Rhizospheric Phosphate Solubilizing Bacillus atrophaeus GQJK17 S8 Increases Quinoa Seedling, Withstands Heavy Metals, and Mitigates Salt Stress
Abstract
:1. Introduction
2. Materials and Methods
2.1. Soil Sampling and PSB Screening
2.2. P Solubilization Measurement
2.3. In Vitro Assessment of PGP Properties
2.3.1. Indole3-Acetic Acid (IAA) Production Assay
2.3.2. Siderophores Production Assay
2.3.3. Production of Hydrocyanic Acid
2.3.4. Ammonia Production Assay
2.3.5. Cellulase and Protease Production Assay
2.3.6. Bacterial Biofilm Formation
2.3.7. Zinc Solubilization Assay
2.4. PSB Tolerance Assessment
2.4.1. Salt and Heat Tolerance
2.4.2. Antibiotic Resistance Profiling
2.4.3. Heavy Metal Tolerance
2.5. Genotypic Identification
2.6. Seed Germination Assay
2.7. Antifungal Assay
2.8. Statistical Analysis
3. Results
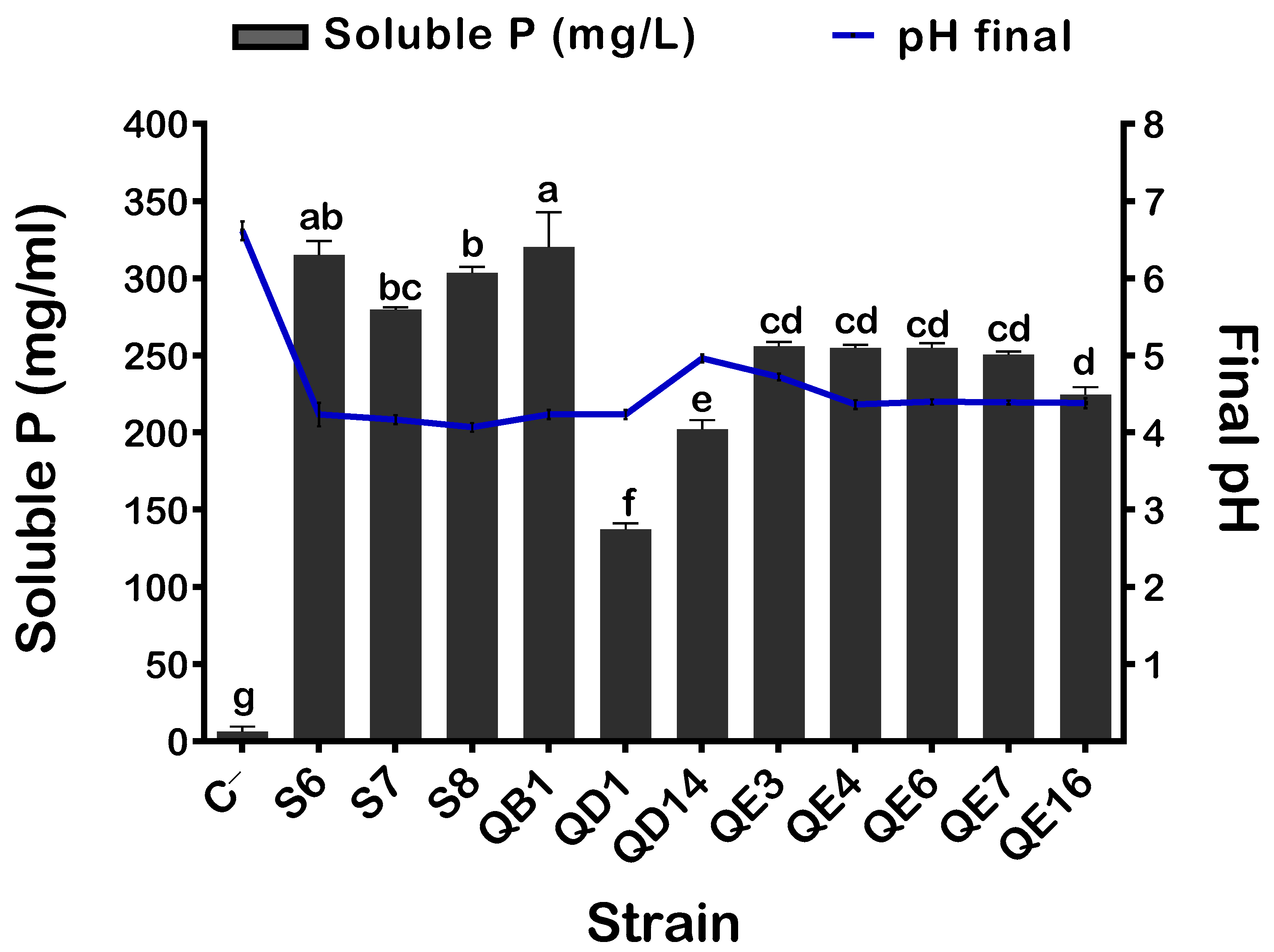
3.1. Isolation of Eleven P Solubilization Rhizobacteria
3.2. The QD1 and QE6 Isolates Are the Best IAA Producers
3.3. The S8 Isolate Tolerated High Salt Concentrations and Is Thermoresistant
3.4. The QB1 Isolate Exhibited a Strong Zn Solubilization
3.5. The QD1, S6, S7, S8 and QB1 Isolates Are The Best Siderophores Producers
3.6. The S6 and S7 Isolates Induced a Strong Biofilm Formation
3.7. Ammonia and HCN Production and the Lack of Cellulase and Protease Activities
3.8. Antibiotic Resistance Profile of Selected PSB Isolates
3.9. Selected PSB Isolates Withstood Heavy Metal Stress
3.10. PSB Isolates Are Related to The Genus of Pseudomonas, Bacillus, and Enterobacter
3.11. Bacillus atrophaeus GQJK17 S8 Strain Induced the Best Seeds Germination and Quinoa Seedling Growth
3.12. Five PSB Strains Exhibit Anti-Fusarium Oxysporum Activity
4. Discussion
4.1. Isolation and Identification of Eleven PSB Strains from Rhizosphere of the Quinoa Plant
4.2. What Are the PGPR Features of Quinoa Isolated PSB Strains?
4.3. Antibiotic, Salt, Heavy Metal, and Rare Elements Resistance Effects on PSB Strain Behavior
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Jacobsen, S.-E. The worldwide potential for quinoa (Chenopodium quinoa Willd.). Food Rev. Int. 2003, 19, 167–177. [Google Scholar] [CrossRef]
- Repo-Carrasco, R.; Espinoza, C.; Jacobsen, S.-E. Nutritional value and use of the Andean crops quinoa (Chenopodium quinoa) and kañiwa (Chenopodium pallidicaule). Food Rev. Int. 2003, 19, 179–189. [Google Scholar] [CrossRef]
- Stikic, R.; Glamoclija, D.; Demin, M.; Vucelic-Radovic, B.; Jovanovic, Z.; Milojkovic-Opsenica, D.; Jacobsen, S.-E.; Milovanovic, M. Agronomical and nutritional evaluation of quinoa seeds (Chenopodium quinoa Willd.) as an ingredient in bread formulations. J. Cereal Sci. 2012, 55, 132–138. [Google Scholar] [CrossRef]
- Benlhabib, O.; Yazar, A.; Qadir, M.; Lourenço, E.; Jacobsen, S.-E. How Can We Improve M editerranean Cropping Systems? J. Agron. Crop Sci. 2014, 200, 325–332. [Google Scholar] [CrossRef]
- Adolf, V.I.; Jacobsen, S.-E.; Shabala, S. Salt tolerance mechanisms in quinoa (Chenopodium quinoa Willd.). Environ. Exp. Bot. 2013, 92, 43–54. [Google Scholar] [CrossRef]
- Jacobsen, S.-E. Adaptation of quinoa (Chenopodium quinoa) to Northern European agriculture: Studies on developmental pattern. Euphytica 1997, 96, 41–48. [Google Scholar] [CrossRef]
- Jacobsen, S.; Bach, A. The influence of temperature on seed germination rate in quinoa (Chenopodium quinoa Willd. Seed Sci. Technol. 1998, 26, 515–523. [Google Scholar]
- Dřímalková, M. Mycoflora of Chenopodium quinoa Willd. seeds. Plant Prot. Sci. 2003, 39, 146–150. [Google Scholar]
- Zablotowicz, R.M.; Tipping, E.M.; Lifshitz, R.; Kloepper, J.W. Plant growth promotion mediated by bacterial rhizosphere colonizers. In The Rhizosphere and Plant Growth; Springer: Berlin/Heidelberg, Germany, 1991; pp. 315–326. [Google Scholar]
- Smith, S.E.; Read, D.J. Mycorrhizal Symbiosis; Academic Press: Cambridge, MA, USA, 2010. [Google Scholar]
- Chen, Y.P.; Rekha, P.D.; Arun, A.B.; Shen, F.T.; Lai, W.A.; Young, C.C. Phosphate solubilizing bacteria from subtropical soil and their tricalcium phosphate solubilizing abilities. Appl. Soil Ecol. 2006, 34, 33–41. [Google Scholar] [CrossRef]
- Wani, P.A.; Zaidi, A.; Khan, A.A.; Khan, M.S. Effect of phorate on phosphate solubilization and indole acetic acid releasing potentials of rhizospheric microorganisms. Ann. Plant Prot. Sci. 2005, 13, 139–144. [Google Scholar]
- Kumar, V.; Kumar Behl, R.; Narula, N. Establishment of phosphate-solubilizing strains of Azotobacter chroococcum in the rhizosphere and their effect on wheat cultivars under green house conditions. Microbiol. Res. 2001, 156, 87–93. [Google Scholar] [CrossRef] [PubMed]
- Chung, H.; Park, M.; Madhaiyan, M.; Seshadri, S.; Song, J.; Cho, H.; Sa, T. Isolation and characterization of phosphate solubilizing bacteria from the rhizosphere of crop plants of Korea. Soil Biol. Biochem. 2005, 37, 1970–1974. [Google Scholar] [CrossRef]
- Pradhan, N.; Sukla, L. Solubilization of inorganic phosphates by fungi isolated from agriculture soil. Afr. J. Biotechnol. 2006, 5, 850–854. [Google Scholar]
- Sharma, K.; Dak, G.; Agrawal, A.; Bhatnagar, M.; Sharma, R. Effect of phosphate solubilizing bacteria on the germination of Cicer arietinum seeds and seedling growth. J. Herb. Med. Toxicol. 2007, 1, 61–63. [Google Scholar]
- Khan, M.S.; Zaidi, A.; Ahemad, M.; Oves, M.; Wani, P.A. Plant growth promotion by phosphate solubilizing fungi–current perspective. Arch. Agron. Soil Sci. 2010, 56, 73–98. [Google Scholar] [CrossRef]
- Hinsinger, P. Bioavailability of soil inorganic P in the rhizosphere as affected by root-induced chemical changes: A review. Plant Soil 2001, 237, 173–195. [Google Scholar] [CrossRef]
- Sharma, S.B.; Sayyed, R.Z.; Trivedi, M.H.; Gobi, T.A. Phosphate solubilizing microbes: Sustainable approach for managing phosphorus deficiency in agricultural soils. Springerplus 2013, 2, 587. [Google Scholar] [CrossRef] [Green Version]
- Hayat, R.; Ali, S.; Amara, U.; Khalid, R.; Ahmed, I. Soil beneficial bacteria and their role in plant growth promotion: A review. Ann. Microbiol. 2010, 60, 579–598. [Google Scholar] [CrossRef]
- Plassard, C.; Robin, A.; Le Cadre, E.; Marsden, C.; Trap, J.; Herrmann, L.; Waithaisong, K.; Lesueur, D.; Blanchart, E.; LARDY, L.; et al. Améliorer la biodisponibilité du phosphore: Comment valoriser les compétences des plantes et les mécanismes biologiques du sol. Innov. Agron. 2015, 43, 115–138. [Google Scholar]
- Rodríguez, H.; Fraga, R. Phosphate solubilizing bacteria and their role in plant growth promotion. Biotechnol. Adv. 1999, 17, 319–339. [Google Scholar] [CrossRef]
- McGill, W.; Cole, C. Comparative aspects of cycling of organic C, N, S and P through soil organic matter. Geoderma 1981, 26, 267–286. [Google Scholar] [CrossRef]
- Khan, N.; Zandi, P.; Ali, S.; Mehmood, A.; Adnan Shahid, M.; Yang, J. Impact of salicylic acid and PGPR on the drought tolerance and phytoremediation potential of Helianthus annus. Front. Microbiol. 2018, 9, 2507. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Biswas, J.; Banerjee, A.; Rai, M.; Naidu, R.; Biswas, B.; Vithanage, M.; Chandra Dash, M.; Sarkar, S.; Meers, E. Potential application of selected metal resistant phosphate solubilizing bacteria isolated from the gut of earthworm (Metaphire posthuma) in plant growth promotion. Geoderma 2018, 330, 117–124. [Google Scholar] [CrossRef]
- Nautiyal, C.S. An efficient microbiological growth medium for screening phosphate solubilizing microorganisms. FEMS Microbiol. Lett. 1999, 170, 265–270. [Google Scholar] [CrossRef] [PubMed]
- Mukhtar, S.; Shahid, I.; Mehnaz, S.; Malik, K. Assessment of two carrier materials for phosphate solubilizing biofertilizers and their effect on growth of wheat (Triticum aestivum L.). Microbiol. Res. 2017, 205. [Google Scholar] [CrossRef] [PubMed]
- Leveau, J.H.; Lindow, S.E. Utilization of the plant hormone indole-3-acetic acid for growth by Pseudomonas putida strain 1290. Appl. Environ. Microbiol. 2005, 71, 2365–2371. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Latif Khan, A.; Ahmed Halo, B.; Elyassi, A.; Ali, S.; Al-Hosni, K.; Hussain, J.; Al-Harrasi, A.; Lee, I.-J. Indole acetic acid and ACC deaminase from endophytic bacteria improves the growth of Solarium lycopersicum. Electron. J. Biotechnol. 2016, 19, 58–64. [Google Scholar] [CrossRef] [Green Version]
- Schwyn, B.; Neilands, J.B. Universal chemical assay for the detection and determination of siderophores. Anal. Biochem. 1987, 160, 47–56. [Google Scholar] [CrossRef]
- Lorck, H. Production of hydrocyanic acid by bacteria. Physiol. Plant. 1948, 1, 142–146. [Google Scholar] [CrossRef]
- Cappuccino, J.C.N.S. Microbiology: A Laboratory Manual, 3rd ed.; Co, B.C.P., Ed.; Pearson: London, UK, 1992. [Google Scholar]
- Chrouqi, L.; Lahcen, O.; Jadrane, I.; Koussa, T.; Alfeddy, M.N. Screening of soil rhizobacteria isolated from wheat plants grown in the Marrakech region (Morocco, North Africa) for plant growth promoting activities. JMES 2017, 8, 3382–3390. [Google Scholar]
- Kasana, R.C.; Salwan, R.; Dhar, H.; Dutt, S.; Gulati, A. A rapid and easy method for the detection of microbial cellulases on agar plates using gram’s iodine. Curr. Microbiol. 2008, 57, 503–507. [Google Scholar] [CrossRef]
- Kavitha, T.; Nelson, R.; Jesi, S.J. Screening of rhizobacteria for plant growth promoting traits and antifungal activity against charcoal rot pathogen Macrophomina phaseolina. Int. J. Pharma Bio Sci. 2013, 4, 177–186. [Google Scholar]
- Smibert, R. Phenotypic characterization. In Methods for General and Molecular Bacteriology; American Society for Microbiology: Washington, DC, USA, 1994; pp. 607–654. [Google Scholar]
- Christensen, G.D.; Simpson, W.A.; Younger, J.; Baddour, L.; Barrett, F.; Melton, D.; Beachey, E. Adherence of coagulase-negative staphylococci to plastic tissue culture plates: A quantitative model for the adherence of staphylococci to medical devices. J. Clin. Microbiol. 1985, 22, 996–1006. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gandhi, A.; Muralidharan, G. Assessment of zinc solubilizing potentiality of Acinetobacter sp. isolated from rice rhizosphere. Eur. J. Soil Biol. 2016, 76, 1–8. [Google Scholar] [CrossRef]
- Khanghahi, M.Y.; Ricciuti, P.; Allegretta, I.; Terzano, R.; Crecchio, C. Solubilization of insoluble zinc compounds by zinc solubilizing bacteria (ZSB) and optimization of their growth conditions. Environ. Sci. Pollut. Res. 2018, 25, 25862–25868. [Google Scholar] [CrossRef] [PubMed]
- Patel, K.S.; Naik, J.H.; Chaudhari, S.; Amaresan, N. Characterization of culturable bacteria isolated from hot springs for plant growth promoting traits and effect on tomato (Lycopersicon esculentum) seedling. C. R. Biol. 2017, 340, 244–249. [Google Scholar] [CrossRef] [PubMed]
- Armalytė, J.; Skerniškytė, J.; Bakienė, E.; Krasauskas, R.; Šiugždinienė, R.; Kareivienė, V.; Kerzienė, S.; Klimienė, I.; Sužiedėlienė, E.; Ružauskas, M. Microbial Diversity and Antimicrobial Resistance Profile in Microbiota From Soils of Conventional and Organic Farming Systems. Front. Microbiol. 2019, 10, 892. [Google Scholar] [CrossRef] [PubMed]
- Halda-Alija, L. Incidence of antibiotic-resistant Klebsiella pneumoniae and Enterobacter species in freshwater wetlands. Lett. Appl. Microbiol. 2004, 39, 445–450. [Google Scholar] [CrossRef]
- Chaoua, S.; Boussaa, S.; El Gharmali, A.; Boumezzough, A. Impact of irrigation with wastewater on accumulation of heavy metals in soil and crops in the region of Marrakech in Morocco. J. Saudi Soc. Agric. Sci. 2019, 18, 429–436. [Google Scholar] [CrossRef]
- Cervantes-Vega, C.; Chavez, J.; Córdova, N.; Amador, J.V. Resistance to metals by Pseudomonas aeruginosa clinical isolates. Microbios 1986, 48, 159–163. [Google Scholar] [PubMed]
- Hill, J.E.; Hemmingsen, S.M.; Town, J.R. Strong PCR Primers and Primer Cocktails. 2009. Available online: https://patents.google.com/patent/US7507535B2/en (accessed on 4 August 2020).
- Letunic, I.; Bork, P. Interactive Tree Of Life (iTOL) v4: Recent updates and new developments. Nucleic Acids Res. 2019, 47, W256–W259. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hussain, M.; Asghar, H.; Arshad, M.; Shahbaz, M. Screening of multi-traits rhizobacteria to improve maize growth under axenic conditions. J. Anim. Plant Sci. 2013, 23, 514–520. [Google Scholar]
- Ji, S.H.; Gururani, M.A.; Chun, S.C. Isolation and characterization of plant growth promoting endophytic diazotrophic bacteria from Korean rice cultivars. Microbiol. Res. 2014, 169, 83–98. [Google Scholar] [CrossRef] [PubMed]
- Suleman, M.; Yasmin, S.; Rasul, M.; Yahya, M.; Atta, B.M.; Mirza, M.S. Phosphate solubilizing bacteria with glucose dehydrogenase gene for phosphorus uptake and beneficial effects on wheat. PLoS ONE 2018, 13, e0204408. [Google Scholar] [CrossRef] [Green Version]
- Islam, S.; Mannan Akanda, A.; Prova, A.; Islam, T.; Hossain, M. Isolation and Identification of Plant Growth Promoting Rhizobacteria from Cucumber Rhizosphere and Their Effect on Plant Growth Promotion and Disease Suppression. Front. Microbiol. 2016, 6, 1360. [Google Scholar] [CrossRef] [Green Version]
- De Boer, W.; Wagenaar, A.-M.; Klein Gunnewiek, P.J.; Van Veen, J.A. In vitro suppression of fungi caused by combinations of apparently non-antagonistic soil bacteria. FEMS Microbiol. Ecol. 2007, 59, 177–185. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ortuño, N.; Castillo, J.A.; Claros, M.; Navia, O.; Angulo, M.; Barja, D.; Gutiérrez, C.; Angulo, V. Enhancing the sustainability of quinoa production and soil resilience by using bioproducts made with native microorganisms. Agronomy 2013, 3, 732–746. [Google Scholar] [CrossRef] [Green Version]
- Beneduzi, A.; Ambrosini, A.; Passaglia, L.M. Plant growth-promoting rhizobacteria (PGPR): Their potential as antagonists and biocontrol agents. Genet. Mol. Biol. 2012, 35, 1044–1051. [Google Scholar] [CrossRef] [Green Version]
- Sella, S.R.; Vandenberghe, L.P.; Soccol, C.R. Bacillus atrophaeus: Main characteristics and biotechnological applications—A review. Crit. Rev. Biotechnol. 2015, 35, 533–545. [Google Scholar] [CrossRef] [PubMed]
- Kloepper, J.W.; Ryu, C.-M.; Zhang, S. Induced systemic resistance and promotion of plant growth by Bacillus spp. Phytopathology 2004, 94, 1259–1266. [Google Scholar] [CrossRef] [Green Version]
- Choudhary, D.K.; Johri, B.N. Interactions of Bacillus spp. and plants—With special reference to induced systemic resistance (ISR). Microbiol. Res. 2009, 164, 493–513. [Google Scholar] [CrossRef]
- Walsh, U.F.; Morrissey, J.P.; O’Gara, F. Pseudomonas for biocontrol of phytopathogens: From functional genomics to commercial exploitation. Curr. Opin. Biotechnol. 2001, 12, 289–295. [Google Scholar] [CrossRef]
- Howell, C.; Beier, R.; Stipanovic, R. Production of ammonia by Enterobacter cloacae and its possible role in the biological control of Pythium preemergence damping-off by the bacterium. Phytopathology 1988, 78, 1075–1078. [Google Scholar] [CrossRef]
- Agbodjato, N.A.; Noumavo, P.A.; Baba-Moussa, F.; Salami, H.A.; Sina, H.; Sèzan, A.; Bankolé, H.; Adjanohoun, A.; Baba-Moussa, L. Characterization of potential plant growth promoting rhizobacteria isolated from Maize (Zea mays L.) in central and Northern Benin (West Africa). Appl. Environ. Soil Sci. 2015, 2015. [Google Scholar] [CrossRef] [Green Version]
- Bensidhoum, L.; Nabti, E.; Tabli, N.; Kupferschmied, P.; Weiss, A.; Rothballer, M.; Schmid, M.; Keel, C.; Hartmann, A. Heavy metal tolerant Pseudomonas protegens isolates from agricultural well water in northeastern Algeria with plant growth promoting, insecticidal and antifungal activities. Eur. J. Soil Biol. 2016, 75, 38–46. [Google Scholar] [CrossRef] [Green Version]
- Orhan, F. Alleviation of salt stress by halotolerant and halophilic plant growth-promoting bacteria in wheat (Triticum aestivum). Braz. J. Microbiol. 2016, 47, 621–627. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Shahid, M.; Hameed, S.; Zafar, M.; Tahir, M.; Ijaz, M.; Tariq, M.; Hussain, K.; Ali, A. Enterobacter sp. strain Fs-11 adapted to diverse ecological conditions and promoted sunflower achene yield, nutrient uptake, and oil contents. Braz. J. Microbiol. 2019, 50, 459–469. [Google Scholar] [CrossRef]
- Mahdi, I.; Fahsi, N.; Hafidi, M.; Allaoui, A.; Biskri, L. Plant Growth Enhancement using Rhizospheric Halotolerant Phosphate Solubilizing Bacterium Bacillus licheniformis QA1 and Enterobacter asburiae QF11 Isolated from Chenopodium quinoa Willd. Microorganisms 2020, 8, 948. [Google Scholar] [CrossRef]
- Lau, Y.Y.; Sulaiman, J.; Chen, J.W.; Yin, W.-F.; Chan, K.-G. Quorum sensing activity of Enterobacter asburiae isolated from lettuce leaves. Sensors 2013, 13, 14189–14199. [Google Scholar] [CrossRef]
- Smith, R.S.; Iglewski, B.H.P. aeruginosa quorum-sensing systems and virulence. Curr. Opin. Microbiol. 2003, 6, 56–60. [Google Scholar] [CrossRef]
- Lalucat, J.; Bennasar, A.; Bosch, R.; García-Valdés, E.; Palleroni, N.J. Biology of Pseudomonas stutzeri. Microbiol. Mol. Biol. Rev. 2006, 70, 510–547. [Google Scholar] [CrossRef] [Green Version]
- Hallmann, J.; Quadt-Hallmann, A.; Mahaffee, W.; Kloepper, J. Bacterial endophytes in agricultural crops. Can. J. Microbiol. 1997, 43, 895–914. [Google Scholar] [CrossRef]
- Barraquio, W.L.; Segubre, E.M.; Gonzalez, M.A.S.; Verma, S.C.; James, E.K.; Ladha, J.K.; Tripathi, A.K. Diazotrophic enterobacteria: What is their role in the rhizosphere of rice. In Quest Nitrogen Fixat Rice; International Rice Research Institute: Los Baños, Philippines, 2000; pp. 93–118. [Google Scholar]
- MEYER, J.-M.; Abdallah, M.A. The siderochromes of non-fluorescent pseudomonads: Production of nocardamine by Pseudomonas stutzeri. Microbiology 1980, 118, 125–129. [Google Scholar] [CrossRef] [Green Version]
- Meyer, J.-M.; Geoffroy, V.A.; Baida, N.; Gardan, L.; Izard, D.; Lemanceau, P.; Achouak, W.; Palleroni, N.J. Siderophore typing, a powerful tool for the identification of fluorescent and nonfluorescent pseudomonads. Appl. Environ. Microbiol. 2002, 68, 2745–2753. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chakraborty, R.N.; Patel, H.N.; Desai, S.B. Isolation and partial characterization of catechol-type siderophore fromPseudomonas stutzeri RC 7. Curr. Microbiol. 1990, 20, 283–286. [Google Scholar] [CrossRef]
- Lim, H.-S.; Kim, Y.-S.; Kim, S.-D. Pseudomonas stutzeri YPL-1 genetic transformation and antifungal mechanism against Fusarium solani, an agent of plant root rot. Appl. Environ. Microbiol. 1991, 57, 510–516. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Seneviratne, G.; Weerasekara, M.; Seneviratne, K.; Zavahir, J.; Kecskés, M.; Kennedy, I. Importance of biofilm formation in plant growth promoting rhizobacterial action. In Plant Growth and Health Promoting Bacteria; Springer: Berlin/Heidelberg, Germany, 2010; pp. 81–95. [Google Scholar]
- Mann, E.E.; Wozniak, D.J. Pseudomonas biofilm matrix composition and niche biology. FEMS Microbiol. Rev. 2012, 36, 893–916. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gupta, G.; Snehi, S.K.; Singh, V. Role of PGPR in biofilm formations and its importance in plant health. Biofilms Plant Soil Health 2017, 27. [Google Scholar] [CrossRef]
- Ali, S.; Shah, A.; Arif, M.; Miraj, G.; Ali, I.; Sajjad, M.; Farhatullah, M. Khan and NM Khan. 2009. Enhanc. Wheat Grain Yield Yield Compon. Foliar Appl. Zinc Boron. Sarhad J. Agric. 2009, 25, 15–19. [Google Scholar]
- Gregory, P.J.; Wahbi, A.; Adu-Gyamfi, J.; Heiling, M.; Gruber, R.; Joy, E.J.M.; Broadley, M.R. Approaches to reduce zinc and iron deficits in food systems. Glob. Food Secur. 2017, 15, 1–10. [Google Scholar] [CrossRef] [Green Version]
- Ahmad, H.R.; Aziz, T.; Hussain, S.; Akraam, M.; Sabir, M.; Hanafi, M.M. Zinc-enriched farm yard manure improves grain yield and grain zinc concentration in rice grown on a saline-sodic soil. Int. J. Agric. Biol. 2012, 14, 787–792. [Google Scholar]
- Isaure, M.-P.; Laboudigue, A.; Manceau, A.; Sarret, G.; Tiffreau, C.; Trocellier, P.; Lamble, G.; Hazemann, J.-L.; Chateigner, D. Quantitative Zn speciation in a contaminated dredged sediment by μ-PIXE, μ-SXRF, EXAFS spectroscopy and principal component analysis. Geochim. Cosmochim. Acta 2002, 66, 1549–1567. [Google Scholar] [CrossRef]
- Adele, N.C.; Ngwenya, B.T.; Heal, K.V.; Mosselmans, J.F.W. Soil bacteria override speciation effects on zinc phytotoxicity in zinc-contaminated soils. Environ. Sci. Technol. 2018, 52, 3412–3421. [Google Scholar] [CrossRef] [Green Version]
- Saravanan, V.; Kumar, M.R.; Sa, T. Microbial zinc solubilization and their role on plants. In Bacteria in Agrobiology: Plant Nutrient Management; Springer: Berlin/Heidelberg, Germany, 2011; pp. 47–63. [Google Scholar]
- Saravanan, V.S.; Subramoniam, S.R.; Raj, S.A. Assessing in vitro solubilization potential of different zinc solubilizing bacterial (ZSB) isolates. Braz. J. Microbiol. 2004, 35, 121–125. [Google Scholar] [CrossRef]
- Khan, N.; Zandi, P.; Ali, S.; Mehmood, A.; Shahid, M.A.; Yang, J. Corrigendum: Impact of Salicylic acid and PGPR on the Drought Tolerance and Phytoremediation potential of Helianthus annus. Front. Microbiol. 2019, 10, 2222. [Google Scholar] [CrossRef] [Green Version]
- Kang, S.M.; Radhakrishnan, R.; You, Y.H.; Khan, A.L.; Lee, K.E.; Lee, J.D.; Lee, I.J. Enterobacter asburiae KE 17 association regulates physiological changes and mitigates the toxic effects of heavy metals in soybean. Plant Biol. 2015, 17, 1013–1022. [Google Scholar] [CrossRef] [PubMed]
- Joonu, J.; Averal, H.I. Heavy metal resistant CZC genes identification in Bacillus cereus, Enterobacter asburiae and Pseudomonas aeruginosa isolated from BHEL industry, Tamilnadu. J. Microbiol. Biotechnol. 2016, 5, 27–31. [Google Scholar]
- Liu, F.; Yang, J.; Xiao, Y.; Li, L.; Yang, F.; Jin, Q. Complete genome sequence of a clinical isolate of Enterobacter asburiae. Genome Announc. 2016, 4. [Google Scholar] [CrossRef] [Green Version]
- Hashem, A.; Abed, K. Arsenic, lead and microorganisms in hair and nails of some women from Saudi Arabia. J. Med. Sci. 2002, 2, 82–84. [Google Scholar] [CrossRef] [Green Version]
- Calomiris, J.J.; Armstrong, J.L.; Seidler, R.J. Association of metal tolerance with multiple antibiotic resistance of bacteria isolated from drinking water. Appl. Environ. Microbiol. 1984, 47, 1238–1242. [Google Scholar] [CrossRef] [Green Version]
- Abelenda-Alonso, G.; Padullés, A.; Rombauts, A.; Gudiol, C.; Pujol, M.; Alvarez-Pouso, C.; Jodar, R.; Carratalà, J. Antibiotic prescription during the COVID-19 pandemic: A biphasic pattern. Infect. Control Hosp. Epidemiol. 2020, 41, 1371–1372. [Google Scholar] [CrossRef]
- Ramakrishna, W.; Yadav, R.; Li, K. Plant growth promoting bacteria in agriculture: Two sides of a coin. Appl. Soil Ecol. 2019, 138, 10–18. [Google Scholar] [CrossRef]
- Kang, Y.; Shen, M.; Xia, D.; Ye, K.; Zhao, Q.; Hu, J. Caution of intensified spread of antibiotic resistance genes by inadvertent introduction of beneficial bacteria into soil. Acta Agric. Scand. Sect. B Soil Plant Sci. 2017, 67, 576–582. [Google Scholar] [CrossRef]
- Numan, M.; Bashir, S.; Khan, Y.; Mumtaz, R.; Shinwari, Z.K.; Khan, A.L.; Khan, A.; Al-Harrasi, A. Plant growth promoting bacteria as an alternative strategy for salt tolerance in plants: A review. Microbiol. Res. 2018, 209, 21–32. [Google Scholar] [CrossRef] [PubMed]
- Bakhshandeh, E.; Rahimian, H.; Pirdashti, H.; Nematzadeh, G.A. Phosphate solubilization potential and modeling of stress tolerance of rhizobacteria from rice paddy soil in northern Iran. World J. Microbiol. Biotechnol. 2014, 30, 2437–2447. [Google Scholar] [CrossRef] [PubMed]









| Bacterial Isolate | Extreme Properties | Zinc Solubilization | Siderophores Production | HCN Production | |||
|---|---|---|---|---|---|---|---|
| NaCl | Temperature | ZnO | Zn3(PO4)2 | CO3Zn | |||
| C− | 3% | 37 °C | + | − | − | − | + |
| S6 | 3% | 37 °C | + | +++ | + | ++ | +++ |
| S7 | 3% | 37 °C | + | ++ | + | ++ | + |
| S8 | 11% | 42 °C | ++ | ++ | + | ++ | ++ |
| QB1 | <5% | 42 °C | +++ | +++ | +++ | ++ | ++ |
| QD1 | <5% | 37 °C | ++ | − | − | +++ | +++ |
| QD14 | <5% | 42 °C | +++ | − | − | + | ++ |
| QE3 | <5% | 42 °C | ++ | + | ++ | − | ++ |
| QE4 | <5% | 42 °C | ++ | + | ++ | − | + |
| QE6 | <5% | 42 °C | ++ | + | ++ | + | ++ |
| QE7 | <5% | 42 °C | ++ | + | ++ | + | ++ |
| QE16 | <5% | 42 °C | +++ | ++ | − | + | +++ |
| Ampicillin | Chloramphenicol | Streptomycin | Spectinomycin | Kanamycin | Tetracycline | |
|---|---|---|---|---|---|---|
| Isolates | (100 μg·mL−1) | (20 μg·mL−1) | (100 μg·mL−1) | (60 μg·mL−1) | (50 μg·mL−1) | (10 μg·mL−1) |
| S6 | + | + | − | − | − | − |
| S7 | + | + | − | − | − | − |
| S8 | − | − | − | − | − | − |
| QB1 | − | − | − | − | − | − |
| QD1 | − | − | − | − | − | − |
| QD14 | − | − | − | − | − | − |
| QE3 | + | − | − | − | − | + |
| QE4 | + | − | − | − | − | + |
| QE6 | − | − | − | − | − | − |
| QE7 | − | − | − | − | − | − |
| QE16 | + | − | − | − | − | + |
| Isolate | Heavy Metal (mg·L−1) | ||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| CdSO4 | N2NiO8 | CuO4S.5H2O | |||||||||||||
| 0.1 | 0.5 | 1 | 1.5 | 2 | 0.1 | 0.5 | 1 | 1.5 | 2 | 0.1 | 0.5 | 1 | 1.5 | 2 | |
| S6 | + | − | − | − | − | + | − | − | − | − | + | − | − | − | − |
| S7 | + | − | − | − | − | + | + | − | − | − | + | − | − | − | − |
| S8 | + | − | − | − | − | + | + | − | − | − | + | − | − | − | − |
| QB1 | + | + | + | + | − | + | + | − | − | − | + | + | − | − | − |
| QD1 | − | − | − | − | − | + | − | − | − | − | + | − | − | − | − |
| QD14 | + | + | + | + | − | + | + | − | − | − | + | + | + | − | − |
| QE3 | + | + | + | + | − | + | + | − | − | − | + | + | + | − | − |
| QE4 | + | + | + | + | − | + | + | − | − | − | + | + | + | − | − |
| QE6 | + | + | + | + | − | + | + | − | − | − | + | + | + | − | − |
| QE7 | + | + | + | + | − | + | + | − | − | − | + | + | + | − | − |
| QE16 | + | + | + | + | − | + | + | − | − | − | + | + | + | − | − |
| Genus | Isolate | Homologous Microorganism | Identity (%) | Accession No. |
|---|---|---|---|---|
| Pseudomonas | S6 | P. frederiksbergensis | 99 | MW295959 |
| S7 | Pseudomonas sp. | 99 | MW295960 | |
| QD1 | P. stutzeri | 100 | MW296888 | |
| Bacillus | S8 | Bacillus atrophaeus GQJK17 | 100 | MW295957 |
| Enterobacter | QB1 | E. asburiae | 99 | MW295958 |
| QD14 | E. asburiae | 99 | MW295961 | |
| QE3 | Enterobacter sp. | 99 | MW295962 | |
| QE4 | E. asburiae | 99 | MW295963 | |
| QE6 | E. asburiae | 99 | MW295964 | |
| QE7 | E. hormaechei subsp. xiangfangensis | 99 | MW296887 | |
| QE16 | E. asburiae | 99 | MW295965 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mahdi, I.; Fahsi, N.; Hafidi, M.; Benjelloun, S.; Allaoui, A.; Biskri, L. Rhizospheric Phosphate Solubilizing Bacillus atrophaeus GQJK17 S8 Increases Quinoa Seedling, Withstands Heavy Metals, and Mitigates Salt Stress. Sustainability 2021, 13, 3307. https://doi.org/10.3390/su13063307
Mahdi I, Fahsi N, Hafidi M, Benjelloun S, Allaoui A, Biskri L. Rhizospheric Phosphate Solubilizing Bacillus atrophaeus GQJK17 S8 Increases Quinoa Seedling, Withstands Heavy Metals, and Mitigates Salt Stress. Sustainability. 2021; 13(6):3307. https://doi.org/10.3390/su13063307
Chicago/Turabian StyleMahdi, Ismail, Nidal Fahsi, Mohamed Hafidi, Saad Benjelloun, Abdelmounaaim Allaoui, and Latefa Biskri. 2021. "Rhizospheric Phosphate Solubilizing Bacillus atrophaeus GQJK17 S8 Increases Quinoa Seedling, Withstands Heavy Metals, and Mitigates Salt Stress" Sustainability 13, no. 6: 3307. https://doi.org/10.3390/su13063307








