Abstract
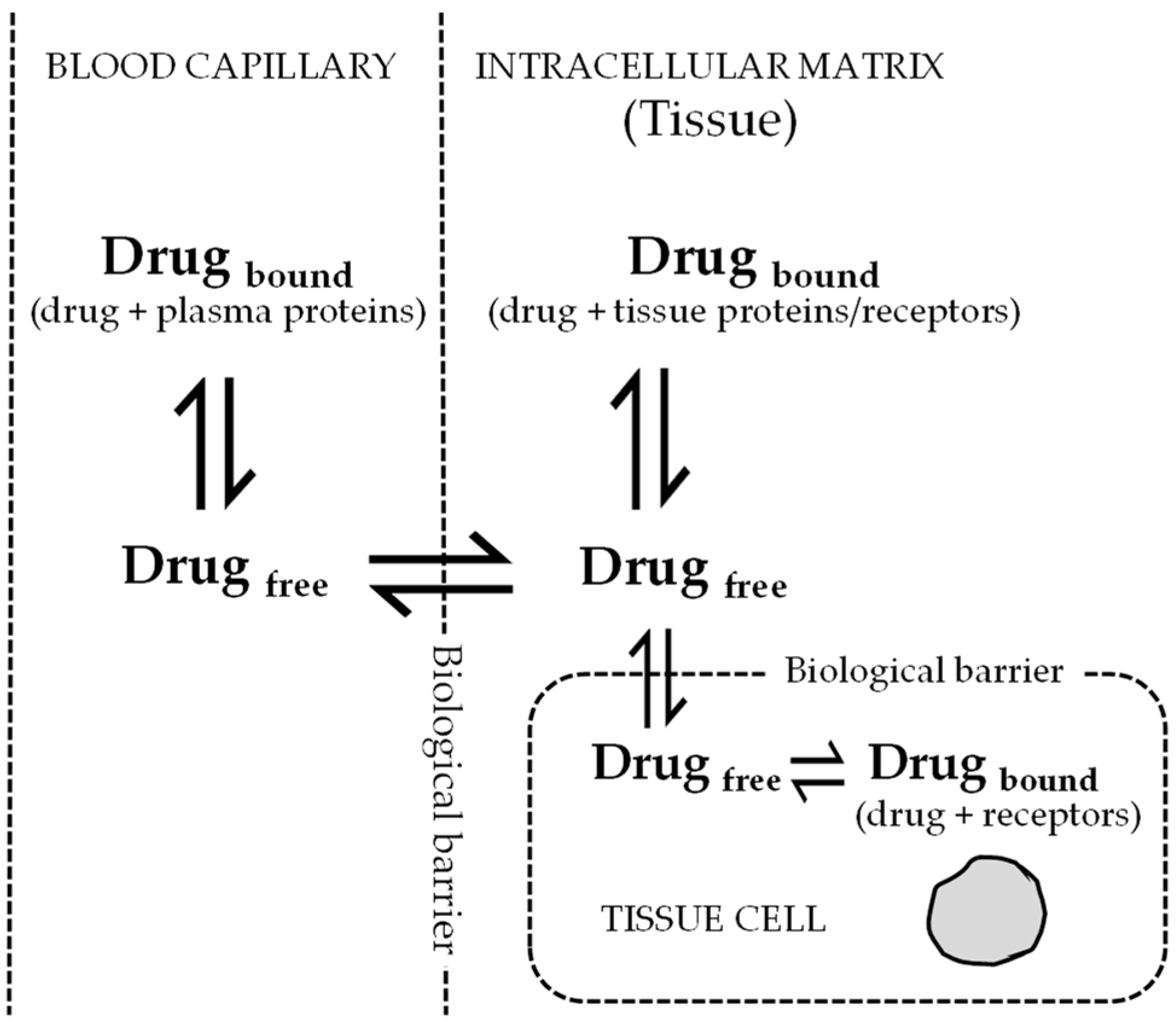
The search for new drugs is an extremely time-consuming and expensive endeavour. Much of that time and money go into generating predictive human pharmacokinetic profiles from preclinical efficacy and safety animal data. These pharmacokinetic profiles are used to prioritize or minimize the attrition at later stages of the drug discovery process. In the area of antiviral drug research, these pharmacokinetic profiles are equally important for the optimization, estimation of half-life, determination of effective dose, and dosing regimen, in humans. In this article we have highlighted three important aspects of these profiles. First, the impact of plasma protein binding on two primary pharmacokinetic parameters—volume of distribution and clearance. Second, interdependence of primary parameters on unbound fraction of the drug. Third, the ability to extrapolate human pharmacokinetic parameters and concentration time profiles from animal profiles.
1. Introduction
Now more than ever, the emergence of deadly viruses and viral pandemics in the last several decades has necessitated the demand for antiviral drug discovery. The discovery and development of new antiviral drugs is one of the most complicated and time-consuming processes which requires a considerable amount of energy and resources. The initial, essential qualification of new chemical entities is in vitro potency. However, the clinical translation with effective efficacy is always challenging due to unfavourable pharmacokinetic (PK) properties and pharmacokinetic-pharmacodynamic (PK-PD) correlation at the pharmacological dose. The process of antiviral drug discovery is summarized graphically in Figure 1. Majority of lead molecules often fail to show the desired efficacy or acceptable safety profiles. The high attrition rates also add to significant increases in resources. Nearly 50% of all drug candidates fail as a consequence of insufficient efficacy [1]—the result of a singular reason, or a combination of several. Nevertheless, the consideration of solubility-permeability relationship helps in the selection of potential oral candidates with desired intestinal absorption. The understanding of absorption, distribution, metabolism, elimination/toxicity (ADME/tox), PK, and calculative prediction of clinical dose at preclinical discovery stage, has ensured the entry of the best antiviral drug candidates into the development phase with optimal ADME properties. Accordingly, more and more attention is drawn toward two main aspects: (i) understanding the correlation and consideration of observed differences in PD/efficacy/safety profiles with PK profiles between animals and humans; and (ii) prediction of human pharmacokinetics at discovery stage and assistance in dialling out potential liabilities in the clinical candidates at an early stage of drug development [2]. The development of oral antiviral drugs is far more complicated and time-consuming compared to parenteral drugs.
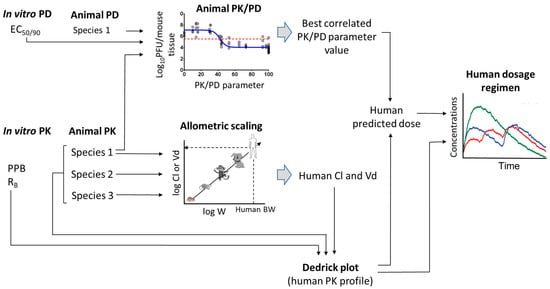
Figure 1.
Graphical representation showing critical pathway from in vitro potency test to human dose prediction in discovery process.
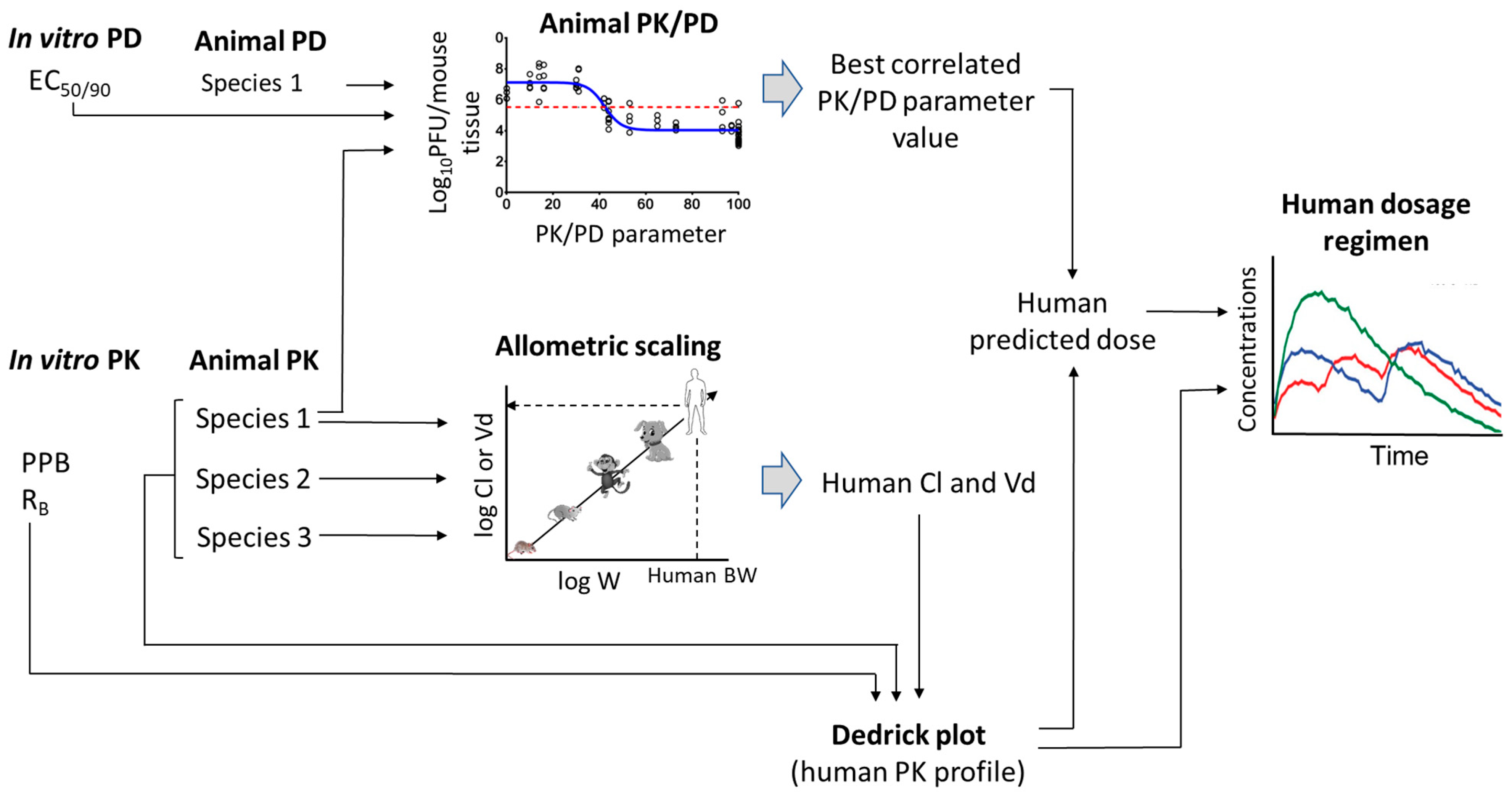
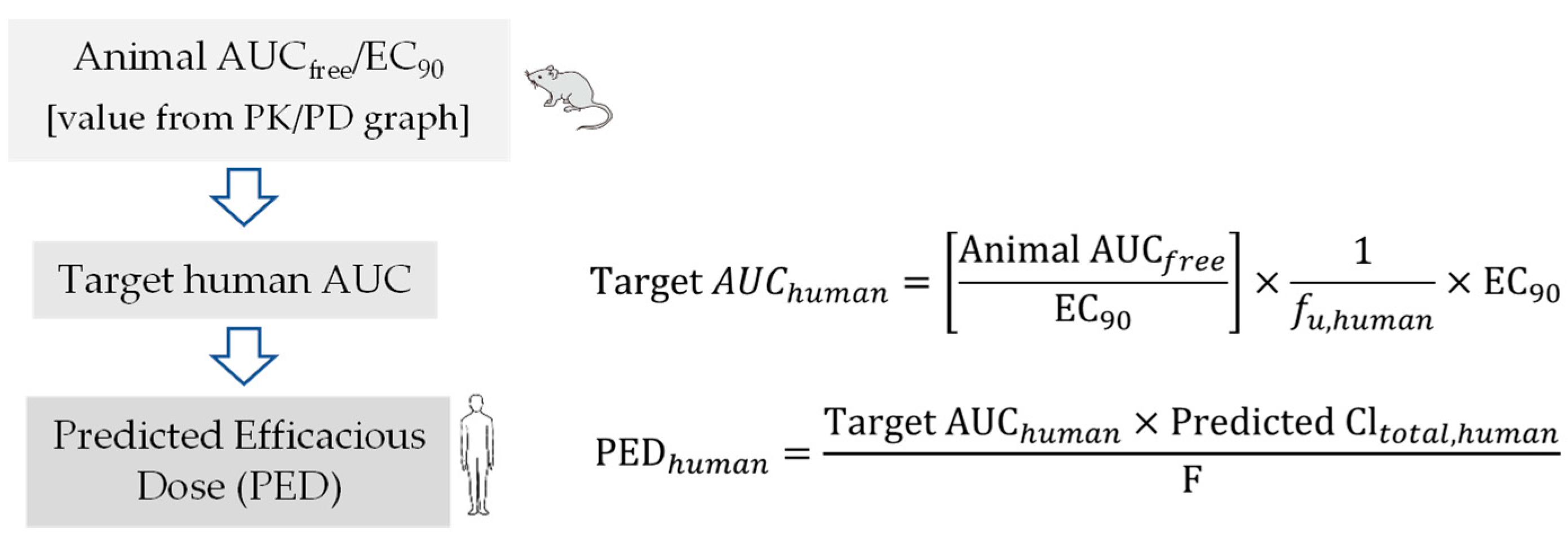
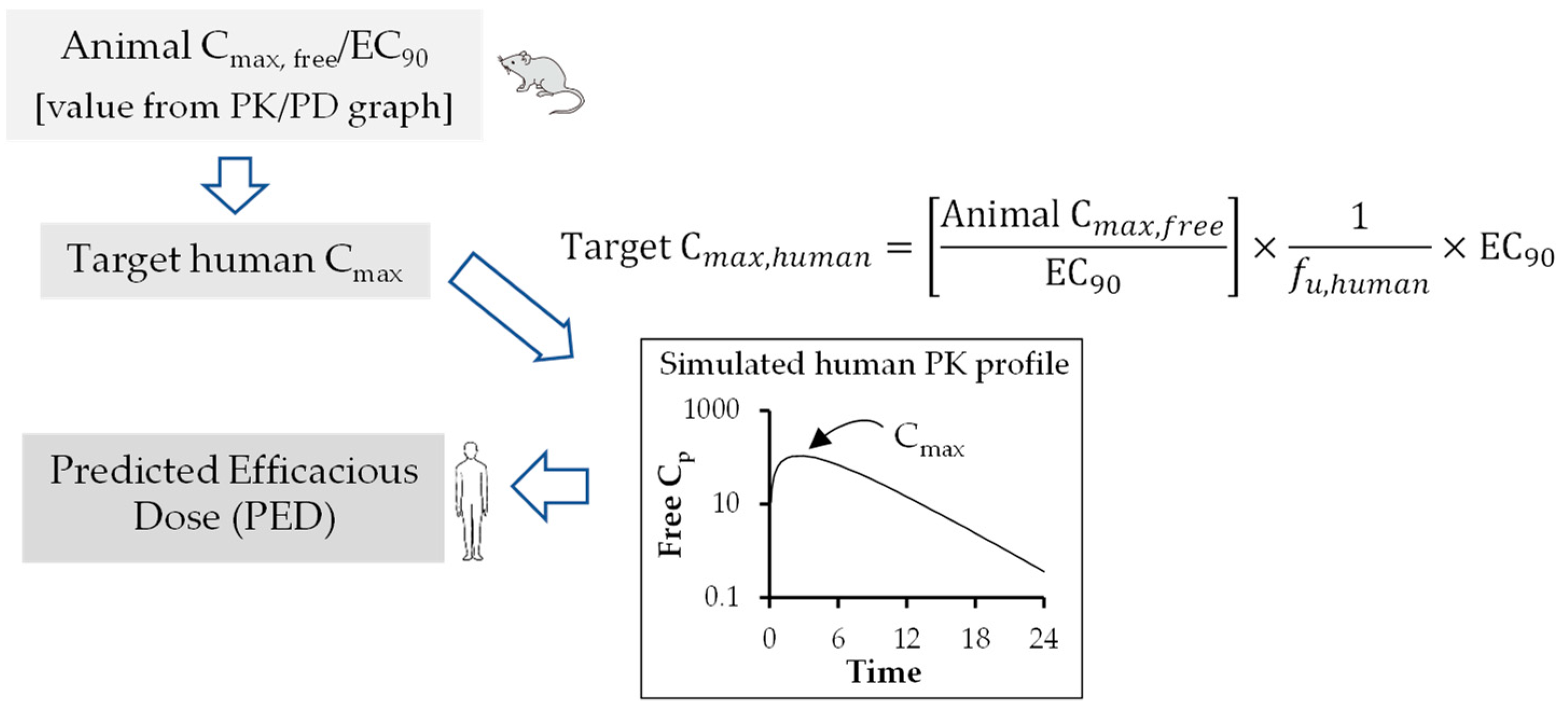
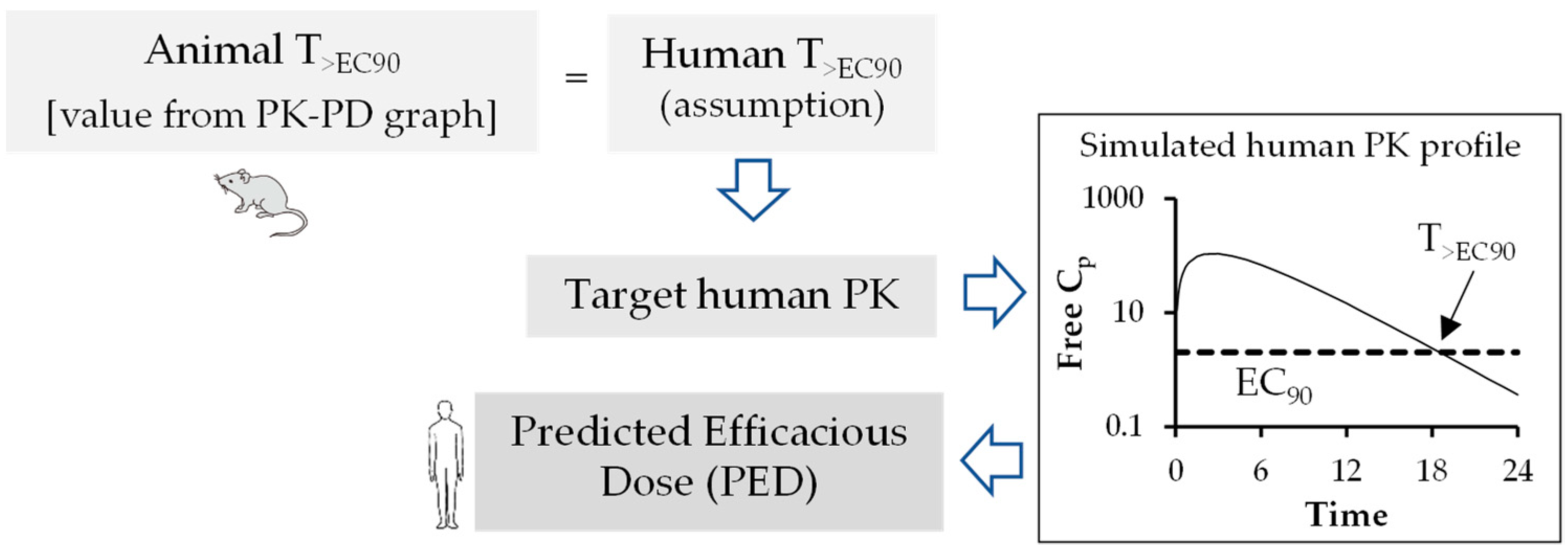
The emergence of drug resistance development has outpaced the discovery of new molecules, thus generating a major cause for concern. One significant reason for suboptimal antiviral response could be the result of inadequate exposure and/or poor PK-PD properties of the investigational drug. Maintenance of sufficient plasma exposure within the therapeutic window is one of the most critical requirements to stop viral replication and the emergence of resistance [3]. It has been demonstrated that setting up the dosage regimen based on the PK-PD relationship of antivirals has increased the probability of successful treatment outcome and reduced emergence of resistance [4]. Therefore, the objectives of this paper are: (i) to highlight the importance of plasma protein binding, volume of distribution, and clearance in the antiviral drug discovery process; (ii) to estimate primary PK parameters in humans, such as volume of distribution and clearance from animal data using allometric scaling; (iii) the prediction of human pharmacokinetic profile; and (iv) estimation of the therapeutic dosage and dosage regimen of antiviral drugs using the PK-PD relationship by applying sound mathematical models.
7. Conclusions
In summary, during the preclinical phase of the drug discovery process, prior information on human pharmacokinetic parameters such as Cl, Vd, and t1/2, provided tremendous value in the compound selection process and simulation of human pharmacokinetic profile from predicted human pharmacokinetic parameters for prediction of dosage regimen. Therefore, in recent years, interspecies scaling of pharmacokinetic parameters and prediction of human pharmacokinetic profile using PK-PD analysis have drawn enormous attention in antiviral drug discovery. However, more robust pharmacodynamic data in the form of reduction in PFUs are to be generated in cell culture and animal models to best correlate the PK-PD parameters. This will help overcome the challenges associated with PK-PD correlations. Two important pharmacokinetic parameters, such as clearance and volume of distribution, are required to simulate time course of drug profiles in humans and to predict human t1/2, a parameter that is better understood by non-pharmacokineticist colleagues in the drug discovery and development field. Estimation of human half-life using predicted human clearance and volume of distribution generally provides more acceptable results in predicting t1/2 rather than direct correlation of animal and human t1/2 values [58]. Over the years, multiple approaches have been suggested to improve the predictive performance of time course profiles, however, there is no method without shortcomings [59]. Hence, consideration of a particular extrapolation method should be conducted based upon physicochemical properties of drugs such as renal secretion or biliary excretion. To improve predictive outcome, careful attention should be given to experimental design, choice of in vivo study species, and analytical errors. In addition, all these factors may have an impact on allometric extrapolation. The animal scaling method for prediction of time course of drug profiles using complex Dedrick plot and human therapeutic dose using various rational PK-PD models (AUC/EC50/90, Cmax/EC50/90, and T>EC50/90) mentioned in this article are simplified, reliable, and a relatively less time-consuming method for extrapolation of preclinical data to humans.
Author Contributions
Conceptualization, T.K.B. and T.C.; writing—review and editing, T.K.B., T.C. and C.S.; supervision, T.K.B. and T.C. All authors have read and agreed to the published version of the manuscript.
Funding
This research received no external funding.
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
Not applicable.
Acknowledgments
We thankfully acknowledge Lloyd, Nicole for English language editing of this manuscript.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Kennedy, T. Managing the drug discovery/development interface. Drug Discov. Today 1997, 2, 436–444. [Google Scholar] [CrossRef]
- Prueksaritanont, T.; Tang, C. ADME of Biologics—What Have We Learned from Small Molecules? AAPS J. 2012, 14, 410–419. [Google Scholar] [CrossRef] [PubMed]
- Gonzalez de Requena, D.; Bonora, S.; Garazzino, S.; Sciandra, M.; D’Avolio, A.; Raiteri, R.; Marrone, R.; Boffito, M.; De Rosa, F.G.; Sinicco, A.; et al. Nevirapine plasma exposure affects both durability of viral suppression and selection of nevirapine primary resistance mutations in a clinical setting. Antimicrob. Agents Chemother. 2005, 49, 3966–3969. [Google Scholar] [CrossRef] [PubMed]
- Boffito, M.; Back, D.; Stainsby-Tron, M.; Hill, A.; Di Perri, G.; Moyle, G.; Nelson, M.; Tomkins, J.; Gazzard, B.; Pozniak, A. Pharmacokinetics of saquinavir hard gel/ritonavir (1000/100 mg twice daily) when administered with tenofovir diproxil fumarate in HIV-1-infected subjects. Br. J. Clin. Pharm. 2005, 59, 38–42. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Avery, L.B.; Zarr, M.A.; Bakshi, R.P.; Siliciano, R.F.; Hendrix, C.W. Increasing Extracellular Protein Concentration Reduces Intracellular Antiretroviral Drug Concentration and Antiviral Effect. Aids Res. Hum. Retrovir. 2013, 29, 1434–1442. [Google Scholar] [CrossRef]
- Fischl, M.A.; Richman, D.D.; Flexner, C.; Fischl, M.A.; Richman, D.D.; Flexner, C.; Para, M.F.; Haubrich, R.; Karim, A.; Yeramian, P.; et al. Phase I/II study of the toxicity, pharmacokinetics, and activity of the HIV protease inhibitor SC-52151. J. Acquir. Immune Defic. Syndr. 1997, 15, 28–34. [Google Scholar] [CrossRef]
- Keogh, J.H.; Rynn, B.C.; Bruno Stieger, B.; Nicholls, G. Membrane Transporters: Fundamentals, Function and Their Role in ADME and Drug Development; Royal Society of Chemistry: London, UK, 2016; Volume 1. [Google Scholar]
- Suzuki, H.; Furukawa, H.; Miyazaki, H.; Hashimoto, M. Absorption, distribution, excretion and metabolism of SC-11800EE, a combined steroid preparation of SC-11800 (ethynodiol diacetate) and ethinyl estradiol in rats and mice. Radioisotopes 1977, 26, 152–157. [Google Scholar] [CrossRef][Green Version]
- Bohnert, T.; Gan, L.S. Plasma protein binding: From discovery to development. J. Pharm. Sci. 2013, 102, 2953–2994. [Google Scholar] [CrossRef]
- Li, P.; Fan, Y.; Wang, Y.; Lu, Y.; Yin, Z. Characterization of plasma protein binding dissociation with online SPE-HPLC. Sci. Rep. 2015, 5, 14866. [Google Scholar] [CrossRef]
- Anusha, G.B.; Ahad, H.A.; Haranath, C.; Suryaprakash, R.C.; Kalpana, K. A technical view on transporters-the drug pharmacokinetics dictators. MOJ Bioequiv. Availab. 2019, 9, 47–52. [Google Scholar] [CrossRef]
- Liu, H.; Sahi, J. Role of Hepatic Drug Transporters in Drug Development. J. Clin. Pharm. 2016, 56 (Suppl. S7), S11–S22. [Google Scholar] [CrossRef]
- Ingelman-Sundberg, M. Pharmacogenetics of cytochrome P450 and its applications in drug therapy: The past, present and future. Trends Pharm. Sci. 2004, 25, 193–200. [Google Scholar] [CrossRef] [PubMed]
- Zanger, U.M.; Schwab, M. Cytochrome P450 enzymes in drug metabolism: Regulation of gene expression, enzyme activities, and impact of genetic variation. Pharm. Ther. 2013, 138, 103–141. [Google Scholar] [CrossRef] [PubMed]
- Fujikura, K.; Ingelman-Sundberg, M.; Lauschke, V.M. Genetic variation in the human cytochrome P450 supergene family. Pharm. Genom. 2015, 25, 584–594. [Google Scholar] [CrossRef] [PubMed]
- Zhou, Y.; Lauschke, V.M. The genetic landscape of major drug metabolizing cytochrome P450 genes—An updated analysis of population-scale sequencing data. Pharm. J. 2022, 22, 284–293. [Google Scholar] [CrossRef]
- Ingelman-Sundberg, M. Implications of polymorphic cytochrome p450-dependent drug metabolism for drug development. Drug Metab. Dispos. 2001, 29, 570–573. [Google Scholar]
- Zhou, S.F.; Liu, J.P.; Chowbay, B. Polymorphism of human cytochrome P450 enzymes and its clinical impact. Drug Metab. Rev. 2009, 41, 89–295. [Google Scholar] [CrossRef]
- Smolders, E.J.; de Kanter, C.T.M.M.; de Knegt, R.J.; van der Valk, M.; Drenth, J.P.H.; Burger, D.M. Drug–drug interactions between direct-acting antivirals and psychoactive medications. Clin. Pharm. 2016, 55, 1471–1494. [Google Scholar] [CrossRef]
- Gong, Y.; Haque, S.; Chowdhury, P.; Cory, T.J.; Kodidela, S.; Yallapu, M.M.; Norwood, J.M.; Kumar, S. Pharmacokinetics and pharmacodynamics of cytochrome P450 inhibitors for HIV treatment. Expert. Opin. Drug Metab. Toxicol. 2019, 15, 417–427. [Google Scholar] [CrossRef]
- Bahap, M.; Kara, E.; Sain Guven, G. Fighting on two fronts: Drug–drug interactions in people living with HIV infected with SARS-CoV-2. Eur. J. Hosp. Pharm. 2020, 28, e3. [Google Scholar] [CrossRef]
- Yagura, H.; Watanabe, D.; Kushida, H.; Tomishima, K.; Togami, H.; Hirano, A.; Takahashi, M.; Hirota, K.; Ikuma, M.; Kasai, D.; et al. Impact of UGT1A1 gene polymorphisms on plasma dolutegravir trough concentrations and neuropsychiatric adverse events in Japanese individuals infected with HIV-1. BMC Infect. Dis. 2017, 17, 622. [Google Scholar] [CrossRef] [PubMed]
- Belkhir, L.; Seguin-Devaux, C.; Elens, L.; Pauly, C.; Gengler, N.; Schneider, S.; Ruelle, J.; Haufroid, V.; Vandercam, B. Impact of UGT1A1 polymorphisms on Raltegravir and its glucuronide plasma concentrations in a cohort of HIV-1 infected patients. Sci. Rep. 2018, 8, 7359. [Google Scholar] [CrossRef] [PubMed]
- Khalilieh, S.; Feng, H.; Hulskotte, E.G.J.; Wenning, L.A.; Butterton, J.R. Clinical pharmacology profile of boceprevir, a hepatitis C virus NS3 protease inhibitor: Focus on drug-drug interactions. Clin. Pharm. 2015, 54, 599–614. [Google Scholar] [CrossRef] [PubMed]
- Abel, S.; Russell, D.; Whitlock, L.A.; Ridgway, C.E.; Nedderman, A.N.R.; Walker, D.K. Assessment of the absorption, metabolism and absolute bioavailability of maraviroc in healthy male subjects. Br. J. Clin. Pharm. 2008, 65, 60–67. [Google Scholar] [CrossRef] [PubMed]
- Huang, Q.; Riviere, J.E. The application of allometric scaling principles to predict pharmacokinetic parameters across species. Expert. Opin. Drug Metab. Toxicol. 2014, 10, 1241–1253. [Google Scholar] [CrossRef]
- Nair, A.B.; Jacob, S. A simple practice guide for dose conversion between animals and human. J. Basic Clin. Pharm. 2016, 7, 27–31. [Google Scholar] [CrossRef]
- Jairam, R.K.; Mallurwar, S.R.; Sulochana, S.P.; Chandrasekhar, D.V.; Todmal, U.; Bhamidipati, R.K.; Richter, W.; Srinivas, N.R.; Mullangi, R. Prediction of Human Pharmacokinetics of Fomepizole from Preclinical Species Pharmacokinetics Based on Normalizing Time Course Profiles. AAPS PharmSciTech 2019, 20, 221. [Google Scholar] [CrossRef]
- Mahmood, I.; Balian, J.D. Interspecies scaling: Predicting clearance of drugs in humans. Three different approaches. Xenobiotica 1996, 26, 887–895. [Google Scholar] [CrossRef]
- Tang, H.; Mayersohn, M. A novel model for prediction of human drug clearance by allometric scaling. Drug Metab. Dispos. 2005, 33, 1297–1303. [Google Scholar] [CrossRef]
- Mahmood, I.; Martinez, M.; Hunter, R.P. Interspecies allometric scaling. Part I: Prediction of clearance in large animals. J. Vet. Pharm. Ther. 2006, 29, 415–423. [Google Scholar] [CrossRef]
- Sawada, Y.; Hanano, M.; Sugiyama, Y.; Iga, T. Prediction of the disposition of beta-lactam antibiotics in humans from pharmacokinetic parameters in animals. J. Pharm. Biopharm. 1984, 12, 241–261. [Google Scholar] [CrossRef]
- Mordenti, J. Man versus beast: Pharmacokinetic scaling in mammals. J. Pharm. Sci. 1986, 75, 1028–1040. [Google Scholar] [CrossRef] [PubMed]
- Boxenbaum, H. Evolutionary biology, animal behavior, fourth-dimensional space, and the raison d’etre of drug metabolism and pharmacokinetics. Drug Metab. Rev. 1983, 14, 1057–1097. [Google Scholar] [CrossRef] [PubMed]
- Boxenbaum, H. Interspecies pharmacokinetic scaling and the evolutionary-comparative paradigm. Drug Metab. Rev. 1984, 15, 1071–1121. [Google Scholar] [CrossRef]
- Chaira, T.; Barman, T.; Samuel Raj, V. In Vitro ADME, Preclinical Pharmacokinetics and Prediction of Human Pharmacokinetics of RBx14255, a Novel Ketolide with Pharmacodynamics Against Multidrug-Resistant Bacterial Pathogens. J. Pharm. Pharm. Sci. 2020, 23, 206–219. [Google Scholar] [CrossRef]
- Mahmood, I.; Yuan, R. A comparative study of allometric scaling with plasma concentrations predicted by species-invariant time methods. Biopharm. Drug Dispos. 1999, 20, 137–144. [Google Scholar] [CrossRef]
- Eagle, H.; Fleischman, R.; Musselman, A.D. The effective concentrations of penicillin in vitro and in vivo for streptococci, pneumococci, and Treponema pallidum. J. Bacteriol. 1950, 59, 625–643. [Google Scholar] [CrossRef]
- Eagle, H.; Fleischman, R.; Musselman, A.D. Effect of schedule of administration on the therapeutic efficacy of penicillin; importance of the aggregate time penicillin remains at effectively bactericidal levels. Am. J. Med. 1950, 9, 280–299. [Google Scholar] [CrossRef]
- Ambrose, P.G.; Bhavnani, S.M.; Rubino, C.M.; Louie, A.; Gumbo, T.; Forrest, A.; Drusano, G.L. Pharmacokinetics-pharmacodynamics of antimicrobial therapy: It’s not just for mice anymore. Clin. Infect. Dis. 2007, 44, 79–86. [Google Scholar] [CrossRef]
- Craig, W.A. Pharmacokinetic/pharmacodynamic parameters: Rationale for antibacterial dosing of mice and men. Clin. Infect. Dis. 1998, 26, 1–10. [Google Scholar] [CrossRef]
- Dudley, M.N.; Ambrose, P.G. Pharmacodynamics in the study of drug resistance and establishing in vitro susceptibility breakpoints: Ready for prime time. Curr. Opin. Microbiol. 2000, 3, 515–521. [Google Scholar] [CrossRef] [PubMed]
- Rodriguez-Gascon, A.; Solinis, M.A.; Isla, A. The Role of PK/PD Analysis in the Development and Evaluation of Antimicrobials. Pharmaceutics 2021, 13, 833. [Google Scholar] [CrossRef] [PubMed]
- Ambrose, P.G.; Grasela, D.M.; Grasela, T.H.; Passarell, J.; Mayer, H.B.; Pierce, P.F. Pharmacodynamics of fluoroquinolones against Streptococcus pneumoniae in patients with community-acquired respiratory tract infections. Antimicrob. Agents Chemother. 2001, 45, 2793–2797. [Google Scholar] [CrossRef] [PubMed]
- Asin-Prieto, E.; Rodriguez-Gascon, A.; Isla, A. Applications of the pharmacokinetic/pharmacodynamic (PK/PD) analysis of antimicrobial agents. J. Infect. Chemother. 2015, 21, 319–329. [Google Scholar] [CrossRef]
- Heffernan, A.J.; Sime, F.B.; Lipman, J.; Roberts, J.A. Individualising Therapy to Minimize Bacterial Multidrug Resistance. Drugs 2018, 78, 621–641. [Google Scholar] [CrossRef]
- Jorda, A.; Zeitlinger, M. Preclinical Pharmacokinetic/Pharmacodynamic Studies and Clinical Trials in the Drug Development Process of EMA-Approved Antibacterial Agents: A Review. Clin. Pharm. 2020, 59, 1071–1084. [Google Scholar] [CrossRef]
- Nicolau, D.P. Optimizing outcomes with antimicrobial therapy through pharmacodynamic profiling. J. Infect. Chemother. 2003, 9, 292–296. [Google Scholar] [CrossRef]
- Drusano, G.L. Antimicrobial pharmacodynamics: Critical interactions of ‘bug and drug’. Nat. Rev. Microbiol. 2004, 2, 289–300. [Google Scholar] [CrossRef]
- McSharry, J.J.; Drusano, G.L. Antiviral pharmacodynamics in hollow fibre bioreactors. Antivir. Chem. Chemother. 2011, 21, 183–192. [Google Scholar] [CrossRef]
- Widmer, N.; Meylan, P.; Ivanyuk, A.; Aouri, M.; Decosterd, L.A.; Buclin, T. Oseltamivir in seasonal, avian H5N1 and pandemic 2009 A/H1N1 influenza: Pharmacokinetic and pharmacodynamic characteristics. Clin. Pharm. 2010, 49, 741–765. [Google Scholar] [CrossRef]
- He, G.; Massarella, J.; Ward, P. Clinical pharmacokinetics of the prodrug oseltamivir and its active metabolite Ro 64-0802. Clin. Pharm. 1999, 37, 471–484. [Google Scholar] [CrossRef] [PubMed]
- Mendel, D.B.; Tai, C.Y.; Escarpe, P.A.; Li, W.; Sidwell, R.W.; Huffman, J.H.; Sweet, C.; Jakeman, K.J.; Merson, J.; Lacy, S.A.; et al. Oral administration of a prodrug of the influenza virus neuraminidase inhibitor GS 4071 protects mice and ferrets against influenza infection. Antimicrob. Agents Chemother. 1998, 42, 640–646. [Google Scholar] [CrossRef] [PubMed]
- Sidwell, R.W.; Smee, D.F. In vitro and in vivo assay systems for study of influenza virus inhibitors. Antivir. Res. 2000, 48, 1–16. [Google Scholar] [CrossRef] [PubMed]
- McSharry, J.J.; Weng, Q.; Brown, A.; Kulawy, R.; Drusano, G.L. Prediction of the pharmacodynamically linked variable of oseltamivir carboxylate for influenza A virus using an in vitro hollow-fiber infection model system. Antimicrob. Agents Chemother. 2009, 53, 2375–2381. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Drusano, G.L.; Preston, S.L.; Smee, D.; Bush, K.; Bailey, K.; Sidwell, R.W. Pharmacodynamic evaluation of RWJ-270201, a novel neuraminidase inhibitor, in a lethal murine model of influenza predicts efficacy for once-daily dosing. Antimicrob. Agents Chemother. 2001, 45, 2115–2118. [Google Scholar] [CrossRef]
- Brown, A.N.; McSharry, J.J.; Weng, Q.; Adams, J.R.; Kulawy, R.; Drusano, G.L. Zanamivir, at 600 milligrams twice daily, inhibits oseltamivir-resistant 2009 pandemic H1N1 influenza virus in an in vitro hollow-fiber infection model system. Antimicrob. Agents Chemother. 2011, 55, 1740–1746. [Google Scholar] [CrossRef]
- Obach, R.S.; Baxter, J.G.; Liston, T.E.; Silber, B.M.; Jones, B.C.; MacIntyre, F.; Rance, D.J.; Wastall, P. The prediction of human pharmacokinetic parameters from preclinical and in vitro metabolism data. J. Pharm. Exp. Ther. 1997, 283, 46–58. [Google Scholar]
- Sharma, A.; Benbrook, D.M.; Woo, S. Pharmacokinetics and interspecies scaling of a novel, orally-bioavailable anti-cancer drug, SHetA2. PLoS ONE 2018, 13, e0194046. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).