Effect of Selected Factors on the Serum 25(OH)D Concentration in Women Treated for Breast Cancer
Abstract
:1. Introduction
2. Materials and Methods
2.1. Studied Groups
2.2. Survey
2.3. Laboratory Determinations
2.4. Statistics
Sample Size Calculation
3. Results
3.1. Demographical and Anthropometrical Characteristic of Studied Groups
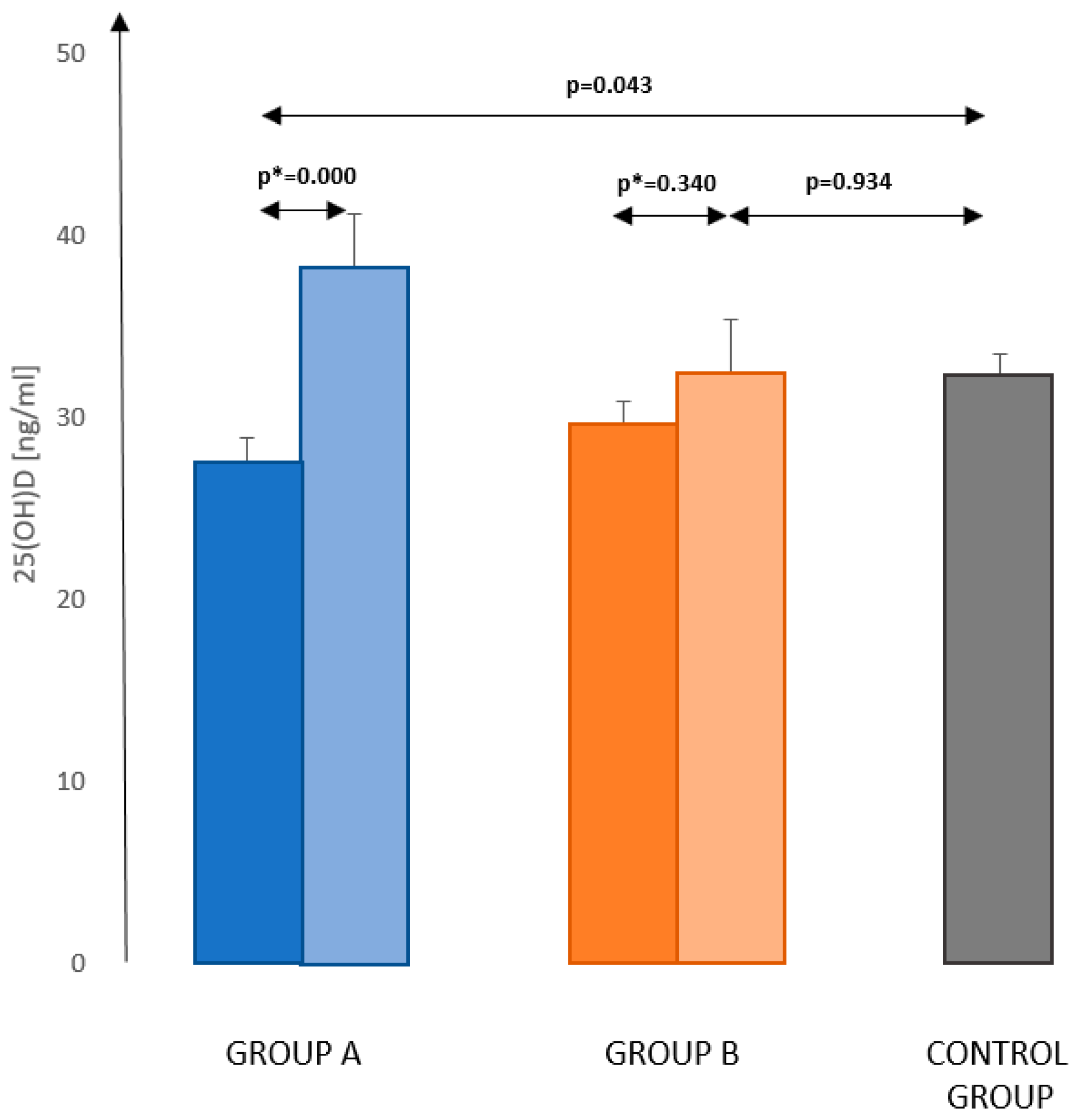
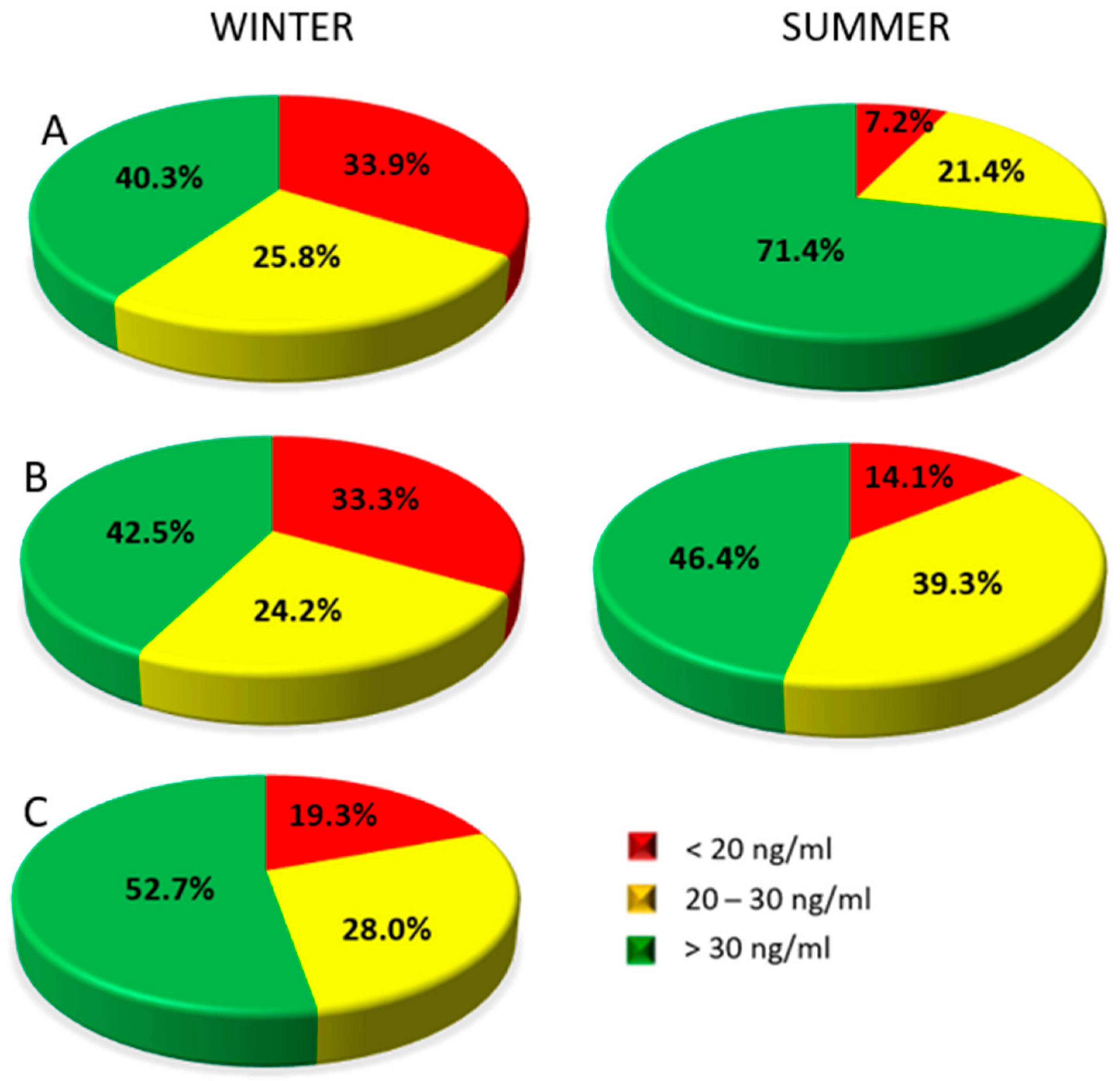
3.2. Vitamin D Concentration in Studied Patients
3.3. The Concentration of Vitamin D and Diet
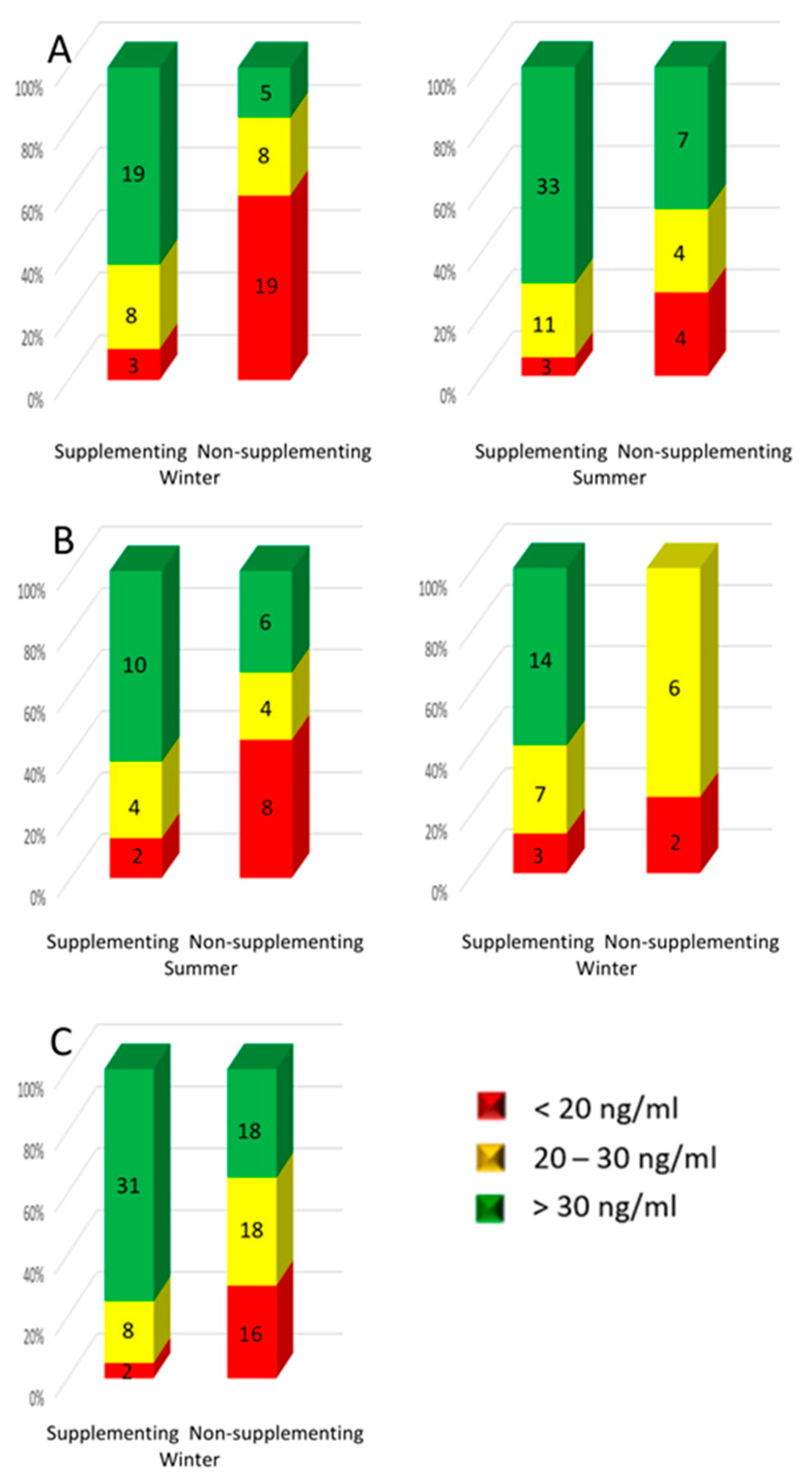
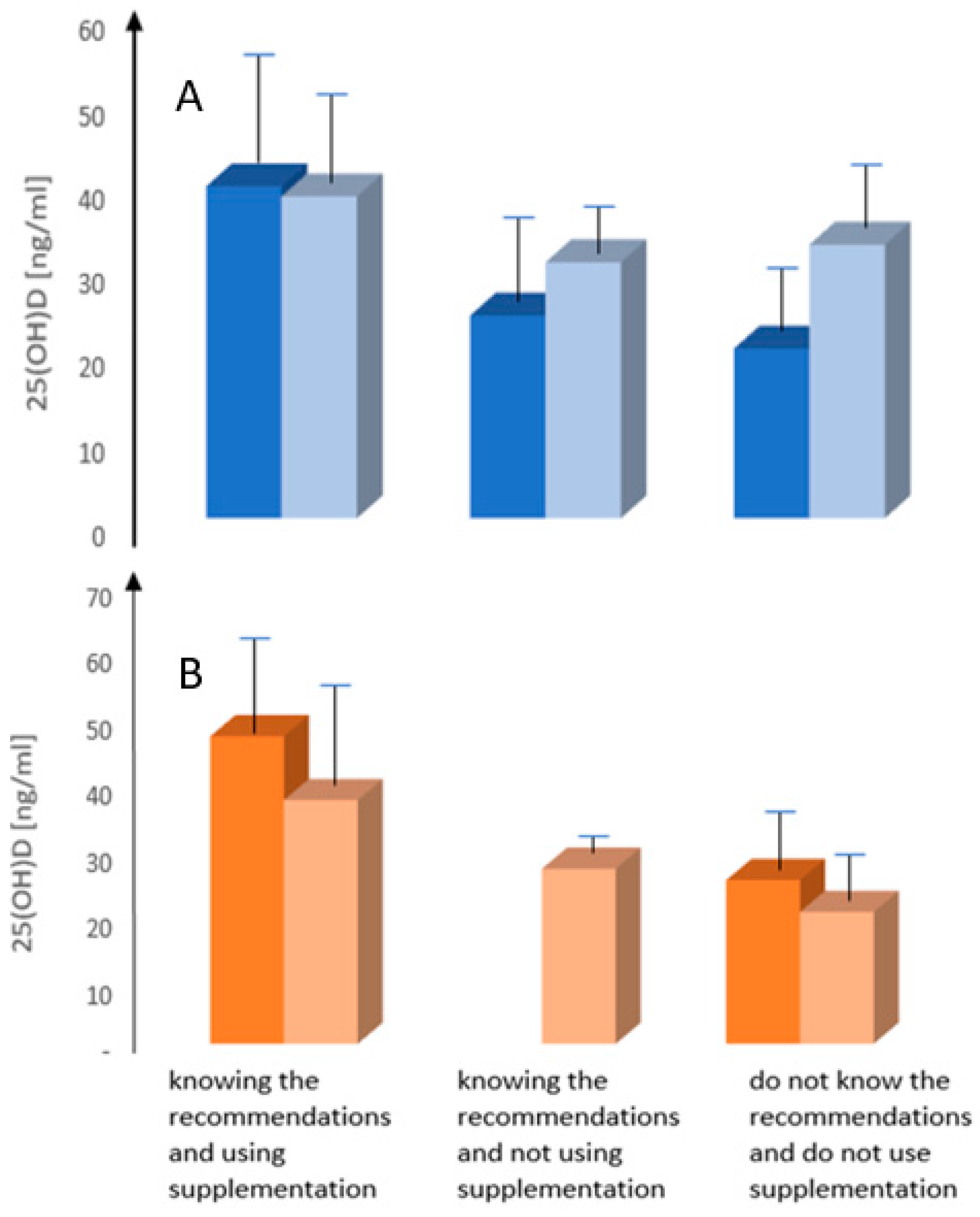
3.4. Supplementation of Vitamin D in Studied Patients
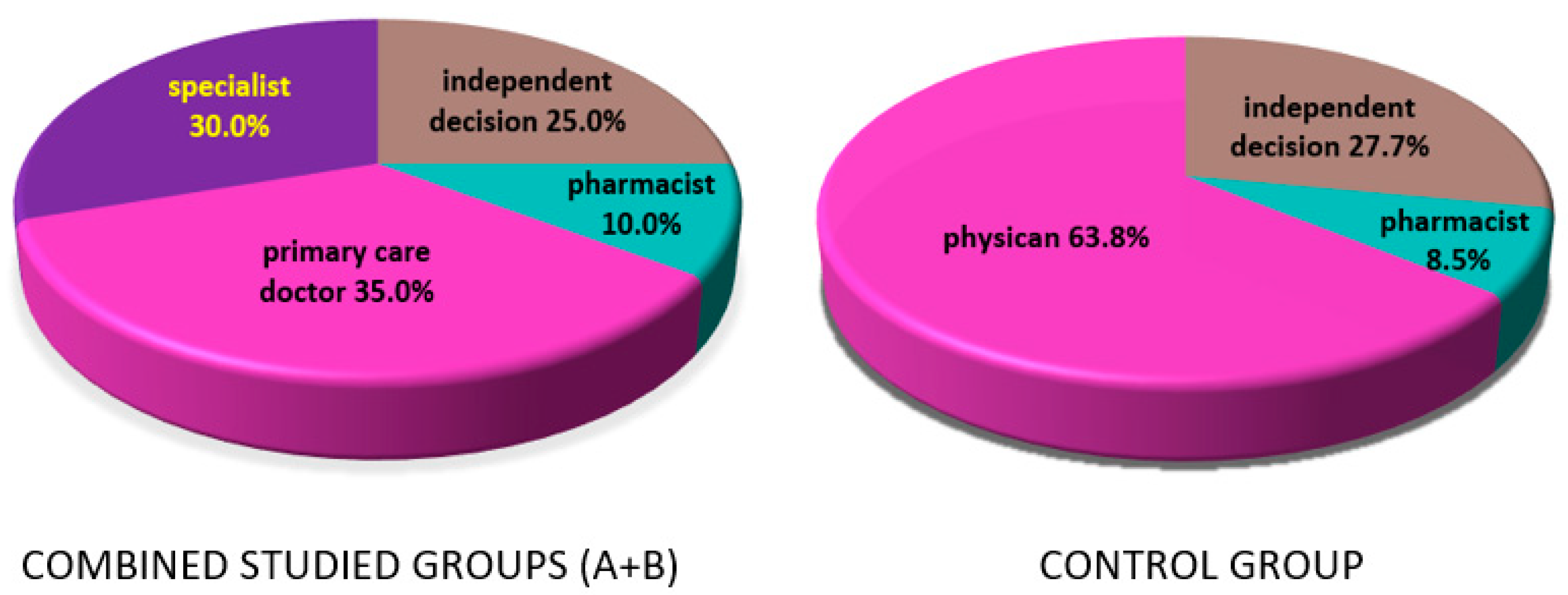
3.5. Knowledge of Recommendations on Vitamin D Supplementation
3.6. Exposure of Patients to Solar Radiation
3.7. Correlation of Vitamin D Concentration with Age
3.8. Correlation of Serum Vitamin D Concentration with Diet
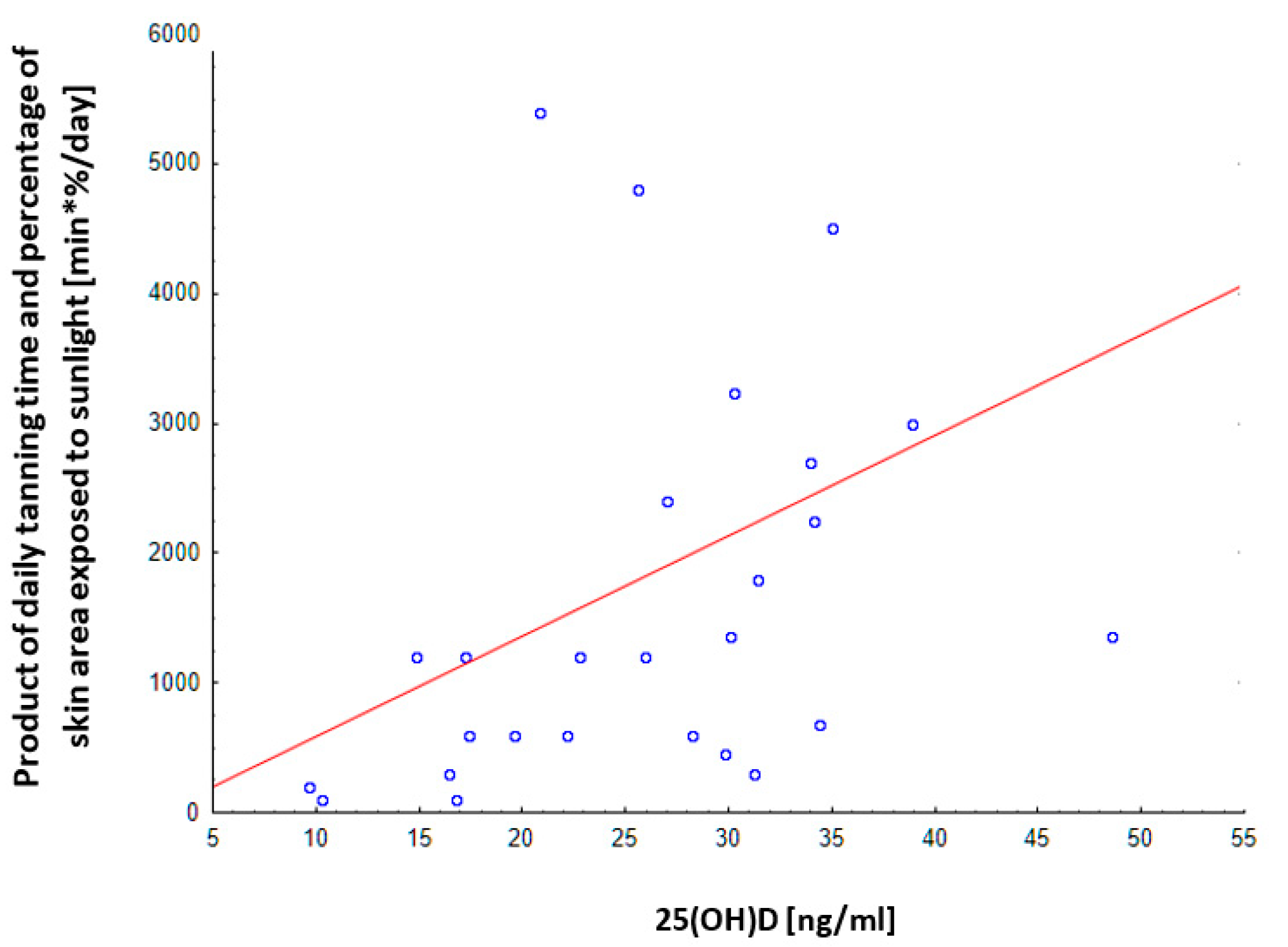
3.9. Correlation of the Declared Tanning Time with the Concentration of Vitamin D
4. Discussion
5. Conclusions
- Women with treated breast cancer are more likely to develop vitamin D deficiency than the healthy population.
- The seasons, and the time and area of the skin exposed to solar radiation, influence the concentration of 25(OH)D in women after treatment for breast cancer.
- Diet is not important in maintaining the proper concentration of vitamin D in women after treatment for breast cancer.
- Supplementation with vitamin D significantly improves its status in women after treatment for breast cancer; therefore, oncologists and GPs should recommend it to patients.
- Knowledge of recommendations on vitamin D supplementation among patients after treatment for breast cancer is directly related to the more frequent use of supplementation and less frequent 25(OH)D deficiency.
- Laboratory monitoring of vitamin D concentration has a positive effect on maintaining optimal vitamin D concentration in women after breast cancer treatment.
- Routine vitamin D testing should be introduced to the screening panel in follow-up patients after breast cancer treatment.
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Janssens, J.; Vandeloo, M. Rak piersi: Bezpośrednie i pośrednie czynniki ryzyka związane z wiekiem i stylem życia. Nowotw. J. Oncol. 2009, 59, 159–167. [Google Scholar]
- Wojciechowska, U.; Czaderny, K.; Ciuba, A.; Olasek, P.; Didkowska, J. Nowotwory Złośliwe w Polsce w 2016 Roku. Krajowy Rejestr Nowotworów. Available online: http://onkologia.org.pl/wp-content/uploads/Nowotwory_2016.pdf (accessed on 11 October 2019).
- Didkowska, J.; Wojciechowska, U.; Olasek, P. Nowotwory Złośliwe w Polsce w 2015 Roku. Krajowy Rejestr Nowotworów. Available online: http://onkologia.org.pl/wp-content/uploads/Nowotwory_2015.pdf (accessed on 11 October 2019).
- Didkowska, J.; Wojciechowska, U.; Czaderny, K.; Olasek, P.; Ciuba, A. Nowotwory Złośliwe w Polsce w 2017 Roku. Krajowy Rejestr Nowotworów. Available online: http://onkologia.org.pl/wp-content/uploads/Nowotwory_2017.pdf (accessed on 11 October 2019).
- Romieu, I.I.; Amadou, A.; Chajes, V. The Role of Diet, Physical Activity, Body Fatness, and Breastfeeding in Breast Cancer in Young Women: Epidemiological Evidence. Rev. Investig. Clin. 2017, 69, 193–203. [Google Scholar] [CrossRef]
- Chun, R.F.; Shieh, A.; Gottlieb, C.; Yacoubian, V.; Wang, J.; Hewison, M.; Adams, J.S. Vitamin D Binding Protein and the Biological Activity of Vitamin D. Front. Endocrinol. 2019, 10, 718. [Google Scholar] [CrossRef]
- De La Puente-Yagüe, M.; Cuadrado-Cenzual, M.A.; Ciudad-Cabañas, M.J.; Hernández-Cabria, M.; Collado, L. Vitamin D: And its role in breast cancer. Kaohsiung J. Med. Sci. 2018, 34, 423–427. [Google Scholar] [CrossRef]
- Garland, F.C.; Garland, C.F.; Gorham, E.D.; Young, J.F. Geographic variation in breast cancer mortality in the United States: A hypothesis involving exposure to solar radiation. Prev. Med. 1990, 19, 614–622. [Google Scholar] [CrossRef]
- Wu, X.; Zhou, T.; Cao, N.; Ni, J.; Wang, X. Role of Vitamin D Metabolism and Activity on Carcinogenesis. Oncol. Res. Featur. Preclin. Clin. Cancer Ther. 2015, 22, 129–137. [Google Scholar] [CrossRef]
- Chen, G.-C.; Zhang, Z.-L.; Wan, Z.; Wang, L.; Weber, P.; Eggersdorfer, M.; Qin, L.-Q.; Zhang, W. Circulating 25-hydroxyvitamin D and risk of lung cancer: A dose–response meta-analysis. Cancer Causes Control 2015, 26, 1719–1728. [Google Scholar] [CrossRef]
- Bauer, S.R.; Hankinson, S.E.; Bertone-Johnson, E.R.; Ding, E.L. Plasma Vitamin D Levels, Menopause, and Risk of Breast Cancer. Medicine 2013, 92, 123–131. [Google Scholar] [CrossRef] [Green Version]
- Shao, T.; Klein, P.; Grossbard, M.L. Vitamin D and Breast Cancer. Oncologist 2011, 17, 36–45. [Google Scholar] [CrossRef] [Green Version]
- Gandini, S.; Boniol, M.; Haukka, J.; Byrnes, G.; Cox, B.; Sneyd, M.J.; Mullie, P.; Autier, P. Meta-analysis of observational studies of serum 25-hydroxyvitamin D levels and colorectal, breast and prostate cancer and colorectal adenoma. Int. J. Cancer 2011, 128, 1414–1424. [Google Scholar] [CrossRef]
- Yin, L.; Grandi, N.; Raum, E.; Haug, U.; Arndt, V.; Brenner, H. Meta-analysis: Serum vitamin D and breast cancer risk. Eur. J. Cancer 2010, 46, 2196–2205. [Google Scholar] [CrossRef]
- Mondul, A.M.; Weinstein, S.J.; Layne, T.M.; Albanes, D. Vitamin D and Cancer Risk and Mortality: State of the Science, Gaps, and Challenges. Epidemiol. Rev. 2017, 39, 28–48. [Google Scholar] [CrossRef] [PubMed]
- Atoum, M.; Alzoughool, F. Vitamin D and Breast Cancer: Latest Evidence and Future Steps. Breast Cancer Basic Clin. Res. 2017, 11, 1–8. [Google Scholar] [CrossRef] [Green Version]
- Pandolfi, F.; Franza, L.; Mandolini, C.; Conti, P. Immune Modulation by Vitamin D: Special Emphasis on Its Role in Prevention and Treatment of Cancer. Clin. Ther. 2017, 39, 884–893. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Cadeau, C.; Fournier, A.; Mesrine, S.; Clavel-Chapelon, F.; Fagherazzi, G.; Boutron-Ruault, M.-C. Interaction between current vitamin D supplementation and menopausal hormone therapy use on breast cancer risk: Evidence from the E3N cohort. Am. J. Clin. Nutr. 2015, 102, 966–973. [Google Scholar] [CrossRef] [Green Version]
- Fathi, N.; Ahmadian, E.; Shahi, S.; Roshangar, L.; Khan, H.; Kouhsoltani, M.; Dizaj, S.M.; Sharifi, S. Role of vitamin D and vitamin D receptor (VDR) in oral cancer. Biomed. Pharmacother. 2019, 109, 391–401. [Google Scholar] [CrossRef]
- O’Brien, K.M.; Sandler, D.P.; Taylor, J.A.; Weinberg, C.R. Serum Vitamin D and Risk of Breast Cancer within Five Years. Environ. Health Perspect. 2017, 125, 077004. [Google Scholar] [CrossRef]
- Takalkar, U.; Asegaonkar, S.; Advani, S. Vitamin D and prevention of breast cancer. J. Cancer Res. Forecast 2018, 1, 1–4. [Google Scholar]
- Runowicz, C.D.; Leach, C.R.; Henry, N.L.; Henry, K.S.; Mackey, H.T.; Cowens-Alvarado, R.L.; Cannady, R.S.; Pratt-Chapman, M.L.; Edge, S.B.; Jacobs, L.A.; et al. American Cancer Society/American Society of Clinical Oncology Breast Cancer Survivorship Care Guideline. CA Cancer J. Clin. 2015, 66, 43–73. [Google Scholar] [CrossRef] [PubMed]
- Coleman, R.; Body, J.J.; Aapro, M.; Hadji, P.; Herrstedt, J. Bone health in cancer patients: ESMO Clinical Practice Guidelines. Ann. Oncol. 2014, 25, iii124–iii137. [Google Scholar] [CrossRef]
- Rusińska, A.; Pludowski, P.; Walczak, M.; Borszewska-Kornacka, M.K.; Bossowski, A.; Chlebna-Sokół, D.; Czech-Kowalska, J.; Dobrzanska, A.; Franek, E.; Helwich, E.; et al. Vitamin D Supplementation Guidelines for General Population and Groups at Risk of Vitamin D Deficiency in Poland—Recommendations of the Polish Society of Pediatric Endocrinology and Diabetes and the Expert Panel with Participation of National Specialist Consultants and Representatives of Scientific Societies—2018 Update. Front. Endocrinol. 2018, 9, 246. [Google Scholar] [CrossRef]
- Kłek, S.; Jankowski, M.; Kruszewski, W.J.; Fijuth, J.; Kapała, A.; Kabata, P.; Wysocki, P.J.; Krzakowski, M.; Rutkowski, P. Standardy leczenia żywieniowego w onkologii. Nowotw. J. Oncol. 2015, 65, 320–337. [Google Scholar] [CrossRef] [Green Version]
- Pilz, S.; Hahn, A.; Schön, C.; Wilhelm, M.; Obeid, R. Effect of Two Different Multimicronutrient Supplements on Vitamin D Status in Women of Childbearing Age: A Randomized Trial. Nutrients 2017, 9, 30. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kotlarczyk, M.P.; Perera, S.; Ferchak, M.A.; Nace, D.A.; Resnick, N.M.; Greenspan, S.L. Vitamin D deficiency is associated with functional decline and falls in frail elderly women despite supplementation. Osteoporos. Int. 2017, 28, 1347–1353. [Google Scholar] [CrossRef]
- Camacho, P.M.; Petak, S.M.; Binkley, N.; Clarke, B.L.; Harris, S.T.; Hurley, D.L.; Kleerekoper, M.; Lewiecki, E.M.; Miller, P.D.; Narula, H.S.; et al. American Association of Clinical Endocrinologists and American College of Endocrinology Clinical Practice Guidelines for the Diagnosis and Treatment of Postmenopausal Osteoporosis—2016. Endocr. Pract. 2016, 22, 1–42. [Google Scholar] [CrossRef] [Green Version]
- German Nutrition Society. New reference values for vitamin D. Ann. Nutr. Metab. 2012, 60, 241–246. [Google Scholar] [CrossRef]
- Jarosz, M. Normy Żywienia dla Populacji Polskiej—Nowelizacja; Wydawnictwa—Instytut Żywności i Żywienia: Warsaw, Poland, 2012. [Google Scholar]
- Stanowisko Zespołu Ekspertów. Polskie zalecenia dotyczące profilaktyki niedoborów witaminy D—2009. Ginekol. Pol. 2010, 81, 149–153. [Google Scholar]
- Mohr, S.B.; Gorham, E.D.; Kim, J.; Hofflich, H.; Garland, C.F. Meta-analysis of vitamin D sufficiency for improving survival of patients with breast cancer. Anticancer Res. 2014, 34, 1163–1166. [Google Scholar]
- Kuzmickiene, I.; Atkocius, V.; Aleknavicius, E.; Ostapenko, V. Impact of season of diagnosis on mortality among breast cancer survivors. J. Cancer Res. Ther. 2018, 14, 1091. [Google Scholar] [CrossRef]
- Shi, L.; Nechuta, S.; Gao, Y.-T.; Zheng, Y.; Dorjgochoo, T.; Wu, J.; Cai, Q.; Zheng, W.; Lu, W.; Shu, X.O. Correlates of 25-Hydroxyvitamin D among Chinese Breast Cancer Patients. PLoS ONE 2014, 9, e86467. [Google Scholar] [CrossRef]
- Lips, P.; Cashman, K.D.; Lamberg-Allardt, C.; Bischoff-Ferrari, H.A.; Obermayer-Pietsch, B.; Bianchi, M.L.; Stepan, J.; Fuleihan, G.E.-H.; Bouillon, R. Current vitamin D status in European and Middle East countries and strategies to prevent vitamin D deficiency: A position statement of the European Calcified Tissue Society. Eur. J. Endocrinol. 2019, 180, P23–P54. [Google Scholar] [CrossRef] [Green Version]
- Płudowski, P.; Ducki, C.; Konstantynowicz, J.; Jaworski, M. Vitamin D status in Poland. Pol. Arch. Intern. Med. 2016, 126, 530–539. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Rabenberg, M.; Scheidt-Nave, C.; A Busch, M.; Rieckmann, N.; Hintzpeter, B.; Mensink, G.B. Vitamin D status among adults in Germany—Results from the German Health Interview and Examination Survey for Adults (DEGS1). BMC Public Health 2015, 15, 1–15. [Google Scholar] [CrossRef] [Green Version]
- Foumani, R.S.; Khodaie, F. The Correlation of Plasma 25-Hydroxyvitamin D Deficiency with Risk of Breast Neoplasms: A Systematic Review. Iran. J. Cancer Prev. 2016, 9, 1–7. [Google Scholar] [CrossRef]
- Andersen, M.R.; Sweet, E.; Hager, S.; Gaul, M.; Dowd, F.; Standish, L.J. Effects of Vitamin D Use on Health-Related Quality of Life of Breast Cancer Patients in Early Survivorship. Integr. Cancer Ther. 2019, 18, 1–12. [Google Scholar] [CrossRef]
- Apoe, O.; Jung, S.-H.; Liu, H.; Seisler, D.K.; Charlamb, J.; Zekan, P.; Wang, L.X.; Unzeitig, G.W.; Garber, J.; Marshall, J.; et al. Effect of Vitamin D Supplementation on Breast Cancer Biomarkers: CALGB 70806 (Alliance) Study Design and Baseline Data. Am. J. Hematol. 2016, 12, 4–9. [Google Scholar]
- Machado, M.R.M.; Almeida-Filho, B.D.S.; Vespoli, H.D.L.; Schmitt, E.B.; Nahas-Neto, J.; Nahas, E.A. Low pretreatment serum concentration of vitamin D at breast cancer diagnosis in postmenopausal women. Menopause 2019, 26, 293–299. [Google Scholar] [CrossRef] [PubMed]
- Valverde, C.N.; Gómez, J.Q. Vitamin D, determinant of bone and extrabone health. Importance of vitamin D supplementation in milk and dairy products. Nutr. Hosp. 2015, 31, 18–25. [Google Scholar]
- Woon, F.C.; Chin, Y.S.; Ismail, I.H.; Batterham, M.; Latiff, A.H.A.; Gan, W.Y.; Appannah, G.; Hussien, S.H.M.; Edi, M.; Tan, M.L.; et al. Vitamin D deficiency during pregnancy and its associated factors among third trimester Malaysian pregnant women. PLoS ONE 2019, 14, e0216439. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sediyama, C.M.O. Lifestyle and vitamin D dosage in women with breast cancer. Nutr. Hosp. 2016, 33, 1179–1186. [Google Scholar] [CrossRef] [Green Version]
- Krzyścin, J.W.; Jarosławski, J.; Sobolewski, P. A mathematical model for seasonal variability of vitamin D due to solar radiation. J. Photochem. Photobiol. B Biol. 2011, 105, 106–112. [Google Scholar] [CrossRef]
- Guo, J.; Lovegrove, J.A.; Givens, D.I. A Narrative Review of The Role of Foods as Dietary Sources of Vitamin D of Ethnic Minority Populations with Darker Skin: The Underestimated Challenge. Nutrients 2019, 11, 81. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zhang, X.; Harbeck, N.; Jeschke, U.; Sixou, S. Influence of vitamin D signaling on hormone receptor status and HER2 expression in breast cancer. J. Cancer Res. Clin. Oncol. 2016, 143, 1107–1122. [Google Scholar] [CrossRef]
- Acevedo, F.; Pérez, V.; Pérez-Sepúlveda, A.; Florenzano, P.; Artigas, R.; Medina, L.; Sánchez, C. High prevalence of vitamin D deficiency in women with breast cancer: The first Chilean study. Breast 2016, 29, 39–43. [Google Scholar] [CrossRef] [PubMed]
- Eliassen, A.H.; Warner, E.T.; Rosner, B.; Collins, L.C.; Beck, A.H.; Quintana, L.M.; Tamimi, R.M.; Hankinson, S.E. Plasma 25-Hydroxyvitamin D and Risk of Breast Cancer in Women Followed over 20 Years. Cancer Res. 2016, 76, 5423–5430. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- O’Sullivan, F.; Raftery, T.; Van Weele, M.; Van Geffen, J.; McNamara, D.; O’Morain, C.; Mahmud, N.; Kelly, D.; Healy, M.; O’Sullivan, M.; et al. Sunshine is an Important Determinant of Vitamin D Status Even among High-dose Supplement Users: Secondary Analysis of a Randomized Controlled Trial in Crohn’s Disease Patients. Photochem. Photobiol. 2019, 95, 1060–1067. [Google Scholar] [CrossRef]
- Hoel, D.G.; De Gruijl, F. Sun Exposure Public Health Directives. Int. J. Environ. Res. Public Health 2018, 15, 2794. [Google Scholar] [CrossRef] [Green Version]
- Directive 2002/46/EC of the European Parliament and of the Council of 10 June 2002. Available online: https://eur-lex.europa.eu/legal-content/EN/ALL/?uri=celex%3A32002L0046 (accessed on 7 May 2020).
- Dymkowska-Malesa, M.; Walczak, Z. Suplementacja w sporcie. Now. Lek. 2011, 80, 3, 199–204. [Google Scholar]
- Madden, J.M.; Murphy, L.; Zgaga, L.; Bennett, K. De novo vitamin D supplement use post-diagnosis is associated with breast cancer survival. Breast Cancer Res. Treat. 2018, 172, 179–190. [Google Scholar] [CrossRef]
- Baumann, M.; Dani, S.; Dietrich, D.; Hochstrasser, A.; Klingbiel, D.; Mark, M.T.; Riesen, W.F.; Ruhstaller, T.; Templeton, A.J.; Thürlimann, B. Vitamin D levels in Swiss breast cancer survivors. Swiss Med. Wkly. 2018, 148, w14576. [Google Scholar] [CrossRef]
- Griffin, N.; Dowling, M. Vitamin D supplementation and clinical outcomes in cancer survivorship. Br. J. Nurs. 2018, 27, 1121–1128. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Grant, W.B. A review of the evidence supporting the Vitamin D-cancer related hypothesis in 2017. Anticancer Res. 2018, 38, 1121–1236. [Google Scholar] [CrossRef] [PubMed]
- Karthikayan, A.; Sureshkumar, S.; Kadambari, D.; Vijayakumar, C. Low serum 25-hydroxy vitamin D levels are associated with aggressive breast cancer variants and poor prognostic factors in patients with breast carcinoma. Arch. Endocrinol. Metab. 2018, 62, 452–459. [Google Scholar] [CrossRef] [PubMed]
- Madden, J.M.; Duffy, M.J.; Zgaga, L.; Bennett, K. Trends in vitamin D supplement use in a general female and breast cancer population in Ireland: A repeated cross-sectional study. PLoS ONE 2018, 13, e0209033. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Tham, Y.-L.; Sexton, K.; Weiss, H.L.; Elledge, R.M.; Friedman, L.C.; Kramer, R.M. The adherence to practice guidelines in the assessment of bone health in women with chemotherapy-induced menopause. J. Support. Oncol. 2006, 4, 295–298. [Google Scholar]
- Bošković, L.; Gašparić, M.; Petković, M.; Gugić, D.; Lovasić, I.B.; Soldić, Ž.; Miše, B.P.; Dabelić, N.; Vazdar, L.; Vrdoljak, E. Bone health and adherence to vitamin D and calcium therapy in early breast cancer patients on endocrine therapy with aromatase inhibitors. Breast 2017, 31, 16–19. [Google Scholar] [CrossRef]
- Kimiafar, K.; Sarbaz, M.; Sales, S.S.; Esmaeili, M.; Ghazvini, Z.J. Breast cancer patients’ information needs and information-seeking behavior in a developing country. Breast 2016, 28, 156–160. [Google Scholar] [CrossRef]
- O’Brien, K.M.; Sandler, D.P.; House, M.; A Taylor, J.; Weinberg, C.R. The Association of a Breast Cancer Diagnosis with Serum 25-Hydroxyvitamin D Concentration over Time. Am. J. Epidemiol. 2019, 188, 637–645. [Google Scholar] [CrossRef]
- Heaney, R.P. Vitamin D in Health and Disease. Clin. J. Am. Soc. Nephrol. 2008, 3, 1535–1541. [Google Scholar] [CrossRef] [Green Version]
- Gallagher, J.C.; Sai, A.; Templin, T.; Smith, L. Dose Response to Vitamin D Supplementation in Postmenopausal Women. Ann. Intern. Med. 2012, 156, 425–437. [Google Scholar] [CrossRef]
- Peñas, R.D.L.; Majem, M.; Perez, J.; Virizuela, J.A.; Cancer, E.; Taín, P.D.; Donnay, O.; Hurtado, A.; Jiménez-Fonseca, P.; Ocon, M.J. SEOM clinical guidelines on nutrition in cancer patients (2018). Clin. Transl. Oncol. 2019, 21, 87–93. [Google Scholar] [CrossRef] [PubMed] [Green Version]
| n Supplementing/ n Non-Supplementing | Patients Supplementing Vitamin D (ng/mL) | Patients Non-SupplementingVitamin D (ng/mL) | p | |
|---|---|---|---|---|
| Group A winter | 30/32 | 34.6 ± 14.2 | 22.4 ± 11.9 | p = 0.0006 |
| Group A summer | 47/15 | 38.9 ± 12.2 | 34.8 ± 12.1 | p = 0.24 |
| Group B summer | 16/18 | 36.8 ± 15.4 | 24.0 ± 9.0 | p = 0.012 |
| Group B winter | 24/8 | 35.3 ± 13.7 | 22.8 ± 5.3 | p = 0.001 |
| Control group winter | 41/36 | 38.3 ± 16.3 | 27.6 ± 10.9 | p = 0.0006 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Radom, A.; Wędrychowicz, A.; Pieczarkowski, S.; Skoczeń, S.; Tomasik, P. Effect of Selected Factors on the Serum 25(OH)D Concentration in Women Treated for Breast Cancer. Nutrients 2021, 13, 564. https://doi.org/10.3390/nu13020564
Radom A, Wędrychowicz A, Pieczarkowski S, Skoczeń S, Tomasik P. Effect of Selected Factors on the Serum 25(OH)D Concentration in Women Treated for Breast Cancer. Nutrients. 2021; 13(2):564. https://doi.org/10.3390/nu13020564
Chicago/Turabian StyleRadom, Agnieszka, Andrzej Wędrychowicz, Stanisław Pieczarkowski, Szymon Skoczeń, and Przemysław Tomasik. 2021. "Effect of Selected Factors on the Serum 25(OH)D Concentration in Women Treated for Breast Cancer" Nutrients 13, no. 2: 564. https://doi.org/10.3390/nu13020564
APA StyleRadom, A., Wędrychowicz, A., Pieczarkowski, S., Skoczeń, S., & Tomasik, P. (2021). Effect of Selected Factors on the Serum 25(OH)D Concentration in Women Treated for Breast Cancer. Nutrients, 13(2), 564. https://doi.org/10.3390/nu13020564