1. Introduction
Arsenic (As) is considered as the most toxic environmental pollutant that causes severe damages to crops when it enters into the soil environment through sewage and industrial waste irrigations (Otero et al., 2016 [
1], Kayode et al., 2021 [
2]). Petrochemical industries and oil refineries are the major sources that contaminate soils with As (González et al., 2021 [
3]). Coal combustion, metallurgical, and artistic glass industries are the other sources of massive As emission into the natural environment (Zevenhoven et al., 2007 [
4]; Formenton et al., 2021 [
5]).
Arsenic occurs in As (III) and As (V) as the most toxic forms in the natural environment. Both these forms are dominant and inter convertible, depending upon chemical and biological processes. As (III) is a soluble form of arsenic (Itaya et al., 2022 [
6]) and dominates in anaerobic environments, while As (V) is mostly found in a solid state under aerobic conditions (Smedley et al., 2002 [
7]; Sharma and Sohn 2009 [
8]; Cai et al., 2020 [
9]). As (V) holds a high affinity with P-transporters, enters into the plant system through a structural analogy with phosphate and competes with Phosphorus (P) by diminishing ATP production that results in stunted growth (Sharma et al., 2020 [
10]). The soluble form of As (III) enables it to associate with fertile soils and increases its bioavailability for plants via contaminated soils. As (III) is a hundred times more toxic than As (V) and binds with protein sulphydryl groups and degraded membranes and even causes cell death (Bhattacharya et al., 2021 [
11]). The extensive As transport through these channels competes with nutrient ions and disturbs essential nutrients’ ion uptake that causes nutrient deficiency in plants (Das et al., 2020 [
12]).
Arsenic toxicity inhibits the physio-biochemical, cellular and molecular functions of plants (Ulhassan et al., 2022 [
13]) and is a death dealing agent even at a very low quantity of about 7 mg L
−1 (De Oliveira et al., 2018 [
14]). Extensive studies prove that As-contamination negatively affects the seed germination and seedling growth (Mridha et al., 2021 [
15]; Nouri et al., 2021 [
16]; Kumar et al., 2022 [
17]). Excessive levels of As disturb the plant water relation and gas exchange attributes due to oxidative damages in plants (Arikan et al., 2022 [
18], Shah et al., 2022 [
19]). It alters the root metabolism and triggers the overproduction of reactive oxygen species (ROS) (Choudhary et al., 2020 [
20]). These over-accumulated ROS are the prominent cause of negative impacts on plant systems (Berni et al., 2019 [
21]). The instigated ROS levels impose oxidative stress that damages the cell structure, i.e., nucleic acids, proteins and lipids (Shahid et al., 2021 [
22]). The antioxidant system is also dependent on ROS concentration and works with coordinate manners (Suhel et al., 2022 [
23]; Fatima et al., 2021 [
24]). Therefore, high levels of As results in increasing levels of ROS that consequently damage the entire system of plants. Additionally, over expression of ROS also increases the levels of antioxidant enzymes (CAT, POS, SOD, APX) and membrane lipid peroxidation MDA and H
2O
2 in plants (Suhel et al., 2022 [
23]) that damage the cell structures.
Meanwhile, the accumulation of secondary metabolites (flavonoids, phenolics, ascorbic acid) is the defensive strategy to lower down oxidative damages (Bhat et al., 2022 [
25], Shamshir et al., 2022 [
26]). The amino acids tend to increase under As induced stress [
27,
28]. The ascorbic acid and phenolics are also secondary metabolites that have an important role in ROS scavenging under oxidative stress (Mishra et al., 2012 [
29]). In addition, secondary metabolite production is considered as a stress coping strategy during the growing stages of plants (Isah 2019 [
30]), while other compounds such as carotenoids and chlorophyll pigments decrease under higher As regimes while increasing under low As concentrations. The decreased levels of these pigments as a result of As toxicity decrease not only the growth of plants but also affect the crop productivity.
The maize (
Zea mays L.) crop is the third most grown crop all over the world for different purposes and fulfills the needs of both animals and human (Bairwa et al. [
31]). It is cultivated as feed and fodder crop all over the world, and the demand for maize crop is increasing rapidly day by day. However, maize production is facing serious challenges of changing environments and heavy metal contamination of soil (Aftab et al., 2020 [
32]; Irfan et al., 2021 [
33]). Arsenic is the top prior hazardous metalloid that decreases the growth and productivity of maize at alarming rates (Kaya et al., 2020 [
34]; Khan et al., 2022 [
35]). Pakistan is at the fourth position in maize cultivation that is facing the definite reduction in yield due to such heavy metals and metalloids’ contamination. Keeping in view the ecofriendly and economically suitable methods, there is a firm need to address As toxicity related hazards.
To control As stress has become challenge for a better and sustainable agri-ecosystem that associates with crop productivity and growth (Khalid et al., 2020 [
36], Shahid et al., 2021 [
22]). However, to overcome the challenges of reduced crop yield, several techniques have been adapted to cope with heavy metal stressed conditions (Pavlikova et al., 2008 [
37]). The most recent strategy of seed priming was recognized as effective against biotic and abiotic stresses. Seed priming is a cheap and easily approachable technology adapted by the previous researchers under stressed environmental cues (Jisha et al., 2013 [
38]). In the context of previous studies, seed primed crops with various substances proved to be effective against abiotic stress (Hussain et al., 2019 [
39]).
The thiamine and IAA seed priming approaches may be a better and easy way to mitigate the detrimental effects of crops growing in As hosted soils’ environment. Previous studies reported that the continuous synthesis of thiamine or its enhanced endogenous levels was supposed to improve the plants’ tolerance against harsh stressed conditions (Abidin et al. 2016 [
40]). Thiamine is associated with abiotic stress tolerance (Jabeen et al., 2021 [
41]; Kaya et al., 2015 [
42]; Ghaffar et al., 2019 [
43]) and helpful in the synthesis of nucleic acids, carbohydrates, adenosine triphosphate and nicotinamide adenine dinucleotide phosphate (Nosaka 2006) [
44]. Moreover, the alternative role of thiamine was recognized as a plant defense activator (Ahn et al., 2005 [
45]). Under stressed conditions, thiamine proved to increase germination percentage in maize (Kaya et al., 2015 [
42]). Being a vitamin of prime importance, it is introduced in medicinal plants for better nutrition and the treatment of thiamine deficiency related diseases (Sunarić et al., 2020 [
46]).
On other hand, the auxins are considered as the organic phyto-hormones that coordinate with developmental processes in plants (Strydhorst et al., 2018 [
47]). Indole-3-acetic acid (IAA) is a plant hormone of the auxin class that increases the photosynthesis in plants and accelerates the source-sink relation (Khan and Mazid 2018 [
48]). IAA induces positive responses against heavy metals (Cd, Pb) and reduces lipid peroxidation MDA with an increase in chlorophyll pigments (Peat et al., 2012 [
49]; Ji et al., 2015 [
50]) that resultantly increases maize plants’ biomass (Hadi et al., 2010 [
51]). Being the main individual of the auxin class and plant growth regulator, IAA has functions in a harmonized manner with other growth regulators (Amoanimaa-Dede et al., 2022 [
52]). In addition, it improves the major developmental processes in plants and is involved in the induction of tolerance against various abiotic stresses (Chen et al., 2020 [
53]). Moreover, it heals the oxidative injuries against As-stress and imparts better growth (Alamri et al., 2021 [
54]).
Therefore, thiamine and IAA as priming agents on maize seeds under As-stress as a combined application were tested. To unlock the hidden role of thiamine and IAA as priming agents under As-stress, the experiment was designed to study two genetically different maize varieties to elucidate the biochemical mechanisms involved to ameliorate the negative role of As in the specific context of growth, oxidative stress, gas exchange, lipid peroxidation and ionic assimilation improvements.
6. Discussion
The As-stress constrained the maize plants to restrict growth (root and shoot length) and biomasses (root and shoot fresh and dry weights) to a significant extent by altering the biochemical mechanisms (Kaya et al., 2020 [
34]; Khan et al., 2022 [
35]). The stunted growth observed in both maize varieties was the result of chlorophyll set up disturbance, anatomical and biochemical changes. The agronomic characteristics (growth and biomass) of maize plants against As-stress represented different trends according to the variety of the different genetic pedigree (Atif and Perveen 2021 [
55]). As a result of disrupted plant physiology under As-stress, the physio-biochemical nature of the plant was disturbed, ultimately leading plants to a remarkable decrease in growth that was already studied in previous research (Khan et al., 2022 [
35], Bidi et al., 2021 [
75]). The depiction of As tolerance against As-stress was already studied in our previous study (Atif and Perveen 2021 [
55]) which correlated with chemical changes and the resistive nature of superior varieties as compared to sensitive ones. This is because of the presence of specific growth and development associated genes that expressed differentially under stress (Khare et al., 2022 [
76]). The seed priming with IAA and thiamine increases the growth (shoot and root length) and biomasses (dry and fresh weight) of maize plants. Thiamine promotes the growth of plants under stress by improving photosynthetic contents, antioxidants and secondary metabolites (Jabeen et al., 2021 [
41]. Correspondingly, endogenous enhanced levels of IAA stimulate the plant growth.
The decrease in chlorophyll pigments (Chl.
a,
b, Chl ratio
a/b, total Chl. and carotenoids) under As-stress toxicity was prominent and was explored as the key indicator of heavy metal stress (
Table 2 and
Table 3). This is because As toxicity includes the photosynthetic degrading hormones instead of synthesizing hormones and compels plants to an oxidative burst (Zemanová et al., 2020 [
77]; Ghorbani et al., 2020 [
78]). The similar depression in chlorophyll attributes was concluded in a previous study by Tanveer et al. (2022) [
79]. The seeds treated with thiamine as priming agents with thiamine and IAA were better in photosynthetic expression (
Table 2 and
Table 3) because thiamine as the priming agent can trigger the plant-defense system (Suohui et al., 2022 [
80]). Hence, being as an essential nutrient, plant roots needed to synthesize it in sufficient amounts under harsh situations; however, exogenously uptake as seed priming could play a vital role in root growth and overall plant development (Goyer 2010 [
81]). Thiamine is also known for combating or scavenging the ROS species, and hence, as a result of over ROS, it maintained the chlorophyll contents (Kaya et al., 2015 [
42]). Accordingly, IAA also controlled the diminishing of chlorophyll pigments under As-stress (
Table 2 and
Table 3). This might be due to IAA as the main auxin that controlled the growth and development by stimulating the growth and having role in the process of photosynthesis and pigment formation. The photosynthetic activity increased with the priming of IAA, confirming the findings by Zhao et al. (2021) [
82]. The novel insight of the current study was the combined priming application of both growth regulators which proved to be the most effective in the recovery of growth and photosynthesis characters.
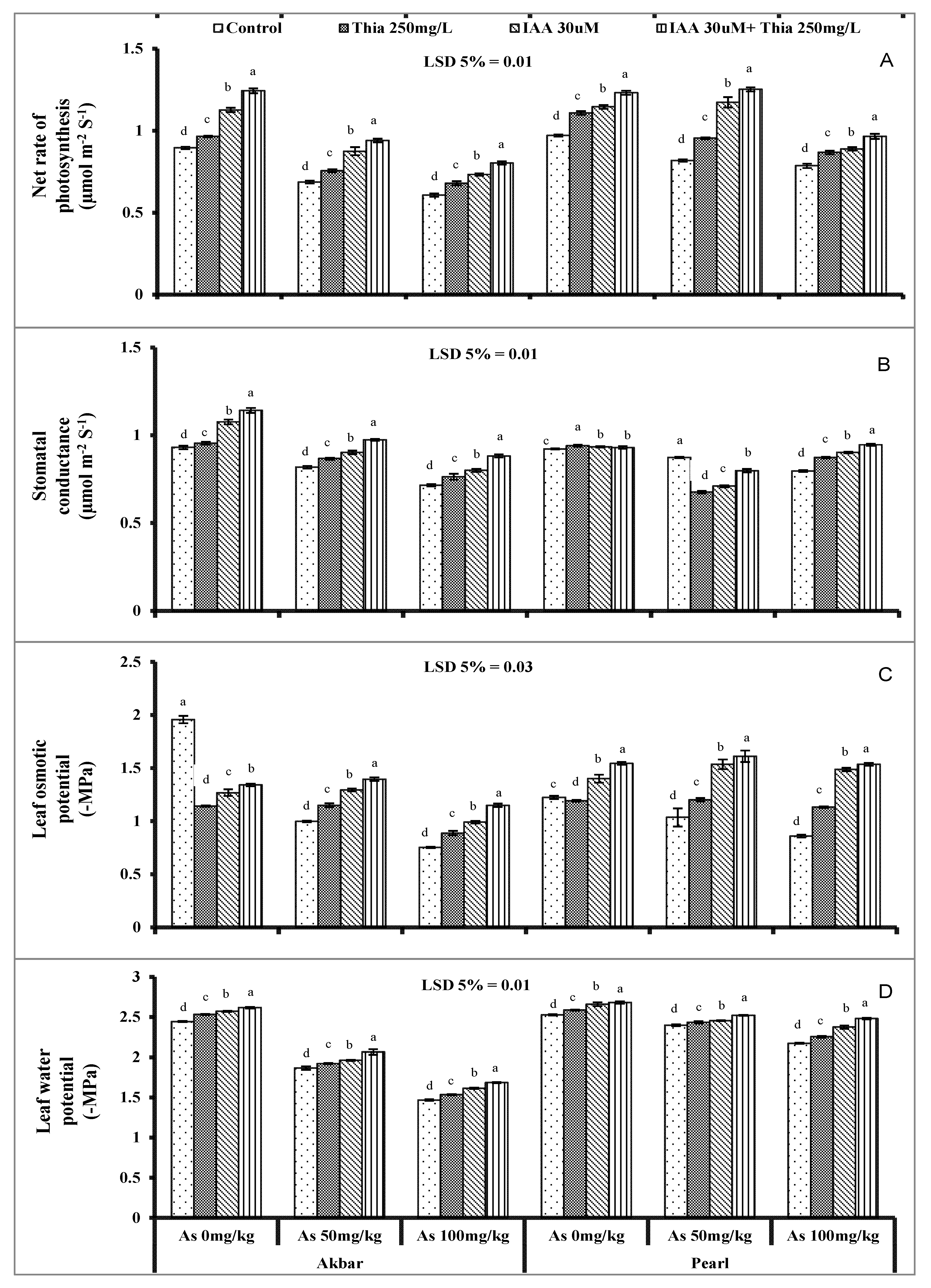
The leaf gas exchange characters—rate of photosynthesis (
A), transpiration rate (
E), stomatal conductance (
gs)—of maize plants when exposed to As-stress showed a substantial decrease (Arikan et al., 2022 [
18]). Different heavy metals including As decreased the gas exchange attributes (Anjum et al., 2017 [
83]) that might be related to chlorophyll degradation and the disturbance of enzymes that take part in CO
2 fixation in the case of the rate of photosynthesis (Li et al., 2007 [
84]). The other reason behind the decreased rate of
E and
A is due to low biomass production that increases the stomatal resistance (Šimonová et al., 2007 [
85]). The improved gas exchange machinery was observed in plants that were primed with thiamine and IAA. Thiamine is the essential nutrient for plants and is recognized as a strong antioxidant agent against the plant stressed environment which in return improves the gas exchange characters.
As-stress also disturbs the plant-water relation and limits the growth (Vezza et al., 2018 [
86]). Disturbance of plant water relation factors—leaf water potential (Ψ
w) and leaf osmotic potential (Ψ
s)—might be due to the altered structure of xylem both in roots and shoots. This might happen due to disturbed intercellular spaces, the water carriage capacity from root to shoot and the number of stomata under heavy metal stress (Rucińska-Sobkowiak 2016 [
87]). The plant water relation attributes’ disturbance may be due to the result of the unbalanced water uptake through altered stomatal behavior (Singh et al., 2019 [
88]). Imposed As-stress also has an obstacle for plants to regulate and retain the water in plant tissues (Anjum et al., 2017 [
83]). Metal toxicity also creates an imbalance in the nutrient uptake that is associated with disturbed plant water relation (Muradoglu et al., 2017 [
89]). The priming application with IAA and thiamine imparts better plant water relation in maize plants of both varieties. An explanation of greater leaf water and osmotic potential (Ψ
w and Ψ
s) by thiamine treatment might be due to the application of thiamine that lowered the osmotic stress by accumulating proline, and similar ameliorating effects of thiamine treatment on leaf water and proline contents have already been reported in maize (Sanjari et al., 2019 [
90]) under Cd stress.
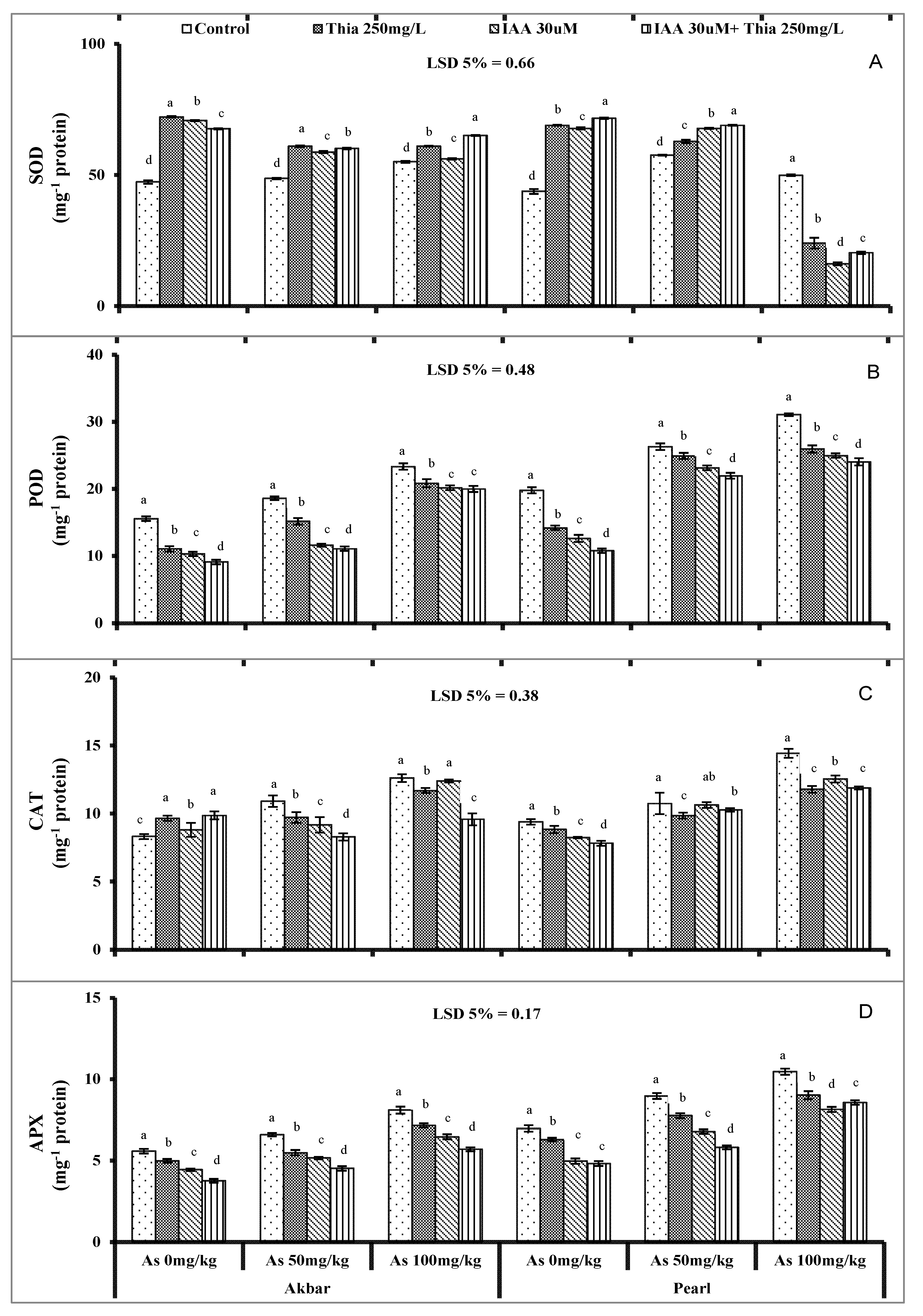
As-stressed maize plants exposed enhancement in enzymes as the result of over expressed ROS and imposed oxidative stress (Bhat et al., 2022 [
25]; Mridha et al., 2022 [
91]). Increases in APX SOD, POD in both maize varieties were in agreement with Suhel et al. (2022) [
23], Atif and Perveen (2021) [
55]. The SOD scavenges the ROS with direct participation against oxidative stress. Over-expression of SOD in plants induced tolerance against stresses. SOD converted the toxic O
2− radicals to molecular oxygen and H
2O
2 which later on was detoxified by GPX or CAT (Jung et al., 2019 [
92]). Under stressed conditions, SOD regulated the intercellular ROS and physiological conditions (Faize et al., 2011 [
93]). It showed amazingly unresponsive behavior in different plant species against stresses (Kliebenstein et al., 1998 [
94]). In a similar way, POD breaks H
2O
2 at the cellular level with its different forms (Ros-Barcelo et al., 2002) [
95]. Seeds primed with thiamine showed improved oxidative enzymes because thiamine limits the oxidative stress (Zhou et al. 2013 [
96]). Thiamine acts as antioxidant after providing the NADH and NADPH (Asensi-Fabado and Munne-Bosch 2010 [
97]). Correspondingly, IAA treated seed also showed minimal damages against antioxidant enzymes against As-stress Because it reduced heavy metals’ toxicity in roots which was ultimate factor for the lowering of damages (Bashri and Parasad 2015 [
98]). Combined treated seeds with thiamine and IAA still provided the novel result of improving the antioxidant activities under As-stress.
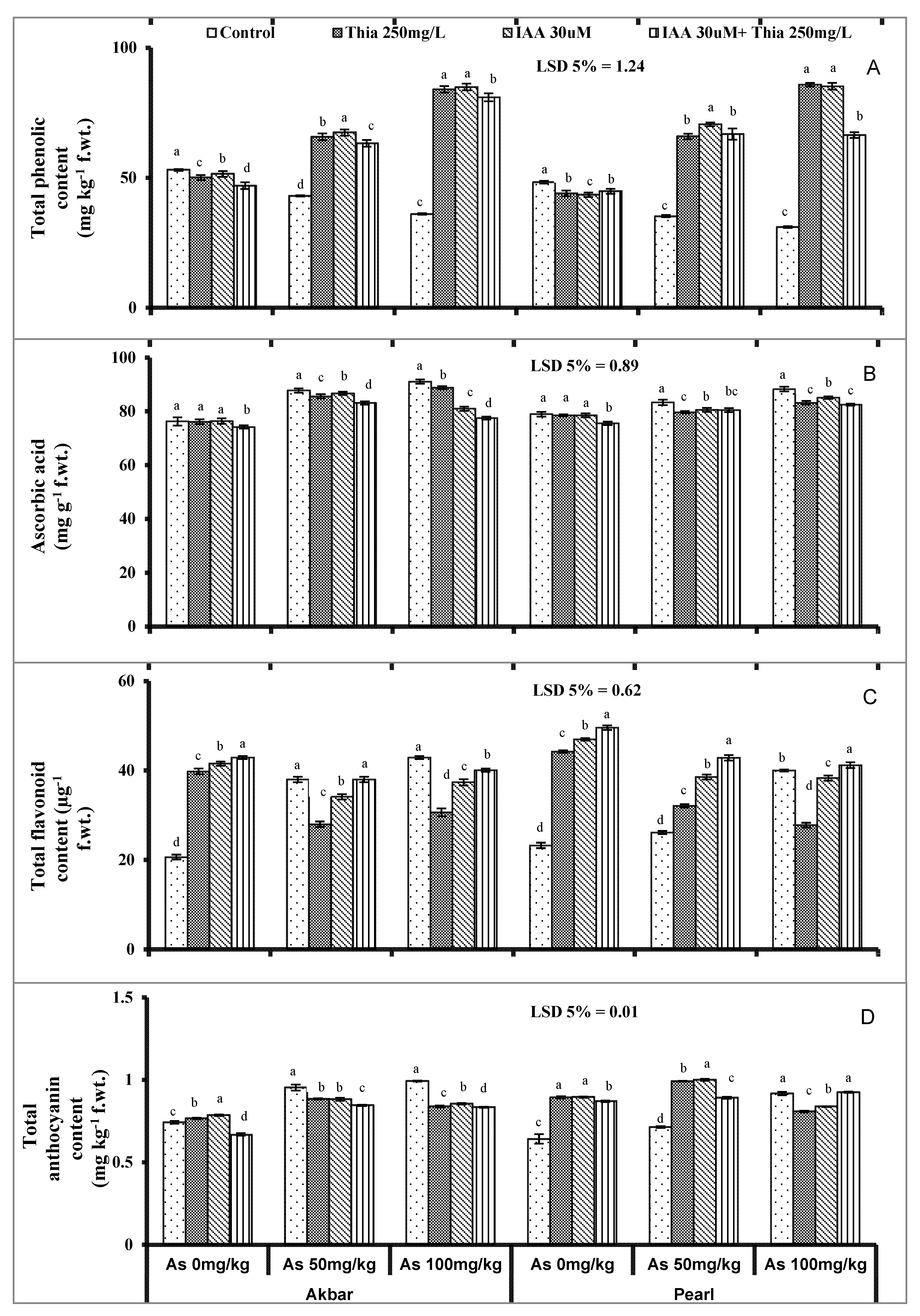
The phenolic compounds including the flavonoids, AsA and anthocyanins are the secondary metabolites that play a key role in plant defense, and their activation depends on the intensity of stress. The current study depicted the decreased phenolic contents in both maize varieties under As-stress (
Figure 3A) that was in agreement with Shamshir et al. (2022) [
26] under As-stress. The phenolic compounds were also improved with the application of thiamine (Boubakri et al. 2013 [
99]). AsA is necessary for ROS scavenging process and showed an increase against As-stress (
Figure 3B). AsA has already been proved as the oxidative stress alleviator in the form of a priming agent (Elkelish et al., 2020 [
100]). AsA accumulation enabled the plants to cope with oxidative stress with increased contents under stress in the current study (
Figure 3B). Seeds primed with IAA improved AsA because IAA has the greater potential for improving the overall antioxidant capacity in oxidative stressed conditions (Madany et al., 2020 [
101]). Anthocyanins are low molecular weight potent compounds that secure plants from heavy-metal-induced oxidative stress to a greater extent (Stambulska et al., 2018 [
102]). The metal-induced increment in anthocyanin was already reported in Arabidopsis (Baek et al., 2012 [
103]).
H
2O
2 and MDA are both considered as the stress indicators in oxidative stress and in terms of lipid peroxidation that reflects the plant strength to oxidative stress. When plants are facing As-toxicity, the oxidative potential was configured with increasing contents of both H
2O
2 and MDA (Bhat et al., 2022 [
25]). The main reason was that the electron transfer during photosynthesis was inhibited, and both these biomolecules were found to be higher than the control, presenting oxidative stress under As-stress (
Table 4) as confirmed by Kaya et al. (2022) [
104] and Mishra et al. (2022) [
29]. The cell membrane functions were disrupted with the over-accumulation of both these biomolecules (Zahra et al., 2018 [
105]). The seed treated plants with thiamine and IAA showed decreases in both biomolecules (
Table 4). Thiamine treated plants initiated the defensive machinery of plant and limited the concentrations of both by mitigating negative effects of As with reduced ROS which was also observed by Kaya et al. (2015) [
42]. Sanjari et al. (2019) [
90] found the role of thiamine to lower the H
2O
2 and MDA contents against oxidative damage by protecting the membranes. Thiamine application is useful in alleviating the toxic effects of stress and reduces H
2O
2 and MDA contents in different plants (Kaya et al. 2015 [
42]). IAA reduces the heavy metal toxicity by improving membrane properties and decreasing the disorder of membrane organization (Bücker-Neto et al., 2017 [
106]). However, the combined treatment of both growth regulators showed the better improvements (
Table 4), while no study yet describes the combined exogenous application role on the biomolecules.
The current study indicated increased contents in osmolytes such as proline under As-stress (Dolui et al., 2022 [
107]). The proline accumulation is being considered as an oxidative stress indicator along with sugars (Bidi et al., 2021 [
75]). The TSS showed a definite increase under As-stress, while NRR and RS exhibited a decrease in their levels compared to the control (
Table 5). The FAA and proline also were considered as the low molecular weight chemical compounds that accumulated in plants under stressed circumstances and coordinated with different plant regulators as a systemic strategy for metal detoxification in plants. Current results also demonstrated a marked increment in the deposition of proline and TFAA in maize plants growing in As-contaminated soil (
Table 4 and
Table 5). These results confirmed the scavenging ROS and antioxidative role of these molecules under oxidative stress and ROS accumulation. The influence of exogenously applied IAA and thiamine displayed a significant increase in endogenous levels of TFAA as alone and combined. The inflated accumulation of proline is correlated with better metal detoxification in plants (Hussain et al., 2019 [
39]). IAA directly increased the protein and proline contents and counteracted with stress (Khalid and Aftab 2020 [
108]). In contrast, RS and NRS decreased under stress, in agreement with Samanta et al. (2020) [
109]. Current results showed that an As-induced increase in soluble sugars was substantially maintained in plants with thiamine and IAA individually and in combination. IAA decreased the protein contents under stress (Khalid and Aftab 2020 [
108]). The mitigating effect of thiamine on maize cultivars might be due to its role as a coenzyme in various metabolic pathways such as sugar and protein metabolism (Goyer 2010 [
81]). All attributes’ improvement through the exogenous application of thiamine and IAA as alone and combined was responsible to enhance the growth against As-stress in both maize varieties.
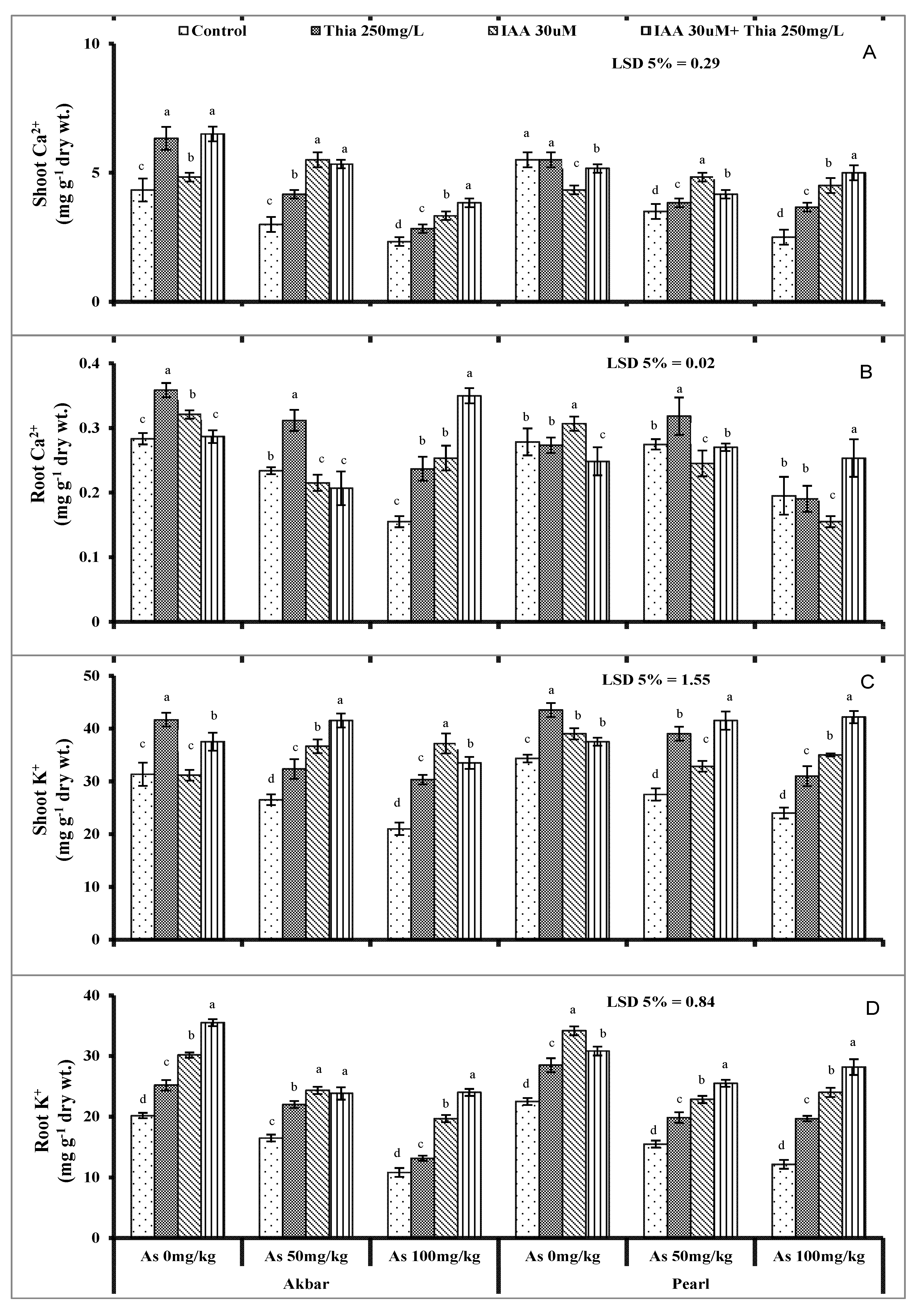
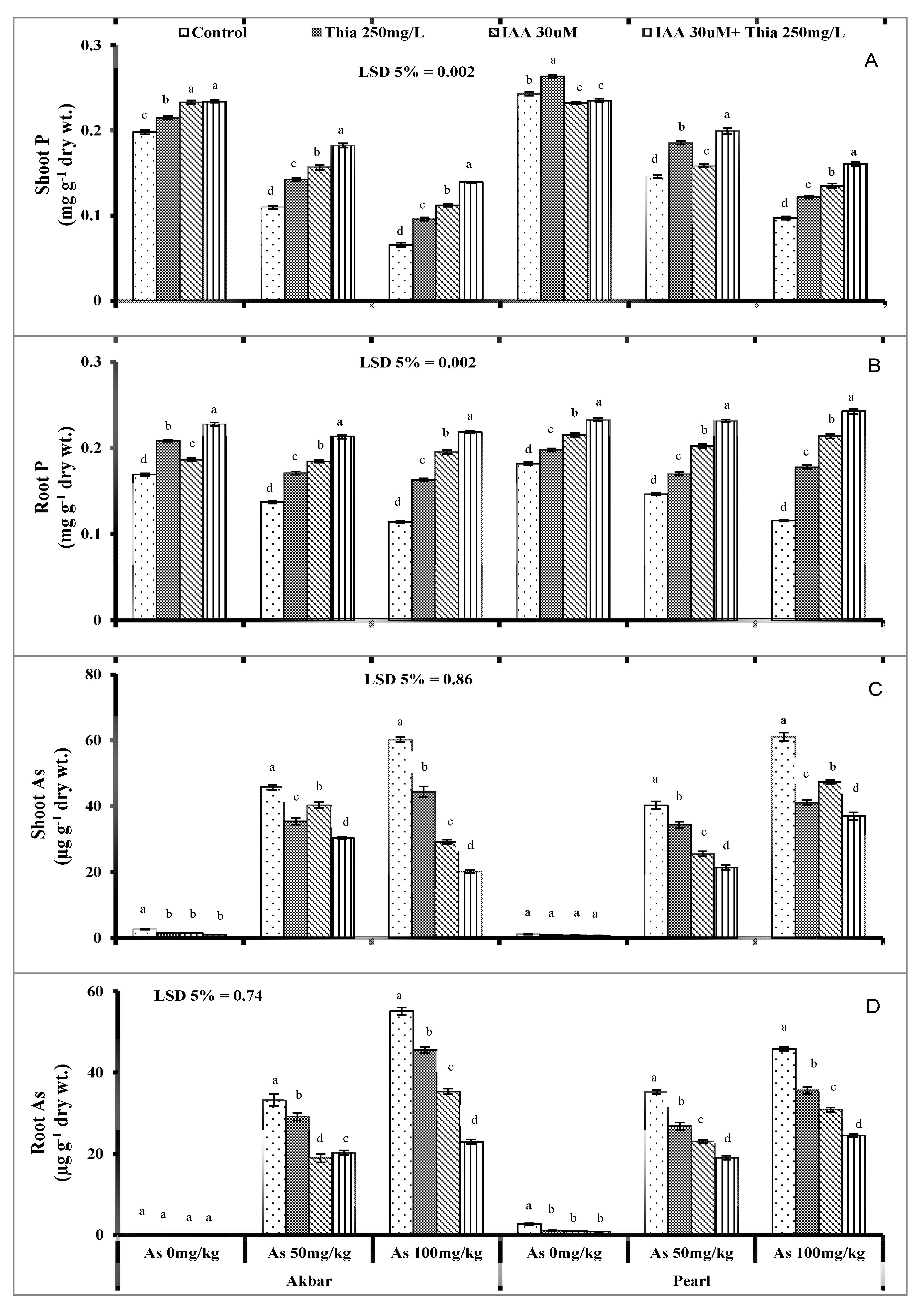
It is well documented that ionic changes reversibly trigger the antioxidant system. The initial set up that was disturbed in As-contaminated soil is a nutrient imbalance, and As travels from root to shoot and ultimately enters into edible parts of plants. The current study also concluded nutrient ions’ (K
+, Ca
2+, P) uptake disturbance when plants faced As toxicity (
Figure 4A–D and
Figure 5A–B). The translocation of different micro- and macronutrients was interrupted with As contamination which interferes negatively with the plant metabolism (Samanta et al., 2022 [
110]). As-contamination significantly altered the nutrient ion uptake (Shamshir et al., 2022 [
26]). Auxins promote stress tolerance and nutrient uptake by controlling the shoot/root growth ratio (Kurepa and Smalle 2022 [
111]). In the present study, exogenously applied thiamine and IAA improved the nutrient uptake in maize plants under As stress and improved the availability of essential nutrients that competed with As. Rapala-Kozik et al. (2012) [
112] suggested that thiamine mediates oxidative stress tolerance via salicylic acid and Ca
2+-related signaling pathways.
As-translocation increased in plants from roots to shoots and in fruits (Kaya et al., 2020 [
34]). As-contents were found more in roots than in shoots (Ruiz-Huerta et al., 2022 [
113]). In sandy loam soils, the As-uptake in maize plants was observed more than in sand because the As translocation varies from soil to soil conditions and plants’ temporary conditions (Gulz et al., 2005 [
114], Zheng et al., 2011 [
115]). As ions were more pronounced in xylem sap as compared to roots in maize plants under As-stress that confirmed As accumulation in shoots (Su et al., 2010 [
116]). The structural similarity of As with P facilitates As to accumulate in plants and travel to aerial parts of plants (
Figure 5A,B). In the current findings, As contents were more in shoots which exhibits the uptake of As ions and accumulation in maize plants which was confirmed by (Kaya et al., 2020 [
34]). Pearson correlation and principal component analysis are shown in
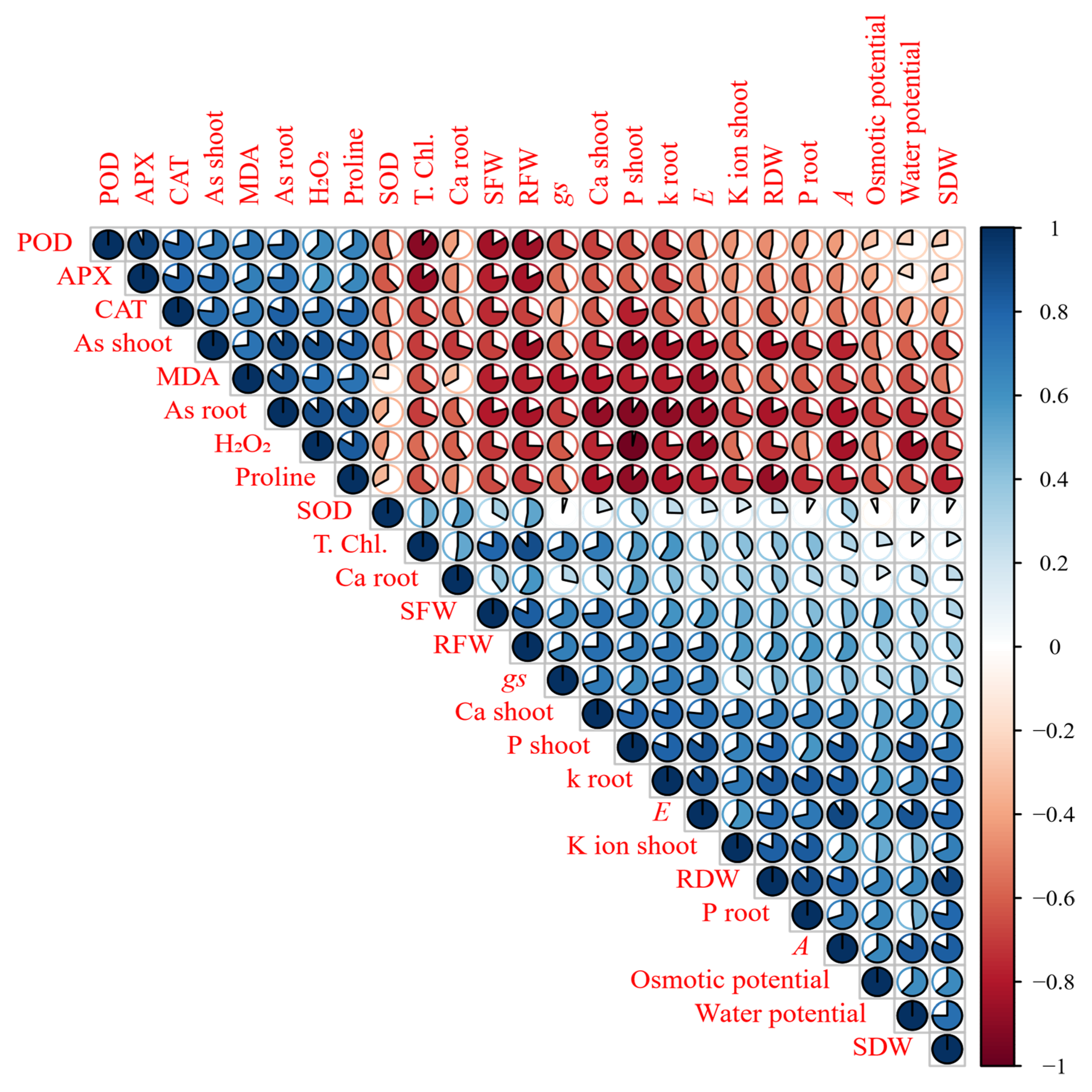
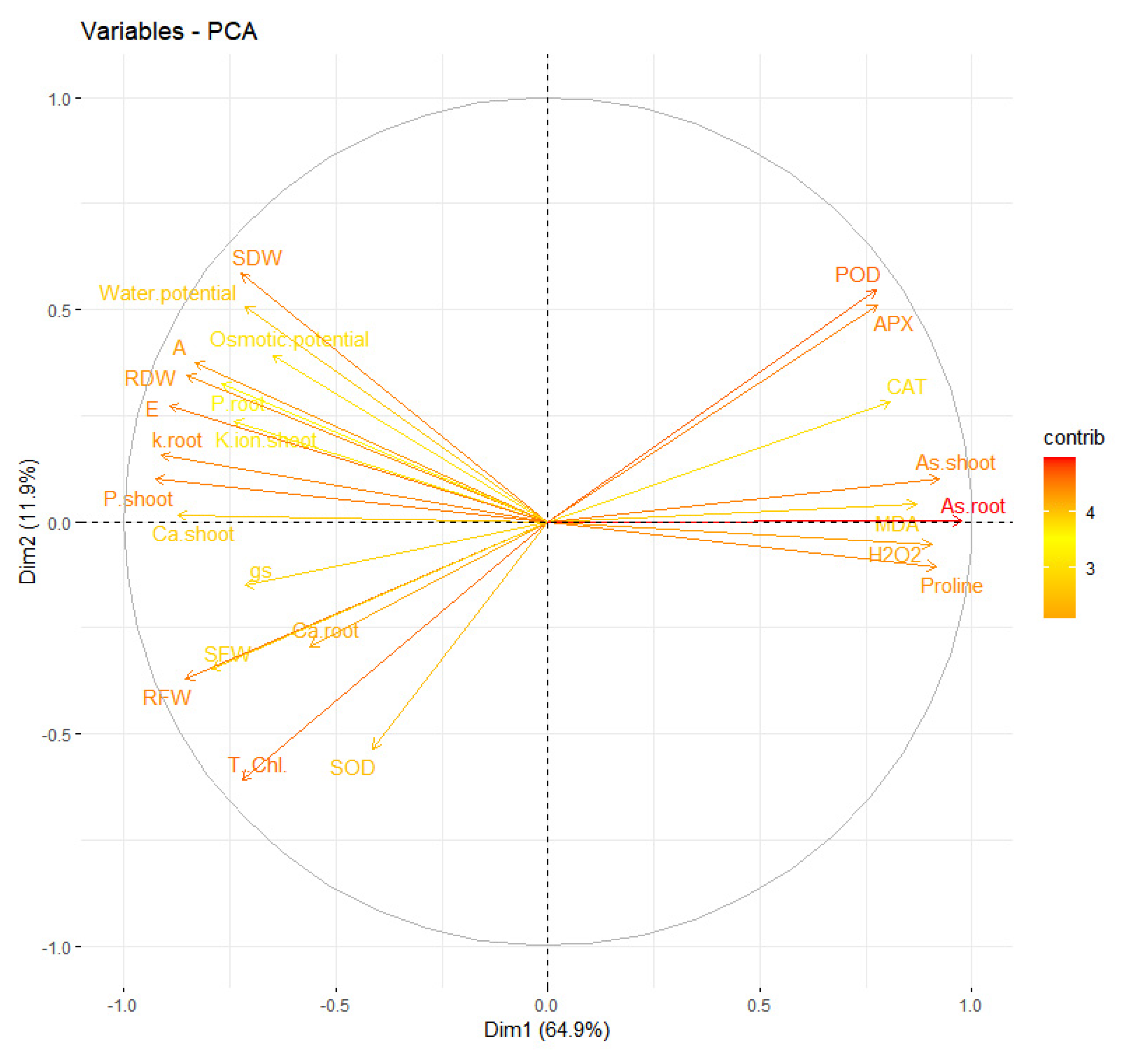
Figure 6 and
Figure 7.