3D Bioprinted Chitosan-Based Hydrogel Scaffolds in Tissue Engineering and Localised Drug Delivery
Abstract
:1. Introduction
2. Chitosan-Based Bioinks for Tissue Engineering
2.1. Bioinks of Composites of Chitosan Hydrogels
2.2. Bioinks of Chitosan-Modified Hydrogels
3. Chitosan-Based Biomaterial-Inks
3.1. Chitosan-Based Biomaterial Inks for Tissue Engineering Applications
3.1.1. Biomaterial Inks of Chitosan and Chitosan Composites
3.1.2. Biomaterial Inks of Polyelectrolyte Complexes of Chitosan
3.1.3. Biomaterial Inks of Modified Chitosan
3.2. Chitosan-Based Biomaterial Inks for Active Substance (API)-Loaded Scaffolds
3.2.1. Biomaterial inks of Loaded Chitosan Composites
3.2.2. Biomaterial Inks of Loaded Chitosan Derivatives or Polyelectrolyte Complexes of Chitosan
4. Regulatory Aspects
5. Expert Opinion and Future Perspectives
6. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| AC | Articular cartilage |
| AcMMT | Acrylated montmorillonite |
| ACP | Amorphous calcium phosphate |
| ADH | Adipic acid dihydrazide |
| AG | Agarose |
| AHA | Aldehyde hyaluronic acid |
| ALP | Alkaline phosphatase |
| AM | Acrylamide |
| ATMPs | Advanced therapy medicinal products |
| BCP | Biphasic calcium phosphate |
| BG | Bioglass |
| BMP | Bone morphogenic protein |
| BMSCs | Bone mesenchymal stem cells |
| C | Succinylated chitosan |
| CAD | Computer aided design |
| CCP | Chitosan cyclodextrin with propolis extract |
| CG | Chitosan crosslinked with genipin |
| Chi-C | Catechol-conjugated chitosan |
| Chi-Ph | Phenol chitosan |
| ChMA | Chitosan methacrylate |
| CMC | Carboxymethyl chitosan |
| CNC | Cellulose nanocrystals |
| CP | Chitosan crosslinked with pectin |
| CS | Chitosan |
| D | Dextran aldehyde |
| DDD | Drug delivery devices |
| DDS | Drug delivery system |
| DLP | Digital light processing |
| EC | European Commission |
| EDC | N-(3-dimethylaminopropyl)-N’-ethyl carbodiimide hydrochloride |
| EDTA | Ethylenediaminetetraacetic acid |
| ECM | Extracellular matrix |
| FBS | Fetal bovine serum |
| FDA | Food and Drug Administration |
| FRESH | Freeform reversible embedding of suspended hydrogels |
| FS | Fluorescein sodium |
| FTIR | Fourier |
| G1Phy | glycerylphytate |
| Gamma-PGA | Poly(gamma-glutamic acid) |
| GC | Glycol chitosan |
| Gel | Gelatin |
| GelMA | Methacrylate gelatin |
| Gly | Glycerol |
| GM | Glycidyl methacrylate |
| GO | Graphene oxide |
| HA | Hyaluronic acid |
| HaCaT | Aneploid immortal keratinocyte cells |
| HAMSCs | Human derived mesenchymal cells |
| HAp | Hydroxyapatite |
| HA-SG | Thiolated hyaluronic acid |
| HBC | Hydroxybutyl chitosan |
| hBMSCs | Human bone mesenchymal stem cells |
| HDF | Human dermal fibroblasts |
| HECS | Hydroxyethyl chitosan |
| HMSCs | Human adipose-derived mesenchymal stem cells |
| HUVECs | Human umbilical vein endothelial cells |
| ION | Iron oxide nanoparticle |
| IPEC | Inter polyelectrolyte complex |
| KC | Keratinocytes |
| K2HPO4 | Potassium phosphate |
| LCST | Low critical solution temperature |
| LEV | Levofloxacin |
| MA | Matrigel |
| MAA | Market authorization application |
| MeGC | Methacrylate glycol chitosan |
| MF | Silk microfibres |
| MG-63 | Osteosarcoma cell line |
| MSCs | Mensechymal stem cells |
| NaHCO3 | Sodium bicarbonate |
| nanoHAp | Nanohydroxyapatite |
| NF | Silk nanofibres |
| Nhdf | Normal dermal human fibroblast cells |
| NIH/3T3 | Fibroblasts |
| NHS | N-hydroxysuccinimide |
| NOCC | N,O Carboxylmethyl chitosan |
| NPs | Nanoparticles |
| NSCs | Neural stem cells |
| OCS | Oxidized chondroitin sulfate |
| OHA | Oxidized hyaluronate |
| PAM | Poly(acrylamide) |
| PBS | Phosphate-buffered saline |
| PCL | Poly(e-caprolactone) |
| PDLSCs | Periodontal ligament stem cells |
| Pec | Pectin |
| PECs | Polyelectrolyte complexes |
| PEG | Polyethylene glycol |
| PEG-SG | Poly(ethylene glycol) succinimidyl glutarate |
| PgGA | Poly(gamma-glutamic acid) |
| PLA | Poly(lactic acid) |
| PLGA | Polylactic-co glycolic acid |
| pNiPAM | Poly(N-isopropyl acrylamide) |
| polyP | Polyphosphate |
| RBC | Red blood cells |
| rhBMP | Recombinant human bone morphogenic protein |
| Saos-2 | Osteoblast-like cells |
| SCI | Spinal cord injury |
| SH-SY5Y | Neuronal-like cells |
| SMSCs | Synovial mesenchymal stem cells |
| SP | Silk powder |
| SPECs | Surfactants and polyelectrolytes complexes |
| SPS | Sodium persulfate |
| TCNF | TEMPO-mediated oxidized cellulose nanofibrils |
| TECs | Tissue engineering constructs |
| TEMPO | 2,2,6,6-tetramethylpiperidinyl-1-oxyl |
| TFNA | Tetrahedral framework nucleic acid |
| TPP | Tripolyphosphate |
| VACS | Vanillin chitosan |
| VitE | α-tocopherol |
| βGP | β-glycerophosphate |
References
- 3D Hubs Manufacturing LLC 3D Printing Trends 2020. Available online: https://downloads.hubs.com/3D_printing_trends_report_2020.pdf (accessed on 19 August 2022).
- Foyt, D.A.; Norman, M.D.A.; Yu, T.T.L.; Gentleman, E. Exploiting Advanced Hydrogel Technologies to Address Key Challenges in Regenerative Medicine. Adv. Healthc. Mater. 2018, 7, e1700939. [Google Scholar] [CrossRef] [PubMed]
- Groll, J.; Burdick, J.A.; Cho, D.W.; Derby, B.; Gelinsky, M.; Heilshorn, S.C.; Jüngst, T.; Malda, J.; Mironov, V.A.; Nakayama, K.; et al. A definition of bioinks and their distinction from biomaterial inks. Biofabrication 2019, 11, 013001. [Google Scholar] [CrossRef] [PubMed]
- Mironov, V. Printing technology to produce living tissue. Expert Opin. Biol. Ther. 2003, 3, 701–704. [Google Scholar] [CrossRef] [PubMed]
- Boland, T.; Mironov, V.; Gutowska, A.; Roth, E.A.; Markwald, R.R. Cell and organ printing 2: Fusion of cell aggregates in three-dimensional gels. Anat. Rec. Part A Discov. Mol. Cell. Evol. Biol. 2003, 272, 497–502. [Google Scholar] [CrossRef] [PubMed]
- Schuurman, W.; Khristov, V.; Pot, M.W.; van Weeren, P.R.; Dhert, W.J.A.; Malda, J. Bioprinting of hybrid tissue constructs with tailorable mechanical properties. Biofabrication 2011, 3, 021001. [Google Scholar] [CrossRef] [PubMed]
- Shim, J.H.; Kim, J.Y.; Park, M.; Park, J.; Cho, D.W. Development of a hybrid scaffold with synthetic biomaterials and hydrogel using solid freeform fabrication technology. Biofabrication 2011, 3, 034102. [Google Scholar] [CrossRef] [PubMed]
- Paxton, N.; Smolan, W.; Böck, T.; Melchels, F.; Groll, J.; Jungst, T. Proposal to assess printability of bioinks for extrusion-based bioprinting and evaluation of rheological properties governing bioprintability. Biofabrication 2017, 9, 044107. [Google Scholar] [CrossRef] [PubMed]
- Zamboulis, A.; Nanaki, S.; Michailidou, G.; Koumentakou, I.; Lazaridou, M.; Ainali, N.M.; Xanthopoulou, E.; Bikiaris, D.N. Chitosan and its derivatives for ocular delivery formulations: Recent advances and developments. Polymers 2020, 12, 1519. [Google Scholar] [CrossRef]
- Koumentakou, I.; Terzopoulou, Z.; Michopoulou, A.; Kalafatakis, I.; Theodorakis, K.; Tzetzis, D.; Bikiaris, D. Chitosan dressings containing inorganic additives and levofloxacin as potential wound care products with enhanced hemostatic properties. Int. J. Biol. Macromol. 2020, 162, 693–703. [Google Scholar] [CrossRef]
- Croisier, F.; Jérôme, C. Chitosan-based biomaterials for tissue engineering. Eur. Polym. J. 2013, 49, 780–792. [Google Scholar] [CrossRef] [Green Version]
- Lazaridou, M.; Nanaki, S.; Zamboulis, A.; Papoulia, C.; Chrissafis, K.; Klonos, P.A.; Kyritsis, A.; Vergkizi-Nikolakaki, S.; Kostoglou, M.; Bikiaris, D.N. Super absorbent chitosan-based hydrogel sponges as carriers for caspofungin antifungal drug. Int. J. Pharm. 2021, 606, 120925. [Google Scholar] [CrossRef]
- Karava, A.; Lazaridou, M.; Nanaki, S.; Michailidou, G.; Christodoulou, E.; Kostoglou, M.; Iatrou, H.; Bikiaris, D.N. Chitosan Derivatives with Mucoadhesive and Antimicrobial Properties for Simultaneous Nanoencapsulation and Extended Ocular Release Formulations of Dexamethasone and Chloramphenicol Drugs. Pharmaceutics 2020, 12, 594. [Google Scholar] [CrossRef]
- Khoeini, R.; Nosrati, H.; Akbarzadeh, A.; Eftekhari, A.; Kavetskyy, T.; Khalilov, R.; Ahmadian, E.; Nasibova, A.; Datta, P.; Roshangar, L.; et al. Natural and Synthetic Bioinks for 3D Bioprinting. Adv. NanoBiomed Res. 2021, 1, 2000097. [Google Scholar] [CrossRef]
- Li, P.; Fu, L.; Liao, Z.; Peng, Y.; Ning, C.; Gao, C.; Zhang, D.; Sui, X.; Lin, Y.; Liu, S.; et al. Chitosan hydrogel/3D-printed poly(ε-caprolactone) hybrid scaffold containing synovial mesenchymal stem cells for cartilage regeneration based on tetrahedral framework nucleic acid recruitment. Biomaterials 2021, 278, 121131. [Google Scholar] [CrossRef]
- Ku, J.; Seonwoo, H.; Park, S.; Jang, K.J.; Lee, J.; Lee, M.; Lim, J.W.; Kim, J.; Chung, J.H. Cell-laden thermosensitive chitosan hydrogel bioinks for 3d bioprinting applications. Appl. Sci. 2020, 10, 2455. [Google Scholar] [CrossRef]
- Intini, C.; Elviri, L.; Cabral, J.; Mros, S.; Bergonzi, C.; Bianchera, A.; Flammini, L.; Govoni, P.; Barocelli, E.; Bettini, R.; et al. 3D-printed chitosan-based scaffolds: An in vitro study of human skin cell growth and an in-vivo wound healing evaluation in experimental diabetes in rats. Carbohydr. Polym. 2018, 199, 593–602. [Google Scholar] [CrossRef]
- Pisani, S.; Dorati, R.; Scocozza, F.; Mariotti, C.; Chiesa, E.; Bruni, G.; Genta, I.; Auricchio, F.; Conti, M.; Conti, B. Preliminary investigation on a new natural based poly(gamma-glutamic acid)/Chitosan bioink. J. Biomed. Mater. Res. Part B Appl. Biomater. 2020, 108, 2718–2732. [Google Scholar] [CrossRef]
- Maturavongsadit, P.; Narayanan, L.K.; Chansoria, P.; Shirwaiker, R.; Benhabbour, S.R. Cell-Laden Nanocellulose/Chitosan-Based Bioinks for 3D Bioprinting and Enhanced Osteogenic Cell Differentiation. ACS Appl. Bio Mater. 2021, 4, 2342–2353. [Google Scholar] [CrossRef]
- Roehm, K.D.; Madihally, S.V. Bioprinted chitosan-gelatin thermosensitive hydrogels using an inexpensive 3D printer. Biofabrication 2018, 10, 015002. [Google Scholar] [CrossRef]
- Rahimnejad, M.; Adoungotchodo, A.; Demarquette, N.R.; Lerouge, S. FRESH bioprinting of biodegradable chitosan thermosensitive hydrogels. Bioprinting 2022, 27, e00209. [Google Scholar] [CrossRef]
- Hafezi, F.; Shorter, S.; Tabriz, A.G.; Hurt, A.; Elmes, V.; Boateng, J.; Douroumis, D. Bioprinting and preliminary testing of highly reproducible novel bioink for potential skin regeneration. Pharmaceutics 2020, 12, 550. [Google Scholar] [CrossRef]
- Chang, H.K.; Yang, D.H.; Ha, M.Y.; Kim, H.J.; Kim, C.H.; Kim, S.H.; Choi, J.W.; Chun, H.J. 3D printing of cell-laden visible light curable glycol chitosan bioink for bone tissue engineering. Carbohydr. Polym. 2022, 287, 119328. [Google Scholar] [CrossRef]
- Tonda-Turo, C.; Carmagnola, I.; Chiappone, A.; Feng, Z.; Ciardelli, G.; Hakkarainen, M.; Sangermano, M. Photocurable chitosan as bioink for cellularized therapies towards personalized scaffold architecture. Bioprinting 2020, 18, e00082. [Google Scholar] [CrossRef]
- Gaihre, B.; Liu, X.; Tilton, M.; Li, L.; Li, Y.; Lu, L. Spatial and uniform deposition of cell-laden constructs on 3D printed composite phosphorylated hydrogels for improved osteoblast responses. J. Mater. Sci. 2021, 56, 17768–17784. [Google Scholar] [CrossRef]
- Lin, F.; Jia, H.R.; Wu, F.G. Glycol chitosan: A water-soluble polymer for cell imaging and drug delivery. Molecules 2019, 24, 4371. [Google Scholar] [CrossRef]
- Roh, H.H.; Kim, H.S.; Kim, C.; Lee, K.Y. 3D Printing of Polysaccharide-Based Self-Healing Hydrogel Reinforced With Alginate for Secondary Cross-Linking. Biomedicines 2021, 9, 1224. [Google Scholar] [CrossRef]
- Butler, H.M.; Naseri, E.; MacDonald, D.S.; Tasker, R.A.; Ahmadi, A. Investigation of rheology, printability, and biocompatibility of N,O-carboxymethyl chitosan and agarose bioinks for 3D bioprinting of neuron cells. Materialia 2021, 18, 101169. [Google Scholar] [CrossRef]
- Neufurth, M.; Wang, S.; Schröder, H.C.; Al-Nawas, B.; Wang, X.; Müller, W.E.G. 3D bioprinting of tissue units with mesenchymal stem cells, retaining their proliferative and differentiating potential, in polyphosphate-containing bio-ink. Biofabrication 2022, 14, 015016. [Google Scholar] [CrossRef] [PubMed]
- Chen, H.; Fei, F.; Li, X.; Nie, Z.; Zhou, D.; Liu, L.; Zhang, J.; Zhang, H.; Fei, Z.; Xu, T. A structure-supporting, self-healing, and high permeating hydrogel bioink for establishment of diverse homogeneous tissue-like constructs. Bioact. Mater. 2021, 6, 3580–3595. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.; Cao, Y.; Shen, Z.; Li, M.; Zhang, W.; Liu, Y.; Zhang, Y.; Duan, J.; Ma, Z.; Sang, S. 3D printed GelMA/carboxymethyl chitosan composite scaffolds for vasculogenesis. Int. J. Polym. Mater. Polym. Biomater. 2022, 1–13. [Google Scholar] [CrossRef]
- Puertas-Bartolomé, M.; Włodarczyk-Biegun, M.K.; del Campo, A.; Vázquez-Lasa, B.; San Román, J. Development of bioactive catechol functionalized nanoparticles applicable for 3D bioprinting. Mater. Sci. Eng. C 2021, 131, 112515. [Google Scholar] [CrossRef]
- Zhao, C.; Qazvini, N.T.; Sadati, M.; Zeng, Z.; Huang, S.; De La Lastra, A.L.; Zhang, L.; Feng, Y.; Liu, W.; Huang, B.; et al. A pH-Triggered, Self-Assembled, and Bioprintable Hybrid Hydrogel Scaffold for Mesenchymal Stem Cell Based Bone Tissue Engineering. ACS Appl. Mater. Interfaces 2019, 11, 8749–8762. [Google Scholar] [CrossRef]
- Liu, Y.; Wong, C.W.; Chang, S.W.; Hsu, S. Hui An injectable, self-healing phenol-functionalized chitosan hydrogel with fast gelling property and visible light-crosslinking capability for 3D printing. Acta Biomater. 2021, 122, 211–219. [Google Scholar] [CrossRef]
- Nie, L.; Li, J.; Lu, G.; Wei, X.; Deng, Y.; Liu, S.; Zhong, S.; Shi, Q.; Hou, R.; Sun, Y.; et al. Temperature responsive hydrogel for cells encapsulation based on graphene oxide reinforced poly(N- isopropylacrylamide)/hydroxyethyl-chitosan. Mater. Today Commun. 2022, 31, 103697. [Google Scholar] [CrossRef]
- Liu, X.; Hao, M.; Chen, Z.; Zhang, T.; Huang, J.; Dai, J.; Zhang, Z. 3D bioprinted neural tissue constructs for spinal cord injury repair. Biomaterials 2021, 272, 120771. [Google Scholar] [CrossRef]
- Wang, W.; Chen, X.; Meng, T.; Liu, L. Multi-network granular hydrogel with enhanced strength for 3D bioprinting. J. Biomater. Appl. 2022, 36, 1852–1862. [Google Scholar] [CrossRef]
- Li, C.; Wang, K.; Zhou, X.; Li, T.; Xu, Y.; Qiang, L.; Peng, M.; Xu, Y.; Xie, L.; He, C.; et al. Controllable fabrication of hydroxybutyl chitosan/oxidized chondroitin sulfate hydrogels by 3D bioprinting technique for cartilage tissue engineering. Biomed. Mater. 2019, 14, 025006. [Google Scholar] [CrossRef]
- Turner, P.R.; Murray, E.; McAdam, C.J.; McConnell, M.A.; Cabral, J.D. Peptide Chitosan/Dextran Core/Shell Vascularized 3D Constructs for Wound Healing. ACS Appl. Mater. Interfaces 2020, 12, 32328–32339. [Google Scholar] [CrossRef]
- Groll, J.; Boland, T.; Blunk, T.; Burdick, J.A.; Cho, D.W.; Dalton, P.D.; Derby, B.; Forgacs, G.; Li, Q.; Mironov, V.A.; et al. Biofabrication: Reappraising the definition of an evolving field. Biofabrication 2016, 8, 013001. [Google Scholar] [CrossRef]
- Sadeghianmaryan, A.; Naghieh, S.; Alizadeh Sardroud, H.; Yazdanpanah, Z.; Afzal Soltani, Y.; Sernaglia, J.; Chen, X. Extrusion-based printing of chitosan scaffolds and their in vitro characterization for cartilage tissue engineering. Int. J. Biol. Macromol. 2020, 164, 3179–3192. [Google Scholar] [CrossRef]
- Bergonzi, C.; Bianchera, A.; Remaggi, G.; Ossiprandi, M.C.; Zimetti, F.; Marchi, C.; Bernini, F.; Bettini, R.; Elviri, L. Biocompatible 3d printed chitosan-based scaffolds containing α-tocopherol showing antioxidant and antimicrobial activity. Appl. Sci. 2021, 11, 7253. [Google Scholar] [CrossRef]
- Fischetti, T.; Celikkin, N.; Contessi Negrini, N.; Farè, S.; Swieszkowski, W. Tripolyphosphate-Crosslinked Chitosan/Gelatin Biocomposite Ink for 3D Printing of Uniaxial Scaffolds. Front. Bioeng. Biotechnol. 2020, 8, 400. [Google Scholar] [CrossRef]
- Wu, X.; Liu, S.; Chen, K.; Wang, F.; Feng, C.; Xu, L.; Zhang, D. 3D printed chitosan-gelatine hydrogel coating on titanium alloy surface as biological fixation interface of artificial joint prosthesis. Int. J. Biol. Macromol. 2021, 182, 669–679. [Google Scholar] [CrossRef]
- Zhang, J.; Allardyce, B.J.; Rajkhowa, R.; Kalita, S.; Dilley, R.J.; Wang, X.; Liu, X. Silk particles, microfibres and nanofibres: A comparative study of their functions in 3D printing hydrogel scaffolds. Mater. Sci. Eng. C 2019, 103, 109784. [Google Scholar] [CrossRef]
- Wu, C.; Yu, Z.; Li, Y.; Zhou, K.; Cao, C.; Zhang, P.; Li, W. Cryogenically printed flexible chitosan/bioglass scaffolds with stable and hierarchical porous structures for wound healing. Biomed. Mater. 2020, 16, 015004. [Google Scholar] [CrossRef]
- Mora-Boza, A.; Włodarczyk-Biegun, M.K.; Del Campo, A.; Vázquez-Lasa, B.; Román, J.S. Glycerylphytate as an ionic crosslinker for 3D printing of multi-layered scaffolds with improved shape fidelity and biological features. Biomater. Sci. 2020, 8, 506–516. [Google Scholar] [CrossRef]
- Suo, H.; Zhang, J.; Xu, M.; Wang, L. Low-temperature 3D printing of collagen and chitosan composite for tissue engineering. Mater. Sci. Eng. C 2021, 123, 111963. [Google Scholar] [CrossRef]
- Heidenreich, A.C.; Pérez-Recalde, M.; González Wusener, A.; Hermida, É.B. Collagen and chitosan blends for 3D bioprinting: A rheological and printability approach. Polym. Test. 2020, 82, 106297. [Google Scholar] [CrossRef]
- Vieira de Souza, T.; Malmonge, S.M.; Santos, A.R. Development of a chitosan and hyaluronic acid hydrogel with potential for bioprinting utilization: A preliminary study. J. Biomater. Appl. 2021, 36, 358–371. [Google Scholar] [CrossRef] [PubMed]
- Maiz-Fernández, S.; Barroso, N.; Pérez-Álvarez, L.; Silván, U.; Vilas-Vilela, J.L.; Lanceros-Mendez, S. 3D printable self-healing hyaluronic acid/chitosan polycomplex hydrogels with drug release capability. Int. J. Biol. Macromol. 2021, 188, 820–832. [Google Scholar] [CrossRef] [PubMed]
- Liu, Q.; Li, Q.; Xu, S.; Zheng, Q.; Cao, X. Preparation and properties of 3D printed alginate-chitosan polyion complex hydrogels for tissue engineering. Polymers 2018, 10, 664. [Google Scholar] [CrossRef]
- Osi, A.R.; Zhang, H.; Chen, J.; Zhou, Y.; Wang, R.; Fu, J.; Müller-Buschbaum, P.; Zhong, Q. Three-Dimensional-Printable Thermo/Photo-Cross-Linked Methacrylated Chitosan-Gelatin Hydrogel Composites for Tissue Engineering. ACS Appl. Mater. Interfaces 2021, 13, 22902–22913. [Google Scholar] [CrossRef]
- He, Y.; Wang, F.; Wang, X.; Zhang, J.; Wang, D.; Huang, X. A photocurable hybrid chitosan/acrylamide bioink for DLP based 3D bioprinting. Mater. Des. 2021, 202, 109588. [Google Scholar] [CrossRef]
- Lee, D.; Park, J.P.; Koh, M.-Y.; Kim, P.; Lee, J.; Shin, M.; Lee, H. Chitosan-catechol: A writable bioink under serum culture media. Biomater. Sci. 2018, 6, 1040–1047. [Google Scholar] [CrossRef]
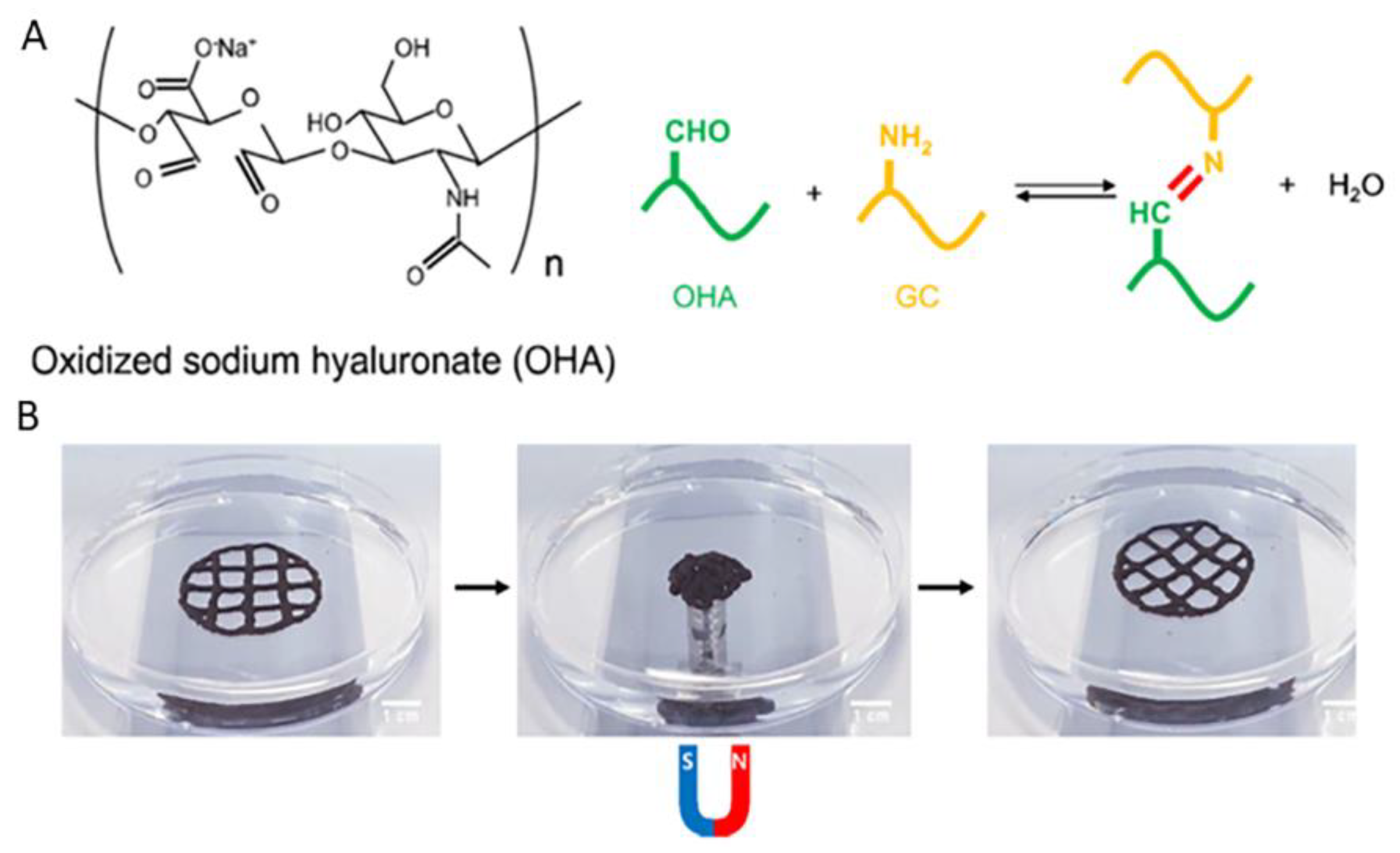
- Ko, E.S.; Kim, C.; Choi, Y.; Lee, K.Y. 3D printing of self-healing ferrogel prepared from glycol chitosan, oxidized hyaluronate, and iron oxide nanoparticles. Carbohydr. Polym. 2020, 245, 116496. [Google Scholar] [CrossRef]
- Michailidou, G.; Koukaras, E.N.; Bikiaris, D.N. Vanillin chitosan miscible hydrogel blends and their prospects for 3D printing biomedical applications. Int. J. Biol. Macromol. 2021, 192, 1266–1275. [Google Scholar] [CrossRef]
- Hidaka, M.; Kojima, M.; Nakahata, M.; Sakai, S. Visible light-curable chitosan ink for extrusion-based and vat polymerization-based 3d bioprintings. Polymers 2021, 13, 1382. [Google Scholar] [CrossRef]
- Xu, H.; Zhang, L.; Bao, Y.; Yan, X.; Yin, Y.; Li, Y.; Wang, X.; Huang, Z.; Xu, P. Preparation and characterization of injectable chitosan-hyaluronic acid hydrogels for nerve growth factor sustained release. J. Bioact. Compat. Polym. 2017, 32, 146–162. [Google Scholar] [CrossRef]
- Biranje, S.S.; Sun, J.; Cheng, L.; Cheng, Y.; Shi, Y.; Yu, S.; Jiao, H.; Zhang, M.; Lu, X.; Han, W.; et al. Development of Cellulose Nanofibril/Casein-Based 3D Composite Hemostasis Scaffold for Potential Wound-Healing Application. ACS Appl. Mater. Interfaces 2022, 14, 3792–3808. [Google Scholar] [CrossRef]
- Lin, H.Y.; Huang, H.Y.; Shiue, S.J.; Cheng, J.K. Osteogenic effects of inductive coupling magnetism from magnetic 3D printed hydrogel scaffold. J. Magn. Magn. Mater. 2020, 504, 166680. [Google Scholar] [CrossRef]
- Hafezi, F.; Scoutaris, N.; Douroumis, D.; Boateng, J. 3D printed chitosan dressing crosslinked with genipin for potential healing of chronic wounds. Int. J. Pharm. 2019, 560, 406–415. [Google Scholar] [CrossRef]
- Ergul, N.M.; Unal, S.; Kartal, I.; Kalkandelen, C.; Ekren, N.; Kilic, O.; Chi-Chang, L.; Gunduz, O. 3D printing of chitosan/poly(vinyl alcohol) hydrogel containing synthesized hydroxyapatite scaffolds for hard-tissue engineering. Polym. Test. 2019, 79, 106006. [Google Scholar] [CrossRef]
- Deng, N.; Sun, J.; Li, Y.; Chen, L.; Chen, C.; Wu, Y.; Wang, Z.; Li, L. Experimental study of rhBMP-2 chitosan nano-sustained release carrier-loaded PLGA/nHA scaffolds to construct mandibular tissue-engineered bone. Arch. Oral Biol. 2019, 102, 16–25. [Google Scholar] [CrossRef]
- Chen, S.; Shi, Y.; Zhang, X.; Ma, J. Evaluation of BMP-2 and VEGF loaded 3D printed hydroxyapatite composite scaffolds with enhanced osteogenic capacity in vitro and in vivo. Mater. Sci. Eng. C 2020, 112, 110893. [Google Scholar] [CrossRef]
- Marques, C.F.; Olhero, S.M.; Torres, P.M.C.; Abrantes, J.C.C.; Fateixa, S.; Nogueira, H.I.S.; Ribeiro, I.A.C.; Bettencourt, A.; Sousa, A.; Granja, P.L.; et al. Novel sintering-free scaffolds obtained by additive manufacturing for concurrent bone regeneration and drug delivery: Proof of concept. Mater. Sci. Eng. C 2019, 94, 426–436. [Google Scholar] [CrossRef]
- Naseri, E.; Cartmell, C.; Saab, M.; Kerr, R.G.; Ahmadi, A. Development of N,O-Carboxymethyl Chitosan-Starch Biomaterial Inks for 3D Printed Wound Dressing Applications. Macromol. Biosci. 2021, 21, 1–9. [Google Scholar] [CrossRef]
- He, Y.; Derakhshanfar, S.; Zhong, W.; Li, B.; Lu, F.; Xing, M.; Li, X. Characterization and Application of Carboxymethyl Chitosan-Based Bioink in Cartilage Tissue Engineering. J. Nanomater. 2020, 2020, 2057097. [Google Scholar] [CrossRef]
- Liu, I.H.; Chang, S.H.; Lin, H.Y. Chitosan-based hydrogel tissue scaffolds made by 3D plotting promotes osteoblast proliferation and mineralization. Biomed. Mater. 2015, 10, 035004. [Google Scholar] [CrossRef]
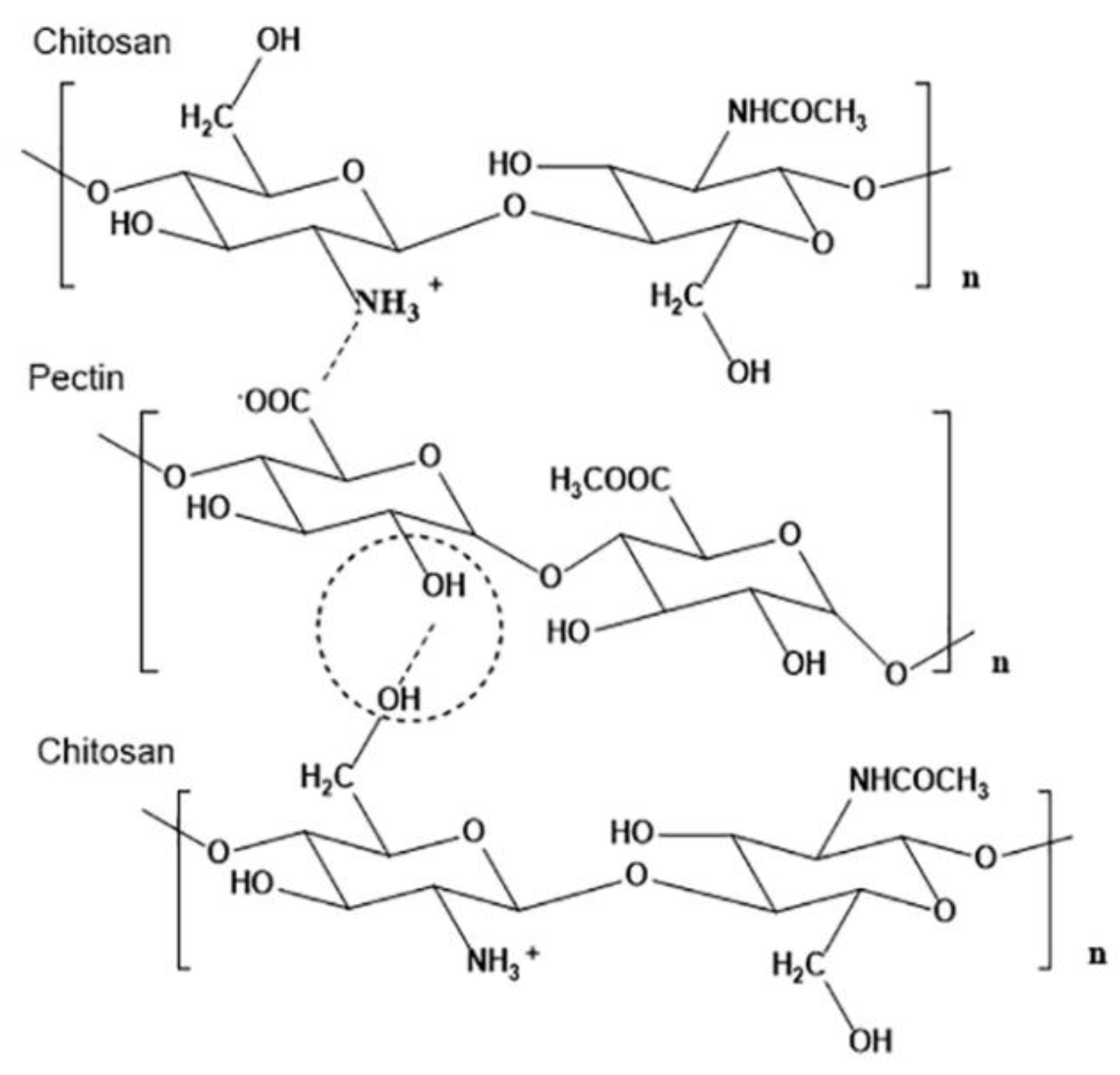
- Long, J.; Etxeberria, A.E.; Nand, A.V.; Bunt, C.R.; Ray, S.; Seyfoddin, A. A 3D printed chitosan-pectin hydrogel wound dressing for lidocaine hydrochloride delivery. Mater. Sci. Eng. C 2019, 104, 109873. [Google Scholar] [CrossRef]
- Andriotis, E.G.; Eleftheriadis, G.K.; Karavasili, C.; Fatouros, D.G. Development of bio-active patches based on Pectin for the treatment of Ulcers and wounds using 3D-bioprinting technology. Pharmaceutics 2020, 12, 56. [Google Scholar] [CrossRef] [Green Version]
- Martinotti, S.; Ranzato, E. Propolis: A new frontier for wound healing? Burn. Trauma 2015, 3, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Mamidi, N.; Velasco Delgadillo, R.M.; Barrera, E.V.; Ramakrishna, S.; Annabi, N. Carbonaceous nanomaterials incorporated biomaterials: The present and future of the flourishing field. Compos. Part B Eng. 2022, 243, 110150. [Google Scholar] [CrossRef]
- Cheng, Z.; Landish, B.; Chi, Z.; Nannan, C.; Jingyu, D.; Sen, L.; Xiangjin, L. 3D printing hydrogel with graphene oxide is functional in cartilage protection by influencing the signal pathway of Rank/Rankl/OPG. Mater. Sci. Eng. C 2018, 82, 244–252. [Google Scholar] [CrossRef] [PubMed]
- Diment, L.E.; Thompson, M.S.; Bergmann, J.H.M. Clinical efficacy and effectiveness of 3D printing: A systematic review. BMJ Open 2017, 7, e016891. [Google Scholar] [CrossRef]
- Trenfield, S.J.; Awad, A.; Goyanes, A.; Gaisford, S.; Basit, A.W. 3D Printing Pharmaceuticals: Drug Development to Frontline Care. Trends Pharmacol. Sci. 2018, 39, 440–451. [Google Scholar] [CrossRef]
- Hourd, P.; Medcalf, N.; Segal, J.; Williams, D.J. A 3D bioprinting exemplar of the consequences of the regulatory requirements on customized processes. Regen. Med. 2015, 10, 863–883. [Google Scholar] [CrossRef]
- Palo, M.; Holländer, J.; Suominen, J.; Yliruusi, J.; Sandler, N. 3D printed drug delivery devices: Perspectives and technical challenges. Expert Rev. Med. Devices 2017, 14, 685–696. [Google Scholar] [CrossRef]
- Mohiuddin, A.K. Extemporaneous compounding: Cautions, controversies and convenience. IP Int. J. Compr. Adv. Pharmacol. 2020, 3, 124–137. [Google Scholar] [CrossRef]
- Pavan Kalyan, B.; Kumar, L. 3D Printing: Applications in Tissue Engineering, Medical Devices, and Drug Delivery. AAPS PharmSciTech 2022, 23, 1–20. [Google Scholar] [CrossRef]
- Bom, S.; Martins, A.M.; Ribeiro, H.M.; Marto, J. Diving into 3D (bio)printing: A revolutionary tool to customize the production of drug and cell-based systems for skin delivery. Int. J. Pharm. 2021, 605, 120794. [Google Scholar] [CrossRef]
- Singh, S.; Choudhury, D.; Yu, F.; Mironov, V.; Naing, M.W. In situ bioprinting–Bioprinting from benchside to bedside? Acta Biomater. 2020, 101, 14–25. [Google Scholar] [CrossRef]
- Ashammakhi, N.; Ahadian, S.; Zengjie, F.; Suthiwanich, K.; Lorestani, F.; Orive, G.; Ostrovidov, S.; Khademhosseini, A. Advances and Future Perspectives in 4D Bioprinting. Biotechnol. J. 2018, 13, 1800148. [Google Scholar] [CrossRef]

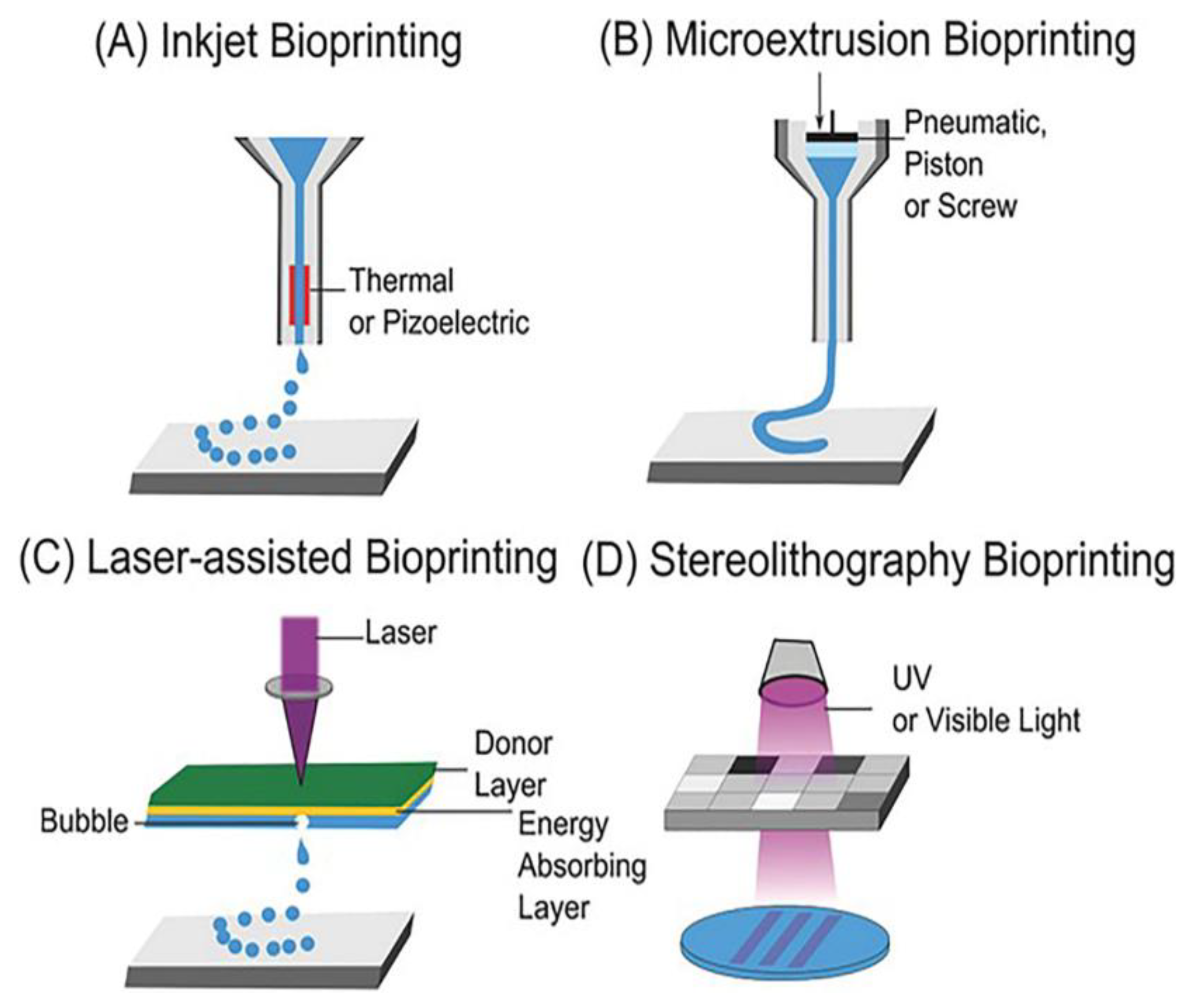

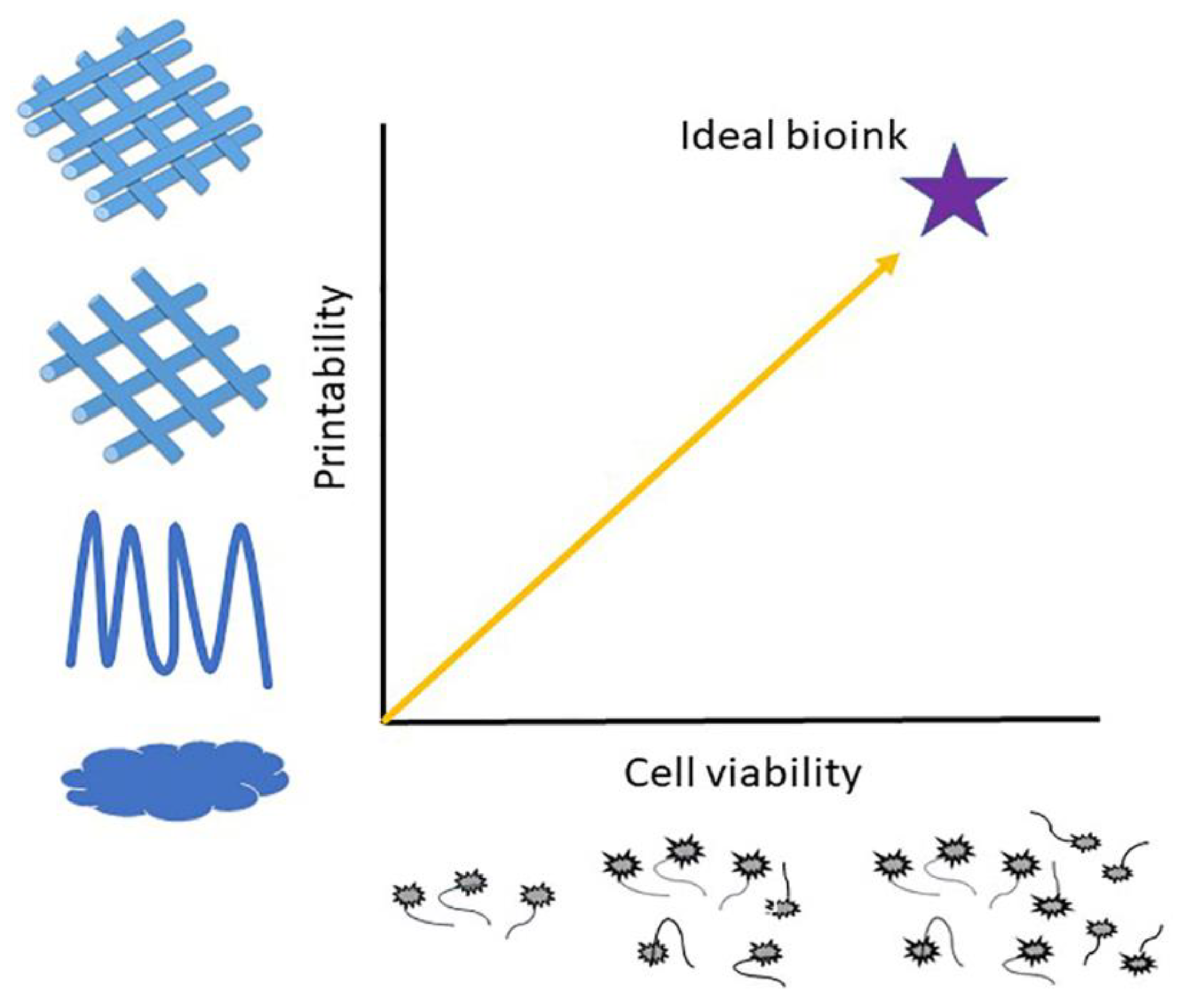




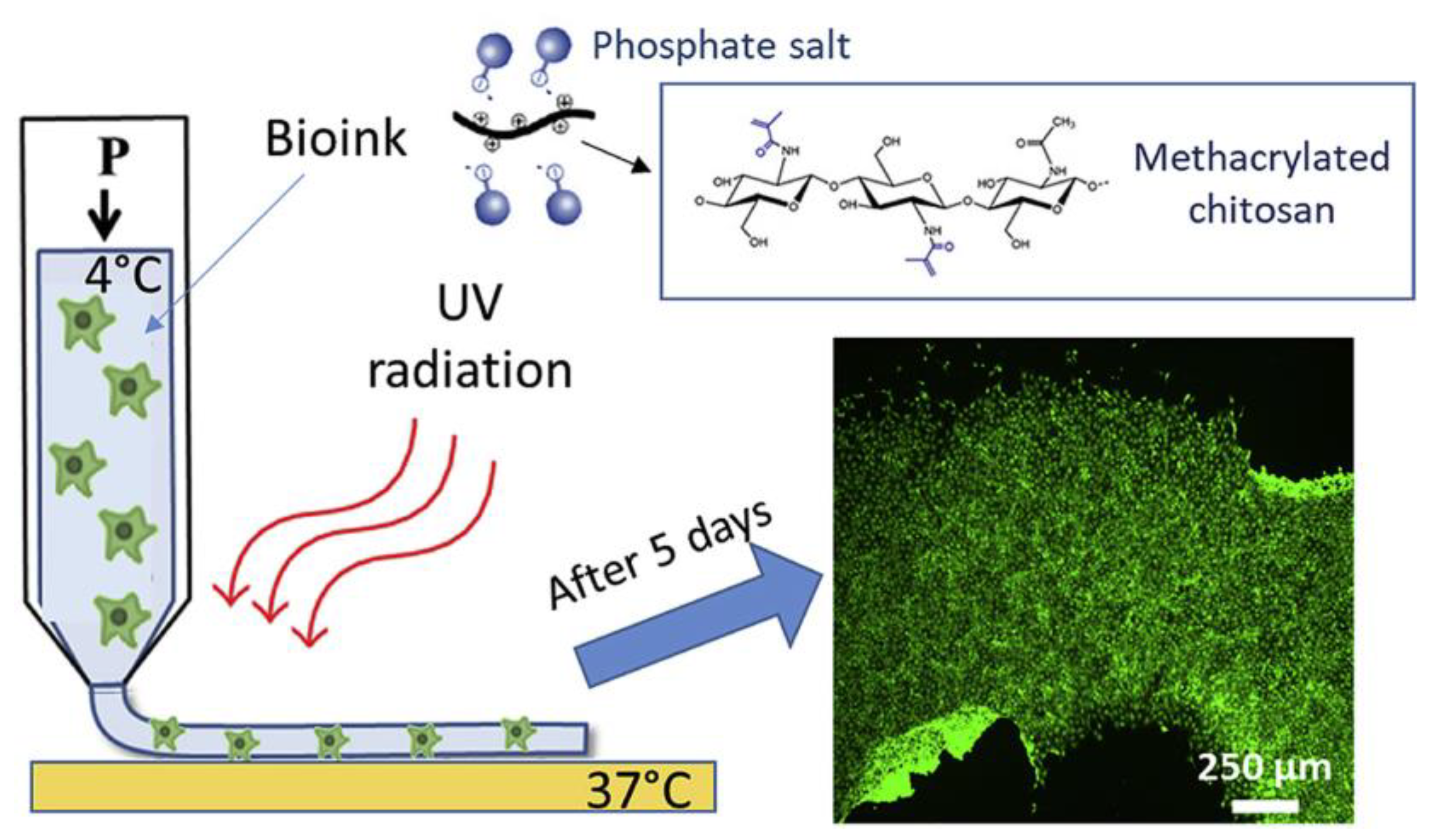

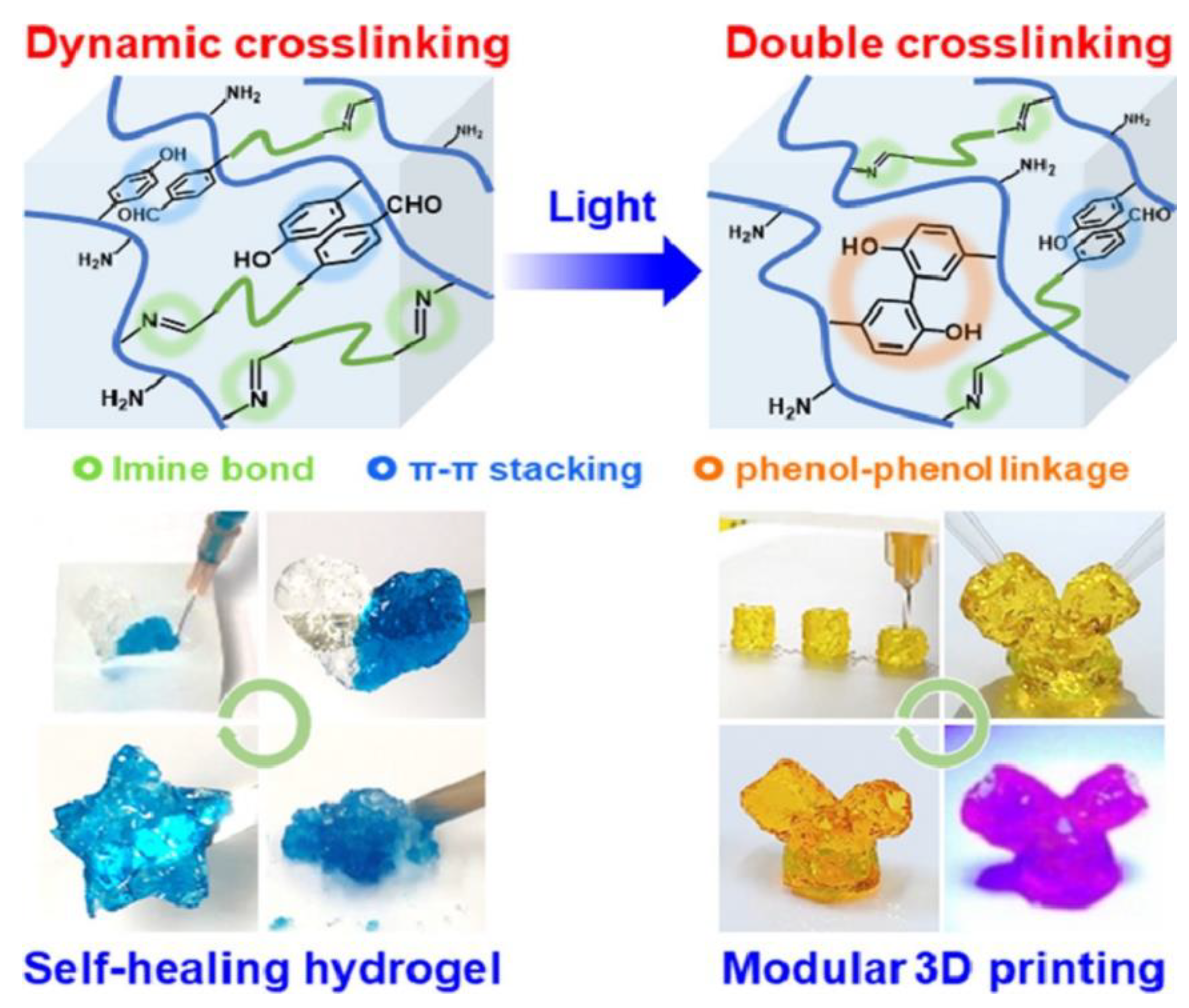
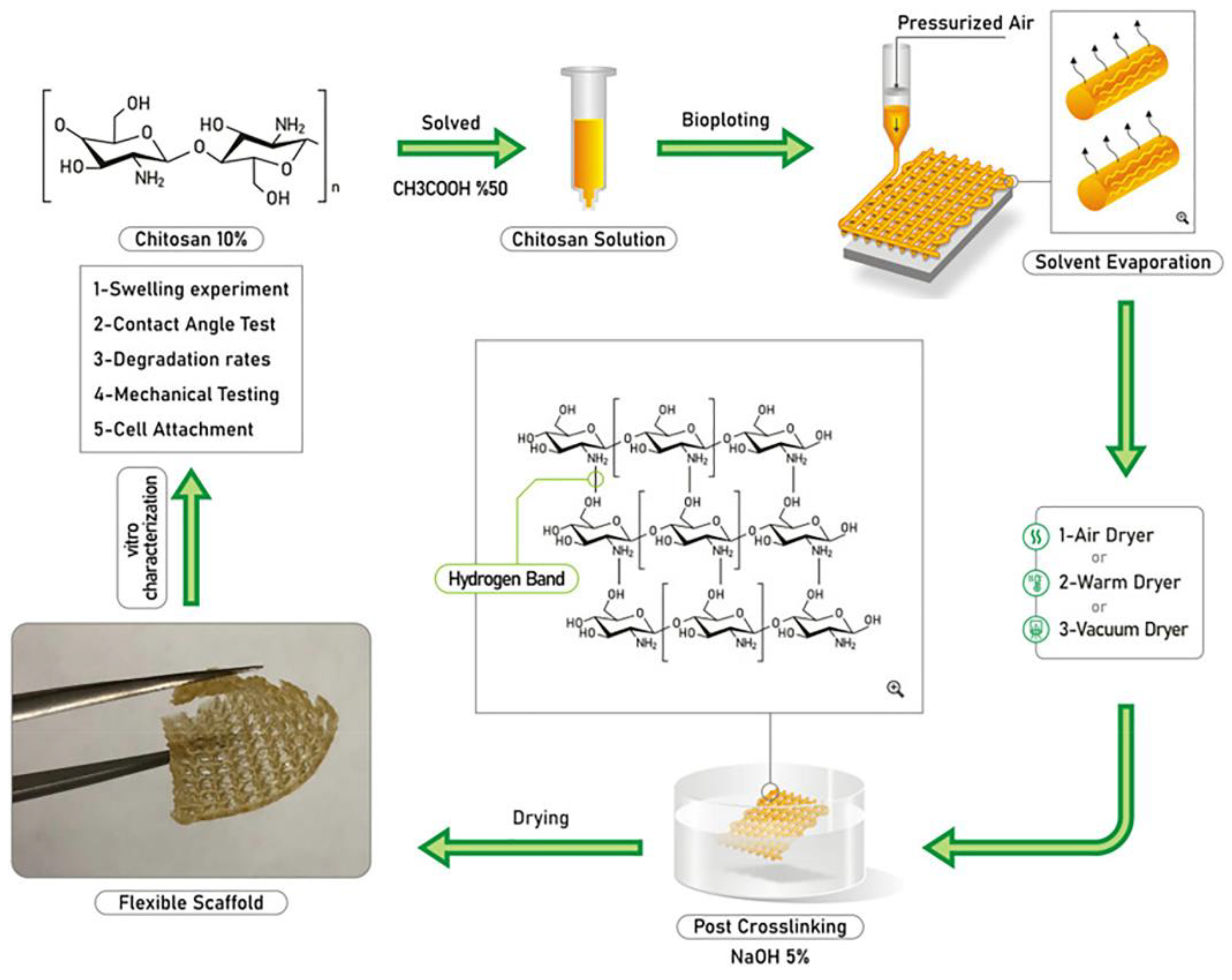







| Sample | Animal Model | Days of Testing | Application |
|---|---|---|---|
| CS-PCL | Rabbits (n = 8 knees for each category) | Test of mechanical properties 3 and 6 months after surgery | Cartilage regeneration of articular cartilage |
| CS-D-(+) raffinose | Adult female Wistar rats | After 7, 10 and 14 days the animals were sacrificed | Wound healing evaluation in experimental diabetes in rats |
| CMC-amorphous calcium phosphate | Athymic nude mice (n = 5) | After 4, 6 and 8 weeks after implantation the animals were sacrificed | Bone regeneration |
| PNiPAM/HECS/GO | Sprague–Dawley (SD) rats | After 1, 7, 14 and 28 days the animals were sacrificed | General drug delivery and tissue engineering applications |
| HBC/HA/MA | Sprague-Dawley (SD) rats (n = 8) | Test of movement ability after week 12 post surgery | Spinal cord injury repair |
| Other Compounds | Cell Type | Bioink Formulation | Bioprinting Method | Condition | Application | Ref. | |
|---|---|---|---|---|---|---|---|
| CS | PCL | Synovial mesenchymal stem cells (SMSCs) (3 × 107 cells/mL) | CS: 2% w/v), β-glycerophosphate: 56% w/v Crosslinker: genipin: 0.5% w/v | Extrusion based | Nozzle: 26 G Temperature: 37 °C | Cartilage regeneration | [15] |
| CS | PCL | Human periodontal stem cells (PDLSCs) (5 × 105 cells/mL) | β-glycerophosphate: 56% (w/v), Crosslinker: sodium bicarbonate: 3.5% | Extrusion based | Temperature: framework printing: 65 °C, cell-laden CS solution: 37 °C | - | [17] |
| CS | D-(+)raffinose | Human fibroblasts (Nhdf) and Keratinocytes (HaCaT) (1 × 105 cells/mL) | CS: 6% w/v, D-(+) raffinose pentahydrate: (290 mM) Crosslinker: KOH (8% w/v) | Extrusion based | Nozzle: 26 G Temperature: 25 °C | Diabetic wound healing | [16] |
| CS | Poly(gamma-glutamic acid) | Human adult fibroblasts (2 × 105 cells/mL) | CS: 4% or 6% w/v PGA: 2% w/v | Extrusion based | Nozzle: 22 G for chitosan and 25 G for Gamma-PGA Pressure: CS: 25–40 kPa Gamma-PGA: 5–10 kPa Temperature: 37 °C Speed: 600 mm/min | - | [18] |
| CS | Cellulose nanocrystals, Hydroxyethyl cellulose | Pre-osteoblast cells (MC3T3-E1) (5 × 106 cells/mL) | CS: 3% w/v CNCs: 0–2% w/v HEC: 0–0.5 mg/mL Crosslinker: β-GP: 100 mM | Extrusion based | Nozzle: 20 G Pressure: 20 kPa Temperature: room temperature Speed: 2 mm/s | Bone tissue engineering | [19] |
| CS | Gelatin, 2GP | IMR-32 cells from neuroblastoma (CCL-127) (2 × 106 cells/mL) | Chitosan: 2% w/v Gelatin: 4% w/v 2GP: 0.56% w/v Crosslinker: genipin | Extrusion based | Temperature: 37 °C | - | [20] |
| CS | Gelatin | Human bone marrow mesenchymal stems cells (MSCs) (5 × 106 cells/mL) | Chitosan: 2% or 3% w/v Gelatin: 2% w/v Crosslinker: β-glycerophosphate/sodium bicarbonate: 0.1 M/0.075 M | Extrusion based | Nozzle: 25 G Temperature: printhead: room temperature, printbed: 37 °C Speed: 3–11 mm/s | - | [21] |
| CS | Gelatin, PEG | Human dermal fibroblasts(HDF) and Keratinocytes (KC) (0.5 × 106 cells/mL) | Chitosan: 0.5% w/v Gelatin: 1% w/v PEG: 2% w/v Crosslinker: genipin 1% w/v | Extrusion based | Nozzle: 22 G Presure: 25 kPa Speed: 4 mm/s | Skin regeneration | [22] |
| CS Derivative | Other Compounds | Cell Type | Bioink Formulation | Bioprinting Method | Condition | Application | Ref. |
|---|---|---|---|---|---|---|---|
| MeGC | - | Human osteosarcoma cell line (1 × 106 cells/mL) | MeGC: 3% w/v, riboflavin: 12 μΜ Crosslinker: UV for 70 s | Extrusion based | Nozzle: 26 G Pressure: 120 ± 10 kPa Temperature: 25 °C Speed: 6 mm/s | Bone tissue engineering | [23] |
| CS-MA | β-glycerophosphate, lithium phenyl-2,4,6-trimethylbenzoylphospinate | Fibroblasts (NIH/3T3) (106 cells/mL), osteoblast-like cells (Saos-2) and neuronal-like cells (SH-SY5Y) | CS-MA: 1.5 % w/v, βGP:0.5 g/mL, LAP: 0.05% w/v Crosslinker: UV | Extrusion based | Nozzle: 25 G Temperature: Printhead: 4 °C Printing bed: 37 °C Speed: 10 mm/s | - | [24] |
| CS-MA | Phosphorylated-oligo [poly(ethylene glycol)fumarate], Acrylated montmorillonite, Methyl cellulose, Gelatin | Pre-osteoblast cells (MC3T3-E1) (1.5 × 106 cells/mL) | MEGC: 2% w/v MMT: 0%, 1%, 4%, 6% Irgacure 2959 1.5 wt.% of total polymer content Crosslinker: UV | Extrusion based | Nozzle: 25 G Pressure: 50 kPa Temperature: 25 °C printing bed: 4 °C Speed:10 mm/s | Bone tissue engineering | [25] |
| GC | Oxidized hyaluronate, Adipic acid dihydrazide, Alginate | ATDC5 cells (1 × 107 cells/mL) | GC: 1 wt.% OHA: 2 wt.% ADH: 0.3% ALG 0.3 wt.% Crosslinker: CaCl2 60 mM | Extrusion based | Nozzle: 25 G Pressure: 300 N Temperature: 25 °C | - | [27] |
| NOCC | Agarose | Neuro2a mouse Neuroblastoma Cells (1 × 105 cells/mL) | Agarose: 40% NOCC: 60% | Extrusion based | Nozzle: 0.41 mm Temperature: 37 °C Speed: 3 mm/s | - | [28] |
| NOCC | Gelatin, Na-Polyphosphate NPs, Alginate | Mesenchymal stem cells (MSC) (3 × 105 cells/mL) | NOCC: 50 mg/mL Gelatin: 25 mg/mL Na-polyP: 100 μg/mL Alginate: 1% Crosslinker: CaCl2 2.5% w/v | Extrusion based | Temperature: 21 °C Pressure: 1 bar Temperature: room temperature Speed: 18 mm/s | - | [29] |
| CMC | Aldehyde hyaluronic acid, Gelatin, 4-arm poly(ethylene glycol) succinimidyl glutarate | NIH/3T3 fibroblasts CRL1658 and C2C12 myoblasts | AHA: 1% w/v CMC: 0.75% w/v GEL: 1% w/v PEG-SG: 0.5% w/v | Extrusion based | Nozzle: 25 G Temperature: 10 °C Speed: linear: 6 mm/s extrusion: 0.07 mL/min | - | [30] |
| CMC | Methacryloyl gelatin | Bone marrow mesenchymal stem cells (BMSCs) (3 × 105 cells/mL) | CMC: 4% w/v LAP: 0.3% w/v 10% GelMA/0.5% CMCS 10% GelMA/1% CMCS 10% GelMA/2% CMCS | Extrusion based | Nozzle: 160 μm Pressure: non cellular inks: 0.8–1.2 bar cell-laden: 0.5–1 bar Speed: Cell-laden: 10–20 mm/s Cell-free: 15 mm/s Temperature: 16 °C | Vascular tissue engineering | [31] |
| CMC | Oxidized and non-oxidized hyaluronic acid | Fibroblasts (L929) (1 × 106 cell/mL) | CMC: 2 wt.% HA: 0.4 wt.% Oxidized HA: 4 wt.% Catechol NPs: 2 and 29 M % Crosslinker: FeCl3 20 mM | Extrusion based | Nozzle: 200 μm Speed: plunger: 0.06 mm/s, printhead: 15 mm/s | - | [32] |
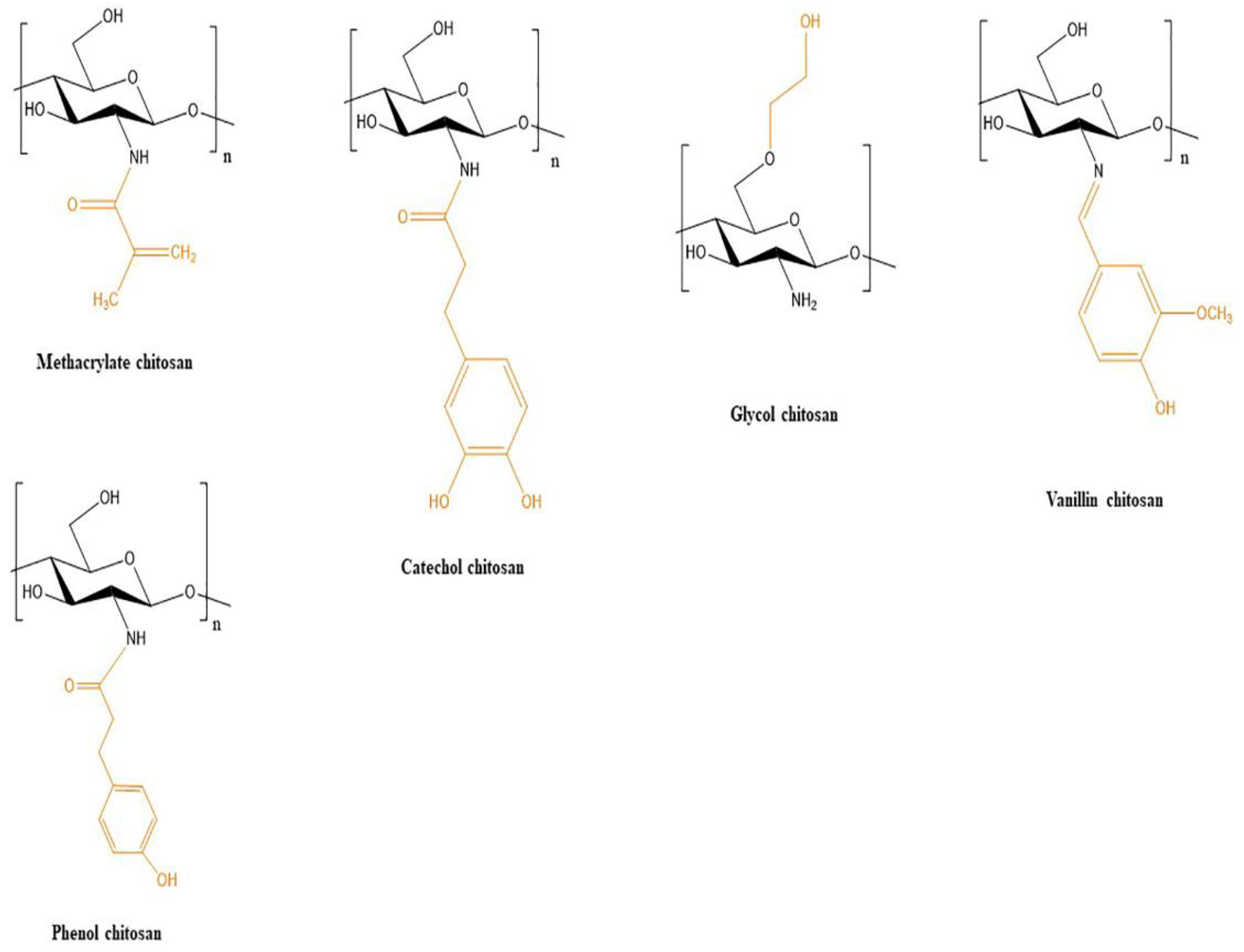
| CS-Ph | Dibenzaldehyde- terminated telechelic poly(ethylene glycol) | Human mesenchymal stem cells (hMSCs) (4 × 106 cells/mL) | Chi-Ph: 1% Crosslinker:DF-PEG 1%, UV (440–460 nm) | Extrusion based | Nozzle: 26 G | Bone tissue engineering | [34] |
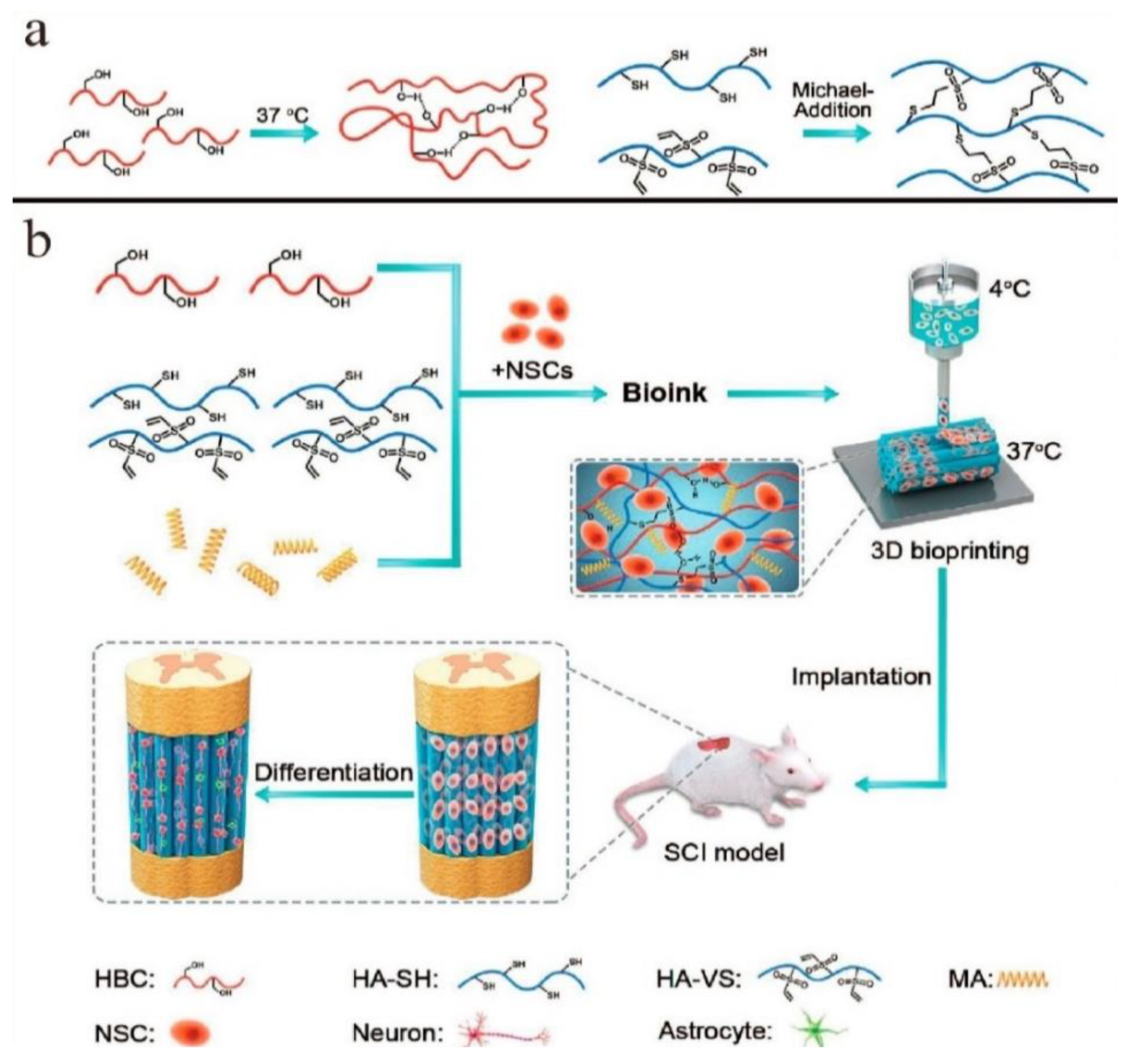
| HBC | Thiolated hyaluronic acid, Vinyl sulfonated hyaluronic acid, Matrigel | Neural stem cells (NSCs) | HBC: 3% w/v, HA-SH: 0.3% w/v, HA-VS: 0.3% w/v, MA: 0.1% w/v | Extrusion based | Nozzle: 260 μm Temperature: printhead: 37 °C printbed: 10 °C, Pressure: 20 kPa | Neural tissue engineering | [36] |
| HPC microspheres | Methacryloyl gelatin, poly(γ-glutamic acid) | Adiposed derived stem cells (ASCs) | GelMA: 5 wt.%, HPC-microspheres:50 mg/mL, PG-microspheres: 50 mg/mL, I2959 Crosslinker: UV | Extrusion based | Nozzle: 18 G Temperature: 25 °C Speed: 5 mm/s | - | [37] |
| HBC | Oxidized chondroitin sulfate | Bone-marrow mesenchymal stem cells (BMSCs) (5 × 106 cells/mL) | GelMA: 13% w/v, Irgacure 2959: 0.2% w/v, Peptide/CD gel concentration: 0.02%/8% SC/0.6% DA, Crosslinker: UV | Extrusion based | Nozzle: core: 400 μm, shell: 800 μm, temperature: printhead: 25 °C, printbed:16 °C, Pressure: core: 225 kPa, shell: 150 kPa, Speed: 5 mm/s | Wound healing | [38] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lazaridou, M.; Bikiaris, D.N.; Lamprou, D.A. 3D Bioprinted Chitosan-Based Hydrogel Scaffolds in Tissue Engineering and Localised Drug Delivery. Pharmaceutics 2022, 14, 1978. https://doi.org/10.3390/pharmaceutics14091978
Lazaridou M, Bikiaris DN, Lamprou DA. 3D Bioprinted Chitosan-Based Hydrogel Scaffolds in Tissue Engineering and Localised Drug Delivery. Pharmaceutics. 2022; 14(9):1978. https://doi.org/10.3390/pharmaceutics14091978
Chicago/Turabian StyleLazaridou, Maria, Dimitrios N. Bikiaris, and Dimitrios A. Lamprou. 2022. "3D Bioprinted Chitosan-Based Hydrogel Scaffolds in Tissue Engineering and Localised Drug Delivery" Pharmaceutics 14, no. 9: 1978. https://doi.org/10.3390/pharmaceutics14091978







