Abstract
Nutritional guidelines suggest specific energy and protein requirements for patients with cancer. However, cancer patients, often malnourished, use self-made or web-based diets to ameliorate the prognosis of their disease. This review aimed to investigate the associations between post-diagnostic diet and prognostic outcomes in cancer patients. A systematic literature search was performed in Pubmed and Web of Science databases from inception to 30 October 2019, based on fixed inclusion and exclusion criteria. The risk of bias was assessed. A total of 29 prospective studies was identified. Breast (n = 11), colorectal (n = 9), prostate (n = 8) cancers are the most studied. Low- fat diet, healthy quality diet, regular consumption of fiber such as vegetables and high-quality protein intake are beneficial while Western diet (WD) and high consumption of saturated fats could be associated with a higher risk of mortality. Bladder (n = 1), gynecological (n = 1), lung, stomach, and pancreatic cancers still remain almost unexplored. This systematic review suggested that detrimental dietary patterns such as WD should be avoided but none of the food categories (meat, dairy products) should be eliminated in cancer patients’ diet. Further large prospective studies are needed to assess the role of post-diagnostic diet in patients with cancer.
1. Introduction
According to the World Health Organization (WHO), the cancer burden rose to 18.1 million new cases and 9.6 million cancer deaths in 2018 [1]. Convincing evidence supports a reduced risk of different types of cancer among healthy populations that follow specific dietary regimens [2,3,4]. Indeed, a recent meta-analysis suggested an association between healthy dietary patterns and decreased risk of colon and breast cancer [2]. Especially, adherence to the Mediterranean diet is associated with a lower incidence of several cancer types such as colorectal cancer [3]. The beneficial effects of Mediterranean diet are mainly driven by higher intakes of fruit, vegetables, and whole grains [4]. In contrast, evidence for the role of post-diagnostic diet in cancer survival remains limited. However, malnutrition, defined as a state resulting from lack of intake of nutrition that leads to altered body composition, is common among cancer patients due to the disease itself and oncologic treatments [5,6]. To prevent malnutrition, energy and protein requirements for cancer patients are largely widespread by international guidelines [7,8,9] but little is known about the food choices and dietary regimen a cancer patient should benefit from. In this context, cancer patients are often motivated to learn how food choices and dietary patterns can improve their nutritional status and response to treatment and reduce risk of cancer recurrence and cancer-specific mortality. Thus, web sources are full of fake prescriptions or confounding statements easily shared by patients without any endorsement by the scientific community [10]. Many “cancer diets” are often restrictive, avoiding a whole nutrient class (i.e., meat or dairy products) in the misleading belief that certain foods “feed the tumor” [10]. In the last decades, a growing number of prospective cohort studies [11,12,13,14,15,16] have investigated the association between post-diagnostic dietary patterns—such as Western diet (WD) and prudent diet (PD)—or food type—such as meat, dairy products, dietary fiber, nuts—and prognostic outcomes among patients with different cancers. This systematic review aims to investigate possible associations between dietary patterns/choices after cancer diagnosis and prognostic outcomes (i.e., mortality, cancer progression, and recurrence) in patients affected by main solid tumors.
2. Methods
This systematic review was performed according to the Cochrane Handbook for systematic reviews [17] and followed the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) statement [18].
2.1. Eligibility Criteria
Inclusion criteria were:
- Population: adult (≥18 years old) patients diagnosed with breast, gastrointestinal (gastric, pancreatic, colorectal), gynecological (uterine, cervical, ovarian, endometrial, vulvar), lung and urological (prostate, bladder) cancers.
- Exposure: any post-diagnostic dietary exposure such as dietary patterns or individual food components exposure (fruit, vegetables, dairy, meat, fish, cereals) or use of diet quality indices.
- Study design: prospective or retrospective cohort studies.
- Outcomes of interest: overall survival (OS) or all-cause mortality (ACM), cancer-specific mortality (CSM), death from a cause other than specific cancer, cancer progression, disease-free survival (DFS), cancer recurrence and recurrence-free survival (RFS).
Exclusion criteria were studies including patients with other types of cancer; reviews, comments, editorials, case series, or meeting abstracts.
2.2. Definitions of Outcomes
OS and ACM were defined as the time from cancer diagnosis to death from any cause. CSM was defined as the time from specific cancer diagnosis to death from this specific cancer. DFS was defined as the time from cancer diagnosis to tumor recurrence or death from any cause. RFS was defined as the time from the cancer diagnosis to tumor recurrence.
2.3. Data Sources and Search Strategy
The search was carried out on 30th October 2019 using two electronic databases, MEDLINE (via PubMed) and ISI Web of Science. The search strategy was limited to English language articles and there were no restrictions on the date of publication. The search string for each database is described in Table S1 (Supplementary File). The reference lists of retrieved articles were manually scrutinized to identify potentially relevant studies.
2.4. Study Selection
The study selection process was independently carried out by three reviewers (P.R.; E.R.; L.S.). All articles generated from the electronic search were imported into Mendeley© (Elsevier, Amsterdam, The Netherlands) a references’ management software, and duplicates were removed. Titles and abstracts of all records were screened for eligibility based on inclusion criteria. All titles assessed as ineligible were excluded. Differences in judgment during the selection process between the three reviewers were settled by discussion and consensus.
2.5. Data Extraction
Information was collected using an Excel© (Microsoft Office, Redmond, WA, USA) spreadsheet specifically developed for this study. Each full-text article was retrieved, and the articles deemed ineligible were excluded and the reasoning reported. Differences in judgment among reviewers were settled by discussion and consensus.
2.6. Quality Assessment
Two reviewers independently assessed the risk of bias of each included study using the Quality In Prognosis Studies (QUIPS) tool as described by Hayden et al. [19]. The QUIPS tool developed to assess the risk of bias in prognostic factor studies has six domains: study participation, study attrition, prognostic factor measurement, outcome measurement, study confounding, and statistical analysis and reporting. Each of the six domains was rated as having a ‘Low’, ‘Moderate’, or ‘High’ risk of bias. Subsequently, the overall risk of bias was established for each study. The overall risk of bias was considered low if ≤2 domains were rated a moderate risk of bias and all others were rated a low risk of bias. The overall risk of bias was considered moderate if >2 domains were rated a moderate risk of bias and all others were rated a low risk of bias. The overall risk of bias was considered high if ≥1 domain was rated the high risk of bias, irrespective of all other domains. Differences in judgment among reviewers were settled by discussion and consensus.
2.7. Data Synthesis
Because of the high heterogeneity of the studies, a systematic review was performed. Indeed, the dietary assessments, dietary factors/dietary patterns, and outcomes of each study were not comparable and consequently, a meta-analysis was unfeasible. The main results of the review were displayed on a summary of findings table. For each study, first author’s last name, year of publication, country of origin, types of outcome, sample size of the population, period time of diagnosis, mean follow-up duration, exposure assessment, diet-quality indices, dietary patterns or other dietary factors, adjustment covariates, and multivariate-adjusted risk estimates—hazards ratios (HR) or risk ratios (RR) for the highest vs. the lowest category with their corresponding 95% confidence intervals (CI) were reported.
3. Results
3.1. Study Selection
The flow diagram in Figure 1 displays the results of the literature search and study selection process. A total of 6372 publications were initially identified, 161 were duplicates. Hand searching allowed the identification of one additional study. Twenty-nine studies were identified for inclusion in the systematic review.
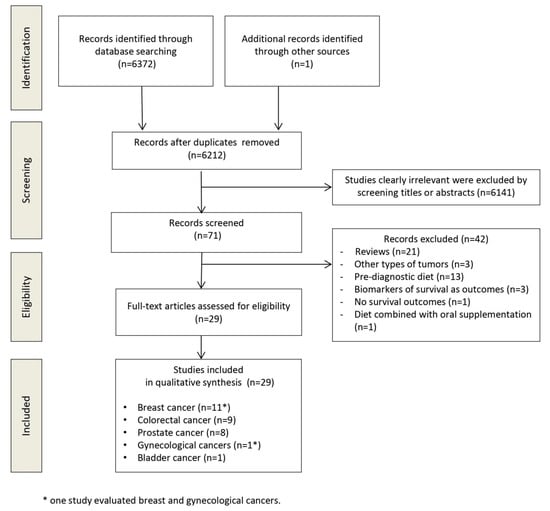
Figure 1.
Preferred reporting items for systematic reviews and meta-analyses (PRISMA) flow diagram.
3.2. Study Characteristics
Table 1 shows the main characteristics of the included studies [11,12,13,14,15,16,20,21,22,23,24,25,26,27,28,29,30,31,32,33,34,35,36,37,38,39,40,41,42]. All included studies are prospective. Eleven studies enrolled patients with breast cancer [10,19,20,21,22,23,24,25,26,27,28], 9 with colorectal cancer [11,12,29,30,31,32,33,34,35], 8 with prostate cancer [13,35,36,37,38,39,40,41], 1 with bladder cancer [16] and 1 with gynecological cancer [29] (ovarian, uterine and cervical cancers). The sample size ranged from 230 [29] to 9514 [26] patients. Twenty-three studies were performed in the USA, 1 in Japan [34], 1 in China [23], 1 in China and USA [26], 1 in the United Kingdom [16], 1 in Germany [15] and 1 in Denmark [28]. The median duration of follow-up ranged from 2 [14] to 28 years [40]. Eight [12,15,21,22,25,29,31,41] out of 29 studies assessed the influence of dietary patterns while all other studies assessed the effect of dietary factors. Twenty studies evaluated ACM, 17 CSM, 8 cancer recurrence, 4 cancer progression, 4 death from non-specific cancer cause, 3 OS, 3 DFS, and 3 RFS. Studies evaluated different dietary patterns, different diet quality indices, or individual dietary factors. The definitions of the dietary patterns and diet quality indices are detailed in Table S2 (Supplementary File).

Table 1.
Main characteristics of the included prospective studies.
3.3. Quality Assessment
According to the QUIPS tool, 13 [11,12,21,24,25,26,27,31,33,35,37,39,41] studies had a low overall risk of bias, 14 studies a moderate overall risk of bias [13,14,15,20,22,26,28,30,32,34,36,38,40,42] and 2 studies a high overall risk of bias [16,29]. The overall risk of bias of each study is presented in Table 2 and the risk of bias of each item is detailed in Table S3 (Supplementary file).

Table 2.
Summary of results of the included studies enrolling patients diagnosed with breast cancer. Statistically significant (p < 0.05) results, HR or RR with 95% CI, are reported.
Fourteen studies provided an adequate description of the source population, baseline study sample, recruitment, and inclusion/exclusion criteria for participants. For two studies [16,29], the risk of bias of study population was high due to a too small sample size. For fourteen studies [13,14,15,20,22,23,28,30,32,34,36,38,40,42], the risk of bias of study population had a moderate risk of bias mainly due to the missing of tumor stage data. Regarding study attrition, all studies had a low risk of bias, providing adequate response rate for study participants, adequate description of attempts to collect information on participants who dropped out and reasons for loss to follow-up. Regarding prognostic factor assessment, all studies had a moderate risk of bias. Indeed, included studies used either food frequency questionnaire (FFQ) or 24-h recall as a dietary assessment tool, providing recall bias in dietary intake assessment. In fact, FFQ or 24-h recall provided measurement errors mainly due to a reporting average intake over a long period of time with under- or over-estimated intake. For the majority of studies, the definitions of outcomes were clear and the methods of outcome measurement were valid and reliable. However, for five studies [21,22,25,31,36], despite a thorough review of records by physicians, some causes of death could have been misclassified. Regarding study confounding, the majority of studies (n = 19) had a moderate risk of bias. Indeed, some studies did not consider the pre-diagnostic diet as confounding factor [14,22,33,38,40,42], or treatment information on 25% of the cohort was missing [30], a too small number of confounders were considered [29,32,34,36], or inconsistent patterns of lifestyle factors between participants lowered the statistical power of the study [26]. Finally, three studies [23,28,29] had a moderate risk of bias in reporting of results.
3.4. Summary of Findings
3.4.1. Breast Cancer
Table 2 reports the statistically significant results (p-value < 0.05), HR or RR with 95% CI, of the included studies enrolling patients diagnosed with breast cancer.
Dietary Patterns
A cohort study [25] of 2729 women from the Nurses’ Health Study with invasive stage I–III breast cancer found, in simple and multivariate-adjusted analyses, no association of CSM with the post- diagnostic Modified Mediterranean Diet Score (MMDS). However, in a multivariate analysis, a higher MMDS was significantly associated with a lower risk of non-breast cancer death in breast cancer women with low physical activity (adjusted RR, 0.39, 95% CI 0.20–0.75) [25].
A recent study [29] of 110 breast cancer women found no significant association between Mediterranean Diet Score (MDS) and ACM. However, a higher Healthy Eating Index (HEI) score (≥70) was significantly associated with lower ACM (adjusted HR 0.49 95% CI 0.25–0.97) [29].
Two cohort studies of 2619 [21] and 1901 [22] breast cancer women assessed after diagnosis, WD and Prudent Diet (PD) in multivariate-adjusted analyses. A significant association between higher adherence to PD and lower risk of ACM (adjusted HR 0.57 95% CI 0.36–0.90) was found in one study [25]. As regards WD, no association was found with ACM or CSM in both studies. Moreover, both studies found a significant association between death from causes other than breast cancer and PD (respective adjusted RR: Q5 vs. Q1 0.54 95% CI 0.31–0.95 [21]; Q4 vs. Q1 HR 0.35 95% CI 0.17–0.73 [22]). In contrast, Kroenke et al. found a significant association between WD and this outcome (Q5 vs. Q1 adjusted RR 2.31, 95% CI 1.23–4.32) [21].
Dietary Factors
A large multi-center prospective cohort study including 4441 breast cancer women [24], after adjustment for factors at diagnosis, suggested that higher intake of saturated (Q5 vs. Q1 adjusted HR 1.41 95% CI 1.06–1.87) and trans fat (Q5 vs. Q1 adjusted HR 1.78 95% CI 1.35–2.32) in the post- diagnostic diet was significantly associated with a higher risk of ACM [24]. No significant association was observed between CSM and the respective intake of meat and dairy products.
A large prospective cohort study included 1982 women with diagnosed breast cancer and followed for 18 years: in multivariate analyses of diet after diagnosis, no significant association was found between low-fat and high-fat dairy intake and ACM [20]. Another large prospective cohort study [27] enrolling 1893 women diagnosed with early-stage invasive breast cancer suggested that, compared with the reference (0 to <0.5 servings/day), those consuming larger amounts of high-fat dairy (≥1.0 servings/day) had a higher risk of ACM (adjusted HR 1.64 95% CI 1.24–2.17) and death from non-breast cancer causes (adjusted HR 1.67 95% CI 1.13–2.47). Recently, a prospective cohort Danish study [28] suggested no associations between post-diagnostic intake of total dairy products (milk yogurt and cheese) and cancer recurrence, CSM, and ACM [28].
A large prospective study of more than 6000 breast cancer patients [11] observed a relationship between higher intake of protein and reduced risk of recurrence (Q4 vs. Q1 adjusted RR 0.75 95% CI 0.61–0.91), which was particularly significant for protein from animal sources (Q4 vs. Q1 adjusted RR 0.78 95% CI 0.63–0.95). Moreover, higher animal protein intake was correlated with a lower risk of CSM (Q4 vs. Q1 adjusted RR 0.77 95% CI 0.62–0.94) [11]. These findings confirmed the results of a large cohort study [20] of 1982 breast cancer women showing a significant association between ACM and total protein intake (Q5 vs. Q1 adjusted RR 0.65 95% CI 0.47–0.88).
In a large study of combined data on 9514 US and Chinese breast cancer women [26], post- diagnosis soy food consumption corresponding to about 10 mg isoflavones/day was associated with a non-significant reduced risk of CSM but a statistically significant reduced risk of disease recurrence (adjusted HR 0.75 95% CI 0.61–0.92). Another cohort study [23] of 5042 female Chinese breast cancer survivors showed that soy food intake, as measured by either soy protein or soy isoflavone intake, was inversely associated with ACM (Q4 vs. Q1 adjusted HR 0.67 95% CI 0.51–0.88) and recurrence (Q4 vs. Q1 adjusted HR 0.66 95% CI 0.52–0.84) [23].
Three studies [20,24,28] investigated post-diagnostic intake of whole-grain products and breast cancer prognosis, neither reporting any associations. However, specifically, Andersen et al. [32] found a significant association between post-diagnostic serving size increment of rye bread per day and higher risk of CSM (adjusted HR 1.29 95% CI 1.02–1.63), hypothetically due to the habit of the Danish population to eat rye bread with high-fat products such as butter, cheese and processed meat [28].
3.4.2. Colorectal Cancer
Table 3 reports the statistically significant results (p-value < 0.05), HR or RR with 95% CI, of the included studies enrolling patients diagnosed with colorectal cancer.

Table 3.
Summary of results of included studies regarding colorectal cancer. Statistically significant (p < 0.05) results (HR or RR; 95% CI) are reported.
Dietary Patterns
One prospective cohort of 1009 stage III colorectal cancer patients [12] reported significant worse OS (adjusted HR 2.32 95% CI 1.36–3.96), DFS (adjusted HR 3.25 95% CI 2.04–5.19) and RFS (adjusted HR 2.85 95% CI 1.75–4.63) in the highest quintile of WD compared to lowest one. Another cohort study [31] of 1201 colorectal cancer patients diagnosed with stage I–III colorectal did not show significant associations between WD and survival. Regarding PD, both studies found no significant association with prognostic endpoints.
One German study [15] of 1404 colorectal cancer patients evaluated MMDs and the Healthy Nordic diet index (HNDI). Higher MMDS was associated with lower ACM (adjusted HR 0.48 95% CI 0.32–0.74).
Dietary Factors
A recent study [33] examined marine ω-3 polyunsaturated fatty acids (PUFAs) and fish intake and survival endpoints among 1011 colorectal cancer patients enrolled in Cancer and Leukemia Group B (CALGB) 89,803 (Alliance) trial (an adjuvant chemotherapy trial) and found that high intake of marine ω-3 PUFAs was associated with longer DFS (adjusted HR 0.72 95% CI 0.54–0.97). Moreover, colorectal cancer patients who consumed dark fish ≥1/week experienced longer DFS (adjusted HR 0.65 95% CI 0.48–0.87), RFS (adjusted HR 0.61 95% CI 0.46–0.86), and OS (adjusted HR 0.68 95% CI 0.48–0.96) [33].
Regarding fiber intake, a Japan cohort study [34] of 5864 colorectal cancer patients demonstrated that a low consumption of green leafy vegetables was correlated with worse ACM (never-consumers vs. everyday consumers, adjusted HR 1.87 95% CI 1.22–2.88). Another cohort of 1575 colorectal cancer patients [35] analyzed total fiber, cereal fiber, vegetable fiber, fruit fiber, and whole-grain intakes after diagnosis. A 5 g/day increment of total fiber and cereal fiber significantly decreased ACM (adjusted HRs total fiber: 0.86 95% CI 0.65–0.93, cereal fiber: 0.78, 95% CI 0.68–0.90) and CSM (adjusted HRs total fiber: 0.78 95% CI 0.65–0.93, cereal fiber 0.67 95% CI 0.50–0.90) [42]. Moreover, a 5 g/day increment of vegetable fiber significantly correlated with ACM (adjusted HR 0.83 95% CI 0.72–0.96) and 20 g/day of whole-grain intake significantly correlated with CSM (adjusted HR 0.72 95% CI 0.59–0.88) [35].
Two observational cohort studies of 1201 [31] and 2315 [30] non-metastatic colorectal cancer patients did not observe any association between red and processed meat intake after diagnosis and both ACM and CSM. However, colorectal cancer patients with consistently high red and processed meat intake before and after diagnosis had a higher risk of CSM (RR 1.79 95% CI 1.11–2.89) compared with those with consistently low intake [30]. Moreover, the above-mentioned Japanese study [40] did not show any significant association between meat and ACM.
One study [32] evaluated associations of dairy product intakes after diagnosis with ACM and CSM among 1111 colorectal cancer patients. An inverse association with ACM was observed for post- diagnosis milk intake (RR 0.72 95% CI 0.55–0.94) [32].
The study of Fung et al. [31] of 1201 women diagnosed with stage I–III colorectal cancer found a higher ACM risk with the consumption of sugar-sweetened beverages and fruit juices (for each additional serving/day, adjusted HR 1.11 95% CI 1.01–1.23).
In a study [13] examining 826 patients with stage III colon cancer enrolled in the CALGB 89,803 trial, a higher nut intake (≥2 servings/week of total nut intake vs. never) was associated with a significant improvement in OS (adjusted HR 0.43 95% CI 0.25–0.74) and DFS (adjusted HR 0.58 95% CI 0.37–0.92). Moreover, a subgroup analysis revealed that higher tree nut intake (≥2 servings/week of total nuts intake vs. never) significantly improved OS (adjusted HR 0.47 95% CI 0.27–0.82) and DFS (adjusted HR 0.54 95% CI 0.34–0.85) [13]. Interestingly, the above-mentioned study of Fung et al. [31] found a significantly lower risk of CSM for each serving/day of nuts consumption (adjusted HR 0.69 95% CI 0.49–0.97).
3.4.3. Prostate Cancer
Table 4 reports the statistically significant results (p-value < 0.05), HR or RR with 95% CI, of the included studies enrolling patients diagnosed with prostate cancer.

Table 4.
Summary of results of included studies regarding prostate cancer. Statistically significant (p < 0.05) results (HR or RR; 95% CI) are reported.
Dietary Patterns
One prospective study examined post-diagnostic Mediterranean diet adherence in relation to CSM and ACM among 4538 men initially diagnosed with prostate cancer (clinical stage T1–T3a) [41]. Lower ACM was associated with greater adherence to the Mediterranean diet after diagnosis (high adherence vs. low adherence adjusted HR 0.78 95% CI 0.67–0.90) [41].
Regarding fat intake after diagnosis, a prospective study of 4577 men with non-metastatic prostate cancer suggested that higher ACM was associated with saturated fats (Q5 vs. Q1 adjusted HR 1.30 95% CI 1.05–1.60) and trans fats (Q5 vs. Q1 adjusted HR 1.25 95% CI 1.05–1.49) intakes after diagnosis [39]. Another study [42] on 926 men with non-metastatic prostate cancer confirmed the adverse effect of saturated fats on ACM and showed that men who obtained more of their daily calories from vegetable fats and less of their daily calories from carbohydrates had a 33% lower risk of ACM (adjusted HR 0.67 95% CI 0.47–0.96) [42].
Dietary Factors
Two studies of 1294 [14] and 1202 men with prostate cancer [36] assessed the association between the consumption of processed red meat (salami, sausage, bacon and hot dogs) after diagnosis and prostate cancer progression; both showed a non-significant increase in the risk of disease progression (p > 0.05) [14,36]. As regards poultry, one study [14] reported that consuming poultry after diagnosis is not significantly associated with prostate cancer progression. However, men who reported consuming higher amounts of poultry with skin after prostate cancer diagnosis had a significantly higher risk of prostate cancer progression (Q3 vs. Q1 adjusted HR 2.26 95% CI 1.36–3.76) [14].
A prospective study [37] including 3918 men with prostate cancer showed that total milk and dairy intakes after diagnosis were not associated with a greater risk of CSM at multivariate analysis. However, men with the highest versus lowest intake of whole milk had increased risk of cancer progression (Q5 vs. Q1 adjusted HR 2.15 95% CI 1.28–3.60) [37]. Song et al. [40] confirmed that higher whole milk intake was associated with worse cancer progression (>2.5 servings/day vs. ≤0.5 servings/day, adjusted HR 2.17 95% CI 1.34–3.51).
As regards fiber intake, a cohort study of 1560 non-metastatic patients with prostate cancer [38] observed that patients consuming ≥5.7 servings/day of cruciferous vegetables after diagnosis—such as broccoli, cauliflower, cabbage, brussels sprouts—had 59% lower risk of cancer progression compared to patients consuming ≤1.4 servings/day of these vegetables (adjusted HR 0.41 95% CI 0.22–0.76). No significant association was found between prostate cancer progression and other vegetables and fruit consumption [38].
Interestingly, a study of 1202 men with non-metastatic prostate cancer in the Health Professionals’ Follow-up Study (HPFS) [36] examined the influence of the post-diagnostic intake of tomato products on prostate cancer progression, suggesting a beneficial effect of tomato sauce consumption (Q1 vs. Q4 adjusted HR 0.56 95% CI 0.38–0.82). However, a study by Richman et al. [38] found no significant associations with prostate cancer progression and tomato sauce.
3.4.4. Bladder Cancer
One recent prospective study [16] of 389 patients with bladder cancer investigated the influence of post-diagnostic vegetables and fruit intake on cancer recurrence. No significant association was found between fruit and vegetable intakes and bladder cancer recurrence (p > 0.05). According to the QUIPS tool, the overall risk of bias in this study is high.
3.4.5. Gynecological Cancers
A recent study [29] of 120 gynecological cancer women (i.e., ovarian (n = 19), cervical (n = 54), and uterine cancer (n = 47)) investigated the association of post-diagnostic diet quality assessed by HEI and MDS with ACM. A high HEI score (≥70) was associated with lower mortality (adjusted HR 0.92 95% CI 0.89–0.96). No significant association between MDS and ACM was found. According to the QUIPS tool, the overall risk of bias in this study is high.
3.4.6. Lung, Gastric and Pancreatic Cancers
We found no data showing an association between post-diagnostic food choices/dietary patterns and survival outcomes in patients diagnosed with lung, gastric, and pancreatic cancers.
4. Discussion
The overall results of this systematic review highlight that none of the food categories should be eliminated by cancer patients. Especially, there is no clear association between consumption of meat or animal products and cancer progression/recurrence or CSM after a cancer diagnosis. However, a significant positive association between detrimental dietary patterns such as WD and cancer progression was found. On the contrary, high consumption of fiber such as whole grain cereals, green and cruciferous vegetables seems to be protective against cancer progression and mortality.
Breast, colorectal and prostate cancers are the most studied. In patients with breast cancer, PD after diagnosis could improve OS. PD is characterized by a diet high in fruits, vegetables, whole grains, legumes, poultry, fish and low-fat products. This finding is consistent with results of studies assessing a positive effect of pre-diagnostic components of PD on breast cancer prognosis. Indeed, previous studies found that, before diagnosis, whole grain [43,44], fruit and vegetable [45] intakes could be associated with a lower risk of mortality after breast cancer. Several studies suggested that a high fiber intake was associated with a reduction in circulating estrogen and androstenedione levels which are involved in the pathogenesis of breast cancer [46,47]. Indeed, fiber may bind estrogens in the colon during the enterohepatic circulation and increase the fecal excretion of estrogens [44]. On the other hand, WD and higher intake of saturated fats/trans after diagnosis could increase ACM, particularly deaths from other causes than breast cancer. WD, based on high intake of refined grains, processed and red meats, desserts, fats, and sweets, is characterized by a high glycemic load which may promote weight gain and an increment of fat mass [48]. Weight gain and increase of fat mass with a concurrent loss in muscle mass have shown to worsen clinical outcomes of breast cancer treatments [24]. Moreover, our results showed that higher intake of protein after diagnosis could be associated with reduced risk of breast cancer recurrence [11,20]. Particularly, in more than 6000 breast cancer patients, protein intake from animal sources (meat, poultry, fish, dairy products, and eggs) could be associated with lower risk of CSM [11,20]. Considering that increased protein intake contributes to greater strength and muscle mass gains when combined with physical exercise [49], higher intake of proteins (particularly animal proteins) with physical activity after diagnosis could counterbalance loss of muscle mass known to jeopardize survival of several cancers [50,51] including breast cancer. However, post-diagnostic consumption of high-fat dairy products could increase ACM and death from other causes than breast cancer [27] whereas total dairy products and low-fat dairy products were not associated with breast cancer prognostic outcomes [20,28]. We can hypothesize that estrogenic hormones found in dairy fat may be detrimental to breast cancer survival [27]. To date, even if further studies are needed to clarify mechanisms of all these findings, clinicians and breast cancer patients can be reassured that consuming animal protein-containing foods is not likely to increase CSM and ACM.
As regards colorectal cancer, high adherence to healthy diet and Mediterranean diet, in particular MMDS for Americans, could decrease cancer mortality and recurrence. Several clinical studies have reported an association of the components of Mediterranean diet—such as olive oil polyphenols and tomato lycopene—with a reduction in cancer initiation and progression [52]. Moreover, Mediterranean diet has been shown to be rich in antioxidants which may be associated with lower plasma concentrations of inflammatory markers [53]. Furthermore, an increment of dietary fiber from vegetables and whole grain after colorectal diagnosis could significantly improve ACM and CSM [34,35]. We can hypothesize that various components in whole grain and vegetables such as phytoestrogens, vitamins, antioxidants and microelements have beneficial influence on colorectal cancer. Moreover, dietary fiber derived from whole grain, fruits and vegetables could lower ACM and CSM after breast cancer by adjusting the gut microbiota and metabolism [54,55]. Indeed, short chain fatty acids (SCFAs) are the end-products of fiber and complex carbohydrates fermentation of most of the bacterial strains in the colon [56]. Several studies [57,58,59] showed a strong association between gut microbiota composition and cancer. Recently, a prospective cohort study [60] showed a significant association between higher concentrations of SCFAs and longer PFS in cancer patients. Additionally, our results suggested that a high consumption of nuts could improve OS and DFS in patients with colorectal cancer. Indeed, nuts—rich in unsaturated fatty acids, fiber, minerals, phenolic antioxidants, and phytosterols—could inhibit colorectal cancer growth by inducing cell cycle arrest, apoptosis, and by inhibiting cell proliferation, angiogenesis, and metastasis [61,62]. Moreover, consumption of marine ω3 PUFAs and dark fish (such as salmon particularly high in marine ω-3 PUFAs) improves DFS and OS [33]. Indeed, in vitro, studies showed that ω3 PUFAs have antiproliferative and apoptotic effects on human colorectal cancer stem-like cells [63,64]. On the other hand, high adherence to WD after diagnosis could worsen OS, DFS, and RFS in colorectal cancer. The study of Fung et al. [65] showed that WD is positively correlated with levels of biomarkers of obesity and cardiovascular disease risk such as serum insulin [65]. Both insulin and insulin like growth factors have been shown to be associated with enhanced tumor growth and antiapoptosis [66]. Hence, WD may increase the risk of cancer progression and recurrence. Notwithstanding red and processed meat intake may increase the risk of developing colorectal cancer [67,68], no significant association was found between mortality and post-diagnostic meat and red meat consumption [30,31,34]. Moreover, a reduction of meat consumption might result in loss of muscle mass which could worsen clinical outcomes and increase risk of colorectal cancer mortality. However, long-term meat consumption may be a more relevant measure and may be better represented by pre-diagnostic diet than by post- diagnostic diet [30].
Regarding prostate cancer, adherence to the Mediterranean diet could decrease ACM [41]. Mediterranean diet is mainly characterized by the consumption of olive oil. Animal and in vitro model studies [69,70] showed that polyphenols found in vegetable fats—such as hydroxytyrosol in olive oil—could induce apoptosis and suppress multiple oncogenic signaling pathways in prostate cancer cells. Also, tomato is a main component of Mediterranean diet, however, a positive association between tomato sauce consumption and prostate cancer prognosis remains under debate [36,38]. Lycopene, a carotenoid present in tomatoes, could inhibit the proliferation of androgen-dependent human prostate tumor cells through specific molecular pathways [71]. Further research on the role of tomatoes after prostate cancer diagnosis is needed. In addition, Richman et al. suggested that patients consuming cruciferous vegetables after prostate cancer diagnosis—such as broccoli, cauliflower, cabbage, brussels sprouts—had lower risk of cancer progression [38]. Cruciferous vegetables are composed of phytochemicals such as glucosinolates. Glucosinolates are hydrolyzed to form isothiocyanates and indoles, which have anticarcinogenic effects in vitro and in vivo [72]. Nevertheless, because of the small sample size of the study of Richman et al. [38] and the lack of assessment of pre-diagnostic diet, these results should be taken with caution. Vegetable fat intake after diagnosis was associated with a lower risk of CSM and ACM [39]. Indeed, animal and in vitro data suggest that components in olive oil inhibit migration, invasion, and adhesion of prostate cancer cells [70], and walnuts reduce prostate tumor growth [73] and inhibit androgen receptor expression in prostate cancer cells [74]. On the contrary, post-diagnostic intake of saturated and trans fat could increase the risk of ACM [39,42]. This could be explained by several biological mechanisms. High-fat diet including saturated and trans fats could modulate androgen signaling [75], upregulate the insulin-like growth factor pathway and increase prostate inflammation, thereby promoting prostate cancer growth [76]. Saturated and trans-fatty acid intake also may raise serum cholesterol levels [77,78]. Cholesterol is required for cell growth and may promote prostate cancer development [79]. Hence, perturbations of cholesterol homeostasis could enhance prostate cancer progression and mortality [79]. Like cholesterol, choline is highly concentrated in prostate cancer cells, and blood concentrations of choline have been associated with an increased risk of prostate cancer [80]. Eggs, milk, and poultry are the main sources of choline. This could explain that high consumption of eggs, poultry with skin, and whole milk could increase cancer progression and ACM. However, in a prospective cohort study of 47,896 men [81] post-diagnostic choline intake was not significantly associated with mortality. Further studies are needed to examine the relation of dietary choline to choline concentrations in the prostate and the effects of dietary choline in malignant prostate cells.
For bladder and gynecological cancers, poor data was available, and the results of the studies did not show any association of food components or diet quality scores [16,29]. Post-diagnostic diet of lung and stomach cancer patients still remains totally unexplored. More research is required to propose an adequate nutritional support to these patients to improve their nutritional status and prognosis [82,83]. Regarding pancreatic cancer, despite exclusion criteria (diet combined with supplementation [84], animal model [85]), few studies [84,85] could not be ignored. In vitro, mice models have shown significantly lower pancreatic tumor weight and higher muscle weight in patients fed with a ketogenic diet (KD) versus a standard diet [85]. KD could be a potential chemotherapy adjuvant in pancreatic cancer patients reducing glucose uptake, necessary for cancer cell cycle [86]. However, the effects of a long-term KD on health and microbiota remain controversial [56]. Consequently, further studies with larger sample size are required to assess the potential effect of KD as adjuvant therapy on patients’ survival and pancreatic tumor progression. A recent study [84] investigated the effects of alkalization diet (with supplementary oral sodium bicarbonate) during chemotherapy on the survival of 28 advanced pancreatic patients. The alkaline diet consists of a daily intake of at least 400 g of fruits and vegetables and no intake of meat and dairy products. From the start of alkalization therapy, the median OS of the patients with high urine pH (>7.0) was significantly longer than those with a low urine pH (≤7.0) [84]. Vegetables and fruits intake may modify tumor metabolism through a reduction of the inflammation, a moderate caloric restriction, and inducing changes in patient’s insulin levels and gut microbiome [84]. However, since the sample size of this study is very small, further studies are needed to confirm these interesting results.
This systematic review has several strengths. The majority of the included studies are cohort studies with large sample size, long follow-up durations, and low to moderate risk of bias. However, some limitations could not be ignored. First, data from the studies, prospective in nature, are observational; therefore, the possibility of residual confounding and causal interference cannot be excluded. Secondly, there is a high heterogeneity between studies due to variations in dietary exposure, dietary assessment methods, and population characteristics, making meta-analyses unfeasible. Third, information on the tumor stage and histology was unfortunately not available in several included studies. Furthermore, in most studies, the exposure to dietary patterns or factors was assessed by FFQs, consequently, errors in measurement need to be considered. Indeed, an imprecise measurement of consumption might have attenuated the true associations. Moreover, most studies did not assess pre-diagnostic dietary data, however, assessing the changes from pre- diagnostic to post-diagnostic diet would be relevant because the association observed after diagnosis may reflect intake before diagnosis.
5. Conclusions
Detrimental dietary patterns such as WD and the high consumption of some food categories (saturated/trans fats, high-fat dairy products) could worsen prognostic outcomes in breast, colorectal and prostate cancer patients. Nevertheless, animal proteins such as fish, poultry, low-fat dairy products and meat should not be excluded from cancer patient’s diet. More research is needed to confirm these findings and better clarify the impact of diet after diagnosis in cancer patients. Further investigation is warranted to explore the role of post-diagnostic diet in the most common cancers such as lung, stomach, gynecological, bladder, and pancreatic cancer.
Supplementary Materials
The following are available online at https://www.mdpi.com/2072-6643/12/8/2345/s1, Table S1: Full search strategies for electronic databases; Table S2: Definitions of dietary patterns and diet quality scores; Table S3: Bias assessment results for each study and each domain using the Quality in Prognostic Studies (QUIPS) tool.
Author Contributions
Conceptualization, E.R. and M.C.M.; data curation, P.R. and L.S.; formal analysis, M.C., P.R. and L.S.; investigation, C.P., E.B.; methodology, G.I. and M.C.; supervision, M.M. and A.M.; validation, A.G.; writing—original draft, P.R. and E.R.; writing—review and editing, E.R., M.C., M.C.M., C.P., E.B., A.G.; All authors have read and agreed to the published version of the manuscript.
Funding
This research received no external funding.
Conflicts of Interest
The authors declare no conflict of interest.
References
- World Health Organization. Press Release n° 263. 12 September 2018. Available online: https://www.who.int/cancer/PRGlobocanFinal.pdf (accessed on 12 February 2020).
- Grosso, G.; Bella, F.; Godos, J.; Sciacca, S.; Del Rio, D.; Ray, S.; Galvano, F.; Giovannucci, E.L. Possible role of diet in cancer: Systematic review and multiple meta-analyses of dietary patterns, lifestyle factors, and cancer risk. Nutr. Rev. 2017, 75, 405–419. [Google Scholar] [CrossRef] [PubMed]
- Schwingshackl, L.; Schwedhelm, C.; Galbete, C.; Hoffmann, G. Adherence to Mediterranean Diet and Risk of Cancer: An Updated Systematic Review and Meta-Analysis. Nutrients 2017, 9, 1063. [Google Scholar] [CrossRef] [PubMed]
- Schwingshackl, L.; Hoffmann, G. Adherence to Mediterranean diet and risk of cancer: A systematic review and meta-analysis of observational studies. Int. J. Cancer 2014, 135, 1884–1897. [Google Scholar] [CrossRef]
- Cederholm, T.; Barazzoni, R.; Austin, P.; Ballmer, P.; Biolo, G.; Bischoff, S.C.; Compher, C.; Correia, I.; Higashiguchi, T.; Holst, M.; et al. ESPEN guidelines on definitions and terminology of clinical nutrition. Clin. Nutr. 2017, 36, 49–64. [Google Scholar] [CrossRef] [PubMed]
- Muscaritoli, M.; Lucia, S.; Farcomeni, A.; Lorusso, V.; Saracino, V.; Barone, C.; Plastino, F.; Gori, S.; Magarotto, R.; Carteni, G.; et al. Prevalence of malnutrition in patients at first medical oncology visit: The PreMiO study. Oncotarget 2017, 45, 79884–79896. [Google Scholar] [CrossRef] [PubMed]
- Muscaritoli, M.; Molfino, A.; Gioia, G.; Laviano, A.; Rossi Fanelli, F. The “parallel pathway”: A novel nutritional and metabolic approach to cancer patients. Int. Emerg. Med. 2011, 6, 105–112. [Google Scholar] [CrossRef] [PubMed]
- Rinninella, E.; Persiani, R.; D’Ugo, D.; Pennestrì, F.; Cicchetti, A.; Di Brino, E.; Cintoni, M.; Miggiano, G.A.D.; Gasbarrini, A.; Mele, M.C. NutriCatt protocol in the Enhanced Recovery After Surgery (ERAS) program for colorectal surgery: The nutritional support improves clinical and cost-effectiveness outcomes. Nutrition 2018, 50, 74–81. [Google Scholar] [CrossRef] [PubMed]
- Arends, J.; Bachmann, P.; Baracos, V.; Barthelemy, N.; Bertz, H.; Bozzetti, F.; Fearon, K.; Hütterer, E.; Isenring, E.; Kaasa, S.; et al. ESPEN guidelines on nutrition in cancer patients. Clin. Nutr. 2017, 36, 11–48. [Google Scholar] [CrossRef]
- Zick, S.M.; Snyder, D.; Abrams, D.I. Pros and cons of dietary strategies popular among cancer patients. Oncology 2018, 32, 542–547. [Google Scholar]
- Holmes, M.D.; Wang, J.; Hankinson, S.E.; Tamimi, R.M.; Chen, W.Y. Protein intake and breast cancer survival in the Nurses’ Health Study. J. Clin. Oncol. 2017, 35, 325–333. [Google Scholar] [CrossRef]
- Meyerhardt, J.A.; Niedzwiecki, D.; Hollis, D.; Saltz, L.B.; Hu, F.B.; Mayer, R.J.; Nelson, H.; Whittom, R.; Hantel, A.; Thomas, J.; et al. Association of dietary patterns with cancer recurrence and survival in patients with stage III colon cancer. JAMA 2007, 298, 754–764. [Google Scholar] [CrossRef] [PubMed]
- Fadelu, T.; Zhang, S.; Niedzwiecki, D.; Ye, X.; Saltz, L.B.; Mayer, R.J.; Mowat, R.B.; Whittom, R.; Hantel, A.; Benson, A.B.; et al. Nut Consumption and Survival in Patients with Stage III Colon Cancer: Results from CALGB 89803 (Alliance). J. Clin. Oncol. 2018, 36, 1112–1120. [Google Scholar] [CrossRef] [PubMed]
- Richman, E.L.; Stampfer, M.J.; Paciorek, A.; Broering, J.M.; Carroll, P.R.; Chan, J.M. Intakes of meat, fish, poultry, and eggs and risk of prostate cancer progression. Am. J. Clin. Nutr. 2010, 91, 712–721. [Google Scholar] [CrossRef]
- Ratjen, I.; Schafmayer, C.; di Giuseppe, R.; Waniek, S.; Plachta-Danielzik, S.; Koch, M.; Nöthlings, U.; Hampe, J.; Schlesinger, S.; Lieb, W. Postdiagnostic Mediterranean, and Healthy Nordic Dietary Patterns Are Inversely Associated with All-Cause Mortality in Long-Term Colorectal Cancer Survivors. J. Nutr. 2017, 147, 636–644. [Google Scholar] [CrossRef] [PubMed]
- Jochems, S.H.J.; van Osch, F.H.M.; Reulen, R.C.; van Hensbergen, M.; Nekeman, D.; Pirrie, S.; Wesselius, A.; van Schooten, F.J.; James, N.D.; Wallace, D.M.A.; et al. Fruit and vegetable intake and the risk of recurrence in patients with non-muscle invasive bladder cancer: A prospective cohort study. Cancer Causes Control 2018, 29, 573–579. [Google Scholar] [CrossRef] [PubMed]
- Cochrane Handbook for Systematic Reviews of Interventions Version 5.1.0 [Updated March 2011]. Available online: www.cochrane-handbook.org (accessed on 28 February 2020).
- Moher, D.; Liberati, A.; Tetzlaff, J.; Altman, D.G.; Prisma Group. Preferred reporting items for systematic reviews and meta-analyses: The Prisma statement. BMJ 2009, 339. [Google Scholar] [CrossRef]
- Hayden, J.; van der Windt, D.A.; Cartwright, J.L.; Côté, P.; Bombardier, C. Assessing bias in studies of prognostic factors. Ann. Int. Med. 2013, 158, 280–286. [Google Scholar] [CrossRef]
- Holmes, M.D.; Stampfer, M.J.; Colditz, G.A.; Rosner, B.; Hunter, D.J.; Willett, W.C. Dietary factors and the survival of women with breast carcinoma. Cancer 1999, 86, 826–835. [Google Scholar] [CrossRef]
- Kroenke, C.H.; Chen, W.Y.; Rosner, B.; Holmes, M.D. Weight, weight gain, and survival after breast cancer diagnosis. J. Clin. Oncol. 2005, 23, 1370–1378. [Google Scholar] [CrossRef]
- Kwan, M.L.; Weltzien, E.; Kushi, L.H.; Castillo, A.; Slattery, M.L.; Caan, B.J. Dietary patterns and breast cancer recurrence and survival among women with early-stage breast cancer. J. Clin. Oncol. 2009, 27, 919–926. [Google Scholar] [CrossRef]
- Shu, X.O.; Zheng, Y.; Cai, H.; Gu, K.; Chen, Z.; Zheng, W.; Lu, W. Soy food and breast cancer survival. JAMA 2009, 302, 2437–2443. [Google Scholar] [CrossRef] [PubMed]
- Beasley, J.M.; Newcomb, P.A.; Trentham-Dietz, A.; Hampton, J.M.; Bersch, A.J.; Passarelli, M.N.; Holick, C.N.; Titus-Ernstof, L.; Egan, K.M.; Holmes, M.D.; et al. Post-diagnosis dietary factor and survival after invasive breast cancer. Breast Cancer Res. Treat. 2011, 128, 229–236. [Google Scholar] [CrossRef]
- Kim, E.H.; Willett, W.C.; Fung, T.; Rosner, B.; Holmes, M.D. Diet quality indices and postmenopausal breast cancer survival. Nutr. Cancer 2011, 63, 381–388. [Google Scholar] [CrossRef] [PubMed]
- Nechuta, S.J.; Caan, B.J.; Chen, W.Y.; Lu, W.; Chen, Z.; Kwan, M.L.; Flatt, S.W.; Zheng, Y.; Zheng, W.; Pierce, J.P.; et al. Soy food intake after diagnosis of breast cancer and survival: An in-depth analysis of combined evidence from cohort studies of US and Chinese women. Am. J. Clin. Nutr. 2012, 96, 123–132. [Google Scholar] [CrossRef] [PubMed]
- Kroenke, C.H.; Kwan, M.L.; Sweeney, C.; Castillo, A.; Caan, B.J. High- and low-fat dairy intake, recurrence, and mortality after breast cancer diagnosis. J. Natl. Cancer Inst. 2013, 105, 616–623. [Google Scholar] [CrossRef]
- Andersen, J.L.M.; Hansen, L.; Thomsen, B.L.R.; Christiansen, L.R.; Dragsted, L.O.; Olsen, A. Pre- and post-diagnostic intake of whole grain and dairy products and breast cancer prognosis: The Danish Diet, Cancer, and Health cohort. Breast Cancer Res. Treat. 2019, 179, 743–753. [Google Scholar] [CrossRef]
- Karavasiloglou, N.; Pestoni, G.; Faeh, D.; Rohrmann, S. Post-Diagnostic Diet Quality and Mortality in Females with Self-Reported History of Breast or Gynecological Cancers: Results from the Third National Health and Nutrition Examination Survey (NHANES III). Nutrients 2019, 11, 2558. [Google Scholar] [CrossRef]
- McCullough, M.L.; Gapstur, S.M.; Shah, R.; Jacobs, E.J.; Campbell, P.T. Association between red and processed meat intake and mortality among colorectal cancer survivors. J. Clin. Oncol. 2013, 31, 2773–2782. [Google Scholar] [CrossRef]
- Fung, T.T.; Kashambwa, R.; Sato, K.; Chiuve, S.E.; Fuchs, C.S.; Wu, K.; Giovannucci, E.; Ogino, S.; Hu, F.B.; Meyerhardt, J.A. Post diagnosis diet quality and colorectal cancer survival in women. PLoS ONE 2014, 9, 115377. [Google Scholar] [CrossRef]
- Yang, B.; McCullough, M.L.; Gapstur, S.M.; Jacobs, E.J.; Bostick, R.M.; Fedirko, V.; Flanders, W.D.; Campbell, P.T. Calcium, vitamin D, dairy products, and mortality among colorectal cancer survivors: The Cancer Prevention Study-II Nutrition Cohort. J. Clin. Oncol. 2014, 32, 2335–2343. [Google Scholar] [CrossRef]
- Van Blarigan, E.L.; Fuchs, C.S.; Niedzwiecki, D.; Ye, X.; Zhang, S.; Song, M.; Saltz, L.B.; Mayer, R.J.; Mowat, R.B.; Whittom, R.; et al. Marine ω-3 Polyunsaturated Fatty Acid and Fish Intake after Colon Cancer Diagnosis and Survival: CALGB 89803 (Alliance). Cancer Epidemiol. Biomark. Prev. 2018, 27, 438–445. [Google Scholar] [CrossRef] [PubMed]
- Tamakoshi, A.; Nakamura, K.; Ukawa, S.; Okada, E.; Hirata, M.; Nagai, A.; Matsuda, K.; Kamatani, Y.; Muto, K.; Kiyohara, Y.; et al. Characteristics and prognosis of Japanese colorectal cancer patients: The BioBank Japan Project. J. Epidemiol. 2017, 27, 36–42. [Google Scholar] [CrossRef] [PubMed]
- Song, M.; Wu, K.; Meyerhardt, J.; Ogino, S.; Wang, M.; Fuchs, C.S.; Giovannucci, E.L.; Chan, A.T. Fiber Intake and Survival After Colorectal Cancer Diagnosis. JAMA Oncol. 2018, 4, 71–79. [Google Scholar] [CrossRef] [PubMed]
- Chan, J.M.; Holick, C.N.; Leitzmann, M.F.; Rimm, E.B.; Willett, W.C.; Stampfer, M.J.; Giovannucci, E.L. Diet after diagnosis and the risk of prostate cancer progression, recurrence, and death (United States). Cancer Causes Control 2006, 17, 199–208. [Google Scholar] [CrossRef]
- Pettersson, A.; Kasperzyk, J.L.; Kenfield, S.A.; Richman, E.L.; Chan, J.M.; Willett, W.C.; Stampfer, M.J.; Mucci, L.A.; Giovannucci, E.L. Milk and dairy consumption among men with prostate cancer and risk of metastases and prostate cancer death. Cancer Epidemiol. Biomark. Prev. 2012, 21, 428–436. [Google Scholar] [CrossRef]
- Richman, E.L.; Carroll, P.R.; Chan, J.M. Vegetable and fruit intake after diagnosis and risk of prostate cancer progression. Int. J. Cancer 2012, 131, 201–210. [Google Scholar] [CrossRef]
- Richman, E.L.; Kenfield, S.A.; Chavarro, J.E.; Stampfer, M.J.; Giovannucci, E.L.; Willett, W.C.; Chan, J.M. Fat intake after diagnosis and risk of lethal prostate cancer and all-cause mortality. JAMA Int. Med. 2013, 173, 1318–1326. [Google Scholar] [CrossRef]
- Song, Y.; Chavarro, J.E.; Cao, Y.; Qiu, W.; Mucci, L.; Sesso, H.D.; Stampfer, M.J.; Giovannucci, E.; Pollak, M.; Liu, S.; et al. Whole milk intake is associated with prostate cancer-specific mortality among U.S. male physicians. J. Nutr. 2013, 143, 189–196. [Google Scholar] [CrossRef]
- Kenfield, S.A.; DuPre, N.; Richman, E.L.; Stampfer, M.J.; Chan, J.M.; Giovannucci, E.L. Mediterranean diet and prostate cancer risk and mortality in the Health Professionals Follow-up Study. Eur. Urol. 2014, 65, 887–894. [Google Scholar] [CrossRef]
- Van Blarigan, E.L.; Kenfield, S.A.; Yang, M.; Sesso, H.D.; Ma, J.; Stampfer, M.J.; Chan, J.M.; Chavarro, J.E. Fat intake after prostate cancer diagnosis and mortality in the Physicians’ Health Study. Cancer Causes Control 2015, 26, 1117–11126. [Google Scholar] [CrossRef]
- Saxe, G.A.; Rock, C.L.; Wicha, M.S.; Schottenfeld, D. Diet and risk for breast cancer recurrence and survival. Breast Cancer Res. Treat. 1999, 53, 241–253. [Google Scholar] [CrossRef] [PubMed]
- Aune, D.; Chan, D.S.; Greenwood, D.C.; Vieira, A.R.; Rosenblatt, D.A.; Vieira, R.; Norat, T. Dietary fiber and breast cancer risk: A systematic review and meta-analysis of prospective studies. Ann. Oncol. 2012, 23, 1394–1402. [Google Scholar] [CrossRef] [PubMed]
- Ingram, D. Diet and subsequent survival in women with breast cancer. Br. J. Cancer 1994, 69, 592–595. [Google Scholar] [CrossRef] [PubMed]
- Monroe, K.R.; Murphy, S.P.; Henderson, B.E.; Kolonel, L.N.; Stanczyk, F.Z.; Adlercreutz, H.; Pike, M.C. Dietary fiber intake and endogenous serum hormone levels in naturally postmenopausal Mexican American women: The Multiethnic Cohort Study. Nutr. Cancer 2007, 58, 127–135. [Google Scholar] [CrossRef] [PubMed]
- Rose, D.P.; Goldman, M.; Connolly, J.M.; Strong, L.E. High-fiber diet reduces serum estrogen concentrations in premenopausal women. Am. J. Clin. Nutr. 1991, 54, 520–525. [Google Scholar] [CrossRef] [PubMed]
- Brand-Miller, J.C.; Holt, S.H.; Pawlak, D.B.; McMillan, J. Glycemic index and obesity. Am. J. Clin. Nutr. 2002, 76, 281–285. [Google Scholar] [CrossRef]
- Carbone, J.W.; Pasiakos, S.M. Dietary Protein and Muscle Mass: Translating Science to Application and Health Benefit. Nutrients 2019, 11, 1136. [Google Scholar] [CrossRef]
- Rinninella, E.; Cintoni, M.; Raoul, P.; Pozzo, C.; Strippoli, A.; Bria, E.; Tortora, G.; Gasbarrini, A.; Mele, M.C. Muscle mass, assessed at diagnosis by L3-CT scan as a prognostic marker of clinical outcomes in patients with gastric cancer: A systematic review and meta-analysis. Clin. Nutr. 2019. [Google Scholar] [CrossRef]
- Rinninella, E.; Fagotti, A.; Cintoni, M.; Raoul, P.; Scaletta, G.; Scambia, G.; Gasbarrini, A.; Mele, M.C. Skeletal muscle mass as a prognostic indicator of outcomes in ovarian cancer: A systematic review and meta-analysis. Int. J. Gynecol. Cancer 2020. [Google Scholar] [CrossRef]
- Farinetti, A.; Zurlo, V.; Manenti, A.; Coppi, F.; Mattioli, A.V. Mediterranean diet and colorectal cancer: A systematic review. Nutrition 2017, 43, 83–88. [Google Scholar] [CrossRef]
- Fung, T.T.; McCullough, M.L.; Newby, P.K.; Manson, J.E.; Meigs, J.B.; Rifai, N.; Willett, W.C.; Hu, F.B. Diet-quality scores and plasma concentrations of markers of inflammation and endothelial dysfunction. Am. J. Clin. Nutr. 2005, 82, 163–173. [Google Scholar] [CrossRef]
- Whisner, C.M.; Athena Aktipis, C. The Role of the Microbiome in Cancer Initiation and Progression: How Microbes and Cancer Cells Utilize Excess Energy and Promote One Another’s Growth. Curr. Nutr. Rep. 2019, 8, 42–51. [Google Scholar] [CrossRef] [PubMed]
- Zeng, H.; Lazarova, D.L.; Bordonaro, M. Mechanisms linking dietary fiber, gut microbiota and colon cancer prevention. World J. Gastrointest Oncol. 2014, 6, 41–51. [Google Scholar] [CrossRef] [PubMed]
- Rinninella, E.; Cintoni, M.; Raoul, P.; Lopetuso, L.R.; Scaldaferri, F.; Pulcini, G.; Miggiano, G.A.D.; Gasbarrini, A.; Mele, M.C. Food Components and Dietary Habits: Keys for a Healthy Gut Microbiota Composition. Nutrients 2019, 11, 2393. [Google Scholar] [CrossRef]
- Wang, T.; Cai, G.; Qiu, Y.; Fei, N.; Zhang, M.; Pang, X.; Jia, W.; Cai, S.; Zhao, L. Structural segregation of gut microbiota between colorectal cancer patients and healthy volunteers. ISME J. 2012, 6, 320–329. [Google Scholar] [CrossRef] [PubMed]
- Shen, X.J.; Rawls, J.F.; Randall, T.; Burcal, L.; Mpande, C.N.; Jenkins, N.; Jovov, B.; Abdo, Z.; Sandler, R.S.; Keku, T.O. Molecular characterization of mucosal adherent bacteria and associations with colorectal adenomas. Gut Microbes 2010, 1, 138–147. [Google Scholar] [CrossRef]
- Kostic, A.D.; Gevers, D.; Pedamallu, C.S.; Michaud, M.; Duke, F.; Earl, A.M.; Ojesina, A.I.; Jung, J.; Bass, A.J.; Tabernero, J.; et al. Genomic analysis identifies association of Fusobacterium with colorectal carcinoma. Genome Res. 2012, 22, 292–298. [Google Scholar] [CrossRef]
- Nomura, M.; Nagatomo, R.; Doi, K.; Shimizu, J.; Baba, K.; Saito, T.; Matsumoto, S.; Inoue, K.; Muto, M. Association of Short-Chain Fatty Acids in the Gut Microbiome With Clinical Response to Treatment With Nivolumab or Pembrolizumab in Patients With Solid Cancer Tumors. JAMA Netw. Open 2020, 3, 202895. [Google Scholar] [CrossRef]
- Nagel, J.M.; Brinkoetter, M.; Magkos, F.; Liu, X.; Chamberland, J.P.; Shah, S.; Zhou, J.; Blackburn, G.; Mantzoros, C.S. Dietary walnuts inhibit colorectal cancer growth in mice by suppressing angiogenesis. Nutrition 2012, 28, 67–75. [Google Scholar] [CrossRef]
- Lee, J.; Kim, Y.S.; Lee, J.; Heo, S.C.; Lee, K.L.; Choi, S.W. Walnut Phenolic Extract and Its Bioactive Compounds Suppress Colon Cancer Cell Growth by Regulating Colon Cancer Stemness. Nutrients 2016, 8, 439. [Google Scholar] [CrossRef]
- Yang, T.; Fang, S.; Zhang, H.X.; Xu, L.X.; Zhang, Z.Q.; Yuan, K.T.; Xue, C.L.; Yu, H.L.; Zhang, S.; Li, Y.F.; et al. N-3 PUFAs have antiproliferative and apoptotic effects on human colorectal cancer stem-like cells in vitro. J. Nutr. Biochem. 2013, 24, 744–753. [Google Scholar] [CrossRef] [PubMed]
- Serini, S.; Cassano, R.; Corsetto, P.A.; Rizzo, A.M.; Calviello, G.; Trombino, S. Omega-3 PUFA Loaded in Resveratrol-Based Solid Lipid Nanoparticles: Physicochemical Properties and Antineoplastic Activities in Human Colorectal Cancer Cells In Vitro. Int. J. Mol. Sci. 2018, 19, 586. [Google Scholar] [CrossRef]
- Fung, T.T.; Rimm, E.B.; Spiegelman, D.; Rifai, N.; Tofler, G.H.; Willett, W.C.; Hu, F.B. Association between dietary patterns and plasma biomarkers of obesity and cardiovascular disease risk. Am. J. Clin. Nutr. 2001, 73, 61–67. [Google Scholar] [CrossRef] [PubMed]
- Sandhu, M.S.; Dunger, D.B.; Giovannucci, E.L. Insulin, insulin-like growth factor-I (IGF-I), IGF binding proteins, their biologic interactions, and colorectal cancer. J. Natl. Cancer Inst. 2002, 94, 972–980. [Google Scholar] [CrossRef] [PubMed]
- Huxley, R.R.; Ansary-Moghaddam, A.; Clifton, P.; Czernichow, S.; Parr, C.L.; Woodward, M. The impact of dietary and lifestyle risk factors on risk of colorectal cancer: A quantitative overview of the epidemiological evidence. Int. J. Cancer 2009, 125, 171–180. [Google Scholar] [CrossRef]
- Muscaritoli, M.; Amabile, M.I.; Molfino, A. Foods and their components promoting gastrointestinal cancer. Curr. Opin. Clin. Nutr. Metab. Care 2016, 19, 377–381. [Google Scholar] [CrossRef]
- Zubair, H.; Bhardwaj, A.; Ahmad, A.; Srivastava, S.K.; Khan, M.A.; Patel, G.K.; Singh, S.; Singh, A.P. Hydroxytyrosol induces apoptosis and cell cycle arrest and suppresses multiple oncogenic signaling pathways in prostate cancer cells. Nutr. Cancer 2017, 69, 932–942. [Google Scholar] [CrossRef]
- Luo, C.; Li, Y.; Wang, H.; Cui, Y.; Feng, Z.; Li, H.; Li, Y.; Wang, Y.; Wurtz, K.; Weber, P.; et al. Hydroxytyrosol promotes superoxide production and defects in autophagy leading to anti-proliferation and apoptosis on human prostate cancer cells. Curr. Cancer Drug Targets. 2013, 13, 625–639. [Google Scholar] [CrossRef]
- Yang, C.M.; Lu, I.H.; Chen, H.Y.; Hu, M.L. Lycopene inhibits the proliferation of androgen-dependent human prostate tumor cells through activation of PPARγ-LXRα-ABCA1 pathway. J. Nutr. Biochem. 2012, 23, 8–17. [Google Scholar] [CrossRef]
- Soundararajan, P.; Kim, J.S. Anti-Carcinogenic Glucosinolates in Cruciferous Vegetables and Their Antagonistic Effects on Prevention of Cancers. Molecules 2018, 23, 2983. [Google Scholar] [CrossRef]
- Reiter, R.J.; Tan, D.X.; Manchester, L.C.; Korkmaz, A.; Fuentes-Broto, L.; Hardman, W.E. A walnut-enriched diet reduces the growth of LNCaP human prostate cancer xenografts in nude mice. Cancer Investig. 2013, 31, 365–373. [Google Scholar] [CrossRef] [PubMed]
- Sanchez-Gonzalez, C.; Ciudad, C.J.; Noe, V.; Izquierdo-Pulido, M. Walnut polyphenol metabolites, urolithins A and B, inhibit the expression of the prostate-specific antigen and the androgen receptor in prostate cancer cells. Food Funct. 2014, 5, 2922–2930. [Google Scholar] [CrossRef] [PubMed]
- Shankar, E.; Bhaskaran, N.; MacLennan, G.T.; Liu, G.; Daneshgari, F.; Gupta, S. Inflammatory Signaling Involved in High-Fat Diet Induced Prostate Diseases. J. Urol. Res. 2015, 2, 1018. [Google Scholar] [PubMed]
- Di Sebastiano, K.M.; Mourtzakis, M. The role of dietary fat throughout the prostate cancer trajectory. Nutrients 2014, 6, 6095–6109. [Google Scholar] [CrossRef] [PubMed]
- Liska, D.J.; Cook, C.M.; Wang, D.D.; Gaine, P.C.; Baer, D.J. Trans fatty acids and cholesterol levels: An evidence map of the available science. Food Chem. Toxicol. 2016, 98, 269–281. [Google Scholar] [CrossRef] [PubMed]
- Hayes, K.C. Saturated fats and blood lipids: New slant on an old story. Can. J. Cardiol. 1995, 11 (Suppl. G), 39–46. [Google Scholar]
- Krycer, J.R.; Brown, A.J. Cholesterol accumulation in prostate cancer: A classic observation from a modern perspective. Biochim. Biophys. Acta 2013, 1835, 219–229. [Google Scholar] [CrossRef]
- Ackerstaff, E.; Pflug, B.R.; Nelson, J.B.; Bhujwalla, Z.M. Detection of increased choline compounds with proton nuclear magnetic resonance spectroscopy subsequent to malignant transformation of human prostatic epithelial cells. Cancer Res. 2001, 61, 3599–3603. [Google Scholar]
- Richman, E.L.; Kenfield, S.A.; Stampfer, M.J.; Giovannucci, E.L.; Zeisel, S.H.; Willett, W.C.; Chan, J.M. Choline intake and risk of lethal prostate cancer: Incidence and survival. Am. J. Clin. Nutr. 2012, 96, 855–863. [Google Scholar] [CrossRef]
- Rinninella, E.; Cintoni, M.; Raoul, P.; Pozzo, C.; Strippoli, A.; Bria, E.; Tortora, G.; Gasbarrini, A.; Mele, M.C. Effects of nutritional interventions on nutritional status in patients with gastric cancer: A systematic review and meta-analysis of randomized controlled trials. Clin. Nutr. ESPEN 2020, 38, 28–42. [Google Scholar] [CrossRef]
- Payne, C.; Larkin, P.J.; McIlfatrick, S.; Dunwoody, L.; Gracey, J.H. Exercise and nutrition interventions in advanced lung cancer: A systematic review. Curr. Oncol. 2013, 20, 321–337. [Google Scholar] [CrossRef] [PubMed]
- Hamaguchi, R.; Narui, R.; Wada, H. Effects of Alkalization Therapy on Chemotherapy Outcomes in Metastatic or Recurrent Pancreatic Cancer. Anticancer Res. 2020, 40, 873–880. [Google Scholar] [CrossRef] [PubMed]
- Shukla, S.K.; Gebregiworgis, T.; Purohit, V.; Chaika, N.V.; Gunda, V.; Radhakrishnan, P.; Mehla, K.; Pipinos, I.I.; Powers, R.; Yu, F.; et al. Metabolic reprogramming induced by ketone bodies diminishes pancreatic cancer cachexia. Cancer Metab. 2014, 2, 18. [Google Scholar] [CrossRef]
- Schwartz, L.; Seyfried, T.; Alfarouk, K.O.; Da Veiga Moreira, J.; Fais, S. Out of Warburg effect: An effective cancer treatment targeting the tumor-specific metabolism and dysregulated pH. Semin. Cancer Biol. 2017, 43, 134–138. [Google Scholar] [CrossRef] [PubMed]
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).