Research Progress of Conjugated Nanomedicine for Cancer Treatment
Abstract
:1. Introduction
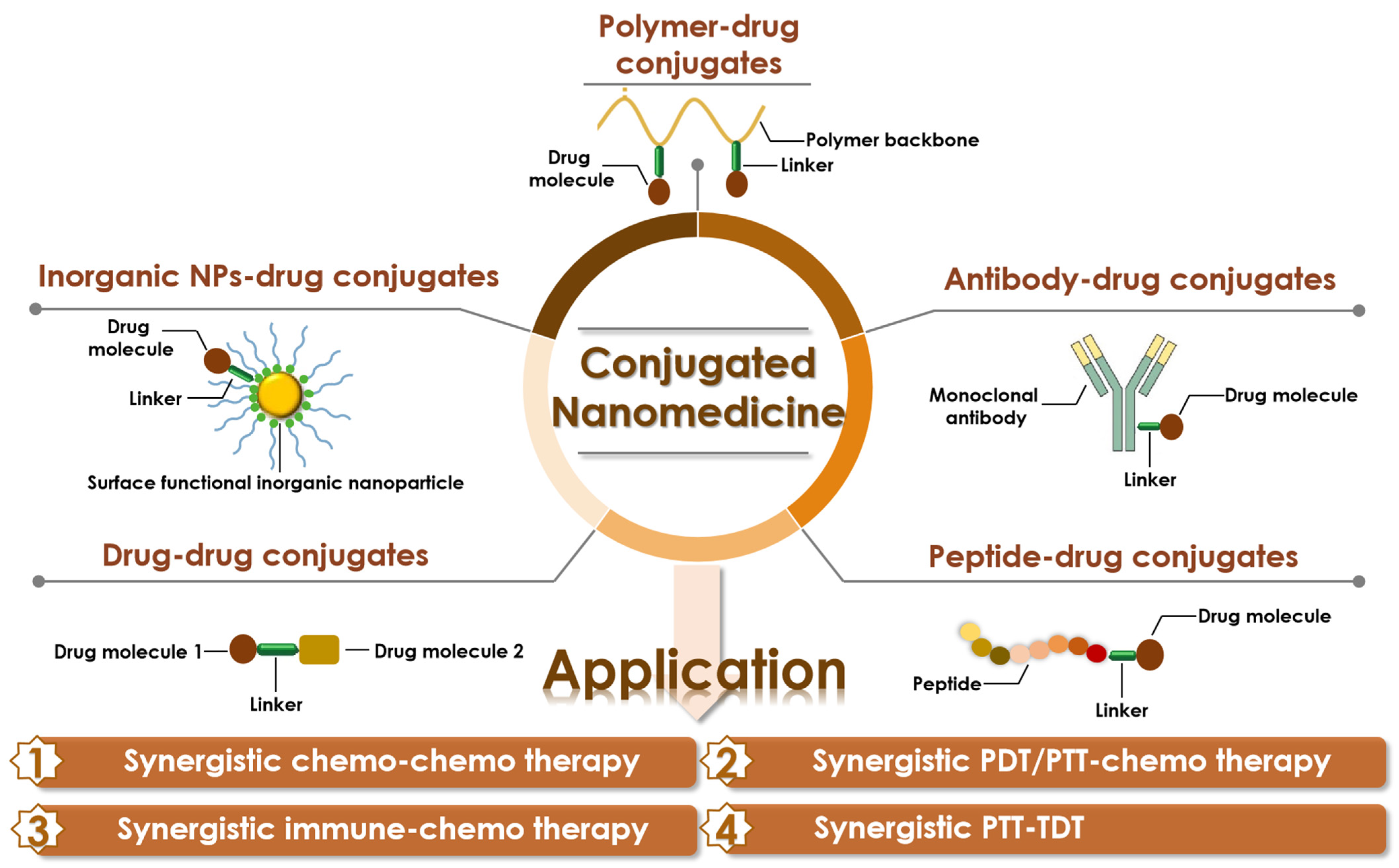
2. Categories of Drug Conjugates
2.1. Polymer-Drug Conjugates
2.2. Antibody-Drug Conjugates
2.3. Peptide-Drug Conjugates
2.4. Drug-Drug Conjugates
2.4.1. Conjugates of Multiple Chemotherapeutic Drugs
2.4.2. Photothermal Agent-Drug Conjugates
2.4.3. Photosensitizer-Drug Conjugates
2.5. Inorganic Nanoparticle-Drug Conjugates
3. Representative Applications of Conjugated Nanomedicine
3.1. Synergistic Chemo-Chemo Therapy
3.2. Synergistic PDT/PTT-Chemo Therapy
3.3. Synergistic Immune-Chemo Therapy
3.4. Synergistic PTT-TDT
4. Challenges and Future Perspectives of Bioconjugation and Nanomedicine
Funding
Institutional Review Board Statement
Informed Consent Statement
Conflicts of Interest
References
- Sung, H.; Ferlay, J.; Siegel, R.L.; Laversanne, M.; Soerjomataram, I.; Jemal, A.; Bray, F. Global cancer statistics 2020: Globocan estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J. Clin. 2021, 71, 209–249. [Google Scholar] [CrossRef] [PubMed]
- Chen, Q.; Ke, H.; Dai, Z.; Liu, Z. Nanoscale theranostics for physical stimulus-responsive cancer therapies. Biomaterials 2015, 73, 214–230. [Google Scholar] [CrossRef] [PubMed]
- Wong, C.; Siah, K.W.; Lo, A.W. Estimation of clinical trial success rates and related parameters. Biostatistics 2019, 20, 273–286. [Google Scholar] [CrossRef] [PubMed]
- Dong, P.; Rakesh, K.P.; Manukumar, H.M.; Mohammed, Y.H.E.; Karthik, C.S.; Sumathi, S.; Mallu, P.; Qin, H.L. Innovative nano-carriers in anticancer drug delivery-a comprehensive review. Bioorg. Chem. 2019, 85, 325–336. [Google Scholar] [CrossRef]
- Shen, J.; Wolfram, J.; Ferrari, M.; Shen, H. Taking the vehicle out of drug delivery. Mater. Today 2017, 20, 95–97. [Google Scholar] [CrossRef] [Green Version]
- Rajitha, B.; Malla, R.R.; Vadde, R.; Kasa, P.; Prasad, G.L.V.; Farran, B.; Kumari, S.; Pavitra, E.; Kamal, M.A.; Raju, G.S.R.; et al. Horizons of nanotechnology applications in female specific cancers. Semin. Cancer Biol. 2021, 69, 376–390. [Google Scholar] [CrossRef]
- Raju, G.S.R.; Dariya, B.; Mungamuri, S.K.; Chalikonda, G.; Kang, S.M.; Khan, I.N.; Sushma, P.S.; Nagaraju, G.P.; Pavitra, E.; Han, Y.K. Nanomaterials multifunctional behavior for enlightened cancer therapeutics. Semin. Cancer Biol. 2021, 69, 178–189. [Google Scholar] [CrossRef]
- de Lázaro, I.; Mooney, D.J. Obstacles and opportunities in a forward vision for cancer nanomedicine. Nat. Mater. 2021, 20, 1469–1479. [Google Scholar] [CrossRef]
- Lammers, T.; Kiessling, F.; Ashford, M.; Hennink, W.; Crommelin, D.; Storm, G. Cancer nanomedicine: Is targeting our target? Nat. Rev. Mater. 2016, 1, 16069. [Google Scholar] [CrossRef] [Green Version]
- Bobo, D.; Robinson, K.J.; Islam, J.; Thurecht, K.J.; Corrie, S.R. Nanoparticle-based medicines: A review of fda-approved materials and clinical trials to date. Pharm. Res. 2016, 33, 2373–2387. [Google Scholar] [CrossRef]
- Pearce, A.K.; O’Reilly, R.K. Insights into active targeting of nanoparticles in drug delivery: Advances in clinical studies and design considerations for cancer nanomedicine. Bioconj. Chem. 2019, 30, 2300–2311. [Google Scholar] [CrossRef] [PubMed]
- Kim, M.T.; Chen, Y.; Marhoul, J.; Jacobson, F. Statistical modeling of the drug load distribution on trastuzumab emtansine (kadcyla), a lysine-linked antibody drug conjugate. Bioconj. Chem. 2014, 25, 1223–1232. [Google Scholar] [CrossRef]
- Barenholz, Y. Doxil®—The first FDA-approved nano-drug: Lessons learned. J. Control. Release 2012, 160, 117–134. [Google Scholar] [CrossRef] [PubMed]
- Cabral, H.; Matsumoto, Y.; Mizuno, K.; Chen, Q.; Murakami, M.; Kimura, M.; Terada, Y.; Kano, M.; Miyazono, K.; Uesaka, M. Accumulation of sub-100 nm polymeric micelles in poorly permeable tumours depends on size. Nat. Nanotechnol. 2011, 6, 815–823. [Google Scholar] [CrossRef] [PubMed]
- Ventola, C.L. Progress in nanomedicine: Approved and investigational nanodrugs. Pharm. Ther. 2017, 42, 742. [Google Scholar]
- Alven, S.; Nqoro, X.; Buyana, B.; Aderibigbe, B.A. Polymer-drug conjugate, a potential therapeutic to combat breast and lung cancer. Pharmaceutics 2020, 12, 406. [Google Scholar] [CrossRef]
- Jatzkewitz, H.P. Bound to blood plasma expander (polyvinylpyrrolidone) as a new depot form of a biologically active primary amine (mescaline) Z. Naturforsch 1955, 10, 27–31. [Google Scholar] [CrossRef]
- Murguia-Favela, L.; Min, W.; Loves, R.; Leon-Ponte, M.; Grunebaum, E. Comparison of elapegademase and pegademase in ada-deficient patients and mice. Clin. Exp. Immunol. 2020, 200, 176–184. [Google Scholar] [CrossRef]
- Alconcel, S.N.; Baas, A.S.; Maynard, H.D. Fda-approved poly (ethylene glycol)–protein conjugate drugs. Polym. Chem. 2011, 2, 1442–1448. [Google Scholar] [CrossRef]
- Amgoth, C.; Dharmapuri, G. Synthesis and characterization of polymeric nanoparticles and capsules as payload for anticancer drugs and nanomedicines. Mater. Today Proc. 2016, 3, 3833–3837. [Google Scholar] [CrossRef]
- De Koker, S.; Hoogenboom, R.; De Geest, B.G. Polymeric multilayer capsules for drug delivery. Chem. Soc. Rev. 2012, 41, 2867–2884. [Google Scholar] [CrossRef] [PubMed]
- Calzoni, E.; Cesaretti, A.; Polchi, A.; Di Michele, A.; Tancini, B.; Emiliani, C. Biocompatible polymer nanoparticles for drug delivery applications in cancer and neurodegenerative disorder therapies. J. Funct. Biomater. 2019, 10, 4. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Mamidi, N.; Delgadillo, R.M.V. Design, fabrication and drug release potential of dual stimuli-responsive composite hydrogel nanoparticle interfaces. Colloids Surf. B Biointerfaces 2021, 204, 111819. [Google Scholar] [CrossRef] [PubMed]
- Mohan, A.; Girdhar, M.; Kumar, R.; Chaturvedi, H.S.; Vadhel, A.; Solanki, P.R.; Mamidi, N. Polyhydroxybutyrate-based nanocomposites for bone tissue engineering. Pharmaceuticals 2021, 14, 1163. [Google Scholar] [CrossRef]
- Palmerston Mendes, L.; Pan, J.; Torchilin, V. Dendrimers as nanocarriers for nucleic acid and drug delivery in cancer therapy. Molecules 2017, 22, 1401. [Google Scholar] [CrossRef] [Green Version]
- Kesharwani, P.; Choudhury, H.; Meher, J.G.; Pandey, M.; Gorain, B. Dendrimer-entrapped gold nanoparticles as promising nanocarriers for anticancer therapeutics and imaging. Prog. Mater. Sci. 2019, 103, 484–508. [Google Scholar] [CrossRef]
- Duncan, R. The dawning era of polymer therapeutics. Nat. Rev. Drug Discov. 2003, 2, 347–360. [Google Scholar] [CrossRef]
- Parveen, S.; Arjmand, F.; Tabassum, S. Clinical developments of antitumor polymer therapeutics. RSC Adv. 2019, 9, 24699–24721. [Google Scholar] [CrossRef] [Green Version]
- Jameson, G.S.; Hamm, J.T.; Weiss, G.J.; Alemany, C.; Anthony, S.; Basche, M.; Ramanathan, R.K.; Borad, M.J.; Tibes, R.; Cohn, A. A multicenter, phase i.; dose-escalation study to assess the safety, tolerability, and pharmacokinetics of etirinotecan pegol in patients with refractory solid tumorsetirinotecan pegol phase i in patients with solid tumors. Clin. Cancer Res. 2013, 19, 268–278. [Google Scholar] [CrossRef] [Green Version]
- Thakor, P.; Bhavana, V.; Sharma, R.; Srivastava, S.; Singh, S.B.; Mehra, N.K. Polymer–drug conjugates: Recent advances and future perspectives. Drug Discov. Today 2020, 25, 1718–1726. [Google Scholar] [CrossRef]
- Ekladious, I.; Colson, Y.L.; Grinstaff, M.W. Polymer–drug conjugate therapeutics: Advances, insights and prospects. Nat. Rev. Drug Discov. 2019, 18, 273–294. [Google Scholar] [CrossRef] [PubMed]
- Greenwald, R.B.; Choe, Y.H.; McGuire, J.; Conover, C.D. Effective drug delivery by pegylated drug conjugates. Adv. Drug Deliv. Rev. 2003, 55, 217–250. [Google Scholar] [CrossRef]
- Ing, M.; Gupta, N.; Teyssandier, M.; Maillère, B.; Pallardy, M.; Delignat, S.; Lacroix-Desmazes, S. Immunogenicity of long-lasting recombinant factor viii products. Cell. Immunol. 2016, 301, 40–48. [Google Scholar] [CrossRef] [Green Version]
- Makadia, H.K.; Siegel, S.J. Poly lactic-co-glycolic acid (plga) as biodegradable controlled drug delivery carrier. Polymers 2011, 3, 1377–1397. [Google Scholar] [CrossRef] [PubMed]
- Vlachopoulos, A.; Karlioti, G.; Balla, E.; Daniilidis, V.; Kalamas, T.; Stefanidou, M.; Bikiaris, N.D.; Christodoulou, E.; Koumentakou, I.; Karavas, E. Poly (lactic acid)-based microparticles for drug delivery applications: An overview of recent advances. Pharmaceutics 2022, 14, 359. [Google Scholar] [CrossRef] [PubMed]
- Espinoza, S.M.; Patil, H.I.; San Martin Martinez, E.; Casañas Pimentel, R.; Ige, P.P. Poly-ε-caprolactone (pcl), a promising polymer for pharmaceutical and biomedical applications: Focus on nanomedicine in cancer. Int. J. Polym. Mater. Polym. Biomater. 2020, 69, 85–126. [Google Scholar] [CrossRef]
- Tinajero-Díaz, E.; de Ilarduya, A.M.; Cavanagh, B.; Heise, A.; Muñoz-Guerra, S. Poly (amino acid)-grafted polymacrolactones. Synthesis, self-assembling and ionic coupling properties. React. Funct. Polym. 2019, 143, 104316. [Google Scholar] [CrossRef]
- Sun, H.; Chang, M.Y.Z.; Cheng, W.-I.; Wang, Q.; Commisso, A.; Capeling, M.; Wu, Y.; Cheng, C. Biodegradable zwitterionic sulfobetaine polymer and its conjugate with paclitaxel for sustained drug delivery. Acta Biomater. 2017, 64, 290–300. [Google Scholar] [CrossRef] [PubMed]
- Feng, X.; Xu, W.; Liu, J.; Li, D.; Li, G.; Ding, J.; Chen, X. Polypeptide nanoformulation-induced immunogenic cell death and remission of immunosuppression for enhanced chemoimmunotherapy. Sci. Bull. 2021, 66, 362–373. [Google Scholar] [CrossRef]
- Ma, S.; Song, W.; Xu, Y.; Si, X.; Zhang, D.; Lv, S.; Yang, C.; Ma, L.; Tang, Z.; Chen, X. Neutralizing tumor-promoting inflammation with polypeptide-dexamethasone conjugate for microenvironment modulation and colorectal cancer therapy. Biomaterials 2020, 232, 119676. [Google Scholar] [CrossRef]
- do Pazo, C.; Nawaz, K.; Webster, R.M. The oncology market for antibody-drug conjugates. Nat. Rev. Drug Discov. 2021, 20, 583–584. [Google Scholar] [CrossRef] [PubMed]
- Woodruff, M.A.; Hutmacher, D.W. The return of a forgotten polymer—polycaprolactone in the 21st century. Prog. Polym. Sci. 2010, 35, 1217–1256. [Google Scholar] [CrossRef] [Green Version]
- Danhier, F.; Ansorena, E.; Silva, J.M.; Coco, R.; Le Breton, A.; Préat, V. Plga-based nanoparticles: An overview of biomedical applications. J. Control. Release 2012, 161, 505–522. [Google Scholar] [CrossRef] [PubMed]
- Nampoothiri, K.M.; Nair, N.R.; John, R.P. An overview of the recent developments in polylactide (pla) research. Bioresour. Technol. 2010, 101, 8493–8501. [Google Scholar] [CrossRef]
- Mamidi, N.; Velasco Delgadillo, R.M.; Barrera, E.V. Covalently functionalized carbon nano-onions integrated gelatin methacryloyl nanocomposite hydrogel containing γ-cyclodextrin as drug carrier for high-performance pH-triggered drug release. Pharmaceuticals 2021, 14, 291. [Google Scholar] [CrossRef]
- Mamidi, N.; Zuníga, A.E.; Villela-Castrejón, J. Engineering and evaluation of forcespun functionalized carbon nano-onions reinforced poly (ε-caprolactone) composite nanofibers for pH-responsive drug release. Mater. Sci. Eng. C 2020, 112, 110928. [Google Scholar] [CrossRef]
- Kumar, R.; Singh, M.; Meena, J.; Singhvi, P.; Thiyagarajan, D.; Saneja, A.; Panda, A.K. Hyaluronic acid-dihydroartemisinin conjugate: Synthesis, characterization and in vitro evaluation in lung cancer cells. Int. J. Biol. Macromol. 2019, 133, 495–502. [Google Scholar] [CrossRef]
- Leung, D.; Wurst, J.M.; Liu, T.; Martinez, R.M.; Datta-Mannan, A.; Feng, Y. Antibody conjugates-recent advances and future innovations. Antibodies 2020, 9, 2. [Google Scholar] [CrossRef] [Green Version]
- Tsuchikama, K.; An, Z. Antibody-drug conjugates: Recent advances in conjugation and linker chemistries. Protein Cell 2018, 9, 33–46. [Google Scholar] [CrossRef] [Green Version]
- Nasiri, H.; Valedkarimi, Z.; Aghebati-Maleki, L.; Majidi, J. Antibody-drug conjugates: Promising and efficient tools for targeted cancer therapy. J. Cell. Physiol. 2018, 233, 6441–6457. [Google Scholar] [CrossRef]
- Petersdorf, S.H.; Kopecky, K.J.; Slovak, M.; Willman, C.; Nevill, T.; Brandwein, J.; Larson, R.A.; Erba, H.P.; Stiff, P.J.; Stuart, R.K. A phase 3 study of gemtuzumab ozogamicin during induction and postconsolidation therapy in younger patients with acute myeloid leukemia. Blood J. Am. Soc. Hematol. 2013, 121, 4854–4860. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Elias, D.J.; Kline, L.E.; Robbins, B.A.; Johnson Jr, H.; Pekny, K.; Benz, M.; Robb, J.A.; Walker, L.E.; Kosty, M.; Dillman, R.O. Monoclonal antibody ks1/4-methotrexate immunoconjugate studies in non-small cell lung carcinoma. Am. J. Respir. Crit. Care Med. 1994, 150, 1114–1122. [Google Scholar] [CrossRef]
- Erickson, H.K.; Park, P.U.; Widdison, W.C.; Kovtun, Y.V.; Garrett, L.M.; Hoffman, K.; Lutz, R.J.; Goldmacher, V.S.; Blaättler, W.A. Antibody-maytansinoid conjugates are activated in targeted cancer cells by lysosomal degradation and linker-dependent intracellular processing. Cancer Res. 2006, 66, 4426–4433. [Google Scholar] [CrossRef] [Green Version]
- Ehrlich, P.; Himmelweit, F.; Dale, H.; Marquardt, M. The Collected Papers of Paul Ehrlich; Franklin Book Co., Inc.: New York, NY, USA, 1957. [Google Scholar]
- Petersen, B.H.; DeHerdt, S.V.; Schneck, D.W.; Bumol, T.F. The human immune response to ks1/4-desacetylvinblastine (ly256787) and ks1/4-desacetylvinblastine hydrazide (ly203728) in single and multiple dose clinical studies. Cancer Res. 1991, 51, 2286–2290. [Google Scholar]
- Kim, E.G.; Kim, K.M. Strategies and advancement in antibody-drug conjugate optimization for targeted cancer therapeutics. Biomol. Ther. 2015, 23, 493. [Google Scholar] [CrossRef] [Green Version]
- LoRusso, P.M.; Weiss, D.; Guardino, E.; Girish, S.; Sliwkowski, M.X. Trastuzumab emtansine: A unique antibody-drug conjugate in development for human epidermal growth factor receptor 2-positive cancer. Clin. Cancer Res. 2011, 17, 6437–6447. [Google Scholar] [CrossRef] [Green Version]
- Verma, S.; Miles, D.; Gianni, L.; Krop, I.E.; Welslau, M.; Baselga, J.; Pegram, M.; Oh, D.-Y.; Diéras, V.; Guardino, E. Trastuzumab emtansine for her 2-positive advanced breast cancer. N. Engl. J. Med. 2012, 367, 1783–1791. [Google Scholar] [CrossRef] [Green Version]
- Beck, A.; Goetsch, L.; Dumontet, C.; Corvaïa, N. Strategies and challenges for the next generation of antibody–drug conjugates. Nat. Rev. Drug Discov. 2017, 16, 315–337. [Google Scholar] [CrossRef]
- Dere, R.; Yi, J.-H.; Lei, C.; Saad, O.M.; Huang, C.; Li, Y.; Baudys, J.; Kaur, S. Pk assays for antibody–drug conjugates: Case study with ado-trastuzumab emtansine. Bioanalysis 2013, 5, 1025–1040. [Google Scholar] [CrossRef]
- Agarwal, P.; Bertozzi, C.R. Site-specific antibody–drug conjugates: The nexus of bioorthogonal chemistry, protein engineering, and drug development. Bioconj. Chem. 2015, 26, 176–192. [Google Scholar] [CrossRef] [Green Version]
- Kung Sutherland, M.S.; Walter, R.B.; Jeffrey, S.; Burke, P.; Yu, C.; Kostner, H.; Stone, I.; Ryan, M.C.; Sussman, D.; Lyon, R.P. Sgn-cd33a: A novel cd33-targeting antibody–drug conjugate using a pyrrolobenzodiazepine dimer is active in models of drug-resistant aml. Blood J. Am. Soc. Hematol. 2013, 122, 1455–1463. [Google Scholar] [CrossRef] [PubMed]
- Bargh, J.D.; Isidro-Llobet, A.; Parker, J.S.; Spring, D.R. Cleavable linkers in antibody–drug conjugates. Chem. Soc. Rev. 2019, 48, 4361–4374. [Google Scholar] [CrossRef] [PubMed]
- Rosario, G. Decoupling stability and release in disulfide bonds with antibody-small molecule conjugates. Chem. Sci. 2017, 8, 366–370. [Google Scholar]
- Gébleux, R.; Wulhfard, S.; Casi, G.; Neri, D. Antibody format and drug release rate determine the therapeutic activity of noninternalizing antibody–drug conjugatesnoninternalizing antibody–drug conjugates. Mol. Cancer Therapeut. 2015, 14, 2606–2612. [Google Scholar] [CrossRef] [Green Version]
- Giansanti, F.; Flavell, D.J.; Angelucci, F.; Fabbrini, M.S.; Ippoliti, R. Strategies to improve the clinical utility of saporin-based targeted toxins. Toxins 2018, 10, 82. [Google Scholar] [CrossRef] [Green Version]
- Pastan, I.; Hassan, R.; FitzGerald, D.J.; Kreitman, R.J. Immunotoxin therapy of cancer. Nat. Rev. Cancer 2006, 6, 559–565. [Google Scholar] [CrossRef]
- Khan, M.; Hossain, M.I.; Hossain, M.K.; Rubel, M.H.K.; Hossain, K.M.; Mahfuz, A.M.U.B.; Anik, M.I. Recent progress in nanostructured smart drug delivery systems for cancer therapy: A review. ACS Appl. Bio. Mater. 2022, 5, 971–1012. [Google Scholar] [CrossRef]
- Lyon, R.; Bovee, T.D.; Doronina, S.O.; Burke, P.J.; Hunter, J.H.; Neff-LaFord, H.D.; Jonas, M.; Anderson, M.E.; Setter, J.R.; Senter, P.D. Reducing hydrophobicity of homogeneous antibody-drug conjugates improves pharmacokinetics and therapeutic index. Nat. Biotechnol. 2015, 33, 733–735. [Google Scholar] [CrossRef]
- Rudin, C.M.; Pietanza, M.C.; Bauer, T.M.; Ready, N.; Morgensztern, D.; Glisson, B.S.; Byers, L.A.; Johnson, M.L.; Burris, H.A., III; Robert, F. Rovalpituzumab tesirine, a dll3-targeted antibody-drug conjugate, in recurrent small-cell lung cancer: A first-in-human, first-in-class, open-label, phase 1 study. Lancet Oncol. 2017, 18, 42–51. [Google Scholar] [CrossRef] [Green Version]
- Hamblett, K.J.; Hammond, P.W.; Barnscher, S.D.; Fung, V.K.; Davies, R.H.; Wickman, G.R.; Hernandez, A.; Ding, T.; Galey, A.S.; Winters, G.C. Zw49, a her2-targeted biparatopic antibody-drug conjugate for the treatment of her2-expressing cancers. Cancer Res. 2018, 78, 3914. [Google Scholar] [CrossRef]
- Koopman, L.A.; Janmaat, M.L.; Jacobsen, K.; Terp, M.G.; Heuvel, E.G.V.D.; Forssman, U.; Lingnau, A.; Parren, P.W.; Ditzel, H.; Breij, E.C. An axl-specific antibody-drug conjugate shows preclinical anti-tumor activity in non-small cell lung cancer, including egfr-inhibitor resistant nsclc. Cancer Res. 2018, 78, 832. [Google Scholar] [CrossRef]
- Fu, Z.; Li, S.; Han, S.; Shi, C.; Zhang, Y. Antibody drug conjugate: The “biological missile” for targeted cancer therapy. Signal Transduct. Target. Ther. 2022, 7, 93. [Google Scholar] [CrossRef] [PubMed]
- Drago, J.Z.; Modi, S.; Chandarlapaty, S. Unlocking the potential of antibody–drug conjugates for cancer therapy. Nat. Rev. Clin. Oncol. 2021, 18, 327–344. [Google Scholar] [CrossRef] [PubMed]
- Huang, P.; Wang, D.; Su, Y.; Huang, W.; Zhou, Y.; Cui, D.; Zhu, X.; Yan, D. Combination of small molecule prodrug and nanodrug delivery: Amphiphilic drug–drug conjugate for cancer therapy. J. Am. Chem. Soc. 2014, 136, 11748–11756. [Google Scholar] [CrossRef]
- Jaiswal, S.K.; Sarathi, V.; Memon, S.S.; Garg, R.; Malhotra, G.; Verma, P.; Shah, R.; Sehemby, M.K.; Patil, V.A.; Jadhav, S. 177lu-dotatate therapy in metastatic/inoperable pheochromocytoma-paraganglioma. Endocr. Connect. 2020, 9, 864–873. [Google Scholar] [CrossRef]
- Zhang, Y.; He, P.; Zhang, P.; Yi, X.; Xiao, C.; Chen, X. Polypeptides–drug conjugates for anticancer therapy. Adv. Healthc. Mater. 2021, 10, 2001974. [Google Scholar] [CrossRef]
- Cooper, B.M.; Iegre, J.; O’Donovan, D.H.; Halvarsson, M.Ö.; Spring, D.R. Peptides as a platform for targeted therapeutics for cancer: Peptide–drug conjugates (pdcs). Chem. Soc. Rev. 2021, 50, 1480–1494. [Google Scholar] [CrossRef]
- Carvalho, M.R.; Carvalho, C.R.; Maia, F.R.; Caballero, D.; Kundu, S.C.; Reis, R.L.; Oliveira, J.M. Peptide-modified dendrimer nanoparticles for targeted therapy of colorectal cancer. Adv. Ther. 2019, 2, 1900132. [Google Scholar] [CrossRef] [Green Version]
- Thorén, P.E.; Persson, D.; Lincoln, P.; Nordén, B. Membrane destabilizing properties of cell-penetrating peptides. Biophys. Chem. 2005, 114, 169–179. [Google Scholar] [CrossRef]
- Fukunaga, K.; Tsutsumi, H.; Mihara, H. Self-assembling peptides as building blocks of functional materials for biomedical applications. Bull. Chem. Soc. Jpn. 2019, 92, 391–399. [Google Scholar] [CrossRef] [Green Version]
- Tesauro, D.; Accardo, A.; Diaferia, C.; Milano, V.; Guillon, J.; Ronga, L.; Rossi, F. Peptide-based drug-delivery systems in biotechnological applications: Recent advances and perspectives. Molecules 2019, 24, 351. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Guidotti, G.; Brambilla, L.; Rossi, D. Cell-penetrating peptides: From basic research to clinics. Trends Pharmacol. Sci. 2017, 38, 406–424. [Google Scholar] [CrossRef] [PubMed]
- Reissmann, S. Cell penetration: Scope and limitations by the application of cell-penetrating peptides. J. Pept. Sci. 2014, 20, 760–784. [Google Scholar] [CrossRef] [PubMed]
- Jang, S.; Hyun, S.; Kim, S.; Lee, S.; Lee, I.S.; Baba, M.; Lee, Y.; Yu, J. Cell-penetrating, dimeric α-helical peptides: Nanomolar inhibitors of hiv-1 transcription. Angew. Chem. 2014, 126, 10250–10253. [Google Scholar] [CrossRef]
- Oh, J.H.; Chong, S.E.; Nam, S.; Hyun, S.; Choi, S.; Gye, H.; Jang, S.; Jang, J.; Hwang, S.W.; Yu, J. Multimeric amphipathic α-helical sequences for rapid and efficient intracellular protein transport at nanomolar concentrations. Adv. Sci. 2018, 5, 1800240. [Google Scholar] [CrossRef]
- Hyun, S.; Choi, Y.; Lee, H.N.; Lee, C.; Oh, D.; Lee, D.-K.; Lee, C.; Lee, Y.; Yu, J. Construction of histidine-containing hydrocarbon stapled cell penetrating peptides for in vitro and in vivo delivery of sirnas. Chem. Sci. 2018, 9, 3820–3827. [Google Scholar] [CrossRef] [Green Version]
- Dougherty, P.G.; Wen, J.; Pan, X.; Koley, A.; Ren, J.-G.; Sahni, A.; Basu, R.; Salim, H.; Appiah Kubi, G.; Qian, Z. Enhancing the cell permeability of stapled peptides with a cyclic cell-penetrating peptide. J. Med. Chem. 2019, 62, 10098–10107. [Google Scholar] [CrossRef]
- LaRochelle, J.R.; Cobb, G.B.; Steinauer, A.; Rhoades, E.; Schepartz, A. Fluorescence correlation spectroscopy reveals highly efficient cytosolic delivery of certain penta-arg proteins and stapled peptides. J. Am. Chem. Soc. 2015, 137, 2536–2541. [Google Scholar] [CrossRef] [Green Version]
- Hughes, J.P.; Rees, S.; Kalindjian, S.B.; Philpott, K.L. Principles of early drug discovery. Br. J. Pharmacol. 2011, 162, 1239–1249. [Google Scholar] [CrossRef] [Green Version]
- Park, S.E.; El-Sayed, N.S.; Shamloo, K.; Lohan, S.; Kumar, S.; Sajid, M.I.; Tiwari, R.K. Targeted delivery of cabazitaxel using cyclic cell-penetrating peptide and biomarkers of extracellular matrix for prostate and breast cancer therapy. Bioconj. Chem. 2021, 32, 1898–1914. [Google Scholar] [CrossRef]
- Tripodi, A.A.P.; Ranđelović, I.; Biri-Kovács, B.; Szeder, B.; Mező, G.; Tóvári, J. In vivo tumor growth inhibition and antiangiogenic effect of cyclic ngr peptide-daunorubicin conjugates developed for targeted drug delivery. Pathol. Oncol. Res. 2020, 26, 1879–1892. [Google Scholar] [CrossRef] [PubMed] [Green Version]
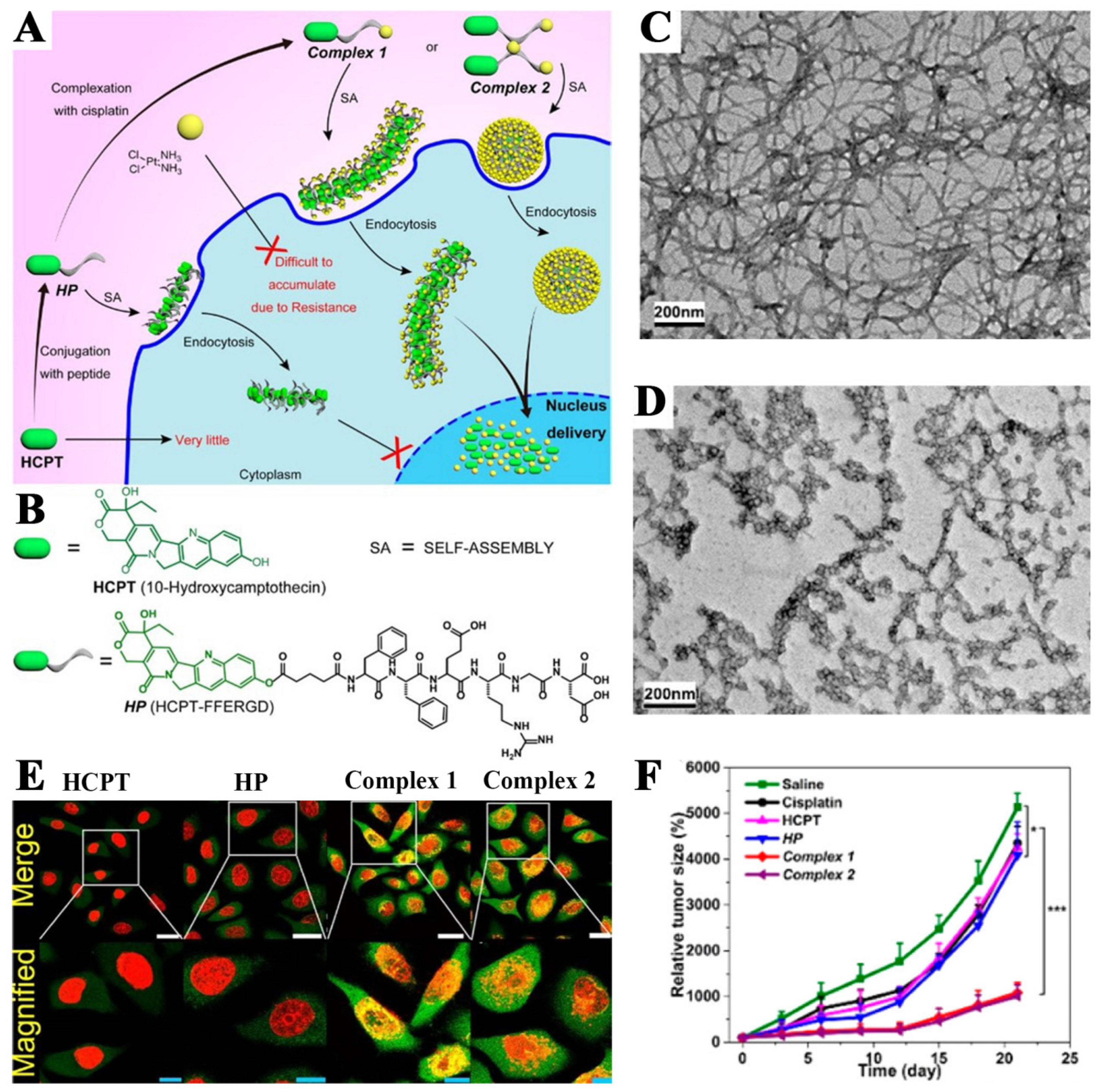
- Cai, Y.; Shen, H.; Zhan, J.; Lin, M.; Dai, L.; Ren, C.; Shi, Y.; Liu, J.; Gao, J.; Yang, Z. Supramolecular “trojan horse” for nuclear delivery of dual anticancer drugs. J. Am. Chem. Soc. 2017, 139, 2876–2879. [Google Scholar] [CrossRef] [PubMed]
- Ma, Y.; Mou, Q.; Zhu, X.; Yan, D. Small molecule nanodrugs for cancer therapy. Mater. Today Chem. 2017, 4, 26–39. [Google Scholar] [CrossRef]
- Firer, M.A.; Gellerman, G. Targeted drug delivery for cancer therapy: The other side of antibodies. J. Hematol. Oncol. 2012, 5, 70. [Google Scholar] [CrossRef] [Green Version]
- Li, J.; Li, X.; Liu, P. Doxorubicin-doxorubicin conjugate prodrug as drug self-delivery system for intracellular ph-triggered slow release. Colloids Surf. B Biointerfaces 2020, 185, 110608. [Google Scholar] [CrossRef]
- Gao, D.; Guo, X.; Zhang, X.; Chen, S.; Wang, Y.; Chen, T.; Huang, G.; Gao, Y.; Tian, Z.; Yang, Z. Multifunctional phototheranostic nanomedicine for cancer imaging and treatment. Mater. Today Bio. 2020, 5, 100035. [Google Scholar] [CrossRef] [PubMed]
- Hou, M.; Li, S.; Xu, Z.; Li, B. A reduction-responsive amphiphilic methotrexate-podophyllotoxin conjugate for targeted chemotherapy. Chem. Asian J. 2019, 14, 3840–3844. [Google Scholar] [CrossRef]
- Cheng, C.; Sui, B.; Wang, M.; Hu, X.; Shi, S.; Xu, P. Carrier-free nanoassembly of curcumin–erlotinib conjugate for cancer targeted therapy. Adv. Healthc. Mater. 2020, 9, 2001128. [Google Scholar] [CrossRef]
- Liu, Y.; Bhattarai, P.; Dai, Z.; Chen, X. Photothermal therapy and photoacoustic imaging via nanotheranostics in fighting cancer. Chem. Soc. Rev. 2019, 48, 2053–2108. [Google Scholar] [CrossRef]
- Beik, J.; Abed, Z.; Ghoreishi, F.S.; Hosseini-Nami, S.; Mehrzadi, S.; Shakeri-Zadeh, A.; Kamrava, S.K. Nanotechnology in hyperthermia cancer therapy: From fundamental principles to advanced applications. J. Control. Release 2016, 235, 205–221. [Google Scholar] [CrossRef]
- Abadeer, N.S.; Murphy, C.J. Recent progress in cancer thermal therapy using gold nanoparticles. In Nanomaterials and Neoplasms; Taylor & Francis Group: Oxfordshire, UK, 2021; pp. 143–217. [Google Scholar]
- Tan, C.; Cao, X.; Wu, X.-J.; He, Q.; Yang, J.; Zhang, X.; Chen, J.; Zhao, W.; Han, S.; Nam, G.-H. Recent advances in ultrathin two-dimensional nanomaterials. Chem. Rev. 2017, 117, 6225–6331. [Google Scholar] [CrossRef] [PubMed]
- Gao, P.; Wang, H.; Cheng, Y. Strategies for efficient photothermal therapy at mild temperatures: Progresses and challenges. Chin. Chem. Lett. 2022, 33, 575–586. [Google Scholar] [CrossRef]
- Li, X.; Lovell, J.F.; Yoon, J.; Chen, X. Clinical development and potential of photothermal and photodynamic therapies for cancer. Nat. Rev. Clin. Oncol. 2020, 17, 657–674. [Google Scholar] [CrossRef]
- Yang, T.; Wang, Q.; Lv, X.; Song, X.; Ke, H.; Guo, Z.; Huang, X.; Hu, J.; Li, Z.; Yang, P. Albumin-coordinated assembly of clearable platinum nanodots for photo-induced cancer theranostics. Biomaterials 2018, 154, 248–260. [Google Scholar]
- Li, X.; Shan, J.; Zhang, W.; Su, S.; Yuwen, L.; Wang, L. Recent advances in synthesis and biomedical applications of two-dimensional transition metal dichalcogenide nanosheets. Small 2017, 13, 1602660. [Google Scholar] [CrossRef]
- Gai, S.; Yang, G.; Yang, P.; He, F.; Lin, J.; Jin, D.; Xing, B. Recent advances in functional nanomaterials for light–triggered cancer therapy. Nano Today 2018, 19, 146–187. [Google Scholar] [CrossRef]
- Wang, T.; Wang, D.; Yu, H.; Feng, B.; Zhou, F.; Zhang, H.; Zhou, L.; Jiao, S.; Li, Y. A cancer vaccine-mediated postoperative immunotherapy for recurrent and metastatic tumors. Nat. Commun. 2018, 9, 1532. [Google Scholar] [CrossRef] [Green Version]
- Wang, P.; Zhang, L.; Zheng, W.; Cong, L.; Guo, Z.; Xie, Y.; Wang, L.; Tang, R.; Feng, Q.; Hamada, Y. Thermo-triggered release of crispr-cas9 system by lipid-encapsulated gold nanoparticles for tumor therapy. Angew. Chem. Int. Ed. 2018, 57, 1491–1496. [Google Scholar] [CrossRef]
- Guo, Z.; Zhu, S.; Yong, Y.; Zhang, X.; Dong, X.; Du, J.; Xie, J.; Wang, Q.; Gu, Z.; Zhao, Y. Synthesis of bsa-coated bioi@ bi2s3 semiconductor heterojunction nanoparticles and their applications for radio/photodynamic/photothermal synergistic therapy of tumor. Adv. Mater. 2017, 29, 1704136. [Google Scholar] [CrossRef]
- Zhang, D.; Zhang, J.; Li, Q.; Tian, H.; Zhang, N.; Li, Z.; Luan, Y. pH-and enzyme-sensitive IR820–paclitaxel conjugate self-assembled nanovehicles for near-infrared fluorescence imaging-guided chemo–photothermal therapy. ACS Appl. Mater. Interfaces 2018, 10, 30092–30102. [Google Scholar] [CrossRef]
- Ao, M.; Yu, F.; Li, Y.; Zhong, M.; Tang, Y.; Yang, H.; Wu, X.; Zhuang, Y.; Wang, H.; Sun, X. Carrier-free nanoparticles of camptothecin prodrug for chemo-photothermal therapy: The making, in vitro and in vivo testing. J. Nanobiotechnol. 2021, 19, 350. [Google Scholar] [CrossRef] [PubMed]
- Du, Q.; Qin, X.; Zhang, M.; Zhao, Z.; Li, Q.; Ren, X.; Wang, N.; Luan, Y. A mitochondrial-metabolism-regulatable carrier-free nanodrug to amplify the sensitivity of photothermal therapy. Chem. Commun. 2021, 57, 8993–8996. [Google Scholar] [CrossRef] [PubMed]
- Shi, X.; Zhang, C.Y.; Gao, J.; Wang, Z. Recent advances in photodynamic therapy for cancer and infectious diseases. Wiley Interdiscip. Rev. Nanomed. Nanobiotechnol. 2019, 11, e1560. [Google Scholar] [CrossRef] [PubMed]
- Felsher, D.W. Cancer revoked: Oncogenes as therapeutic targets. Nat. Rev. Cancer 2003, 3, 375–379. [Google Scholar] [CrossRef] [PubMed]
- Letokhov, V. Laser biology and medicine. Nature 1985, 316, 325–330. [Google Scholar] [CrossRef]
- Rui, L.-L.; Cao, H.-L.; Xue, Y.-D.; Liu, L.-C.; Xu, L.; Gao, Y.; Zhang, W.-A. Functional organic nanoparticles for photodynamic therapy. Chin. Chem. Lett. 2016, 27, 1412–1420. [Google Scholar] [CrossRef]
- Maas, A.L.; Carter, S.L.; Wileyto, E.P.; Miller, J.; Yuan, M.; Yu, G.; Durham, A.C.; Busch, T.M. Tumor vascular microenvironment determines responsiveness to photodynamic therapy. Cancer Res. 2012, 72, 2079–2088. [Google Scholar] [CrossRef] [Green Version]
- Mroz, P.; Yaroslavsky, A.; Kharkwal, G.B.; Hamblin, M.R. Cell death pathways in photodynamic therapy of cancer. Cancers 2011, 3, 2516–2539. [Google Scholar] [CrossRef] [Green Version]
- Preise, D.; Oren, R.; Glinert, I.; Kalchenko, V.; Jung, S.; Scherz, A.; Salomon, Y. Systemic antitumor protection by vascular-targeted photodynamic therapy involves cellular and humoral immunity. Cancer Immunol. Immunother. 2009, 58, 71–84. [Google Scholar] [CrossRef]
- Castano, A.P.; Mroz, P.; Wu, M.X.; Hamblin, M.R. Photodynamic therapy plus low-dose cyclophosphamide generates antitumor immunity in a mouse model. Proc. Natl. Acad. Sci. USA 2008, 105, 5495–5500. [Google Scholar] [CrossRef] [Green Version]
- Pye, H.; Stamati, I.; Yahioglu, G.; Butt, M.A.; Deonarain, M. Antibody-directed phototherapy (ADP). Antibodies 2013, 2, 270–305. [Google Scholar] [CrossRef]
- Hao, Y.; Chen, Y.; He, X.; Yu, Y.; Han, R.; Li, Y.; Yang, C.; Hu, D.; Qian, Z. Polymeric nanoparticles with ros-responsive prodrug and platinum nanozyme for enhanced chemophotodynamic therapy of colon cancer. Adv. Sci. 2020, 7, 2001853. [Google Scholar] [CrossRef] [PubMed]
- Ha, S.Y.; Zhou, Y.; Fong, W.-P.; Ng, D.K. Multifunctional molecular therapeutic agent for targeted and controlled dual chemo-and photodynamic therapy. J. Med. Chem. 2020, 63, 8512–8523. [Google Scholar] [CrossRef] [PubMed]
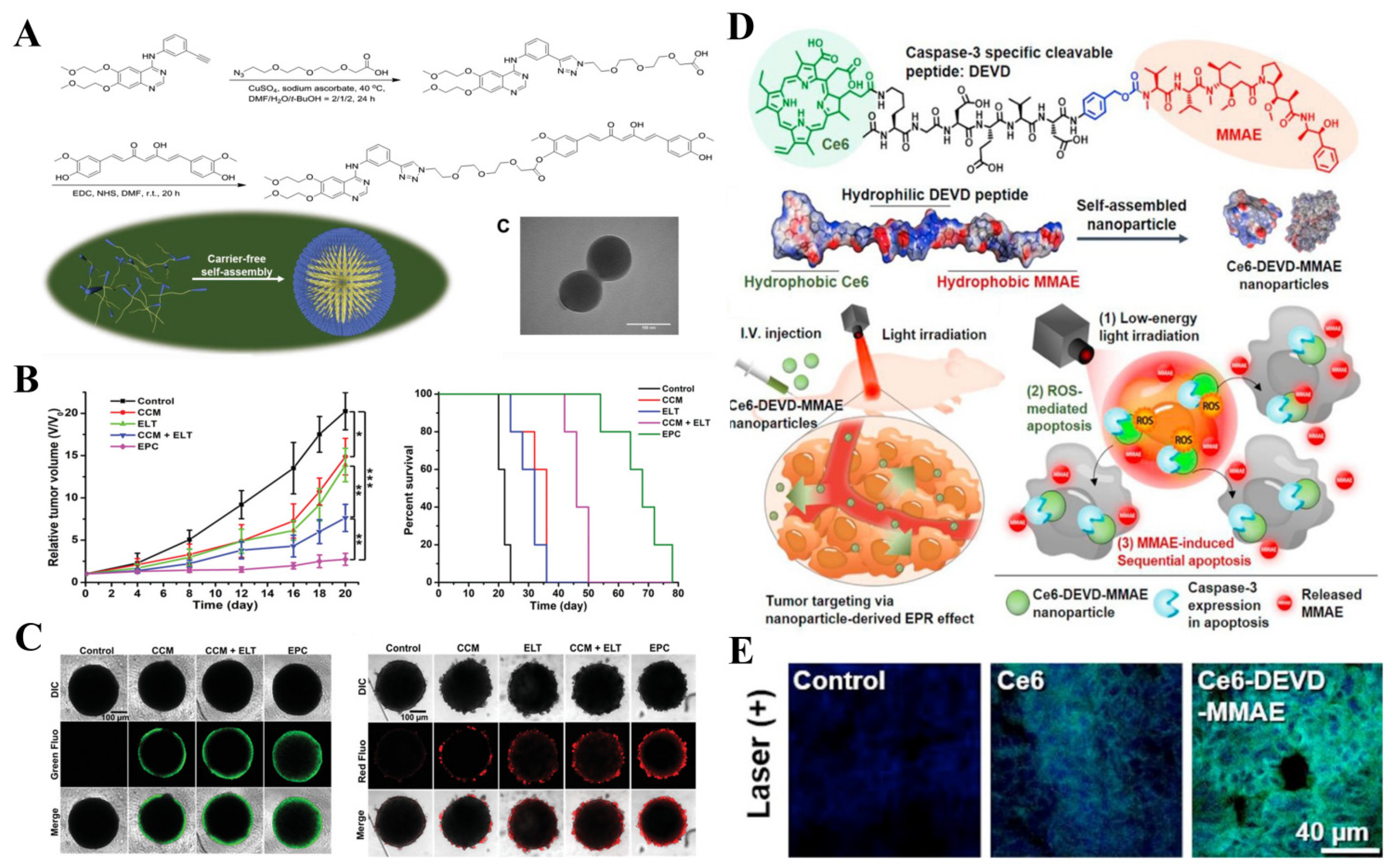
- Um, W.; Park, J.; Ko, H.; Lim, S.; Yoon, H.Y.; Shim, M.K.; Lee, S.; Ko, Y.J.; Kim, M.J.; Park, J.H. Visible light-induced apoptosis activatable nanoparticles of photosensitizer-devd-anticancer drug conjugate for targeted cancer therapy. Biomaterials 2019, 224, 119494. [Google Scholar] [CrossRef] [PubMed]
- Thankarajan, E.; Tuchinsky, H.; Aviel-Ronen, S.; Bazylevich, A.; Gellerman, G.; Patsenker, L. Antibody guided activatable NIR photosensitizing system for fluorescently monitored photodynamic therapy with reduced side effects. J. Control. Release 2022, 343, 506–517. [Google Scholar] [CrossRef] [PubMed]
- Liu, X.; Zheng, C.; Kong, Y.; Wang, H.; Wang, L. An in situ nanoparticle recombinant strategy for the enhancement of photothermal therapy. Chin. Chem. Lett. 2022, 33, 328–333. [Google Scholar] [CrossRef]
- Prasher, P.; Sharma, M.; Mudila, H.; Gupta, G.; Sharma, A.K.; Kumar, D.; Bakshi, H.A.; Negi, P.; Kapoor, D.N.; Chellappan, D.K. Emerging trends in clinical implications of bio-conjugated silver nanoparticles in drug delivery. Colloid Interface Sci. Commun. 2020, 35, 100244. [Google Scholar] [CrossRef]
- Power, S.; Slattery, M.M.; Lee, M.J. Nanotechnology and its relationship to interventional radiology. Part ii: Drug delivery, thermotherapy, and vascular intervention. Cardiovasc. Interv. Radiol. 2011, 34, 676–690. [Google Scholar] [CrossRef]
- Sanna, V.; Pala, N.; Sechi, M. Targeted therapy using nanotechnology: Focus on cancer. Int. J. Nanomed. 2014, 9, 467. [Google Scholar]
- Parra-Nieto, J.; Del Cid, M.A.G.; de Cárcer, I.A.; Baeza, A. Inorganic porous nanoparticles for drug delivery in antitumoral therapy. Biotechnol. J. 2021, 16, 2000150. [Google Scholar] [CrossRef]
- Abdellatif, A.A.; Khan, R.A.; Alhowail, A.H.; Alqasoumi, A.; Sajid, S.M.; Mohammed, A.M.; Alsharidah, M.; Al Rugaie, O.; Mousa, A.M. Octreotide-conjugated silver nanoparticles for active targeting of somatostatin receptors and their application in a nebulized rat model. Nanotechnol. Rev. 2022, 11, 266–283. [Google Scholar] [CrossRef]
- Gormley, A.J.; Larson, N.; Banisadr, A.; Robinson, R.; Frazier, N.; Ray, A.; Ghandehari, H. Plasmonic photothermal therapy increases the tumor mass penetration of hpma copolymers. J. Control. Release 2013, 166, 130–138. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Vigderman, L.; Khanal, B.P.; Zubarev, E.R. Functional gold nanorods: Synthesis, self-assembly, and sensing applications. Adv. Mater. 2012, 24, 4811–4841. [Google Scholar] [CrossRef] [PubMed]
- Kennedy, L.C.; Bickford, L.R.; Lewinski, N.A.; Coughlin, A.J.; Hu, Y.; Day, E.S.; West, J.L.; Drezek, R.A. A new era for cancer treatment: Gold-nanoparticle-mediated thermal therapies. Small 2011, 7, 169–183. [Google Scholar] [CrossRef] [PubMed]
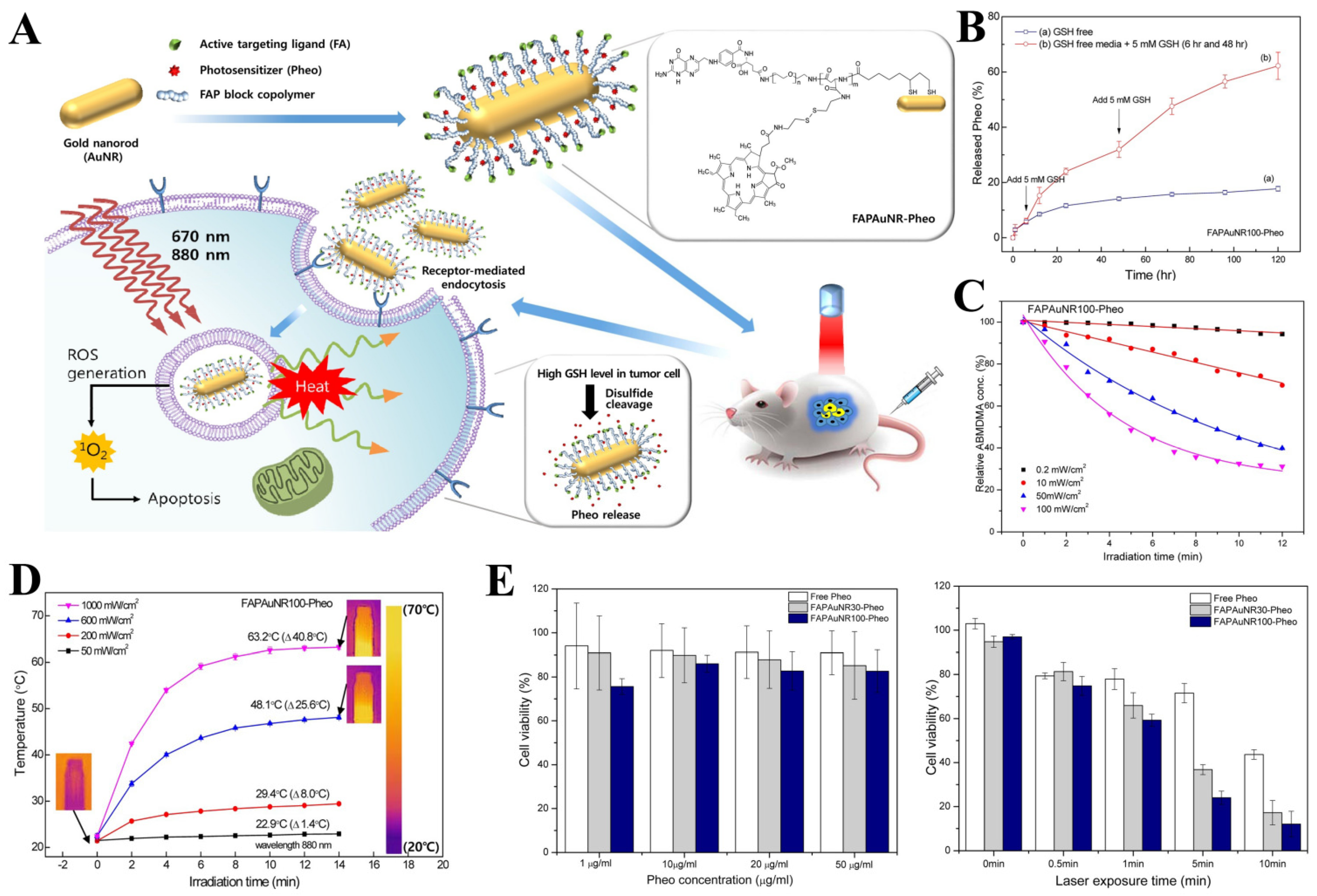
- Choi, J.; Kim, S.Y. Photothermally enhanced photodynamic therapy based on glutathione-responsive pheophorbide a-conjugated gold nanorod formulations for cancer theranostic applications. J. Ind. Eng. Chem. 2020, 85, 66–74. [Google Scholar] [CrossRef]
- Santos, H.A.; Mäkilä, E.; Airaksinen, A.J.; Bimbo, L.M.; Hirvonen, J. Porous silicon nanoparticles for nanomedicine: Preparation and biomedical applications. Nanomedicine 2014, 9, 535–554. [Google Scholar] [CrossRef]
- Duncan, R. Polymer therapeutics: Top 10 selling pharmaceuticals—what next? J. Control. Release 2014, 190, 371–380. [Google Scholar] [CrossRef]
- Peng, S.; Zhang, F.; Huang, B.; Wang, J.; Zhang, L. Mesoporous silica nanoprodrug encapsulated with near-infrared absorption dye for photothermal therapy combined with chemotherapy. ACS Appl. Bio. Mater. 2021, 4, 8225–8235. [Google Scholar] [CrossRef]
- Thakare, V.S.; Das, M.; Jain, A.K.; Patil, S.; Jain, S. Carbon nanotubes in cancer theragnosis. Nanomedicine 2010, 5, 1277–1301. [Google Scholar] [CrossRef]
- Dhar, S.; Liu, Z.; Thomale, J.; Dai, H.; Lippard, S.J. Targeted single-wall carbon nanotube-mediated pt (iv) prodrug delivery using folate as a homing device. J. Am. Chem. Soc. 2008, 130, 11467–11476. [Google Scholar] [CrossRef] [Green Version]
- Lotfollahzadeh, S.; Hosseini, E.S.; Mahmoudi Aznaveh, H.; Nikkhah, M.; Hosseinkhani, S. Trail/s-layer/graphene quantum dot nanohybrid enhanced stability and anticancer activity of trail on colon cancer cells. Sci. Rep. 2022, 12, 5851. [Google Scholar] [CrossRef] [PubMed]
- Andreou, C.; Weissleder, R.; Kircher, M.F. Multiplexed imaging in oncology. Nat. Biomed. Eng. 2022, 6, 527–540. [Google Scholar] [CrossRef] [PubMed]
- Nanayakkara, A.K.; Follit, C.A.; Chen, G.; Williams, N.S.; Vogel, P.D.; Wise, J.G. Targeted inhibitors of p-glycoprotein increase chemotherapeutic-induced mortality of multidrug resistant tumor cells. Sci. Rep. 2018, 8, 967. [Google Scholar] [CrossRef] [Green Version]
- Marusyk, A.; Almendro, V.; Polyak, K. Intra-tumour heterogeneity: A looking glass for cancer? Nat. Rev. Cancer 2012, 12, 323–334. [Google Scholar] [CrossRef] [PubMed]
- Kerbel, R. Molecular and physiologic mechanisms of drug resistance in cancer: An overview. Cancer Metastasis Rev. 2001, 20, 1. [Google Scholar] [CrossRef]
- Wang, F.; Zhang, D.; Zhang, Q.; Chen, Y.; Zheng, D.; Hao, L.; Duan, C.; Jia, L.; Liu, G.; Liu, Y. Synergistic effect of folate-mediated targeting and verapamil-mediated P-gp inhibition with paclitaxel-polymer micelles to overcome multi-drug resistance. Biomaterials 2011, 32, 9444–9456. [Google Scholar] [CrossRef]
- Jadia, R.; Scandore, C.; Rai, P. Nanoparticles for effective combination therapy of cancer. Int. J. Nanotechnol. Nanomed. 2016, 1, 1–26. [Google Scholar]
- Hu, C.-M.J.; Zhang, L. Nanoparticle-based combination therapy toward overcoming drug resistance in cancer. Biochem. Pharmacol. 2012, 83, 1104–1111. [Google Scholar] [CrossRef]
- Chiu, G.N.; Wong, M.-Y.; Ling, L.-U.; Shaikh, I.M.; Tan, K.-B.; Chaudhury, A.; Tan, B.-J. Lipid-based nanoparticulate systems for the delivery of anti-cancer drug cocktails: Implications on pharmacokinetics and drug toxicities. Curr. Drug Metab. 2009, 10, 861–874. [Google Scholar] [CrossRef]
- Hu, C.-M.J.; Aryal, S.; Zhang, L. Nanoparticle-assisted combination therapies for effective cancer treatment. Ther. Deliv. 2010, 1, 323–334. [Google Scholar] [CrossRef]
- Wang, S.; Deng, H.; Huang, P.; Sun, P.; Huang, X.; Su, Y.; Zhu, X.; Shen, J.; Yan, D. Real-time self-tracking of an anticancer small molecule nanodrug based on colorful fluorescence variations. RSC Adv. 2016, 6, 12472–12478. [Google Scholar] [CrossRef]
- Feng, L.; Gao, M.; Tao, D.; Chen, Q.; Wang, H.; Dong, Z.; Chen, M.; Liu, Z. Cisplatin-prodrug-constructed liposomes as a versatile theranostic nanoplatform for bimodal imaging guided combination cancer therapy. Adv. Funct. Mater. 2016, 26, 2207–2217. [Google Scholar] [CrossRef]
- Wang, D.; Yu, C.; Xu, L.; Shi, L.; Tong, G.; Wu, J.; Liu, H.; Yan, D.; Zhu, X. Nucleoside analogue-based supramolecular nanodrugs driven by molecular recognition for synergistic cancer therapy. J. Am. Chem. Soc. 2018, 140, 8797–8806. [Google Scholar] [CrossRef]
- Sun, M.; Qian, Q.; Shi, L.; Xu, L.; Liu, Q.; Zhou, L.; Zhu, X.; Yue, J.-M.; Yan, D. Amphiphilic drug-drug conjugate for cancer therapy with combination of chemotherapeutic and antiangiogenesis drugs. Sci. China Chem. 2020, 63, 35–41. [Google Scholar] [CrossRef]
- Wang, L.; Guo, B.; Wang, R.; Jiang, Y.; Qin, S.; Liang, S.; Zhao, Y.; Guo, W.; Li, K.; Fan, X. Inhibition of cell growth and up-regulation of mad2 in human oesophageal squamous cell carcinoma after treatment with the src/abl inhibitor dasatinib. Clin. Sci. 2012, 122, 13–24. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Yang, L.; Xu, J.; Xie, Z.; Song, F.; Wang, X.; Tang, R. Carrier-free prodrug nanoparticles based on dasatinib and cisplatin for efficient antitumor in vivo. Asian J. Pharm. Sci. 2021, 16, 762–771. [Google Scholar] [CrossRef] [PubMed]
- Shan, X.; Zhao, Z.; Wang, C.; Sun, J.; He, Z.; Luo, C.; Zhang, S. Emerging prodrug-engineered nanomedicines for synergistic chemo-phototherapy. Chem. Eng. J. 2022, 442, 136383. [Google Scholar] [CrossRef]
- Yang, Y.; Zhang, Y.; Wang, R.; Rong, X.; Liu, T.; Xia, X.; Fan, J.; Sun, W.; Peng, X. A glutathione activatable pro-drug-photosensitizer for combined chemotherapy and photodynamic therapy. Chin. Chem. Lett. 2022, 33, 4583–4586. [Google Scholar] [CrossRef]
- Castano, A.P.; Mroz, P.; Hamblin, M.R. Photodynamic therapy and anti-tumour immunity. Nat. Rev. Cancer 2006, 6, 535–545. [Google Scholar] [CrossRef] [Green Version]
- Ji, C.; Gao, Q.; Dong, X.; Yin, W.; Gu, Z.; Gan, Z.; Zhao, Y.; Yin, M. A size-reducible nanodrug with an aggregation-enhanced photodynamic effect for deep chemo-photodynamic therapy. Angew. Chem. 2018, 130, 11554–11558. [Google Scholar] [CrossRef]
- Chen, H.; Zeng, X.; Tham, H.P.; Phua, S.Z.F.; Cheng, W.; Zeng, W.; Shi, H.; Mei, L.; Zhao, Y. Nir-light-activated combination therapy with a precise ratio of photosensitizer and prodrug using a host–guest strategy. Angew. Chem. Int. Ed. 2019, 58, 7641–7646. [Google Scholar] [CrossRef] [PubMed]
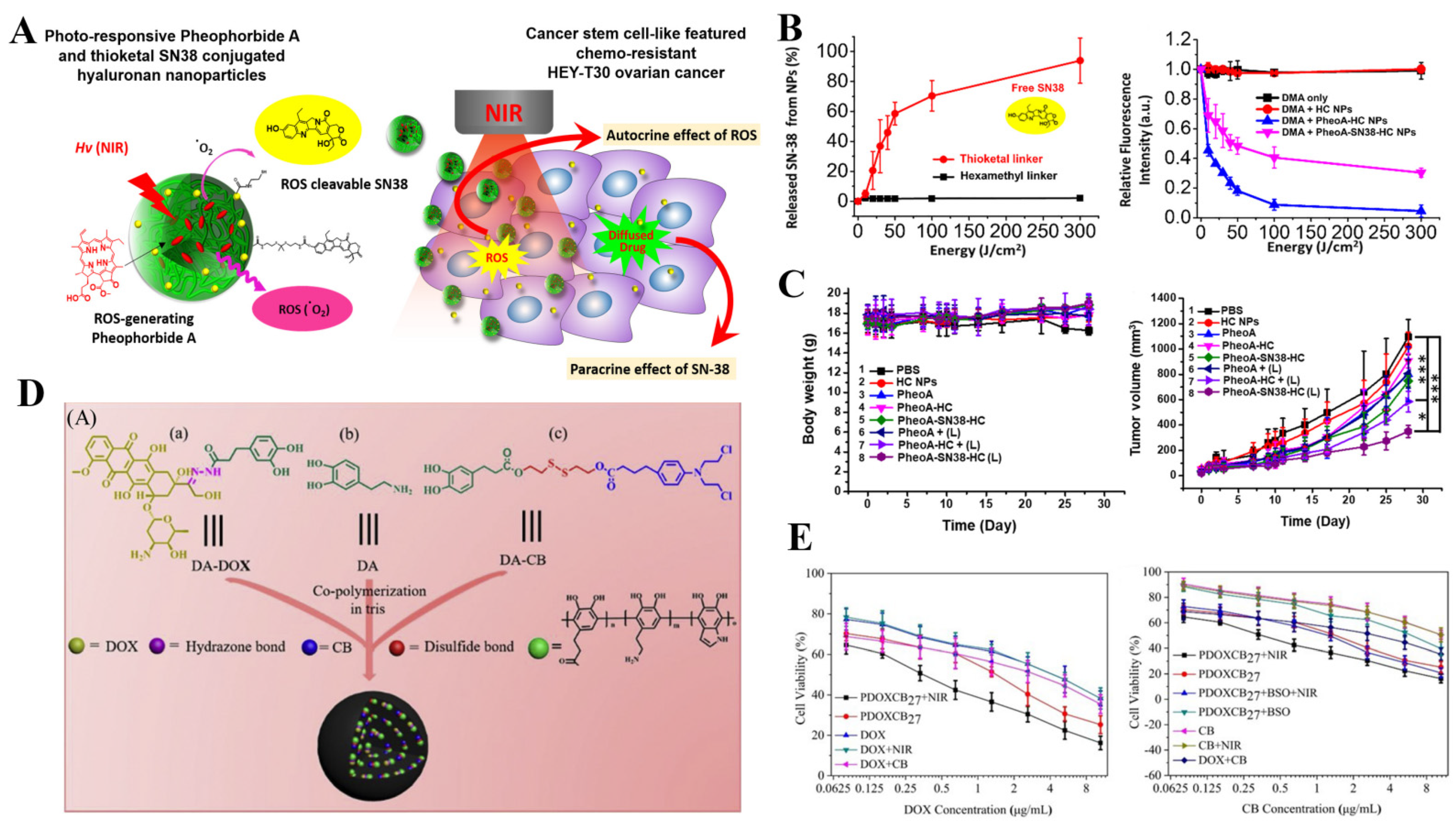
- Lee, J.; Davaa, E.; Jiang, Y.; Shin, K.-J.; Kim, M.H.; An, H.; Kim, J.; Cho, S.K.; Yang, S.-G. Pheophorbide a and sn38 conjugated hyaluronan nanoparticles for photodynamic-and cascadic chemotherapy of cancer stem-like ovarian cancer. Carbohydr. Polym. 2022, 289, 119455. [Google Scholar] [CrossRef] [PubMed]
- Xu, S.; Zhu, X.; Zhang, C.; Huang, W.; Zhou, Y.; Yan, D. Oxygen and pt (ii) self-generating conjugate for synergistic photo-chemo therapy of hypoxic tumor. Nat. Commun. 2018, 9, 2053. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; Yang, M.; Cao, J.; Gu, H.; Liu, W.; Xia, T.; Sun, W.; Fan, J.; Peng, X. H-aggregates of prodrug-hemicyanine conjugate for enhanced photothermal therapy and sequential hypoxia-activated chemotherapy. ACS Mater. Lett. 2022, 4, 724–732. [Google Scholar] [CrossRef]
- Zhou, L.; Du, C.; Zhang, R.; Dong, C. Stimuli-responsive dual drugs-conjugated polydopamine nanoparticles for the combination photothermal-cocktail chemotherapy. Chin. Chem. Lett. 2021, 32, 561–564. [Google Scholar] [CrossRef]
- Nam, J.; Son, S.; Park, K.S.; Zou, W.; Shea, L.D.; Moon, J.J. Cancer nanomedicine for combination cancer immunotherapy. Nat. Rev. Mater. 2019, 4, 398–414. [Google Scholar] [CrossRef]
- Abdel-Wahab, N.; Alshawa, A.; Suarez-Almazor, M.E. Adverse events in cancer immunotherapy. In Immunotherapy; Springer: Berlin/Heidelberg, Germany, 2017; pp. 155–174. [Google Scholar]
- June, C.H.; Warshauer, J.T.; Bluestone, J.A. Is autoimmunity the achilles’ heel of cancer immunotherapy? Nat. Med. 2017, 23, 540–547. [Google Scholar] [CrossRef] [Green Version]
- Geng, Z.; Wang, L.; Liu, K.; Liu, J.; Tan, W. Enhancing anti-pd-1 immunotherapy by nanomicelles self-assembled from multivalent aptamer drug conjugates. Angew. Chem. 2021, 133, 15587–15593. [Google Scholar] [CrossRef]
- Hu, D.; Zhang, W.; Xiang, J.; Li, D.; Chen, Y.; Yuan, P.; Shao, S.; Zhou, Z.; Shen, Y.; Tang, J. A ros-responsive synergistic delivery system for combined immunotherapy and chemotherapy. Mater. Today Bio. 2022, 14, 100284. [Google Scholar] [CrossRef]
- Bai, S.; Yang, L.L.; Wang, Y.; Zhang, T.; Fu, L.; Yang, S.; Wan, S.; Wang, S.; Jia, D.; Li, B. Prodrug-based versatile nanomedicine for enhancing cancer immunotherapy by increasing immunogenic cell death. Small 2020, 16, 2000214. [Google Scholar] [CrossRef]
- Libutti, S.K.; Paciotti, G.F.; Byrnes, A.A.; Alexander, H.R.; Gannon, W.E.; Walker, M.; Seidel, G.D.; Yuldasheva, N.; Tamarkin, L. Phase i and pharmacokinetic studies of cyt-6091, a novel pegylated colloidal gold-rhtnf nanomedicinepegylated colloidal gold-rhtnf nanomedicine phase i trial. Clin. Cancer Res. 2010, 16, 6139–6149. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Xue, G.; Wang, Z.; Zheng, N.; Fang, J.; Mao, C.; Li, X.; Jin, G.; Ming, X.; Lu, Y. Elimination of acquired resistance to pd-1 blockade via the concurrent depletion of tumour cells and immunosuppressive cells. Nat. Biomed. Eng. 2021, 5, 1306–1319. [Google Scholar] [CrossRef] [PubMed]
- Luo, Y.; Chen, Z.; Sun, M.; Li, B.; Pan, F.; Ma, A.; Liao, J.; Yin, T.; Tang, X.; Huang, G. IL-12 nanochaperone-engineered CAR T cell for robust tumor-immunotherapy. Biomaterials 2022, 281, 121341. [Google Scholar] [CrossRef]
- Li, Q.; Zhang, Y.; Huang, X.; Yang, D.; Weng, L.; Ou, C.; Song, X.; Dong, X. An NIR-II light responsive antibacterial gelation for repetitious photothermal/thermodynamic synergistic therapy. Chem. Eng. J. 2021, 407, 127200. [Google Scholar] [CrossRef]
- Liu, Y.; Tang, P.; Xiao, P.; Luo, S.; Zhang, S.; Zhang, H.; Yang, Y.; Wu, D. Molecular stacking composite nanoparticles of gossypolone and thermodynamic agent for elimination of large tumor in mice via electrothermal-thermodynamic-chemo trimodal combination therapy. Adv. Funct. Mater. 2022, 32, 2201666. [Google Scholar] [CrossRef]
- Xia, R.; Zheng, X.; Hu, X.; Liu, S.; Xie, Z. Photothermal-controlled generation of alkyl radical from organic nanoparticles for tumor treatment. ACS Appl. Mater. Interfaces 2019, 11, 5782–5790. [Google Scholar] [CrossRef]
- Gao, D.; Chen, T.; Chen, S.; Ren, X.; Han, Y.; Li, Y.; Wang, Y.; Guo, X.; Wang, H.; Chen, X. Targeting hypoxic tumors with hybrid nanobullets for oxygen-independent synergistic photothermal and thermodynamic therapy. Nano-Micro Lett. 2021, 13, 99. [Google Scholar] [CrossRef] [PubMed]
- Gao, D.; Shi, Y.; Ni, J.; Chen, S.; Wang, Y.; Zhao, B.; Song, M.; Guo, X.; Ren, X.; Zhang, X. Nir/mri-guided oxygen-independent carrier-free anti-tumor nano-theranostics. Small 2021, 2106000. [Google Scholar] [CrossRef]



| Trade Name | Active Principle | Nanotechnology Platform | Indication | Approved Status |
|---|---|---|---|---|
| Doxil/Caelyx | Doxorubicin | PEGylated liposomes | Breast cancer, ovarian cancer, myeloma | FDA 1, EMA 2 |
| DaunoXome | Daunorubicin | Liposomes | Kaposi sarcoma | FDA |
| Myocet | Doxorubicin | Liposomes | Breast cancer | FDA |
| Lipusu | Paclitaxel | Liposomes | breast cancer, non-small-cell lung cancer | NMPA 3 |
| Abraxane | Paclitaxel | Albumin-bound nanoparticles | Metastatic breast cancer, metastatic pancreatic cancer, advanced non-small-cell lung cancer | FDA, EMA |
| Genexol-PM | Paclitaxel | Polymeric micelles | Non-small-cell lung cancer | KFDA 4 |
| MEPACT | Mifamurtide | Liposomes | Osteosarcoma | EMA |
| Marqibo | Vincristine | Liposomes | Philadelphia chromosome-negative acute lymphoblastic leukemia | FDA |
| PICN | Paclitaxel | Polymer/lipid nanoparticles | Metastatic breast cancer | India |
| Onivyde (MM-398) | Irinotecan | PEGylated liposomes | Metastatic pancreatic cancer | FDA |
| VYXEOS | Cytarabine/daunorubicin (5:1) | Liposomes | Acute myeloid leukaemia | FDA, EMA |
| Paclical | Paclitaxel | Polymeric micelles | Ovarian cancer | Russia |
| Hensify | N/A | Hafnium oxide NP | Locally advanced soft tissue sarcoma | EMA |
| DHP107 | Paclitaxel | Lipid nanoparticles | Advanced gastric cancer | KFDA |
| NanoTherm | N/A | Iron oxide nanoparticles | Recurrent glioblastoma | EMA |
| Nanoxel | Paclitaxel | Polymeric micelles | Breast cancer, ovarian cancer | India |
| Depocyt | Cytarabine | Liposomes | Acute Nonlymphocytic Leukemia, Meningeal Leukemia, Lymphomatous Meningitis | FDA |
| Trade Name | ADC | Target Antigen | Indication | Approved Status |
|---|---|---|---|---|
| Mylotarg | Gemtuzumab ozogamicin | CD33 | CD33 positive AML | FDA |
| Adcetris | Brentuximab vedotin | CD30 | Hodgkin lymphoma and anaplastic large cell lymphoma | FDA |
| Kadcyla | Ado-trastuzumab emtansine | HER2 | HER2-positive breast cancer | FDA |
| Besponsa | Inotuzumab ozogamicin | CD22 | Relapsed or refractory B cell precursor acute lymphoblastic leukemia | FDA |
| Lumoxiti | Moxetumomab pasudotox | CD22 | Relapsed or refractory B-cell precursor acute lymphoblastic leukemia | FDA |
| Polivy | Polatuzumab vedotin | CD79b | Relapsed or refractory diffuse large B cell lymphoma | FDA |
| Padcev | Enfortumab vedotin | Nectin-4 | Advanced or metastatic urothelial | FDA |
| Enhertu | Trastuzumab deruxtecan | HER2 | Relapsed or refractory diffuse large B cell lymphoma | FDA |
| Trodelvy | Sacituzumab govitecan | Trop-2 | HER2-triple-negative breast cancer | FDA |
| Blenrep | Belantamab mafodotin | BCMA | Relapsed or refractory multiple myeloma | FDA |
| Akalux | Cetuximab IRDye700DX | EGFR | Head and neck tumors, esophageal tumors, lung tumors, colon cancers | PMSB 1 |
| Zvnionta | Loncastuximab tesirine | CD19 | Relapsed or refractory diffuse large B cell lymphoma | FDA |
| Aidixi | Disitamab vedotin | HER-2 | HER-2 positive metastatic gastric cancer | NMPA |
| Tivdak | Tisotumab vedotin-tftv | TF | Relapsed or metastatic cervical cancer | FDA |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zhao, B.; Chen, S.; Hong, Y.; Jia, L.; Zhou, Y.; He, X.; Wang, Y.; Tian, Z.; Yang, Z.; Gao, D. Research Progress of Conjugated Nanomedicine for Cancer Treatment. Pharmaceutics 2022, 14, 1522. https://doi.org/10.3390/pharmaceutics14071522
Zhao B, Chen S, Hong Y, Jia L, Zhou Y, He X, Wang Y, Tian Z, Yang Z, Gao D. Research Progress of Conjugated Nanomedicine for Cancer Treatment. Pharmaceutics. 2022; 14(7):1522. https://doi.org/10.3390/pharmaceutics14071522
Chicago/Turabian StyleZhao, Bin, Sa Chen, Ye Hong, Liangliang Jia, Ying Zhou, Xinyu He, Ying Wang, Zhongmin Tian, Zhe Yang, and Di Gao. 2022. "Research Progress of Conjugated Nanomedicine for Cancer Treatment" Pharmaceutics 14, no. 7: 1522. https://doi.org/10.3390/pharmaceutics14071522
APA StyleZhao, B., Chen, S., Hong, Y., Jia, L., Zhou, Y., He, X., Wang, Y., Tian, Z., Yang, Z., & Gao, D. (2022). Research Progress of Conjugated Nanomedicine for Cancer Treatment. Pharmaceutics, 14(7), 1522. https://doi.org/10.3390/pharmaceutics14071522







