Highlights
What are the main findings?
- MFC breakdown, electrochemical principles for generating bioelectricity.
- MFC operation mechanism and applications.
- MFCs previous perspectives.
- RSA energy crisis, MFC technology viable solution.
What is the implication of the main finding?
- MFC suggests that, in theory, bioelectricity can be produced from organic content using just chemical energy.
- MFC can be used in plant operations on a daily basis to cut operational costs.
- Existing research shows that MFCs are viable, but scaling-up is very necessary.
- For the current energy crisis in South Africa, MFCs may be a workable sustainable energy source.
Abstract
Microbial fuel cell (MFC) technology turns chemical energy into bioelectricity in a clean and efficient manner, lowering carbon emissions and increasing bioenergy production. It is a multifaceted technique that has the potential to be a panacea for clean water scarcity and sustainable, renewable energy. In this review, the approach focuses on scaling-up and application prospects at a commercial scale. An outlook on various, previously tried methodologies was generated in order to establish a viable bioelectricity scaling-up approach that is also cost-effective in its design. Precise themes were followed to evaluate previously produced models and methodologies for MFCs: principle and anatomical mechanisms, basic applications, bioelectricity scaling-up potentials from previous work and limitations, then an outlook on MFC feasibility and its wastewater treatment plant (WWTP) energy supply chain. The goal of this paper is to derive a viable approach from prior research in order to comprehend how MFC technology may be scaled-up for commercial and practical power output. Essentially, this article summarizes the current energy predicaments faced by South Africa and proposes MFCs as a new knowledge-contributing technology with electricity scaling-up potential. Conclusively, more research on MFC technique scaling-up operating factors is recommended.
1. Introduction
The creation of a sustainable society will necessitate minimizing carbon footprints, which will reduce the amount of pollution produced and the excessive use of carbon sources. In a specialized area such as wastewater treatment, these two aspects should be addressed simultaneously. As it stands, the paradigm has shifted from disposing of wastewater/waste matter to turning its organic matter into electrical energy. METs have now gained popularity as a viable solution for dealing with this problem. MFCs and MECs are both fundamental disciplines of METs [,].
An MFC is a process unit that biodegrades complex substrates and produces bioenergy concurrently []. This technology henceforth produces multiple bioenergy products such as bioelectricity, hydrogen, methane, etc. Several rapidly biodegradable chemicals, such as glucose and acetate, as well as several types of wastewater, including residential, starching, and paper recycling plant effluent, have been adopted as a source of electrogenes in MFCs [,,]. Most may remove a significant amount of chemical oxygen demand (COD) while also producing power. The United States (US) space program sparked the growth of MFCs in the 1960s as a way to dispose of garbage during space flights while simultaneously providing power [,]. MFC technology has been carefully studied, with a focus on current developments, practical applications, and a future roadmap. Due to their viable novelty and multifaceted approach to generating bioenergy concurrent to wastewater treatment, MFCs have since been studied immensely. Furthermore, some recent MFC modifications that used an anoxic cathode enhanced the external voltage at the cathode. Phototrophic MFCs and solar-powered MFCs are also noteworthy attempts at upscaling MFC technology for electricity generation []. MFC technology provides a flexible way to generate energy while also treating wastewater. MFCs are a renewable energy technology that can meet the needs for both clean, reliable energy and fresh water. More effort is needed to elevate MFC technology to a commercial level [].
Energy generation, the utilization of carbon-free sources, the biodegradation of chemical matter, wastewater treatment, bioenergy generation, and the remediation of pollutants such as nitrates are all advantages of MFCs [,,,,,,]. For full-scale commercial deployment and the exploitation of its capacities, inordinate knowledge is required to understand the electrochemical active microbial community, as well as intensive efforts to boost electricity generation potential. Specifically, this field of MFCs still requires ongoing research and development. The extensive application of these microbial fuel cell technologies to scale up energy generation is one of the field’s challenges. These systems exhibited power densities of only 2–20 mW/m2 under optimum laboratory circumstances [,]. Microbial processes, on the other hand, use extremely little biomass-based energy. Their full potential for real-world or pilot-scale power generation has not yet been realized. As a result, the present possibilities for scaling up MFC technology for both power production and efficient industrial wastewater treatment shall be discussed in this paper, as well as its potential for actual commercial applications with appropriate operating and cost-effective designs. This study focuses on the use of MFCs for bioenergy production, with a particular emphasis on the current possibilities for scaling up to commercial applications. The overall goal of this review manuscript is to analyze various aspects developed in previous studies. Hence, it aims to determine how MFC technology may be scaled-up in order to generate more bioenergy. In addition, the study will evaluate MFC technology as a viable option to exterminate the current energy crisis in South Africa and its energy sector. A detailed analysis of events leading to the current power shedding predicaments currently occurring in the South African nation at large has been carried out. Specifically, a viable MFC design for possible application towards contributing to the South African national energy grid is proposed. Precisely, this work has been structured as per the following: MET blueprint, MFC layout and operational mechanisms, practical applications of MFCs, MFCs’ previous scaling-up attempts, recent advances in MFC scaling-up potential and challenges, and then, consequently, a discussion on the current South African energy predicament with MFC technology posing a viable solution to the grid. The future prospects of this technology are also discussed in closing in order to uncover possible bioenergy scaling-up concepts in MFCs.
2. Microbial Electrochemical Technologies Blueprint
METs connect bacterial respiratory mechanisms to an electrochemical system []. The following portion of this paper provides an overview of some of the configurations and prospective applications of METs: the MET layout, with its subsidiary bio-electrochemical systems, MFCs and MECs, is clearly outlined to be a technical bioconversion of wastewater in the form of complex substrates into bioenergy, either in the form of bio-hydrogen, bioelectricity, etc. The primary product solely depends on the basic MET operating system objective and methodological approach.
There are numerous potential uses that are more specialized than the two main proposed paths for the industrial application of microbial electrochemical technologies, MFCs and MECs [,,,]. To list a few, bio-sensing, biocomputing, and fundamental studies of microbial metabolism. Because microbial electron transfer enables direct interaction between biological processes and electrical circuits, MET is helpful for bio-sensing because it can be integrated with conventional computing systems and produce short response times. Utilizing MET for biocomputing has the same drawback and furthermore has the ability to incorporate the intricate regulatory apparatus of bacterial cells into the biocomputing circuit []. The microbe–electrode interface is useful in fundamental metabolic research because it enables the direct, real-time assessment of a population of cells’ respiratory activity, a characteristic not present in any experimental system. New concepts and applications occasionally surface in the vast and dynamic field of MET research [].
To create chemicals such as hydrogen, methane, ethanol, or hydrogen peroxide, MECs use electrode-respiring microorganisms [,]. Despite having a similar construction to MFCs, MECs use a resistor or power source to circumvent thermodynamic restrictions by introducing an external potential into the system. Due to the low cost of fossil fuels, the use of MFCs to produce energy in the real world is unlikely, but the increased value of MEC products (e.g., methane, hydrogen, hydrogen peroxide) makes MECs a more promising technology []. Biological, chemical, and physical restrictions that limit MFC scale-up also restrict the development of MECs; therefore, there are still numerous application-related obstacles to overcome []. The cathode’s accumulation of gaseous products, such as hydrogen and methane, also poses difficulties []. MEC technologies are promising, but they are still in the experimental stage, and they need to be shown to be overall efficient and reliable in the long term before they can be used in real-world applications [].
MFCs use bacterial metabolism to produce a current. In order to transport electrons from a usable substrate (typically suggested as industrial effluent) through a circuit that eventually reduces a terminal electron acceptor, electrode-respiring bacteria must be able to donate electrons to negatively poised electrodes [,,,,,]. If a load is introduced to the circuit, the generated bacterial current can be used for work. Bacteria can breathe or accumulate charged electrodes in a variety of ways, including by direct respiration via extracellular or outer-membrane proteins, mediator-based respiration via endogenous or exogenous mediators, and mediator-based respiration via endogenous or exogenous mediators. This review manuscript’s focus is on the investigation of the bioelectricity mechanisms in microbial fuel cells as well as their optimization for scalable and up-scaled bioelectricity production. In a microbial fuel cell, electricity is only produced if the whole process is thermodynamically favorable []. Gibbs free energy, which is measured in Joules, can be used to analyze the reaction []. This is a measure of the most work that can be carried out because of the reaction and is calculated as follows:
Given that, Gr (J) presents Gibbs free energy at a stated process range. Gor (J) relates to Gibbs free energy within a normal range, mostly set at 298.15 K, 1 bar pressure, and 1 M assays for all components []. R = 8.31447 (J.mol/K) presents the universal gas constant, T (K) is absolute temperature, and π relates to the reaction quotient commonly found as the activities of the products divided by those of the reactants []. The standard Gibbs free energy is then found from the energies of formation for organic compounds in water. In MFCs, It is more practical to compare the reaction to the entire electromotive force of the cell (Eemf), (V) [], defined as the voltage difference between the cathode and the anode. This relates to the work done (Energy) (J) in the MFC;
Given that Q = nF, n is the number of electrons per reaction mole, Q is the charge transferred in the reaction represented in Coulomb (C), and F is the Faraday constant, (9.64853 × 104 C/mol) []. Hence, we have:
This model above simplifies to the following model, which is useful because it is positive for a favorable reaction and instantly generates a value of the Eemf for the reaction []. The MFC voltage is capped by this Emf, but due to various potential losses, the actual potential from the MFC will be lower:
2.1. Microbial Fuel Cell (MFC) Principle and Anatomical Mechanisms
MFCs use bacterial metabolism to generate electricity. Electrode-breathing microorganisms that can donate electrons to negatively positioned electrodes transfer electrons from, typically, wastewater via a circuit that oxidizes the catholyte. If a load is presented to the circuit, the bacterial current can be used for work. Bacteria can breathe or populate with charged electrodes in a variety of ways, such as directly via extracellular or outer-membrane proteins, as well as via internal or external mediators. Figure 1 illustrates the traditional layout and bio-electrochemical steps that occur in an MFC during the biodegradation of high-strength organic contaminated substrates for electricity production.
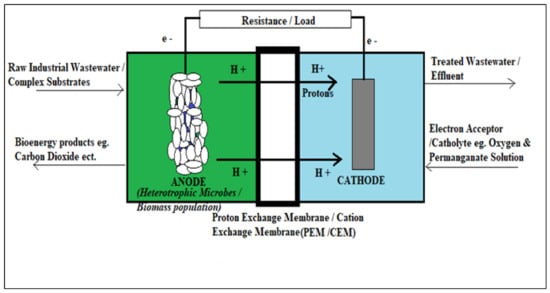
Figure 1.
Summarizes typical process functioning of an MFC chamber, all compartments outlined.
The basic compartments of a conventional MFC unit are outlined in Figure 1. In general, proton exchange membrane (PEM) and cation exchange membrane (CEM) refer to semi-permeable membranes. Other porous separators can also be considered to allow the permselectivity of protons and ions []. The most commonly used CEM or PEM is Nafion from DuPont (Wilmington, DE, USA). Perflourosulfonic acid membranes can also be considered but are limited due to their expensive nature in terms of the material of construction and its unfriendly environmental waste issue. The CEM or PEM exudes a certain permselectivity, which only allows passage for protons, in order to avoid the diffusion of trace amounts of oxygen into the anodic chamber. Frankly, the CEM or PEM serves as a barrier to undesired active species during the electrochemical and biodegradation process of the anolyte to produce bioenergy. In the anodic biofilm, the bacterial community mostly oxidizes the organic substrate’s organic compounds []. Due to the biodegradation process, electrons are discharged onto the anodic electrode and flow towards the cathodic compartment through a resistor load wire material [,,,,,]. Protons are released simultaneously with electrons in the anodic chamber, allowing them to pass through the semi-permeable film known as a PEM. This layer only enables protons to flow through and prevents oxygen from diffusing back into the anodic chamber. Protons migrate from the anode to the cathode chamber during a process known as electrogenesis, which is enabled by the PEM or CEM specialized membrane unit. As a result, the electrogenes are the viable active biomass cultures that release these electrons. This process underpins the basic principle of establishing green technology relevant to the MFC. MFC technology is based on the straightforward change of waste biological substances in the form of biological power into electrical energy. This biological conversion is ascertained in the presence of active electrogenes [,,,]. Figure 2 summarizes the block process diagram of the typical mechanism of bioelectricity generation in an MFC. It is imperative to note that complex substrates harvested from different industrial sources with common organic characteristics of high CODs, high TOCs, and high BOD summarize the nature of the source of fuel fed into the MFC. Figure 2 summarizes a clear description of both biodegradation and electrochemical process transpiring in a typical MFC system.
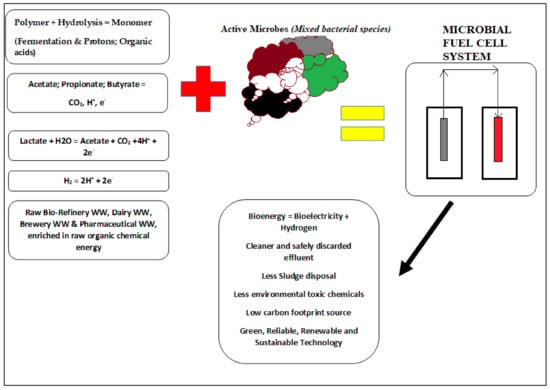
Figure 2.
Summary of the wastewater treatment framework in an MFC for bioelectricity production.
Electrodes, separators, and electrogenes are the three main components of a standard MFC. Some carbon-based, graphite-based, and metal-based electrodes have been recommended in previous studies. Carbon cloth, carbon paper, carbon felt [,], graphite granules, carbon mesh [,], platinum, platinum black, activated carbon single, tubular, or a design with many electrodes. Biocompatibility, stability, good electrical conductivity, and a broad surface area are all desirable characteristics for these electrodes [,,,,]. The cathode can be exposed to air or other electron acceptors such as permanganate, chromium hexacynoferate, azo dye, and so on []. To maintain the chamber’s cleanliness, a cation exchange membrane or a salt bridge has been utilized as a separator [,,,,,,,,,,,,,,,,]. While the microbial breakdown of waste materials provided as substrate generates bioelectricity, the electromotive force formed between the anode and cathode chambers pushes electrons to run on the circuit [,,,,,,,,,,].
2.2. Analysis of the MFC Compartments
The major components of a typical MFC unit utilized in the treatment of wastewater while producing energy are itemized in Table 1. The essential aspects that have been studied are anticipated to highlight the most important factors to consider while designing, constructing, and utilizing MFC prototypes for any given industrial application. The most prevalent building materials, current market suppliers, and advantages and downsides of these components have all been thoroughly analyzed in Table 1.

Table 1.
Perspectives on Key Design MFC Components.
2.2.1. Anode Chamber
The anode chamber acts as sort of the only anaerobic zone of any typical MFC bioreactor. The succinct biodegradation and flow of electrons from the complex sourced substrate to the external biofilm of the anodic electrode transpires here. For increased power generation, there is a need for properly cultured and well screened and selected active electrogenes, which are the source catalyst for the electrogenation process in this chamber. A properly operated and well-commissioned anode chamber will definitely result in an optimized overall potential difference in the MFC unit, hence upscaling the production of bioelectricity within this technique. In any case, one has to investigate the basic contributing factors towards the scaling-up of electricity production within an MFC in this chamber []. These factors may include the flow of electrons from the anode to the cathode chamber, hence resulting in smooth and improved potential difference, and the microbial activity rate mechanism of this chamber. The microbial respiratory principles are monitored and effective in the anode chamber. The pH medium and the effect of temperature have a much more impactful contribution in this chamber of the MFC unit. Precisely for efficient MFC unit scaling-up towards the production of increased power densities, the anode chamber has to be properly designed with effective materials, design layout, and mechanical configuration, and the solution chemistry for the microbes or electrogenes in this chamber has to be properly observed.
The sequence of existing augmentations with total inner surface area is as follows: (i) carbon felt, (ii) carbon foam, and (iii) graphite, according to [,,,]. There have not been any studies done on the long-term effects of biofilm formation or particles in the flow on any of the above surfaces. Manganese, Mn (IV) and Iron, Fe (III) were included, and covalently bonded neutral red was used to mediate electron transport to the anode [,,]. In order for the anode to produce electrons, microbes are crucial. Due to the effectiveness of substrate oxidation and its impact on microbial activity rates, the structural and basic properties of the anode have a major impact on MFC efficiency [,,]. One of the most important factors affecting MFC performance is anodic microbial electron flow, which improves the rate of microbial electron transfer utilizing a variety of practical techniques and modifies the electrode and cell design [,,,]. As a result, anodic electrodes are essential parts of an MFC and play a vital role in improving its effectiveness. Therefore, when establishing a standard anode in an MFC, appropriate anode materials should be considered. It should have a big surface area and be inexpensive, noncorrosive, and highly conductive, as shown in Table 1 [].
The most typical anode material, as shown in Table 1, is carbon-based and includes metal electrodes as well as graphite rods, felt, brushes, and fibers. Due to their low cost, simple operation, and high pore stability, carbon-based electrode materials (plates or rods) are crucial components for anode electrodes [,]. Due to their large surface area and porosity, compact materials such as carbon-based electrodes are promising for the development of biocompatible and active microorganisms that can viably produce energy from complex substrates []. Carbon felt and carbon brushes have higher overall power outputs of 2437 and 2110 mW/m2 (90% COD elimination) [,,]. Several studies on the suitability of anode materials have demonstrated and proved that improvement in MFC performance can be accomplished primarily through a highly porous structure. Despite the fact that carbon-based materials are commonly used as an anode, nitrogen-pre-treated carbon electrodes can achieve even higher power densities [,,]. Corrosion resistance and cost-effectiveness are vital aspects of metal-based electrode materials []. Various types of anode materials are alternatives, especially carbon-structured electrode materials [,,,,,,,,,,,,,,,,,,,,,,,,,].
2.2.2. Cathode Chamber
This compartment sort of acts as the final stage of the MFC electricity generation stepwise process. It is the last unit operation section of the MFC technique. This chamber, as aforementioned in the above section, aids the completion of the flow of electrons and completes the reduction of the treated wastewater towards basic cleaner effluent, free of high organic or biodegradable pollutants and perhaps particulate or non-biodegradable pollutants with the aid of exogenous mediums, e.g., permanganate solution. Although cathode electrodes have made significant strides, they still have drawbacks such as high cost, surface toxicity of microbes, and insufficient re-oxidation, necessitating the routine maintenance of the catholyte []. These factors have led to research into more appealing materials to increase MFC power output. Among these are materials with a carbon basis, metal oxides and complexes, and others. This solution improves the electron reception of this chamber while treating inorganic contents from the complex substrate such as phosphates and nitrates. A good cathodic chamber with optimized operation factors and proper configuration would assist in upscaling the production of bioelectricity in the MFC unit.
The practice of oxygen cathodes is restricted for MFC designs that can stand low performance due to the comparatively low oxygen acceptance rates of plain carbon and the associated significant overpotential. In saltwater, microbial assistance for oxygen reduction on carbon cathodes has been shown [,,]. Stainless steel cathodes, which rapidly decrease oxygen when aided by a bacterial biofilm, have also been reported to undergo microbial-supported decline. Platinum (Pt) catalysts are commonly used for dissolved oxygen [,,,,,,,,,,,,,] or open-air cathodes, enhancing oxygen reduction rates. The platinum load for the MFC can be retained as low as 0.1 mg/cm2 to reduce costs []. Platinum’s long-term strength needs to be further examined, and new types of low-cost catalysts are still needed. MFC cathodes made of pyrolyzed iron (II) phthalocyanine have recently been recommended as noble-metal-free catalysts [,].
In numerous studies, carbon-based cathode electrodes have assisted in= significantly advancing high catalytic activity and performance. One investigation on heteroatom-doped carbon, for instance, showed significant cathodic performance (1328.9 mW/m2) that was comparable to the traditional catalyst Pt/C utilized in cathode cells (1337.7 mW/m2) []. It has been suggested that cathodes made from carbon fiber cloth, polyvinylidene fluoride, and the catalysts Mn, O, Fe, and C could serve as cathode-viable electrodes for wastewater treatment, with low operating costs and appropriate removal efficiency. According to research, 90% of the COD, 80% of the ammonium, and 65% of the total phosphorous were successfully eliminated []. A study carried out in a stacked MFC with a fed-batch operating time of 48 h successfully removed 97% COD from the wastewater while achieving a maximum power density of 1.7 W/m3. Granular activated carbon (GAC) compact electrodes were utilized in the experiment []. Remarkable MFC performance was attained when graphite plates were used in both electrodes, resulting in a power density of 1771 mW/m2 []. Additionally, 90% oxidative removal of tetracycline hydrochloride by reactive oxygen species was achieved using incapacitated GAC on the cathode. There has been a lot of attention on carbon and metal nanoparticles with novel combinations and varied dimensional structures []. A thorough study was conducted on the utilization of a bio-cathode to remove carbon and nitrogen while also generating electricity [].
2.2.3. Cation Exchange Membrane (CEM)/Proton Exchange Membrane (PET)
This section acts as the rudimentary passage to the precise protons that are released during the reduction of the wastewater as the suitable substrate biodegrades and produces the viable electrons and protons in the process of electrogenation. A CEM is required in mainstream MFC designs to partition the anode and cathode chambers. Natural separation systems, such as sediment MFCs [,,,] and, particularly, fabricated single-compartment MFCs [,,], are exceptions. The concept and implementation of ion exchange membranes are gradually rising, demanding more broad and vital studies to measure the membrane’s impact on performance and lasting steadiness [,,,,,,,,,,,,,,,,,,,,,,,,,,,,,,]. Rossi et al. [,,,,,] studied the effect of an anion exchange membrane (AEM) utilized to create a membrane electrode assembly (MEA) in an MFC, with the anode, AEM, and cathode placed closely together to improve the movement of hydroxide ions from the cathode to the anode, eliminate pH imbalances, and shorten electrode distance. The MFC produced 5.7 W/m2 using a flow-through felt anode. Due to the effects of localized pH on the performance of the electrode, MFCs may be restricted to low power densities. The anodic biofilm’s acidification reduces the amount of current the bacteria can produce, and the oxygen reduction reaction’s increase in cathode pH lowers the potential of the entire cell. In light of the aforementioned facts about some aspects of ion exchange membrane technology, it can be concluded that most MFC applications can achieve practical scale-up power by using this technology, which is better suited for high power densities and more compatible with the thermodynamics of bioenergy production by active bacterial species with few restrictions.
2.3. Summary of Challenges and Improvements of Electrode Materials of Construction towards Scaling Up
The procedure, application, and general design of the CEM/PEM membranes and electrodes largely affect the overall performance and cost-effectiveness of the MFC system. The performance of MFC can be enhanced by using efficient electrode materials. This is such that activation polarization losses in a fuel cell might vary depending on the anode materials employed. Evaluating an effective electrode material is one of the biggest difficulties for the MFC to operate as an affordable and accessible technology []. As such, both in anode and cathode configurations, Pt and Pt electrodes outperform carbon and graphite-based electrodes, despite being much more expensive [,]. Studies reveal that the least resistant electrode materials are the most effective, so it is vital to measure their resistance power in order to select the most effective electrode materials []. The ideal electrode material, applicable to both anode and cathode, should be non-flammable, conductive, non-fouling, and affordable. In large-scale applications, adopting a high-efficiency electrode material such as platinum is not economically viable and perhaps impractical, according to the literature and aforementioned sections [18. As a result, current MFC research has shifted focus towards improving the overall fabrication, installation, and operational cost-effectiveness of this system with commercially viable electrodes and membranes in overall a more feasible and realistic operation methodology of this technique. Wei et al. [,,] concentrated on the significance of the high conductivity and mechanical strength required for effective electron transport in the realm of material properties. To increase bacterial adherence, the electrode’s surface area needs to be augmented and alienated into several different conformations []. The literature claims that a nanoparticle-modified electrode produces more power than a regular electrode [,].
3. Current Applications of Microbial Fuel Cells Commercially
In earlier studies, MFCs have been demonstrated to be a feasible and adaptable technology that has been explored across a wide range of applications []. This method is supposed to convey a strong sense of sustainability and dependability because it produces very little waste and requires no new raw materials. Instead, it is a full cleaning technology. MFCs utilize organic waste matter as a source of fuel in the cell to generate power and other bioenergy products such as hydrogen, methane, and biosensors, rather than discarding it, while also purifying wastewater for industrial reuse and maintaining zero liquid effluent discharge potentials []. Figure 3 summarizes the various applications and major uses of MFC technology. This technology stands out as a promising future solution with several clean, reliable, and sustainable bioenergy potentials.
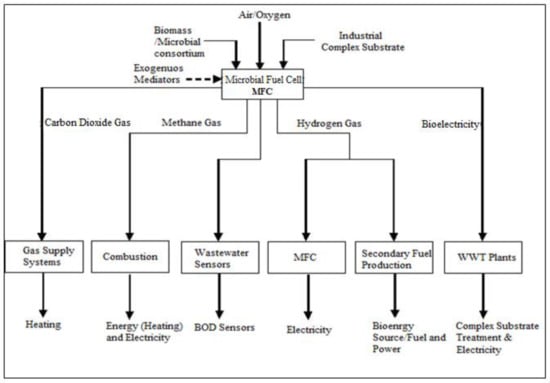
Figure 3.
Outline process block diagram (PBD) for the practical applications of MFCs.
From Figure 3, we can clearly identify the various applications of MFCs as a green technology with multiple renewable and recycled products in the form of energy, heat, biological oxygen demand sensors, etc. Logan et al. [], presenting MFC technology as a multifaceted approach towards the global energy crisis and continued climate change issues, have reported this theorem. More versatility and application of MFCs are summarized in Figure 4 and Figure 5 for MFC applications in a typical wastewater treatment plant at different phase stages, sufficing the same principle of the basic conversion of organic waste into renewable energy. In these figures, the MFC is applicable for wastewater treatment simultaneous to achieving continued bioelectricity generation and zero liquid effluent discharge (ZLED), free of high organic pollutant strengths.
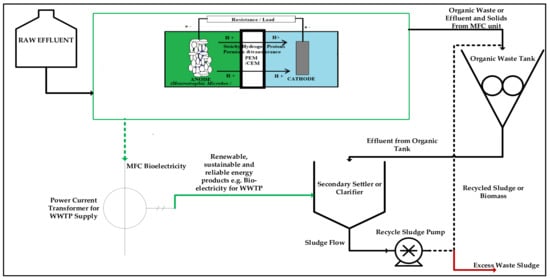
Figure 4.
Summarized flowchart of the possible practical installation of an MFC at the secondary treatment stage, feeding into a downstream solid contacting vessel, a secondary settler, solids pumps, and a recycled sludge stream to the solids tank.
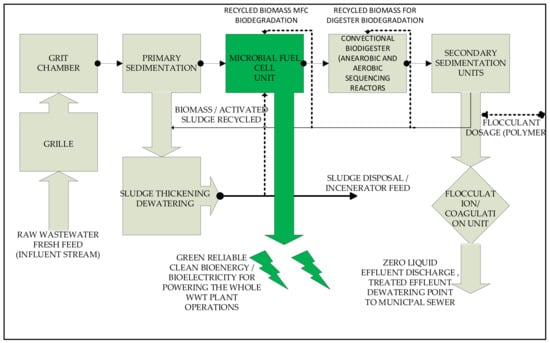
Figure 5.
Process block diagram for MFC installation in a convectional wastewater treatment plant at the primary sedimentation stage for enhanced substrate concentrations.
Figure 4 and Figure 5 pictorially outline the typical process path in the form of a process flow diagram and block process flow diagram. This pictorial view summarizes the detailed stages whereby the MFC unit is applied in a typical wastewater treatment plant. It is critical to note that the MFC unit is applicable just before the convectional and usual bio-digesters and physicochemical treatment units. This is primarily to harness the fresh effluent or industrial complex substrate upstream before processing, such that the substrates act as the perfect source of electrogenes and a source of fuel in the MFC unit. Moreso, it is imperative to note that the MFC at this point and stage of the plant treatment is in a position to generate power to operate the whole wastewater plant unit. At this stage, the source of clean, reliable, and renewable bioenergy can be tapped in the form of bioelectricity, methane, hydrogen, and carbon dioxide, which can and are commonly used as reliable, renewable, and sustainable energy sources. MFCs are typically positioned upstream in a wastewater treatment plant. This is to target the high-strength pollutants that are considered electron transfer and growth boosters. This quality of industrial wastewater help bio-electrochemically active microorganisms grow faster during wastewater treatment as it acts as a source of fuel to the active biomass to produce and convert the contained adenosine triphosphate (ATP) energy from chemical energy into bio-electrochemical energy [,,,,].
Another application of MFCs, summarized in Figure 3, is for secondary fuel production, which has been reported as viable and practicable commercially. MFCs can be implemented in the generation of by-products such as hydrogen (H2) as an option for bioelectricity with modest adjustments. The cathode receives any electrons and protons generated in the anodic chamber, which consequently combines with electrons produced in the water under conventional experimental conditions. Hydrogen creation is thermodynamically unfavorable; this implies that it is a challenging process for a cell to totally overcome the reaction’s thermodynamic barrier and generate H2. As a result, at the cathode, the ions created by the anolyte unite to make H2. When compared to the amount of H2 produced by the traditional glucose fermentation process, MFC is likely to yield more. Using the heterotrophs and autotrophs of Shewenella oneidensis MR-1, single-chamber membrane-free MECs were developed and engineered with a high success rate of hydrogen production from organic substrate material [,,,,,].
MFCs can also be applicable as reliable wastewater biosensors, as presented in Figure 3. MFCs with a changeable anaerobic consortium cannot be instigated as a biosensor to measure organic matter in real time. Although a variety of traditional approaches have been instigated to determine organic content in wastewater in terms of biological oxygen demand (BOD), the majority of them are inadequate for the operational monitoring and management of raw wastewater treatment processes. MFC can function as a BOD sensor due to a direct correlation between Coulombic efficacy and pollutant strength in the complex substrate [,,,,,,,,,,,,,,,,,,,,,,,]. The MFC’s Coulombic yield efficiency offers an estimate of BOD that has proven to be a reliable and accurate method for measuring BOD values in process wastewater treatment operations over a wide testing range of organic matter []. As a continuous BOD sensor, a mediator-free microbial fuel cell has been reported [,]. BOD levels of up to 100 mg BOD/L were assessed using a direct correlation between Coulombic yield and organic matter strength at an incoming flow of 0.35 mL/min (retention time of 1.05 h) [,,,,,,,,,,,,,,,,,,,,,,,,,,,,,,,,,,,,,,,,,,,,,,,,,,,,,,,,,].
Waste Management Hierarchy That Incorporates MFC Technology as a Viable Solution to Both Waste and Energy Shortage
A logical starting point for understanding pollution prevention concepts is the concepts of waste and wastewater management in any typical practical plant environment. The wastewater management concept has a defined and applicable hierarchy in a WWTP. It is vital to follow the imperativeness of wastewater in a WWTP as not just a waste commodity but also a natural resource to be converted into bioelectricity, as it has the potential to exude an adequate adenosine triphosphate capacity that can easily converge into electricity in the presence of good biodegrading biomass and other biological mediums such as the MFC unit. This is summarized in a sequential manner in Figure 6. The versatility and sustainability of an MFC unit are presented to supply the whole WWTP with just the adequate amount of power, with the practical approach of being the stand-alone weekend power supply source, as per Logan et al. [].
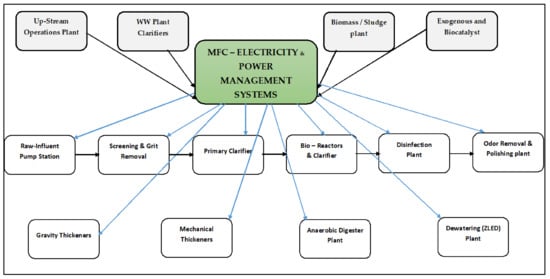
Figure 6.
Typical waste management hierarchy that incorporates MFC technology as a viable solution for both waste disposal and energy shortages.
4. Previous Outlooks on MFC Electricity Generation and Scaling-Up Capacity
Waste biomass provides an affordable and abundant source of electrons for bacteria capable of producing electrical currents outside the cell []. MFCs are rapidly gaining traction and are part of a broad range of essential future energy and chemical production technologies [,]. This study is interested in learning operating conditions that will allow exo-electrogene bacteria to produce biofuels, hydrogen, methane, and other valuable inorganic and organic chemicals in the future. It will also look at the major challenges of putting these systems in place, as well as compare them to other renewable energy technology options. Despite the fact that commercial development in a number of applications ranging from wastewater treatment to industrial chemical synthesis is underway, more studies in the areas of efficiency, scalability, system lifetimes, and dependability are recommended.
In an MFC, the anaerobic oxidation of organic molecules by bacteria can be a reliable source of energy. An anode chamber and a cathode chamber are the two compartments in a conventional MFC system [,]. In the anode chamber, microorganisms decompose biodegradable organic molecules and deliver electrons to the anode electrode (negative electrode) [,,,,,,,,,,,,,,,,]. Electrons travel through a circuit to the cathode chamber (positive electrode), where oxygen or other chemicals such as ferricyanide accept them [,,,,,,,,,,,,,]. Water (from oxygen) or ferricyanide is formed when these electrons mix with protons that diffuse from the anode chamber to the cathode chamber (ferricyanide) [,,,,,,]. Essentially, this process of protons diffusing from the anodic chamber to the cathodic chamber effectively relies on the quality and type of PEM that separates these chambers and optimizes the chances of achieving a bigger load upon the completion of the load on the cathode side. This chamber determines the feasibility of the thermodynamic favorability of the MFC unit, hence the capacity of this technique to generate bioelectricity [,,,,,,,,]. Table 2 summarizes current viewpoints gleaned from recent investigations, which are quickly emphasized and clearly summarized based on the various experimental approaches and operating parameters used to carry out these methods. Across the board, it was perceived that there is an imminent issue of very low power densities. This has severely limited the feasibility and efficiency of this operation at the experimental scale. A critical description of the evaluated work’s merits and weaknesses was also highlighted in order to comprehend the MFC technique’s future prospects and use.

Table 2.
Summarized perspectives on MFC studies.
From the analysis of Table 2, it can be perceived that MFC technology has the potential of being a reliable source of energy concurrent to wastewater treatment. It is gathered that high COD removal efficiencies were accomplished with reasonably low overall power densities that still need scaling up towards commercial applications. More research work is imperative based on the findings of Table 2.
According to Feng et al. [], the development of microbial fuel cell electric power was investigated in order to contribute to MFC technology applications and space research. The applied microbial cultures responsible for producing bioelectricity from chemical energy contained in substrates were discovered to be one of the decisive variables in MFC technology. Despite intensive growth over the previous decade, the knowledge gap related to microbial power generation is an issue. The quick screening approach relied on microbial iron (III) reduction and did not necessitate the use of MFC infrastructure []. The approach can be used to evaluate multiple microbe species or strains at the same time, allowing researchers to expand the spectrum of potential MFC biocatalysts and anticipate how much power the cultures would generate []. The knowledge derived from this work on growth, iron (III) reduction, substrate utilization, adhering, biofilm-forming properties, extracellular conductive proteins, and redox mediator production measurements is critical for the use of G. toluenoxydans and S. xiamenensis species in various MFC applications (wastewater remediation or energy generation) [,,]. A promising MFC approach with such good energy outcomes was presented in this study. However, a need for scaling up this technology is evident, based on the quality of microbes and its solution chemistry knowledge, to best utilize the biomass or microbe chemical energy towards efficient bioenergy-producing electricity.
Min et al. [] demonstrated that a flat plate system for power generation from residential wastewater could constantly produce energy from biological species in wastewater while performing complex substrate remediation processes. After a one-month acclimation period, the FPMFC generated steady electricity from wastewater throughout the 5-month treatment period. For a fresh feed influent stream of 2463 mg COD/L, an average power density of 560 mW/m2 was produced at an HRT of 2.0 h (0.22 mL/min flow rate; 164 mg/L log mean COD), an airflow rate of 2 mL/min, and a 470 ohms resistor [,,]. Under these operating conditions, the COD removal rate was 1.2 mg/L min (58% COD removal), []. The highest power density was attained at a flow rate of 0.22 mL/min. Under typical operating conditions, this power density is roughly 10% higher than that provided by a 470 ohms resistor []. These results showed the substantial potential of the MFC technology’s ability to produce bioelectricity while treating complex substrates. A practical need for scaling up the overall power density and the unit’s pragmatic installation was observed from this work.
Furthermore, Feng et al. [] used a traditional double chamber system and a typical PEM, which also allowed oxygen to diffuse to the anode. Without a deeper understanding of the features of the bacteria that produce energy in MFCs, there are many possible causes of electricity generation in these conditions [,]. Bacteria adhering directly to the electrode may be able to create electricity even in the presence of low oxygen concentrations. The carbon electrode’s uneven surface, on the other hand, might enable bacteria to grow in the deeper areas of the electrode (near the cathode) to scavenge oxygen before it reaches the bacteria producing electricity on the anodic biofilm. As aspects of continuous bioenergy production are unknown, the ability to have these electrodes so close to one other is a major discovery since it opens the door to more efficient MFC reactor designs [,,,,,,,,,]. A crucial finding of electrode spacing and electrogenation surface area was established in this work, hence adding a positive spark towards the scaling-up potential based on the design and configuration of the MFC unit. This work definitely catalyzed an interest in experimenting with the effect of electrode spacing on increasing the overall output power in MFCs.
Cheng et al. [] have reported an SCMFC for continuous wastewater treatment and energy generation. It was feasible to achieve the highest power density of 26 mW/m2 while removing up to 80% of COD from a complex substrate []. Domestic wastewater might generate up to 28 mW/m2 of power, according to a newly developed smaller batch system [,,,]. When the proton exchange membrane (PEM) was removed, they were able to produce a maximum of 146 mW/m2 of electricity in the form of power density. However, the effluent was not treated continuously in these tests []. The anode remained separate from the cation membrane/cathodic chamber. As a result, it is uncertain if a single electrode/PEM assembly can improve power generation in an MFC or whether oxygen transfer from the cathode to the anode chamber would impede efficient power production in an MFC [,]. From the findings of this work, the above-articulated research questions have to suffice towards the positive prospects of MFC power scaling-up.
In another study, domestic wastewater treatment was investigated using an SCMFC at different temperatures (23 °C and 30 °C) and flow modes (fed-batch and continuous), as reported by Ahn et al. []. The significance of temperature on treatment efficiency and electricity generation was vital and evident. At an OLR of 54 g COD/L-d, [], the maximum power density of 422 mW/m2 (12.8 W/m3) was achieved at constant flow and mesophilic settings, achieving 25.8% COD elimination []. The reactor design, as well as the operational conditions (flow mode, temperature, organic loading rate, and HRT), were found to have a significant impact on energy recovery. According to the findings, using temperature-phased, in-series MFC designs for residential wastewater treatment has various benefits, including decreased power consumption, lower solids production, and increased treatment efficiency.
It is known that MFCs are utilized to produce power from a range of chemicals, with acetate, lactate, and glucose, as recounted by Rabaey et al. []. The feasibility of producing electricity in an MFC from domestic wastewater while also performing biological wastewater treatment was stated. The efficiency of the cathode was the primary determinant of current generation. Allowing passive airflow rather than forced airflow 4.5–5.5 L/min resulted in optimal cathode performance []. Based on COD removal and energy generation, the system’s Coulombic efficiency was reported to be 12%, indicating that a sizeable amount of the biological material was lost without energy production []. Based on the findings of this study, it can be perceived that scaling up electricity generation in MFC systems could give a new alternative to offset wastewater treatment facility operating expenses and hence save many of these chemical plants’ overall utility costs. Therefore, MFCs are a good, reliable, and sustainable technology needed to address these current commercial plant-operating issues.
In another study, Lu et al. [] presented COD and NH4+-N removal. It was reported that removal efficiencies are proportional to retention time, reaching 98.0% and 90.6% by the membrane and electrode assembly air-cathode MFC []. More so, a study of concurrent complex substrate treatment and biological electricity production using starch-processing wastewater (SPW) as a substrate was successfully completed. The findings could have induced high nitrate removal values. The viability of applying MFC technology to produce energy while instantaneously treating SPW with high-range COD and nitrate reductions was established. This study presented an enticing option for reducing wastewater inorganic pollutants while generating power from a renewable resource. Unlike conventional wastewater treatment methods, MFCs are posed as a complete approach that not only biodegrades organic constituents from wastewater but also critically addresses the inorganics for continued zero liquid effluent discharge to local wastewater receiving bodies. This technology indirectly shows its other superior quality of being viable enough to address environmental issues such as eutrophication due to continued nitrates and phosphates dewatering into municipal bodies and running river streams.
5. Current Outlooks on MFC Scaling-Up—Application Challenges
The MFC’s design and mechanical layout are critical for achieving ultimate power densities, whether at the research scale or in practical implementation. This section critically examines the basic, commonly used experimental designs both currently/previously used in MFCs for ultimate power generation. The idea is to clearly summarize the recurring economic challenges based on the design configuration concurrent to economic applications for bigger-scale implementation—either at the local wastewater treatment level or at the national grid level, as an alternative source of bioelectricity to curb possible power outages in the case of recurrent power meltdowns in nations such as South Africa. From an analysis of Table 3, it is observed that the scalability and economic applicability of MFC technology are directly proportional to each other. This implies that in most studies, the complexity of the design simply relates to the real-life possibility of economic application in terms of fabrication costs, operation costs, and general maintenance and overview of the plant as per the normal sequence. It is vital to maintain a simple yet effective and scalable design to curb the overall economic costs of practically employing that particular design of MFC unit operation.

Table 3.
Summary of recent MFC scaling-up attempts.
From the analysis of Table 3, it is evident that there is a serious need to develop a viable MFC prototype that can solve the recurrent issue of electricity scaling-up and its practical application potential. The following sections briefly summarize the current predicaments encountered in MFCs, as outlined in Table 3 above. Based on the above findings, MFCs can be scaled up, but the impact of reactor design and experimental conditions must be better understood [25,40,64,89–91. Logan et al. [] linked the batch enactment of a smaller MFC (SMFC 28 mL) to a larger MFC (SMFC 48 mL) (LMFC 520 mL). The SMFC produced 14 W/m3 with a 4 cm electrode spacing, which is in line with earlier studies. The LMFC produced 16 W/m3 due to its shorter average electrode spacing of 2.6 cm and larger anode surface area per volume (150 m2/m3 vs. 25 m2/m3 for the SMFC). With the addition of graphite granules or graphite fiber brushes to the LMFC anode chamber, the effect of a higher anode surface area on power was proven to be relatively small. Regardless of the fact that the granules and graphite brushes augmented the surface area by a factor of 6 and 56, the highest power density in the ML-MFC improved by 8% and 4%, respectively, according to Sharif et al. [,].
Using NaCl to increase the LMFC’s ionic strength from 100 to 300 mM, on the other hand, increased the power density by 25% to 20 W/m3. In continuous flow mode, the LMFC generated a maximum power density of 22 W/m3 after an HRT of 11.3 h. According to Logan et al. [], a thick biofilm formed on the cathodic chamber. However, at power densities less than 1.0 mA/cm2, the cathode potentials were unaffected. According to Logan et al. [], overall power densities can be sustained during MFC scale-up; anode surface area increase and biofilm formation on the cathode compartment have no significant impact on MFC reactor outcome, and electrode spacing is a critical design parameter for maximizing overall power generation in an MFC unit. Logan et al. [] accentuate the critical difficulties of optimizing power generation in an MFC, described as follows:
- ○
- Need for the influent to reach the entire anode matrix without disturbance.
- ○
- Protons must diffuse rapidly towards the membrane.
- ○
- Suitable electrical contact must be made between the suspended bacterial masses and the anode.
- ○
- To have functional power, sufficient voltage has to be achieved across the MFC.
- ○
- The installation of an aeration device should primarily be re-examined.
Application Challenges
Activation losses, concentration losses, aeration, high initializing costs, and low power densities are only a few of the application’s major restrictions. In light of the foregoing, Logan et al. [,,,,,,,,,,,,,,,,,,,,,] severely dismiss the issue of lower power densities as part of the application issues, citing the fact that MFC technology is still developing and in its early stages, requiring optimized procedures to reach commercial viability. According to the literature, the highest power density is 3600 mW/m2. This was accomplished by treating glucose with adjusted anaerobic consortia in a double-compartment MFC. This process took place in an anode chamber and a cathode chamber that was continuously aerated and contained an electrolyte solution. Oxygen transmission in the cathode compartment was made better by the formulation of the catholyte. One of the most significant barriers to MFC adoption is the high initial cost of design and fabrication, as well as commissioning and operation [,,,]. This technology is generally made of high-cost materials that are essential for electricity regeneration, such as the expensive Nafion membrane, which has been described as the most efficient CEM for increasing power densities. It is reported that numerous attempts have been made to construct a low-cost version of a PEM that would produce the same effective outcomes as the Nafion prototype [,].
6. Perspectives on the Current South African Energy Crisis: MFC Technology as a Potential Solution
The ongoing South African energy crisis is characterized by back-to-back blackouts and load shedding because the country’s electricity supply is insufficient compared to the demand. The prevailing consensus on this situation is that it is the result of repeated national grid failures brought on by poor maintenance and engineering on the part of the national power generator, Eskom. This aspect has clearly posed a threat to the national grid stability and future status, as attempts to redeem this predicament are currently obtuse and seem unfeasible. The government-based national power grid by the national generator, Eskom, and a number of various state officials perhaps also contributed to these ongoing power blackouts due to the poor stability of the national grid sustenance. A reported reserve quantity of about 8% or less of such power blackouts is envisaged every time generating units are deemed offline due to poor scheduled maintenance and repairs [,,,,,,,,,,,,,,,,]. The resolution calls for the construction of new power plants and generators based on information from Eskom and the South African government []. Lately, the current president announced and clearly assured the country is not in a state of emergency regarding these recurrent electricity load-shedding rollouts, but there is a need for the nation to be sympathetic and cooperative to bring the country out of this minor glitch. A need for skilled and veteran personnel in the energy and power industry was announced as a viable direction []. Regardless, the nation always points out the lack of adequate national governance, Eskom governance due to unskilled and scrupulous managerial personnel, and the exorbitant squandering of maintenance and repair funds to have deteriorated the credibility and capacity of Eskom to supply the grid sustainable and reliably.
The South African load-shedding periods, to list a few, include 2007–2008, 2014–2015, 2019–2020, and March 2021 until the present. Generally, Eskom has been criticized, as mentioned above, for exporting abundant power capacities to local Southern African neighboring states, thought to be one of the predicaments leading to electricity power shortages in the national grid [,,,,,,,,,,,,,]. During this time, the average annual growth rate of the gross domestic product (GDP) was 3.1% []. Consequently, predictions of the end of power shortages in 2012 were common [,,,,,,,,,,]. Sadly, Eskom, around the period of the end of 2015, fell short of massive megawatts of power, which was later captured as an outcome of an unplanned maintenance schedule and overall extremely poor planning of this national electricity power generating entity, Eskom.
Around early 2020, the chief operating officer of Eskom at the time announced that the root cause of this unreliability and energy crisis was due to poor maintenance [] and planned unit operations such as power turbines’ timely service, overhaul, and repairs. This was estimated to have prevailed over a twelve-year-long period hence resulting in such a backlog of failures to meet the national grid demand []. To date, the local and sole power generating stations of Eskom have been reported to be under continuous service and repair challenges, breakdowns, shortages of raw fuel supply such as quality coal, etc. [,,,,,,,,,,,,,]. An overview layout in a form of a simple trend in Figure 7 demonstrates the recurrent power blackouts in South Africa and clearly summarizes the need for reliable and sustainable, clean energy from innovative techniques and methodologies.
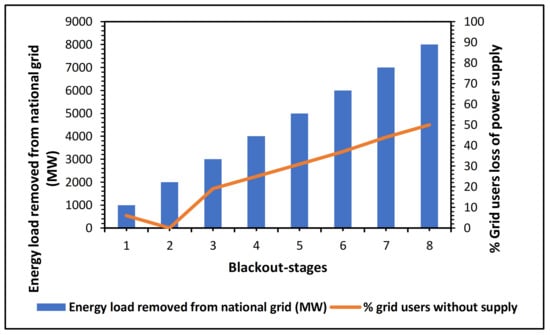
Figure 7.
South African load-shedding stages and national grid users’ impact [].
In general, the South African cents per kWh of electricity over the above-reported periods, marking the start of the energy crisis, as presented in Figure 7 above, was reported by [,,,,,,,,,,,,,] in detail as the beginning of very high increases in the electricity unit cost. This continued failure and unreliability of the local South African energy-generating entity due to a reported misuse of maintenance funds, unskilled management system, continued hiring of unskilled and reliable engineering personnel in the power units, political sabotage, and so forth, is calling for a reliable solution [,,,,,,,,,,,,,].
As observed by the national local government, the national grid is to remain with the state and hence the nation’s outburst on Eskom’s unreliable power supply remains a mystery to be resolved. The current president also made a clear call for the additional input of power to the grid by other sustainable and novel technologies. The need for nuclear power and other hydro-powered and solar-powered technologies has been implored towards contributing to the national grid. However, these techniques have their drawbacks. For example, there are hidden dangers in the safety and environmental friendliness of nuclear technology. Even if one considers relatively green renewable energy sources, wind and solar techniques are unstable and intermittent during generation, and energy storage systems are needed to improve the utilization and stabilization of renewable energy. The above-stated fundamental reasons undoubtedly increase the application and commercial viability complexities of these systems, hence limiting the options of the South African energy sector to consider implementing them. Frankly, these well-articulated drawbacks perfectly present the significance of the development of MFC technology towards answering the current issues of South African national grid shortfalls emanating from recurrent national power shedding. Most of all, this study proposes the use of clean, sustainable, and reliable MFC technology as one novel yet potential solution to the recurrent energy crisis South Africa is facing. MFC technology, with the use of absolutely fewer carbon sources, fossil fuels, etc., not only appears to be a solution for substantiating power but also a resolution to the current global environmental crisis of water scarcity and climate change. The authors in the above sections have discussed the MFC technique proposed in this study as a practical magic bullet and multifaceted approach that can solve the above-underpinned South African energy predicament and possibly provide a global solution to the current worldwide energy and water scarcity [,,,,,,,,,,,,,].
Proposed—Hybrid Technology: MFC Integrated Method for Electricity Scaling–Up Possibilities and Future Work
Based on the aforementioned challenges, it is mandatory to develop a new knowledge-contributing prototype of MFC technology that can viably attempt to address the looming issues of both scaling-up and application potential. From a practical and realistic perspective, the South African energy grid shortage crisis has been an ongoing predicament without feasible mitigating strategies implemented to curb this phenomenon. As summarized in the above section, large loads of national energy are required to eradicate the mismanagement that persists in the national energy grid platform and provider, Eskom, hence a critical need for alternative methods to generate electricity to back up the grid is required. This MFC technology has been existing, but as aforementioned above, with massive shortfalls in scaling-up and practical application potentials. The below-proposed prototype is simple in design and already exists but has been developed by integrating two process unit operations to avoid complex bio-electrochemical designs while improving the overall potential of the MFC in generating clean and reliable electricity. In this regard, Figure 8 and Figure 9 present the alternative methodology attempting to address the most significant grey area in MFC technology, which is bioelectricity scaling-up and practical application potential. As discussed in earlier studies, the type of biomass that will be used as the mediator-free electron donor in the anode chamber has a big impact on how much energy is produced.
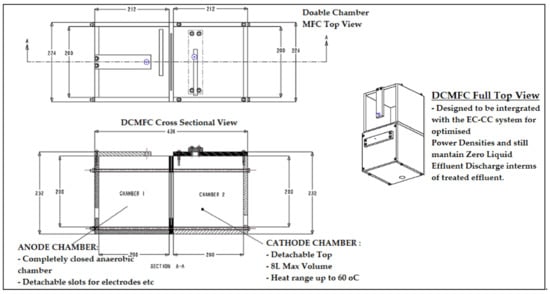
Figure 8.
Proposed novel large double-chamber open-top air-cathode MFC (LDCMFC).
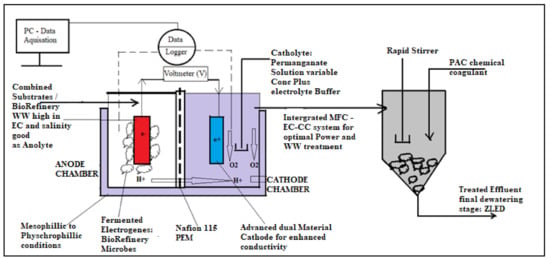
Figure 9.
Summary of the proposed integrated lab–scale DCMFC–flocculator system for enhanced bioelectricity and WWT.
In this prototype, a unique combined DCMFC–flocculator system using an anaerobic, heterotrophic mixed culture as a mediator and an electron donor will be implemented. Based on a particular chosen aeration configuration (anoxic), it is expected that very high rates of nitrification and denitrification, as well as simultaneous nitrification and denitrification, will occur in the cathodic chamber, mostly favoring the nitrates and total phosphate removal. Bakare et al. [,,,,] have previously demonstrated this element of biochemical kinetics. In order to scale up bio-energy production, this MFC method will look into numerous essential issues. The above-mentioned combined unique DCMFC–flocculator strategy will improve the removal of non-biodegradable contaminants, primarily particulate COD and total phosphates, in raw complex industrial wastewater, boosting efficient bioenergy production and reliable WWT, achieving continued zero liquid-effluent discharge standards. In comparison to prior studies, the currently proposed alternative is likely to produce effective outcomes; hence, it will undoubtedly add fresh knowledge to the currently impending research gaps related to the commercial use and scaling-up possibilities of this technology.
7. Future Work
According to the aforementioned research, the concentrations of organic substrate and the overall design parameters of the system’s units determine the amount of energy produced in MFCs through various configurations and operations. More crucially, the expense of these biological processes is inversely correlated with fundamental design considerations and experimental techniques, such as longer HRTs, that seem to be monetarily unviable in terms of application prospects. From an economic standpoint, one may argue that this system still has a long way to go before it can be scaled up for commercial applications. It is necessary to improve and optimize its overall power generation capacity from mWatts to kWatts. Future imperatives include a more in-depth investigation of the impact of the key optimization elements. Finding a sweet spot to boost a computational version of MFCs is precisely advised in order to introduce a fresh understanding of scaling-up chances. It is necessary to conduct a thorough analysis of the chemical energy of the biomass and its capacity to scale up based on its total ATP from extractive cell ATP and dissolved ATP, modeled over critical activity rate monitoring parameters of the active biomass such as BSI (biomass stress index), AVSS (active volatile suspended solids), and active biomass ration (ABR). The information above will help in the development of an MFC model for power generation that will be optimized using MATLAB—Simulink R2022a, which will feasibly predict the potential for the scaling-up of the MFC system based on these components.
8. Conclusions
MFC technology has shown promising potential for both wastewater treatment and electricity generation. It does this through the straightforward conversion of organic and chemical energy in wastewater, primarily through active electrogenes that biodegrade high-strength pollutant constituents into bioelectricity. The fundamental limits of scaling up and applications have yet to be resolved in most MFC experiments, indicating that a scalable MFC unit is still a long way off. MFCs appear to be the most viable means of generating energy, particularly when combined with synchronous wastewater treatment to achieve zero liquid effluent discharge regulations. The goal of this review was to present current upscaling and application potentials, considering the nature of MFC technology, which still requires additional studies to solve these grey areas related to scaling up and commercialization. However, it can be consistently stated that MFC technology is a rapidly emerging niche area that is simple, robust, and entirely renewable and green. More research is required to address the persistent problem of low power densities, which must be increased to at least 1 kW/m3 of complex substrates to assure the method’s long-term viability. In view of the above-stated shortcomings, more studies on MFC technology are in demand and highly recommended as an effort to address these limitations. It is also strongly advised that hybrid and novel integrated techniques and process unit operation configurations be investigated in light of scaling up the overall energy and electricity generation in an MFC because MFCs are a promising technology that needs to be assimilated with convectional existing types. Categorically stated from the above-covered main sections, one concludes the following:
- ○
- For both the anode and cathode compartments, a good electrode material of construction that is highly conducive, less corrosive, affordable, and easily accessible would be a good choice.
- ○
- Previous authors have vehemently argued that a solid grasp of microbial electrochemistry is essential for scaling up bioenergy and obtaining realistic power densities for MFC commercial applications.
- ○
- Since the MFC technology converts simple waste into bioenergy—specifically electricity—it is more dependable, sustainable, and multifarious, which can substantially benefit the current local South African national grid deficits.
- ○
- Achieving practicable power densities to make MFC technology an economically feasible strategy is one of its present limits, in addition to its expensive effectual process operating units.
Author Contributions
Conceptualization, K.P.S., J.K.B. and B.F.B.; methodology, K.P.S. and J.K.B.; validation, K.P.S.; formal analysis, K.P.S.; investigation, K.P.S.; resources, B.F.B.; data curation, K.P.S. and J.K.B.; writing—original draft preparation, K.P.S.; writing—review and editing, K.P.S. and J.K.B.; visualization, K.P.S.; supervision, B.F.B.; project administration, B.F.B.; funding acquisition, B.F.B. and K.P.S. All authors have read and agreed to the published version of the manuscript.
Funding
Mangosuthu University of Technology (MUT), staff research grant, funded this research and The APC charges.
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
Not applicable.
Acknowledgments
B.F. Bakare, who has been supportive, guiding, and patient with my work and proofread the contents of my review manuscript. J.K. Bwapwa (Senior Lecturer and Researcher, Department of Civil Engineering, MUT) played a significant role in advising and proofreading this article. Finally, I would like to express my heartfelt gratitude to the Departments of Research and Innovation at Mangosuthu University of Technology (MUT) and Durban University of Technology (DUT) for sponsoring and funding the publication costs of this review article.
Conflicts of Interest
The authors declare no conflict of interest.
Abbreviations
| AVSS | Active volatile suspended solids |
| ATP | Adenosine triphosphates |
| ABR | Active biomass ration |
| MFC | Microbial fuel cell |
| SMFC | Single-chamber microbial fuel cell |
| DCMFC | Double-chamber microbial fuel cell |
| LMFC | Large microbial fuel cell |
| SMFC | Small microbial fuel cell |
| FPMFC | Flat-plate microbial fuel cell |
| COD | Chemical oxygen demand (mgCOD/L) |
| BOD | Biological oxygen demand (mgBOD/L) |
| TOC | Total organic carbon (mgTOC/L) |
| CE | Coulombic efficiency (%) |
| OLR | Organic loading rates (kgCOD/L.day) |
| EMF | Electromotive force potential |
| MET | Microbial electrochemical technologies |
| MEC | Microbial electrolysis cell |
| WWT | Wastewater treatment plant |
| O2 | Oxygen |
| CO2 | Carbon dioxide |
| H2 | Hydrogen |
| HRT | Hydraulic retention time (hours or days) |
| PEM | Proton exchange membrane |
| CEM | Cation exchange membrane |
| ML-MFC | Membraneless microbial fuel cell |
| H2O | Water |
| NH4-N | Ammonium nitrate |
References
- Rahimnejad, M.; Najafpour, G.D.; Ghoreyshi, A.A.; Jafari, T.; Haghparast, F. Microbial Fuel Cell a Reliable Source for Recovery of Electrical Power from Synthetic Wastewater. Linnaeus Eco-Tech. 2010, 627–635. [Google Scholar] [CrossRef]
- Tamboli, E.; Eswari, J.S. Microbial fuel cell configurations: An overview. In Microbial Electrochemical Technology; Springer: Berlin/Heidelberg, Germany, 2019; pp. 407–435. [Google Scholar]
- Wang, H.; Ren, Z.J. A comprehensive review of microbial electrochemical systems as a platform technology. Biotechnol. Adv. 2013, 31, 1796–1807. [Google Scholar] [CrossRef] [PubMed]
- Shah, S.; Venkatramanan, V.; Prasad, R. Microbial fuel cell: Sustainable green technology for bioelectricity generation and wastewater treatment. In Sustainable Green Technologies for Environmental Management; Springer: Berlin/Heidelberg, Germany, 2019. [Google Scholar]
- Rahimnejad, M.; Ghoreyshi, A.A.; Najafpour, G.; Jafary, T. Power generation from organic substrate in batch and continuous flow microbial fuel cell operations. Appl. Energy 2011, 88, 3999–4004. [Google Scholar] [CrossRef]
- Gudiukaite, R.; Nadda, A.K.; Gricajeva, A.; Shanmugam, S.; Nguyen, D.D.; Lam, S.S. Bioprocesses for the recovery of bioenergy and value-added products from wastewater: A review. J. Environ. Manag. 2021, 300, 113831. [Google Scholar] [CrossRef] [PubMed]
- Ahn, Y.; Logan, B.E. Effectiveness of domestic wastewater treatment using microbial fuel cells at ambient and mesophilic temperatures. Bioresour. Technol. 2010, 101, 469–475. [Google Scholar] [CrossRef]
- Rabaey, K.; Rodríguez, J.; Blackall, L.L.; Keller, J.; Gross, P.; Batstone, D.; Verstraete, W.; Nealson, K.H. Microbial ecology meets electrochemistry: Electricity-driven and driving communities. ISME J. 2007, 1, 9–18. [Google Scholar] [CrossRef]
- Du, Z.; Li, H.; Gu, T. A state of the art review on microbial fuel cells: A promising technology for wastewater treatment and bioenergy. Biotechnol. Adv. 2007, 25, 464–482. [Google Scholar] [CrossRef]
- Lay, C.H.; Kokko, M.E.; Puhakka, J.A. Power generation in fed-batch and continuous up-flow microbial fuel cell from synthetic wastewater. Energy 2015, 91, 235–241. [Google Scholar] [CrossRef]
- Azwar, M.Y.; Hussain, M.A.; Abdul-Wahab, A.K. Development of biohydrogen production by photobiological, fermentation and electrochemical processes: A review. Renew. Sustain. Energy Rev. 2014, 31, 158–173. [Google Scholar] [CrossRef]
- Kumar, G.; Saratale, R.G.; Kadier, A.; Sivagurunathan, P.; Zhen, G.; Kim, S.H.; Saratale, G.D. A review on bio-electrochemical systems (BESs) for the syngas and value added biochemicals production. Chemosphere 2017, 177, 84–92. [Google Scholar] [CrossRef]
- Min, B.; Kim, J.R.; Oh, S.E.; Regan, J.M.; Logan, B.E. Electricity generation from swine wastewater using microbial fuel cells. Water Res. 2005, 39, 4961–4968. [Google Scholar] [CrossRef] [PubMed]
- Noori, M.T.; Ganta, A.; Tiwari, B.R. Recent Advances in the Design and Architecture of Bioelectrochemical Systems to Treat Wastewater and to Produce Choice-Based Byproducts. J. Hazard. Toxic Radioact. Waste 2020, 24, 04020023. [Google Scholar] [CrossRef]
- Pant, D.; Singh, A.; Van Bogaert, G.; Gallego, Y.A.; Diels, L.; Vanbroekhoven, K. An introduction to the life cycle assessment (LCA) of bioelectrochemical systems (BES) for sustainable energy and product generation: Relevance and key aspects. Renew. Sustain. Energy Rev. 2011, 15, 1305–1313. [Google Scholar] [CrossRef]
- McCarty, P.L.; Bae, J.; Kim, J. Domestic wastewater treatment as a net energy producer-can this be achieved? Environ. Sci. Technol. 2011, 45, 7100–7106. [Google Scholar] [CrossRef] [PubMed]
- Tabassum, N.; Islam, N.; Ahmed, S. Progress in microbial fuel cells for sustainable management of industrial effluents. Process Biochem. 2021, 106, 20–41. [Google Scholar] [CrossRef]
- Ahmed, S.F.; Mofijur, M.; Islam, N.; Parisa, T.A.; Rafa, N.; Bokhari, A.; Klemeš, J.J.; Mahlia, T.M.I. Insights into the development of microbial fuel cells for generating biohydrogen, bioelectricity, and treating wastewater. Energy 2022, 254, 124163. [Google Scholar] [CrossRef]
- Wang, H.; Ren, Z.J. Bioelectrochemical metal recovery from wastewater: A review. Water Res. 2014, 66, 219–232. [Google Scholar] [CrossRef]
- Ogugbue, C.J.; Ebode, E.E.; Leera, S. Electricity generation from swine wastewater using microbial fuel cell. J. Ecol Eng. 2015, 16, 26–33. [Google Scholar] [CrossRef]
- Wang, H.; Park, J.; Do, R.Z.J. Practical energy harvesting for microbial fuel cells: A review. Environ. Sci. Technol. 2015, 49, 3267–3277. [Google Scholar] [CrossRef]
- Wang, H.; Luo, H.; Fallgren, P.H.; Jin, S.; Ren, Z.J. Bioelectrochemical system platform for sustainable environmental remediation and energy generation. Biotechnol. Adv. 2015, 33, 317–334. [Google Scholar] [CrossRef]
- Ren, L.; Ahn, Y.; Logan, B.E. A two-stage microbial fuel cell and anaerobic fluidized bed membrane bioreactor (MFC-AFMBR) system for effective domestic wastewater treatment. Environ. Sci. Technol. 2014, 48, 4199–4206. [Google Scholar] [CrossRef] [PubMed]
- Fan, Y.; Hu, H.; Liu, H. Enhanced Coulombic efficiency and power density of air-cathode microbial fuel cells with an improved cell configuration. J. Power Sources 2007, 171, 348–354. [Google Scholar] [CrossRef]
- Sharif, H.M.A.; Farooq, M.; Hussain, I.; Ali, M.; Mujtaba, M.A.; Sultan, M.; Yang, B. Recent innovations for scaling up microbial fuel cell systems: Significance of physicochemical factors for electrodes and membranes materials. J. Taiwan Inst. Chem. Eng. 2021, 129, 207–226. [Google Scholar] [CrossRef]
- Zhuang, L.; Zheng, Y.; Zhou, S.; Yuan, Y.; Yuan, H.; Chen, Y. Scalable microbial fuel cell (MFC) stack for continuous real wastewater treatment. Bioresour. Technol. 2012, 106, 82–88. [Google Scholar] [CrossRef]
- Cheng, S.; Logan, B.E. Ammonia treatment of carbon cloth anodes to enhance power generation of microbial fuel cells. Electrochem. Commun. 2007, 9, 492–496. [Google Scholar] [CrossRef]
- Wang, C.-T.; Huang, R.-Y.; Lee, Y.-C.; Zhang, C.-D. Electrode Material of Carbon Nanotube/Polyaniline Carbon Paper Applied in Microbial Fuel Cells. J. Clean Energy Technol. 2013, 1, 206–210. [Google Scholar] [CrossRef]
- Zhou, M.; Chi, M.; Luo, J.; He, H.; Jin, T. An overview of electrode materials in microbial fuel cells. J. Power Sources 2011, 196, 4427–4435. [Google Scholar] [CrossRef]
- Pattanayak, P.; Pramanik, N.; Kumar, P.; Kundu, P.P. Fabrication of cost-effective non-noble metal supported on conducting polymer composite such as copper/polypyrrole graphene oxide (Cu2O/PPy–GO) as an anode catalyst for methanol oxidation in DMFC. Int. J. Hydrog. Energy 2018, 43, 11505–11519. [Google Scholar] [CrossRef]
- Zhang, F.; Brastad, K.S.; He, Z. Integrating forward osmosis into microbial fuel cells for wastewater treatment, water extraction and bioelectricity generation. Environ. Sci. Technol. 2011, 45, 6690–6696. [Google Scholar] [CrossRef]
- Cabrera, J.; Irfan, M.; Dai, Y.; Zhang, P.; Zong, Y.; Liu, X. Bioelectrochemical system as an innovative technology for treatment of produced water from oil and gas industry: A review. Chemosphere 2021, 285, 131428. [Google Scholar] [CrossRef]
- Kumar, G.; Bakonyi, P.; Zhen, G.; Sivagurunathan, P.; Koók, L.; Kim, S.H.; Tóth, G.; Nemestóthy, N.; Bélafi-Bakó, K. Microbial electrochemical systems for sustainable biohydrogen production: Surveying the experiences from a start-up viewpoint. Renew. Sustain. Energy Rev. 2017, 70, 589–597. [Google Scholar] [CrossRef]
- Mathuriya, A.S. Eco-affectionate face of microbial fuel cells. Crit. Rev. Environ. Sci. Technol. 2014, 44, 97–153. [Google Scholar] [CrossRef]
- Lai, M.F.; Lou, C.W.; Lin, J.H. Improve 3D electrode materials performance on electricity generation from livestock wastewater in microbial fuel cell. Int. J. Hydrog. Energy 2018, 43, 11520–11529. [Google Scholar] [CrossRef]
- Kalathil, S.; Lee, J.; Cho, M.H. Granular activated carbon based microbial fuel cell for simultaneous decolorization of real dye wastewater and electricity generation. N. Biotechnol. 2011, 29, 32–37. [Google Scholar] [CrossRef] [PubMed]
- Kim, B.H.; Chang, I.S.; Gadd, G.M. Challenges in microbial fuel cell development and operation. Appl. Microbiol. Biotechnol. 2007, 76, 485–494. [Google Scholar] [CrossRef]
- Chung, K.; Okabe, S. Continuous power generation and microbial community structure of the anode biofilms in a three-stage microbial fuel cell system. Appl. Microbiol. Biotechnol. 2009, 83, 965–977. [Google Scholar] [CrossRef]
- Scott, K.; Rimbu, G.A.; Katuri, K.P.; Prasad, K.K.; Head, I.M. Application of modified carbon anodes in microbial fuel cells. Process Saf. Environ. Prot. 2007, 85, 481–488. [Google Scholar] [CrossRef]
- Cheng, S.; Liu, H.; Logan, B.E. Increased power generation in a continuous flow MFC with advective flow through the porous anode and reduced electrode spacing. Environ. Sci. Technol. 2006, 40, 2426–2432. [Google Scholar] [CrossRef]
- Logan, B.; Cheng, S.; Watson, V.; Estadt, G. Graphite fiber brush anodes for increased power production in air-cathode microbial fuel cells. Environ. Sci. Technol. 2007, 41, 3341–3346. [Google Scholar] [CrossRef]
- Huggins, T.M.; Pietron, J.J.; Wang, H.; Ren, Z.J.; Biffinger, J.C. Graphitic biochar as a cathode electrocatalyst support for microbial fuel cells. Bioresour. Technol. 2015, 195, 147–153. [Google Scholar] [CrossRef]
- Ter Heijne, A.; Liu, F.; Van Rijnsoever, L.S.; Saakes, M.; Hamelers, H.V.M.; Buisman, C.J.N. Performance of a scaled-up Microbial Fuel Cell with iron reduction as the cathode reaction. J. Power Sources 2011, 196, 7572–7577. [Google Scholar] [CrossRef]
- HaoYu, E.; Cheng, S.; Scott, K.; Logan, B. Microbial fuel cell performance with non-Pt cathode catalysts. J. Power Sources 2007, 171, 275–281. [Google Scholar] [CrossRef]
- Salahuddin, M.; Uddin, M.N.; Hwang, G.; Asmatulu, R. Superhydrophobic PAN nanofibers for gas diffusion layers of proton exchange membrane fuel cells for cathodic water management. Int. J. Hydrog. Energy 2018, 43, 11530–11538. [Google Scholar] [CrossRef]
- Gu, H.Y.; Zhang, X.W.; Li, Z.J.; Lei, L.C. Studies on treatment of chlorophenol-containing wastewater by microbial fuel cell. Chin. Sci. Bull. 2007, 52, 3448–3451. [Google Scholar] [CrossRef]
- Xiao, B.; Yang, F.; Liu, J. Enhancing simultaneous electricity production and reduction of sewage sludge in two-chamber MFC by aerobic sludge digestion and sludge pretreatments. J. Hazard. Mater. 2011, 189, 444–449. [Google Scholar] [CrossRef] [PubMed]
- Jung, R.K.; Dec, J.; Bruns, M.A.; Logan, B.E. Removal of odors from swine wastewater by using microbial fuel cells. Appl. Environ. Microbiol. 2008, 74, 2540–2543. [Google Scholar]
- Anwar, A.; Siddique, M.; Eyup Dogan Sharif, A. The moderating role of renewable and non-renewable energy in environment-income nexus for ASEAN countries: Evidence from Method of Moments Quantile Regression. Renew. Energy 2021, 164, 956–967. [Google Scholar] [CrossRef]
- Muchtar, A.; Tambunan, A.H.; Machfud, M. Preliminary Analysis of Single-Flash Geothermal Power Plant by Using Exergy Method: A Case Study from Ulubelu Geothermal Power Plant in Indonesia Bioethanol from Brown Seaweed View project Production System Design of Banana based Energy Bar Industry View Project. 2018. Available online: https://www.researchgate.net/publication/335175433 (accessed on 21 January 2022).
- Huggins, T.; Wang, H.; Kearns, J.; Jenkins, P.; Ren, Z.J. Biochar as a sustainable electrode material for electricity production in microbial fuel cells. Bioresour. Technol. 2014, 157, 114–119. [Google Scholar] [CrossRef]
- Wang, B.; Han, J.I. A single chamber stackable microbial fuel cell with air cathode. Biotechnol. Lett. 2009, 31, 387–393. [Google Scholar] [CrossRef]
- Kaku, N.; Yonezawa, N.; Kodama, Y.; Watanabe, K. Plant/microbe cooperation for electricity generation in a rice paddy field. Appl. Microbiol. Biotechnol. 2008, 79, 43–49. [Google Scholar] [CrossRef]
- Feng, Y.; Wang, X.; Logan, B.E.; Lee, H. Brewery wastewater treatment using air-cathode microbial fuel cells. Appl. Microbiol. Biotechnol. 2008, 78, 873–880. [Google Scholar] [CrossRef] [PubMed]
- Zuo, Y.; Cheng, S.; Call, D.; Logan, B.E. Tubular membrane cathodes for scalable power generation in microbial fuel cells. Environ. Sci. Technol. 2007, 41, 3347–3353. [Google Scholar] [CrossRef] [PubMed]
- Logan, B.E.; Regan, J.M. Electricity-producing bacterial communities in microbial fuel cells. Trends Microbiol. 2006, 14, 512–518. [Google Scholar] [CrossRef] [PubMed]
- Rabaey, K.; Clauwaert, P.; Aelterman, P.; Verstraete, W. Tubular microbial fuel cells for efficient electricity generation. Environ. Sci. Technol. 2005, 39, 8077–8082. [Google Scholar] [CrossRef]
- Zhang, B.; Zhao, H.; Zhou, S.; Shi, C.; Wang, C.; Ni, J. A novel UASB-MFC-BAF integrated system for high strength molasses wastewater treatment and bioelectricity generation. Bioresour. Technol. 2009, 100, 5687–5693. [Google Scholar] [CrossRef]
- Sivasankaran, A.; Sangeetha, D.; Ahn, Y.H. Nanocomposite membranes based on sulfonated polystyrene ethylene butylene polystyrene (SSEBS) and sulfonated SiO2 for microbial fuel cell application. Chem. Eng. J. 2016, 289, 442–451. [Google Scholar] [CrossRef]
- Sil, A.; Ijeri, V.S.; Srivastava, A.K. Coated-wire iron(III) ion-selective electrode based on iron complex of 1,4,8,11-tetraazacyclotetradecane. Sens. Actuators B Chem. 2005, 106, 648–653. [Google Scholar]
- Rossi, R.; Wang, X.; Logan, B.E. High performance flow through microbial fuel cells with anion exchange membrane. J. Power Sources 2020, 475, 228633. [Google Scholar] [CrossRef]
- Nandy, A.; Kumar, V.; Kundu, P.P. Effect of electric impulse for improved energy generation in mediatorless dual chamber microbial fuel cell through electroevolution of Escherichia coli. Biosens. Bioelectron. 2016, 79, 796–801. [Google Scholar] [CrossRef]
- Pham, T.H.; Rabaey, K.; Aelterman, P.; Clauwaert, P.; De Schamphelaire, L.; Boon, N.; Verstraete, W. Microbial fuel cells in relation to conventional anaerobic digestion technology. Eng. Life Sci. 2006, 6, 285–292. [Google Scholar] [CrossRef]
- Suzuki, K.; Tanaka, Y.; Osada, T.; Waki, M. Removal of phosphate, magnesium and calcium from swine wastewater through crystallization enhanced by aeration. Water Res. 2002, 36, 2991–2998. [Google Scholar] [CrossRef]
- Bakare, B.F.; Shabangu, K.; Chetty, M. Brewery wastewater treatment using laboratory scale aerobic sequencing batch reactor. S. Afr. J Chem Eng. 2017, 24, 128–134. [Google Scholar] [CrossRef]
- Dosta, J.; Rovira, J.; Galí, A.; Macé, S.; Mata-Álvarez, J. Integration of a Coagulation/Flocculation step in a biological sequencing batch reactor for COD and nitrogen removal of supernatant of anaerobically digested piggery wastewater. Bioresour. Technol. 2008, 99, 5722–5730. [Google Scholar] [CrossRef] [PubMed]
- Ringsmuth, A.K.; Landsberg, M.J.; Hankamer, B. Can photosynthesis enable a global transition from fossil fuels to solar fuels, to mitigate climate change and fuel-supply limitations? Renew. Sustain. Energy Rev. 2016, 62, 134–163. [Google Scholar] [CrossRef]
- Cheng, S.; Liu, H.; Logan, B.E. Increased performance of single-chamber microbial fuel cells using an improved cathode structure. Electrochem. Commun. 2006, 8, 489–494. [Google Scholar] [CrossRef]
- Logan, B.E.; Rabaey, K. Conversion of wastes into bioelectricity and chemicals by using microbial electrochemical technologies. Science 2012, 337, 686–690. [Google Scholar] [CrossRef]
- Allami, S.; Hasan, B.; Redah, M.; Hamody, H.; Abd Ali, Z.D. Using Low Cost Membrane in Dual-Chamber Microbial Fuel Cells (MFCs) for Petroleum Refinery Wastewater Treatment. J. Phys. Conf. Ser. 2018, 1032, 012061. [Google Scholar] [CrossRef]
- Jafary, T.; Ghoreyshi, A.A.; Najafpour, G.D.; Fatemi, S.; Rahimnejad, M. Investigation on performance of microbial fuel cells based on carbon sources and kinetic models. Int. J. Energy Res. 2013, 37, 1539–1549. [Google Scholar] [CrossRef]
- Tessema, T.D.; Yemata, T.A. Experimental dataset on the effect of electron acceptors in energy generation from brewery wastewater via a microbial fuel cell. Data Brief 2021, 37, 107272. [Google Scholar] [CrossRef]
- Rozendal, R.A.; Hamelers, H.V.M.; Rabaey, K.; Keller, J.; Buisman, C.J.N. Towards practical implementation of bioelectrochemical wastewater treatment. Trends Biotechnol. 2008, 26, 450–459. [Google Scholar] [CrossRef]
- Lu, N.; Zhou, S.G.; Zhuang, L.; Zhang, J.T.; Ni, J.R. Electricity generation from starch processing wastewater using microbial fuel cell technology. Biochem. Eng. J. 2009, 43, 246–251. [Google Scholar] [CrossRef]
- Puig, S.; Serra, M.; Coma, M.; Balaguer, M.D.; Colprim, J. Simultaneous domestic wastewater treatment and renewable energy production using microbial fuel cells (MFCs). Water Sci. Technol. 2011, 64, 904–909. [Google Scholar] [CrossRef] [PubMed]
- Min, B.; Logan, B.E. Continuous Electricity Generation from Domestic Wastewater and Organic Substrates in a Flat Plate Microbial Fuel Cell. Environ. Sci. Technol. 2004, 38, 5809–5814. [Google Scholar] [CrossRef]
- Dong, J.; Wu, Y.; Wang, C.; Lu, H.; Li, Y. Three-dimensional electrodes enhance electricity generation and nitrogen removal of microbial fuel cells. Bioprocess Biosyst. Eng. 2020, 43, 2165–2174. [Google Scholar] [CrossRef] [PubMed]
- Butti, S.K.; Velvizhi, G.; Sulonen, M.L.K.; Haavisto, J.M.; Oguz Koroglu, E.; Yusuf Cetinkaya, A.; Singh, S.; Arya, D.; Modestra, J.A.; Krishna, K.V.; et al. Microbial electrochemical technologies with the perspective of harnessing bioenergy: Maneuvering towards upscaling. Renew. Sustain. Energy Rev. 2016, 53, 462–476. [Google Scholar] [CrossRef]
- Venkatramanan, V.; Shah, S.; Prasad, R. A Critical Review on Microbial Fuel Cells Technology: Perspectives on Wastewater Treatment. Open Biotechnol. J. 2021, 15, 131–141. [Google Scholar] [CrossRef]
- Gul, H.; Raza, W.; Lee, J.; Azam, M.; Ashraf, M.; Kim, K.H. Progress in microbial fuel cell technology for wastewater treatment and energy harvesting. Chemosphere 2021, 281, 130828. [Google Scholar] [CrossRef]
- Wang, J.; Li, Q.; Qi, R.; Tandoi, V.; Yang, M. Sludge Bulking Impact on Relevant Bacterial Populations in a Full-Scale Municipal Wastewater Treatment Plant. Process Biochem. 2014, 49, 2258–2265. [Google Scholar] [CrossRef]
- Filamentous and Non-Filamentous Bulking of Activated Sludge Encountered Under Nutrients Limitation or Deficiency Conditions|Elsevier Enhanced Reader. Available online: https://reader.elsevier.com/reader/sd/pii/S1385894714008067?token=4D0DE471E017CBA9AFFE44200063C1294125B65E92E7653D829FB6F2D253FF651E32660F281D947ED9A6B45D6ED437F0&originRegion=eu-west-1&originCreation=20220121175337 (accessed on 21 January 2022).
- Sato, C.; Paucar, N.E.; Chiu, S.; Mahmud, M.Z.I.M.; Dudgeon, J. Single-Chamber Microbial Fuel Cell with Multiple Plates of Bamboo Charcoal Anode: Performance Evaluation. Processes 2021, 9, 2194. [Google Scholar] [CrossRef]
- Yang, Z.; Tsapekos, P.; Zhang, Y.; Zhang, Y.; Angelidaki, I.; Wang, W. Bio-electrochemically extracted nitrogen from residual resources for microbial protein production. Bioresour. Technol. 2021, 337, 125353. [Google Scholar] [CrossRef]
- Complete Nitrogen Removal by Simultaneous Nitrification and Denitrification in Flat-Panel Air-Cathode Microbial Fuel Cells Treating Domestic Wastewater|Elsevier Enhanced Reader. Available online: https://reader.elsevier.com/reader/sd/pii/S1385894717301626?token=5F85DD284D224B87D998AB8F229CC975D821C421C01C566B48F471370C637BA9844E70E2926BD9580FD01518DD7E4350&originRegion=eu-west-1&originCreation=20220121162726 (accessed on 21 January 2022).
- Negassa, L.W.; Mohiuddin, M.; Tiruye, G.A. Treatment of brewery industrial wastewater and generation of sustainable bioelectricity by microbial fuel cell inoculated with locally isolated microorganisms. J. Water Process. Eng. 2021, 41, 102018. [Google Scholar] [CrossRef]
- Zhao, F.; Slade, R.C.T.; Varcoe, J.R. Techniques for The Study and Development of Microbial Fuel Cells: An Electrochemical Perspective. Chem. Soc. Rev. 2009, 38, 1926–1939. [Google Scholar] [CrossRef] [PubMed]
- Vinayak, V.; Khan, M.J.; Varjani, S.; Saratale, G.D.; Saratale, R.G.; Bhatia, S.K. Microbial fuel cells for remediation of environmental pollutants and value addition: Special focus on coupling diatom microbial fuel cells with photocatalytic and photoelectric fuel cells. J. Biotechnol. 2021, 338, 5–19. [Google Scholar] [CrossRef] [PubMed]
- Apollon, W.; Luna-Maldonado, A.I.; Kamaraj, S.K.; Vidales-Contreras, J.A.; Rodríguez-Fuentes, H.; Gómez-Leyva, J.F.; Aranda-Ruíz, J. Progress and recent trends in photosynthetic assisted microbial fuel cells: A review. Biomass Bioenergy 2021, 148, 106028. [Google Scholar] [CrossRef]
- Munoz-Cupa, C.; Hu, Y.; Xu, C.; Bassi, A. An overview of microbial fuel cell usage in wastewater treatment, resource recovery and energy production. Sci. Total Environ. 2021, 754, 142429. [Google Scholar] [CrossRef]
- Bensaida, K.; Maamoun, I.; Eljamal, R.; Falyouna, O.; Sugihara, Y.; Eljamal, O. New insight for electricity amplification in microbial fuel cells (MFCs) applying magnesium hydroxide coated iron nanoparticles. Energy Convers. Manag. 2021, 249, 114877. [Google Scholar] [CrossRef]
- Sumisha, A.; Haribabu, K. Energy Generation and Iron Removal in Batch and Continuous Single-Chamber Microbial Fuel Cells. Chem. Eng. Technol. 2021, 44, 258–264. [Google Scholar] [CrossRef]
- Jung, R.K.; Zuo, Y.; Regan, J.M.; Logan, B.E. Analysis of ammonia loss mechanisms in microbial fuel cells treating animal wastewater. Biotechnol. Bioeng. 2008, 99, 1120–1127. [Google Scholar]
- Ye, Y.; Ngo, H.H.; Guo, W.; Chang, S.W.; Nguyen, D.D.; Varjani, S.; Liu, Q.; Bui, X.T.; Hoang, N.B. Bio-membrane integrated systems for nitrogen recovery from wastewater in circular bioeconomy. Chemosphere 2022, 289, 133175. [Google Scholar] [CrossRef]
- Kong, W.; Guo, Q.; Wang, X.; Yue, X. Electricity generation from wastewater using an anaerobic fluidized bed microbial fuel cell. Ind. Eng. Chem. Res. 2011, 50, 12225–12232. [Google Scholar] [CrossRef]
- Khandaker, S.; Das, S.; Hossain, M.T.; Islam, A.; Miah, M.R.; Awual, M.R. Sustainable approach for wastewater treatment using microbial fuel cells and green energy generation–A comprehensive review. J. Mol. Liq. 2021, 344, 117795. [Google Scholar] [CrossRef]
- An, J.; Kim, D.; Chun, Y.; Lee, S.J.; Ng, H.Y.; Chang, I.S. Floating-type microbial fuel cell (FT-MFC) for treating organic-contaminated water. Environ. Sci. Technol. 2009, 43, 1642–1647. [Google Scholar] [CrossRef] [PubMed]
- Santoro, C.; Ieropoulos, I.; Greenman, J.; Cristiani, P.; Vadas, T.; Mackay, A.; Li, B. Power generation and contaminant removal in single chamber microbial fuel cells (SCMFCs) treating human urine. Int. J. Hydrog. Energy 2013, 38, 11543–11551. [Google Scholar] [CrossRef]
- Wang, X.; Feng, Y.J.; Lee, H. Electricity production from beer brewery wastewater using single chamber microbial fuel cell. Water Sci. Technol. 2008, 57, 1117–1121. [Google Scholar] [CrossRef] [PubMed]
- Ting, M.B.; Byrne, R. Eskom and the rise of renewables: Regime-resistance, crisis and the strategy of incumbency in South Africa’s electricity system. Energy Res. Soc. Sci. 2020, 60, 101333. [Google Scholar] [CrossRef]
- Mbuli, N.; Mendu, B.; Seitshiro, M.; Pretorius, J.H.C. Statistical analysis of forced outage durations of Eskom subtransmission transformers. Energy Rep. 2020, 6, 321–329. [Google Scholar] [CrossRef]
- Valet, R. Factors Influencing the Societal Acceptance of Renewable Energies Related Papers Background Paper on Technology Roadmaps for UNFCCC. Available online: www.createacceptance.net (accessed on 25 August 2022).
- Khimulu, R.M. Assesment of The Nexus Of Energy, Climate Change, Urbanization And Industrialization For Africa’s Transformation. 2017. Available online: http://repository.pauwes-cop.net/handle/1/133 (accessed on 25 August 2022).
- Eskom: Electricity and Technopolitics in South Africa-Sylvy Jaglin, Alain Dubresson-Google Books. Available online: https://books.google.co.za/books?hl=en&lr=&id=bfZyDwAAQBAJ&oi=fnd&pg=PP5&dq=Eskom+captured&ots=VicQqqSAQu&sig=EJHDUCUWSNUY-oohlOo307yDOK8&redir_esc=y#v=onepage&q=Eskomcaptured&f=false (accessed on 25 August 2022).
- South Africa Comes to Standstill with Eskom’s Load Shedding-ProQuest. Available online: https://www.proquest.com/openview/88be753c6894745beeaa1cea8e53d700/1?pq-origsite=gscholar&cbl=39 (accessed on 25 August 2022).
- Du Venage, G. South Africa Comes to Standstill with Eskom’s Load Shedding. 2020. Available online: www.e-mj.com (accessed on 25 August 2022).
- Inglesi, R. Aggregate electricity demand in South Africa: Conditional forecasts to 2030. Appl. Energy 2010, 87, 197–204. [Google Scholar] [CrossRef]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).