Essential Oil of Ipomoea carnea: Chemical Profile, Chemometric Analysis, Free Radical Scavenging, and Antibacterial Activities
Abstract
:1. Introduction
2. Materials and Methods
2.1. Plant Materials
2.2. EO Extraction Analysis and Characterization
2.3. Antioxidant Activity
2.4. Antibacterial Activity
2.5. Data Analysis
3. Results and Discussion
3.1. Chemical Profile of I. carnea EO
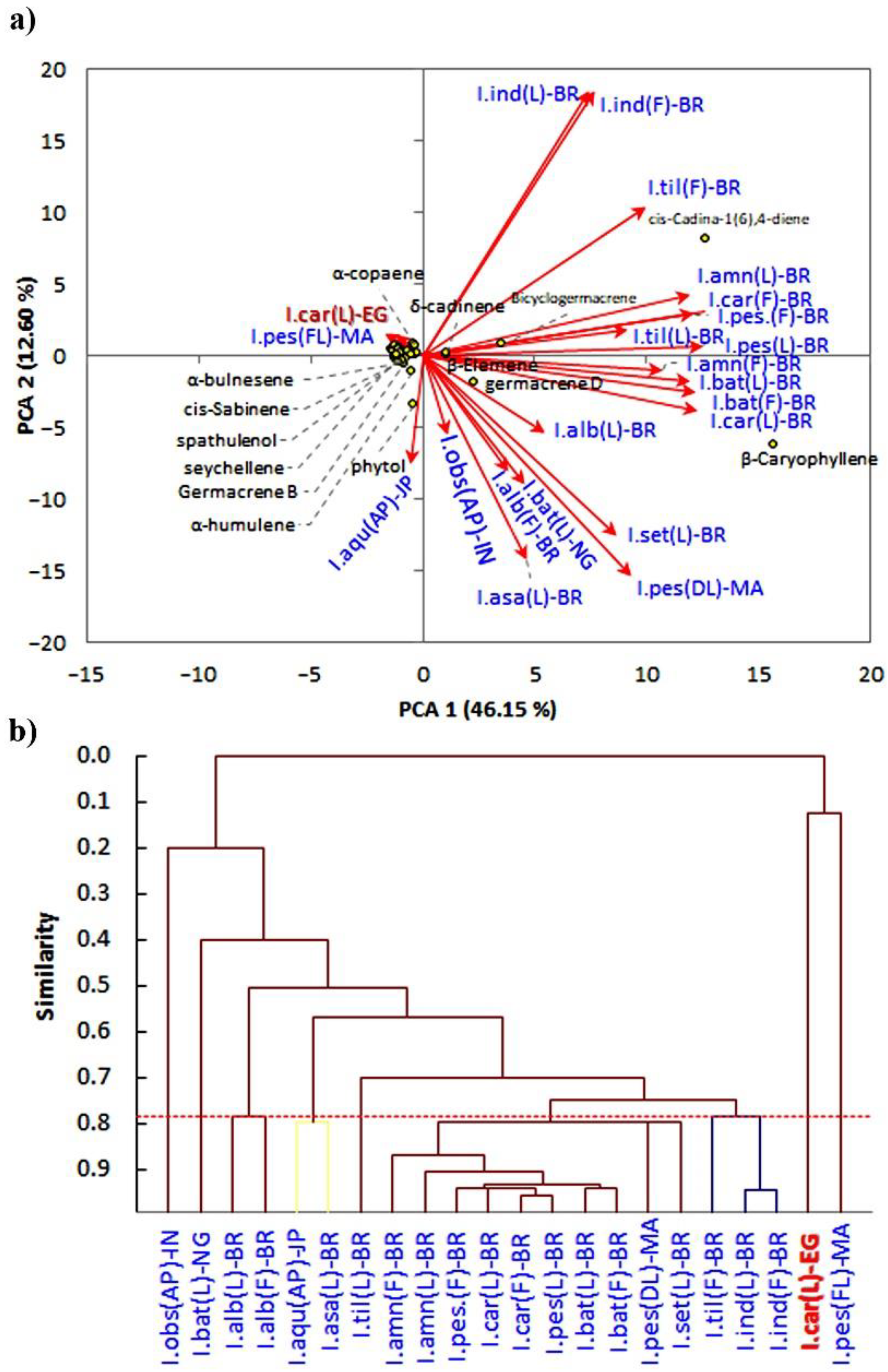
3.2. Chemometric Analysis of the EOs of Ipomea Specie
3.3. Free Radical Scavenging Activity of I. carnea EO
3.4. Antibacterial Activity of I. carnea EO
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Salmerón-Manzano, E.; Garrido-Cardenas, J.A.; Manzano-Agugliaro, F. Worldwide research trends on medicinal plants. Int. J. Environ. Res. Public Health 2020, 17, 3376. [Google Scholar] [CrossRef]
- Sharma, S.; Barkauskaite, S.; Jaiswal, A.K.; Jaiswal, S. Essential oils as additives in active food packaging. Food Chem. 2021, 343, 128403. [Google Scholar] [CrossRef]
- Maurya, A.; Prasad, J.; Das, S.; Dwivedy, A.K. Essential oils and their application in food safety. Front. Sustain. Food Syst. 2021, 5, 133. [Google Scholar] [CrossRef]
- Abd-ElGawad, A.M.; El Gendy, A.E.-N.G.; Assaeed, A.M.; Al-Rowaily, S.L.; Alharthi, A.S.; Mohamed, T.A.; Nassar, M.I.; Dewir, Y.H.; Elshamy, A.I. Phytotoxic effects of plant essential oils: A systematic review and structure-activity relationship based on chemometric analyses. Plants 2020, 10, 36. [Google Scholar] [CrossRef]
- Ammar, N.M.; Hassan, H.; Ahmed, R.; El Gendy, A.E.-N.; Abd-ElGawad, A.; Farrag, A.R.; Farag, A.R.; Elshamy, A.; Afifi, S. Gastro-protective effect of Artemisia sieberi essential oil against ethanol-induced ulcer in rats as revealed via biochemical, histopathological and metabolomics analysis. Biomarkers 2022, 27, 247–257. [Google Scholar] [CrossRef]
- Fayed, E.M.; Abd-EIGawad, A.M.; Elshamy, A.I.; El-Halawany, E.S.F.; EI-Amier, Y.A. Essential oil of Deverra tortuosa aerial parts: Detailed chemical profile, allelopathic, antimicrobial, and antioxidant activities. Chem. Biodivers. 2021, 18, e2000914. [Google Scholar] [CrossRef]
- Abd-ElGawad, A.M.; Elgamal, A.M.; Ei-Amier, Y.A.; Mohamed, T.A.; El Gendy, A.G.; Elshamy, A.I. Chemical composition, allelopathic, antioxidant, and anti-inflammatory activities of sesquiterpenes rich essential oil of Cleome amblyocarpa Barratte & Murb. Plants 2021, 10, 1294. [Google Scholar]
- Abd-ElGawad, A.M.; Elshamy, A.I.; El-Nasser El Gendy, A.; Al-Rowaily, S.L.; Assaeed, A.M. Preponderance of oxygenated sesquiterpenes and diterpenes in the volatile oil constituents of Lactuca serriola L. revealed antioxidant and allelopathic activity. Chem. Biodivers. 2019, 16, e1900278. [Google Scholar] [CrossRef]
- Chouhan, S.; Sharma, K.; Guleria, S. Antimicrobial activity of some essential oils—Present status and future perspectives. Medicines 2017, 4, 58. [Google Scholar] [CrossRef] [Green Version]
- Fisher, K.; Phillips, C. Potential antimicrobial uses of essential oils in food: Is citrus the answer? Trends Food Sci. Technol. 2008, 19, 156–164. [Google Scholar] [CrossRef]
- Austin, D.F.; Staples, G.W.; Simão-Bianchini, R. A synopsis of Ipomoea (Convolvulaceae) in the Americas: Further corrections, changes, and additions. Taxon 2015, 64, 625–633. [Google Scholar] [CrossRef]
- Bovell-Benjamin, A.C. Sweet potato: A review of its past, present, and future role in human nutrition. Adv. Food Nutr. Res. 2007, 52, 1–59. [Google Scholar] [PubMed]
- Marie, D.E.; Dejan, B.; Quetin-Leclercq, J. GC-MS Analysis of the leaf essential oil of Ipomea pes-caprae, a traditional herbal medicine in mauritius. Nat. Prod. Commun. 2007, 2, 1225–1228. [Google Scholar] [CrossRef] [Green Version]
- Jenett-Siems, K.; Weigl, R.; Kaloga, M.; Schulz, J.; Eich, E. Ipobscurines C and D: Macrolactam-type indole alkaloids from the seeds of Ipomoea obscura. Phytochemistry 2003, 62, 1257–1263. [Google Scholar] [CrossRef]
- Kourouma, V.; Mu, T.H.; Zhang, M.; Sun, H.N. Comparative study on chemical composition, polyphenols, flavonoids, carotenoids and antioxidant activities of various cultivars of sweet potato. Int. J. Food Sci. Technol. 2020, 55, 369–378. [Google Scholar] [CrossRef]
- Fatima, N.; Rahman, M.M.; Khan, M.A.; Fu, J. A review on Ipomoea carnea: Pharmacology, toxicology and phytochemistry. J. Complement. Integr. Med. 2014, 11, 55–62. [Google Scholar] [CrossRef]
- Wadnerwar, N.; Deogade, M. Future perspectives of therapeutic claims of an ethnopharmacological drug Ipomoea carnea Jacq.-A critical review. Int. J. Ayurvedic Med. 2022, 12, 177–184. [Google Scholar] [CrossRef]
- Padhi, S.; Tayung, K. Antimicrobial activity and molecular characterization of an endophytic fungus, Quambalaria sp. isolated from Ipomoea carnea. Ann. Microbiol. 2013, 63, 793–800. [Google Scholar] [CrossRef]
- Khan, M.Z.I.; Zahra, S.S.; Ahmed, M.; Fatima, H.; Mirza, B.; Haq, I.-u.; Khan, S.U. Polyphenolic profiling of Ipomoea carnea Jacq. by HPLC-DAD and its implications in oxidative stress and cancer. Nat. Prod. Res. 2019, 33, 2099–2104. [Google Scholar] [CrossRef]
- Ambiga, S.; Jeyaraj, M. Evaluation of in vitro antioxidant activity of Ipomoea carnea Jacq. Int. J. Curr. Microbiol. Appl. Sci. 2015, 4, 327–338. [Google Scholar]
- Khalid, M.S.; Singh, R.K.; Kumar, S.J.; Suresh, D.; Rao, S.K.; Reddy, N.I. Antidiabetic activity of aqueous extract of Ipomoea carnea leaves in streptozotocin induced diabetic rats. Int. J. Pharmacol. Biol. Sci. 2011, 5, 45–54. [Google Scholar]
- Hueza, I.M.; Górniak, S.L. The immunomodulatory effects of Ipomoea carnea in rats vary depending on life stage. Hum. Exp. Toxicol. 2011, 30, 1690–1700. [Google Scholar] [CrossRef] [PubMed]
- Shukla, R.; Gupta, G.; Kashaw, S.K.; Jain, A.P.; Lodhi, S. Wound healing effect of ethanolic extract from morning glory (Ipomoea carnea Jacq.) leaves by using different models in rats. Pak. J. Pharm. Sci. 2018, 31, 1355–1361. [Google Scholar] [PubMed]
- Khatiwora, E.; Adsul, V.B.; Kulkarni, M.M.; Deshpande, N.; Kashalkar, R. Spectroscopic determination of total phenol and flavonoid contents of Ipomoea carnea. Int. J. ChemTech Res. 2010, 2, 1698–1701. [Google Scholar]
- Haraguchi, M.; Gorniak, S.L.; Ikeda, K.; Minami, Y.; Kato, A.; Watson, A.A.; Nash, R.J.; Molyneux, R.J.; Asano, N. Alkaloidal components in the poisonous plant, Ipomoea carnea (Convolvulaceae). J. Agric. Food Chem. 2003, 51, 4995–5000. [Google Scholar] [CrossRef]
- Tenório, T.M.; Moraes, M.M.; Camara, C.A.; Araujo, C.A.; Silva, M.M.; Rodrigues, L.V. Scents from the Brazilian Atlantic Forest Biome: Chemical composition of essential oils from the leaves and flowers of seven species of Ipomoea (Convolvulaceae). J. Essent. Oil Res. 2021, 33, 567–583. [Google Scholar] [CrossRef]
- Kameoka, H.; Kubo, K.; Miyazawa, M. Essential oil components of water-convolvulus (Ipomoea aquatica Forsk.). J. Essent. Oil Res. 1992, 4, 219–222. [Google Scholar] [CrossRef]
- Joshi, R.K. Sesquiterpene-rich volatile constituents of Ipomoea obscura (L.) Ker-Gawl. Nat. Prod. Res. 2015, 29, 1935–1937. [Google Scholar] [CrossRef]
- da Silva Júnior, O.S.; Franco, C.d.J.P.; de Moraes, A.A.B.; Cruz, J.N.; da Costa, K.S.; do Nascimento, L.D.; de Aguiar Andrade, E.H. In silico analyses of toxicity of the major constituents of essential oils from two Ipomoea L. species. Toxicon 2021, 195, 111–118. [Google Scholar] [CrossRef]
- Ogunmoye, A.; Adebayo, M.A.; Inikpi, E.; Ogunwande, I.A. Chemical constituents of essential oil from the leaves of Ipomoea batatas L. (Lam.). Int. Res. J. Pure Appl. Chem. 2015, 7, 42–48. [Google Scholar] [CrossRef]
- Tackholm, V. Students’ Flora of Egypt, 2nd ed.; Cairo University Press: Cairo, Egypt, 1974. [Google Scholar]
- Boulos, L. Flora of Egypt; All Hadara Publishing: Cairo, Egypt, 1995; Volume 1. [Google Scholar]
- Abd-ELGawad, A.M.; Al-Rowaily, S.L.; Assaeed, A.M.; Ei-Amier, Y.A.; El Gendy, A.E.-N.G.; Omer, E.; Al-Dosari, D.H.; Bonanomi, G.; Kassem, H.S.; Elshamy, A.I. Comparative chemical profiles and phytotoxic activity of essential oils of two ecospecies of Pulicaria undulata (L.) CA Mey. Plants 2021, 10, 2366. [Google Scholar] [CrossRef] [PubMed]
- Miguel, M.G. Antioxidant activity of medicinal and aromatic plants. Flavour Fragr. J. 2010, 25, 219–312. [Google Scholar] [CrossRef]
- Re, R.; Pellegrini, N.; Proteggente, A.; Pannala, A.; Yang, M.; Rice-Evans, C. Antioxidant activity applying an improved ABTS radical cation decolorization assay. Free Radic. Biol. Med. 1999, 26, 1231–1237. [Google Scholar] [CrossRef]
- Lorian, V. Antibiotics in Laboratory Medicine; Lippincott Williams & Wilkins: Baltimore, PA, USA, 2005. [Google Scholar]
- Abd-ElGawad, A.M.; El-Amier, Y.A.; Assaeed, A.M.; Al-Rowaily, S.L. Interspecific variations in the habitats of Reichardia tingitana (L.) Roth leading to changes in its bioactive constituents and allelopathic activity. Saudi J. Biol. Sci. 2020, 27, 489–499. [Google Scholar] [CrossRef] [PubMed]
- Figueiredo, A.C.; Barroso, J.G.; Pedro, L.G.; Scheffer, J.J. Factors affecting secondary metabolite production in plants: Volatile components and essential oils. Flavour Fragr. J. 2008, 23, 213–226. [Google Scholar] [CrossRef]
- Barra, A. Factors affecting chemical variability of essential oils: A review of recent developments. Nat. Prod. Commun. 2009, 4, 1147–1154. [Google Scholar] [CrossRef] [Green Version]
- Al-Rowaily, S.L.; Abd-ElGawad, A.M.; Assaeed, A.M.; Elgamal, A.M.; Gendy, A.E.-N.G.E.; Mohamed, T.A.; Dar, B.A.; Mohamed, T.K.; Elshamy, A.I. Essential oil of Calotropis procera: Comparative chemical profiles, antimicrobial activity, and allelopathic potential on weeds. Molecules 2020, 25, 5203. [Google Scholar] [CrossRef]
- Saleh, I.; Abd-ElGawad, A.; El Gendy, A.E.-N.; Abd El Aty, A.; Mohamed, T.; Kassem, H.; Aldosri, F.; Elshamy, A.; Hegazy, M.-E.F. Phytotoxic and antimicrobial activities of Teucrium polium and Thymus decussatus essential oils extracted using hydrodistillation and microwave-assisted techniques. Plants 2020, 9, 716. [Google Scholar] [CrossRef]
- da Silva, W.M.F.; Kringel, D.H.; de Souza, E.J.D.; da Rosa Zavareze, E.; Dias, A.R.G. Basil essential oil: Methods of extraction, chemical composition, biological activities, and food applications. Food Bioprocess Technol. 2021, 15, 1–27. [Google Scholar] [CrossRef]
- Elshamy, A.; Abd-ElGawad, A.; Mohamed, T.; El Gendy, A.E.N.; Abd El Aty, A.A.; Saleh, I.; Moustafa, M.F.; Hussien, T.A.; Pare, P.W.; Hegazy, M.E.F. Extraction development for antimicrobial and phytotoxic essential oils from Asteraceae species: Achillea fragrantissima, Artemisia judaica and Tanacetum sinaicum. Flavour Fragr. J. 2021, 36, 352–364. [Google Scholar] [CrossRef]
- Alam, I.; Forid, S.; Roney, M.; Aluwi, F.F.M.; Huq, M. Antioxidant and antibacterial activity of Ipomoea mauritiana Jacq.: A traditionally used medicinal plant in Bangladesh. Clin. Phytosci. 2020, 6, 35. [Google Scholar] [CrossRef]
- Qasim, M.; Abideen, Z.; Adnan, M.; Gulzar, S.; Gul, B.; Rasheed, M.; Khan, M. Antioxidant properties, phenolic composition, bioactive compounds and nutritive value of medicinal halophytes commonly used as herbal teas. S. Afr. J. Bot. 2017, 110, 240–250. [Google Scholar] [CrossRef]
- Abd-ElGawad, A.M.; El-Amier, Y.A.; Bonanomi, G.; Gendy, A.E.-N.G.E.; Elgorban, A.M.; Alamery, S.F.; Elshamy, A.I. Chemical composition of Kickxia aegyptiaca essential oil and its potential antioxidant and antimicrobial activities. Plants 2022, 11, 594. [Google Scholar] [CrossRef] [PubMed]
- Hammad, H.M.; Matar, S.A.; Litescu, S.-C.; Abuhamdah, S.; Al-Jaber, H.I.; Afifi, F.U. Biological activities of the hydro-alcoholic and aqueous extracts of Achillea fragrantissima (Forssk.) grown in Jordan. Nat. Sci. 2014, 6, 23–30. [Google Scholar]
- Abd-ElGawad, A.M.; El Gendy, A.E.-N.G.; Assaeed, A.M.; Al-Rowaily, S.L.; Omer, E.A.; Dar, B.A.; Al-Taisan, W.a.A.; Elshamy, A.I. Essential oil enriched with oxygenated constituents from invasive plant Argemone ochroleuca exhibited potent phytotoxic effects. Plants 2020, 9, 998. [Google Scholar] [CrossRef] [PubMed]
- Abd El-Gawad, A.M. Chemical constituents, antioxidant and potential allelopathic effect of the essential oil from the aerial parts of Cullen plicata. Ind. Crops Prod. 2016, 80, 36–41. [Google Scholar] [CrossRef]
- Maizi, Y.; Meddah, B.; Tir Touil Meddah, A.; Gabaldon Hernandez, J.A. Seasonal variation in essential oil content, chemical composition and antioxidant activity of Teucrium polium L. growing in Mascara (North West of Algeria). J. Appl. Biotechnol. Rep. 2019, 6, 151–157. [Google Scholar] [CrossRef]
- Nafis, A.; Kasrati, A.; Jamali, C.A.; Mezrioui, N.; Setzer, W.; Abbad, A.; Hassani, L. Antioxidant activity and evidence for synergism of Cannabis sativa (L.) essential oil with antimicrobial standards. Ind. Crops Prod. 2019, 137, 396–400. [Google Scholar] [CrossRef]
- Gursoy, N.; Tepe, B.; Akpulat, H.A. Chemical composition and antioxidant activity of the essential oils of Salvia palaestina (Bentham) and S. ceratophylla (L.). Rec. Nat. Prod. 2012, 6, 278–287. [Google Scholar]
- Abd-ElGawad, A.M.; Elshamy, A.I.; Al-Rowaily, S.L.; El-Amier, Y.A. Habitat affects the chemical profile, allelopathy, and antioxidant properties of essential oils and phenolic enriched extracts of the invasive plant Heliotropium curassavicum. Plants 2019, 8, 482. [Google Scholar] [CrossRef] [Green Version]
- Boligon, A.A.; Schwanz, T.G.; Piana, M.; Bandeira, R.V.; Frohlich, J.K.; Brum, T.F.d.; Zadra, M.; Athayde, M.L. Chemical composition and antioxidant activity of the essential oil of Tabernaemontana catharinensis A. DC. leaves. Nat. Prod. Res. 2013, 27, 68–71. [Google Scholar] [CrossRef]
- González, A.M.; Tracanna, M.I.; Amani, S.M.; Schuff, C.; Poch, M.J.; Bach, H.; Catalán, C.A. Chemical composition, antimicrobial and antioxidant properties of the volatile oil and methanol extract of Xenophyllum poposum. Nat. Prod. Commun. 2012, 7, 1663–1666. [Google Scholar] [CrossRef] [Green Version]
- Amorati, R.; Foti, M.C.; Valgimigli, L. Antioxidant activity of essential oils. J. Agric. Food Chem. 2013, 61, 10835–10847. [Google Scholar] [CrossRef]
- Rao, U. Pharmacological potential of Ipomea pes-caprae (L.) R. Br. whole plant extracts. Pelagia Res. Lib. 2015, 6, 52–60. [Google Scholar]
- Pongprayoon, U.; Baeckström, P.; Jacobsson, U.; Lindström, M.; Bohlin, L. Compounds inhibiting prostaglandin synthesis isolated from Ipomoea pes-caprae. Plant. Med. 1991, 57, 515–518. [Google Scholar] [CrossRef] [PubMed]
- Ghaneian, M.T.; Ehrampoush, M.H.; Jebali, A.; Hekmatimoghaddam, S.; Mahmoudi, M. Antimicrobial activity, toxicity and stability of phytol as a novel surface disinfectant. Environ. Health Eng. Manag. J. 2015, 2, 13–16. [Google Scholar]
- Yuan, B.; Xue, L.-W.; Zhang, Q.-Y.; Kong, W.-W.; Peng, J.; Kou, M.; Jiang, J.-H. Essential oil from sweet potato vines, a potential new natural preservative, and an antioxidant on sweet potato tubers: Assessment of the activity and the constitution. J. Agric. Food Chem. 2016, 64, 7481–7491. [Google Scholar] [CrossRef]
- Stefanello, M.É.A.; Cervi, A.C.; Ito, I.Y.; Salvador, M.J.; Wisniewski Jr, A.; Simionatto, E.L. Chemical composition and antimicrobial activity of essential oils of Eugenia chlorophylla (Myrtaceae). J. Essent. Oil Res. 2008, 20, 75–78. [Google Scholar] [CrossRef]
- Ho, C.-L.; Liao, P.-C.; Wang, E.I.-C.; Su, Y.-C. Composition and antimicrobial activity of the leaf and twig oils of Litsea acutivena from Taiwan. Nat. Prod. Commun. 2011, 6, 1755–1758. [Google Scholar] [CrossRef] [Green Version]
- Su, Y.-C.; Hsu, K.-P.; Wang, E.I.-C.; Ho, C.-L. Composition, in vitro cytotoxic, and antimicrobial activities of the flower essential oil of Diospyros discolor from Taiwan. Nat. Prod. Commun. 2015, 10, 1311–1314. [Google Scholar] [CrossRef] [Green Version]
- Cazella, L.N.; Glamoclija, J.; Soković, M.; Gonçalves, J.E.; Linde, G.A.; Colauto, N.B.; Gazim, Z.C. Antimicrobial activity of essential oil of Baccharis dracunculifolia DC (Asteraceae) aerial parts at flowering period. Front. Plant Sci. 2019, 10, 27. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ghavam, M.; Manca, M.L.; Manconi, M.; Bacchetta, G. Chemical composition and antimicrobial activity of essential oils obtained from leaves and flowers of Salvia hydrangea DC. ex Benth. Sci. Rep. 2020, 10, 15647. [Google Scholar] [CrossRef]
- Azaz, D.; Demirci, F.; Satıl, F.; Kürkçüoğlu, M.; Hüsnü, K.; Bașerb, C. Antimicrobial activity of some Satureja essential oils. Z. Naturforsch. C 2002, 57, 817–821. [Google Scholar] [CrossRef] [PubMed]
- Schmidt, E.; Bail, S.; Friedl, S.M.; Jirovetz, L.; Buchbauer, G.; Wanner, J.; Denkova, Z.; Slavchev, A.; Stoyanova, A.; Geissler, M. Antimicrobial activities of single aroma compounds. Nat. Prod. Commun. 2010, 5, 1365–1368. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Claeson, P.; Rådström, P.; Sköld, O.; Nilsson, Å.; Höglund, S. Bactericidal effect of the sesquiterpene T-cadinol on Staphylococcus aureus. Phytother. Res. 1992, 6, 94–98. [Google Scholar] [CrossRef]
- Dahham, S.S.; Tabana, Y.M.; Iqbal, M.A.; Ahamed, M.B.; Ezzat, M.O.; Majid, A.S.; Majid, A.M. The anticancer, antioxidant and antimicrobial properties of the sesquiterpene β-caryophyllene from the essential oil of Aquilaria crassna. Molecules 2015, 20, 11808–11829. [Google Scholar] [CrossRef] [PubMed]
- Chinou, I.B.; Bougatsos, C.; Perdetzoglou, D. Chemical composition and antimicrobial activities of Helichrysum amorginum cultivated in Greece. J. Essent. Oil Res. 2004, 16, 243–245. [Google Scholar] [CrossRef]
- Bajpai, V.K.; Sharma, A.; Baek, K.-H. Antibacterial mode of action of Cudrania tricuspidata fruit essential oil, affecting membrane permeability and surface characteristics of food-borne pathogens. Food Control 2013, 32, 582–590. [Google Scholar] [CrossRef]


| No | Compound name | KIp 1 | KIc 2 | Conc. (%) | Identification 3 |
|---|---|---|---|---|---|
| Oxygenated Monoterpenes | |||||
| 1 | Linalool | 1095 | 1095 | 0.49 ± 0.01 | MS, KI |
| 2 | α-Fenchyl alcohol | 1114 | 1115 | 0.57 ± 0.02 | MS, KI |
| 3 | 4-Terpineol | 1177 | 1175 | 0.69 ± 0.01 | MS, KI |
| Sesquiterpene Hydrocarbons | |||||
| 4 | β-Patchoulene | 1381 | 1379 | 0.77 ± 0.03 | MS, KI |
| 5 | α-Gurjunene | 1409 | 1411 | 0.53 ± 0.01 | MS, KI |
| 6 | E-Caryophyllene | 1417 | 1417 | 0.67 ± 0.02 | MS, KI |
| 7 | trans-α-Bergamotene | 1432 | 1435 | 0.33 ± 0.01 | MS, KI |
| 8 | Aromandendrene | 1439 | 1441 | 0.69 ± 0.02 | MS, KI |
| 9 | α-Humulene | 1452 | 1449 | 0.31 ± 0.01 | MS, KI |
| 10 | ar-Curcumene | 1479 | 1476 | 1.83 ± 0.04 | MS, KI |
| 11 | γ-Muurolene | 1480 | 1483 | 1.76 ± 0.03 | MS, KI |
| 12 | α-Muurolene | 1500 | 1502 | 0.56 ± 0.01 | MS, KI |
| 13 | δ-Cadinene | 1522 | 1520 | 0.97 ± 0.03 | MS, KI |
| 14 | α-Calacorene | 1544 | 1541 | 1.04 ± 0.03 | MS, KI |
| 15 | Guaiazulene | 1780 | 1784 | 0.37 ± 0.01 | MS, KI |
| Oxygenated Sesquiterpenes | |||||
| 17 | Citronellyl 3-methylbutanoate | 1531 | 1529 | 1.55 ± 0.04 | MS, KI |
| 18 | Spathulenol | 1578 | 1581 | 8.11 ± 0.08 | MS, KI |
| 19 | Caryophyllene oxide | 1583 | 1585 | 6.56 ± 0.06 | MS, KI |
| 20 | Geranyl isovalerate | 1607 | 1604 | 0.81 ± 0.02 | MS, KI |
| 21 | Humulene epoxide II | 1608 | 1612 | 0.97 ± 0.03 | MS, KI |
| 22 | tau-Cadinol | 1640 | 1643 | 35.68 ± 0.16 | MS, KI |
| 23 | α-Cadinol | 1654 | 1650 | 26.76 ± 0.12 | MS, KI |
| 24 | α-Santalol | 1674 | 1677 | 0.63 ± 0.02 | MS, KI |
| 25 | 6-Epishyobunone | 1680 | 1680 | 0.35 ± 0.01 | MS, KI |
| 26 | Ledene oxide-(II) | 1682 | 1683 | 0.69 ± 0.02 | MS, KI |
| 27 | Bisabolone | 1742 | 1746 | 0.77 ± 0.02 | MS, KI |
| Diterpene Hydrocarbons | |||||
| 28 | Beyerene | 1931 | 1936 | 0.36 ± 0.01 | MS, KI |
| Carotenoid-Derived Compounds | |||||
| 29 | E-β-Damascenone | 1383 | 1382 | 0.77 ± 0.02 | MS, KI |
| 30 | E-α-Ionone | 1428 | 1430 | 0.18 ± 0.01 | MS, KI |
| Apocarotenoid-Derived Compounds | |||||
| 31 | Hexahydrofarnesyl acetone | 1845 | 1849 | 1.81 ± 0.03 | MS, KI |
| Oxygenated monoterpenes | 1.75 | ||||
| Sesquiterpene hydrocarbons | 9.83 | ||||
| Oxygenated sesquiterpenes | 82.88 | ||||
| Diterpene hydrocarbons | 0.36 | ||||
| Carotenoid-derived compounds | 0.95 | ||||
| Apocarotenoid-derived compounds | 1.81 | ||||
| Treatment | Conc. (mg L−1) | Scavenging Activity (%) | |||
|---|---|---|---|---|---|
| DPPH | IC50 (mg L−1) | ABTS | IC50 (mg L−1) | ||
| Ipomoea carnea EO | 5 | 11.37 ± 0.32 I * | 33.69 | 6.52 ± 0.19 J | 40.86 |
| 10 | 17.21 ± 0.49 H | 12.66 ± 0.46 I | |||
| 20 | 31.07 ± 0.89 F | 24.15 ± 0.69 G | |||
| 30 | 52.54 ± 1.64 D | 38.41 ± 1.10 E | |||
| 40 | 63.29 ± 1.81 B | 50.6 ± 1.88 D | |||
| 50 | 74.31 ± 2.12 A | 59.69 ± 1.97 C | |||
| LSD0.05 | 3.21 *** | ||||
| F-value | 436.40 | ||||
| Ascorbic acid | 1 | 5.25 ± 0.12 K | 11.51 | 2.14 ± 0.08 L | 12.94 |
| 2.5 | 14.47 ± 0.42 I | 10.36 ± 0.35 J | |||
| 5 | 41.19 ± 1.28 G | 37.08 ± 1.26 H | |||
| 10 | 53.73 ± 1.67 E | 45.62 ± 1.58 F | |||
| 15 | 60.02 ± 1.79 C | 55.91 ± 2.11 D | |||
| 20 | 73.17 ± 2.17 A | 69.06 ± 2.63 B | |||
| LSD0.05 | 2.03 *** | ||||
| F-value | 1307.67 | ||||
| Bacterial Strain | EO (10 mg mL−1) | Reference Antibiotics IZ mm | ||||
|---|---|---|---|---|---|---|
| IZ mm | MIC (mg mL−1) | Cephalexin | Tetracycline | Ofloxacin | Ampicillin | |
| Gram-negative bacteria | ||||||
| Escherichia coli | 19.62 ± 0.59 C # | 84.0 | 10.29 ± 0.31 D | 21.08 ± 0.64 A | 24.91 ± 0.75 A | 20.06 ± 0.63 D |
| Klebsiella pneumoniae | 23.56 ± 0.71 A | 94.0 | 10.05 ± 0.30 D | 20.62 ± 0.62 A | 20.11 ± 0.61 C | 6.71 ± 0.21 E |
| Pseudomonas aeruginosa | 16.41 ± 0.50 D | 124 | 0.00 E | 0.00 E | 10.36 ± 0.31 E | 0.00 F |
| Salmonella typhimurium | 12.39 ± 0.38 E | 158 | 0.00 E | 9.88 ± 0.30 D | 0.00 F | 0.00 F |
| Gram-positive bacteria | ||||||
| Bacillus cereus | 21.36 ± 0.65 B | 82 | 19.13 ± 0.58 B | 10.34 ± 0.31 D | 19.66 ± 0.60 CD | 7.53 ± 0.21 E |
| Staphylococcus epidermidis | 19.05 ± 0.58 C | 84 | 14.07 ± 0.43 C | 17.65 ± 0.53 C | 23.05 ± 0.70 B | 27.96 ± 0.74 A |
| Staphylococcus haemolyticus | 8.47 ± 0.26 F | 1442 | 23.74 ± 0.72 A | 19.51 ± 0.59 B | 23.48 ± 0.61 B | 21.07 ± 0.61 C |
| Staphylococcus xylosus | 22.69 ± 0.69 A | 82 | 18.39 ± 0.56 B | 17.04 ± 0.52 C | 19.03 ± 0.58 D | 23.64 ± 0.81 B |
| LSD0.05 | 0.94 *** | 0.89 *** | 0.92 *** | 0.90 *** | 0.82 *** | |
| F-value | 279.11 | 867.02 | 565.54 | 765.49 | 1408.00 | |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Abd-ElGawad, A.M.; Elshamy, A.I.; Elgorban, A.M.; Hassan, E.M.; Zaghloul, N.S.; Alamery, S.F.; El Gendy, A.E.-N.G.; Elhindi, K.M.; EI-Amier, Y.A. Essential Oil of Ipomoea carnea: Chemical Profile, Chemometric Analysis, Free Radical Scavenging, and Antibacterial Activities. Sustainability 2022, 14, 9504. https://doi.org/10.3390/su14159504
Abd-ElGawad AM, Elshamy AI, Elgorban AM, Hassan EM, Zaghloul NS, Alamery SF, El Gendy AE-NG, Elhindi KM, EI-Amier YA. Essential Oil of Ipomoea carnea: Chemical Profile, Chemometric Analysis, Free Radical Scavenging, and Antibacterial Activities. Sustainability. 2022; 14(15):9504. https://doi.org/10.3390/su14159504
Chicago/Turabian StyleAbd-ElGawad, Ahmed M., Abdelsamed I. Elshamy, Abdallah M. Elgorban, Emad M. Hassan, Nouf S. Zaghloul, Salman F. Alamery, Abd El-Nasser G. El Gendy, Khalid M. Elhindi, and Yasser A. EI-Amier. 2022. "Essential Oil of Ipomoea carnea: Chemical Profile, Chemometric Analysis, Free Radical Scavenging, and Antibacterial Activities" Sustainability 14, no. 15: 9504. https://doi.org/10.3390/su14159504
APA StyleAbd-ElGawad, A. M., Elshamy, A. I., Elgorban, A. M., Hassan, E. M., Zaghloul, N. S., Alamery, S. F., El Gendy, A. E.-N. G., Elhindi, K. M., & EI-Amier, Y. A. (2022). Essential Oil of Ipomoea carnea: Chemical Profile, Chemometric Analysis, Free Radical Scavenging, and Antibacterial Activities. Sustainability, 14(15), 9504. https://doi.org/10.3390/su14159504









