Supplemental Effects of Biochar and Foliar Application of Ascorbic Acid on Physio-Biochemical Attributes of Barley (Hordeum vulgare L.) under Cadmium-Contaminated Soil
Abstract
:1. Introduction
2. Materials and Methods
2.1. Seed Sterilization
2.2. Experimental Design
2.3. Seeds Sowing and Pot Preparation
2.4. Cadmium (Cd) Contamination
2.5. Ascorbic Acid (Asa)
2.6. Growth Attributes
2.7. Measurement of Chlorophyll Contents and Gas Exchange Characteristics
2.8. Electrolyte Leakage
2.9. Oxidative Stress Indicators
2.10. Determination of Antioxidant Enzymatic Activities
2.11. Estimation of Proline, Sugars, and Non-Enzymatic Antioxidants
2.12. Analysis of Cadmium Contents
2.13. Statistical Analysis
3. Results
3.1. Root and Shoot Length
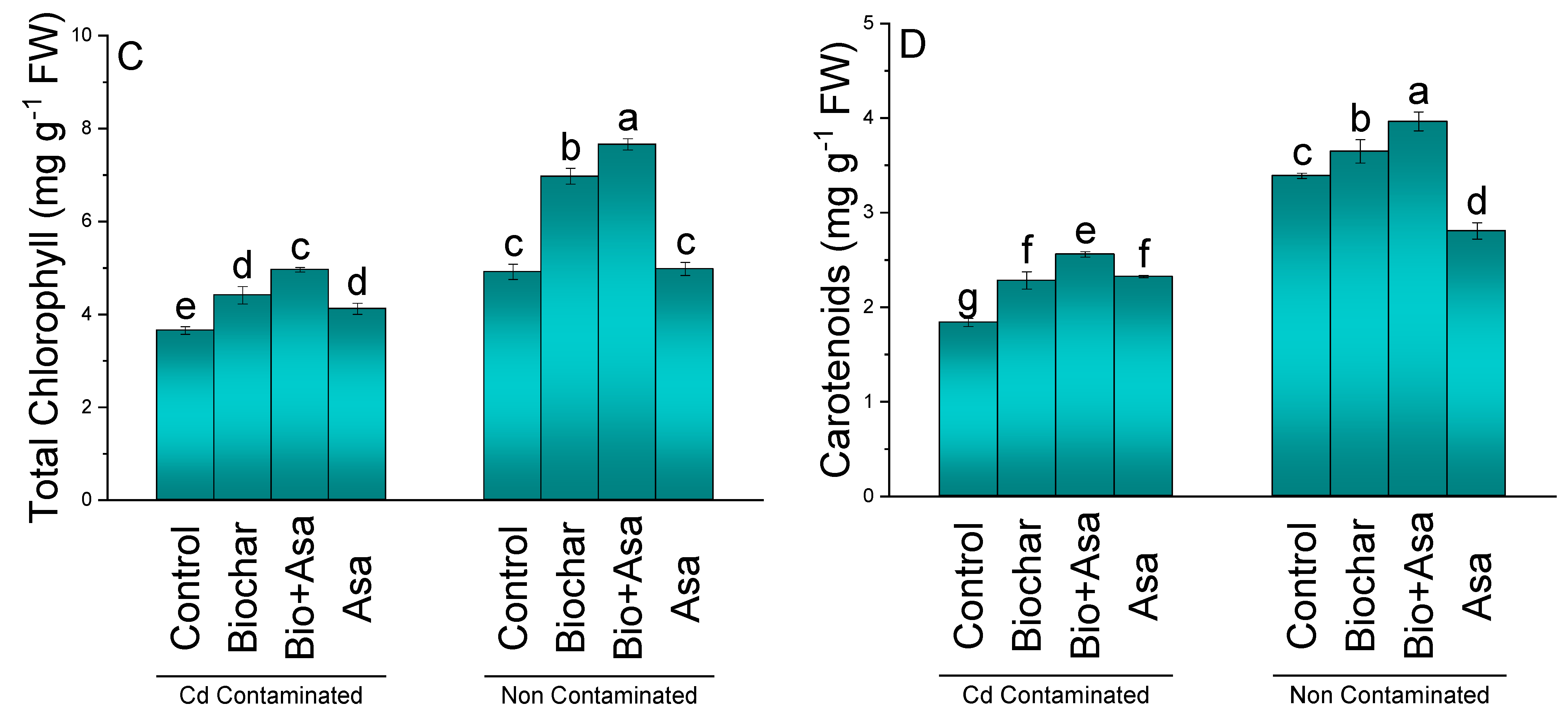
3.2. Chlorophyll Contents
3.3. Gas Exchange Attributes
3.4. SOD, POD, and APX
3.5. Catalase, Phenolics, Flavonoids, and Anthocyanin
3.6. Ascorbic Acid, MDA, H2O2, and Electrolyte Leakage
3.7. Proline, Total Soluble Proteins, Root Cd Concentration, and Shoot Cd Concentration
3.8. Pearson Correlation
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Hassan, A.; Amjad, S.F.; Saleem, M.H.; Yasmin, H.; Imran, M.; Riaz, M.; Ali, Q.; Joyia, F.A.; Mobeen; Ahmed, S.; et al. Foliar application of ascorbic acid enhances salinity stress tolerance in barley (Hordeum vulgare L.) through modulation of morpho-physio-biochemical attributes, ions uptake, osmo-protectants and stress response genes expression. Saudi J. Biol. Sci. 2021, 28, 4276–4290. [Google Scholar] [CrossRef]
- Rizwan, M.; Ali, S.; Qayyum, M.F.; Ibrahim, M.; Rehman, M.Z.U.; Abbas, T.; Ok, Y.S. Mechanisms of biochar-mediated alleviation of toxicity of trace elements in plants: A critical review. Environ. Sci. Pollut. Res. 2015, 23, 2230–2248. [Google Scholar] [CrossRef]
- Ahmed, N.; Shah, A.R.; Danish, S.; Fahad, S.; Ali, M.A.; Zarei, T.; Vranová, V.; Datta, R. Immobilization of Cd in soil by biochar and new emerging chemically produced carbon. J. King Saud Univ. Sci. 2021, 33, 101472. [Google Scholar] [CrossRef]
- Younis, U.; Qayyum, M.F.; Shah, M.H.R.; Danish, S.; Shahzad, A.N.; Malik, S.A.; Mahmood, S. Growth, survival, and heavy metal (Cd and Ni) uptake of spinach(Spinacia oleracea)and fenugreek(Trigonella corniculata)in a biochar-amended sewage-irrigated contaminated soil. J. Plant Nutr. Soil Sci. 2015, 178, 209–217. [Google Scholar] [CrossRef]
- Awad, M.; El-Desoky, M.; Ghallab, A.; Kubes, J.; Abdel-Mawly, S.; Danish, S.; Ratnasekera, D.; Islam, M.S.; Skalicky, M.; Brestic, M.; et al. Ornamental Plant Efficiency for Heavy Metals Phytoextraction from Contaminated Soils Amended with Organic Materials. Molecules 2021, 26, 3360. [Google Scholar] [CrossRef] [PubMed]
- Danish, S.; Zafar-ul-Hye, M.; Fahad, S.; Saud, S.; Brtnicky, M.; Hammerschmiedt, T.; Datta, R. Drought stress alleviation by ACC deaminase producing Achromobacter xylosoxidans and Enterobacter cloacae, with and without timber waste biochar in maize. Sustainability 2020, 12, 6286. [Google Scholar] [CrossRef]
- Younis, U.; Danish, S.; Malik, S.A.; Ahmed, N.; Munir, T.M.; Rasheed, M.K. Role of cotton sticks biochar in immobilization of nickel under induced toxicity condition and growth indices of Trigonella corniculata L. Environ. Sci. Pollut. Res. 2019, 27, 1752–1761. [Google Scholar] [CrossRef] [PubMed]
- Malik, K.; Khan, K.; Rukh, S.; Khan, A.; Akbar, S.; Billah, M.; Bashir, S.; Danish, S.; Alwahibi, M.; Elshikh, M.; et al. Immobilization of Cd, Pb and Zn through Organic Amendments in Wastewater Irrigated Soils. Sustainability 2021, 13, 2392. [Google Scholar] [CrossRef]
- Keller, C.; Rizwan, M.; Davidian, J.-C.; Pokrovsky, O.S.; Bovet, N.; Chaurand, P.; Meunier, J.-D. Effect of silicon on wheat seedlings (Triticum turgidum L.) grown in hydroponics and exposed to 0 to 30 µM Cu. Planta 2014, 241, 847–860. [Google Scholar] [CrossRef] [Green Version]
- Zaheer, I.; Ali, S.; Rizwan, M.; Farid, M.; Shakoor, M.B.; Gill, R.A.; Najeeb, U.; Iqbal, N.; Ahmad, R. Citric acid assisted phytoremediation of copper by Brassica napus L. Ecotoxicol. Environ. Saf. 2015, 120, 310–317. [Google Scholar] [CrossRef]
- Younis, U.; Shah, M.H.R.; Danish, S.; Malik, S.A.; Ameer, A. Biochar role in improving biometric and growth attributes of S. oleracea and T. corniculata under cadmium stress. Int. J. Biosci. 2014, 5, 84–90. [Google Scholar]
- Abid, M.; Danish, S.; Zafar-ul-Hye, M.; Shaaban, M.; Iqbal, M.M.; Rehim, A.; Qayyum, M.F.; Naqqash, M.N. Biochar in-creased photosynthetic and accessory pigments in tomato (Solanum lycopersicum L.) plants by reducing cadmium con-centration under various irrigation waters. Environ. Sci. Pollut. Res. 2017, 24, 22111–22118. [Google Scholar] [CrossRef] [PubMed]
- Zafar-Ul-Hye, M.; Shahjahan, A.; Danish, S.; Abid, M.; Qayyum, M.F. Mitigation of cadmium toxicity induced stress in wheat by ACC-deaminase containing PGPR isolated from cadmium polluted wheat rhizosphere. Pak. J. Bot. 2018, 50, 1727–1734. [Google Scholar]
- Zafar-Ul-Hye, M.; Naeem, M.; Danish, S.; Khan, M.J.; Fahad, S.; Datta, R.; Brtnicky, M.; Kintl, A.; Hussain, G.S.; El-Esawi, M.A. Effect of Cadmium-Tolerant Rhizobacteria on Growth Attributes and Chlorophyll Contents of Bitter Gourd under Cadmium Toxicity. Plants 2020, 9, 1386. [Google Scholar] [CrossRef]
- Shah, A.; Bibi, F.; Hussain, I.; Yasin, N.; Akram, W.; Tahir, M.; Ali, H.; Salem, M.; Siddiqui, M.; Danish, S.; et al. Synergistic Effect of Bacillus thuringiensis IAGS 199 and Putrescine on Alleviating Cadmium-Induced Phytotoxicity in Capsicum annum. Plants 2020, 9, 1512. [Google Scholar] [CrossRef]
- Tamás, L.; Mistrík, I.; Alemayehu, A.; Zelinová, V.; Bočová, B.; Huttová, J. Salicylic acid alleviates cadmium-induced stress responses through the inhibition of Cd-induced auxin-mediated reactive oxygen species production in barley root tips. J. Plant Physiol. 2015, 173, 1–8. [Google Scholar] [CrossRef]
- Mehdizadeh, L.; Farsaraei, S.; Moghaddam, M. Biochar application modified growth and physiological parameters of Ocimum ciliatum L. and reduced human risk assessment under cadmium stress. J. Hazard. Mater. 2020, 409, 124954. [Google Scholar] [CrossRef] [PubMed]
- Zafar-ul-Hye, M.; Tahzeeb-ul-Hassan, M.; Abid, M.; Fahad, S.; Brtnicky, M.; Dokulilova, T.; Datta, R.; Danish, S. Potential role of compost mixed biochar with rhizobacteria in mitigating lead toxicity in spinach. Sci. Rep. 2020, 10, 12159. [Google Scholar] [CrossRef]
- Zafar-Ul-Hye, M.; Tahzeeb-Ul-Hassan, M.; Wahid, A.; Danish, S.; Khan, M.J.; Fahad, S.; Brtnicky, M.; Hussain, G.S.; Battaglia, M.L.; Datta, R. Compost mixed fruits and vegetable waste biochar with ACC deaminase rhizobacteria can minimize lead stress in mint plants. Sci. Rep. 2021, 11, 6606. [Google Scholar] [CrossRef] [PubMed]
- Danish, S.; Tahir, F.A.; Rasheed, M.K.; Ahmad, N.; Ali, M.A.; Kiran, S.; Younis, U.; Irshad, I.; Butt, B. Effect of foliar application of Fe and banana peel waste biochar on growth, chlorophyll content and accessory pigments synthesis in spinach under chromium (IV) toxicity. Open Agric. 2019, 4, 381–390. [Google Scholar] [CrossRef]
- Noreen, S.; Sultan, M.; Akhter, M.S.; Shah, K.H.; Ummara, U.; Manzoor, H.; Ulfat, M.; Alyemeni, M.N.; Ahmad, P. Foliar fertigation of ascorbic acid and zinc improves growth, antioxidant enzyme activity and harvest index in barley (Hordeum vulgare L.) grown under salt stress. Plant Physiol. Biochem. 2020, 158, 244–254. [Google Scholar] [CrossRef]
- Billah, M.; Rohman, M.M.; Hossain, N.; Uddin, M.S. Exogenous ascorbic acid improved tolerance in maize (Zea mays L.) by increasing antioxidant activity under salinity stress. Afr. J. Agric. Res. 2017, 12, 1437–1446. [Google Scholar] [CrossRef] [Green Version]
- Agami, R.A. Applications of ascorbic acid or proline increase resistance to salt stress in barley seedlings. Biol. Plant. 2014, 58, 341–347. [Google Scholar] [CrossRef]
- Alayafi, A.A.M. Exogenous ascorbic acid induces systemic heat stress tolerance in tomato seedlings: Transcriptional regula-tion mechanism. Environ. Sci. Pollut. Res. 2020, 27, 19186–19199. [Google Scholar] [CrossRef]
- Wang, Y.-H.; Zhang, G.; Chen, Y.; Gao, J.; Sun, Y.-R.; Sun, M.-F.; Chen, J.-P. Exogenous application of gibberellic acid and ascorbic acid improved tolerance of okra seedlings to NaCl stress. Acta Physiol. Plant. 2019, 41, 1–10. [Google Scholar] [CrossRef]
- Naheed, S.; Raza, I.; Anwar, M.Z.; Habib, N.; Zahra, N.; Siddiqui, S. Forecasting area and production of barley in Punjab, Pakistan. Pakistan J. Agric. Res. 2015, 28, 304–309. [Google Scholar]
- Eskin, V.; Bougay, V. Pakistan Barley Production Quantity. Available online: https://knoema.com/atlas/Pakistan/topics/Agriculture/Crops-Production-Quantity-tonnes/Barley-production (accessed on 30 July 2021).
- Meints, B.; Hayes, P.M. Breeding naked barley for food, feed, and malt. Plant Breed. Rev. 2019, 43, 95–119. [Google Scholar]
- Fuster, V.; Rydén, L.E.; Cannom, D.S.; Crijns, H.J.; Curtis, A.B.; Ellenbogen, K.A.; Halperin, J.L.; Le Heuzey, J.Y.; Kay, G.N.; Lowe, J.E.; et al. Erratum: ACC/AHA/ESC 2006 guidelines for the management of patients with atrial fibrillation-executive summary. Eur. Heart J. 2007, 28, 2046. [Google Scholar] [CrossRef] [Green Version]
- Awashthi, S.K. Prevention of Food Adultration; Ashoka Law House: New Delhi, India, 2000. [Google Scholar]
- WHO/FAO. Report of the Thirty Eight Session of the Codex Committee on Food Hygiene; Joint FAO/WHO Food Standards Programme: Rome, Italy, 2007. [Google Scholar]
- Lutts, S.; Kinet, J.M.; Bouharmont, J. NaCl-induced senescence in leaves of rice (Oryza sativa L.) cultivars differing in salinity resistance. Ann. Bot. 1996, 78, 389–398. [Google Scholar] [CrossRef]
- Heath, R.L.; Packer, L. Photoperoxidation in isolated chloroplasts. Arch. Biochem. Biophys. 1968, 125, 189–198. [Google Scholar] [CrossRef]
- Jana, S.; Choudhuri, M.A. Glycolate metabolism of three submersed aquatic angiosperms during ageing. Aquat. Bot. 1982, 12, 345–354. [Google Scholar] [CrossRef]
- Beauchamp, C.; Fridovich, I. Superoxide dismutase: Improved assays and an assay applicable to acrylamide gels. Anal. Biochem. 1971, 44, 276–287. [Google Scholar] [CrossRef]
- Bradford, M.M. A rapid and sensitive method for the quantitation of microgram quantities of protein using the principle of protein dye binding. Anal. Biochem. 1976, 72, 248–254. [Google Scholar] [CrossRef]
- E Lewis, C.; Walker, J.R.L.; E Lancaster, J.; Sutton, K.H. Determination of anthocyanins, flavonoids and phenolic acids in potatoes. I: Coloured cultivars of Solanum tuberosum L. J. Sci. Food Agric. 1998, 77, 45–57. [Google Scholar] [CrossRef]
- Bray, H.G.; Thorpe, W.V. Analysis of Phenolic Compounds of Interest in Metabolism. Methods Biochem. Anal. 2006, 1, 27–52. [Google Scholar] [CrossRef]
- Pękal, A.; Pyrzynska, K. Evaluation of Aluminium Complexation Reaction for Flavonoid Content Assay. Food Anal. Methods 2014, 7, 1776–1782. [Google Scholar] [CrossRef] [Green Version]
- Dubois, M.; Gilles, K.A.; Hamilton, J.K.; Rebers, P.A.; Smith, F. Colorimetric Method for Determination of Sugars and Related Substances. Anal. Chem. 1956, 28, 350–356. [Google Scholar] [CrossRef]
- Azuma, K.; Nakayama, M.; Koshioka, M.; Ippoushi, K.; Yamaguchi, Y.; Kohata, K.; Yamauchi, Y.; Ito, H.; Higashio, H. Phenolic Antioxidants from the Leaves of Corchorus olitorius L. J. Agric. Food Chem. 1999, 47, 3963–3966. [Google Scholar] [CrossRef]
- Bates, L.S.; Waldren, R.P.; Teare, I.D. Rapid determination of free proline for water-stress studies. Plant Soil 1973, 39, 205–207. [Google Scholar] [CrossRef]
- Miller, O. Nitric-Perchloric Acid Wet Digestion In an Open Vessel. In Reference Methods for Plant Analysis; Kalra, Y., Ed.; CRC Press: Washington, DC, USA, 1998; pp. 57–62. [Google Scholar]
- Suarez, D.L. Beryllium, Magnesium, Calcium, Strontium, and Barium. In Methods of Soil Analysis: Part 3 Chemical Methods, 5.3; Sparks, D.L., Page, A.L., Helmke, P.A., Loeppert, R.H., Soltanpour, P.N., Tabatabai, M.A., Johnston, C.T., Sumner, M.E., Eds.; John Wiley & Sons, Ltd.: Hoboken, NJ, USA, 2018; pp. 575–601. [Google Scholar]
- Steel, R.G.; Torrie, J.H.; Dickey, D.A. Principles and Procedures of Statistics: A Biometrical Approach, 3rd ed.; McGraw Hill Book International Co.: Singapore, 1997. [Google Scholar]
- OriginLab Corporation. OriginPro; OriginLab: Northampton, MA, USA, 2021. [Google Scholar]
- Younis, U.; Malik, S.A.; Rizwan, M.; Qayyum, M.F.; Ok, Y.S.; Shah, M.H.R.; Rehman, R.A.; Ahmad, N. Biochar enhances the cadmium tolerance in spinach (Spinacia oleracea) through modification of Cd uptake and physiological and biochemical attributes. Environ. Sci. Pollut. Res. 2016, 23, 21385–21394. [Google Scholar] [CrossRef]
- Rizwan, M.; Ali, S.; Adrees, M.; Rizvi, H.; Zia-Ur-Rehman, M.; Hannan, F.; Qayyum, M.F.; Hafeez, F.; Ok, Y.S. Cadmium stress in rice: Toxic effects, tolerance mechanisms, and management: A critical review. Environ. Sci. Pollut. Res. 2016, 23, 17859–17879. [Google Scholar] [CrossRef] [PubMed]
- Siddiqui, M.H.; Alamri, S.; Al-Khaishany, M.Y.; Khan, M.N.; Al-Amri, A.; Ali, H.M.; Alaraidh, I.A.; Alsahli, A.A. Exogenous melatonin counteracts NaCl-induced damage by regulating the antioxidant system, proline and carbohydrates metabolism in tomato seedlings. Int. J. Mol. Sci. 2019, 20, 353. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Barzegar, T.; Fateh, M.; Razavi, F. Enhancement of postharvest sensory quality and antioxidant capacity of sweet pepper fruits by foliar applying calcium lactate and ascorbic acid. Sci. Hortic. 2018, 241, 293–303. [Google Scholar] [CrossRef]
- Zhou, F.; Wang, H.; Fang, S.; Zhang, W.; Qiu, R. Pb(II), Cr(VI) and atrazine sorption behavior on sludge-derived biochar: Role of humic acids. Environ. Sci. Pollut. Res. 2015, 22, 16031–16039. [Google Scholar] [CrossRef]
- Puga, A.; Abreu, C.; Melo, L.; Beesley, L. Biochar application to a contaminated soil reduces the availability and plant uptake of zinc, lead and cadmium. J. Environ. Manag. 2015, 159, 86–93. [Google Scholar] [CrossRef]
- Khan, S.; Reid, B.J.; Li, G.; Zhu, Y.-G. Application of biochar to soil reduces cancer risk via rice consumption: A case study in Miaoqian village, Longyan, China. Environ. Int. 2014, 68, 154–161. [Google Scholar] [CrossRef] [Green Version]
- Kamal, M.A.; Saleem, M.F.; Shahid, M.; Awais, M.; Khan, H.Z.; Ahmed, K. Ascorbic acid triggered physiochemical trans-formations at different phenological stages of heat-stressed Bt cotton. J. Agron. Crop Sci. 2017, 203, 323–331. [Google Scholar] [CrossRef]
- Naz, H.; Akram, N.A.; Ashraf, M. Impact of ascorbic acid on growth and some physiological attributes of cucumber (Cu-cumis sativus) plants under water-deficit conditions. Pak. J. Bot. 2016, 48, 877–883. [Google Scholar]
- Bose, J.; Rodrigo-Moreno, A.; Shabala, S. ROS homeostasis in halophytes in the context of salinity stress tolerance. J. Exp. Bot. 2013, 65, 1241–1257. [Google Scholar] [CrossRef]
- Noreen, S.; Faiz, S.; Akhter, M.S.; Shah, K.H. Influence of Foliar Application of Osmoprotectants to Ameliorate Salt Stress in Sunflower (Helianthus annuus L.). Sarhad J. Agric. 2019, 35, 1316–1325. [Google Scholar] [CrossRef]
- Noreen, S.; Shaheen, A.; Shah, K.H.; Ammara, U. Effects of Aerial Application of Salicylic Acid on Growth, Pigment Concentration, Ions Uptake and Mitigation of Salinity Stress in Two Varieties of Wheat (Triticum aestivum L.). Pak. J. Life Soc. Sci. 2019, 17, 78–85. [Google Scholar]
- Zelinová, V.; Mistrík, I.; Pavlovkin, J.; Tamás, L. Glutathione peroxidase expression and activity in barley root tip after short-term treatment with cadmium, hydrogen peroxide and t-butyl hydroperoxide. Protoplasma 2013, 250, 1057–1065. [Google Scholar] [CrossRef]








Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Yaseen, S.; Amjad, S.F.; Mansoora, N.; Kausar, S.; Shahid, H.; Alamri, S.A.M.; Alrumman, S.A.; Eid, E.M.; Ansari, M.J.; Danish, S.; et al. Supplemental Effects of Biochar and Foliar Application of Ascorbic Acid on Physio-Biochemical Attributes of Barley (Hordeum vulgare L.) under Cadmium-Contaminated Soil. Sustainability 2021, 13, 9128. https://doi.org/10.3390/su13169128
Yaseen S, Amjad SF, Mansoora N, Kausar S, Shahid H, Alamri SAM, Alrumman SA, Eid EM, Ansari MJ, Danish S, et al. Supplemental Effects of Biochar and Foliar Application of Ascorbic Acid on Physio-Biochemical Attributes of Barley (Hordeum vulgare L.) under Cadmium-Contaminated Soil. Sustainability. 2021; 13(16):9128. https://doi.org/10.3390/su13169128
Chicago/Turabian StyleYaseen, Samia, Syeda Fasiha Amjad, Nida Mansoora, Shameem Kausar, Huma Shahid, Saad A. M. Alamri, Sulaiman A. Alrumman, Ebrahem M. Eid, Mohammad Javed Ansari, Subhan Danish, and et al. 2021. "Supplemental Effects of Biochar and Foliar Application of Ascorbic Acid on Physio-Biochemical Attributes of Barley (Hordeum vulgare L.) under Cadmium-Contaminated Soil" Sustainability 13, no. 16: 9128. https://doi.org/10.3390/su13169128









