Changes in Soil Chemistry and Soil Nutrient Stocks after 30 Years of Treated Municipal Wastewater Land Disposal: A Natural Experiment
Abstract
:1. Introduction
2. Materials and Methods
2.1. Study Site and Soil Collection

2.2. Chemical Analysis
2.3. Data Analysis
| Parameter 1 | # Analysis (N =) | Mean ± SE 2 | Median | Range | DW 3 Standards | WW 4 Standards | Application 5 kg ha−1 y−1 |
|---|---|---|---|---|---|---|---|
| TSS | 2 | 3.3, 3.8 | 10–150 | ||||
| EC (mS m−1) | 121 | 68 ± 1.9 | 66 | (29–167) | 100–700 | ||
| TN | 2 | 46, 47 | 15–70 | 1860 | |||
| NO3−-N | 123 | 12 ± 1.2 | 3.9 | (<0.005–61.8) | 11.3 | 10–50 | 480 |
| NH4+-N | 123 | 6.2 ± 0.99 | 0.32 | (<0.005–37) | 248 | ||
| Inorganic N | 125 | 18 ± 1.2 | 20 | (<0.005–73) | 720 | ||
| TP | 2 | 6.1, 7.1 | 264 | ||||
| DR-P 6 | 123 | 1.3 ± 0.2 | 0.1 | (<0.005–10) | 2–30 | 52 | |
| Ca | 2 | 11, 12 | 460 | ||||
| Mg | 2 | 3.1, 3.3 | 128 | ||||
| K | 2 | 23, 24 | 940 | ||||
| Na | 2 | 63, 65 | 2560 | ||||
| Zn (μg L−1) | 45 | 5.8 ± 1.8 | 1.0 | (1–54) | 231 | ||
| Cu (μg L−1) | 45 | 6.0 ± 1.8 | 2.0 | (<1–59) | 2000 | 0.23 | |
| As (μg L−1) | 45 | 1.4 ± 0.3 | <1 | (<1–10) | 10 | 0.06 | |
| B (μg L−1) | 43 | 163 ± 12 | 160 | (<5–614) | 2400 | 6.5 | |
| Cd (μg L−1) | 45 | 0.1 ± 0 | <0.06 | (<0.06–2) | 4 | 4 (g ha−1 y−1) | |
| Cr (μg L−1) | 45 | 1.4 ± 0.3 | 0.9 | (<0.15–12) | 50 | 0.06 | |
| Hg (μg L−1) | 45 | <0.5 | <0.5 | <0.5 | 7 | <20 (g ha−1 y−1) | |
| Ni (μg L−1) | 45 | <8 | <8 | <8 | 80 | <0.32 | |
| Pb (μg L−1) | 125 | 1 | <1 | (<1–2) | 10 | 0.04 | |
| SAR 7 | 1 | 4.4 | 8–10 |
3. Results
3.1. Changes in Soil Chemical Properties
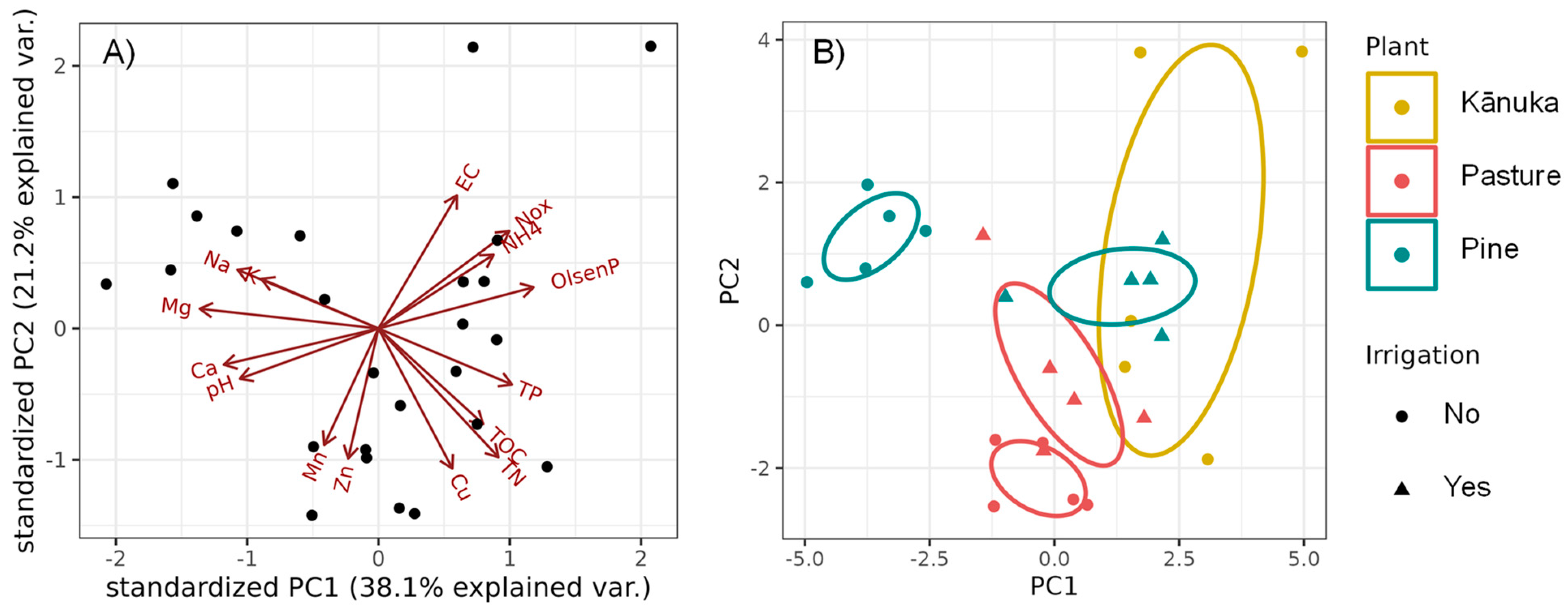
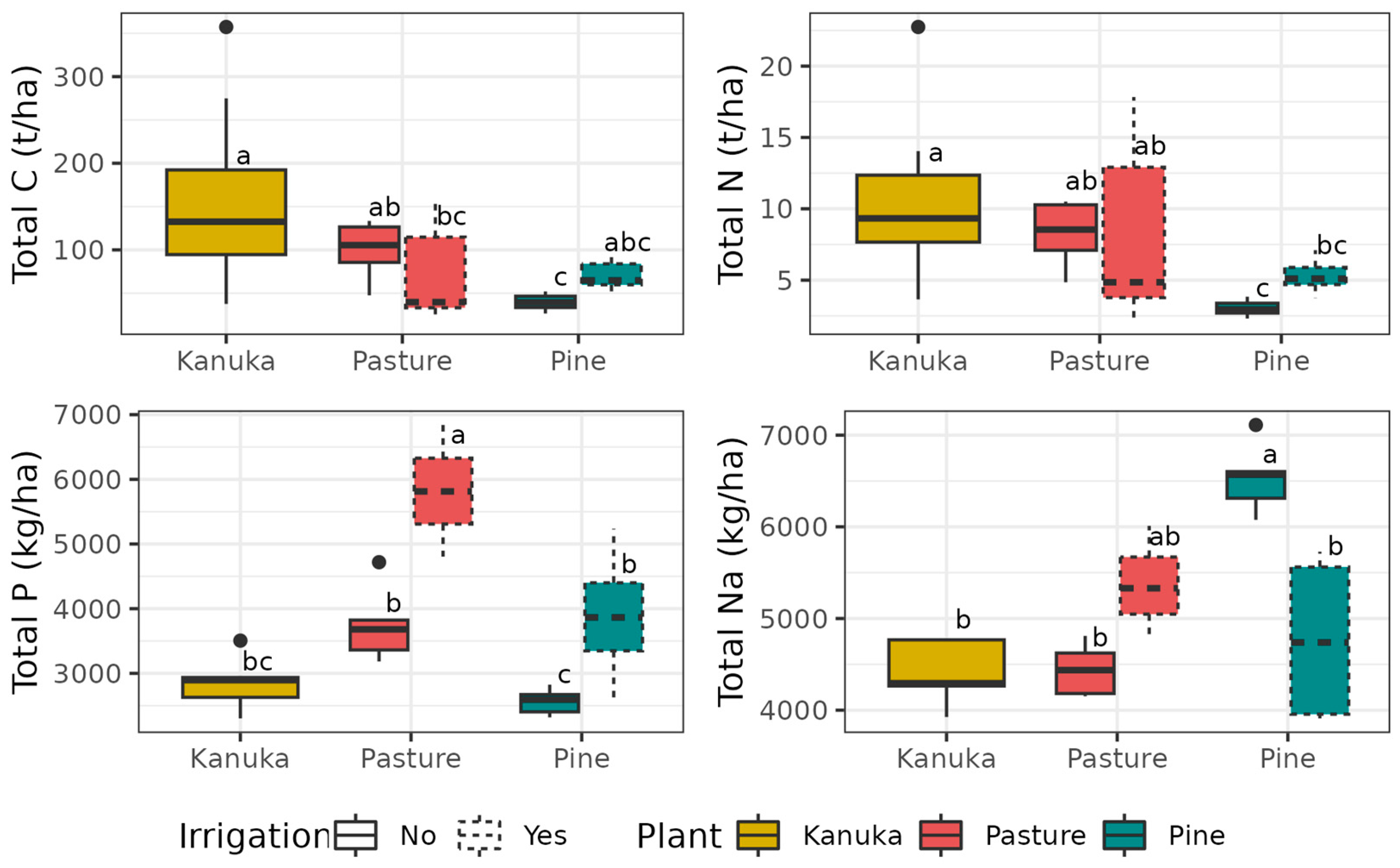
3.2. Changes in Soil Stocks
4. Discussion
4.1. Changes in Soil Chemical Properties
4.2. Exports of Contaminants and Soil Nutrient Stocks
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- WWAP (United Nations World Water Assessment Programme). The United Nations World Water Development Report 2017. Wastewater: The Untapped Resource; UNESCO: Paris, France, 2017. [Google Scholar]
- Krzmarzick, M.J.; Novak, P.J. Removal of chlorinated organic compounds during wastewater treatment: Achievements and limits. Appl. Microbiol. Biotechnol. 2014, 98, 6233–6242. [Google Scholar] [CrossRef]
- Preisner, M.; Neverova-Dziopak, E.; Kowalewski, Z. An Analytical Review of Different Approaches to Wastewater Discharge Standards with Particular Emphasis on Nutrients. Environ. Manag. 2020, 66, 694–708. [Google Scholar] [CrossRef] [PubMed]
- Rout, P.R.; Shahid, M.K.; Dash, R.R.; Bhunia, P.; Liu, D.; Varjani, S.; Zhang, T.C.; Surampalli, R.Y. Nutrient removal from domestic wastewater: A comprehensive review on conventional and advanced technologies. J. Environ. Manag. 2021, 296, 113246. [Google Scholar] [CrossRef] [PubMed]
- Pedrero, F.; Kalavrouziotis, I.; Alarcón, J.J.; Koukoulakis, P.; Asano, T. Use of treated municipal wastewater in irrigated agriculture—Review of some practices in Spain and Greece. Agric. Water Manag. 2010, 97, 1233–1241. [Google Scholar] [CrossRef]
- Becerra-Castro, C.; Lopes, A.R.; Vaz-Moreira, I.; Silva, E.F.; Manaia, C.M.; Nunes, O.C. Wastewater reuse in irrigation: A microbiological perspective on implications in soil fertility and human and environmental health. Environ. Int. 2015, 75, 117–135. [Google Scholar] [CrossRef]
- Overcash, M.; Sims, R.C.; Sims, J.L.; Nieman, J.K.C. Beneficial Reuse and Sustainability: The Fate of Organic Compounds in Land-Applied Waste. J. Environ. Qual. 2005, 34, 29–41. [Google Scholar] [CrossRef]
- Adrover, M.; Farrús, E.; Moyà, G.; Vadell, J. Chemical properties and biological activity in soils of Mallorca following twenty years of treated wastewater irrigation. J. Environ. Manag. 2012, 95, S188–S192. [Google Scholar] [CrossRef]
- Alguacil, M.d.M.; Torrecillas, E.; Torres, P.; García-Orenes, F.; Roldán, A. Long-Term Effects of Irrigation with Waste Water on Soil AM Fungi Diversity and Microbial Activities: The Implications for Agro-Ecosystem Resilience. PLoS ONE 2012, 7, e47680. [Google Scholar] [CrossRef]
- Bedbabis, S.; Trigui, D.; Ben Ahmed, C.; Clodoveo, M.L.; Camposeo, S.; Vivaldi, G.A.; Ben Rouina, B. Long-terms effects of irrigation with treated municipal wastewater on soil, yield and olive oil quality. Agric. Water Manag. 2015, 160, 14–21. [Google Scholar] [CrossRef]
- Dimitriou, I.; Aronsson, P. Wastewater and sewage sludge application to willows and poplars grown in lysimeters–Plant response and treatment efficiency. Biomass Bioenergy 2011, 35, 161–170. [Google Scholar] [CrossRef]
- Magesan, G.N.; McLay, C.D.A.; Lal, V.V. Nitrate leaching from a free-draining volcanic soil irrigated with municipal sewage effluent in New Zealand. Agric. Ecosyst. Environ. 1998, 70, 181–187. [Google Scholar] [CrossRef]
- Mohammad Rusan, M.J.; Hinnawi, S.; Rousan, L. Long term effect of wastewater irrigation of forage crops on soil and plant quality parameters. Desalination 2007, 215, 143–152. [Google Scholar] [CrossRef]
- Qian, Y.L.; Mecham, B. Long-Term Effects of Recycled Wastewater Irrigation on Soil Chemical Properties on Golf Course Fairways. Agron. J. 2005, 97, 717–721. [Google Scholar] [CrossRef]
- Segal, E.; Dag, A.; Ben-Gal, A.; Zipori, I.; Erel, R.; Suryano, S.; Yermiyahu, U. Olive orchard irrigation with reclaimed wastewater: Agronomic and environmental considerations. Agric. Ecosyst. Environ. 2011, 140, 454–461. [Google Scholar] [CrossRef]
- Zavadil, J. The effect of municipal wastewater irrigation on the yield and quality of vegetables and crops. Soil Water Res. 2009, 4, 91–103. [Google Scholar] [CrossRef]
- Asgari, K.; Cornelis, W.M. Heavy metal accumulation in soils and grains, and health risks associated with use of treated municipal wastewater in subsurface drip irrigation. Environ. Monit. Assess. 2015, 187, 410. [Google Scholar] [CrossRef]
- Angin, I.; Yaganoglu, A.V.; Turan, M. Effects of Long-Term Wastewater Irrigation on Soil Properties. J. Sustain. Agric. 2005, 26, 31–42. [Google Scholar] [CrossRef]
- Assouline, S.; Narkis, K. Effects of long-term irrigation with treated wastewater on the hydraulic properties of a clayey soil. Water Resour. Res. 2011, 47, W08530. [Google Scholar] [CrossRef]
- Bedbabis, S.; Ben Rouina, B.; Boukhris, M.; Ferrara, G. Effect of irrigation with treated wastewater on soil chemical properties and infiltration rate. J. Environ. Manag. 2014, 133, 45–50. [Google Scholar] [CrossRef]
- Lal, K.; Minhas, P.S.; Yadav, R.K. Long-term impact of wastewater irrigation and nutrient rates II. Nutrient balance, nitrate leaching and soil properties under peri-urban cropping systems. Agric. Water Manag. 2015, 156, 110–117. [Google Scholar] [CrossRef]
- Abrol, I.P.; Yadav, J.S.P.; Massoud, F.I. Salt-Affected Soils and Their Management; FAO Soils Bulletin; Food and Agriculture Organization of the United Nations, Ed.; FAO: Rome, Italy, 1988; Volume 39. [Google Scholar]
- Esperschuetz, J.; Anderson, C.; Bulman, S.; Katamian, O.; Horswell, J.; Dickinson, N.; Robinson, B. Response of Leptospermum scoparium, Kunzea robusta and Pinus radiata to contrasting biowastes. Sci. Total Environ. 2017, 587–588, 258–265. [Google Scholar] [CrossRef] [PubMed]
- Halford, S.; Gutiérrez-Ginés, M.J.; Rees, A.; Alderton, I.; Schaller, K.; Smith, R.; Horswell, J. Effect of mānuka (Leptospermum scoparium) on nitrogen and Escherichia coli reductions in soils: A field experiment. Plant Soil 2021, 465, 491–502. [Google Scholar] [CrossRef]
- Meister, A.; Li, F.; Gutierrez-Gines, M.J.; Dickinson, N.; Gaw, S.; Bourke, M.; Robinson, B. Interactions of treated municipal wastewater with native plant species. Ecol. Eng. 2022, 183, 106741. [Google Scholar] [CrossRef]
- Davies, G.M.; Gray, A. Don’t let spurious accusations of pseudoreplication limit our ability to learn from natural experiments (and other messy kinds of ecological monitoring). Ecol. Evol. 2015, 5, 5295–5304. [Google Scholar] [CrossRef]
- Landcare Research. New Zealand Soil Classifiation. Available online: https://soils-maps.landcareresearch.co.nz/#maps (accessed on 1 September 2023).
- Blakemore, L.C.; Searle, P.L.; Daly, B.K. Methods for chemical analysis of soils. In NZ Soil Bureau Scientific Report 80; Dept. Scientific and Industrial Research: Lower Hutt, New Zealand, 1987. [Google Scholar]
- Saunders, W.M.H. Phosphate retention by New Zealand soils and its relationship to free sesquioxides, organic matter, and other soil properties. N. Z. J. Agric. Res. 1965, 8, 30–57. [Google Scholar] [CrossRef]
- New Zealand Government. Water Services (Drinking Water Standards for New Zealand) Regulations 2022; New Zealand Government: Wellington, New Zealand, 2022.
- Ayers, R.S.; Westcot, D.W. Water Quality for Agriculture; FAO Irrigation and Drainage Paper; Food and Agriculture Organization of the United Nations: Rome, Italy, 1985; Volume 29. [Google Scholar]
- R Core Team. R: A Language and Environment for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria, 2021. [Google Scholar]
- Mendiburu, F.d.; Yaseen, M.; Agricolae: Statistical Procedures for Agricultural Research. R Package Version 1.4.0. 2020. Available online: https://cran.r-project.org/web/packages/agricolae/index.html (accessed on 1 September 2023).
- Wickham, H. ggplot2: Elegant Graphics for Data Analysis; Springer: New York, NY, USA, 2016. [Google Scholar]
- Meyer, F.; Perrier, V.; Carroll, I.; Dang, X.; Bevacqua, N.; Choi, D.Y.; Rocha, Z. Package ‘Esquisse’ Explore and Visualize Your Data Interactively. R Package Version 1.1.0. 2022. Available online: https://github.com/dreamRs/esquisse/ (accessed on 1 September 2023).
- Garnier, S.; Ross, N.; Rudis, B.; Sciaini, M.; Camargo, A.P.; Scherer, C. Colorblind-Friendly Color Maps for R. 2023. Available online: https://sjmgarnier.github.io/viridis/ (accessed on 1 September 2023).
- Lizarralde, C.A.; McDowell, R.; Condron, L.; Brown, J. The potential for phosphorus loss to groundwater from soils irrigated with dairy factory wastewater. N. Z. J. Agric. Res. 2023, 66, 189–207. [Google Scholar] [CrossRef]
- Sparling, G.; Lilburne, L.; Vojvodić-Vuković, M. Provisional Targets for Soil Quality Indicators in New Zealand; Landcare Research Science Series No. 34; Manaaki Whenua Press: Lincoln, New Zealand, 2008. [Google Scholar]
- Gutierrez-Gines, M.J.; Mishra, M.; McIntyre, C.; Chau, H.W.; Esperschuetz, J.; McLenaghen, R.; Bourke, M.P.; Robinson, B.H. Risks and benefits of pasture irrigation using treated municipal effluent : A lysimeter case study, Canterbury, New Zealand. Environ. Sci. Pollut. Res. 2020, 27, 11830–11841. [Google Scholar] [CrossRef]
- Hu, C.; Zhang, T.C.; Huang, Y.H.; Dahab, M.F.; Surampalli, R. Effects of Long-Term Wastewater Application on Chemical Properties and Phosphorus Adsorption Capacity in Soils of a Wastewater Land Treatment System. Environ. Sci. Technol. 2005, 39, 7240–7245. [Google Scholar] [CrossRef]
- Xu, J.; Wu, L.; Chang, A.C.; Zhang, Y. Impact of long-term reclaimed wastewater irrigation on agricultural soils: A preliminary assessment. J. Hazard. Mater. 2010, 183, 780–786. [Google Scholar] [CrossRef]
- Liu, Y.Y.; Haynes, R.J. Influence of land application of dairy factory effluent on soil nutrient status and the size, activity, composition and catabolic capability of the soil microbial community. Appl. Soil Ecol. 2011, 48, 133–141. [Google Scholar] [CrossRef]
- Ndour, N.Y.B.; Baudoin, E.; Guissé, A.; Seck, M.; Khouma, M.; Brauman, A. Impact of irrigation water quality on soil nitrifying and total bacterial communities. Biol. Fertil. Soils 2008, 44, 797–803. [Google Scholar] [CrossRef]
- Lal, K.; Minhas, P.S.; Yadav, R.K.; Rosin, K.G. GHG mitigation from sewage effluents: C sequestration after reuse in different urban cropping systems. Carbon Manag. 2020, 11, 281–290. [Google Scholar] [CrossRef]
- Somenahally, A.C.; McLawrence, J.; Chaganti, V.N.; Ganjegunte, G.K.; Obayomi, O.; Brady, J.A. Response of soil microbial Communities, inorganic and organic soil carbon pools in arid saline soils to alternative land use practices. Ecol. Indic. 2023, 150, 110227. [Google Scholar] [CrossRef]
- Cameron, K.C.; Di, H.J.; Moir, J.L. Nitrogen losses from the soil/plant system: A review. Ann. Appl. Biol. 2013, 162, 145–173. [Google Scholar] [CrossRef]
- Gelsomino, A.; Badalucco, L.; Ambrosoli, R.; Crecchio, C.; Puglisi, E.; Meli, S.M. Changes in chemical and biological soil properties as induced by anthropogenic disturbance: A case study of an agricultural soil under recurrent flooding by wastewaters. Soil Biol. Biochem. 2006, 38, 2069–2080. [Google Scholar] [CrossRef]
- de Lange, P.J. A revision of the New Zealand Kunzeaericoides (Myrtaceae) complex. PhytoKeys 2014, 40, 1–185. [Google Scholar] [CrossRef] [PubMed]
- MWLR. S-Map Online. 2023. Available online: https://smap.landcareresearch.co.nz/ (accessed on 1 September 2023).
- Baldi, M.; Burgess, S.; Mullan, B.; Salinger, J.; Ramsay, D.; Bell, R. Updated Climate Change Scenarios for the Kapiti Coast; NIWA: Wellington, New Zealand, 2007; p. 56. [Google Scholar]
- Rowe, L.; Jackson, R.B.; Fahey, B. Land Use and Water Resources: Hydrological Effects of Different Vegetation Covers; SMF2167: Report No 5.; Landcare Research: Lincoln, New Zealand, 2002; p. 142. [Google Scholar]
- Gutierrez-Gines, M.J.; Bishit, A.; Meister, A.; Robinson, B.H.; Clarke, D.; Tupuhi, G.; Alderton, I.; Horswell, J.; Wang, K.I.K.; Bohm, K.; et al. Maanuka Dominated Ecosystems to Improve Water and Soil Quality in Lake Waikare Catchment; ESR: Christchurch, New Zealand, 2022; Available online: https://waikatoriver.org.nz/wp-content/uploads/2022/06/WRA16-008-Final-Report-WRA-2022_10.pdf (accessed on 1 September 2023).
- Hart, M.R.; Quin, B.F.; Nguyen, M.L. Phosphorus Runoff from Agricultural Land and Direct Fertilizer Effects: A Review. J. Environ. Qual. 2004, 33, 1954–1972. [Google Scholar] [CrossRef] [PubMed]
- Gwenzi, W.; Munondo, R. Long-term impacts of pasture irrigation with treated sewage effluent on shallow groundwater quality. Water Sci. Technol. 2008, 58, 2443–2452. [Google Scholar] [CrossRef] [PubMed]
- Kimberley, M.O.; Bergin, D.; Beets, P. Carbon Sequestration by Planted Native Trees and Shrubs. In Technical Handbook Section 10: Native Tree Plantations; Tane’s Tree Trust: Hamilton, New Zealand, 2014; Volume 10, pp. 1–12. [Google Scholar]
- Piaszczyk, W.; Błońska, E.; Lasota, J.; Lukac, M. A comparison of C:N:P stoichiometry in soil and deadwood at an advanced decomposition stage. CATENA 2019, 179, 1–5. [Google Scholar] [CrossRef]
- Tang, Z.; Xu, W.; Zhou, G.; Bai, Y.; Li, J.; Tang, X.; Chen, D.; Liu, Q.; Ma, W.; Xiong, G.; et al. Patterns of plant carbon, nitrogen, and phosphorus concentration in relation to productivity in China’s terrestrial ecosystems. Proc. Natl. Acad. Sci. USA 2018, 115, 4033–4038. [Google Scholar] [CrossRef]
- Will, G.M. Increased Phosphorus Uptake by Radiata Pine in Riverhead Forest following Superphosphate Applications. N. Z. J. For. 1965, 10, 33–42. [Google Scholar]
- Pan, B.; Xia, L.; Lam, S.K.; Wang, E.; Zhang, Y.; Mosier, A.; Chen, D. A global synthesis of soil denitrification: Driving factors and mitigation strategies. Agric. Ecosyst. Environ. 2022, 327, 107850. [Google Scholar] [CrossRef]
- Scheer, C.; Fuchs, K.; Pelster, D.E.; Butterbach-Bahl, K. Estimating global terrestrial denitrification from measured N2O:(N2O + N2) product ratios. Curr. Opin. Environ. Sustain. 2020, 47, 72–80. [Google Scholar] [CrossRef]
- Sánchez–González, A.; Chapela–Lara, M.; Germán–Venegas, E.; Fuentes-García, R.; Río-Portilla, F.d.; Siebe, C. Changes in quality and quantity of soil organic matter stocks resulting from wastewater irrigation in formerly forested land. Geoderma 2017, 306, 99–107. [Google Scholar] [CrossRef]
- Oliver, G.R.; Beets, P.N.; Garrett, L.G.; Pearce, S.H.; Kimberly, M.O.; Ford-Robertson, J.B.; Robertson, K.A. Variation in soil carbon in pine plantations and implications for monitoring soil carbon stocks in relation to land-use change and forest site management in New Zealand. For. Ecol. Manag. 2004, 203, 283–295. [Google Scholar] [CrossRef]

| Parameter and Transformations | Veg. | Irrig. | Depth | Veg × Irrig | Veg × Depth | Irrig × Depth | V × I × D |
|---|---|---|---|---|---|---|---|
| TP | *** | *** | *** | * | ° | ||
| Log10(OlsenP + 1) | *** | *** | *** | ° | ** | ||
| Log10(P_ret) | *** | *** | |||||
| pH | *** | *** | *** | *** | |||
| Log10(NO3 + 1) | *** | *** | *** | *** | ° | * | |
| Log10(Na) | ** | *** | *** | *** | * | ** | |
| Log10(EC) | *** | *** | *** | ** | |||
| Log10(TOC) | *** | *** | *** | ||||
| Log10(TN) | *** | *** | *** | ||||
| NH4- | *** | * | *** | ° | |||
| Mg | *** | ** | ** | ° | * | ||
| Ca | ° | *** | * | * | |||
| K | * | *** | |||||
| Log10(Cu) | *** | * | * | ° | |||
| Mn | *** | * | *** | ° | |||
| Log10(Zn) | *** | *** | ** |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gutiérrez-Ginés, M.J.; Robinson, B.H.; Halford, S.; Alderton, I.; Ambrose, V.; Horswell, J.; Lowe, H. Changes in Soil Chemistry and Soil Nutrient Stocks after 30 Years of Treated Municipal Wastewater Land Disposal: A Natural Experiment. Sustainability 2023, 15, 16230. https://doi.org/10.3390/su152316230
Gutiérrez-Ginés MJ, Robinson BH, Halford S, Alderton I, Ambrose V, Horswell J, Lowe H. Changes in Soil Chemistry and Soil Nutrient Stocks after 30 Years of Treated Municipal Wastewater Land Disposal: A Natural Experiment. Sustainability. 2023; 15(23):16230. https://doi.org/10.3390/su152316230
Chicago/Turabian StyleGutiérrez-Ginés, María Jesús, Brett H. Robinson, Sky Halford, Izzie Alderton, Vikki Ambrose, Jacqui Horswell, and Hamish Lowe. 2023. "Changes in Soil Chemistry and Soil Nutrient Stocks after 30 Years of Treated Municipal Wastewater Land Disposal: A Natural Experiment" Sustainability 15, no. 23: 16230. https://doi.org/10.3390/su152316230
APA StyleGutiérrez-Ginés, M. J., Robinson, B. H., Halford, S., Alderton, I., Ambrose, V., Horswell, J., & Lowe, H. (2023). Changes in Soil Chemistry and Soil Nutrient Stocks after 30 Years of Treated Municipal Wastewater Land Disposal: A Natural Experiment. Sustainability, 15(23), 16230. https://doi.org/10.3390/su152316230







