High Fat Rodent Models of Type 2 Diabetes: From Rodent to Human
Abstract
:1. Introduction
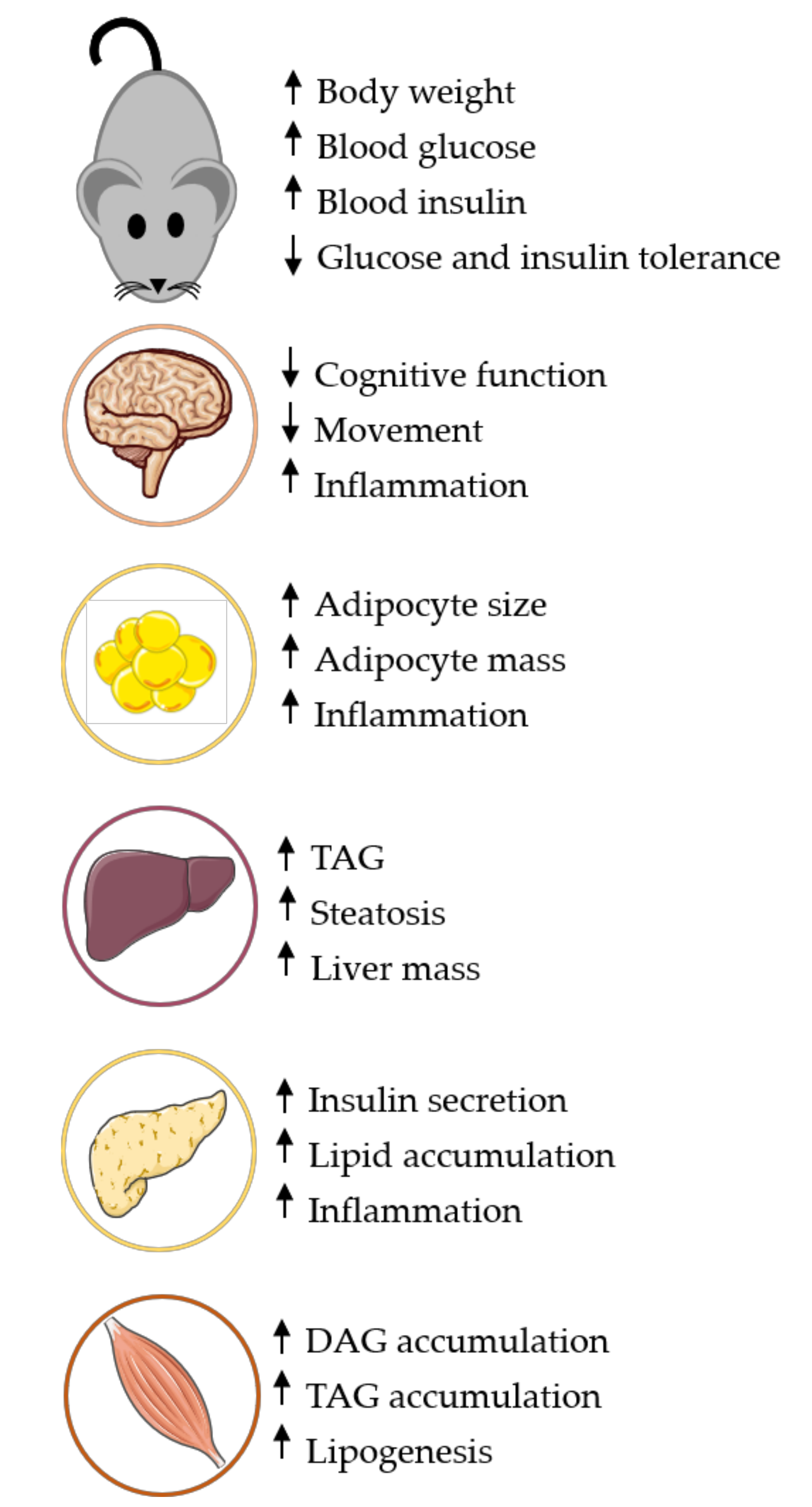
2. Tissue-Specific Effects of HFD Models
2.1. Liver
2.2. Adipose
2.3. Pancreas
2.4. Brain
2.5. Skeletal Muscle
3. Choosing the Most Appropriate Model
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Agriculture, USA. 2015–2020 Dietary Guidelines for Americans; U.S. Department of Health and Human Services and U.S. Department of Agriculture: Washington, DC, USA, 2015; pp. 1–144.
- Mazidi, M.; Kengne, A.-P.; Mikhailidis, D.P.; Toth, P.P.; Ray, K.K.; Banach, M. Dietary food patterns and glucose/insulin homeostasis: A cross-sectional study involving 24,182 adult Americans. Lipids Health Dis. 2017, 16, 192. [Google Scholar] [CrossRef] [Green Version]
- Hariri, N.; Gougeon, R.; Thibault, L. A highly saturated fat-rich diet is more obesogenic than diets with lower saturated fat content. Nutr. Res. 2010, 30, 632–643. [Google Scholar] [CrossRef]
- Licholai, J.A.; Nguyen, K.P.; Fobbs, W.C.; Schuster, C.J.; Ali, M.A.; Kravitz, A.V. Why Do Mice Overeat High-Fat Diets? How High-Fat Diet Alters the Regulation of Daily Caloric Intake in Mice. Obesity (Silver Spring) 2018, 26, 1026–1033. [Google Scholar] [CrossRef] [Green Version]
- Kubant, R.; Poon, A.N.; Sanchezhernandez, D.; Domenichiello, A.F.; Huot, P.S.P.; Pannia, E.; Cho, C.E.; Hunschede, S.; Bazinet, R.P.; Anderson, G.H. A comparison of effects of lard and hydrogenated vegetable shortening on the development of high-fat diet-induced obesity in rats. Nutr. Diabetes 2015, 5, e188. [Google Scholar] [CrossRef]
- Lee, R.K.; Hittel, D.S.; Nyamandi, V.Z.; Kang, L.; Soh, J.; Sensen, C.W.; Shearer, J. Unconventional microarray design reveals the response to obesity is largely tissue specific: Analysis of common and divergent responses to diet-induced obesity in insulin-sensitive tissues. Appl. Physiol. Nutr. Metab. 2012, 37, 257–268. [Google Scholar] [CrossRef]
- De Fourmestraux, V.; Neubauer, H.; Poussin, C.; Farmer, P.; Falquet, L.; Burcelin, R.; Delorenzi, M.; Thorens, B. Transcript Profiling Suggests That Differential Metabolic Adaptation of Mice to a High Fat Diet Is Associated with Changes in Liver to Muscle Lipid Fluxes. J. Biol. Chem. 2004, 279, 50743–50753. [Google Scholar] [CrossRef] [Green Version]
- Tajima, K.; Shirakawa, J.; Okuyama, T.; Kyohara, M.; Yamazaki, S.; Togashi, Y.; Terauchi, Y. Effects of metformin on compensatory pancreatic β-cell hyperplasia in mice fed a high-fat diet. Am. J. Physiol. Endocrinol. Metab. 2017, 313, E367–E380. [Google Scholar] [CrossRef] [Green Version]
- Moretto, T.L.; Benfato, I.D.; De Carvalho, F.P.; Barthichoto, M.; Le Sueur-Maluf, L.; De Oliveira, C.A.M. The effects of calorie-matched high-fat diet consumption on spontaneous physical activity and development of obesity. Life Sci. 2017, 179, 30–36. [Google Scholar] [CrossRef]
- Kleinert, M.; Clemmensen, C.; Hofmann, S.M.; Moore, M.C.; Renner, S.; Woods, S.C.; Huypens, P.; Beckers, J.; De Angelis, M.H.; Schürmann, A.; et al. Animal models of obesity and diabetes mellitus. Nat. Rev. Endocrinol. 2018, 14, 140–162. [Google Scholar] [CrossRef] [Green Version]
- Islam, S.; Wilson, R.D. Experimentally Induced Rodent Models of Type 2 Diabetes. Animal Models Diabetes Res. 2012, 933, 161–174. [Google Scholar] [CrossRef]
- Kanuri, G.; Bergheim, I. In Vitro and in Vivo Models of Non-Alcoholic Fatty Liver Disease (NAFLD). Int. J. Mol. Sci. 2013, 14, 11963–11980. [Google Scholar] [CrossRef] [Green Version]
- Warden, C.H.; Fisler, J.S. Comparisons of Diets Used in Animal Models of High-Fat Feeding. Cell Metab. 2008, 7, 277. [Google Scholar] [CrossRef] [Green Version]
- Bullen, J.W.; Bluher, S.; Kelesidis, T.; Mantzoros, C.S. Regulation of adiponectin and its receptors in response to development of diet-induced obesity in mice. Am. J. Physiol. Endocrinol. Metab. 2007, 292, E1079–E1086. [Google Scholar] [CrossRef]
- Kim, S.; Sohn, I.; Ahn, J.-I.; Lee, K.-H.; Lee, Y.S.; Lee, Y.S. Hepatic gene expression profiles in a long-term high-fat diet-induced obesity mouse model. Gene 2004, 340, 99–109. [Google Scholar] [CrossRef]
- Flanagan, A.M.; Brown, J.L.; Santiago, C.A.; Aad, P.Y.; Spicer, L.J.; Spicer, M.T. High-fat diets promote insulin resistance through cytokine gene expression in growing female rats. J. Nutr. Biochem. 2008, 19, 505–513. [Google Scholar] [CrossRef]
- Xavier, G.D.S.; Hodson, D.J. Mouse models of peripheral metabolic disease. Best Pr. Res. Clin. Endocrinol. Metab. 2018, 32, 299–315. [Google Scholar] [CrossRef]
- Hintze, K.J.; Benninghoff, A.D.; Cho, C.E.; Ward, R.E. Modeling the Western Diet for Preclinical Investigations. Adv. Nutr. 2018, 9, 263–271. [Google Scholar] [CrossRef] [Green Version]
- Timmers, S.; Bosch, J.D.V.-V.D.; De Wit, N.; Schaart, G.; Van Beurden, D.; Hesselink, M.K.C.; Van Der Meer, R.; Schrauwen, P. Differential effects of saturated versus unsaturated dietary fatty acids on weight gain and myocellular lipid profiles in mice. Nutr. Diabetes 2011, 1, e11. [Google Scholar] [CrossRef] [Green Version]
- An, X.; Liu, J.; Li, Y.; Dou, Z.; Li, N.; Suo, Y.; Ma, Y.; Sun, M.; Tian, Z.; Xu, L. Chemerin/CMKLR1 ameliorates nonalcoholic steatohepatitis by promoting autophagy and alleviating oxidative stress through the JAK2-STAT3 pathway. Peptides 2020, 135, 170422. [Google Scholar] [CrossRef]
- Lu, Z.; Li, Y.; Syn, W.-K.; Li, A.-J.; Ritter, S.; Wank, S.A.; Lopes-Virella, M.F.; Huang, Y. GPR40-Deficiency Is Associated with Hepatic FAT/CD36 Upregulation, Steatosis, Inflammation and Cell Injury in C57BL/6 Mice. Am. J. Physiol. Endocrinol. Metab. 2020. [Google Scholar] [CrossRef]
- Sardi, C.; Martini, E.; Mello, T.; Camelliti, S.; Sfondrini, L.; Marcucci, F.; Kallikourdis, M.; Sommariva, M.; Rumio, C. Effect of acetylsalicylic acid on inflamed adipose tissue. Insulin resistance and hepatic steatosis in a mouse model of diet-induced obesity. Life Sci. 2020, 118618. [Google Scholar] [CrossRef]
- Shen, H.H.; Alex, R.; Bellner, L.; Raffaele, M.; Licari, M.; Vanella, L.; Stec, D.E.; Abraham, N.G. Milk thistle seed cold press oil attenuates markers of the metabolic syndrome in a mouse model of dietary-induced obesity. J. Food Biochem. 2020, e13522. [Google Scholar] [CrossRef] [PubMed]
- Peck, B.; Huot, J.; Renzi, T.; Arthur, S.; Turner, M.J.; Marino, J.S. Mice Lacking PKCtheta in Skeletal Muscle Have Reduced Intramyocellular Lipid Accumulation and Increased Insulin Responsiveness in Skeletal Muscle. Am. J. Physiol. Regul. Integr. Comp. Physiol. 2017, 314, 468–477. [Google Scholar] [CrossRef] [Green Version]
- Bezan, P.; Holland, H.; De Castro, G.S.; Cardoso, J.; Ovidio, P.; Calder, P.C.; Jordão, A. High Dose of A Conjugated Linoleic Acid Mixture Increases Insulin Resistance in Rats Fed Either A Low Fat or A High Fat Diet. Exp. Clin. Endocrinol. Diabetes 2018, 126, 379–386. [Google Scholar] [CrossRef] [Green Version]
- Baek, K.-W.; Gim, J.-A.; Park, J.-J. Regular moderate aerobic exercise improves high-fat diet-induced nonalcoholic fatty liver disease via monoacylglycerol O-acyltransferase 1 pathway suppression. J. Sport Health Sci. 2020, 9, 472–478. [Google Scholar] [CrossRef]
- Abdulmalek, S.A.; Fessal, M.; El-Sayed, M. Effective amelioration of hepatic inflammation and insulin response in high fat diet-fed rats via regulating AKT/mTOR signaling: Role of Lepidium sativum seed extracts. J. Ethnopharmacol. 2021, 266, 113439. [Google Scholar] [CrossRef] [PubMed]
- Saltiel, A.R.; Olefsky, J.M. Inflammatory mechanisms linking obesity and metabolic disease. J. Clin. Investig. 2017, 127, 1–4. [Google Scholar] [CrossRef]
- Inoue, M.; Ohtake, T.; Motomura, W.; Takahashi, N.; Hosoki, Y.; Miyoshi, S.; Suzuki, Y.; Saito, H.; Kohgo, Y.; Okumura, T. Increased expression of PPARgamma in high fat diet-induced liver steatosis in mice. Biochem. Biophys. Res. Commun. 2005, 336, 215–222. [Google Scholar] [CrossRef] [Green Version]
- Wikan, N.; Tocharus, J.; Sivasinprasasn, S.; Kongkaew, A.; Chaichompoo, W.; Suksamrarn, A.; Tocharus, C. Capsaicinoid nonivamide improves nonalcoholic fatty liver disease in rats fed a high-fat diet. J. Pharmacol. Sci. 2020, 143, 188–198. [Google Scholar] [CrossRef]
- Ramalho, L.; Da Jornada, M.N.; Antunes, L.D.C.; Hidalgo, M.P.L. Metabolic disturbances due to a high-fat diet in a non-insulin-resistant animal model. Nutr. Diabetes 2017, 7, e245. [Google Scholar] [CrossRef] [Green Version]
- Erion, D.M.; Park, H.-J.; Lee, H.-Y. The role of lipids in the pathogenesis and treatment of type 2 diabetes and associated co-morbidities. BMB Rep. 2016, 49, 139–148. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Finucane, O.M.; Lyons, C.L.; Murphy, A.M.; Reynolds, C.M.; Klinger, R.; Healy, N.P.; Cooke, A.A.; Coll, R.C.; McAllan, L.; Nilaweera, K.N.; et al. Monounsaturated Fatty Acid–Enriched High-Fat Diets Impede Adipose NLRP3 Inflammasome–Mediated IL-1β Secretion and Insulin Resistance Despite Obesity. Diabetes 2015, 64, 2116–2128. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Shen, H.H.; Peterson, S.J.; Bellner, L.; Choudhary, A.; Levy, L.; Gancz, L.; Sasson, A.; Trainer, J.; Rezzani, R.; Resnick, A.; et al. Cold-Pressed Nigella Sativa Oil Standardized to 3% Thymoquinone Potentiates Omega-3 Protection against Obesity-Induced Oxidative Stress, Inflammation, and Markers of Insulin Resistance Accompanied with Conversion of White to Beige Fat in Mice. Antioxidants 2020, 9, 489. [Google Scholar] [CrossRef]
- Strissel, K.J.; Stancheva, Z.; Miyoshi, H.; Perfield, J.W.; DeFuria, J.; Jick, Z.; Greenberg, A.S.; Obin, M.S. Adipocyte death, adipose tissue remodeling, and obesity complications. Diabetes 2007, 56, 2910–2918. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lyons, C.L.; Kennedy, E.B.; Roche, H.M. Metabolic Inflammation-Differential Modulation by Dietary Constituents. Nutrition 2016, 8, 247. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Lu, X.; Li, X.; Du, P.; Qin, G. High-fat diet triggers obesity-related early infiltration of macrophages into adipose tissue and transient reduction of blood monocyte count. Mol. Immunol. 2020, 117, 139–146. [Google Scholar] [CrossRef] [PubMed]
- Hu, Q.; Lu, Y.; Hu, F.; He, S.; Xu, X.; Niu, Y.; Zhang, H.; Li, X.; Su, Q. Resistant dextrin reduces obesity and attenuates adipose tissue inflammation in high-fat diet-fed mice. Int. J. Med Sci. 2020, 17, 2611–2621. [Google Scholar] [CrossRef]
- Eom, J.; Thomas, S.S.; Sung, N.-Y.; Kim, D.-S.; Cha, Y.-S.; Kim, K.-A. Abeliophyllum distichum Ameliorates High-Fat Diet-Induced Obesity in C57BL/6J Mice by Upregulating the AMPK Pathway. Nutrients 2020, 12, 3320. [Google Scholar] [CrossRef]
- Liang, H.; Jiang, F.; Cheng, R.; Luo, Y.; Wang, J.; Luo, Z.; Li, M.; Shen, X.; He, F. A high-fat diet and high-fat and high-cholesterol diet may affect glucose and lipid metabolism differentially through gut microbiota in mice. Exp. Anim. 2020, 20. [Google Scholar] [CrossRef]
- Kim, S.-Y.; Lee, M.-S.; Chang, E.; Jung, S.; Ko, H.; Lee, E.; Lee, S.; Kim, C.-T.; Kim, I.-H.; Kim, Y. Tartary Buckwheat Extract Attenuated the Obesity-Induced Inflammation and Increased Muscle PGC-1a/SIRT1 Expression in High Fat Diet-Induced Obese Rats. Nutrition 2019, 11, 654. [Google Scholar] [CrossRef] [Green Version]
- Vasconcelos, R.P.; Peixoto, M.S.; De Oliveira, K.A.; Ferreira, A.C.F.; Coelho-De-Souza, A.N.; Carvalho, D.P.; De Oliveira, A.C.; Fortunato, R.S. Sex differences in subcutaneous adipose tissue redox homeostasis and inflammation markers in control and high-fat diet fed rats. Appl. Physiol. Nutr. Metab. 2019, 44, 720–726. [Google Scholar] [CrossRef] [PubMed]
- Grycel, S.; Markowski, A.R.; Hady, H.R.; Zabielski, P.; Kojta, I.; Imierska, M.; Górski, J.; Błachnio-Zabielska, A. Metformin treatment affects adipocytokine secretion and lipid composition in adipose tissues of diet-induced insulin-resistant rats. Nutrition 2019, 63, 126–133. [Google Scholar] [CrossRef] [PubMed]
- Kolahdouzi, S.; Talebi-Garakani, E.; Hamidian, G.; Safarzade, A. Exercise training prevents high-fat diet-induced adipose tissue remodeling by promoting capillary density and macrophage polarization. Life Sci. 2019, 220, 32–43. [Google Scholar] [CrossRef]
- Ellenbroek, J.H.; Töns, H.A.; De Graaf, N.; Loomans, C.J.; Engelse, M.A.; Vrolijk, H.; Voshol, P.J.; Rabelink, T.J.; Carlotti, F.; De Koning, E.J.P. Topologically Heterogeneous Beta Cell Adaptation in Response to High-Fat Diet in Mice. PLoS ONE 2013, 8, e56922. [Google Scholar] [CrossRef] [Green Version]
- Faleck, D.M.; Ali, K.; Roat, R.; Graham, M.J.; Crooke, R.M.; Battisti, R.; Garcia, E.; Ahima, R.S.; Imai, Y. Adipose differentiation-related protein regulates lipids and insulin in pancreatic islets. Am. J. Physiol. Endocrinol. Metab. 2010, 299, E249–E257. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gupta, D.; Jetton, T.L.; LaRock, K.; Monga, N.; Satish, B.; Lausier, J.; Peshavaria, M.; Leahy, J.L. Temporal characterization of beta cell-adaptive and -maladaptive mechanisms during chronic high-fat feeding in C57BL/6NTac mice. J. Biol. Chem. 2017, 292, 12449–12459. [Google Scholar] [CrossRef] [Green Version]
- Li, H.; Park, H.-M.; Ji, H.-S.; Han, J.; Kim, S.K.; Park, H.-Y.; Jeong, T.-S. Phenolic-enriched blueberry-leaf extract attenuates glucose homeostasis, pancreatic β-cell function, and insulin sensitivity in high-fat diet–induced diabetic mice. Nutr. Res. 2020, 73, 83–96. [Google Scholar] [CrossRef]
- Seferovic, M.D.; Beamish, C.A.; Mosser, R.E.; Townsend, S.; Pappan, K.; Poitout, V.; Aagaard, K.M.; Gannon, M.A. Increases in bioactive lipids accompany early metabolic changes associated with β-cell expansion in response to short-term high-fat diet. Am. J. Physiol. Endocrinol. Metab. 2018, 315, E1251–E1263. [Google Scholar] [CrossRef]
- Ye, T.; Chen, Y.-H.; Gao, J.-H.; Wang, X.-X.; Qiang, O.; Tang, C.-W.; Liu, R. Effect of octreotide on pancreatic fibrosis in rats with high-fat diet-induced obesity. Int. J. Clin. Exp. Pathol. 2018, 11, 4784–4794. [Google Scholar] [CrossRef]
- Sun, Q.; Nie, S.; Wang, L.; Yang, F.; Meng, Z.; Xiao, H.; Xiang, B.; Li, X.; Fu, X.; Wang, S. Factors that Affect Pancreatic Islet Cell Autophagy in Adult Rats: Evaluation of a Calorie-Restricted Diet and a High-Fat Diet. PLoS ONE 2016, 11, e0151104. [Google Scholar] [CrossRef] [Green Version]
- Teleman, A. Faculty Opinions recommendation of Glutamine oxidation maintains the TCA cycle and cell survival during impaired mitochondrial pyruvate transport. Fac. Opin. 2015, 56, 414–424. [Google Scholar] [CrossRef] [Green Version]
- Abdel-Hamid, A.A.M.; Firgany, A.E.-D.L. Correlation between pancreatic mast cells and the low grade inflammation in adipose tissue of experimental prediabetes. Acta Histochem. 2019, 121, 35–42. [Google Scholar] [CrossRef] [PubMed]
- Butler, A.E.; Janson, J.; Bonner-Weir, S.; Ritzel, R.A.; Rizza, R.A.; Butler, P.C. Cell Deficit and Increased -Cell Apoptosis in Humans With Type 2 Diabetes. Diabetes 2003, 52, 102–110. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Parilla, J.H.; Willard, J.R.; Barrow, B.M.; Zraika, S. A Mouse Model of Beta-Cell Dysfunction as Seen in Human Type 2 Diabetes. J. Diabetes Res. 2018, 2018, 6106051. [Google Scholar] [CrossRef] [Green Version]
- Lu, B.; Wu, H.; Gu, P.; Du, H.; Shao, J.; Wang, J.; Zou, D. Improved glucose-stimulated insulin secretion by intra-islet inhibition of protein-tyrosine phosphatase 1B expression in rats fed a high-fat diet. J. Endocrinol. Investig. 2011, 35, 63–70. [Google Scholar]
- Cordner, Z.A.; Tamashiro, K.L. Effects of high-fat diet exposure on learning & memory. Physiol. Behav. 2015, 152 Pt B, 363–371. [Google Scholar] [CrossRef] [Green Version]
- Kanoski, S.E.; Davidson, T.L. Western diet consumption and cognitive impairment: Links to hippocampal dysfunction and obesity. Physiol. Behav. 2011, 103, 59–68. [Google Scholar] [CrossRef] [Green Version]
- Beilharz, J.E.; Maniam, J.; Morris, M.J. Diet-Induced Cognitive Deficits: The Role of Fat and Sugar, Potential Mechanisms and Nutritional Interventions. Nutrients 2015, 7, 6719–6738. [Google Scholar] [CrossRef] [Green Version]
- Chesnokova, V.; Pechnick, R.N.; Wawrowsky, K. Chronic peripheral inflammation, hippocampal neurogenesis, and behavior. Brain Behav. Immun. 2016, 58, 1–8. [Google Scholar] [CrossRef] [Green Version]
- Kowiański, P.; Lietzau, G.; Czuba, E.; Waśkow, M.; Steliga, A.; Moryś, J. BDNF: A Key Factor with Multipotent Impact on Brain Signaling and Synaptic Plasticity. Cell. Mol. Neurobiol. 2018, 38, 579–593. [Google Scholar] [CrossRef]
- Marino, J.S.; Xu, Y.; Hill, J.W. Central insulin and leptin-mediated autonomic control of glucose homeostasis. Trends Endocrinol. Metab. 2011, 22, 275–285. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lizarbe, B.; Soares, A.F.; Larsson, S.; Duarte, J.M.N.; Lizarbe, B.; Soares, A.F.; Larsson, S.; Duarte, J.M.N. Neurochemical Modifications in the Hippocampus, Cortex and Hypothalamus of Mice Exposed to Long-Term High-Fat Diet. Front. Neurosci. 2019, 12, 985. [Google Scholar] [CrossRef] [PubMed]
- Klein, C.; Jonas, W.; Wiedmer, P.; Schreyer, S.; Akyüz, L.; Spranger, J.; Hellweg, R.; Steiner, B. High-fat Diet and Physical Exercise Differentially Modulate Adult Neurogenesis in the Mouse Hypothalamus. Neuroscience 2019, 400, 146–156. [Google Scholar] [CrossRef]
- Ruegsegger, G.N.; Vanderboom, P.M.; Dasari, S.; Klaus, K.A.; Kabiraj, P.; McCarthy, C.B.; Lucchinetti, C.F.; Nair, K.S. Exercise and metformin counteract altered mitochondrial function in the insulin-resistant brain. JCI Insight 2019, 4. [Google Scholar] [CrossRef] [PubMed]
- Hassan, A.M.; Mancano, G.; Kashofer, K.; Fröhlich, E.E.; Matak, A.; Mayerhofer, R.; Reichmann, F.; Olivares, M.; Neyrinck, A.M.; Delzenne, N.M.; et al. High-fat diet induces depression-like behaviour in mice associated with changes in microbiome, neuropeptide Y, and brain metabolome. Nutr. Neurosci. 2019, 22, 877–893. [Google Scholar] [CrossRef] [Green Version]
- Scherer, T.; Lindtner, C.; Zielinski, E.; O’Hare, J.; Filatova, N.; Buettner, C. Short Term Voluntary Overfeeding Disrupts Brain Insulin Control of Adipose Tissue Lipolysis*. J. Biol. Chem. 2012, 287, 33061–33069. [Google Scholar] [CrossRef] [Green Version]
- Top, M.V.D.; Zhao, F.-Y.; Viriyapong, R.; Michael, N.J.; Munder, A.C.; Pryor, J.T.; Renaud, L.P.; Spanswick, D. The impact of ageing, fasting and high-fat diet on central and peripheral glucose tolerance and glucose-sensing neural networks in the arcuate nucleus. J. Neuroendocr. 2017, 29, e12528. [Google Scholar] [CrossRef]
- Fu, Z.; Wu, J.; Nesil, T.; Li, M.D.; Aylor, K.W.; Liu, Z. Long-term high-fat diet induces hippocampal microvascular insulin resistance and cognitive dysfunction. Am. J. Physiol. Endocrinol. Metab. 2017, 312, E89–E97. [Google Scholar] [CrossRef]
- Greenwood, E.C.; Winocur, G. Cognitive impairment in rats fed high-fat diets: A specific effect of saturated fatty-acid intake. Behav. Neurosci. 1996, 110, 451–459. [Google Scholar] [CrossRef]
- Ribeiro, M.; Castelhano, J.; Petrella, L.; Sereno, J.; Rodrigues, T.; Neves, C.; Letra, L.; Baptista, F.I.; Seiça, R.; Matafome, P.; et al. High-fat diet induces a neurometabolic state characterized by changes in glutamate and N-acetylaspartate pools associated with early glucose intolerance: An in vivo multimodal MRI study. J. Magn. Reson. Imaging 2018, 48, 757–766. [Google Scholar] [CrossRef]
- Moffett, J.R.; Arun, P.; Ariyannur, P.S.; Namboodiri, A.M.A. N-Acetylaspartate reductions in brain injury: Impact on post-injury neuroenergetics, lipid synthesis, and protein acetylation. Front. Neuroenerg. 2013, 5, 11. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- van der Graaf, M.; Janssen, S.W.; van Asten, J.J.; Hermus, A.R.; Sweep, C.G.; Pikkemaat, J.A.; Martens, G.J.; Heerschap, A. Metabolic profile of the hippocampus of Zucker Diabetic Fatty rats assessed by in vivo 1H magnetic resonance spectroscopy. NMR Biomed. 2004, 17, 405–410. [Google Scholar] [CrossRef] [PubMed]
- Bouchard-Mercier, A.; Rudkowska, I.; Lemieux, S.; Couture, P.; Vohl, M.-C. The metabolic signature associated with the Western dietary pattern: A cross-sectional study. Nutr. J. 2013, 12, 158. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Tsai, S.-F.; Chen, Y.-W.; Kuo, Y.-M. High-fat diet reduces the hippocampal content level of lactate which is correlated with the expression of glial glutamate transporters. Neurosci. Lett. 2018, 662, 142–146. [Google Scholar] [CrossRef]
- Krssak, M.; Petersen, K.F.; Dresner, A.; DiPietro, L.; Vogel, S.M.; Rothman, D.L.; Shulman, G.I.; Roden, M. Intramyocellular lipid concentrations are correlated with insulin sensitivity in humans: A 1 H NMR spectroscopy study. Diabetology 1999, 42, 113–116. [Google Scholar] [CrossRef] [Green Version]
- Jiang, L.Q.; de Castro Barbosa, T.; Massart, J.; Deshmukh, A.S.; Löfgren, L.; Duque-Guimaraes, D.E.; Ozilgen, A.; Osler, M.E.; Chibalin, A.V.; Zierath, J.R. Diacylglycerol kinase-delta regulates AMPK signaling, lipid metabolism, and skeletal muscle energetics. Am. J. Physiol. Endocrinol. Metab. 2016, 310, E51–E60. [Google Scholar] [CrossRef] [Green Version]
- Gao, Z.; Wang, Z.; Zhang, X.; Butler, A.A.; Zuberi, A.; Gawronska-Kozak, B.; Lefevre, M.; York, D.; Ravussin, E.; Berthoud, H.R.; et al. Inactivation of PKCtheta leads to increased susceptibility to obesity and dietary insulin resistance in mice. Am. J. Physiol. Endocrinol. Metab. 2007, 292, E84–E91. [Google Scholar] [CrossRef]
- Mormeneo, E.; Jimenez-Mallebrera, C.; Palomer, X.; De Nigris, V.; Vázquez-Carrera, M.; Orozco, A.; Nascimento, A.; Colomer, J.; Lerin, C.; Gómez-Foix, A.M. PGC-1α Induces Mitochondrial and Myokine Transcriptional Programs and Lipid Droplet and Glycogen Accumulation in Cultured Human Skeletal Muscle Cells. PLoS ONE 2012, 7, e29985. [Google Scholar] [CrossRef]
- De Mendonça, M.; De Sousa, É.; Da Paixão, A.O.; Dos Santos, B.A.; Spagnol, A.R.; Murata, G.M.; Araújo, H.N.; De Lima, T.I.; Guimarães, D.S.P.S.F.; Silveira, L.R.; et al. MicroRNA miR-222 mediates pioglitazone beneficial effects on skeletal muscle of diet-induced obese mice. Mol. Cell. Endocrinol. 2019, 501, 110661. [Google Scholar] [CrossRef]
- Gao, Z.; Wang, Z.; Zhang, X.; Butler, A.A.; Zuberi, A.; Gawronska-Kozak, B.; Lefevre, M.; York, D.; Ravussin, E.; Berthoud, H.R.; et al. Role of diacylglycerol activation of PKCtheta in lipid-induced muscle insulin resistance in humans. Proc. Natl. Acad. Sci. USA 2014, 111, 9597–9602. [Google Scholar]
- Bergman, B.C.; Hunerdosse, D.M.; Kerege, A.; Playdon, M.C.; Perreault, L. Localisation and composition of skeletal muscle diacylglycerol predicts insulin resistance in humans. Diabetology 2012, 55, 1140–1150. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kim, J.K.; Fillmore, J.J.; Sunshine, M.J.; Albrecht, B.; Higashimori, T.; Kim, D.-W.; Liu, Z.-X.; Soos, T.J.; Cline, G.W.; O’Brien, W.R.; et al. PKC knockout mice are protected from fat-induced insulin resistance. J. Clin. Investig. 2004, 114, 823–827. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Perreault, L.; Newsom, S.A.; Strauss, A.; Kerege, A.; Kahn, D.E.; Harrison, K.A.; Snell-Bergeon, J.K.; Nemkov, T.; D’Alessandro, A.; Jackman, M.R.; et al. Intracellular localization of diacylglycerols and sphingolipids influences insulin sensitivity and mitochondrial function in human skeletal muscle. JCI Insight 2018, 3. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Rudnicki, M.; Abdifarkosh, G.; Rezvan, O.; Nwadozi, E.; Roudier, E.; Haas, T.L. Female Mice Have Higher Angiogenesis in Perigonadal Adipose Tissue Than Males in Response to High-Fat Diet. Front. Physiol. 2018, 9, 1452. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kim, B.; Kwon, J.; Kim, M.-S.; Park, H.; Ji, Y.; Holzapfel, W.; Hyun, C.-K. Protective effects of Bacillus probiotics against high-fat diet-induced metabolic disorders in mice. PLoS ONE 2018, 13, e0210120. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Cao, Y.; Chang, S.; Dong, J.; Zhu, S.; Zheng, X.; Li, J.; Long, R.; Zhou, Y.; Cui, J.; Zhang, Y. Emodin ameliorates high-fat-diet induced insulin resistance in rats by reducing lipid accumulation in skeletal muscle. Eur. J. Pharmacol. 2016, 780, 194–201. [Google Scholar] [CrossRef]
- Andrich, D.E.; Melbouci, L.; Ou, Y.; Auclair, N.; Mercier, J.; Grenier, J.-C.; Lira, F.S.; Barreiro, L.B.; Danialou, G.; Comtois, A.-S.; et al. A Short-Term High-Fat Diet Alters Glutathione Levels and IL-6 Gene Expression in Oxidative Skeletal Muscles of Young Rats. Front. Physiol. 2019, 10, 372. [Google Scholar] [CrossRef] [Green Version]
- Andrich, D.E.; Ou, Y.; Melbouci, L.; Leduc-Gaudet, J.-P.; Auclair, N.; Mercier, J.; Secco, B.; Tomaz, L.M.; Gouspillou, G.; Danialou, G.; et al. Altered Lipid Metabolism Impairs Skeletal Muscle Force in Young Rats Submitted to a Short-Term High-Fat Diet. Front. Physiol. 2018, 9. [Google Scholar] [CrossRef]
- Pataky, M.W.; Wang, H.; Yu, C.S.; Arias, E.B.; Ploutz-Snyder, R.J.; Zheng, X.; Cartee, G.D. High-Fat Diet-Induced Insulin Resistance in Single Skeletal Muscle Fibers is Fiber Type Selective. Sci. Rep. 2017, 7, 13642. [Google Scholar] [CrossRef] [Green Version]
- Pataky, M.W.; Yu, C.S.; Nie, Y.; Arias, E.B.; Singh, M.; Mendias, C.L.; Ploutz-Snyder, R.J.; Cartee, G.D. Skeletal muscle fiber type-selective effects of acute exercise on insulin-stimulated glucose uptake in insulin-resistant, high-fat-fed rats. Am. J. Physiol. Endocrinol. Metab. 2019, 316, E695–E706. [Google Scholar] [CrossRef]
- Soares, A.F.; Duarte, J.M.N.; Gruetter, R. Increased hepatic fatty acid polyunsaturation precedes ectopic lipid deposition in the liver in adaptation to high-fat diets in mice. MAGMA 2017, 31, 341–354. [Google Scholar] [CrossRef] [Green Version]
- Lai, M.; Chandrasekera, P.C.; Barnard, N.D. You are what you eat, or are you? The challenges of translating high-fat-fed rodents to human obesity and diabetes. Nutr. Diabetes 2014, 4, e135. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kowalski, G.M.; Bruce, C.R. The regulation of glucose metabolism: Implications and considerations for the assessment of glucose homeostasis in rodents. Am. J. Physiol. Endocrinol. Metab. 2014, 307, E859–E871. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Burgess, S.M.; Jeffrey, F.M.H.; Storey, C.; Milde, A.; Hausler, N.; Merritt, M.E.; Mulder, H.; Holm, C.; Sherry, A.D.; Malloy, C.R. Effect of murine strain on metabolic pathways of glucose production after brief or prolonged fasting. Am. J. Physiol. Endocrinol. Metab. 2005, 289, E53–E61. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gannon, K.S.; Smith, J.C.; Henderson, R.; Hendrick, P. A system for studying the microstructure of ingestive behavior in mice. Physiol. Behav. 1992, 51, 515–521. [Google Scholar] [CrossRef]
- Ellacott, K.L.; Morton, G.J.; Woods, S.C.; Tso, P.; Schwartz, M.W. Assessment of Feeding Behavior in Laboratory Mice. Cell Metab. 2010, 12, 10–17. [Google Scholar] [CrossRef] [Green Version]
- Perelis, M.; Ramsey, K.M.; Marcheva, B.; Bass, J. Circadian Transcription from Beta Cell Function to Diabetes Pathophysiology. J. Biol. Rhythm. 2016, 31, 323–336. [Google Scholar] [CrossRef]
- Qian, J.; Yeh, B.; Rakshit, K.; Colwell, C.S.; Matveyenko, A.V. Circadian Disruption and Diet-Induced Obesity Synergize to Promote Development of β-Cell Failure and Diabetes in Male Rats. Endocrinology 2015, 156, 4426–4436. [Google Scholar] [CrossRef] [Green Version]
- Johnston, J.D.; Ordovás, J.M.; Scheer, F.A.; Turek, F.W. Circadian Rhythms, Metabolism, and Chrononutrition in Rodents and Humans. Adv. Nutr. 2016, 7, 399–406. [Google Scholar] [CrossRef] [Green Version]
- Luo, S.; Zhang, Y.; Ezrokhi, M.; Li, Y.; Tsai, T.-H.; Cincotta, A.H. Circadian peak dopaminergic activity response at the biological clock pacemaker (suprachiasmatic nucleus) area mediates the metabolic responsiveness to a high-fat diet. J. Neuroendocr. 2018, 30, e12563. [Google Scholar] [CrossRef] [Green Version]
- Vadiveloo, M.; Scott, M.; Quatromoni, P.; Jacques, P.; Parekh, N. Trends in dietary fat and high-fat food intakes from 1991 to 2008 in the Framingham Heart Study participants. Br. J. Nutr. 2014, 111, 724–734. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kalupahana, N.S.; Voy, B.H.; Saxton, A.M.; Moustaid-Moussa, N. Energy-Restricted High-Fat Diets Only Partially Improve Markers of Systemic and Adipose Tissue Inflammation. Obesity (Silver Spring) 2011, 19, 245–254. [Google Scholar] [CrossRef] [PubMed]
- Salinero, A.E.; Anderson, B.M.; Zuloaga, K.L. Sex differences in the metabolic effects of diet-induced obesity vary by age of onset. Int. J. Obes. 2018, 42, 1088–1091. [Google Scholar] [CrossRef] [PubMed]
| Source | Fat Source | Macronutrients (% kcal) | Duration | Strain; Sex | Tissue Findings | T2D Status |
|---|---|---|---|---|---|---|
| [19] | (a) Cocoa butter (b) Palm oil (c) Olive oil (d) Safflower oil | 45% fat 20% protein 35% carbohydrate All diet formulations maintained same ratio | 8 weeks | C57BL/6 mice; Male | ↑ Liver TAG in palm and olive oil groups | ↑ Body weight in palm oil compared to cocoa butter. ↓ Glucose tolerance: cocoa, palm and safflower oils |
| [6] | Hydrogenated coconut oil | 59% fat 15% protein 26% carbohydrate | 20 weeks | C57BL/6 mice; Male | ↑ FABP mRNA ↑ Inflammation ↑ Increased BTNL2 | ↑ Fat mass ↓ Insulin sensitivity ↑ Plasma insulin |
| [22] | Lard and Soybean oil | 60% fat 20% protein 20% carbohydrate | 20 weeks | C57BL/6 mice; Female | ↑ Lipid accumulation ↑ Hepatic steatosis | ↑ Body weight ↓ Glucose tolerance ↑Serum insulin ↑ Serum glucose |
| [20,21,23,24] | Lard and Soybean oil | 60% fat ~20% protein ~20% carbohydrate | 8 weeks [23] 15 weeks [24] 18 weeks [20] 20 weeks [21] | C57BL/6 mice; Male | ↑ Inflammation ↑ Lipid accumulation ↑ Fibrosis | ↑ Body weight ↑ Serum glucose ↑ Serum insulin ↑ HOMA-IR |
| [25] | Lard | 45% fat 30% protein 25% carbohydrate | 4 weeks | Wister rats; Male | ↑ Liver mass ↑ Liver TAG ↑ Hepatic steatosis | ↑ Body weight ↓Insulin sensitivity ←→ Plasma glucose |
| [26] | Lard and Soybean oil | 45% fat 20% protein 35% carbohydrate | 12 weeks | Sprague-Dawley rats; Male | ↑ Lipid accumulation ↑ Lipogenic gene and protein expression | ↑ Body weight |
| [27] | Butter | 58% fat 25% protein 17% carbohydrate | 18 weeks | Sprague-Dawley rats; Male | ↑ Inflammation ↑ Lipid accumulation ↓ Insulin signaling ↑ Hepatic necrosis ↑ Oxidative stress | ↑ Body weight ↑ Serum glucose ↑ Serum insulin ↑ HOMA-IR |
| Source | Fat Source | Macronutrients (% kcal) | Duration | Strain; Sex | Findings | T2D Status |
|---|---|---|---|---|---|---|
| [38] | Lard and Soybean oil | 45% fat 20% protein 35% carbohydrate | 12 weeks | C57BL/6; Male | ↑ Body weight ↑ Epididymal fat mass and adipocyte size ↑ Inflammation | ↑ Body weight ↑ Blood glucose |
| [33] | (1) Palm oil (2) Sunflower oil | (1) 45% fat 20% protein 35% carbohydrate (2) 45% fat 20% protein35% carbohydrate | 24 weeks | C57BL/6; Male | ↓ SFA: Adipose insulin signaling ↑ SFA: Inflammation ↑ SFA: Adipocyte size ↑ SFA and MUFA: Epididymal, visceral, subcutaneous, and perirenal fat pad mass | ↓ Palm and sunflower oils: Glucose tolerance ↑ Sunflower oil: Hyperinsulinemia ↑ Palm oil: Hyperinsulinemia above sunflower oil |
| [37,39] | Lard and Soybean oil | 60% fat 20% protein 20% carbohydrate | 7 to 8 weeks | C57BL/6; Male | ↑ Subcutaneous fat ↑ Visceral fat ↑ Adipocyte size ↑ Lipogenic gene expression | ↑ Body weight ↑ Serum glucose |
| [40] | Lard and Soybean oil | 60% fat 20% protein 20% carbohydrate | 12 weeks | C57BL/6; female | ↑ Perirenal fat ↑ Gonadal fat ↑ Mesenteric fat | ↑ Body weight ←→ Glucose tolerance |
| [35] | Lard and Soybean oil | 60% fat 20% protein20% carbohydrate | 20 weeks | C57BL/6; Male | ↑ Adipose weight ↑ Adipocyte size peaks at 12 weeks ↑ Adipocyte death peaks at 16 weeks ↑ Inflammation | ↑ Body weight ↑ Serum insulin by 8 weeks ↑ Insulin resistance by 8 weeks ↑ HOMA-IR by 8 weeks |
| [41] | Lard and Soybean oil | 45% fat 20% protein 35% carbohydrate | 13 weeks | Sprague-Dawley rats; Male | ↑ Retroperitoneal and epididymal fat mass ↑ Adipocyte size ↑ Adipogenic gene expression ↑ Macrophage accumulation ↑ Inflammation | ↑ Body weight |
| [42] | Unknown | 57% fat 10% protein 31% carbohydrate | 11 weeks | Wister rats; Male and Female | ↑ Subcutaneous and retroperitoneal fat mass and adipocyte diameter in males and females ↑ Oxidative stress in males only ↑ Subcutaneous fat inflammation in males only | ↑ Body weight males only |
| [43] | Lard and Soybean oil | 60% fat 20% protein 20% carbohydrate | 8 weeks | Wister rats; Male | ↑ Ceramide content ↑ DAG content ↑ Plasma free fatty acids | ←→ Body weight ↑ Plasma glucose ↑ Plasma insulin ↑ HOMA-IR |
| [44] | UFA Mix: Sheep rump fat | 62.1% fat 16% protein 28.2% fat | 20 weeks | Wister rats; Males | ↑ Inguinal, mesenteric, epididymal, retroperitoneal, and perirenal fat mass ↓ Capillary density ↑ Increased macrophage crown-like structures | ↑ Body weight ↑ Blood glucose ↑ HOMA-IR ←→ Insulin |
| Source | Fat Source | Macronutrients (% kcal) | Duration | Strain; Sex | Findings | T2D Status |
|---|---|---|---|---|---|---|
| [45] | Lard and Soybean oil | 45% fat 20% protein 35% carbohydrate | 6 weeks | C57BL/6; Male | ↑ Beta cell proliferation in splenic region ↑ Increased insulin secretion from isolated islets | ↑ Body weight ↑ Plasma insulin ↓ Glucose tolerance ↓ Insulin tolerance |
| [46] | Lard and Soybean oil | 45% fat 20% protein 35% carbohydrate | 12 weeks | C57BL/6; Male | ↑ Lipid accumulation in acinar cells ↑ Adipose differentiation-related protein (ADFP) | ↑ Body weight ↑ Serum insulin ↑ Blood glucose |
| [48] | Lard and Soybean oil | 60% fat 20% protein 20% carbohydrate | 8 weeks | C57BL/6; Male | ↑ Islet size ↑ Islet Insulin | ↑ Body weight ↑ Blood glucose ↑ HbA1c ↑ Serum Insulin ↑ HOMA-IR |
| [53] | Not reported | 46% fat 20.3% protein 24% carbohydrate | 12 weeks | Sprague-Dawley rats; Male | ↑ Mast cell accumulation ↑ Islet area and proliferation ↑ β and α cell area | ↑ Plasma insulin ↓ Glucose tolerance |
| [56] | Not reported | 60% fat 18% protein 22% carbohydrate | 8 weeks | Sprague-Dawley rats; Male | Glucose-stimulated islet insulin secretion | Glucose tolerance |
| [51] | Not reported | 66.43% fat 18.08% protein 15.48% carbohydrate | 8 and 16 weeks | Sprague-Dawley rats; Male | ↑ Islet cell insulin at 8 and 16 weeks ↑ Glucagon at 16 weeks ↑ β and α cell area at 16 weeks ↑ β cell autophagy at 16 weeks | ↑ Body weight at 16 weeks ↑ Plasma glucose at 16 weeks ↑ Serum insulin at 8 and 16 weeks ↑ HOMA-IR at 8 and 16 weeks |
| Source | Fat Source | Macronutrients (% kcal) | Duration | Strain; Sex | Findings | T2D Status |
|---|---|---|---|---|---|---|
| [63] | Lard and soybean oil | 10, 45, or 60% fat 20% protein 70, 35, or 20 % carbohydrates | 24 weeks | C57BL/6; Male | 45% and 60% fat ↓ Spontaneous activity ↓ Locomotion ↑ Neuroinflammation 60% fatAltered metabolite profile | 45% and 60% fat ↑ Body weight: 60% > 45% ↑ Plasma glucose: 60% > 45% ↑ Plasma insulin ↑ Plasma Leptin ↓ Glucose tolerance: 60% > 45% at 2 h |
| [65] | Lard and soybean oil | 60% fat 20% protein 20% carbohydrates | 4 weeks | C57BL/6; Male | ↓ Insulin signaling in isolated hypothalamic and hippocampal neurons ↓ Mitochondrial function ↑ Oxidative stress | ↑ Body weight ↑ Fat mass ↑ Plasma glucose ↑ Plasma insulin ↑ HOMA-IR |
| [66] | Palm oil | 60% fat 16% protein 24% carbohydrates | 8 weeks | C57BL/6; Male | ↓ Socialization behavior ↑ Disruption of normal circadian feeding Hypothalamic NPY expression | ↑ Body weight |
| [64] | Lard and soybean oil | 60% fat 20% protein 20% carbohydrates | 12 weeks | C57BL/6; Female | ↑ Microglia in hypothalamic arcuate nucleus ↑ Trend in neurogenesis of POMC neurons | ↑ Body weight ↑ Fat mass |
| [67] | Lard | 10 % added to standard diet | 3 days | Sprague-Dawley Rats; Male | ↓ Hypothalamic insulin sensitivity ↓ Hypothalamic insulin-stimulated adipose lipolysis ↓ Hypothalamic insulin-stimulated hepatic glucose production | ←→ Body weight ←→ Plasma glucose ←→ Plasma insulin |
| [68] | Not reported | 45% fat 20% protein 35% carbohydrates | 20 weeks | Wistar-Han Rats; Male | ←→ CSF glucose ↓ CSF glucose tolerance | ↑ Body weight Plasma glucose ↓ Glucose tolerance |
| [69] | Lard and soybean oil | 60% fat 20% protein 20% carbohydrates | 24 weeks | Sprague-Dawley Rats; Male | ↓ Cognitive function ↓ Insulin-stimulated hippocampal perfusion ↓ Hippocampal insulin signaling ↓ Hippocampal eNOS | ↓ Whole body glucose disposal ↑ Plasma insulin |
| Source | Fat Source | Macronutrients (%kcal) | Duration | Strain; Sex | Findings | T2D Status |
|---|---|---|---|---|---|---|
| [19] | (a) Cocoa butter (b) Palm oil (c) Olive oil (d) Safflower oil | 45% fat 20% protein 35% carbohydrateAll diet formulations maintained same ratio | 8 weeks | C57BL/6; Male | ↑ Gastrocnemius TAG and DAG: cocoa butter, palm oil, and olive oil | ↑ Body weight in palm oil compared to cocoa butter. ↑ Glucose tolerance: cocoa, palm and safflower oils |
| [85] | Coconut oil and soybean oil | 58% fat 17% protein 25% carbohydrate | 16 weeks | FVB; B6;Male and Female | ↓ Muscle insulin sensitivity: Males and Females | ↑ Body weight: Males only ↑ Plasma glucose: Males only ↓ Glucose and insulin tolerance: Males only |
| [6,80] | Hydrogenated coconut oil | 59% fat 15% protein 26% carbohydrate | 8 weeks [83] 20 weeks [6] | C57BL/6; Male | ↑ Fatty acid transport ↑ Lipogenesis ↑ Muscle Adipocyte differentiation ↓ Fatty acid oxidation ↓ Mitochondrial function | ↑ Body weight ↑ Blood glucose ↑ Plasma insulin ↓ Insulin sensitivity |
| [86] | Lard and soybean oil | 60% fat 20% protein 20% carbohydrate | 15 weeks | C57BL/6; Male | ↓ Insulin sensitivity ↑ Muscle weight ↑ Inflammatory mRNA profile | ↑ Body weight ↑ Serum insulin ↕ Glucose tolerance |
| [87] | Lard | 45% fat 20% protein 35% carbohydrate | 15 weeks | Sprague-Dawley rats; Male | ↑ Soleus lipid accumulation ↑ Protein and mRNA supporting fatty acid transport and lipogenesis | ↑ Body weight ↑ Plasma glucose ↑ Serum insulin ↑ HOMA-IR ↓ Insulin sensitivity |
| [88,89] | Lard and soybean oil | 60% fat 20% protein 20% carbohydrate | 2 weeks | Wister rats; Male | ←→ Soleus and extensor digitorum longus weight ←→ Soleus and extensor digitorum longus insulin signaling ↓ Soleus force production ↓ Soleus glutathione ↑ Soleus IL-6 mRNA ↑ Lipid droplet size in soleus ↑ Percentage of large lipid droplets in soleus ↑ Lipogenic mRNA in soleus | ←→ Body weight |
| [90,91] | Lard and soybean oil | 60% fat 20% protein 20% carbohydrate | 2 weeks | Wister rats; Male | ↓ Glucose uptake by isolated type IIAX, IIX, IIBX, and IIB fibers ↑ Lipid droplet density in type I, IIA, and IIAX fibers ↑ Lipid droplet size in type I and IIA fibers ↓ Insulin-stimulated glucose uptake in whole muscle ↓ Insulin-stimulated glucose uptake in isolated type IIA, IIAX, and IIB fibers | ←→ Body weight |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Stott, N.L.; Marino, J.S. High Fat Rodent Models of Type 2 Diabetes: From Rodent to Human. Nutrients 2020, 12, 3650. https://doi.org/10.3390/nu12123650
Stott NL, Marino JS. High Fat Rodent Models of Type 2 Diabetes: From Rodent to Human. Nutrients. 2020; 12(12):3650. https://doi.org/10.3390/nu12123650
Chicago/Turabian StyleStott, Nicole L., and Joseph S. Marino. 2020. "High Fat Rodent Models of Type 2 Diabetes: From Rodent to Human" Nutrients 12, no. 12: 3650. https://doi.org/10.3390/nu12123650





