Abstract
This study investigated the characteristics of using a cost-effective, amine-modified biochar (BC) derived from chicken manure for removing dimethyl sulfide (DMS) from an aqueous solution. The amine-modified BC showed much higher adsorption of DMS compared to commercial activated carbons under varying conditions of contact time, initial concentration, and adsorbent dosage. The DMS removal efficiency increased as the adsorbent dosage was increased from 0.01 to 0.25 g and reached 92.4% even at the relatively low adsorbent dose of 0.015 g. The DMS adsorption capacity of the amine-modified BC (mg/g) increased with increasing DMS concentration, while the incremental rate of the removal efficiency decreased. The adsorption process was well explained by a pseudo-second-order kinetics model. The adsorption of DMS is more appropriately described by the Freundlich isotherm (R2 = 0.989) than by the Langmuir isotherm (R2 = 0.942). The DMS removal efficiency was only reduced by 23.4% even after 10 recovery cycles. The surface area of the amine-modified BC was much higher (9.4 ± 1.2 times) than that of the unmodified BC. The amine-modified BC with a high surface area of 334.6 m2/g can be utilized as a cheap and effective alternative adsorbent to commercial activated carbon for DMS removal.
1. Introduction
Volatile organic sulfur compounds, such as dimethyl sulfide (DMS), dimethyl polysulfides, and carbon disulfide, are toxic and corrosive [1]. These compounds are formed from the anaerobic decay and heating of organic matter, leading to emissions that can raise the ambient concentrations well above the odor threshold level in some situations [2].
DMS is a very powerful odor pollutant owing to its offensive smells with a very low odor threshold value of 0.6–40 ppb [3]. The exposure to DMS can cause human health threats, causing headaches, lung congestion, skin and respiratory irritation, vomiting and several other body ailments, suffocation, and even death at high concentration [4,5]. DMS is released from the cooking of seafood and petroleum refining, and is also generated in the off-gas from paper mills, wastewater treatment, sewage sludge disposal, Kraft process, organic synthesis, dyeing of acetate textiles, pharmaceuticals, insecticides, and fungicide [1]. It also is becoming a metabolic product of many bio-systems. DMS is produced abundantly by marine algae and becomes the principal volatile sulfur compound in seawater [6]. The resulting massive production of atmospheric DMS over the oceans may significantly affect the Earth’s climate [7,8]. It is thus necessary to develop a suitable method to reduce the DMS concentration in ambient air and wastewater.
Consideration of cost and simplicity supports the use of an adsorption process that can be easily operated under ambient conditions. Porous materials such as molecular sieves, zeolites, active carbon, and biochar (BC) can be used for ambient adsorption [9,10]. Several studies have been reported about the removal of DMS on zeolite or activated carbon. Wang et al. [11] used bamboo charcoal of Dendrocalamus to remove DMS from aqueous solution. Wakita et al. [12] have studied the removal of DMS and t-butylmercaptan (TBM) by using of some zeolites and concluded that the main adsorption site on Na-Y was presumably Na+. Cui and Turn [13] used iron chloride-impregnated activated carbon to remove DMS and they confirmed that the addition of FeCl3 created some new active sites and these new active sites likely had stronger affinity with DMS molecule. Tanada et al. and Miyoshi et al. [14,15] investigated the adsorption of DMS and several other organic vapors on silica gel, zeolites and activated carbons. Both research groups reported that the adsorption is determined mainly by the pore structure of the adsorbents rather than by their surface properties.
Carbon materials, such as activated carbon (AC), carbon nanotubes, fullerenes, and graphene, have been widely used in pollution control because of their structural stability and increased micropores, mesopores and surface areas. Due to their remarkable adsorption capability, ACs have been commonly used as an adsorbent for the removal of various contaminants. However, the manufacture of ACs requires high temperature, high pressure, and costly activation processes [16]. The resulting high operational costs have limited the practical applicability of ACs for large plants or facilities. Thus, inexpensive and abundant adsorbents, such as agriculture wastes, nature materials, and non-toxic industrial wastes, should be used as alternative adsorbents for pollutant removal.
As a precursor of AC, biochars have received considerable attention in the past few decades [17,18,19,20]. BC is the carbon-rich product of the thermal decomposition of organic material under a limited supply of O2 and at relatively low temperatures (<700 °C) [21]. BC has been known to act as a super-sorbent for organic contaminants in soil/sediment or wastewater [22,23,24,25]. BC and AC differ primarily in their preparation method, source material, and the resulting physiochemical properties of the products. In contrast to AC, BC use could be a cheaper remediation technology because the source or waste materials are potentially free and BC production at lower temperatures is more energy-efficient and less cost-intensive [26] than AC production.
Thus, BC is a new and promising adsorbent that can be utilized for pollutant removal because of its excellent porous structure, specific surface properties, reusability, minimal costs, and environmentally friendly nature. However, it is hard to selectively adsorb pollutants from a system that contains a mixture of pollutants [27]. To enhance their capacity and selectivity for pollutant removal or adsorbent stability, the modification of the carbon materials in BC has been proposed [11,28]. Although pretreatments or modification of carbon materials such as BCs increase cost, they are necessary steps for improving the selective adsorption of pollutants and for increasing adsorption capacity. Several methods (e.g., acid and alkali modification, oxidation, and chemical grafting) have been used for the pretreatment or modification of BC in order to improve the adsorption capacity [29,30,31]. Generally, these techniques modify the BC’s interfacial regions by increasing its specific surface area, porous structure or chemical reactivity obtained through the formation of surface functional groups that can chemically bond or interact (chelate) with the DMS.
In this work, BC was thermally produced from the slow pyrolysis of chicken manure, a common organic waste, and then modified with HNO3/NH3. We hypothesized that HNO3/NH3 treatment of the BC would increase the number of amine functional groups on its surface and enhance the porous structure of the BCs, thereby enhancing its ability to remove DMS from wastewater. The purpose of this study was to develop a new technology to modify BC derived from chicken manure and to develop value-added and BC-based sorbents for the effective removal of DMS in wastewater.
2. Materials and Methods
2.1. Production of BC
Chicken manure waste collected from a local chicken farm in Ulsan, Korea was used as feedstock for BC production. The chicken manure was pretreated at 110 °C for 48 h to remove moisture and then pyrolyzed to produce BC using a laboratory-scale fluidized bed reactor (FBR) maintained at 450 °C for 1 h. The FBR system consisted of parts for the storage and loading of samples, a fluidized bed reactor, a pre-heated gas (N2) input, a cyclone, and condensation and cooling mechanisms in order to minimize the emission of pollutants generated from the pyrolysis system. The sample feeding rates ranged from 2.1 g/min to 3.7 g/min for lab scale experiments in order to minimize sample clogging inside the throat section of the FBR. The BC was collected from the reactor after being cooled to room temperature of around 22 °C–24 °C.
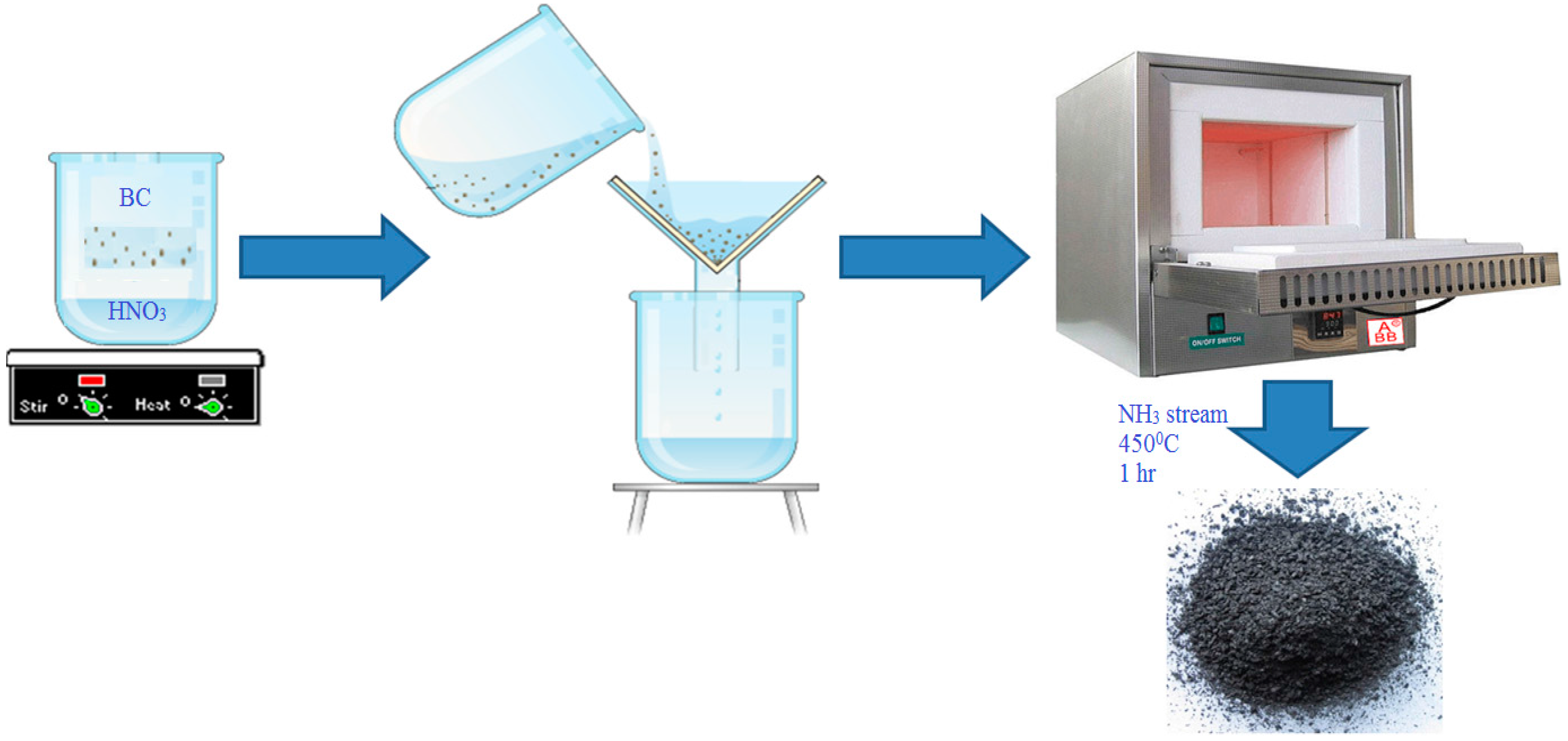
The collected BC was initially mixed with a HNO3 solution (15.7 N) at a 1:10 solid/liquid ratio and stirred for 1 h at room temperature. Upon completion of the HNO3 treatment, the BC was separated from the aqueous phase by centrifugation, washed thoroughly with a mixture of 0.1 M NaOH and subsequently washed with deionized water until the pH of the washing solution reached 6–7 to remove the excess nitric acid. Finally, the acid-treated BC was placed in a quartz tube reactor and modified with anhydrous ammonia gas (min. purity > 99.99%) for 1 h at 450 °C. The amine-modified carbon was dried at 40 °C overnight and stored in a desiccator at room temperature until the adsorption and characterization experiments of the amine-modified BC were conducted (Figure 1).
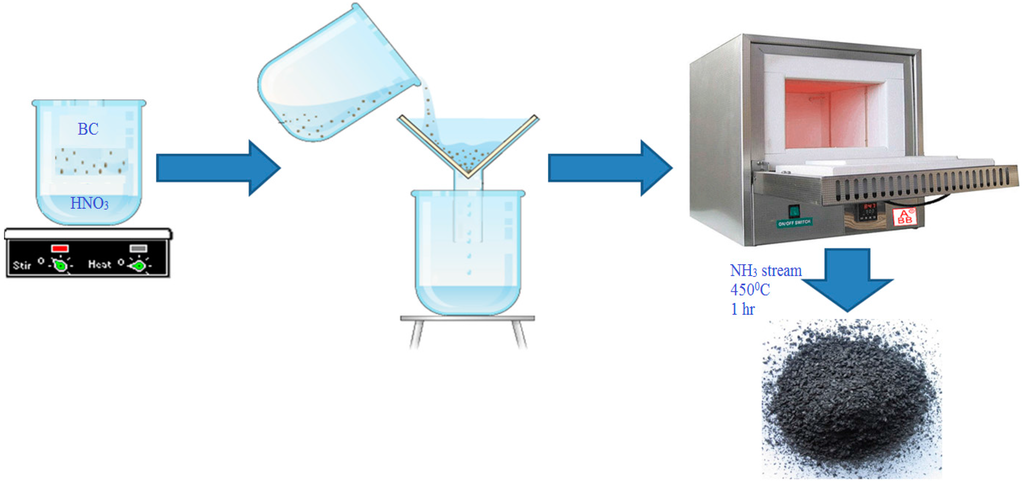
Figure 1.
Schematic of study procedure for pollutant removal by the amine-modified biochar.
Figure 1.
Schematic of study procedure for pollutant removal by the amine-modified biochar.
2.2. Characterization of Biochar (BC)
The elemental composition (carbon, hydrogen, and nitrogen contents) of the amine-modified BC derived from chicken manure was determined using an Elementar Analyzer (LECO-CHNS 932, St. Joseph, MI, USA). The specific surface area of the amine-modified BC was measured with a surface area analyzer (ASAP 2020, Micromeritics Instrument Co., Norcross, GA, USA) using N2 (BET) adsorption methods to determine the macro- and micro-pore surface area. The surface functional ligands were qualitatively analyzed using Fourier transform infrared spectroscopy (5700-FTIR spectrometer of Thermo Nicolet).
2.3. Batch Sorption of DMS
Laboratory batch sorption experiments were utilized to determine the sorption of DMS onto the amine-modified BC. About 0.20 g of the amine-modified BC was mixed with 100 mL of a given concentration of DMS solution (0.001, 0.005, 0.01, 0.015, 0.02, 0.03, 0.04, and 0.05 mg/L) for each adsorption experiment. The mixture was shaken by a mechanical shaker (120 rpm) at the room temperature (22 °C–24 °C). At the end of each experiment, the mixture was filtered out and the DMS concentrations in the filtrate were determined using gas chromatography (GC; Agilent 7890 A Series, Agilent Technologies, Wilmington, DE, USA) equipped with a flame photometric detector.
Blank solutions were included in all sorption experiments. The adsorbed DMS concentrations were calculated based on the differences between the initial and final concentrations. All the sorption experiments were performed in triplicate and their average values were calculated. Additional analyses were repeated whenever the error between two experiments was larger than 10%. All chemical reagents used in the experiments were high purity grades purchased from Aldrich–Sigma, South Korea.
The amount of DMS adsorbed per gram of the amine-modified BC (adsorbent) was calculated by the difference between the initial and final concentrations using Equation (1).
where qe is the equilibrium amount of the DMS adsorbed (mg/g) on the amine-modified BC, Co the initial concentration of DMS (mg/L), Ce the equilibrium concentration of DMS in the aqueous solution (mg/L), V the volume of the used aqueous solution (L), and W the weight of the used BC (g).
2.4. Recycling of Amine-Modified BC
For regeneration-desorption studies, 100 mL of 0.005 mg/L DMS solution was adsorbed with 0.015 g of amine-modified BC using a temperature-controlled shaker incubator at 200 rpm for 2 h. After a 2-h adsorption reaction, the DMS-loaded amino-modified BC was filtered off and washed several times with distilled water to remove excess DMS. Then, the DMS-loaded amine-modified BC was desorbed with 50 mL of 0.01 M NaOH solution. After the alkaline desorption reaction for 60 min, the solution was filtered and the concentration of DMS remaining in the desorption solution was determined by GC, as described above. The same procedure was repeated for 10 consecutive cycles of adsorption and desorption. The DMS in the regenerated solution can be degradable by some microorganism (Microbacterium sp., Thiobacillus novellus, or Pseudomonas sp.) [12,13].
2.5. Adsorption Isotherms
The adsorption isotherm indicates how the adsorbates are partitioned in the adsorbent and the liquid phase at equilibrium. In this study, the equilibrium data obtained from the sorption of DMS was analyzed using the Langmuir and Freundlich isotherms. The Langmuir isotherm is utilized for monolayer sorption and described in the following equation:
where qmax (mg/g) is the monolayer adsorption capacity and KL (L/mg) the heat of adsorption determined from the plot of Ce/qe versus Ce. The favorability or unfavorability of the Langmuir isotherm is expressed by RL (shown in Equation (3)).
An RL value between 0 and 1 indicates favorable adsorption.
The Freundlich isotherm describes multilayer adsorption and is expressed in the following Equation (4):
where KF shows the adsorption capacity (mg/g) and n is an empirical parameter related to the adsorption intensity, which varies with the heterogeneity of the adsorbent. Higher values of 1/n show improved adsorption favorability.
3. Results and Discussion
3.1. Physical Properties
The physical and chemical properties of amine-modified BC (Table 1) showed that the ammoxidation process significantly increased the nitrogen content, which confirmed that nitrogen moieties were inserted into the BC structure by the oxidation reaction with HNO3/NH3 after the triplicate experiments with the standard deviation from these experiments [32,33]. The nitrogen percentage after the 450 °C ammoxidation of the chicken manure-BC was as high as 3.75%, compared to only 2.19% for the BC without the ammoxidation. The process of ammoxidation also significantly changed the contents of carbon, hydrogen, and oxygen. Carbonization of the ammoxidized BC sample increased the carbon content accompanied by a decrease in the oxygen content [29]. This was attributed to loss/evaporation of oxidized volatile components through the high-temperature treatment. Through ammoxidation, the nitrogen content significantly increased (1.56%), indicating nitrogen impregnation into the BC structure. There was a slight increase in hydrogen content (2.53% to 3.07%) after the ammoxidation process, which may have been caused by the incorporation of a considerable amount of nitrogen functional groups, such as amine moieties (-NH2) into the carbon structure [33].
The surface area is an important factor for pollutant adsorption and separation processes. The obtained surface areas of the amine-modified BCs were much higher (9.4 ± 1.2 times) than that of the chicken manure-derived BC. This large increase in surface area of the amine-modified BC was attributed to the decomposition of some acidic functional groups in the BC structure and chemical activation processes of the BC by HNO3 and NH3, accompanied by an improvement in the pore structure. The ammoxidation process can generate several alkaline active sites that may promote the introduction of nitrogen-containing functional groups [33,34]. The Vmico/Vtotal of BC was only 38.5% meaning it has mainly a mesoporous distribution. However, the pore structure characteristics of the amine-modified BCs were primarily attributed to the micropores as the percent contribution of micropores was more than 65% of the Vtotal.

Table 1.
Physical and chemical properties of the amine-modified biochar.
| Physical Properties | BC | Amine-modified BC |
|---|---|---|
| C (%) | 80.25 ± 3.46 | 84.16 ± 4.79 |
| O (%) | 8.64 ± 1.02 | 7.06 ± 0.94 |
| H (%) | 2.53 ± 0.21 | 3.07 ± 0.26 |
| N (%) | 2.19 ± 0.15 | 3.75 ± 0.29 |
| BET (m2/g) | 34.6 ± 4.82 | 334.7 ± 24.57 |
| pHpzc | 3.6 ± 0.21 | 7.7 ± 0.45 |
| Vmicro (cm3/g) | 0.036 ± 0.009 | 0.093 ± 0.011 |
| Vtotal (cm3/g) | 0.101 ± 0.017 | 0.154 ± 0.023 |
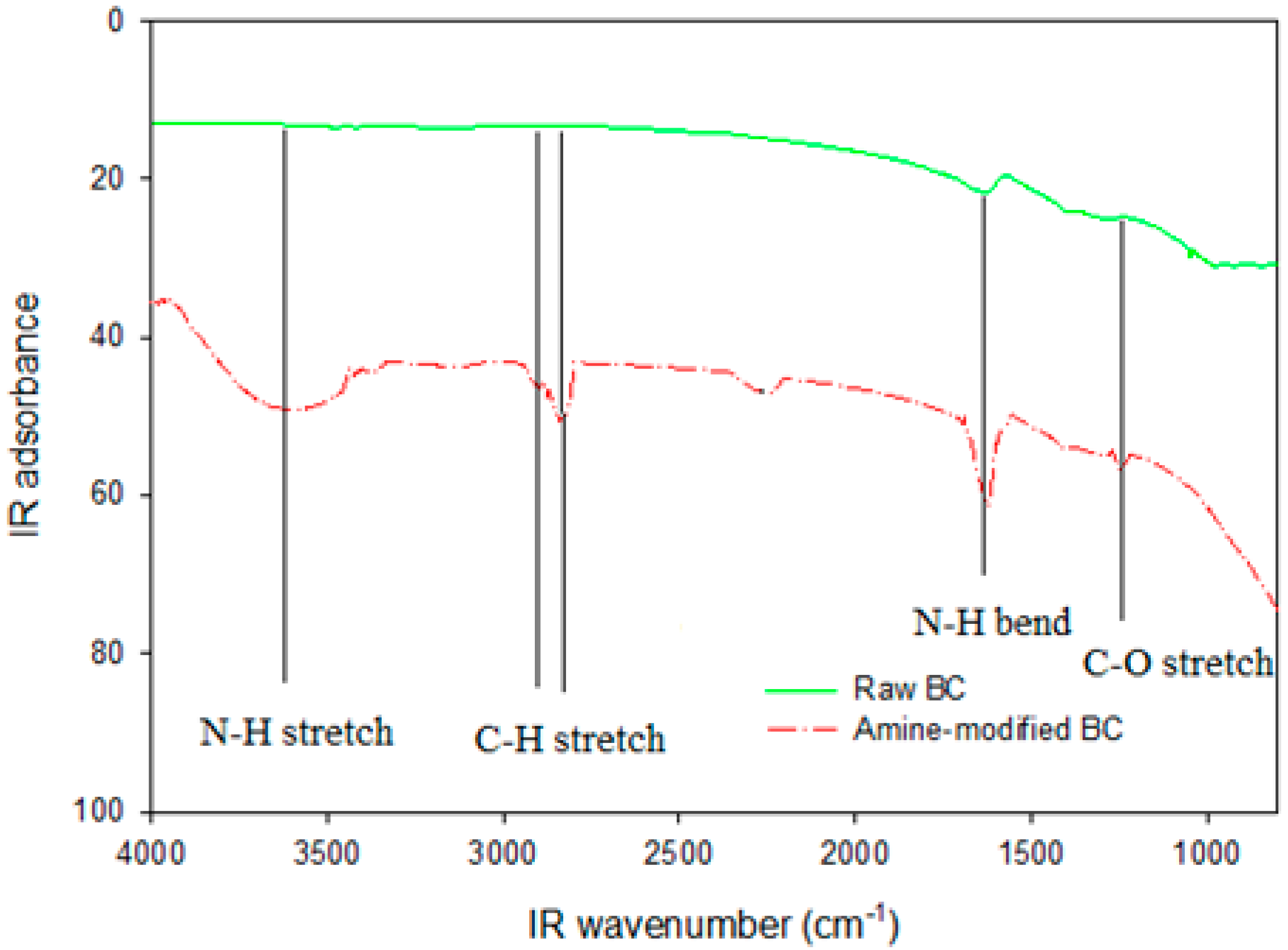
To investigate the functional groups introduced by ammoxidation, the Fourier transform infrared spectroscopy (FTIR) spectra of amine-modified BC and chicken manure-derived BC were analyzed (Figure 2). In comparison with the chicken manure-derived BC spectrum, new bands appeared on the amine-modified BC surface at 1673 cm−1 which was assigned to C≡N peaks. The additional band at 1386 cm−1 was ascribed to C-N stretching. Alkyl CH2 stretching showed peaks at 2968 and 2915 cm−1, while C–O stretching was identified at 1052 cm−1, and 2259 cm−1 may be CO2, carboxyls and lactones functional groups [35,36]. These results suggested that HNO3/NH3 plays a catalytic role in the modification on the amine-modified BC surface, causing amine functionality. HNO3/NH3 treatment also demonstrated that the amine groups had been successfully introduced onto the amine-modified BC surfaces, which would generate new sites for DMS adsorption.
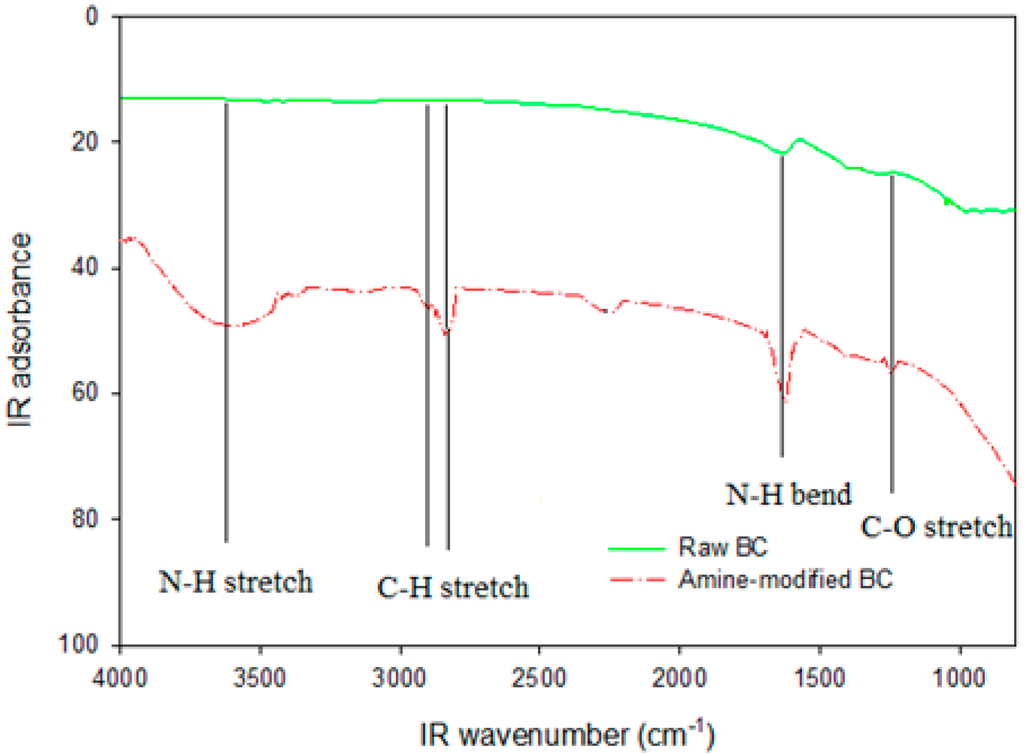
Figure 2.
FTIR spectrum of chicken manure biochar without modification (upper) and amine modified biochar (lower).
Figure 2.
FTIR spectrum of chicken manure biochar without modification (upper) and amine modified biochar (lower).
3.2. Effect of Initial Concentration
Figure 3 shows the effect of initial concentration on the adsorption of DMS by the amine-modified BC derived from chicken manure with varying DMS solution concentrations (0.001, 0.005, 0.01, 0.015, 0.02, 0.03, 0.04, and 0.05 mg/L) using 0.20 g of amine-modified BC. The adsorption capacity (μg/g) of DMS increased with increasing initial concentration of the interest solution, while the incremental rate of the removal efficiency decreased. The increase in the initial DMS concentration raised the number of available molecules per adsorption site. At a low initial solution concentration, the increased surface area and the availability of adsorption sites of the amine-modified BC readily contributed to adsorption/removal of DMS. With an excessive increase in concentration of the interest solution, the more available adsorption sites were limited, which decreased the incremental rate of the DMS removal efficiency [37]. The increased adsorption capacity (mg/g) at the increasing initial concentration is due to the increased concentration gradient between the DMS concentration and the surface of the adsorbent [36,38].
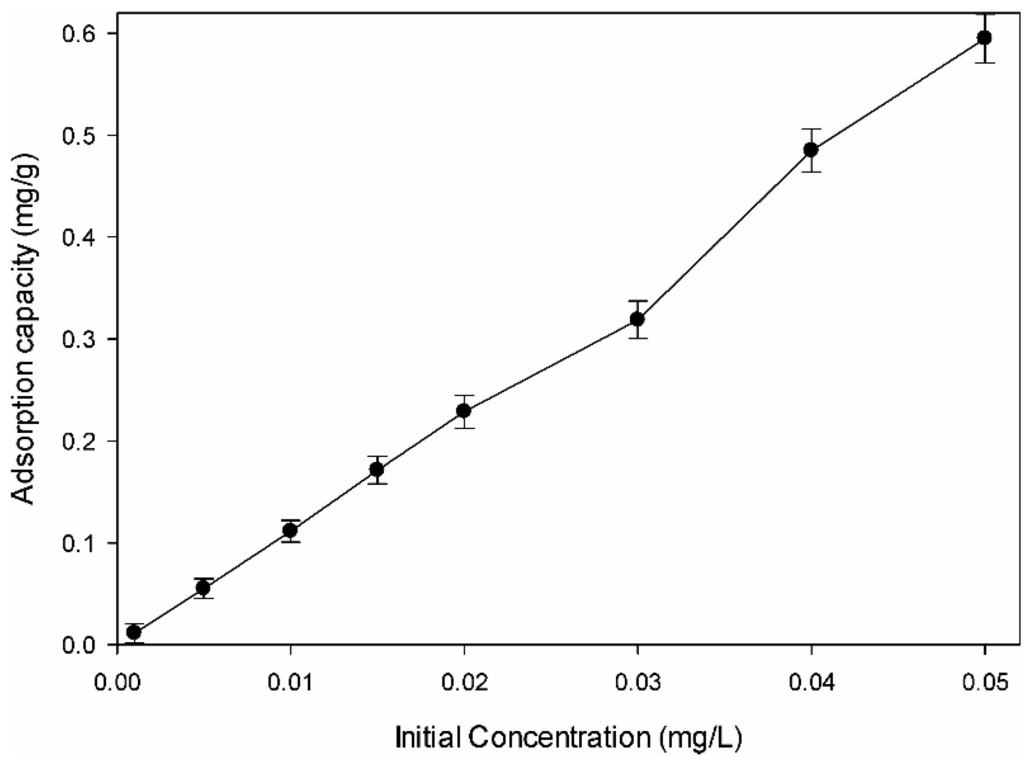
Figure 3.
Effect of initial concentration on adsorption removal of dimethyl sulfide.
Figure 3.
Effect of initial concentration on adsorption removal of dimethyl sulfide.
3.3. Effect of Adsorbent Dosage
The effect of adsorbent dosage of the amine-modified BC on the DMS adsorption was investigated with an initial DMS concentration of 0.05 mg/L. The removal efficiency of DMS increased as the adsorbent dosage was increased from 0.01 to 0.25 g. In particular, the efficiency reached up to 92.4% even at a low adsorbent dosage of 0.015 g (Figure 4). The increase in the DMS removal with increasing adsorbent dosage rate was expected because more surface area and more adsorption sites are available for DMS capture (due to the higher amount of adsorbent in the solution) [39]. The maximum removal was almost achieved as the surface and the solution concentrations of DMS came close to equilibrium with each other [40]. The decrease in the adsorption capacity of the amine-modified BC was due to the splitting effect of flux (concentration gradient) between adsorbates with increasing adsorbent concentration causing a decrease in the amount of DMS adsorbed onto a unit weight of biomass [36,37,41]. A further reason might be due to the particle interaction, such as aggregation or agglomeration, resulting from the high adsorbent dose. Such aggregation/agglomeration decreases the total surface area of the adsorbent and increases the diffusional path length [40].
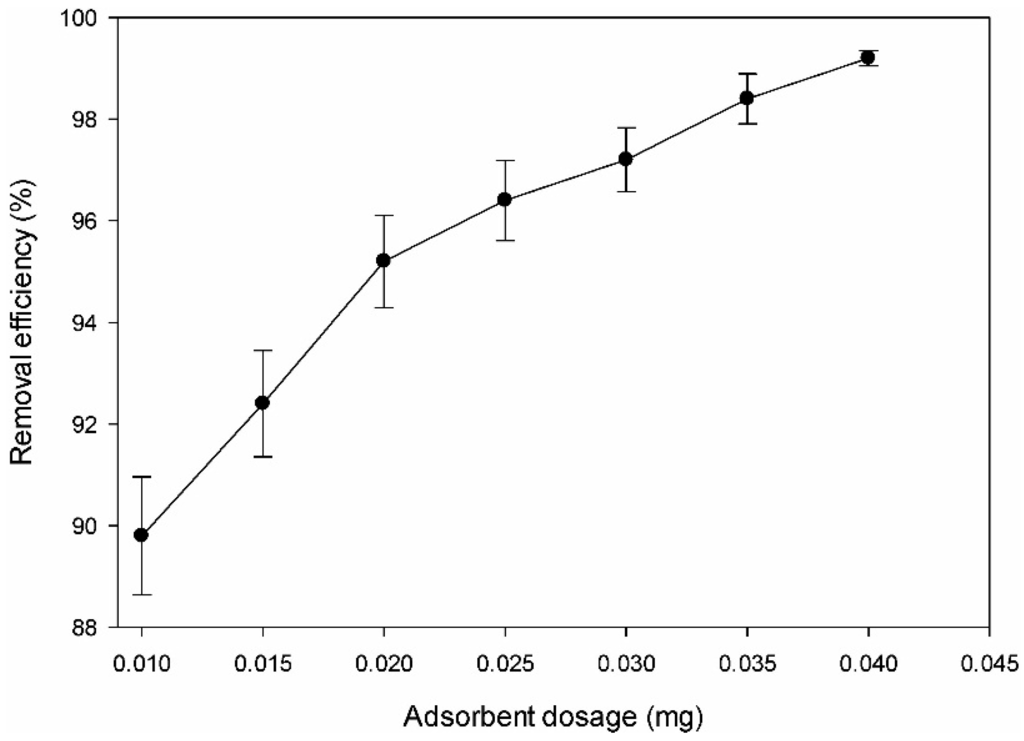
Figure 4.
Effect of adsorbent dosage on adsorption removal of dimethyl sulfide.
Figure 4.
Effect of adsorbent dosage on adsorption removal of dimethyl sulfide.
3.4. Effect of Reaction Time
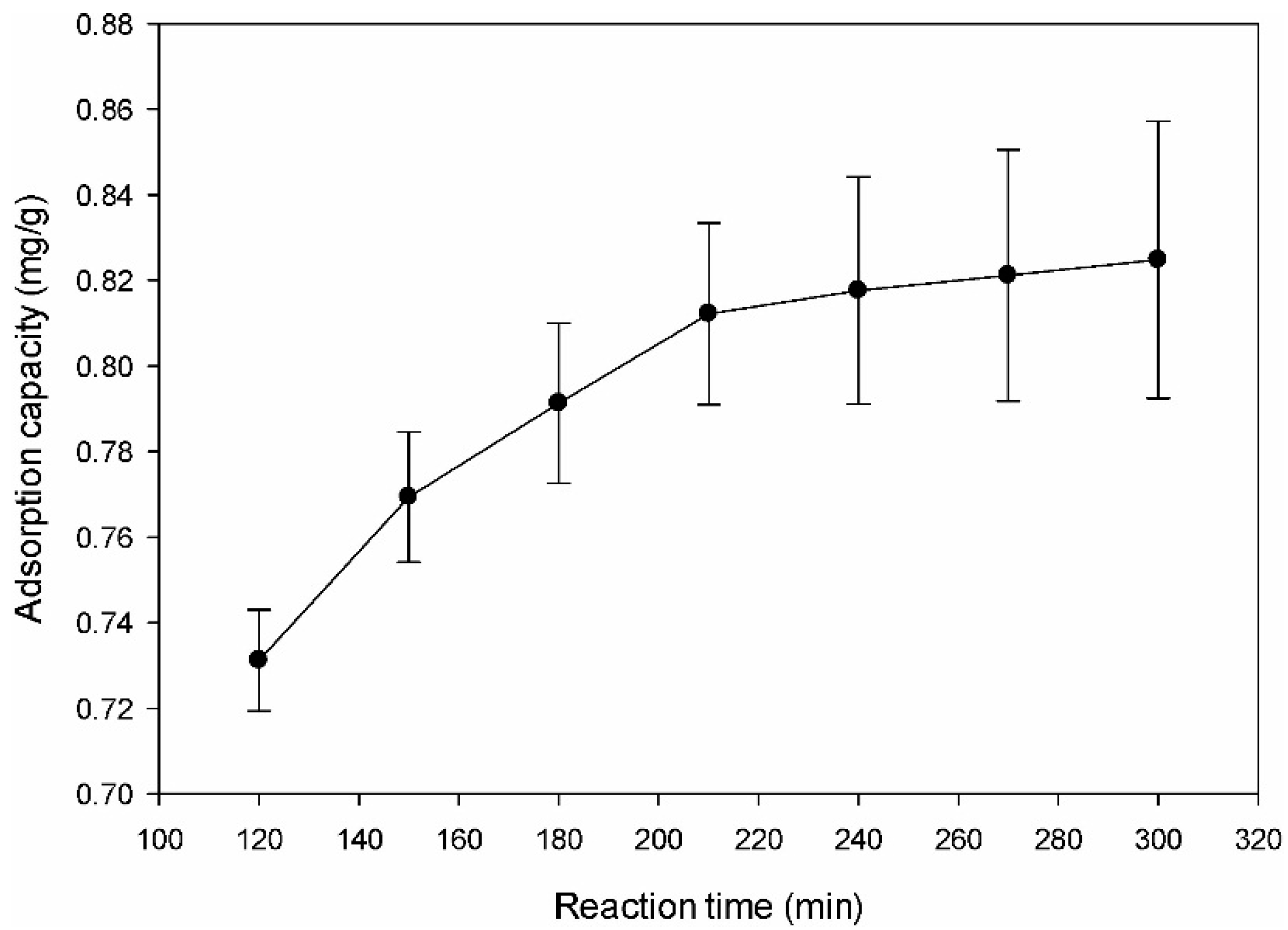
The effect of the contact time on the adsorption of DMS by the amine-modified BC was examined over the time range from 120 min to 300 min. Figure 5 shows that the removal efficiency increased with increasing adsorption time and that the DMS adsorption percentage increased with increasing contact time. The DMS adsorption removal on amine-modified BC was rapid at the initial reaction period but then slowed to a stagnant level with increasing contact time. The adsorption rate determined from the slope of the curve was relatively fast, initially. About 98% of DMS was removed within 3 h, after which the adsorption capacity increased slowly. The initial high adsorption rate was attributed to the abundance of adsorption sites of the amine-modified BC. After the initial stage of the adsorption process, the DMS must transfer further and deeper into the micropores that are subject to a degree of resistance. As a result of further transfer, the sample exhibited a decreased further driving force and adsorption rate [42]. The steep slope of DMS adsorption indicates instantaneous adsorption, which may have been due to the effects of surface functional groups on the surface of adsorbents [42]. Therefore, the adsorption of DMS was thought to have taken place probably via surface adsorption until the surface functional sites were fully occupied, after which the DMS molecules diffused into the pores of the adsorbents for further adsorption.
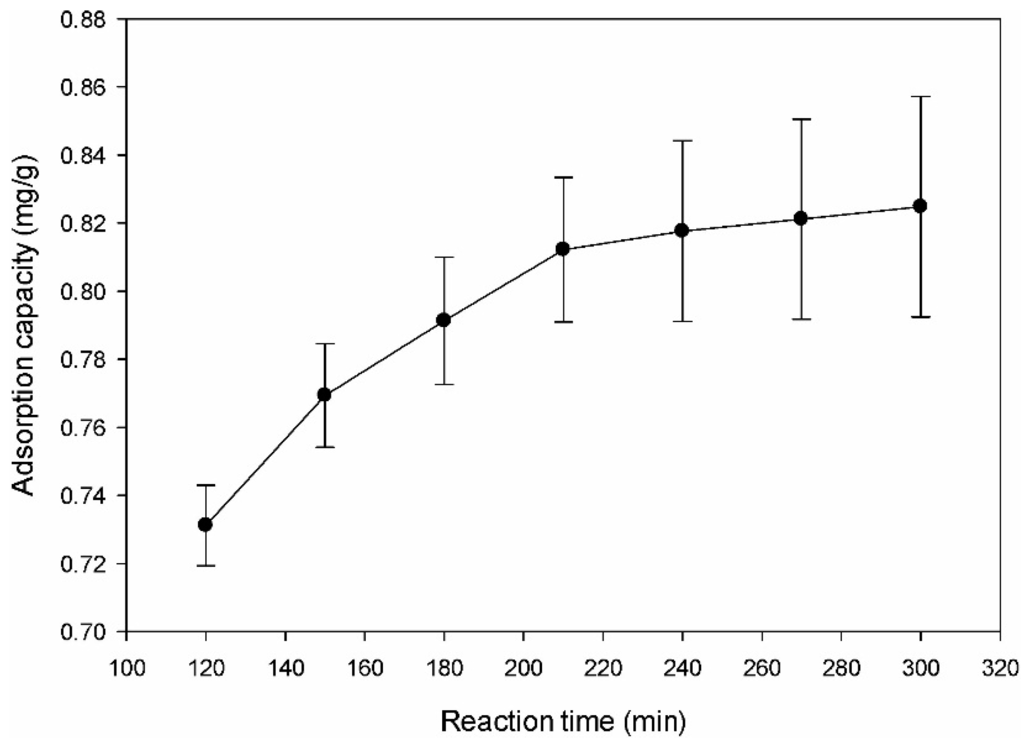
Figure 5.
Effect of reaction time on adsorption removal of dimethyl sulfide.
Figure 5.
Effect of reaction time on adsorption removal of dimethyl sulfide.
3.5. Adsorption Isotherm and Kinetics
The adsorption equilibrium data of DMS on amine-modified BC were analyzed using the Langmuir and Freundlich isotherm models. The adsorption parameters from the two isotherms are shown in Table 2. A comparison of regression coefficients indicates the adsorption of DMS is more appropriately fitted by the Freundlich isotherm (R2 = 0.989) than by the Langmuir isotherm (R2 = 0.942). The Freundlich 1/n was less than one, indicating that DMS was preferentially adsorbed onto the adsorption sites of the amine-modified BC. The adsorption capacity of the amine-modified BC for DMS was much higher than that of the other adsorbents, such as metal organic framework, zeolite, and charcoal (Table 3). This demonstrated that amine-modified BC was more effective for DMS removal than other alternative adsorbents.

Table 2.
Adsorption isotherm constants of the amine-modified biochar for dimethyl sulfide removal.
| Langmuir Isotherm | Freundlich Isotherm | |||||
|---|---|---|---|---|---|---|
| Qmax (mg/g) | KL | RL | R2 | Kf | 1/n | R2 |
| 1.14 | 0.052 | 0.0284 | 0.942 | 12.58 | 0.85 | 0.989 |

Table 3.
Adsorption capacity comparison of dimethyl sulfide by various adsorbents.
| Adsorbent | Methods | Adsorption Capacity (mg/g) | Characteristics | Reference |
|---|---|---|---|---|
| Modified activated carbon | HNO3/O3 | 8.0 | High manufacturing cost Smell and treatment of O3 problems | [43] |
| Amine-modified Biochar | HNO3/NH3 | 1.14 | Cheap | This Study |
| IRMOF-3 | - | 0.87 | Low capacity | [44] |
| Zeolite | AgNO3 | 0.497 | Low capacity | [45] |
| Commercial activated carbon | HNO3 | 0.32 | Low capacity | [46] |
| Bamboo Charcoal | - | 0.266 | Low capacity | [47] |
| Bentonite | Silver | 0.155 | Low capacity | [48] |
The poor regression from the pseudo-first-order model (R2, 0.817) and the difference between qe and qe,cal indicate that the adsorption of DMS onto amine-modified BC does not follow first-order kinetics. The qe,cal value using the pseudo-second-order model is in good agreement with the experimental qe value. In addition, the good regression (R2, 0.994) from the pseudo-second-order model shows that the adsorption process confirmed well to the pseudo-second-order kinetics model (Table 4). This result suggests that DMS and the amine-modified BC are conceivably involved in the rate-determining step, which may be a chemisorption process [49,50,51]. In the following section, the specific interaction between adsorbate and adsorbent is found to be designed for the enhanced adsorption of DMS onto the amine-modified BC.

Table 4.
Adsorption kinetic constants.
| DMS Conc (mg/L) | Qcal (mg/g) | Pseudo First-order Model | Pseudo Second-order Model | ||||
|---|---|---|---|---|---|---|---|
| qe (mg/g) | K1 (g/mg/min) | R2 | qe (mg/g) | K2 (g/mg/min) | R2 | ||
| 0.05 | 0.75 | 1.02 | 0.2307 | 0.817 | 0.87 | 0.1688 | 0.994 |
3.6. Desorption and Regeneration
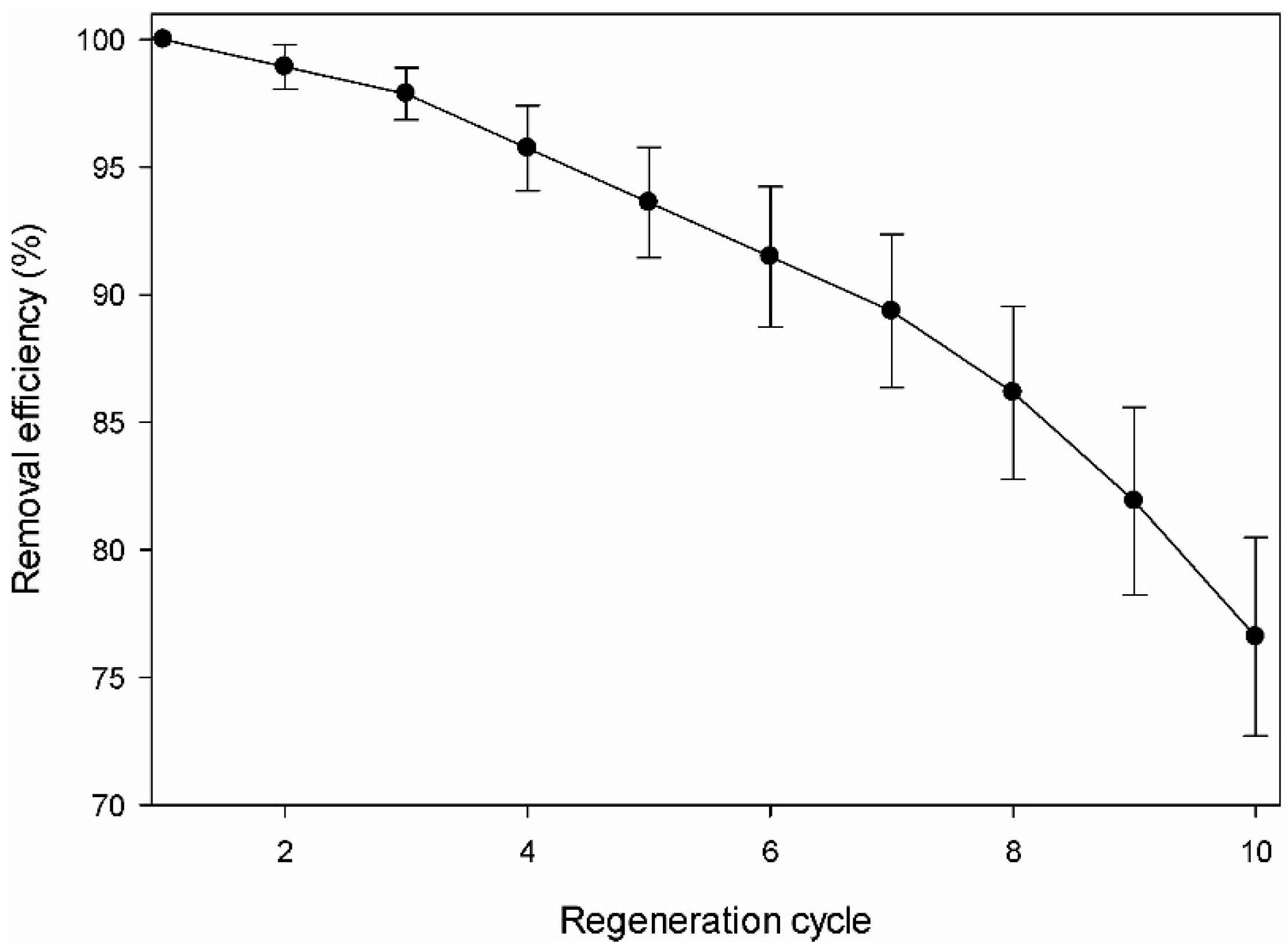
The regeneration or recovery characteristics for the reuse of the pollutant-loaded adsorbent are important features in determining the economic efficiency of the adsorbent. Figure 6 shows the DMS adsorption capacity of the amine-modified BC adsorbent through 10 cycles of repeated recovery (adsorption–desorption cycles) in series. Even after 10 cycles, the adsorption efficiency of DMS was maintained at approximately 76.6% of the initial efficiency. The adsorption capacity of DMS in the first cycle of 0.0677 mg/g was only decreased by 1.1% of the initial efficiency in the second cycle. In subsequent cycles, the adsorption capacity decreased slowly, so that the total loss of adsorption capacity over the 10 cycles was only 23.4%. This result indicates that the adsorbent still showed almost 76.6% of the initial adsorption capacity even after being recovered 10 times. The DMS remaining in the aqueous solution after the adsorption process was lower than the regulated standard by the US EPA. Consideration of the advantageous features of the amine-modified BC adsorbent in repeated cycles of DMS loading and elution (adsorption and desorption) supports the use of the adsorbent as an inexpensive candidate for the effective removal of DMS from the environment.
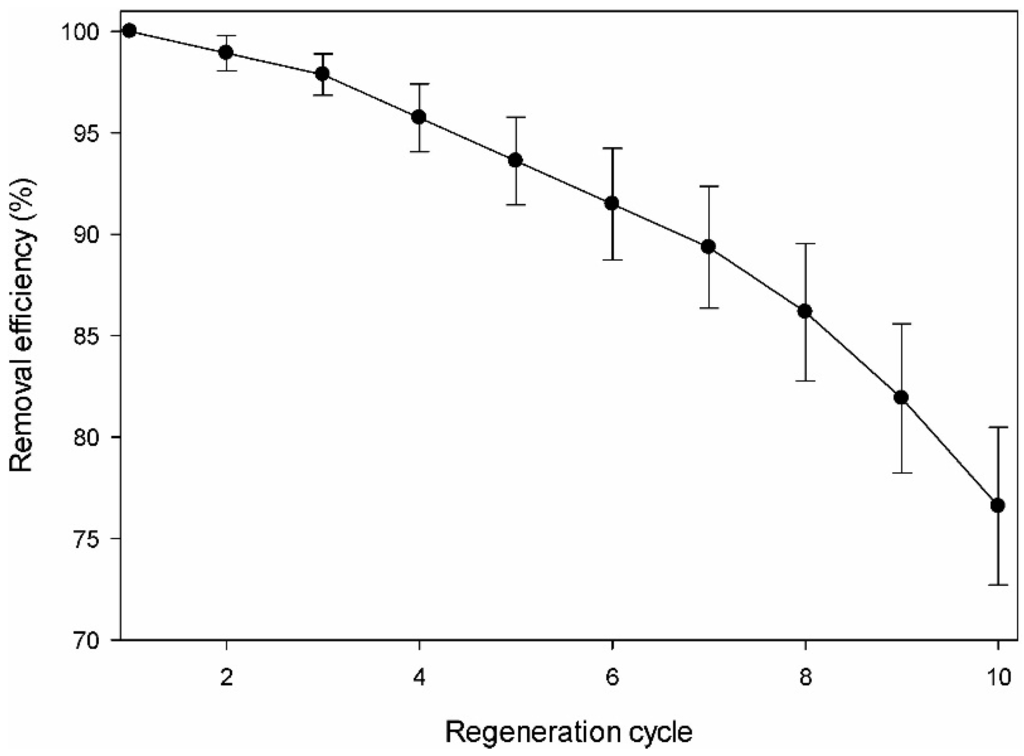
Figure 6.
Desorption and regeneration of the amine-modified biochar.
Figure 6.
Desorption and regeneration of the amine-modified biochar.
3.7. Adsorption Cost Estimation
The adsorption cost for DMS removal by the amine-modified BC derived from chicken manure was an important factor for evaluating the BC that can be utilized as an alternative adsorbent for industrial applications. The economic feasibility of the adsorptive removal of DMS from water and wastewater depends on the availability, efficacy, and cost effectiveness of adsorbents.
A simplified cost comparison for DMS removal of the previously reported adsorbents and the amine-modified BC is given in Table 5. Numerous commercially-available activated carbons or low-cost adsorbents have been used for the adsorption of pesticides and DMS by adsorbents, both as-received and after chemical or physical modifications. Direct adsorption cost varies when these adsorbents are made in well-developed, developing, or underdeveloped countries. However, unfortunately, the adsorption cost for removal of pesticides and DMS by the adsorbents developed from waste materials has seldom been reported because it strongly depends on the local availability, processing required, treatment conditions, and both regeneration and lifetime characteristics of the adsorbent.
In this study, chicken manure, a waste poultry biomass, was collected at no cost from a local farm in Ulsan, South Korea. Such chicken manure is freely available in Korea as its collection helps to reduce the waste disposal burden for the farm. After considering the expenses for transport, required chemicals, and electrical energy requirements, the preparation cost of chicken manure-derived BC is approximately US Dollar (USD) $1.60/kg. The manufacturing costs for the amine-modified BC only comprise purchasing the chemical cost for the modification process after obtaining BC from chicken manure by pyrolysis.
The roughly calculated adsorption costs of the amine-modified BC, commercial AC which is purchased from Korean chemical company [52,53], natural zeolite and charcoal were $2.28 ± 0.66, $2.63 ± 0.63, $3.10 ± 0.72, and $3.15 ± 0.55 USD for adsorption removal of 1 g of DMS contained in aqueous solution, respectively. The simply estimated adsorption cost by the amine-modified BC was 13.1%, 26.4%, and 27.6% less than that of the commercial AC, natural zeolite and charcoal, respectively. However, this cost comparison was calculated based on laboratory-scale experiments. The adsorption cost could be further reduced for industrial application by minimizing transportation and handling costs on a larger scale, particularly for the amine-modified BC.
BC can also be more economically manufactured in a larger production facility. Increasing the size of the manufacturing plant will raise the initial capital costs, but subsequently lower the production costs due to higher product output. Ideally, the proper size of manufacturing equipment must be installed to maximize throughput and minimize production or manufacturing cost.
Based on the simply estimated the adsorption cost, the amine-modified BC can be used as a low-cost and highly effective adsorbent for DMS removal from aqueous solutions. In addition, the amine-modified BC can be used as an alternative to commercial AC or natural zeolite (Table 5).

Table 5.
Adsorption cost estimation of various adsorbents
| This Study | Commercially Available | |||||
|---|---|---|---|---|---|---|
| Cost/capacity | Chicken Manure-derived BC | Amine-Modified BC | AC | Zeolite | Charcoal | |
| Adsorbent Preparation | Purchase and operation cost (USD/kg) | 0.60 ± 0.20 | - | 21 ± 2.5 | 1.55 ± 0.50 | 0.85 ± 0.15 |
| Chemical consumption (USD/kg) | 1 ± 0.1 | 1 ± 0.1 | - | - | - | |
| Total manufacturing cost (USD/kg) | 1.30 ± 0.30 | 2.60 ± 0.40 * | 21 ± 2.5 | 1.55 ± 0.50 | 0.85 ± 0.15 | |
| Adsorption capacity (g/kg) | - | 1.14 ± 0.12 | 8 ± 0.8 | 0.50 ± 0.05 | 0.27 ± 0.03 | |
| Adsorption cost (USD/g) | 2.28 ± 0.66 | 2.63 ± 0.63 | 3.10 ± 0.72 | 3.15 ± 0.55 | ||
* Total Manufacturing cost of amine-modified BC = Total manufacturing cost of BC + Adsorbent preparation cost of amine-modified. The data was the average of three replicated experiments and the error was the standard deviation of the three replicated experiments. BC: Biochar; AC: activated carbon.
4. Conclusions
Amine-modified BC derived from chicken manure exhibited remarkably enhanced adsorption of DMS from aqueous solution when compared with other adsorbents. The ammonia treatment improved the physiochemical properties of the chicken manure-derived BC, by providing more surface area and adding more amine functional groups. The amine-modification of chicken manure BC greatly increased the surface area (9.4 times) as compared with the unmodified chicken manure-derived BC and, thus, significantly improved its adsorption capacity for DMS removal from aqueous solution. Under the optimum adsorption conditions, the maximum DMS removal capacity by the amine-modified BC was 1.14 mg/g. The adsorption of DMS by the amine-modified BC followed the pseudo-second-order model and was better described by the Freundlich isotherm. After even 10 cycles of the sorption-desorption process, the amine-modified BC retained 76.6% of the original adsorption capacity. Based on the roughly-estimated adsorption costs of the adsorbent for DMS removal, the amine-modified chicken manure BC provides a simple, low-cost, and promising option for DMS treatment.
Acknowledgments
This study was supported by the 2014 research fund of the Ulsan Science Park funded through Ulsan Technopark, Korea.
Author Contributions
All authors contributed equally to this work.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Smet, E.; van Langenhove, H. Abatement of volatile organic sulfur compounds in odorous emissions from the bio-industry. Biodegradation 1988, 9, 273–284. [Google Scholar] [CrossRef]
- Lens, P.N.L.; Pol, L.H. Environmental Technologies to Treat Sulfur Pollution, Principles and Engineering; IWA Publishing: London, UK, 2000; pp. 281–304. [Google Scholar]
- Mudliar, S.N.; Deshmukh, S.C.; Banerjee, S.; Pandey, R.A. Treatment of waste gas containing low concentration of dimethyl sulphide (DMS) in a bench-scale biofilter. Bioresour. Technol. 2010, 101, 2185–2190. [Google Scholar] [CrossRef] [PubMed]
- Jäppinen, P.; Kangas, J.; Silakoski, L.; Savolainen, H. Volatile metabolites in occupational exposure to organic sulfur compounds. Arch. Toxicol. 1993, 67, 104–106. [Google Scholar] [CrossRef] [PubMed]
- Terazawa, K.; Mizukami, K.; Wu, B.; Takatori, T. Fatality due to inhalation of dimethyl sulfide in a confined space: A case report and animal experiments. Int. J. Legal Med. 1991, 104, 141–144. [Google Scholar] [CrossRef] [PubMed]
- Opdyke, D.L.J. Monographs on fragrance raw materials. Dimethyl sulfide. Food Cosmet. Toxicol. 1979, 17, 365–368. [Google Scholar]
- Malin, G.; Turner, S.M.; Liss, P.S. Sulfur: The plankton/climate connection. J. Phycol. 1992, 28, 590–597. [Google Scholar] [CrossRef]
- Simpson, D.; Winiwarter, W.; Börjesson, G.; Cinderby, S.; Ferreiro, A.; Guenther, A.; Hewitt, C.N.; Janson, R.; Khalil, M.; Aslam, K.; et al. Inventorying emissions from nature in Europe. J. Geophys. Res. 1999, 104, 8113–8152. [Google Scholar] [CrossRef]
- Crespo, D.; Wang, Y.; Yang, F.H.; Yang, R.T. Superior sorbent for natural gas desulfurization. Ind. Eng. Chem. Res. 2008, 47, 1238–1244. [Google Scholar] [CrossRef]
- Khosravi, M.; Gholikandi, G.B.; Bali, A.S.; Riahi, R.; Tashaouei, H.R. Membrane Process Design for the Reduction of Wastewater Color of the Mazandaran Pulp-Paper Industry, Iran. Water. Resour. Manag. 2011, 25, 2989–3004. [Google Scholar] [CrossRef]
- Wang, M.; Huang, Z.H.; Liu, G.; Kang, F. Adsorption of dimethyl sulfide from aqueous solution by a cost-effective bamboo charcoal. J. Hazard. Mater. 2011, 190, 1009–1015. [Google Scholar] [CrossRef] [PubMed]
- Wakita, H.; Tachibana, Y.; Hosaka, M. Removal of dimethyl sulfide and t-butylmercaptan from city gas by adsorption on zeolites. Microporous Mesoporous Mater. 2011, 46, 237–247. [Google Scholar] [CrossRef]
- Cui, H.; Turn, S.Q. Adsorption/desorption of dimethylsulfide on activated carbon modified with iron chloride. Appl. Catal. B 2009, 88, 25–31. [Google Scholar] [CrossRef]
- Tanada, S.; Boki, K.; Matsumoto, K. Adsorption properties of methyl sulfide and methyl disulfide on activated carbon, zeolite, and silicate, and their porous structure. Chem. Pharm. Bull. 1978, 26, 1527–1532. [Google Scholar] [CrossRef]
- Miyoshi, T.; Tanada, S.; Boki, K. Studies on adsorption isotherms of eight odorous pollutants. Sangyo Igaku 1979, 21, 178–179. [Google Scholar] [CrossRef] [PubMed]
- Boehm, H.P. Some aspects of the surface chemistry of carbon blacks and other carbons. Carbon 1994, 32, 759–769. [Google Scholar] [CrossRef]
- Azargohar, R.; Dalai, A.K. Biochar as a precursor of activated carbon. Appl. Biochem. Biotechnol. 2006, 131, 762–773. [Google Scholar] [CrossRef]
- Hayes, M.H.B. Biochar and biofuels for a brighter future. Nature 2006. [Google Scholar] [CrossRef] [PubMed]
- Renner, R. Rethinking biochar. Environ. Sci. Technol. 2007, 41, 5932–5933. [Google Scholar] [CrossRef] [PubMed]
- Moussavi, G.; Khosravi, R. Preparation and characterization of a biochar from pistachio hull biomass and its catalytic potential for ozonation of water recalcitrant contaminants. Bioresour. Technol. 2012, 119, 66–71. [Google Scholar] [CrossRef] [PubMed]
- Hale, S.E.; Hanley, K.; Lehmann, J.; Zimmerman, A.; Cornelissen, G. The effects of chemical, biological and physical aging as well as soil addition on the sorption of pyrene to activated carbon and biochar. Environ. Sci. Technol. 2011, 45, 10445–10453. [Google Scholar] [CrossRef] [PubMed]
- Sattar, A.; Gilani, A.M.; Saeed, M.A. Gas chromatographic examination of the essential oil of Cinnamomum camphora. Pak. J. Sci. Ind. Res. 1991, 34, 135–136. [Google Scholar]
- Pandey, A.K.; Bora, H.R.; Deka, S.C.; Rastaji, R.C.; Banch, A.K.S. Composition of the essential oil of the bark of Cinnamomum camphora. J. Med. Arom. Plant. Sci. 1997, 19, 408–409. [Google Scholar]
- Lou, L.P.; Luo, L.; Cheng, G.H.; Wei, Y.F.; Mei, R.W.; Xun, B.; Xu, X.U.; Hu, B.L.; Chen, Y.X. The sorption of pentachlorophenol by aged sediment supplemented with black carbon produced from rice straw and fly ash. Bioresour. Technol. 2012, 112, 61–66. [Google Scholar] [CrossRef] [PubMed]
- Inyang, M.; Gao, B.; Yao, Y.; Xue, Y.W.; Zimmerman, A.; Pullammanappallil, P.; Cao, X.D. Removal of heavy metals from aqueous solution by biochars derived from anaerobically digested biomass. Bioresour. Technol. 2012, 110, 50–56. [Google Scholar] [CrossRef] [PubMed]
- Kumar, Y.P.; King, P.; Prasad, V.S.R.K. Equilibrium and kinetic studies for the bio-sorption system of copper (II) ion from aqueous solution using Tectona grandis L.f. leaves powder. J. Hazard. Mater. 2006, 137, 1211–1217. [Google Scholar] [CrossRef] [PubMed]
- Nowack, K.O.; Cannon, F.S.; Mazyck, D.W. Enhancing Activated Carbon Adsorption of 2-methylisoborneol: Methane and Steam Treatments. Environ. Sci. Technol. 2004, 38, 276–284. [Google Scholar] [CrossRef] [PubMed]
- Chen, J.P.; Wu, S.; Chong, K. Surface modification of a granular activated carbon by citric acid for enhancement of copper adsorption. Carbon 2003, 41, 1979–1986. [Google Scholar] [CrossRef]
- Xue, Y.W.; Gao, B.; Yao, Y.; Inyang, M.; Zhang, M.; Zimmerman, A.R.; Ro, K.S. Hydrogen peroxide modification enhances the ability of biochar (hydrochar) produced from hydrothermal carbonization of peanut hull to remove aqueous heavy metals: Batch and column tests. Chem. Eng. J. 2012, 200–202, 673–680. [Google Scholar] [CrossRef]
- Liu, P.; Liu, W.; Jiang, H.; Chen, J.J.; Li, W.W.; Yu, H.Q. Modification of bio-char derived from fast pyrolysis of biomass and its application in removal of tetracycline from aqueous solution. Bioresour. Technol. 2012, 121, 235–240. [Google Scholar] [CrossRef] [PubMed]
- Gutiérrez-Valero, M.D.; Godino-Salido, M.L.; Arranz-Mascarós, P.; López-Garzón, R.; Cuesta, R.; García-Martín, J. Adsorption of designed pyrimidine derivative ligands on an activated carbon for the removal of Cu(II) ions from aqueous solution. Langmuir 2007, 23, 5995–6003. [Google Scholar] [CrossRef] [PubMed]
- Deng, S.; Ting, Y.P. Fungal biomass with grafted poly(acrylic acid) for enhancement of Cu(II) and Cd(II) biosorption. Langmuir 2005, 21, 5940–5948. [Google Scholar] [CrossRef] [PubMed]
- Nowicki, P.; Pietrzak, R.; Wachowska, H. Influence of the precursor metamorphism degree on preparation of nitrogen-enriched ACs by ammoxidation and chemical activation of coals. Energy Fuel 2009, 23, 2205–2212. [Google Scholar] [CrossRef]
- Starcka, J.; Burga, P.; Mullera, S.; Bimerb, J.; Furdinc, G.; Fiouxd, P.; Guterld, C.V.; Begine, D.; Fauref, P.; Azambre, B. The influence of demineralisation and ammoxidation on the adsorption properties of an activated carbon prepared from a Polish lignite. Carbon 2006, 44, 2549–2557. [Google Scholar] [CrossRef]
- Chen, Y.L.; Pan, B.C.; Li, H.Y.; Zhang, W.M.; Lv, L.; Wu, J. Selective removal of Cu(II) ions by using cation-exchange resin-supported polyethyleneimine (PEI) nanoclusters. Environ. Sci. Technol. 2010, 44, 3508–3513. [Google Scholar] [CrossRef] [PubMed]
- Yu, L.J.; Shukla, S.S.; Dorris, K.L.; Shukla, A.; Margrave, J.L. Adsorption of chromium from aqueous solutions by maple sawdust. J. Hazard. Mater. B 2003, 100, 53–63. [Google Scholar] [CrossRef]
- Gupta, V.K.; Ali, I. Removal of lead and chromium from wastewater using bagasse fly ash—A sugar industry waste. J. Colloid Interface Sci. 2004, 271, 321–328. [Google Scholar] [CrossRef] [PubMed]
- Shukla, A.; Zhang, Y.H.; Dubey, P.; Margrave, J.L.; Shukla, S.S. The role of sawdust in the removal of unwanted materials from water. J. Hazard. Mater. B 2002, 95, 137–152. [Google Scholar] [CrossRef]
- Bhargava, D.S.; Sheldarkar, S.B. Effects of adsorbent dose and size on phosphate-removal from wastewaters. Environ. Pollut. 1992, 76, 51–60. [Google Scholar] [CrossRef]
- Naiya, T.K.; Bhattacharya, A.K.; Das, S.K. Adsorption of Cd(II) and Pb(II) from aqueous solutions on activated alumina. J. Colloid Interface Sci. 2009, 333, 14–26. [Google Scholar] [CrossRef] [PubMed]
- Wang, F.Y.; Wang, H.; Ma, J.W. Adsorption of cadmium(II) ions from aqueous solution by a new low-cost adsorbent-bamboo charcoal. J. Hazard. Mater. 2010, 177, 300–306. [Google Scholar] [CrossRef] [PubMed]
- Denga, H.; Lua, J.; Lib, G.; Zhanga, G.; Wang, X. Adsorption of methylene blue on adsorbent materials produced from cotton stalk. Chem. Eng. J. 2011, 172, 326–334. [Google Scholar] [CrossRef]
- Vega, E.; Lemus, J.; Anfruns, A.; Gonzalez-Olmos, R.; Palomar, J.; Martin, M.J. Adsorption of volatile sulphur compounds onto modified activated carbons: Effect of oxygen functional groups. J. Hazard. Mater. 2013, 258–259, 77–83. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.L.; Fan, H.L.; Tian, Z.; He, E.Y.; Li, Y.; Shangguan, J. Adsorptive removal of sulfur compounds using IRMOF-3 at ambient temperature. Appl. Surf. Sci. 2014, 289, 107–113. [Google Scholar] [CrossRef]
- Hwang, C.L.; Tai, N.H. Removal of dimethylsulfide by adsorption on ion-exchanged zeolites. Appl. Catal. B 2010, 93, 363–367. [Google Scholar] [CrossRef]
- Goyal, M.; Dhawan, R.; Bhagat, M. Adsorption of dimethyl sulfide vapors by activated carbons. Colloid Surf. A 2008, 322, 164–169. [Google Scholar] [CrossRef]
- Huang, H.; Yi, D.; Lu, Y.; Wu, X.; Bai, Y.; Meng, X.; Shi, L. Study on the adsorption behavior and mechanism of dimethyl sulfide on silver modified bentonite by in situ FTIR and temperature-programmed desorption. Chem. Eng. J. 2013, 225, 447–455. [Google Scholar] [CrossRef]
- Shu, C.C.H.; Chen, C.K. Enhanced removal of dimethyl sulfide from a synthetic waste gas stream using a bioreactor inoculated with Microbacterium sp. NTUT26 and Pseudomonas putida. J Ind Microbiol. Biotechnol. 2009, 36, 95–104. [Google Scholar] [CrossRef] [PubMed]
- Ho, Y.S.; McKay, G. A comparison of chemisorption kinetic models applied to pollutant removal on various sorbents. Trans. Inst. Chem. Eng. 1998, 76, 332–340. [Google Scholar] [CrossRef]
- Rao, M.M.; Rao, G.P.C.; Seshaiah, K.; Choudary, N.V.; Wang, M.C. Activated carbon from Ceiba pentandra hulls, an agricultural waste, as an adsorbent in the removal of lead and zinc from aqueous solutions. Waste Manag. 2008, 28, 849–858. [Google Scholar] [CrossRef] [PubMed]
- Sin, Y.H.; Lin, Y.H.; Hou, K.Y.; Hwang, S.C.J. Degradation of dimethyl-sulfoxide-containing wastewater using airlift bioreactor by polyvinyl—Alcohol-immobilized cell beads. Bioresour. Technol. 2011, 102, 5609–5616. [Google Scholar] [CrossRef] [PubMed]
- Carbon, Activated, Powder. Available online: http://www.daejungchem.co.kr/eng/product/search_popup.asp?idx=23402 (accessed on 9 November 2015).
- Carbon, Activated, Granular. Available online: http://www.daejungchem.co.kr/eng/product/search_popup.asp?idx=23399 (accessed on 9 November 2015).
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
