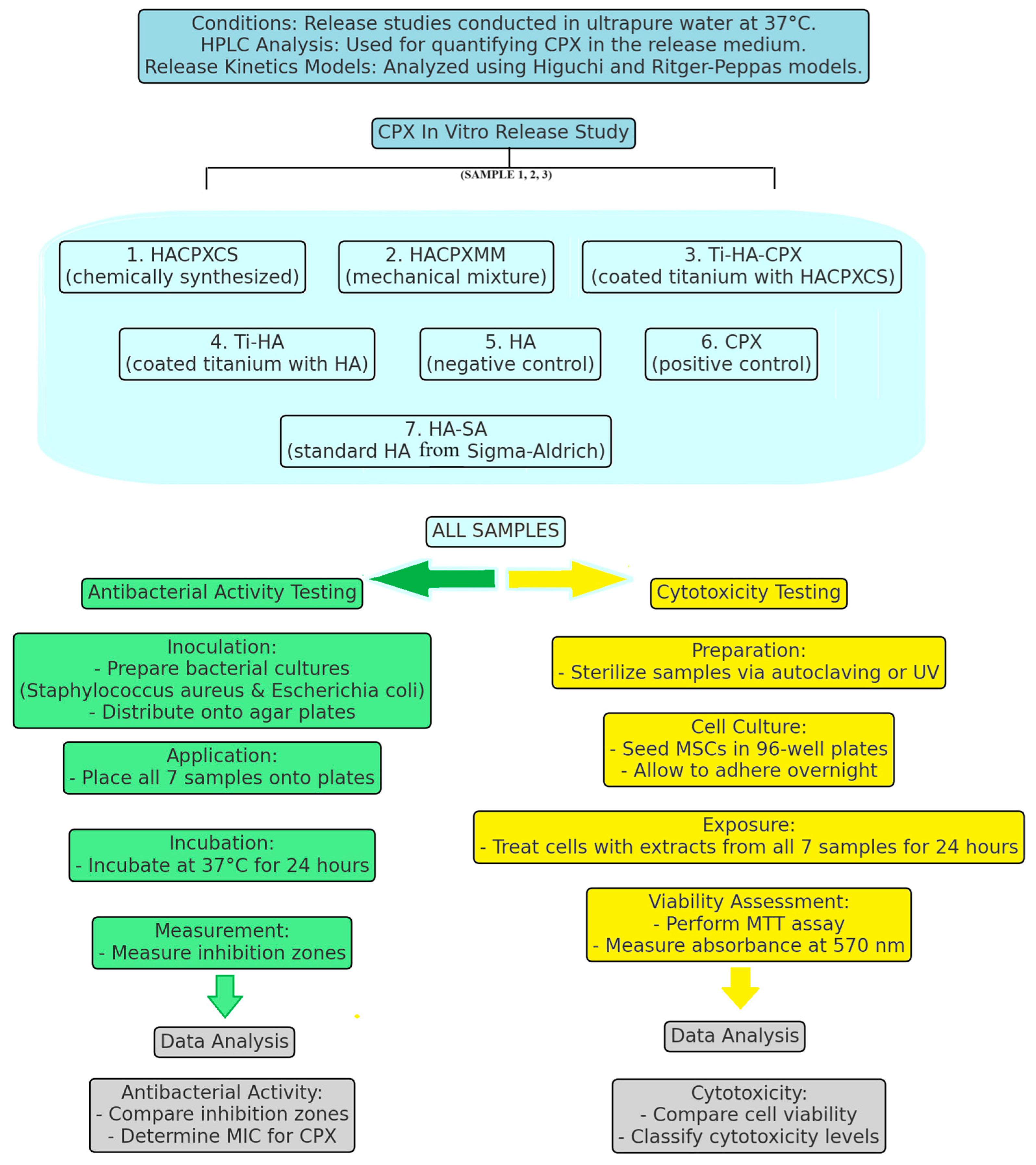
1. Introduction
Bone and joint infections contribute significantly to global morbidity and healthcare costs. Chronic osteomyelitis, in particular, presents a persistent clinical challenge due to its recurrent nature and complex treatment requirements. In the United States, the incidence of osteomyelitis is estimated at 21.8 cases per 100,000 person-years, with higher rates in individuals with diabetes or peripheral vascular disease. In Europe, the incidence ranges from 20 to 30 cases per 100,000 person-years, varying by region and population demographics [
1,
2]. The financial burden of osteomyelitis treatment is substantial, with hospitalization, prolonged antibiotic therapy, and potential surgical interventions significantly impacting healthcare expenditures. The average cost per patient ranges from
$20,000 to
$60,000, depending on the severity and complications [
3].
Treating bone and joint infections, such as osteomyelitis, septic arthritis, and prosthetic joint infections, presents significant challenges, necessitating prolonged antibiotic administration and supplementary surgical intervention. Despite advancements in medical treatments, there remains a substantial research gap in developing effective localized treatments that minimize systemic side effects and reduce the need for multiple surgical procedures. The functionalization of implantable biocomposites with antibiotics and their localized release offer advantages in the treatment of chronic osteomyelitis and infected fractures. Furthermore, the antibiotic-functionalized bioceramic implant enhances the feasibility of a single-stage surgical intervention, obviating the need for subsequent surgery to remove the spacer.
The application of ceramic biocomposites for delivering antibiotics, along with bone morphogenetic proteins, bisphosphonates, growth factors, or living cells, is currently under investigation and requires further study [
4]. Recent studies have shown that combining antibiotics with hydroxyapatite (HA) can significantly enhance their antibacterial properties. For instance, silver-doped HA has demonstrated enhanced antibacterial activity against multidrug-resistant bacterial strains [
5]. Moreover, the incorporation of antibiotics such as vancomycin and tetracycline into HA matrices has shown improved antibacterial efficacy compared to the antibiotics alone [
6]. These findings suggest that HA can serve as an effective carrier for antibiotics, enhancing their delivery and potency.
In particular, the development of novel tetracycline and CPX-loaded silver-doped HA suspensions has shown promising results for biomedical applications. These suspensions exhibit strong antibacterial activity, making them suitable for preventing infections in various medical settings [
7]. Additionally, the fabrication of bioactive rifampicin-loaded κ-Car-MA-INH/nano HA composites has demonstrated significant potential for treating tuberculosis, osteomyelitis, and regenerating infected tissue. These composites effectively deliver rifampicin, maintaining its antibacterial efficacy while supporting tissue regeneration [
8].
Furthermore, the use of levofloxacin-loaded mesoporous silica microspheres, nano-HA, and polyurethane composite scaffolds has proven effective in treating chronic osteomyelitis with bone defects. These scaffolds provide sustained release of levofloxacin, ensuring prolonged antibacterial activity and supporting bone healing [
9]. The primary cause of both acute and chronic hematogenous osteomyelitis in adults and children is
Staphylococcus aureus infections. Chronic osteomyelitis, which may result from neighboring infections, has shown isolates of
Staphylococcus epidermidis,
Pseudomonas aeruginosa,
Serratia marcescens, and
Escherichia coli. Fungal and mycobacterial infections causing osteomyelitis are less common and are typically reported in patients with immune deficiencies. Treatment typically involves oral and injectable routes of administration for CPX, a broad-spectrum antibiotic that is effective against a wide range of Gram-negative and Gram-positive bacteria. Its mechanism of action involves inhibiting DNA topoisomerases, enzymes that are crucial for bacterial DNA replication and repair. This bactericidal activity makes CPX particularly effective in treating severe and persistent infections [
10]. CPX has excellent tissue penetration, including into bone tissue, which is critical for effectively treating osteomyelitis. The ability to reach high concentrations at the site of infection enhances its therapeutic efficacy. Plants coated with CPX can provide a sustained release of the antibiotic over an extended period. This prolonged release helps maintain therapeutic drug levels at the infection site, which is crucial for effectively eradicating chronic infections and preventing recurrence. Localized delivery of CPX from implants reduces the need for high systemic doses, thereby lowering the risk of systemic toxicity and adverse effects. This is particularly important for patients who may require long-term antibiotic therapy. CPX-functionalized implants can help prevent the formation of bacterial biofilms on the implant surface. Biofilms are complex communities of bacteria that are highly resistant to antibiotics and are a common cause of persistent infections in implant-related osteomyelitis.
Given the promising results from recent research, this study aims to explore the antibacterial efficacy and cytotoxicity of CPX-functionalized bioceramic implants. Specifically, we synthesized HA-ciprofloxacin (HACPXCS) composites and applied them to titanium substrates (Ti-HA-CPX), evaluating their performance against Staphylococcus aureus (ATCC 25923) and Escherichia coli (ATCC 25922). Our approach includes molecular docking studies to understand the binding affinities between CPX and bacterial proteins, correlating these findings with enhanced antibacterial efficacy. By addressing the current gaps in research, this study contributes to the development of more effective treatment strategies for chronic bone infections, balancing antibacterial effectiveness with manageable cytotoxicity. This approach not only facilitates a deeper understanding of antibiotic action at the molecular level but also offers a novel perspective on enhancing antibiotic delivery directly to the infection site, potentially improving treatment efficacy.
3. Results
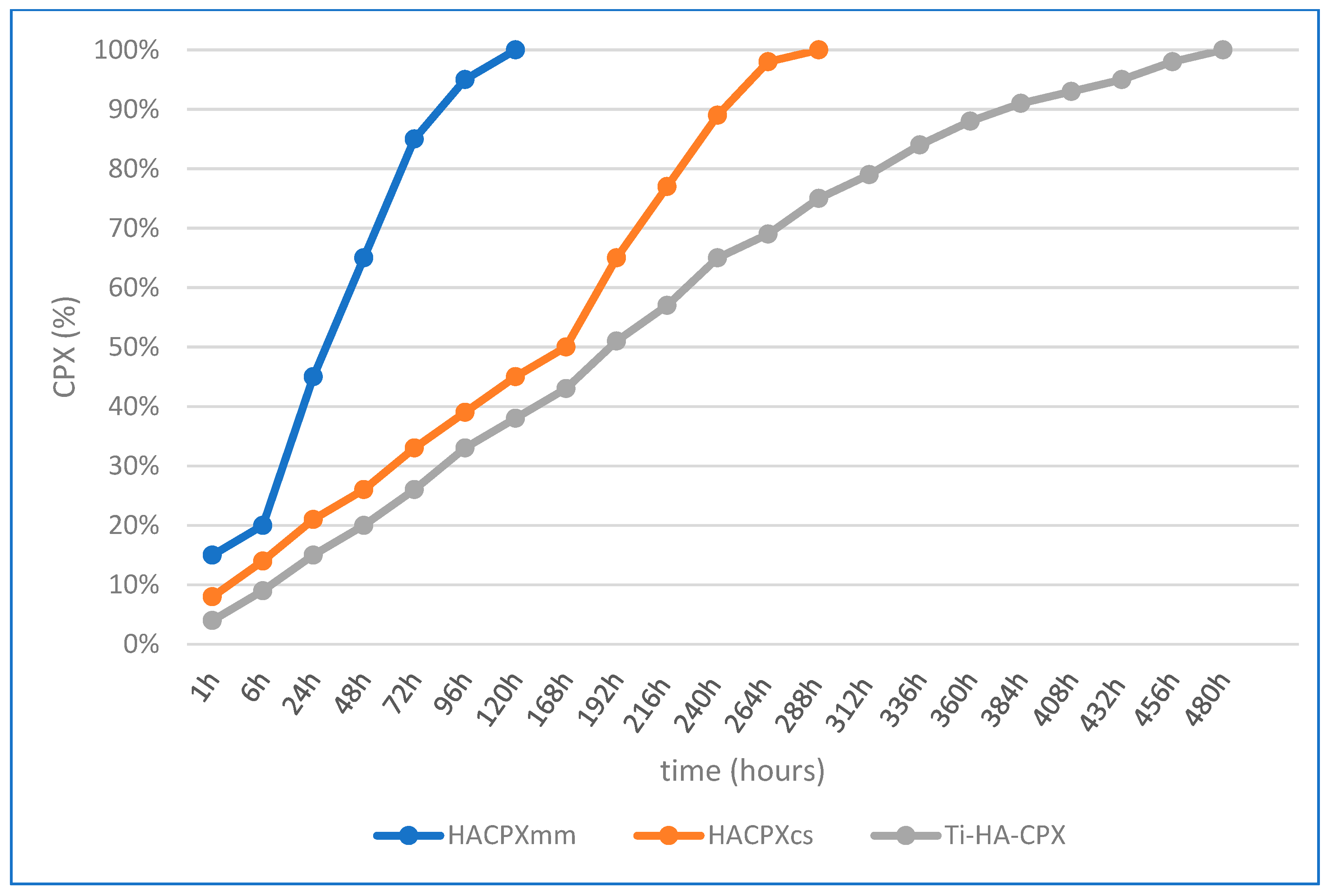
For HA-CPX tablets prepared by mechanical mixing and pressing, the release profile was rapid, with 85% of CPX released within the first 3 days. The Higuchi model showed a higher correlation coefficient (R
2 = 0.987) compared to the Ritger-Peppas model (R
2 = 0.975), indicating that diffusion was the main release mechanism. For HA-CPX tablets synthesized chemically, 50% of CPX was released within the first 7 days, with 98% released by the 11th day. The higher correlation coefficient for the Higuchi model (R
2 = 0.980) compared to the Ritger-Peppas model (R
2 = 0.972) also suggested diffusion as the main release process. For Ti-HA-CPX coatings on titanium plates, the initial release was higher due to desorption from the HA surface, followed by slower diffusion from the HA matrix. The Higuchi model showed a correlation coefficient of 0.988, while the Ritger-Peppas model showed a slightly higher correlation coefficient of 0.992, suggesting that alongside diffusion, other processes such as surface erosion and desorption also contributed to the drug release (
Figure 4).
The results show that the heat of formation for CPX, HACPX
CS, and Ti-HA-CPX-CPX is −98.52 kcal/mol, −395.03 kcal/mol, and −514.53 kcal/mol, respectively. The increased stability (more negative values) of HACPX
CS and Ti-HA-CPX correlates with their enhanced antibacterial activity compared to CPX alone, as evidenced by larger inhibition zones in antibacterial tests (Ti-HA-CPX had an inhibition zone diameter of 33.5 mm for
Staphylococcus aureus (ATCC 25923)) (
Table 2). In the study, the dipole moment values are 7.85 Debye for CPX, 15.22 Debye for HACPX
CS, and 10.96 Debye for Ti-HA-CPX. The higher dipole moment of HACPX
CS suggests greater reactivity, which can enhance its ability to disrupt bacterial cell membranes, contributing to its higher antibacterial activity. The total energy values in the study are −945.24 kcal/mol for CPX, −1968.04 kcal/mol for HACPX
CS, and −2025.25 kcal/mol for Ti-HA-CPX. The significantly lower total energy of HACPX
CS and Ti-HA-CPX indicates greater stability and is associated with better antibacterial performance compared to CPX alone (
Table 2).
In
Table 3, the frontier orbitals of the CPX, HACPX
CS, and Ti-HA-CPX are presented. The investigation of molecular surface areas for the two types of molecular orbitals reveals the contribution of atomic orbitals to their formation. Molecules with the highest E
HOMO values are most susceptible to electrophilic attack, while those with the highest E
LUMO values are more susceptible to nucleophilic attack. Based on the values provided in
Table 2, the most stable molecule is Ti-HA-CPX, followed by HACPX
CS.
Table 4 provides the following ΔE values: 11.13 eV for CPX, 7.64 eV for HACPX
CS, and 5.72 eV for Ti-HA-CPX. The smaller ΔE for HACPX
CS and Ti-HA-CPX correlates with their enhanced antibacterial activity, as these compounds are more reactive and capable of interacting more effectively with bacterial cells (
Table 4).
Molecular docking studies show the binding modes and energies of CPX, HACPXCS, and Ti-HA-CPX with bacterial proteins. The binding energies are −4.68 ± 0.01 kcal/mol for Escherichia coli and −5.46 ± 0.03 kcal/mol for Staphylococcus aureus.
Ti-HACPX
CS shows the highest antibacterial efficacy against both
Staphylococcus aureus (ATCC 25923) (33.5 ± 0.3 mm) and
Escherichia coli (ATCC 25922) (27.5 ± 0.4 mm), indicating a strong synergistic effect of CPX when combined with Ti-HA. HACPX
CS and HACPX
MM show similar inhibition zones, suggesting that both methods of combining HA with CPX are effective. Both treatments exhibit high antibacterial activity, with inhibition zones indicating sensitivity to both bacterial strains. The standard CPX (CPX-SA) also shows high antibacterial activity, with inhibition zones of 29 ± 0.4 mm for
Staphylococcus aureus (ATCC 25923) and 27.5 ± 0.3 mm for
Escherichia coli (ATCC 25922), reinforcing CPX’s effectiveness as an antibiotic. HA-synthesis and HA-SA show much lower antibacterial activity, with HA-SA being particularly ineffective against
Staphylococcus aureus (ATCC 25923) (14.5 ± 0.2 mm, resistant) and only intermediate against
Escherichia coli (ATCC 25922) (17 ± 0.2 mm). This underscores the fact that HA has limited antibacterial properties on its own. The Ti-HA composite shows intermediate antibacterial activity, better than HA alone but significantly less effective than Ti-HA-CPX. This indicates that the antibacterial properties are primarily due to CPX’s incorporation. The molecular docking results in
Table 5 align well with the antibacterial efficacy results in
Table 6. Strong binding energies for CPX, HACPX
CS, and Ti-HA-CPX correspond to high antibacterial activity, confirming the effectiveness of these composites in inhibiting bacterial growth. The superior performance of Ti-HA-CPX highlights the synergistic benefits of combining CPX with titanium and HA, leading to enhanced stability and release of the antibiotic, thereby improving its antibacterial efficacy.
Ti-HA, HA-synthesis, and HA-SA samples showed the highest cell viability at 95%, indicating that they are highly biocompatible with MSCs. Ti-HA-CPX, HACPX
CS, and HACPX
MM exhibited moderate cytotoxicity with cell viability in the range of 75% to 80%. The presence of CPX in these composites likely contributed to the observed cytotoxic effects, as antibiotics can affect cellular metabolism and viability. The CPX-SA sample demonstrated the lowest cell viability at 50%, indicating high cytotoxicity. Direct exposure to pure CPX significantly affected the MSCs, likely due to the antibiotic’s potent activity interfering with cellular functions (
Table 7).
The Tukey HSD test reveals that Ti-HA-CPX has significantly larger inhibition zones compared to other products, including HACPXCS, HACPXMM, and CPX-SA, indicating superior antibacterial efficacy against Staphylococcus aureus (ATCC 25923). Similar results were observed, with Ti-HA-CPX showing significantly higher antibacterial activity compared to other samples against Escherichia coli (ATCC 25922). For both Staphylococcus aureus (ATCC 25923) and Escherichia coli (ATCC 25922), Ti-HA-CPX and CPX-SA consistently show significantly higher antibacterial activity compared to other products (p < 0.001). For the cytotoxicity test, the Tukey HSD test shows that Ti-HA, HA-synthesis, and HA-SA have significantly higher cell viability compared to Ti-HA-CPX and CPX-SA. Ti-HA-CPX and CPX-SA exhibit lower cell viability, indicating higher cytotoxicity. Pairwise t-tests confirm the significant differences observed in the Tukey HSD test. Products such as Ti-HA-CPX and CPX-SA show significant differences in cell viability compared to other products, suggesting higher cytotoxicity. Ti-HA, HA-synthesis, and HA-SA are highly biocompatible, showing high cell viability and low cytotoxicity. Ti-HA-CPX and CPX-SA exhibit moderate to high cytotoxicity, which might limit their use in certain clinical applications where cell compatibility is crucial. The results suggest a trade-off between antibacterial efficacy and cytotoxicity. While Ti-HA-CPX is highly effective against bacteria, its higher cytotoxicity needs to be carefully managed.
4. Discussion
The enthalpy of formation is a measure of the heat energy released or absorbed when a chemical substance is formed; a low value indicates that the substance is more stable. The electric dipole moment reflects the partial separation of electric charge within a molecule, with a high value indicating increased reactivity [
18,
19,
20]. The total energy expresses the stability of a molecule, with a low value of this descriptor being correlated with increased stability. By calculating the electrostatic potential, a three-dimensional map of the electron density was obtained, with electrostatic potential values indicated by different colors. The drug-substance-to-protein-target interaction, which produces the corresponding therapeutic effect, is only possible for a certain form of the molecule and a specific distribution of electron density within the molecule. This distribution generates regions rich in electrons (
Table 3). The electrostatic potential can be used to assess molecular lipophilicity, as it characterizes the polarity of a specific region on the van der Waals surface of the molecule. Regions with high potential values will strongly attract polar molecules, while regions with low values will not and can be considered hydrophobic [
21]. The presence of atoms with partial negative charges (such as halogens, sulfur, nitrogen, and oxygen) in the ligand molecule results in a large number of ligand-receptor interactions [
22]. The value of the E
HOMO descriptor indicates the donor properties of a molecule, that is, its tendencies towards oxidation, while the value of the E
LUMO descriptor allows for the estimation of a molecule’s acceptor properties, that is, its reduction tendencies. The energy difference between the HOMO and LUMO levels (ΔE = E
LUMO-E
HOMO) is an important chemico-biological molecular descriptor, as it explains the stability of the molecule, with a low value indicating that the molecule has increased reactivity.
Research studies have reported that the HOMO-LUMO energy gap (ΔE) is an important indicator of antibacterial activity. In a study by Obot et al., it was shown that a smaller HOMO-LUMO gap often correlates with higher antibacterial activity, as it suggests greater chemical reactivity and potential for interaction with bacterial cells. Their research demonstrated that molecules with smaller ΔE values had better antibacterial efficacy, as confirmed through both in vitro and docking studies [
23].
Other researchers have found that the heat of formation of a molecule can correlate with its antibacterial activity. For instance, a study by Chen et al. demonstrated that molecules with lower heat of formation values tend to exhibit higher antibacterial activity. This is because a lower heat of formation often indicates greater stability of the molecule, which may enhance its interaction with bacterial targets [
24].
The dipole moment is an expression of the molecular system’s polarization and serves as a predictor of the chemical reactivity of the molecules, which affects their reactivity and interaction with bacterial cell membranes. Other researchers have shown that a higher dipole moment can improve the binding affinity of molecules in docking studies. This improved binding affinity is often linked to better antibacterial activity as stronger interactions with bacterial proteins are facilitated. Ishihara et al. highlighted that molecules with higher dipole moments showed enhanced docking scores against bacterial enzymes, correlating with their observed antibacterial efficacy [
25].
Other researchers have observed a correlation between the total energy of a molecule and its antibacterial properties. Shah et al. found that molecules with lower total energy values tend to exhibit higher antibacterial activity. This is attributed to the fact that lower total energy reflects greater molecular stability and potentially more effective interaction with bacterial cell components [
26].
Molecular docking results suggest that HACPX
CS and Ti-HA-CPX have strong binding affinities, which likely contribute to their higher antibacterial activities observed in vitro. The molecular docking study highlights the binding mode and energy of CXP, HACPX
CS, and Ti-HA-CPX as being almost equal to those of the pure antibiotic, as detailed in
Table 5. The 2D diagrams illustrate the types of bonds established between CPX and the protein targets (
Figure 5 and
Figure 6). These correlations demonstrate that more stable, reactive, and effectively binding molecules, as indicated by their molecular descriptors and docking studies, tend to exhibit enhanced antibacterial activity.
The sustained release profile of CPX from HACPXCS and Ti-HA-CPX composites, maintaining prolonged antibacterial activity, is particularly significant for clinical applications. This feature could reduce the need for repeated antibiotic administration and minimize systemic side effects, which is crucial for treating chronic osteomyelitis and infected fractures. The ability of these composites to provide localized antibiotic delivery directly to the infection site offers a promising approach for single-stage surgical interventions, potentially improving treatment outcomes and reducing healthcare costs associated with prolonged antibiotic therapy and multiple surgeries.
Although the antimicrobial activity of CPX against the germs studied in this experiment is known, the binding of this compound to HA could modify the antimicrobial activity of the chemical compound [
27]. Following the performance of antibiograms to determine the MIC, we have established that a dose of 5 µg CPX is the lowest amount of antibiotic that inhibits bacterial growth on the tested germs. Data on the antimicrobial activity of HA are quite contradictory. While initial studies on the chemical synthesis of HA did not refer to its antibacterial activity, using it as a negative control, more recently, there has been an increasing notion that it may have a weak antibacterial effect. A research group found that HA nanoparticles exhibit significant antibacterial activity against both Gram-positive and Gram-negative bacteria, suggesting their potential application in biomedical devices to prevent infections. This study demonstrated that the antibacterial properties were due to the physical disruption of bacterial cell membranes and the release of calcium and phosphate ions, which interfered with bacterial metabolism [
28]. In another study, researchers investigated the synergistic effects of HA and various antibiotics. They found that the combination of HA with antibiotics such as tetracycline and vancomycin significantly enhanced antibacterial efficacy compared to antibiotics alone. The study concluded that HA could serve as an effective carrier to enhance the delivery and potency of antibiotics [
29]. Another investigation revealed that silver-doped HA exhibits enhanced antibacterial activity against multi-drug-resistant bacterial strains. The incorporation of silver ions into the HA matrix provided a sustained release of silver, which effectively inhibited bacterial growth and biofilm formation. This study highlights the potential of silver-doped HA as a multifunctional material for use in bone implants to reduce the risk of post-surgical infections [
30]. In another study, a vancomycin-loaded HA/collagen/PLA bone graft substitute was developed that exhibited a high bacterial inhibition rate and strong adherence to the damaged site without causing inflammatory reactions. This study highlights the infection-inhibition properties of HA composites when combined with antibiotics, supporting their use in preventing infections in orthopedic applications [
31]. As observed in the results presented in
Table 6, HA deposits on substrates are characterized by a weak antibacterial effect. The inhibition zone diameters for HA samples used in the study are approximately the same, with higher values in the case of
Escherichia coli (ATCC 25922). This is consistent with literature data indicating that HA nanoparticles exhibit a more pronounced antibacterial character against
Escherichia coli (ATCC 25922), explained by the relatively thin cell wall thickness characteristic of this bacterial species and its specific chemical structure. Although the antimicrobial activity of CPX against the studied bacteria is well known, binding this compound to HA could potentially modify the antimicrobial activity of the chemically synthesized compound.
Our results with HACPX
CS and HACPX
MM showed similar antibacterial activities, with inhibition zones of 26 mm for both
Staphylococcus aureus (ATCC 25923) and
Escherichia coli (ATCC 25922). This suggests that, like gentamicin in Suchý et al.’s study, CPX retains its antibacterial efficacy when combined with HA. However, the 30% reduction seen with vancomycin in Suchý et al.’s study is not observed in our results, indicating a stable integration of CPX within the HA matrix [
32]. Gomes et al.’s findings support our observations of sustained antibacterial activity in HACPX
CS composites. Both HACPX
CS and HACPX
MM demonstrated substantial antibacterial zones, indicating effective and sustained release of CPX akin to the amoxicillin release observed in Gomes et al.’s study [
33]. Prasanna et al. found that the antimicrobial properties of CPX and tetracycline embedded in silver-doped HA (AgHA-C and AgHA-T) suspensions were tested against
Staphylococcus aureus (ATCC 25923),
Escherichia coli (ATCC 25922), and
Candida albicans. The results showed that both AgHA-C and AgHA-T suspensions exhibited exceptional antibacterial activity, which increased with longer incubation times. Our study did not observe a significant decrease in antibacterial activity in HACPX
CS composites compared to CPX alone. This suggests that the interaction between CPX and HA in our composites may be more stable, preserving CPX’s efficacy [
34]. The high antibacterial activity of Ti-HA-CPX in our study, with inhibition zones of 33.5 mm for
Staphylococcus aureus (ATCC 25923) and 27.5 mm for
Escherichia coli (ATCC 25922), aligns well with Predoi et al.’s findings. The integration of CPX in HA on Ti substrates enhances antibacterial efficacy, similar to the vancomycin-coated Ti implants in their study [
35]. Our HACPX
CS composites’ effective antibacterial activity and sustained release are consistent with Amarnath et al.’s findings. The retention of antibacterial properties in our HACPX
CS samples mirrors the sustained efficacy of rifampicin in HA-gelatin composites.
Staphylococcus aureus (ATCC 25923) [
36]. The prolonged antibacterial activity of Ti-HA-CPX in our study aligns with Wang et al.’s findings. The effective integration of CPX in HA and Ti substrates supports its potential for treating chronic infections, similar to levofloxacin in HA beads. The moderate cytotoxicity observed in our HACPX
CS composites (75–80% cell viability) is consistent with other researchers findings of acceptable cytotoxicity levels in HA-antibiotic biocomposites. This suggests that HA can effectively control antibiotic release while maintaining biocompatibility [
37]. The sustained antibacterial activity observed in our HACPX
CS composites parallels Alvarez et al.’s findings, indicating that HA matrices can effectively manage antibiotic release and maintain antimicrobial efficacy [
38]. Unlike Shinde et al.’s findings, our study did not observe a significant reduction in CPX’s antibacterial activity when incorporated into HA. This indicates a more stable integration of CPX in HA, preserving its efficacy [
39]. Our results differed from Ghosh et al.’s observations as CPX maintained its antibacterial efficacy when combined with HA, suggesting a stable incorporation that prevents significant loss of activity [
40]. Therefore, we prepared samples in the form of tablets, obtained both from the chemically synthesized composite and the mechanical mixture of HA and CPX, maintaining the same ratio as in our synthesized compound (20%). The antibiogram results indicate that binding CPX to HA minimally modifies antibacterial activity (inhibition zone diameter decreases by less than 2% upon binding to HA). An aspect observed in this study is a slight increase in the inhibition zone when the studied compounds are deposited on Ti substrates, compared to when they are in tablet form. It is well known that Ti-based alloys are covered by a spontaneously forming TiO
2 layer due to oxidation reactions under light influence.
Several studies in the literature show that this oxide layer has antimicrobial properties. Given that the film deposited through MAPLE is found only on one side of the Ti discs, the other side (which contains TiO
2) could enhance the antimicrobial effect [
41].
Another study shows significant limitations regarding the use of the antibacterial effect of the TiO
2 layer in biomedical applications, indicating that this layer must be designed according to the type of bacteria, their attachment to the surface, their growth mechanism, and the environment in which they are found [
42].
The antibacterial efficacy of bioceramic samples, as indicated by larger inhibition zones (
Table 6), must be balanced against their cytotoxic effects (
Table 7). For instance, Ti-HA-CPX showed the highest antibacterial activity but moderate cytotoxicity, suggesting its potential for clinical use where infection prevention is paramount, but careful consideration of dosage and release mechanisms is necessary to minimize cytotoxic effects. The incorporation of antibiotics such as CPX into bioceramic matrices should be optimized to enhance antibacterial efficacy while maintaining acceptable cytotoxicity levels. Techniques such as controlled release, surface modifications, and composite formulations can help achieve this balance. Our study showed that Ti-HA, HA-synthesis, and HA-SA exhibited low cytotoxicity with a cell viability of 95%. This high viability suggests that these bioceramics do not significantly affect cell growth and proliferation, making them suitable for applications where biocompatibility is crucial, such as in bone regeneration and repair. This high viability indicates excellent biocompatibility, consistent with the findings of Pupo et al., who reported that HA scaffolds without antibiotics supported high cell viability and were suitable for bone regeneration applications. Similarly, Song et al. found that HA-gelatin composites without antibiotics showed high biocompatibility, supporting cell adhesion and proliferation [
43].
Ti-HA-CPX, HACPX
CS, and HACPX
MM samples demonstrated moderate cytotoxicity, with cell viability ranging from 75% to 80%. This is in line with the findings of Patel et al., who observed that HA-alginate biocomposites loaded with clindamycin showed moderate cytotoxic effects but remained within an acceptable range for clinical applications. Additionally, Zheng et al. reported that HA-chitosan composites loaded with doxycycline exhibited moderate cytotoxicity while maintaining effective antibacterial properties. CPX alone exhibited high cytotoxicity, with cell viability at 50%. This is consistent with the observations by Shinde et al., who reported significant cytotoxic effects of cefazolin when not embedded in a biocompatible matrix, reducing cell viability substantially. Kim et al. also found that azithromycin combined with HA showed reduced cell viability due to the adsorption properties of HA, which affected the antibiotic’s bioavailability and increased cytotoxicity [
30,
44,
45].
The present study is limited by a relatively small sample size, which impacts the generalizability and statistical power of the findings. Future research with larger sample sizes is necessary to validate these findings and provide more comprehensive insights into the antibacterial efficacy and cytotoxicity of CPX-functionalized bioceramic implants. Expanding the sample size would enhance the reliability of the results and improve the applicability of the study’s conclusions to a broader context.