Potential Applications of Native Cyanobacterium Isolate (Arthrospira platensis NIOF17/003) for Biodiesel Production and Utilization of Its Byproduct in Marine Rotifer (Brachionus plicatilis) Production
Abstract
:1. Introduction
2. Materials and Methods
2.1. Sampling
2.2. Isolation and Purification
2.3. Phylogenetic Identification
2.4. Growth, Biomass, and Biochemical Constituent
2.5. Determination of Biodiesel Properties
2.6. Application of Algal-Free Lipid as Aquaculture Feed
2.7. Statistical Analysis
3. Results
3.1. Water Sample Characterizations
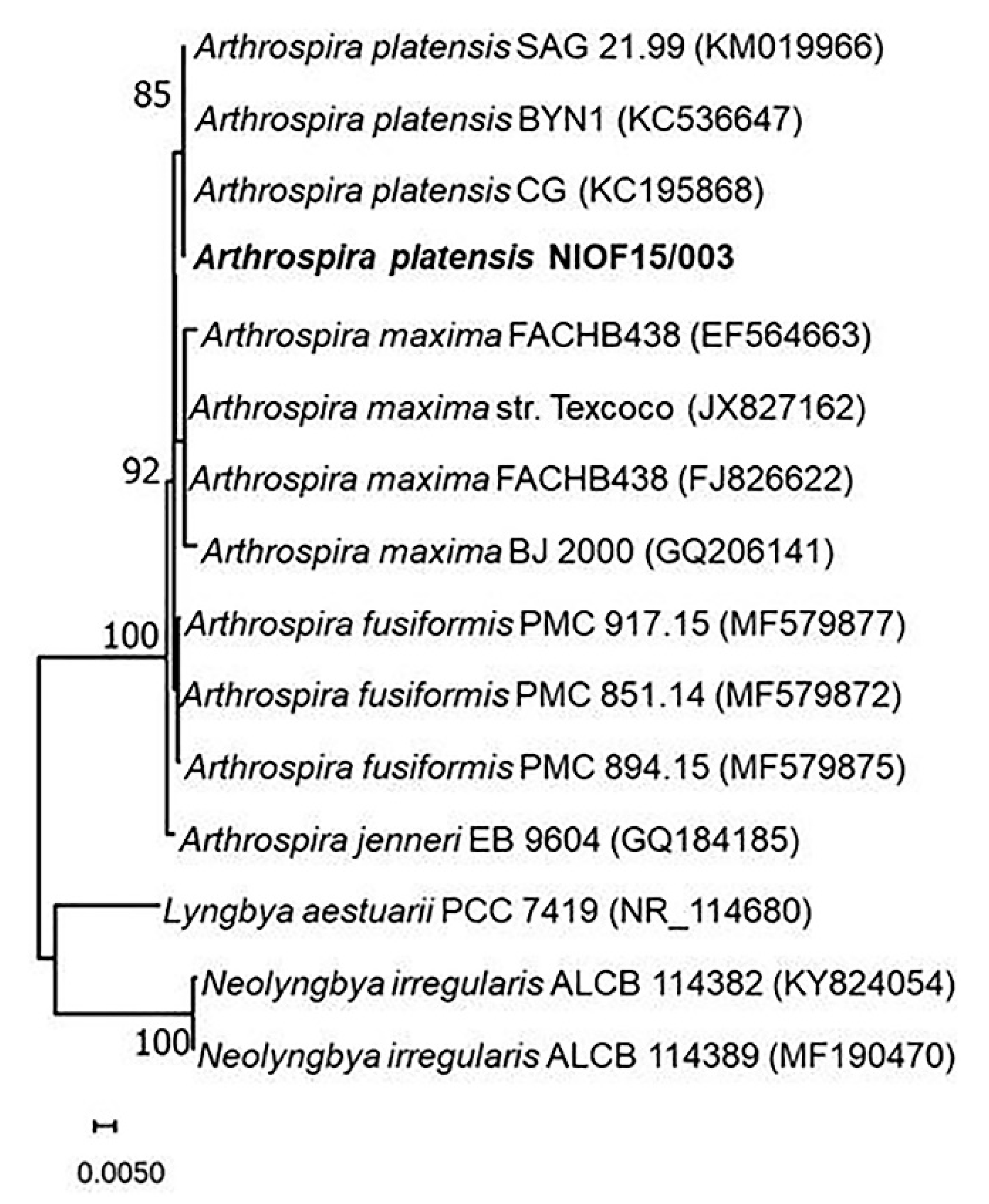
3.2. Phylogenetic Identification
3.3. Growth and Biochemical Composition
3.4. Fatty Acid Profiles and Biodiesel Characteristics
3.5. Nutritional Value of Biodiesel Byproduct (FL) as Aquaculture Feed
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| ANOVA | analysis of variance |
| CFPP | cold filter plugging point |
| CN | cetane number |
| DU | unsaturation degree |
| DW | dry weight |
| EC | electrical conductivity |
| EEP | early exponential phase |
| FA | fatty acid |
| FAME | fatty acid methylated ester |
| FL | free lipid biomass |
| GC-MS | gas chromatography-mass spectrometry |
| IV | iodine value |
| KV | kinematic viscosity |
| LCSF | long-chain saturation factor |
| LEP | late exponential phase |
| MUFA | monounsaturated fatty acid |
| NIOF | National Institute of Oceanography and Fisheries |
| NJ | neighbor-joining |
| OD | optical density |
| PD | parameter distance |
| PUFA | polyunsaturated fatty acids |
| RT | retention time |
| SD | standard deviation |
| SV | saponification value |
| TSS | total soluble salts |
| WB | whole biomass |
References
- Chisti, Y. Biodiesel from microalgae. Biotechnol. Adv. 2007, 25, 294–306. [Google Scholar] [CrossRef] [PubMed]
- Rahman, M.A.; Aziz, M.A.; Al-khulaidi, R.A.; Sakib, N.; Islam, M. Biodiesel production from microalgae Spirulina maxima by two step process: Optimization of process variable. J. Rad. Res. Appl. Sci. 2017, 10, 140–147. [Google Scholar] [CrossRef] [Green Version]
- El-Shenody, R.A.; Ashour, M.; Ghobara, M.M.E. Evaluating the chemical composition and antioxidant activity of three Egyptian seaweeds Dictyota dichtoma, Turbinaria decurrens, and Laurencia obtuse. Braz. J. Food Technol. 2019, 22, e2018203. [Google Scholar] [CrossRef] [Green Version]
- Shao, W.; Ebaid, R.; El-Sheekh, M.; Abomohra, A.; Eladel, H. Pharmaceutical applications and consequent environmental impacts of Spirulina (Arthrospira): An overview. Grasas Aceites 2019, 70, e292. [Google Scholar] [CrossRef] [Green Version]
- Barra, L.; Chandrasekaran, R.; Corato, F.; Brunet, C. The challenge of ecophysiological biodiversity for biotechnological applications of marine microalgae. Mar. Drugs 2014, 12, 1641–1675. [Google Scholar] [CrossRef]
- Zabed, H.M.; Akter, S.; Yun, J.; Zhang, G.; Zhang, Y.; Qi, X. Biogas from microalgae: Technologies, challenges and opportunities. Renew. Sustain. Energy Rev. 2020, 117, 109503. [Google Scholar] [CrossRef]
- Ashour, M. Marine Microalgae: Aquaculture and Biodiesel Production; LAP Lambert Academic Publisher: Saarbrücken, Germany, 2015; pp. 1–240, ISBN-13: 978-3659691683. [Google Scholar]
- Elshobary, M.; El-Shenody, R.; Ashour, M.; Zabed, H.M.; Qi, X. Antimicrobial and antioxidant characterization of bioactive components from Chlorococcum minutum, a newly isolated green microalga. Food Biosci. 2020, 35, 100567. [Google Scholar] [CrossRef]
- Sharawy, Z.Z.; Ashour, M.; Abbas, E.; Ashry, O.; Helal, M.; Nazmi, H.; Kelany, M.; Kamel, A.; Hassaan, M.; Rossi, W.J.; et al. Effects of dietary marine microalgae, Tetraselmis suecica on production, gene expression, protein markers and bacterial count of Pacific white shrimp Litopenaeus vannamei. Aquac. Res. 2020, 51, 2216–2228. [Google Scholar] [CrossRef]
- Ashour, M.; Mabrouk, M.M.; Ayoub, H.F.; El-Feky, M.M.M.; Sharawy, Z.Z.; Van Doan, H.; Goda, A.M.A.-S.; El-Haroun, E. Effect of dietary seaweed extract supplementation on growth, feed utilization, hematological indices and non-specific immunity of Nile Tilapia, Oreochromis niloticus, challenged with Aeromonas hydrophila. J. Appl. Phycol. 2020, 32, 3467–3479. [Google Scholar] [CrossRef]
- Abdel Rahman, S.H.; Abdel Razek, F.A.; Abou-Zeid, A.E.; Ashour, M. Optimum growth conditions of three isolated diatoms species; Skeletonema costatum, Chaetoceros calcitrans and Detonula confervacea and their utilization as feed for marine penaeid shrimp larvae. EJAR 2010, 36, 161–183. [Google Scholar]
- Abo-Taleb, H.A.; Zeina, A.F.; Ashour, M.; Mabrouk, M.M.; Sallam, A.E.; El-feky, M.M.M. Isolation and cultivation of the freshwater amphipod Gammarus pulex (Linnaeus, 1758), with an evaluation of its chemical and nutritional content. Egypt. J. Aquat. Biol. Fish. 2020, 24, 69–82. [Google Scholar] [CrossRef] [Green Version]
- Ashour, M.; El-Shafei, A.; Khairy, H.M.; Abd-Elkader, D.Y.; Mattar, M.A.; Alataway, A.; Hassan, S.M. Effect of Pterocladia capillacea seaweed extracts on growth parameters and biochemical constituents of Jew’s Mallow. Agronomy 2020, 10, 420. [Google Scholar] [CrossRef] [Green Version]
- Hassan, S.M.; Ashour, M.; Soliman, A.A.F. Anticancer activity, antioxidant activity, mineral contents, vegetative and yield of Eruca sativa using foliar application of autoclaved cellular extract of Spirulina platensis extract, comparing to N-P-K fertilizers. J. Plant Prod. 2017, 8, 529–536. [Google Scholar] [CrossRef] [Green Version]
- Ashour, M. Current and future perspectives of microalgae-aquaculture in Egypt, case study: SIMAF-prototype-project. Egypt. J. Anim. Prod. 2020, 57, 163–170. [Google Scholar] [CrossRef]
- Han, S.; Jin, W.; Chen, Y.; Tu, R.; Abomohra, A. Enhancement of Lipid Production of Chlorella Pyrenoidosa Cultivated in Municipal Wastewater by Magnetic Treatment. Appl. Biochem. Biotechnol. 2016, 180, 1043–1055. [Google Scholar] [CrossRef] [PubMed]
- Ashour, M.; Elshobary, M.E.; El-Shenody, R.; Kamil, A.W.; Abomohra, A.E.F. Evaluation of a native oleaginous marine microalga Nannochloropsis oceanica for dual use in biodiesel production and aquaculture feed. Biomass Bioenergy 2019, 120, 439–447. [Google Scholar] [CrossRef]
- Schenk, P.M.; Thomas-Hall, S.R.; Stephens, E.; Marx, U.C.; Mussgnug, J.H.; Posten, C.; Kruse, O.; Hankamer, B. Second generation biofuels: High-efficiency microalgae for biodiesel production. Bioenergy Res. 2008, 1, 20–43. [Google Scholar] [CrossRef]
- Scott, S.A.; Davey, M.P.; Dennis, J.S.; Horst, I.; Howe, C.J.; Lea-Smith, D.J.; Smith, A.G. Biodiesel from algae: Challenge and prospects. Curr. Opin. Biotechnol. 2010, 21, 277–286. [Google Scholar] [CrossRef] [PubMed]
- Mostafa, S.S.M.; El-Gendy, N.S. Evaluation of fuel properties for microalgae Spirulina platensis bio-diesel and its blends with Egyptian petro-diesel. Arab. J. Chem. 2017, 10, S2040–S2050. [Google Scholar] [CrossRef] [Green Version]
- Habib, M.A.B.; Parvin, M.; Huntington, T.C.; Hasan, M.R. A Review on Culture, Production and Use of Spirulina as Food for Humans and Feeds for Domestic Animals and Fish; FAO Fisheries and Aquaculture Circular, No 1034; FAO: Rome, Italy, 2008; 33p. [Google Scholar]
- United Nations Food and Agriculture Organization. The State of World Fisheries and Aquaculture 2018: Meeting the Sustainable Development Goals; Licence: CC BY-NC-SA 3.0 IGO; FAO: Rome, Italy, 2018. [Google Scholar]
- Kalsum, L.; Dewi, E.; Margarety, E.; Ningsih, A.S. Lipid Extraction from Microalgae Spirulina platensis for Raw Materials of Biodiesel. In Journal of Physics: Conference Series, Proceedings of 2nd Forum in Research, Science, and Technology, Palembang, Indonesia, 30–31 October 2018; IOP Publishing: Bristol, UK, 2018; Volume 1167, p. 012051. [Google Scholar] [CrossRef]
- Wuang, S.C.; Khin, M.C.; Chua, P.Q.D.; Luo, Y.D. Use of Spirulina biomass produced from treatment of aquaculture waste water as agricultural fertilizers. Algal Res. 2016, 15, 59–64. [Google Scholar] [CrossRef]
- Rajak, U.; Nashine, P.; Verma, T.N. Characteristics of microalgae Spirulina biodiesel with the impact of n-butanol addition on a CI engine. Energy 2019, 189, 116311. [Google Scholar] [CrossRef]
- Murad, M.E.; Al-Dawody, M.F. Biodiesel Production from Spirulina Microalgae and its impact on Diesel Engine Characteristics-Review. Al-Qadisiyah J. Eng. Sci. 2020, 13, 158–166. [Google Scholar] [CrossRef]
- Shirazia, H.M.; Karimi-Sabet, J.; Ghotbia, C. Biodiesel production from Spirulina microalgae feedstock using direct Transesterification near supercritical methanol condition. Bioresour. Technol. 2017, 239, 378–386. [Google Scholar] [CrossRef] [PubMed]
- Volkmann, H.; Imianovsky, U.; Oliveira, J.L.; Sant’Anna, E.S. Cultivation of Arthrospira (Spirulina) platensis in desalinator wastewater and salinated synthetic medium: Protein content and amino-acid profile. Braz. J. Microbiol. 2008, 39, 98–101. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Costa, L.A.V.; Colla, L.M.; Filho, P.D. Spirulina platensis growth in open raceway ponds using fresh water supplemented with carbon, nitrogen and metal ions. Z. Nat. C 2002, 58, 76–80. [Google Scholar] [CrossRef]
- Nogueira, S.M.S.; Junior, J.S.; Maia, H.D.; Saboya, J.P.S.; Farias, W.R.L. Use of Spirulina platensis in treatment of fish farming wastewater. Rev. Ciência Agronômica 2018, 49, 599–606. [Google Scholar] [CrossRef]
- El-Kassas, H.Y.; Heneash, A.M.M.; Hussein, N.R. Cultivation of Arthrospira (Spirulina) platensis using confectionary wastes for aquaculture feeding. J. Genet. Eng. Biotech. 2015, 13, 145–155. [Google Scholar] [CrossRef] [Green Version]
- Cheunbarn, S.; Peerapornpisal, Y. Cultivation of Spirulina platensis using anaerobically swine wastewater treatment effluent. Int. J. Agric. Biol. 2010, 12, 586–590. [Google Scholar]
- Chavan, R.; Mutnuri, S. Tertiary treatment of domestic wastewater by Spirulina platensis integrated with microalgal biorefinery. Biofuels 2019, 10, 33–44. [Google Scholar] [CrossRef]
- Hoballah, E.M.; Attallah, A.G.; Abd-El-Aal, S.K. Genetic diversity of some new local strains of cyanobacteria isolated from Wadi El-natrun, Egypt. Int. J. Acad. Res. 2014, 4, 314–326. [Google Scholar]
- Selim, I.M.; Barakat, O.S.; Aly, M.S.; Higazy, A.M. Ecological evaluation of marine cyanobacteria of El-Khadra Lake in Egypt. Int. J. Curr. Microbiol. App. Sci. 2014, 3, 468–485. [Google Scholar]
- Aly, M.S.; Gad, A.S. Chemical Composition and Potential Application of Spirulina platensis Biomass. J. Am. Sci. 2010, 6, 1291–1293. [Google Scholar]
- Marrez, D.A.L.; Naguib, M.M.; Sultan, Y.Y.; Daw, Z.Y.; Higazy, A.M. Evaluation of Chemical Composition for Spirulina platensis in Different Culture Media. Res. J. Pharm. Biol. Chem. Sci. 2014, 5, 1162. [Google Scholar]
- Hamed, A.F.; Salem, B.B.; Abd El-Fatah, H.M. Floristic Survey of Blue-Green Algae/Cyanobacteria in Saline-Alkaline Lakes of Wadi El- Natrun (Egypt) by Remote Sensing Application. J. Appl. Sci. Res. 2007, 3, 495–506. [Google Scholar]
- American Public Health Association. Standard Methods for the Examination of Water and Wastewater, 16th ed.; American Public Health Association: Washington, DC, USA, 1985; p. 1268. [Google Scholar]
- Zarrouk, C. Contribution a L’etude D’une Cyanophycee. Influence de Divers Facteurs Physiques et Chimiques sur la Croissance et la Photosynthese de Spirulina mixima. Ph.D. Thesis, University of Paris, Paris, France, 1966. [Google Scholar]
- Robert, A. Algal Culturing Techniques: Traditional Microalgae Isolation Techniques; Elsevier Academic Press: Cambridge, MA, USA, 2005; pp. 83–98. [Google Scholar]
- Komárek, J.; Anagnostidis, K. Modern Approach to the Classification System of Cyanophytes 4. Nostocales. Arch. Hydrobiol. 1989, 82, 247–345. [Google Scholar]
- Komárek, J. The modern classification of cyanoprokaryotes (Cyanobacteria). Oceanol. Hydrobiol. Stud. 2005, 34, 3. [Google Scholar]
- Komárek, J. Recent changes (2008) in cyanobacteria taxonomy based on a combination of molecular background with phenotype and ecological consequences (genus and species concept). Hydrobiologia 2010, 639, 245–259. [Google Scholar] [CrossRef]
- Grube, M.; DePriest, P.T.; Gargas, A.; Hafellner, J. DNA isolation from lichen ascomata. Mycol. Res. 1995, 99, 1321–1324. [Google Scholar] [CrossRef]
- Elshobary, M.E.; Osman, M.E.H.; Abushady, A.M.; Piercey-Normore, M.D. Comparison of lichen-forming cyanobacterial and green algal photobionts with free-living algae. Cryptogam. Algol. 2015, 36, 81–100. [Google Scholar] [CrossRef]
- Kumar, S.; Stecher, G.; Li, M.; Knyaz, C.; Tamura, K. MEGA X: Molecular Evolutionary Genetics Analysis across computing platforms. Mol. Biol. Evol. 2018, 35, 1547–1549. [Google Scholar] [CrossRef]
- Madkour, F.F.; Kamil, A.E.W.; Nasr, H.S. Production and nutritive value of Spirulina platensis in reduced cost media. Egypt. J. Aquat. Res. 2012, 38, 51–57. [Google Scholar] [CrossRef] [Green Version]
- Abomohra, A.; Wagner, M.; El-Sheekh, M.; Hanelt, D. Lipid and total fatty acid productivity in photoautotrophic fresh water microalgae: Screening studies towards biodiesel production. J. Appl. Phycol. 2013, 25, 931–936. [Google Scholar] [CrossRef]
- Rausch, T. The estimation of micro-algal protein content and its meaning to the evaluation of algal biomass I. Comparison of methods for extracting protein. Hydrobiologia 1981, 7, 237–251. [Google Scholar] [CrossRef]
- Hartree, E. Determination of protein: A modification of the Lowry method that gives a linear photometric response. Anal. Biochem. 1972, 48, 422–427. [Google Scholar] [CrossRef]
- Myklestad, S.; Haug, A. Production of carbohydrates by the marine diatom Chaetoceros affinis var. willei (Gran) Hustedt. I. Effect of the concentration of nutrients in the culture medium. J. Exp. Mar. Biol. Ecol. 1972, 9, 125–136. [Google Scholar] [CrossRef]
- DuBois, M.; Gilles, K.; Hamilton, J.; Rebers, P.; Smith, F. Colorimetric method for determination of sugars and related substances. Anal. Chem. 1956, 28, 350–356. [Google Scholar] [CrossRef]
- Elshobary, M.E.; Abo-Shady, A.M.; Khairy, H.M.; Essa, D.; Zabed, H.M.; Qi, X.; Abomohra, A.E.-F.F. Influence of nutrient supplementation and starvation conditions on the biomass and lipid productivities of Micractinium reisseri grown in wastewater for biodiesel production. J. Environ. Manag. 2019, 250, 109529. [Google Scholar] [CrossRef] [PubMed]
- Wen, X.; Du, K.; Wang, Z.; Peng, X.; Luo, L.; Tao, H.; Xu, Y.; Zhang, D.; Geng, Y.; Li, Y. Effective cultivation of microalgae for biofuel production: A pilot‑scale evaluation of a novel oleaginous microalga Graesiella sp. WBG‑1. Biotechnol. Biofuels 2016, 9, 123. [Google Scholar] [CrossRef] [Green Version]
- Ramirez-Verduzco, L.F.; Rodriguez-Rodriguez, J.E.; Jaramillo-Jacob, A.R. Predicting cetane number, kinematic viscosity, density and higher heating value of biodiesel from its fatty acid methyl ester composition. Fuel 2012, 91, 102–111. [Google Scholar] [CrossRef]
- Can, S.S.; Koru, E.; Cirik, S. Effect of temperature and nitrogen concentration on the growth and lipid content of Spirulina platensis and biodiesel production. Aquacult. Int. 2017, 25, 1485–1493. [Google Scholar] [CrossRef]
- Ma, Y.; Wang, Z.; Yu, C.; Yin, Y.; Zhou, G. Evaluation of the potential of 9 Nannochloropsis strains for biodiesel production. Bioresour. Technol. 2014, 167, 503–509. [Google Scholar] [CrossRef]
- Mitra, M.; Shah, F.; Bharadwaj, S.; Patidar, S.; Mishra, S. Cultivation of Nannochloropsis oceanica biomass rich in eicosapentaenoic acid utilizing wastewater as nutrient resource. Bioresour. Technol. 2016, 218, 1178–1186. [Google Scholar] [CrossRef]
- American Society for Testing and Materials. Standard Specification for Biodiesel Fuel Blend Stock (B100) for Middle Distillate Fuels; ASTM D6751-08; ASTM International: West Conshohocken, PA, USA, 2008. [Google Scholar]
- European Committee for Standardization. Automotive Fuels D Fatty Acid Methylesters (FAME) for Diesel Engines D Requirements and Test Methods; EN14214; European Committee for Standardization: Brussels, Belgium, 2008. [Google Scholar]
- Abdulqader, G.; Barsanti, L.; Tredici, M.R. Harvest of Arthrospira platensis from Lake Kossorom (Chad) and its household usage among the Kanembu. J. Appl. Phycol. 2000, 12, 493–498. [Google Scholar] [CrossRef]
- Venkataraman, L. Spirulina platensis (Arthrospira): Physiology, Cell Biology and Biotechnologym, edited by Avigad Vonshak. J. Appl. Phycol. 1997, 9, 295–296. [Google Scholar] [CrossRef]
- Hamed, I. The Evolution and Versatility of Microalgal Biotechnology: A Review. Compr. Rev. Food Sci. Food Saf. 2016, 15, 1104–1123. [Google Scholar] [CrossRef]
- Gouveia, L.; Batista, A.P.; Sousa, I.; Raymundo, A.; Bandarra, N.M. Microalgae in novel food products. In Food Chemistry Research Developments; Papadopoulos, K.N., Ed.; Nova Publishers: Hauppauge, NY, USA, 2008; pp. 75–111. [Google Scholar]
- Abbas, E.M.; Ali, F.S.; Desouky, M.G.; Ashour, M.; El-Shafei, A.; Maaty, M.M.; Sharawy, Z.Z. Novel Comprehensive Molecular and Ecological Study Introducing Coastal Mud Shrimp (Solenocera Crassicornis) Recorded at the Gulf of Suez, Egypt. J. Mar. Sci. Eng. 2021, 9, 9. [Google Scholar] [CrossRef]
- Abo-Taleb, H.; Ashour, M.; El-Shafei, A.; Alataway, A.; Maaty, M.M. Biodiversity of Calanoida Copepoda in different habitats of the North-Western Red Sea (Hurghada Shelf). Water 2020, 12, 656. [Google Scholar] [CrossRef] [Green Version]
- Albert, N.; Wague, R.; Mbaïlao, M.; Fabienne, N. Changes in the physico-chemical properties of Spirulina platensis from three production sites in Chad. J. Ani. Plant Sci. 2012, 13, 1811–1822. [Google Scholar]
- Gershwin, M.E.; Belay, A. Spirulina in Human Nutrition and Health; CRC Press: Boca Raton, FL, USA, 2008. [Google Scholar] [CrossRef]
- Becker, E.W. Micro-algae as a source of protein. Biotechnol. Adv. 2007, 25, 207–210. [Google Scholar] [CrossRef]
- Yoshida, M.; Hoshii, H. Nutritive value of Spirulina, green algae, for poultry feed. Jpn. Poult. Sci. 1980, 17, 2730. [Google Scholar] [CrossRef] [Green Version]
- Matta, T.M.; Martin, A.A.; Caetano, N.S. Microalgae for Biodiesel Production and Aplication: A Review. Renew. Sustain. Energy Rev. 2017, 14, 217–232. [Google Scholar] [CrossRef] [Green Version]
- Ashour, M.; Kamel, A. Enhance growth and biochemical composition of Nannochloropsis oceanica, cultured under nutrient limitation, using commercial agricultural fertilizers. J. Mar. Sci. Res. Dev. 2017, 7, 233. [Google Scholar] [CrossRef] [Green Version]
- Ashour, M.; Abo-Taleb, H.A.; Abou-Mahmoud, M.M.; El-Feky, M.M.M. Effect of the integration between plankton natural productivity and environmental assessment of irrigation water, El-Mahmoudia Canal, on aquaculture potential of Oreochromis niloticus. Turk. J. Fish. Aquat. Sci. 2018, 18, 1163–1175. [Google Scholar] [CrossRef]
- Metwally, A.S.; El-Naggar, H.A.; El-Damhougy, K.A.; Bashar, M.A.E.; Ashour, M.; Abo-Taleb, H.A.H. GC-MS analysis of bioactive components in six different crude extracts from the Soft Coral (Sinularia maxim) collected from Ras Mohamed, Aqaba Gulf, Red Sea, Egypt. Egy. J. Aqua. Biol. Fisher. 2020, 24, 425–434. [Google Scholar] [CrossRef]
- Cohen, Z. The chemicals of Spirulina. In Spirulina platensis (Arthrospira): Physiology, Cell-Biology and Biotechnology; Vonshak, A., Ed.; Taylor & Francis Ltd.: London, UK, 1997; pp. 175–204. [Google Scholar]
- El-Sheekh, M.; Abomohra, A.; Hanelt, D. Optimization of biomass and fatty acid productivity of Scenedesmus obliquus as a promising microalga for biodiesel production, World. J. Microbiol. Biotechnol. 2013, 29, 915–922. [Google Scholar] [CrossRef] [PubMed]
- Adams, C.; Godfrey, V.; Wahlen, B.; Seefeldt, L.; Bugbee, B. Understanding precision nitrogen stress to optimize the growth and lipid content tradeoff in oleaginous green microalgae. Bioresour. Technol. 2013, 131, 188–194. [Google Scholar] [CrossRef] [Green Version]
- Udayan, A.; Kathiresan, S.; Arumugam, M. Kinetin and Gibberellic acid (GA3) act synergistically to produce high value polyunsaturated fatty acids in Nannochloropsis oceanica CASA CC201. Algal Res. 2018, 32, 82–192. [Google Scholar] [CrossRef]
- Lee, S.J.; Go, S.; Jeong, G.T.; Kim, S.K. Oil production from five marine microalgae for the production of biodiesel. Biotechnol. Bioproc. Eng. 2011, 16, 561–566. [Google Scholar] [CrossRef]
- Heneash, A.; Ashour, M.; Matar, M. Effect of Un-live Microalgal diet, Nannochloropsis oculata and Arthrospira (Spirulina) platensis, comparing to yeast on population of rotifer, Brachionus plicatilis. Mediterr. Aquac. J. 2015, 7, 48–54. [Google Scholar] [CrossRef]
- Abdel Rahman, S.H.; Abdel Razek, F.A.; Abou-Zeid, A.E.; Ashour, M. Population growth rate, fecundity, filtration and ingestion rate of marine rotifer Brachionus plicatilis fed with motile and immotile microalgae. Egypt. J. Aquat. Res. 2008, 34, 426–439. [Google Scholar]
- Abomohra, A.; Elsayed, M.; Esakkimuthud, S.; El-Sheekh, M.; Hanelte, D. Potential of fat, oil and grease (FOG) for biodiesel production: A critical review on the recent progress and future perspectives. Prog. Energy Combust. Sci. 2020, 81, 100868. [Google Scholar] [CrossRef]
- Abomohra, A.; Zhenga, X.; Wang, Q.; Huang, J.; Ebaid, R. Enhancement of biodiesel yield and characteristics through in-situ solvo-thermal co-transesterification of wet microalgae with spent coffee grounds. Bior. Tech. 2021, 323, 124640. [Google Scholar] [CrossRef] [PubMed]
- El-Khodary, G.M.; El-Sayed, H.S.; Khairy, H.M.; El-Sheikh, M.A.; Qi, X.; Elshobary, M.E. Comparative study on growth, survival and pigmentation of Solea aegyptiaca larvae by using four different microalgal species with emphasize on water quality and nutritional value. Aquac. Nutr. 2020, 1–15. [Google Scholar] [CrossRef]
- Abomohra, A.; El-Sheekh, M.; Hanelt, D. Pilot cultivation of the chlorophyte microalga Scenedesmus obliquus as a promising feedstock for biofuel. Biomass Bioenergy 2014, 64, 237–244. [Google Scholar] [CrossRef]
- El-Kassas, H.; Mohammady, N.; El-Sayed, H.; ELSherbiny, B. Growth and biochemical variability of complete and lipid extracted Chlorella species (application for Artemia franciscana feeding). Rend. Fis. Acc. Lincei. 2016, 27, 761–774. [Google Scholar] [CrossRef]



| Characteristics | Values |
|---|---|
| Temperature (°C) | 27.50 ± 0.75 |
| pH | 9.36 ± 0.17 |
| CO3 (ppt) | 2.80 ± 0.18 |
| HCO3 (ppt) | 8.55 ± 260 |
| Conductivity (EC, ds/m) | 87.70 ± 1.90 |
| Total Soluble Salts (TSS, ppt) | 67.30 ± 1.10 |
| Parameter | Values |
|---|---|
| Dry Weight (DW, g L−1) | 0.845 ± 0.038 |
| Proteins (% DW) | 52.03 ± 0.77 |
| Lipids (% DW) | 8.52 ± 1.04 |
| Carbohydrates (% DW) | 14.00 ± 1.67 |
| Biomass productivity (mg L−1 day−1) | 143.83 ± 3.69 |
| Lipid productivity (mg L−1 day−1) | 14.37 ± 0.37 |
| Fatty Acids | Percentage (%) of Total FA |
|---|---|
| C6:0 | 0.04 |
| C8:0 | 0.23 |
| C10:0 | 0.13 |
| C11:0 | 0.24 |
| C12:0 | 0.05 |
| C13:0 | 0.08 |
| C14:0 | 1.96 |
| C14:1 | 0.45 |
| C15:0 | 1.75 |
| C15:1 | 0.18 |
| C16:0 | 33.60 |
| C16:1 | 11.34 |
| C17:0 | 1.86 |
| C17:1 | 0.48 |
| C18:0 | 2.33 |
| C18:1 | 14.26 |
| C18:2ω6 | 27.88 |
| C18:3 ω3 | 1.99 |
| C20:3ω3 | 0.46 |
| C20:5 ω3 | 0.32 |
| C22:6 ω3 | 0.39 |
| ΣSTFA | 42.27 |
| ΣMUFA | 26.71 |
| ΣPUFA | 31.04 |
| Strain/Standard | DU | CN | IV | SV | LCSF | CFPP | KV | Db ≥ 4 | Reference |
|---|---|---|---|---|---|---|---|---|---|
| A. platensis NIOF17/003 | 88.7 | 52.9 | 85.5 | 210.7 | 4.5 | −2.2 | 19.3 | 0.71 | This study |
| A. platensis | 12.53 * | 70 | 102 | 191.9 * | 4.28 * | −3 | 12.4 | 0 | [20] |
| A. platensis | 99.08 * | 71.02 * | 109.64 * | 110.50 * | 8.35 * | 9.75 * | 4.67 | 0 | [57] |
| N. oceanica CCMP531 | 58 | 58 | 66 | 207 * | 5 * | 10 | 4.2 | 5.4 | [58] |
| N. oceanica CCNM 1081 | 70–102 * | 36–54 | 88–169 | 195–200 | 4.4–6 | 2.5–2.8 | 4.6 * | 12–36 | [59] |
| N. oceanica NIOF15/001 | 57 | 52 | 94 | 200 | 6.7 | 4.6 | 4 | 19.5 | [17] |
| US (ASTM6751−03) # | - | ≥47 | - | - | - | - | 1.9–6.0 | - | [60] |
| Europe (EN 14214) # | - | ≥51 | ≤120 | - | - | ≤5/≤−20 | 3.5–5.0 | - | [61] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zaki, M.A.; Ashour, M.; Heneash, A.M.M.; Mabrouk, M.M.; Alprol, A.E.; Khairy, H.M.; Nour, A.M.; Mansour, A.T.; Hassanien, H.A.; Gaber, A.; et al. Potential Applications of Native Cyanobacterium Isolate (Arthrospira platensis NIOF17/003) for Biodiesel Production and Utilization of Its Byproduct in Marine Rotifer (Brachionus plicatilis) Production. Sustainability 2021, 13, 1769. https://doi.org/10.3390/su13041769
Zaki MA, Ashour M, Heneash AMM, Mabrouk MM, Alprol AE, Khairy HM, Nour AM, Mansour AT, Hassanien HA, Gaber A, et al. Potential Applications of Native Cyanobacterium Isolate (Arthrospira platensis NIOF17/003) for Biodiesel Production and Utilization of Its Byproduct in Marine Rotifer (Brachionus plicatilis) Production. Sustainability. 2021; 13(4):1769. https://doi.org/10.3390/su13041769
Chicago/Turabian StyleZaki, Mohamed A., Mohamed Ashour, Ahmed M. M. Heneash, Mohamed M. Mabrouk, Ahmed E. Alprol, Hanan M. Khairy, Abdelaziz M. Nour, Abdallah Tageldein Mansour, Hesham A. Hassanien, Ahmed Gaber, and et al. 2021. "Potential Applications of Native Cyanobacterium Isolate (Arthrospira platensis NIOF17/003) for Biodiesel Production and Utilization of Its Byproduct in Marine Rotifer (Brachionus plicatilis) Production" Sustainability 13, no. 4: 1769. https://doi.org/10.3390/su13041769











