93, 161–171. [Google Scholar] [PubMed]
- Zhanel, G.G.; Simor, A.E.; Vercaigne, L.; Mandell, L.; Canadian Carbapenem Discussion Group. Imipenem and meropenem: Comparison of in vitro activity, pharmacokinetics, clinical trials and adverse effects. Can. J. Infect. Dis. 1998, 9, 215–228. [Google Scholar] [CrossRef] [PubMed]
- Soudeiha, M.; Dahdouh, E.A.; Azar, E.; Sarkis, D.K.; Daoud, Z. In vitro Evaluation of the Colistin-Carbapenem Combination in Clinical Isolates of A. baumannii Using the Checkerboard, Etest, and Time-Kill Curve Techniques. Front. Cell Infect. Microbiol. 2017, 7, 209. [Google Scholar] [CrossRef] [PubMed]
- Horan, T.C.; Andrus, M.; Dudeck, M.A. CDC/NHSN surveillance definition of health care-associated infection and criteria for specific types of infections in the acute care setting. Am. J. Infect. Control. 2008, 36, 309–332. [Google Scholar] [CrossRef]
- CLSI. Performance Standards for Antimicrobial Susceptibility Testing, Twenty-Fifth Informational Supplement. In CLSI Document M100-S25; Clinical and Laboratory Standards Institute: Wayne, PA, USA, 2015. [Google Scholar]
- Lee, S.Y.; Shin, J.H.; Lee, K.; Joo, M.Y.; Park, K.H.; Shin, M.G.; Suh, S.P.; Ryang, D.W.; Kim, S.H. Comparison of the Vitek 2, MicroScan, and Etest methods with the agar dilution method in assessing colistin susceptibility of bloodstream isolates of Acinetobacter species from a Korean university hospital. J. Clin. Microbiol. 2013, 51, 1924–1926. [Google Scholar] [CrossRef] [Green Version]
- Patrick, A.R.; Schneeweiss, S.; Brookhart, M.A.; Glynn, R.J.; Rothman, K.J.; Avorn, J.; Stürmer, T. The implications of propensity score variable selection strategies in pharmacoepidemiology: An empirical illustration. Pharmacoepidemiol. Drug Saf. 2011, 20, 551–559. [Google Scholar] [CrossRef] [Green Version]
- Cai, Y.; Chai, D.; Wang, R.; Liang, B.; Bai, N. Colistin resistance of Acinetobacter baumannii: Clinical reports, mechanisms and antimicrobial strategies. J. Antimicrob. Chemother. 2012, 67, 1607–1615. [Google Scholar] [CrossRef]
- Isler, B.; Doi, Y.; Bonomo, R.A.; Paterson, D.L. New Treatment Options against Carbapenem-Resistant Acinetobacter baumannii Infections. Antimicrob. Agents Chemother. 2018, 63, e01110–e01118. [Google Scholar] [CrossRef] [Green Version]
- Montero, M.M.; Domene Ochoa, S.; López-Causapé, C.; VanScoy, B.; Luque, S.; Sorlí, L.; Campillo, N.; Padilla, E.; Prim, N.; Segura, C.; et al. Colistin plus meropenem combination is synergistic in vitro against extensively drug-resistant Pseudomonas aeruginosa, including high-risk clones. J. Glob. Antimicrob. Resist. 2019, 18, 37–44. [Google Scholar] [CrossRef] [PubMed]
- Bergen, P.J.; Tsuji, B.T.; Bulitta, J.B.; Forrest, A.; Jacob, J.; Sidjabat, H.E.; Paterson, D.L.; Nation, R.L.; Li, J. Synergistic killing of multidrug-resistant Pseudomonas aeruginosa at multiple inocula by colistin combined with doripenem in an in vitro pharmacokinetic/pharmacodynamic model. Antimicrob. Agents Chemother. 2011, 55, 5685–5695. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Karakonstantis, S.; Ioannou, P.; Samonis, G.; Kofteridis, D.P. Systematic Review of Antimicrobial Combination Options for Pandrug-Resistant Acinetobacter baumannii. Antibiotics 2021, 10, 1344. [Google Scholar] [CrossRef] [PubMed]
- Jaruratanasirikul, S.; Sudsai, T. Comparison of the pharmacodynamics of imipenem in patients with ventilator-associated pneumonia following administration by 2 or 0.5 h infusion. J. Antimicrob. Chemother. 2009, 63, 560–563. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Song, J.Y.; Cheong, H.J.; Lee, J.; Sung, A.K.; Kim, W.J. Efficacy of monotherapy and combined antibiotic therapy for carbapenem-resistant Acinetobacter baumannii pneumonia in an immunosuppressed mouse model. Int. J. Antimicrob. Agents 2009, 31, 33–39. [Google Scholar] [CrossRef]
- Fan, B.; Guan, J.; Wang, X.; Cong, Y. Activity of Colistin in Combination with Meropenem, Tigecycline, Fosfomycin, Fusidic Acid, Rifampin or Sulbactam against Extensively Drug-Resistant Acinetobacter baumannii in a Murine Thigh-Infection Model. PLoS ONE 2016, 11, e0157757. [Google Scholar] [CrossRef] [Green Version]
- Daoud, Z.; Mansour, N.; Masri, K. Synergistic combination of carbapenems and colistin against P. aeruginosa and A. baumannii. Open J. Med. Microbiol. 2013, 3, 253–258. [Google Scholar] [CrossRef] [Green Version]
- Zusman, O.; Avni, T.; Leibovici, L.; Adler, A.; Friberg, L.; Stergiopoulou, T.; Carmeli, Y.; Paul, M. Systematic review and meta-analysis of in vitro synergy of polymyxins and carbapenems. Antimicrob. Agents Chemother. 2013, 57, 5104–5111. [Google Scholar] [CrossRef] [Green Version]
- Scudeller, L.; Righi, E.; Chiamenti, M.; Bragantini, D.; Menchinelli, G.; Cattaneo, P.; Giske, C.G.; Lodise, T.; Sanguinetti, M.; Piddock, L.; et al. Systematic review and meta-analysis of in vitro efficacy of antibiotic combination therapy against carbapenem-resistant Gram-negative bacilli. Int. J. Antimicrob. Agents 2021, 57, 106344. [Google Scholar] [CrossRef]
- Santimaleeworagun, W.; Wongpoowarak, P.; Chayakul, P.; Pattharachayakul, S.; Tansakul, P.; Garey, K.W. In vitro activity of colistin or sulbactam in combination with fosfomycin or imipenem against clinical isolates of carbapenem-resistant Acinetobacter baumannii producing OXA-23 carbapenemases. Southeast Asian J. Trop. Med. Public Health 2011, 42, 890–900. [Google Scholar]
| Characteristic | Colistin–Meropenem (n = 311) | Colistin–Imipenem (n = 68) | p-Value |
|---|---|---|---|
| Sex, n (%) | |||
| Male | 129 (41.48) | 25 (36.76) | 0.499 |
| Female | 182 (58.52) | 43 (63.24) | |
| Age, years, mean ± SD | 65.98 ± 17.52 | 63.14 ± 15.60 | 0.218 |
| Duration of treatment, days, mean ± SD | 9.20 ± 6.17 | 10.04 ± 5.73 | 0.307 |
| Comorbidities *, n (%) | |||
| Hypertension | 149 (47.91) | 24 (35.29) | 0.061 |
| Cardiovascular disease | 113 (36.45) | 13 (19.12) | 0.007 |
| Diabetes mellitus | 76 (24.44) | 9 (13.24) | 0.053 |
| Chronic kidney disease | 72 (23.23) | 13 (19.12) | 0.524 |
| Chronic obstructive pulmonary disease | 57 (18.33) | 6 (8.82) | 0.071 |
| Malignancy | 72 (23.15) | 25 (36.76) | 0.031 |
| Chronic liver disease | 21 (6.77) | 1 (1.47) | 0.147 |
| Septic shock | 229 (73.63) | 41 (60.29) | 0.038 |
| Mechanical ventilation | 216 (83.92) | 56 (82.35) | 0.720 |
| Charlson score, median, mean ± SD | 2.69 ± 2.26 | 2.29 ± 1.72 | 0.167 |
| APACHE II score a, mean ± SD | 7.94 ± 4.29 | 7.91 ± 3.83 | 0.948 |
| ICU admission | 227 (72.99) | 44 (64.71) | 0.183 |
| Baseline SCr, mg/dL, median (IQR) | 0.90 (0.60–1.50) | 0.80 (0.50–1.50) | 0.175 |
| Baseline GFR, mL/min, median (IQR) | 54.59 (18.57–95.74) | 69.27 (33.45–103.55) | 0.101 |
| Total colistin dose, g, median (IQR) | 1.60 (0.90–2.85) | 1.85 (1.05–3.12) | 0.175 |
| Type of nephrotoxic medications #, n (%) | |||
| Aminoglycosides | 2 (0.64) | 3 (4.41) | 0.042 |
| Diuretics | 245 (78.78) | 57 (83.82) | 0.408 |
| Amphotericin B | 28 (9.00) | 11 (16.18) | 0.120 |
| Vasopressors | 226 (72.67) | 43 (63.24) | 0.140 |
| Vancomycin | 199 (63.99) | 37 (54.41) | 0.167 |
| Site of CRAB infection | |||
| Pneumonia | 260 (83.60) | 53 (77.94) | 0.290 |
| Bacteraemia | 15 (4.82) | 4 (5.88) | 0.758 |
| UTI | 34 (10.93) | 9 (13.24) | 0.535 |
| Other * | 17 (5.47) | 5 (7.35) | 0.567 |
| MIC colistin µg/mL, median (min–max) | 0.50 (0.50–1.00) | 0.50 (0.50–1.00) | 1.000 |
| Outcomes | Colistin–Meropenem (n = 311) | Colistin–Imipenem (n = 68) | p-Value |
|---|---|---|---|
| Primary outcome | |||
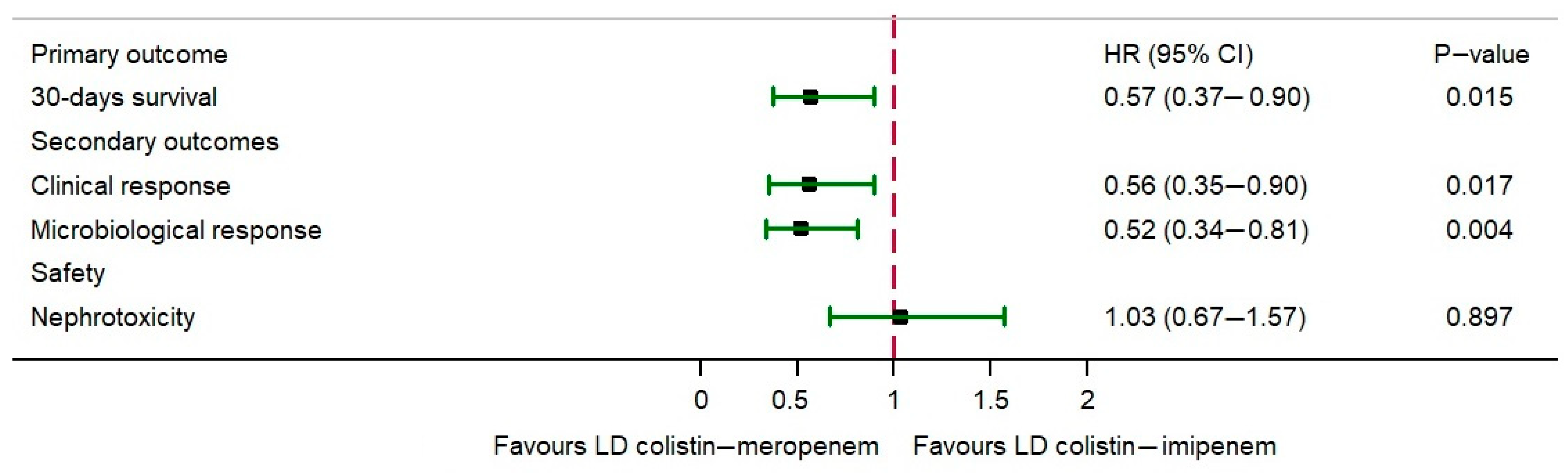
| 30-day survival rate | 167 (53.70) | 33 (48.53) | 0.503 |
| Secondary outcomes | |||
| Clinical response | 170 (54.66) | 30 (44.12) | 0.140 |
| Microbiological response | 194 (62.38) | 37 (54.41) | 0.272 |
| Safety | |||
| Nephrotoxicity | 138 (44.37) | 41 (60.29) | 0.022 |
| Outcome and Variable * | Colistin–Meropenem (n = 311) | Colistin–Imipenem (n = 68) | Crude HR (95% CI) | p-Value | Adjusted HR ** (95% CI) | p-Value |
|---|---|---|---|---|---|---|
| Safety | ||||||
| Nephrotoxicity | 138 (44.37) | 41 (60.29) | 1.18 (0.84–1.68) | 0.343 | 1.03 (0.67–1.57) | 0.897 |
| - Previous CKD | 19 (26.39) | 3 (26.39) | 0.87 (0.26–2.94) | 0.821 | 0.02 (0.01–3.42) | 0.268 |
| - Non-CKD | 119 (49.79) | 38 (69.09) | 1.19 (0.83–1.72) | 0.347 | 1.38 (0.88–2.15) | 0.159 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Katip, W.; Oberdorfer, P.; Kasatpibal, N. Effectiveness and Nephrotoxicity of Loading Dose Colistin–Meropenem versus Loading Dose Colistin–Imipenem in the Treatment of Carbapenem-Resistant Acinetobacter baumannii Infection. Pharmaceutics 2022, 14, 1266. https://doi.org/10.3390/pharmaceutics14061266
Katip W, Oberdorfer P, Kasatpibal N. Effectiveness and Nephrotoxicity of Loading Dose Colistin–Meropenem versus Loading Dose Colistin–Imipenem in the Treatment of Carbapenem-Resistant Acinetobacter baumannii Infection. Pharmaceutics. 2022; 14(6):1266. https://doi.org/10.3390/pharmaceutics14061266
Chicago/Turabian StyleKatip, Wasan, Peninnah Oberdorfer, and Nongyao Kasatpibal. 2022. "Effectiveness and Nephrotoxicity of Loading Dose Colistin–Meropenem versus Loading Dose Colistin–Imipenem in the Treatment of Carbapenem-Resistant Acinetobacter baumannii Infection" Pharmaceutics 14, no. 6: 1266. https://doi.org/10.3390/pharmaceutics14061266
APA StyleKatip, W., Oberdorfer, P., & Kasatpibal, N. (2022). Effectiveness and Nephrotoxicity of Loading Dose Colistin–Meropenem versus Loading Dose Colistin–Imipenem in the Treatment of Carbapenem-Resistant Acinetobacter baumannii Infection. Pharmaceutics, 14(6), 1266. https://doi.org/10.3390/pharmaceutics14061266






