Potential Bioinoculants for Sustainable Agriculture Prospected from Ferruginous Caves of the Iron Quadrangle/Brazil
Abstract
:1. Introduction
2. Materials and Methods
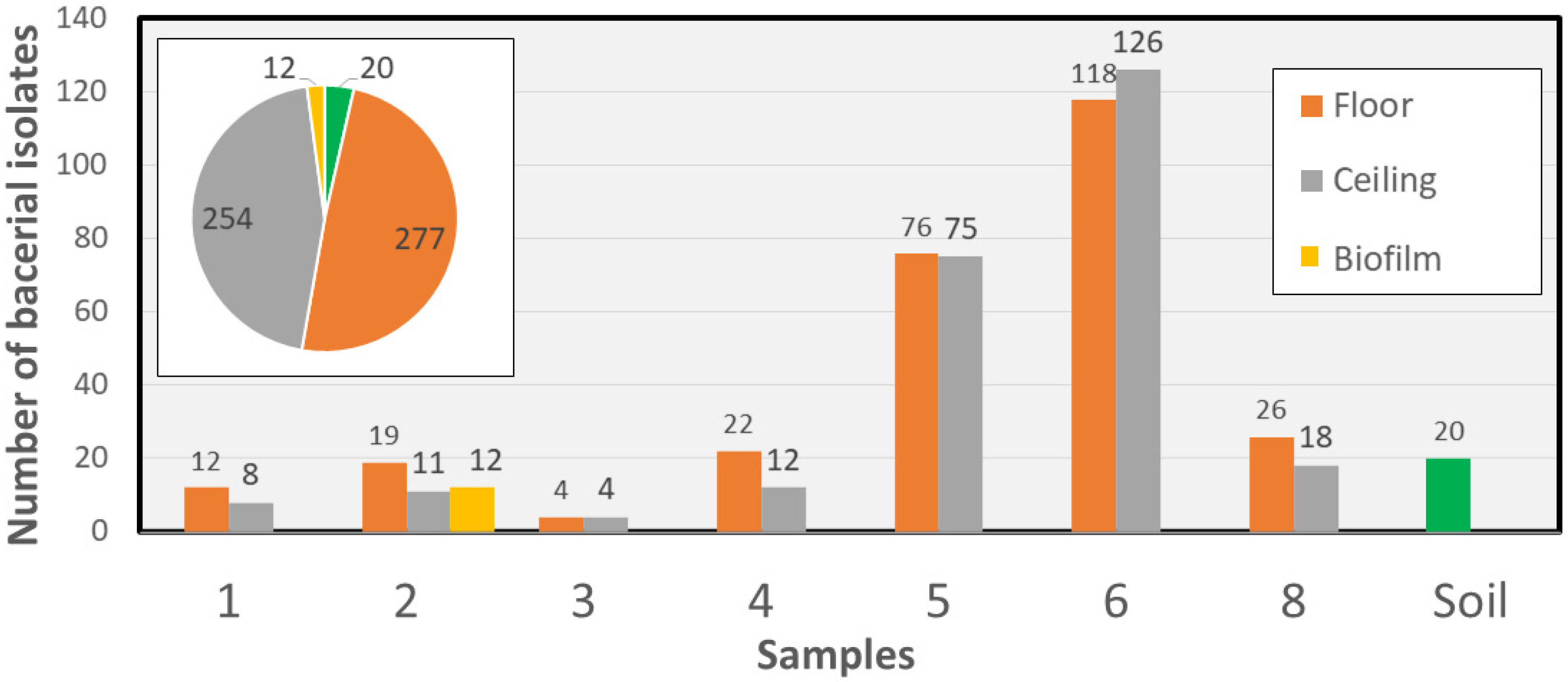
2.1. Cave Selection and Collection of Sample Material
2.2. Isolation and Preservation of Culturable Bacterial Isolates
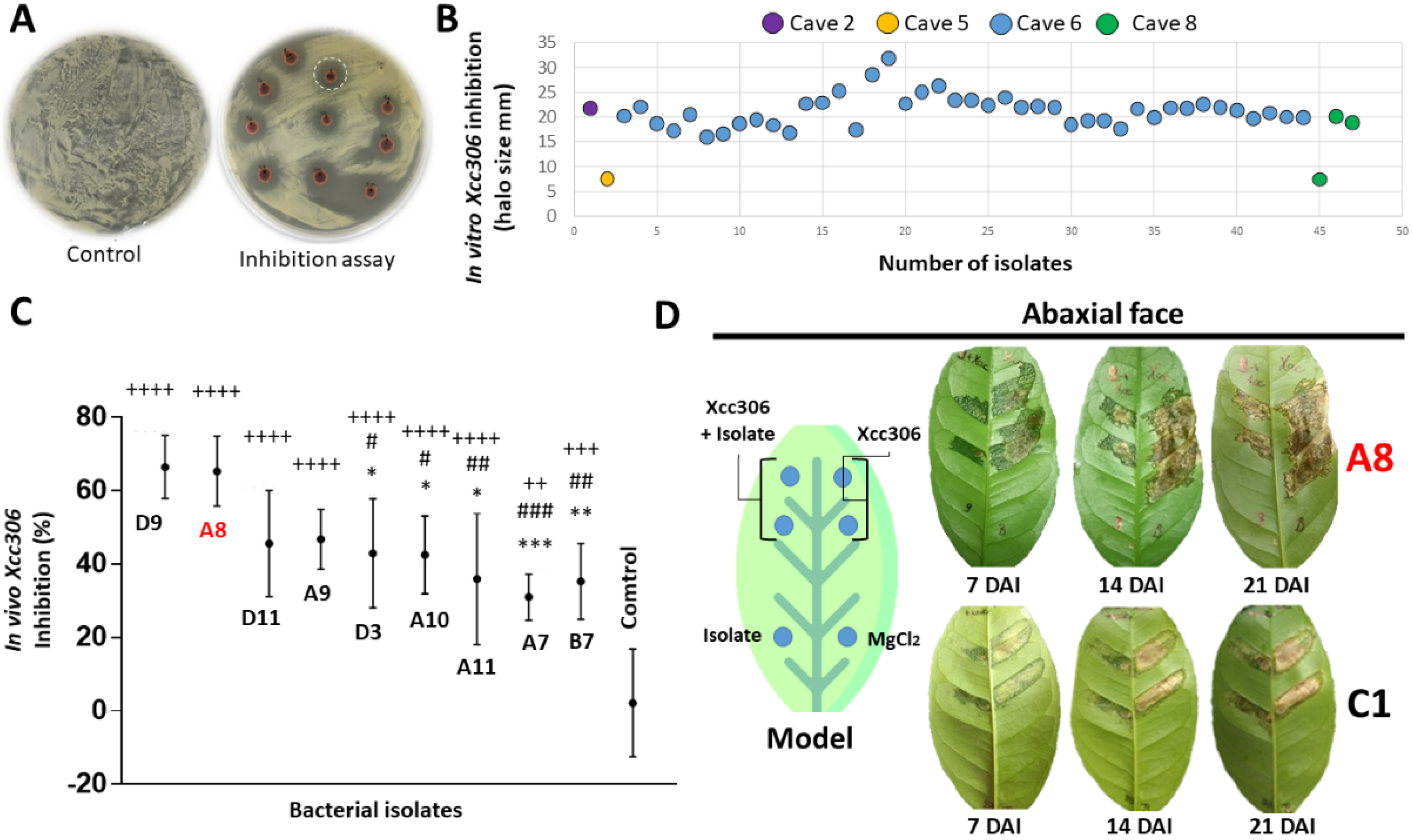
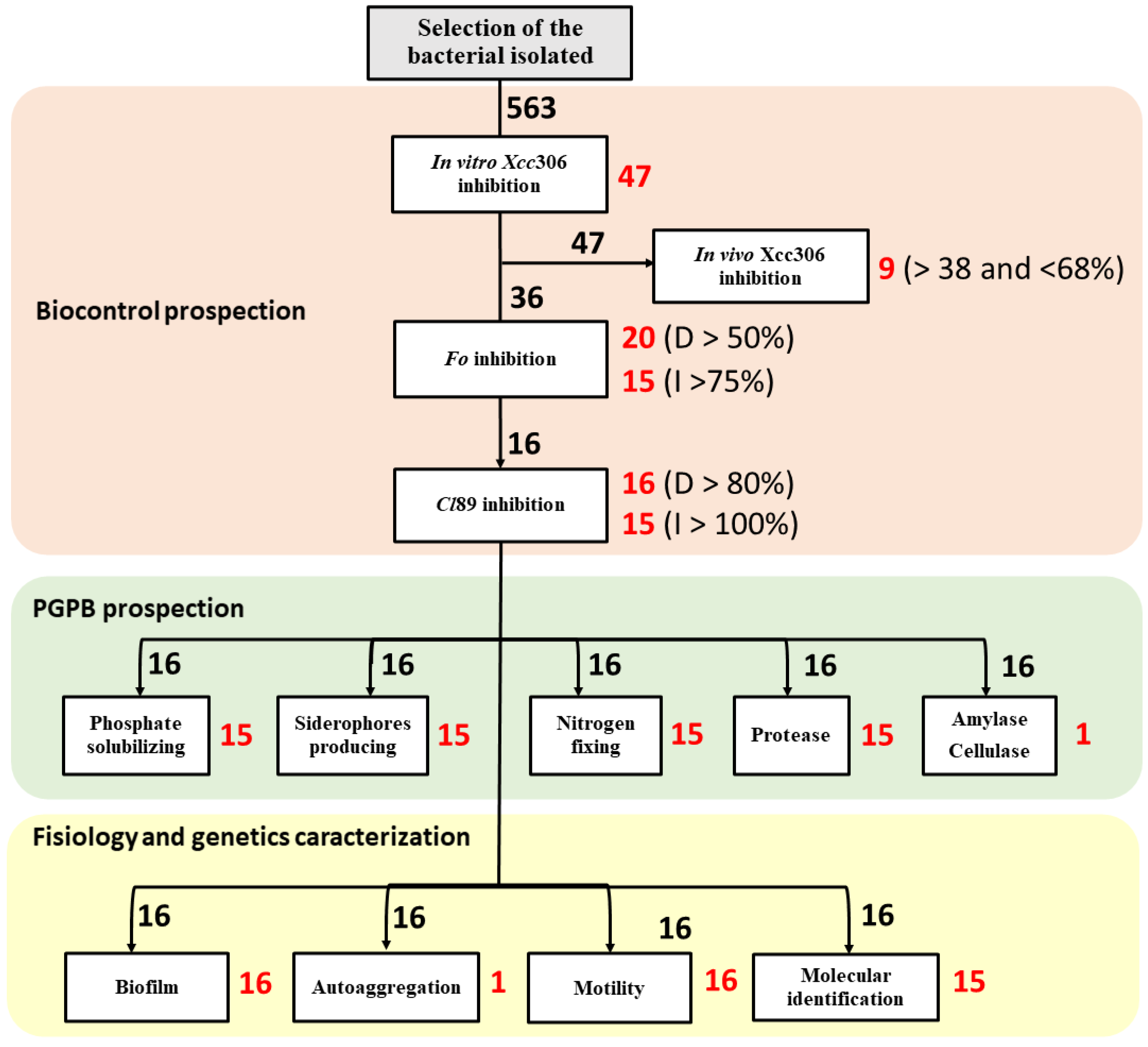
2.3. In Vitro Inhibition Assays against Xanthomonas citri subsp. citri (Xcc306)
2.4. In Vivo Inhibition Assay against Xcc306
2.5. In Vitro Inhibition Test against Fusarium oxysporum (Fo)
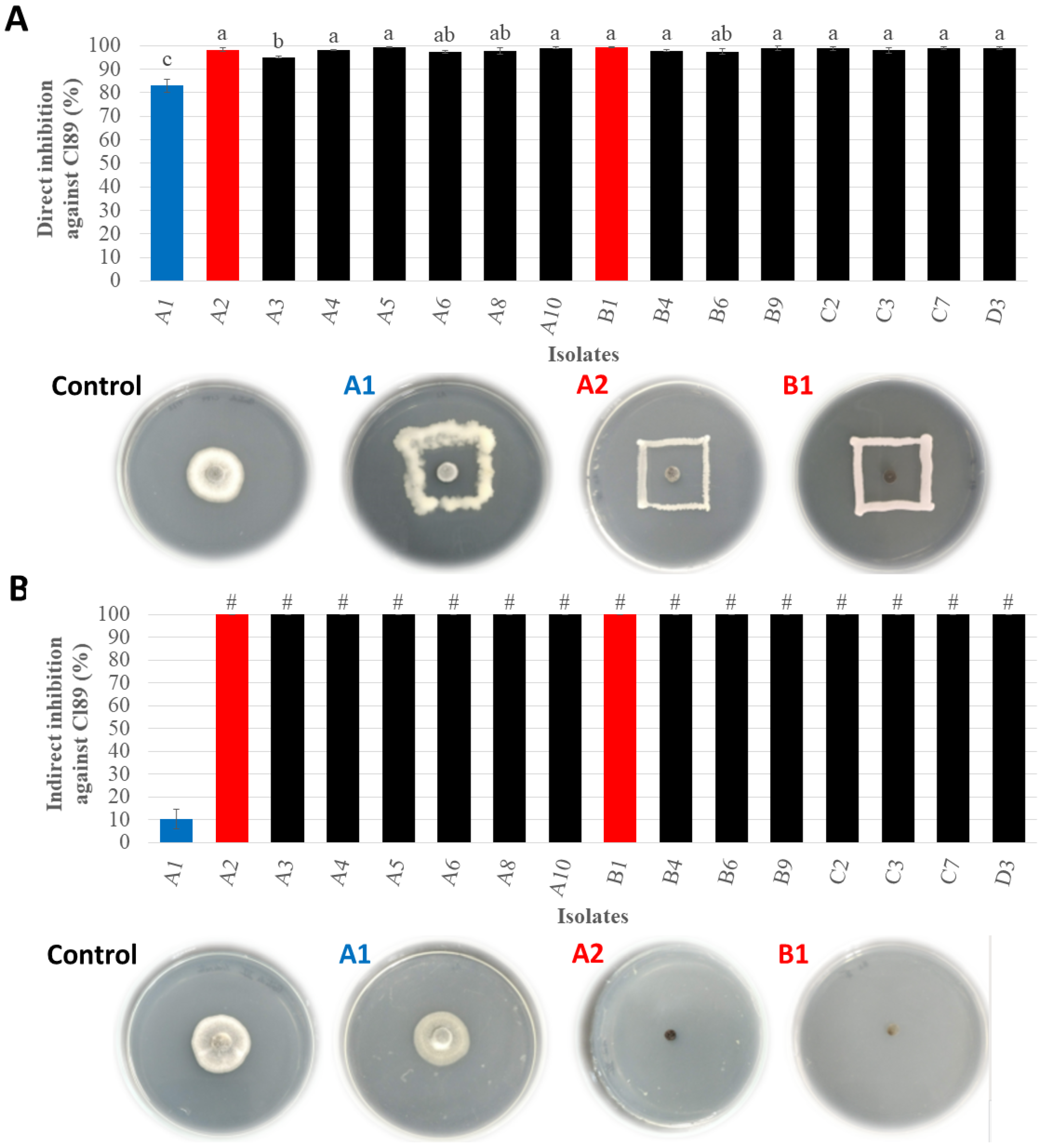
2.6. In Vitro Inhibition Assay against Colletotrichum lindemuthianum Strain 89 (Cl89)
2.7. Calculation of the Inhibition Rate of Phytopathogenic Fungi
2.8. Plant Growth Promotion Assays
2.9. Calcium Phosphate Solubilization Assay
2.10. Siderophore Production Assay
2.11. Nitrogen Fixation Assay
2.12. Hydrolase Production Assay
2.13. Bacterial Motility Assay
2.14. Test of Biofilm Production and Bacterial Self-Aggregation Capacity
2.15. Molecular Identification of Potential Bioinoculants
2.16. Statistical Analysis
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Roeser, H.; Roeser, P. O Quadrilátero Ferrífero-MG, Brasil: Aspectos sobre sua história, seus recursos minerais e problemas ambientais relacionados. Rev. Geonomos 2013, 18, 33–37. [Google Scholar] [CrossRef] [Green Version]
- Azevedo, Ú.; Machado, M. Geological and mining heritage of iron quadrangle, Minas Gerais—Characterization and strategies for education and geotourism. Bol. Parana. Geosci. 2013, 70, 120–136. [Google Scholar]
- Jacobi, C.M.; Carmo, F.F. The contribution of ironstone outcrops to plant diversity in the Iron Quadrangle, a threatened Brazilian landscape. Ambio 2008, 37, 324–326. [Google Scholar] [CrossRef]
- Fernandes, G.W.; Goulart, F.F.; Ranieri, B.D.; Coelho, M.S.; Dales, K.; Boesche, N.; Bustamante, M.; Carvalho, F.A.; Carvalho, D.C.; Dirzo, R.; et al. Deep into the mud: Ecological and socio-economic impacts of the dam breach in Mariana, Brazil. Nat. Conserv. 2016, 14, 35–45. [Google Scholar] [CrossRef]
- Carmo, F.F.D.; Kamino, L.H.Y.; Junior, R.T.; Campos, I.C.D.; Carmo, F.F.D.; Silvino, G.; Castro, K.J.D.S.X.D.; Mauro, M.L.; Rodrigues, N.U.A.; Miranda, M.P.D.S.; et al. Fundão tailings dam failures: The environment tragedy of the largest technological disaster of Brazilian mining in global context. Perspect. Ecol. Conserv. 2017, 15, 145–151. [Google Scholar] [CrossRef]
- Cionek, V.M.; Alves, G.H.Z.; Tófoli, R.M.; Rodrigues-Filho, J.L.; Dias, R.M. Brazil in the mud again: Lessons not learned from Mariana dam collapse. Biodivers. Conserv. 2019, 28, 1935–1938. [Google Scholar] [CrossRef]
- Pereira, M.C.; Ruchkys, Ú.; Pereira, E.O. Panorama do Patrimônio Espeleológico do Quadrilátero Ferrífero. In Patrimônio Espeleológico em Rochas Ferruginosas: Propostas Para sua Conservação no Quadrilátero Ferrífero, Minas Gerais; Ruchkys, Ú., Travassos, L.E.P., Rasteiro, M.A., Faria, L.E., Eds.; Sociedade Brasileira de Espeleologia: Campinas, Brasil, 2015; Volume 1, pp. 276–287. [Google Scholar]
- CECAV. Anuário Estatístico do Patrimônio Espeleológico Brasileiro. Available online: http://www.icmbio.gov.br/cecav/ (accessed on 4 July 2021).
- Auler, A.S.; Piló, L.B. Aspectos morfológicos e evolução de cavernas em minério de ferro no Quadrilátero Ferrífero, Minas Gerais. In Proceedings of the Encontro Brasileiro de Estudos do Instituto de Geociências; IG-USP: São Paulo, Brazil, 2007; p. 8. [Google Scholar]
- Simmons, G.C. Canga Caves in the Quadrilátero Ferrífero, Minas Gerais, Brazil. Bull. Natl. Speleol. Soc. 1963, 22, 66–72. [Google Scholar]
- Culver, D.C. Cave Life: Evolution and Ecology; Harvard University Press: Cambridge, MA, USA, 1982; p. 189. [Google Scholar]
- Davila-Ramos, S.; Castelan-Sanchez, H.G.; Martinez-Avila, L.; Sanchez-Carbente, M.D.R.; Peralta, R.; Hernandez-Mendoza, A.; Dobson, A.D.W.; Gonzalez, R.A.; Pastor, N.; Batista-Garcia, R.A. A Review on Viral Metagenomics in Extreme Environments. Front. Microbiol. 2019, 10, 2403. [Google Scholar] [CrossRef] [Green Version]
- Orellana, R.; Macaya, C.; Bravo, G.; Dorochesi, F.; Cumsille, A.; Valencia, R.; Rojas, C.; Seeger, M. Living at the Frontiers of Life: Extremophiles in Chile and Their Potential for Bioremediation. Front. Microbiol. 2018, 9, 2309. [Google Scholar] [CrossRef]
- Nunez-Montero, K.; Barrientos, L. Advances in Antarctic Research for Antimicrobial Discovery: A Comprehensive Narrative Review of Bacteria from Antarctic Environments as Potential Sources of Novel Antibiotic Compounds Against Human Pathogens and Microorganisms of Industrial Importance. Antibiotics 2018, 7, 90. [Google Scholar] [CrossRef] [Green Version]
- Orcutt, B.N.; Larowe, D.E.; Biddle, J.F.; Colwell, F.S.; Glazer, B.T.; Reese, B.K.; Kirkpatrick, J.B.; Lapham, L.L.; Mills, H.J.; Sylvan, J.B.; et al. Microbial activity in the marine deep biosphere: Progress and prospects. Front. Microbiol. 2013, 4, 189. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Rooney, D.; Hutchens, E.; Clipson, N.; Baldini, J.; McDermott, F. Microbial Community Diversity of Moonmilk Deposits at Ballynamintra Cave, Co. Waterford, Ireland. Microb. Ecol. 2010, 60, 753–761. [Google Scholar] [CrossRef] [PubMed]
- Yasir, M. Analysis of bacterial communities and characterization of antimicrobial strains from cave microbiota. Braz. J. Microbiol. 2018, 49, 248–257. [Google Scholar] [CrossRef] [PubMed]
- Gabriel, C.; Northup, D. Microbial Ecology: Caves as an Extreme Habitat; Springer: New York, NY, USA, 2013; Volume 1, pp. 85–108. [Google Scholar]
- Hershey, O.S.; Barton, H.A. The Microbial Diversity of Caves. In Cave Ecology; Moldovan, O.T., Kováč, Ľ., Halse, S., Eds.; Springer International Publishing: Cham, Switzerland, 2018; pp. 69–90. [Google Scholar]
- Rangseekaew, P.; Pathomaree, W. Cave Actinobacteria as Producers of Bioactive Metabolites. Front. Microbiol. 2019, 10, 387. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Cheeptham, N.; Kay, S.; Pathom-aree, W. Screening of Volcanic Cave Actinomycetes for Antimicrobial Activity Against Paenibacillus larvae, A Honey Bee Pathogen. Chiang Mai J. Sci. 2012, 40, 26. [Google Scholar]
- Riquelme, C.; Enes Dapkevicius, M.L.; Miller, A.Z.; Charlop-Powers, Z.; Brady, S.; Mason, C.; Cheeptham, N. Biotechnological potential of Actinobacteria from Canadian and Azorean volcanic caves. Appl. Microbiol. Biotechnol. 2017, 101, 843–857. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ghosh, S.; Kuisiene, N.; Cheeptham, N. The cave microbiome as a source for drug discovery: Reality or pipe dream? Biochem. Pharmacol. 2017, 134, 18–34. [Google Scholar] [CrossRef]
- Pawlowski, A.C.; Wang, W.; Koteva, K.; Barton, H.A.; McArthur, A.G.; Wright, G.D. A diverse intrinsic antibiotic resistome from a cave bacterium. Nat. Commun. 2016, 7, 13803. [Google Scholar] [CrossRef] [Green Version]
- Bhullar, K.; Waglechner, N.; Pawlowski, A.; Koteva, K.; Banks, E.D.; Johnston, M.D.; Barton, H.A.; Wright, G.D. Antibiotic Resistance Is Prevalent in an Isolated Cave Microbiome. PLoS ONE 2012, 7, e34953. [Google Scholar] [CrossRef]
- Montano, E.; Henderson, L. Studies of Antibiotic Production by Cave Bacteria. In Cave Microbiomes: A Novel Resource for Drug Discovery; SpringerBriefs in Microbiology; Cheeptham, N., Ed.; Springer: New York, NY, USA, 2013; pp. 109–130. [Google Scholar] [CrossRef]
- Venkadesaperumal, G.; Amaresan, N.; Kumar, K. Plant growth promoting capability and genetic diversity of bacteria isolated from mud volcano and lime cave of Andaman and Nicobar Islands. Braz. J. Microbiol. 2014, 45, 1271–1281. [Google Scholar] [CrossRef] [Green Version]
- Sessitsch, A.; Brader, G.; Pfaffenbichler, N.; Gusenbauer, D.; Mitter, B. The contribution of plant microbiota to economy growth. Microb. Biotechnol. 2018, 11, 801–805. [Google Scholar] [CrossRef]
- Diguță, C.F.; Nițoi, G.D.; Matei, F.; Luță, G.; Cornea, C.P. The Biotechnological Potential of Pediococcus spp. Isolated from Kombucha Microbial Consortium. Foods 2020, 9, 1780. [Google Scholar] [CrossRef]
- Ramakrishna, W.; Yadav, R.; Li, K. Plant growth promoting bacteria in agriculture: Two sides of a coin. Appl. Soil Ecol. 2019, 138, 10–18. [Google Scholar] [CrossRef]
- Compant, S.; Duffy, B.; Nowak, J.; Clement, C.; Barka, E.A. Use of plant growth-promoting bacteria for biocontrol of plant diseases: Principles, mechanisms of action, and future prospects. Appl. Environ. Microbiol. 2005, 71, 4951–4959. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lemes, C.G.C. Diversidade Metagenômica e Potencial Biotecnológico de Cavernas de Canga do Quadrilátero Ferrífero. Master’s Thesis, Universidade Federal de Ouro Preto, Ouro Preto, Brazil, 2018. [Google Scholar]
- Felestrino, E.B.; Santiago, I.F.; Freitas, L.D.; Rosa, L.H.; Ribeiro, S.P.; Moreira, L.M. Plant Growth Promoting Bacteria Associated with Langsdorffia hypogaea-Rhizosphere-Host Biological Interface: A Neglected Model of Bacterial Prospection. Front. Microbiol. 2017, 8, 172. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Caneschi, W.L.; Felestrino, É.B.; Fonseca, N.P.; Villa, M.M.; Lemes, C.G.C.; Cordeiro, I.F.; Assis, R.D.A.B.; Sanchez, A.B.; Vieira, I.T.; Kamino, L.H.Y.; et al. Brazilian Ironstone Plant Communities as Reservoirs of Culturable Bacteria With Diverse Biotechnological Potential. Front. Microbiol. 2018, 9, 1638. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Caicedo, J.C.; Villamizar, S.; Ferro, M.I.T.; Kupper, K.C.; Ferro, J.A. Bacteria from the citrus phylloplane can disrupt cell–cell signalling in Xanthomonas citri and reduce citrus canker disease severity. Plant Pathol. 2016, 65, 782–791. [Google Scholar] [CrossRef]
- Schneider, C.A.; Rasband, W.S.; Eliceiri, K.W. NIH Image to ImageJ: 25 years of image analysis. Nat. Methods 2012, 9, 671–675. [Google Scholar] [CrossRef]
- Cruz, D.R.; Leandro, L.F.S.; Munkvold, G.P. Effects of Temperature and pH on Fusarium oxysporum and Soybean Seedling Disease. Plant Dis. 2019, 103, 3234–3243. [Google Scholar] [CrossRef] [PubMed]
- Nourozian, J.; Etebarian, H.R.; Khodakaramian, G. Biological control of Fusarium graminearum on wheat by antagonistic bacteria. Songklanakarin J. Sci. Technol. 2006, 28, 29–38. [Google Scholar]
- Tozze-Júnior, H.J.; Firmino, A.C.; Fischer, I.H.; Furtado, E.L.; Júnior, N.S.M. Caracterização de isolados de Colletotrichum spp. associados às frutíferas no Estado de São Paulo. Summa Phytopathol. 2015, 41, 270–280. [Google Scholar] [CrossRef]
- Nautiyal, C.S. An efficient microbiological growth medium for screening phosphate solubilizing microorganisms. FEMS Microbiol. Lett. 1999, 170, 265–270. [Google Scholar] [CrossRef] [PubMed]
- Nguyen, C.; Yan, W.; Tacon, F.L.; Lapeyrie, F. Genetic variability of phosphate solubilizing activity by monocaryotic and dicaryotic mycelia of the ectomycorrhizal fungus Laccaria bicolor (Maire) P.D. Orton. Plant Soil 1992, 143, 193–199. [Google Scholar] [CrossRef]
- Schwyn, B.; Neilands, J.B. Universal chemical assay for the detection and determination of siderophores. Anal. Biochem. 1987, 160, 47–56. [Google Scholar] [CrossRef]
- Estrada-De Los Santos, P.; Bustillos-Cristales, R.; Caballero-Mellado, J. Burkholderia, a genus rich in plant-associated nitrogen fixers with wide environmental and geographic distribution. Appl. Environ. Microbiol. 2001, 67, 2790–2798. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Strauss, M.L.; Jolly, N.P.; Lambrechts, M.G.; van Rensburg, P. Screening for the production of extracellular hydrolytic enzymes by non-Saccharomyces wine yeasts. J. Appl. Microbiol. 2001, 91, 182–190. [Google Scholar] [CrossRef] [PubMed]
- Menegatto, M.B.D.S. Avaliação do Potencial Celulolítico e Fermentativo de Bactérias do Gênero Clostridium e da Microbiota Autóctone na Fermentação do Bagaço de Cana Bruto. Master’s Thesis, Universidade Federal de Ouro Preto, Ouro Preto, Brazil, 2019. [Google Scholar]
- Hankin, L.; Anagnostakis, S.L. The Use of Solid Media for Detection of Enzyme Production by Fungi. Mycologia 1975, 67, 597–607. [Google Scholar] [CrossRef]
- Montenegro, S.; Araújo, J.; Stamford, N. Atividade enzimática de microrganismos isolados do jacatupé (Pachyrhizus erosus L. Urban). Ciênc. Tecnol. Aliment. 1998, 18, 382–385. [Google Scholar] [CrossRef]
- Lealem, F.; Gashe, B.A. Amylase production by a Gram-positive bacterium isolated from fermenting tef (Eragrostis tef). J. Appl. Bacteriol. 1994, 77, 348–352. [Google Scholar] [CrossRef]
- Park, S.Y.; Pontes, M.H.; Groisman, E.A. Flagella-independent surface motility in Salmonella enterica serovar Typhimurium. Proc. Natl. Acad. Sci. USA 2015, 112, 1850–1855. [Google Scholar] [CrossRef] [Green Version]
- O’Toole, G.A. Microtiter dish biofilm formation assay. J. Vis. Exp. 2011, e2437. [Google Scholar] [CrossRef] [PubMed]
- Muzzolón, J. Uso de Bacterias Lácticas y/o Levaduras en la Prevención de Aflatoxicosis en Animales de Compañía. Monografia. Universidad Nacional de Río Cuarto, Río Cuarto, Brazil, 2010. [Google Scholar]
- Lima, C.E. Potencial Probiótico de Bactérias Ácido-Láticas Isoladas de Ração de Peixe. Master’s Thesis, Universidade Federal do Piauí, Teresina, Brazil, 2016. [Google Scholar]
- Fouhy, F.; Clooney, A.G.; Stanton, C.; Claesson, M.J.; Cotter, P.D. 16S rRNA gene sequencing of mock microbial populations- impact of DNA extraction method, primer choice and sequencing platform. BMC Microbiol. 2016, 16, 123. [Google Scholar] [CrossRef] [PubMed]
- Hall, T.A. BioEdit: A user-friendly biological sequence alignment editor and analysis program for Windows 95/98/NT. Nucl. Acids. Symp. Ser. 1999, 41, 95–98. [Google Scholar]
- Altschul, S.F.; Gish, W.; Miller, W.; Myers, E.W.; Lipman, D.J. Basic local alignment search tool. J. Mol. Biol. 1990, 215, 403–410. [Google Scholar] [CrossRef]
- Sas, I. SAS® 9.4 ODS Graphics: Procedures Guide, 4th ed.; SAS Institute Inc.: Cary, NC, USA, 2014. [Google Scholar]
- Bank, T.W. Agriculture and Food. Available online: https://www.worldbank.org/en/topic/agriculture/overview (accessed on 30 March 2021).
- CEPEA; CNA. PIB do Agronegócio Alcança Participação de 26.6% no PIB Brasileiro em 2020; Centro de Estudos Avançados em Economia Aplicada (CEPEA), Confederação da Agricultura e Pecuária do Brasil (CNA): Piracicaba, Brazil, 2021; p. 18. [Google Scholar]
- Shang, Y.; Hasan, M.K.; Ahammed, G.J.; Li, M.; Yin, H.; Zhou, J. Applications of Nanotechnology in Plant Growth and Crop Protection: A Review. Molecules 2019, 24, 2558. [Google Scholar] [CrossRef] [Green Version]
- Chen, P.; Du, Q.; Liu, X.; Zhou, L.; Hussain, S.; Lei, L.; Song, C.; Wang, X.; Liu, W.; Yang, F.; et al. Effects of reduced nitrogen inputs on crop yield and nitrogen use efficiency in a long-term maize-soybean relay strip intercropping system. PLoS ONE 2017, 12, e0184503. [Google Scholar] [CrossRef] [Green Version]
- Cooper, J.; Dobson, H. The benefits of pesticides to mankind and the environment. Crop. Prot. 2007, 26, 1337–1348. [Google Scholar] [CrossRef]
- Rosell, G.; Quero, C.; Coll, J.; Guerrero, A. Biorational insecticides in pest management. J. Pestic. Sci. 2008, 33, 103–121. [Google Scholar] [CrossRef] [Green Version]
- Liu, Y.; Li, S.; Ni, Z.; Qu, M.; Zhong, D.; Ye, C.; Tang, F. Pesticides in persimmons, jujubes and soil from China: Residue levels, risk assessment and relationship between fruits and soils. Sci. Total Environ. 2016, 542, 620–628. [Google Scholar] [CrossRef]
- Tilman, D.; Cassman, K.G.; Matson, P.A.; Naylor, R.; Polasky, S. Agricultural sustainability and intensive production practices. Nature 2002, 418, 671–677. [Google Scholar] [CrossRef]
- Ma, L.; Ma, W.Q.; Velthof, G.L.; Wang, F.H.; Qin, W.; Zhang, F.S.; Oenema, O. Modeling nutrient flows in the food chain of China. J. Environ. Quality 2010, 39, 1279–1289. [Google Scholar] [CrossRef] [PubMed]
- Zahid, M.; Abbasi, M.K.; Hameed, S.; Rahim, N. Isolation and identification of indigenous plant growth promoting rhizobacteria from Himalayan region of Kashmir and their effect on improving growth and nutrient contents of maize (Zea mays L.). Front. Microbiol. 2015, 6, 207. [Google Scholar] [CrossRef]
- Sarkar, S.; Gil, J.D.B.; Keeley, J.; Möhring, N.; Jansen, K. The Use of Pesticides in Developing Countries and Their Impact on Health and the Right to Food; Oxford Analytica Europe: Brussels, Belgium, 2021; p. 44. ISBN 978-92-846-7673-6. [Google Scholar]
- Singh, D.P.; Singh, H.B.; Prabha, R. Microbial Inoculants in Sustainable Agricultural Productivity; Singh, D.P., Singh, H.B., Prabha, R., Eds.; Springer: New Delhi, India, 2016; Volume 2, p. 308. [Google Scholar]
- Malusà, E.; Pinzari, F.; Canfora, L. Efficacy of Biofertilizers: Challenges to Improve Crop Production. In Microbial Inoculants in Sustainable Agricultural Productivity: Vol. 2: Functional Applications; Singh, D.P., Singh, H.B., Prabha, R., Eds.; Springer: New Delhi, India, 2016; pp. 17–40. [Google Scholar]
- Dong, L.; Li, Y.; Xu, J.; Yang, J.; Wei, G.; Shen, L.; Ding, W.; Chen, S. Biofertilizers regulate the soil microbial community and enhance Panax ginseng yields. Chin. Med. 2019, 14, 20. [Google Scholar] [CrossRef] [Green Version]
- Bhardwaj, D.; Ansari, M.W.; Sahoo, R.K.; Tuteja, N. Biofertilizers function as key player in sustainable agriculture by improving soil fertility, plant tolerance and crop productivity. Microb. Cell Factories 2014, 13, 66. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Dal Cortivo, C.; Ferrari, M.; Visioli, G.; Lauro, M.; Fornasier, F.; Barion, G.; Panozzo, A.; Vamerali, T. Effects of Seed-Applied Biofertilizers on Rhizosphere Biodiversity and Growth of Common Wheat (Triticum aestivum L.) in the Field. Front. Plant Sci. 2020, 11, 72. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Soumare, A.; Boubekri, K.; Lyamlouli, K.; Hafidi, M.; Ouhdouch, Y.; Kouisni, L. From Isolation of Phosphate Solubilizing Microbes to Their Formulation and Use as Biofertilizers: Status and Needs. Front. Bioeng. Biotechnol. 2019, 7, 425. [Google Scholar] [CrossRef] [PubMed]
- Newitt, J.T.; Prudence, S.M.M.; Hutchings, M.I.; Worsley, S.F. Biocontrol of Cereal Crop Diseases Using Streptomycetes. Pathogens 2019, 8, 78. [Google Scholar] [CrossRef] [Green Version]
- Dagher, F.; Olishevska, S.; Philion, V.; Zheng, J.; Deziel, E. Development of a novel biological control agent targeting the phytopathogen Erwinia amylovora. Heliyon 2020, 6, e05222. [Google Scholar] [CrossRef]
- Bubici, G.; Kaushal, M.; Prigigallo, M.I.; Gómez-Lama Cabanás, C.; Mercado-Blanco, J. Biological Control Agents Against Fusarium Wilt of Banana. Front. Microbiol. 2019, 10, 616. [Google Scholar] [CrossRef] [Green Version]
- Sethi, S.K.; Adhikary, S.P. Cost effective pilot scale production of biofertilizer using Rhizobium and Azotobacter. Afr. J. Biotechnol. 2012, 11, 13490–13493. [Google Scholar] [CrossRef]
- Ortega, H.E.; Torres-Mendoza, D.; Cubilla-Rios, L. Patents on Endophytic Fungi for Agriculture and Bio- and Phytoremediation Applications. Microorganisms 2020, 8, 1237. [Google Scholar] [CrossRef]
- Shahzad, R.; Bilal, S.; Imran, M.; Khan, A.L.; Alosaimi, A.A.; Al-Shwyeh, H.A.; Almahasheer, H.; Rehman, S.; Lee, I.-J. Amelioration of heavy metal stress by endophytic Bacillus amyloliquefaciens RWL-1 in rice by regulating metabolic changes: Potential for bacterial bioremediation. Biochem. J. 2019, 476, 3385–3400. [Google Scholar] [CrossRef]
- Khan, N.; Martinez-Hidalgo, P.; Humm, E.A.; Maymon, M.; Kaplan, D.; Hirsch, A.M. Inoculation With a Microbe Isolated From the Negev Desert Enhances Corn Growth. Front. Microbiol. 2020, 11, 1149. [Google Scholar] [CrossRef]
- Alsharif, W.; Saad, M.M.; Hirt, H. Desert Microbes for Boosting Sustainable Agriculture in Extreme Environments. Front. Microbiol. 2020, 11, 1666. [Google Scholar] [CrossRef] [PubMed]
- Pranaw, K.; Pidlisnyuk, V.; Trogl, J.; Malinska, H. Bioprospecting of a Novel Plant Growth-Promoting Bacterium Bacillus altitudinis KP-14 for Enhancing Miscanthus x giganteus Growth in Metals Contaminated Soil. Biology 2020, 9, 305. [Google Scholar] [CrossRef]
- Portillo, M.C.; Saiz-Jimenez, C.; Gonzalez, J.M. Molecular characterization of total and metabolically active bacterial communities of “white colonizations” in the Altamira Cave, Spain. Res. Microbiol. 2009, 160, 41–47. [Google Scholar] [CrossRef]
- Gonzalez, J.M.; Portillo, M.C.; Saiz-Jimenez, C. Metabolically active Crenarchaeota in Altamira Cave. Naturwissenschaften 2006, 93, 42–45. [Google Scholar] [CrossRef]
- Kalita, P.; Bora, L.C.; Bhagabati, K.N. Phylloplane microflora of citrus and their role in management of citrus canker. Indian Phytopathol. 1996, 49, 234–237. [Google Scholar]
- Grosu, A.I.; Sicuia, O.-A.; Dobre, A.; Voaideş, C.; Cornea, C.P. Evaluation of Some Bacillus spp. Strains for the Biocontrol of Fusarium graminearum and F. culmorum in Wheat. Agric. Agric. Sci. Procedia 2015, 6, 559–566. [Google Scholar] [CrossRef] [Green Version]
- Das, R.; Mondal, B.; Mondal, P.; Khatua, D.C.; Mukherjee, N. Biological management of citrus canker on acid lime through Bacillus subtilis (S-12) in West Bengal, India. J. Biopestic. 2014, 7, 38–41. [Google Scholar]
- Islam, M.N.; Ali, M.S.; Choi, S.J.; Hyun, J.W.; Baek, K.H. Biocontrol of Citrus Canker Disease Caused by Xanthomonas citri subsp. citri Using an Endophytic Bacillus thuringiensis. Plant Pathol. J. 2019, 35, 486–497. [Google Scholar] [CrossRef]
- Villamizar, S.; Ferro, J.A.; Caicedo, J.C.; Alves, L.M.C. Bactericidal Effect of Entomopathogenic Bacterium Pseudomonas entomophila Against Xanthomonas citri Reduces Citrus Canker Disease Severity. Front. Microbiol. 2020, 11, 1431. [Google Scholar] [CrossRef]
- Rabbee, M.F.; Ali, M.S.; Baek, K.-H. Endophyte Bacillus velezensis Isolated from Citrus spp. Controls Streptomycin-Resistant Xanthomonas citri subsp. citri That Causes Citrus Bacterial Canker. Agronomy 2019, 9, 470. [Google Scholar] [CrossRef] [Green Version]
- Daungfu, O.; Youpensuk, S.; Lumyong, S. Endophytic Bacteria Isolated from Citrus Plants for Biological Control of Citrus Canker in Lime Plants. Trop. Life Sci. Res. 2019, 30, 73–88. [Google Scholar] [CrossRef]
- Roach, R.; Mann, R.; Gambley, C.G.; Shivas, R.G.; Chapman, T.; Rodoni, B. Pathogenicity and copper tolerance in Australian Xanthomonas species associated with bacterial leaf spot. Crop. Prot. 2020, 127, 104923. [Google Scholar] [CrossRef]
- Marin, T.G.S.; Galvanin, A.L.; Lanza, F.E.; Behlau, F. Description of copper tolerant Xanthomonas citri subsp. citri and genotypic comparison with sensitive and resistant strains. Plant Pathol. 2019, 68, 1088–1098. [Google Scholar] [CrossRef]
- Gordon, T.R. Fusarium oxysporum and the Fusarium Wilt Syndrome. Annu. Rev. Phytopathol. 2017, 55, 23–39. [Google Scholar] [CrossRef]
- McGovern, R.J. Management of tomato diseases caused by Fusarium oxysporum. Crop. Prot. 2015, 73, 78–92. [Google Scholar] [CrossRef]
- López-Aranda, J.M.; Domínguez, P.; Miranda, L.; de los Santos, B.; Talavera, M.; Daugovish, O.; Soria, C.; Chamorro, M.; Medina, J.J. Fumigant Use for Strawberry Production in Europe: The Current Landscape and Solutions. Int. J. Fruit Sci. 2016, 16, 1–15. [Google Scholar] [CrossRef]
- de Lamo, F.J.; Takken, F.L.W. Biocontrol by Fusarium oxysporum Using Endophyte-Mediated Resistance. Front. Plant Sci. 2020, 11, 37. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Takken, F.; Rep, M. The arms race between tomato and Fusarium oxysporum. Mol. Plant Pathol. 2010, 11, 309–314. [Google Scholar] [CrossRef]
- Tehrani, A.; Ramezani, M. Biological control of Fusarium oxysporum, the causal agent of onion wilt by antagonistic bacteria. Commun. Agric. Appl. Biol. Sci. 2003, 68, 543–547. [Google Scholar]
- Moreno-Velandia, C.A.; Izquierdo-García, L.F.; Ongena, M.; Kloepper, J.W.; Cotes, A.M. Soil sterilization, pathogen and antagonist concentration affect biological control of Fusarium wilt of cape gooseberry by Bacillus velezensis Bs006. Plant Soil 2019, 435, 39–55. [Google Scholar] [CrossRef] [Green Version]
- Malathi, S. Biological control of onion basal rot caused by Fusarium oxysporum f. sp. cepae. Asian J. Bio Sci. 2015, 10, 21–26. [Google Scholar] [CrossRef]
- Zain, M.; Yasmin, S.; Hafeez, F.Y. Isolation and Characterization of Plant Growth Promoting Antagonistic Bacteria from Cotton and Sugarcane Plants for Suppression of Phytopathogenic Fusarium Species. Iran. J. Biotechnol. 2019, 17, e1974. [Google Scholar] [CrossRef] [PubMed]
- Yang, W. Components of rhizospheric bacterial communities of barley and their potential for plant growth promotion and biocontrol of Fusarium wilt of watermelon. Braz. J. Microbiol. 2019, 50, 749–757. [Google Scholar] [CrossRef] [PubMed]
- Pastor-Corrales, M.A. Enfermedades del frijol causadas por hongos. In Frijol: Investigación y Producción. Programa de las Naciones Unidas (PNUD); Marceliano, L.G., Fernando, O., Fernández, O., Schoonhoven, A., Eds.; Centro Internacional de Agricultura Tropical (CIAT): Cali, Colombia, 1985; pp. 169–206. [Google Scholar]
- Bianchini, A.; Maringoni, A.C.; Carneiro, B.S.M.T.G. Doenças do feijoeiro (Phaseolus vulgaris L.). In Manual de Fitopatologia. Doenças das Plantas Cultivadas; Agronômica Ceres: São Paulo, Brazil, 1997; Volume 3, pp. 376–399. [Google Scholar]
- Bassanezi, R.B.; Amorim, L.; Filho, A.B.; Hau, B.; Berger, R.D. Accounting for photosynthetic efficiency of bean leaves with rust, angular leaf spot and anthracnose to assess crop damage. Plant Pathol. 2001, 50, 443–452. [Google Scholar] [CrossRef]
- Pereira, A.C.; Paula, R.G.D.; Júnior, T.J.D.P.; Silva, L.C.D.; Carneiro, J.E.D.S.; Carneiro, P.C.S. Selection of anthracnose resistant common beans using detached leaves in partially controlled environment. Rev. Ceres 2014, 61, 518–522. [Google Scholar] [CrossRef] [Green Version]
- Amin, M.; Fitsum, S.; Selvaraj, T.; Mulugeta, N. Field Management of Anthracnose (Colletotrichum lindemuthianum) in Common Bean through Fungicides and Bioagents. Adv. Crop Sci. Technol. 2014, 2, 1000124. [Google Scholar] [CrossRef] [Green Version]
- Gillard, C.L.; Ranatunga, N.K.; Conner, R.L. The control of dry bean anthracnose through seed treatment and the correct application timing of foliar fungicides. Crop. Prot. 2012, 37, 81–90. [Google Scholar] [CrossRef]
- Schwartz, H.F.; Steadman, J.R.; Hall, R.; Forster, R.L. Compendium of Bean Diseases; APS Press: St. Paul, Brazil, 2005; p. 109. [Google Scholar]
- Dildey, O.D.F. Indução de Resistência à Antracnose do Feijoeiro por Frações de Filtrado de Cultura e Extrato de Micélio de Trichoderma longibrachiatum. Ph.D. Thesis, Universidade Estadual do Oeste do Paraná, Marechal Cândido Rondon, Brazil, 2017. [Google Scholar]
- Martins, S.J.; Faria, A.F.; Pedroso, M.P.; Cunha, M.G.; Rocha, M.R.; Medeiros, F.H.V. Microbial volatiles organic compounds control anthracnose (Colletotrichum lindemuthianum) in common bean (Phaseolus vulgaris L.). Biol. Control 2019, 131, 36–42. [Google Scholar] [CrossRef]
- Christmann, P.; Dalla Pria, M.; Hennipman, H.; Godoy, A. In vitro control of Colletotrichum lindemuthianum by Trichoderma spp. and in vivo with Alternative Products. Int. J. Adv. Eng. Res. Sci. 2019, 6, 74–80. [Google Scholar] [CrossRef]
- Pedro, E.A.D.S.; Harakava, R.; Lucon, C.M.M.; Guzzo, S.D. Promoção do crescimento do feijoeiro e controle da antracnose por Trichoderma spp. Pesqui. Agropecuária Bras. 2012, 47, 1589–1595. [Google Scholar] [CrossRef] [Green Version]
- Garcia, F.A.D.O.; Romeiro, R.D.S. Biocontrole da mancha-angular do feijoeiro por antagonistas bacterianos. Pesqui. Agropecuária Bras. 2011, 46, 1603–1608. [Google Scholar] [CrossRef]
- Corrêa, B.; Andrea, M.; Denardin, N.; Soares, V.; Schäfer, J.; Ludwig, J. Influência da microbiolização de sementes de feijão sobre a transmissão de Colletrichum lindemuthianum (Saac e Magn). Rev. Bras. Sementes 2008, 30, 156–163. [Google Scholar] [CrossRef]
- Gholami, M.; Khakvar, R.; Aliasgharzad, N. Application of endophytic bacteria for controlling anthracnose disease (Colletotrichum lindemuthianum) on bean plants. Arch. Phytopathol. Plant Prot. 2013, 46, 1831–1838. [Google Scholar] [CrossRef]
- Pandey, N.; Ravi, S.; Gusain, V.M. In vitro efficacy of biocontrol agents and fungicides against anthracnose of french bean caused by Colletotrichum lindemuthianum (Sacc. and Magn.). Int. J. Chem. Stud. 2019, 7, 3266–3269. [Google Scholar]
- Zhang, Y.; Chen, F.S.; Wu, X.Q.; Luan, F.G.; Zhang, L.P.; Fang, X.M.; Wan, S.Z.; Hu, X.F.; Ye, J.R. Isolation and characterization of two phosphate-solubilizing fungi from rhizosphere soil of moso bamboo and their functional capacities when exposed to different phosphorus sources and pH environments. PLoS ONE 2018, 13, e0199625. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Adnan, M.; Shah, Z.; Fahad, S.; Arif, M.; Alam, M.; Khan, I.A.; Mian, I.A.; Basir, A.; Ullah, H.; Arshad, M.; et al. Phosphate-Solubilizing Bacteria Nullify the Antagonistic Effect of Soil Calcification on Bioavailability of Phosphorus in Alkaline Soils. Sci. Rep. 2017, 7, 16131. [Google Scholar] [CrossRef]
- Turan, M.; Ataoğlu, N.; Sahin, F. Effects of Bacillus FS3 on growth of tomato (Lycopersicon esculentum L.) plants and availability of phosphorus in soil. Plant Soil Environ. 2007, 53, 58–64. [Google Scholar] [CrossRef] [Green Version]
- Alori, E.T.; Glick, B.R.; Babalola, O.O. Microbial Phosphorus Solubilization and Its Potential for Use in Sustainable Agriculture. Front. Microbiol. 2017, 8, 971. [Google Scholar] [CrossRef] [Green Version]
- Ali, H.; Ahmad, M.I. Agronomic efficiency and profitability of cotton on integrated use of phosphorus and plant microbes. Braz. J. Biol. 2021, 81, 484–494. [Google Scholar] [CrossRef] [PubMed]
- Munda, S.; Shivakumar, B.G.; Rana, D.S.; Gangaiah, B.; Manjaiah, K.M.; Dass, A.; Layek, J.; Lakshman, K. Inorganic phosphorus along with biofertilizers improves profitability and sustainability in soybean (Glycine max)–potato (Solanum tuberosum) cropping system. J. Saudi Soc. Agric. Sci. 2018, 17, 107–113. [Google Scholar] [CrossRef]
- Perez-Rodriguez, M.M.; Piccoli, P.; Anzuay, M.S.; Baraldi, R.; Neri, L.; Taurian, T.; Lobato Ureche, M.A.; Segura, D.M.; Cohen, A.C. Native bacteria isolated from roots and rhizosphere of Solanum lycopersicum L. increase tomato seedling growth under a reduced fertilization regime. Sci. Rep. 2020, 10, 15642. [Google Scholar] [CrossRef] [PubMed]
- Saha, M.; Sarkar, S.; Sarkar, B.; Sharma, B.K.; Bhattacharjee, S.; Tribedi, P. Microbial siderophores and their potential applications: A review. Environ. Sci. Pollut. Res. Int. 2016, 23, 3984–3999. [Google Scholar] [CrossRef]
- Aznar, A.; Dellagi, A. New insights into the role of siderophores as triggers of plant immunity: What can we learn from animals? J. Exp. Bot. 2015, 66, 3001–3010. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ahmad, F.; Ahmad, I.; Aqil, F.; Khan, M.S.; Hayat, S. Diversity and Potential of Nonsymbiotic Diazotrophic Bacteria in Promoting Plant Growth, Chapter 5. In Plant-Bacteria Interactions; Wiley: Hoboken, NJ, USA, 2008; pp. 81–109. [Google Scholar] [CrossRef]
- Greenfield, L.M.; Hill, P.W.; Paterson, E.; Baggs, E.M.; Jones, D.L. Do plants use root-derived proteases to promote the uptake of soil organic nitrogen? Plant Soil 2020, 456, 355–367. [Google Scholar] [CrossRef] [PubMed]
- Bogino, P.C.; Oliva Mde, L.; Sorroche, F.G.; Giordano, W. The role of bacterial biofilms and surface components in plant-bacterial associations. Int. J. Mol. Sci. 2013, 14, 15838–15859. [Google Scholar] [CrossRef] [Green Version]
- Seneviratne, G.; Weerasekara, M.; Seneviratne, C.; Ismail, J.; Kecskés, M.; Kennedy, I. Importance of Biofilm Formation in Plant Growth Promoting Rhizobacterial Action. In Plant Growth and Health Promoting Bacteria; Microbiology Monographs; Springer: Berlin/Heidelberg, Germany, 2010; Volume 18, pp. 81–95. [Google Scholar] [CrossRef]
- Herrera, J.J.; Cabo, M.L.; Gonzalez, A.; Pazos, I.; Pastoriza, L. Adhesion and detachment kinetics of several strains of Staphylococcus aureus subsp. aureus under three different experimental conditions. Food Microbiol. 2007, 24, 585–591. [Google Scholar] [CrossRef] [PubMed]
- Meziane, H.; Gavriel, S.; Ismailov, Z.; Chet, I.; Chernin, L.; Höfte, M. Control of green and blue mould on orange fruit by Serratia plymuthica strains IC14 and IC1270 and putative modes of action. Postharvest Biol. Technol. 2006, 39, 125–133. [Google Scholar] [CrossRef]
- Queiroz, B.P.V.D.; Melo, I.S.D. Antagonism of Serratia marcescens towards Phytophthora parasitica and its effects in promoting the growth of citrus. Braz. J. Microbiol. 2006, 37, 448–450. [Google Scholar] [CrossRef] [Green Version]
- Kar, N.; Gharbi, S.; Mebrouk, K.; Hen, J. Biological Control of Fusarium oxysporum f.p. lycopersici Isolated from Algerian Tomato by Pseudomonas fluorescens, Bacillus cereus, Serratia marcescens and Trichoderma harzianum. Res. J. Agron. 2010, 4, 31–34. [Google Scholar] [CrossRef]
- Singh, G.; Bhalla, A.; Hoondal, G. Effect of Environmental Factors on Chitinase Production by Serratia Marcescens GG5. IUP J. Life Sci. 2010, 4, 16–24. [Google Scholar]
- Jamsari, A.; Kamelia, R.; Syukur, S.; Syukriani, L.; Ferita, I. Biochemical and pathogenic potential characterization of Serratia plymuthica UBCR_12 as promising biological agents for controlling Colletotrichum gloeosporioides. Asian J. Agric. Biol. 2018, 6, 95–102. [Google Scholar]
- Wei, G. Induced Systemic Resistance to Cucumber Diseases and Increased Plant Growth by Plant Growth-Promoting Rhizobacteria Under Field Conditions. Phytopathology 1996, 86, 221. [Google Scholar] [CrossRef]
- Samson, R.; Legendre, J.B.; Christen, R.; Saux, M.F.-L.; Achouak, W.; Gardan, L. Transfer of Pectobacterium chrysanthemi (Burkholder et al., 1953) Brenner et al. 1973 and Brenneria paradisiaca to the genus Dickeya gen. nov. as Dickeya chrysanthemi comb. nov. and Dickeya paradisiaca comb. nov. and delineation of four novel species, Dickeya dadantii sp. nov., Dickeya dianthicola sp. nov., Dickeya dieffenbachiae sp. nov. and Dickeya zeae sp. nov. Int. J. Syst. Evol. Microbiol. 2005, 55, 1415–1427. [Google Scholar] [CrossRef]
- Mlaga, K.D.; Lotte, R.; Montaudie, H.; Rolain, J.M.; Ruimy, R. ‘Nissabacter archeti’ gen. nov., sp. nov., a new member of Enterobacteriaceae family, isolated from human sample at Archet 2 Hospital, Nice, France. New Microbes New Infect. 2017, 17, 81–83. [Google Scholar] [CrossRef] [PubMed]



| Isolate | Source | CC | Biocontrol Activity (%) | PGP Activity | Molecular Identification | |||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Cave | Samples | Xcc306 (iVt) (mm) | Xcc306 (iVv) | Fo (DI iVt) | Fo (II iVt) | Cl89 (DI iVt) | Cl89 (II iVt) | PS | NF (mm) | P/C/A | Sid | Genus | Accession | Ident. (%) | Cov. (%) | NCBI Ref | ||
| A1 | 2 | Ceiling | White | 21.78 | 36.4 | 23.7 | 2.7 | 83.1 | 10.3 | 0 | 0 | − + + | − | Bacillus sp. | MZ468598 | 96.09% | 97% | NR_075005.2 |
| A2 | 6 | Floor | Red | 20.18 | −12.6 | 55.0 | 79.4 | 98.3 | 100.0 | 82.9 | 3.5 | + − − | + | NI | − | − | − | − |
| A3 | 6 | Floor | Red | 22.07 | 4.6 | 56.7 | 76.9 | 95.0 | 100.0 | 49.3 | 5.5 | + − − | + | Nissabacter sp. | MZ468599 | 94.42% | 95% | NR_147393.1 |
| A4 | 6 | Floor | Red | 18.60 | 54.9 | 79.6 | 98.2 | 100.0 | 5.5 | + − − | + | Nissabacter sp. | MZ468600 | 96.50% | 924% | NR_147393.1 | ||
| A5 | 6 | Floor | Red | 17.19 | 35.0 | 56.1 | 76.2 | 99.3 | 100.0 | 157.9 | 7.5 | + − − | + | Nissabacter sp. | MZ468601 | 98.78% | 93% | NR_147393.1 |
| A6 | 6 | Floor | Red | 20.56 | 40.7 | 56.4 | 77.9 | 97.3 | 100.0 | 301.8 | 6.5 | + − − | + | Nissabacter sp. | MZ468602 | 99.39% | 94% | NR_147393.1 |
| A8 | 6 | Floor | Red | 16.65 | 67.8 | 53.9 | 79.5 | 97.7 | 100.0 | 151.5 | 7 | + − − | + | Dickeya sp. | MZ468603 | 98.47% | 98% | NR_151914.1 |
| A10 | 6 | Floor | Red | 18.36 | 44.2 | 57.6 | 77.3 | 99.0 | 100.0 | 24.9 | 7 | + − − | + | Serratia sp. | MZ468604 | 99.28% | 95% | NR_114157.1 |
| B1 | 6 | Floor | Red | 22.86 | 0.46 | 56.1 | 79.8 | 99.3 | 100.0 | 41.5 | 6 | + − − | + | Serratia sp. | MZ468605 | 99.64% | 89% | NR_114157.1 |
| B4 | 6 | Ceiling | Red | 22.69 | 17.2 | 58.4 | 79.5 | 97.8 | 100.0 | 260.4 | 3.5 | + − − | + | Serratia sp. | MZ468606 | 98.48% | 87% | NR_114043.1 |
| B6 | 6 | Ceiling | Red | 23.39 | 28.0 | 57.4 | 78.1 | 97.5 | 100.0 | 49.3 | 5.5 | + − − | + | Serratia sp. | MZ468607 | 98.20% | 95% | NR_114157.1 |
| B9 | 6 | Ceiling | Red | 18.38 | 20.5 | 67.0 | 78.0 | 99.0 | 100.0 | 20.4 | 4.5 | + − − | + | Serratia sp. | MZ468608 | 97.93% | 94% | NR_114157.1 |
| C2 | 6 | Ceiling | Red | 21.66 | −2.1 | 62.7 | 80,0 | 98.8 | 100.0 | 65.5 | 5 | + − − | + | Serratia sp. | MZ468609 | 99.64% | 97% | NR_114157.1 |
| C3 | 6 | Ceiling | Red | 19.88 | 40.8 | 66.0 | 81.4 | 83.7 | 100.0 | 46.9 | 5.5 | + − − | + | Serratia sp. | MZ468610 | 98.93% | 90% | NR_114157.1 |
| C7 | 6 | Ceiling | Red | 22.02 | −8.0 | 67.0 | 79.5 | 99.0 | 100.0 | 52.8 | 4 | + − − | + | Serratia sp. | MZ468611 | 99.64% | 94% | NR_114157.1 |
| D3 | 8 | Ceiling | Red | 20.07 | 46.0 | 68.4 | 80.6 | 98.9 | 100.0 | 39.0 | 4 | + − − | + | Serratia sp. | MZ468612 | 99.65% | 95% | NR_114157.1 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lemes, C.G.C.; Cordeiro, I.F.; de Paula, C.H.; Silva, A.K.; do Carmo, F.F.; Kamino, L.H.Y.; Carvalho, F.M.S.; Caicedo, J.C.; Ferro, J.A.; Moreira, L.M. Potential Bioinoculants for Sustainable Agriculture Prospected from Ferruginous Caves of the Iron Quadrangle/Brazil. Sustainability 2021, 13, 9354. https://doi.org/10.3390/su13169354
Lemes CGC, Cordeiro IF, de Paula CH, Silva AK, do Carmo FF, Kamino LHY, Carvalho FMS, Caicedo JC, Ferro JA, Moreira LM. Potential Bioinoculants for Sustainable Agriculture Prospected from Ferruginous Caves of the Iron Quadrangle/Brazil. Sustainability. 2021; 13(16):9354. https://doi.org/10.3390/su13169354
Chicago/Turabian StyleLemes, Camila G. C., Isabella F. Cordeiro, Camila H. de Paula, Ana K. Silva, Flávio F. do Carmo, Luciana H. Y. Kamino, Flávia M. S. Carvalho, Juan C. Caicedo, Jesus A. Ferro, and Leandro M. Moreira. 2021. "Potential Bioinoculants for Sustainable Agriculture Prospected from Ferruginous Caves of the Iron Quadrangle/Brazil" Sustainability 13, no. 16: 9354. https://doi.org/10.3390/su13169354






