Supplementation of Omega 3 during Pregnancy and the Risk of Preterm Birth: A Systematic Review and Meta-Analysis
Abstract
:1. Introduction
2. Materials and Methods
2.1. Eligibility Criteria
2.2. Outcomes
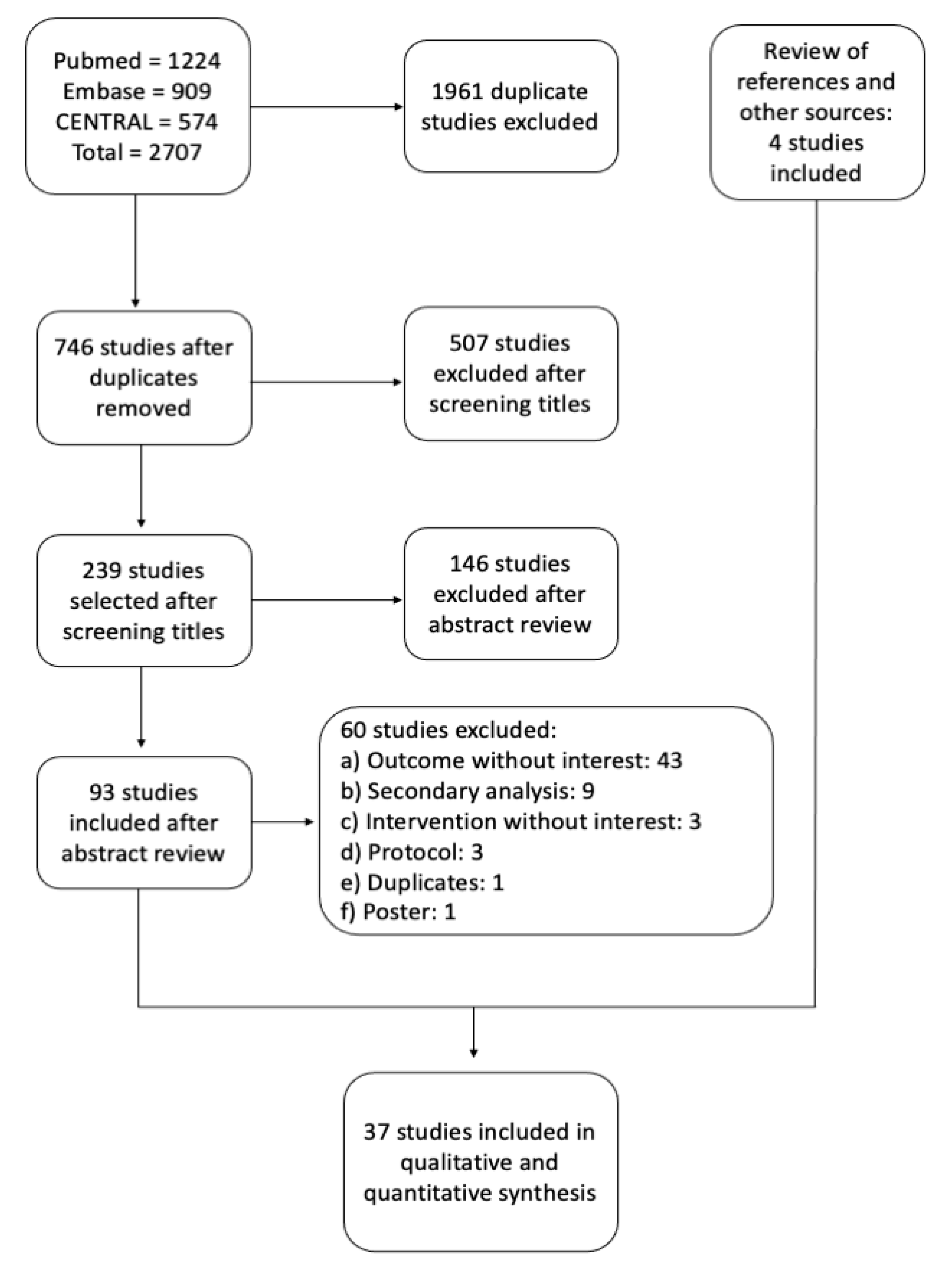
2.3. Literature Search and Selection of Studies
2.4. Data Collection
2.5. Risk of Bias
2.6. Statistical Analyses
3. Results
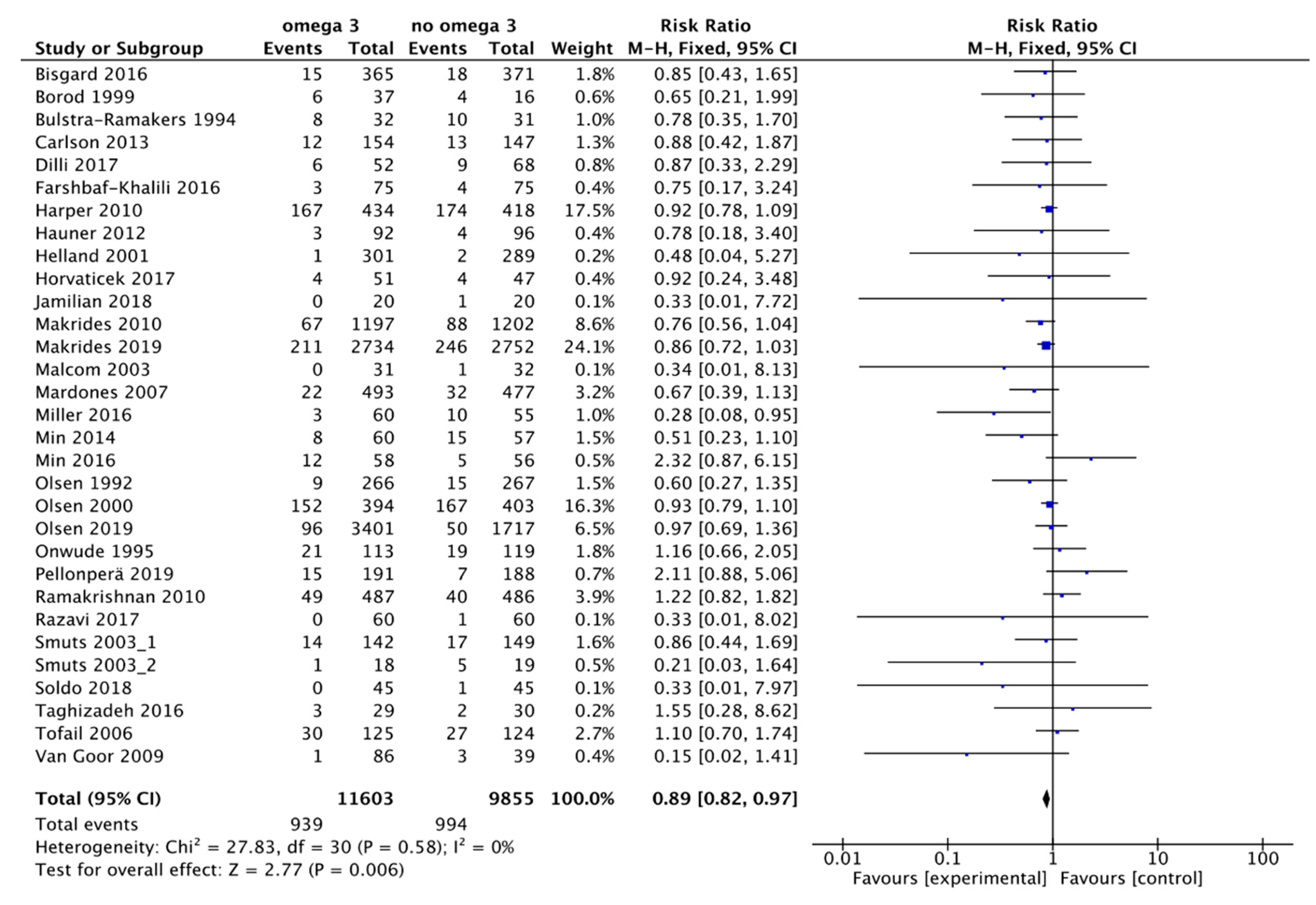
3.1. Main Outcome: Preterm Birth Less Than 37 Weeks
3.2. Secondary Outcomes
3.2.1. Early Preterm Delivery
3.2.2. Preeclampsia or Pregnancy-Induced Hypertension
3.2.3. Intrauterine Growth Restriction (IUGR)
3.2.4. Fetal Death
3.2.5. Neonatal Death
3.3. Sensitivity Analysis
3.4. Risk of Bias
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Blencowe, H.; Cousens, S.; Oestergaard, M.Z.; Chou, D.; Moller, A.B.; Narwal, R.; Adler, A.; Vera Garcia, C.; Rohde, S.; Say, L.; et al. National, regional, and worldwide estimates of preterm birth rates in the year 2010 with time trends since 1990 for selected countries: A systematic analysis and implications. Lancet 2012, 379, 2162–2172. [Google Scholar] [CrossRef] [Green Version]
- Blencowe, H.; Cousens, S.; Chou, D.; Oestergaard, M.; Say, L.; Moller, A.B.; Kinney, M.; Lawn, J. Born Too Soon: The global epidemiology of 15 million preterm births Understanding the data Preterm birth—What is it? Defining preterm birth. Reprod. Health 2013, 10, 1–13. [Google Scholar] [CrossRef] [Green Version]
- Adams, M.M.; Elam-Evans, L.D.; Wilson, H.G.; Gilbertz, D.A. Rates of and Factors Associated with Recurrence of Preterm Delivery. Obstet. Gynecol. Surv. 2000, 55, 534–536. [Google Scholar] [CrossRef]
- Spong, C.Y. Prediction and Prevention of Recurrent Spontaneous Preterm Birth. Obstet. Gynecol. 2007, 110, 405–415. [Google Scholar] [CrossRef]
- Carlson, S.E.; Colombo, J. Docosahexaenoic Acid and Arachidonic Acid Nutrition in Early Development. Adv. Pediatr. 2016, 63, 453–471. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Institute of Medicine. Dietary Reference Intakes for Energy, Carbohydrate, Fiber, Fat, Fatty Acids, Cholesterol, Protein, and Amino Acids; Institute of Medicine: Washington, DC, USA, 2005; ISBN 030908525X. [Google Scholar]
- Food and Agriculture Organization FAO. Fats and Fatty Acids in Human Nutrition. Proceedings of the Joint FAO/WHO Expert Consultation; FAO: Rome, Italy, 2008; Volume 55. [Google Scholar]
- Olsen, S.F.; Halldorsson, T.I.; Thorne-Lyman, A.L.; Strøm, M.; Gørtz, S.; Granstrøm, C.; Nielsen, P.H.; Wohlfahrt, J.; Lykke, J.A.; Langhoff-Roos, J.; et al. Plasma Concentrations of Long Chain N-3 Fatty Acids in Early and Mid-Pregnancy and Risk of Early Preterm Birth. EBioMedicine 2018, 35, 325–333. [Google Scholar] [CrossRef] [Green Version]
- Wadhwani, N.; Patil, V.; Pisal, H.; Joshi, A.; Mehendale, S.; Gupte, S.; Wagh, G.; Joshi, S. Altered maternal proportions of long chain polyunsaturated fatty acids and their transport leads to disturbed fetal stores in preeclampsia. Prostaglandins Leukot. Essent. Fat. Acids 2014, 91, 21–30. [Google Scholar] [CrossRef] [PubMed]
- Olsen, S.F.; Secher, N.J.; Tabor, A.; Weber, T.; Walker, J.J.; Gluud, C. Randomised clinical trials of fish oil supplementation in high risk pregnancies. BJOG Int. J. Obstet. Gynaecol. 2000, 107, 382–395. [Google Scholar] [CrossRef] [PubMed]
- Borod, E.; Atkinson, R.; Barclay, W.R.; Carlson, S.E. Effects of third trimester consumption of eggs high in docosahexaenoic acid on docosahexaenoic acid status and pregnancy. Lipids 1999, 34, 1999. [Google Scholar] [CrossRef] [PubMed]
- Carlson, S.E.; Colombo, J.; Gajewski, B.J.; Gustafson, K.M.; Mundy, D.; Yeast, J.; Georgieff, M.K.; Markley, L.A.; Kerling, E.H.; Shaddy, D.J. DHA supplementation and pregnancy outcomes. Am. J. Clin. Nutr. 2013, 97, 808–815. [Google Scholar] [CrossRef] [Green Version]
- Harper, M.; Thom, E.; Klebanoff, M.A.; Thorp, J.; Sorokin, Y.; Varner, M.W.; Wapner, R.J.; Caritis, S.N.; Iams, J.D.; Carpenter, M.W.; et al. Omega-3 fatty acid supplementation to prevent recurrent preterm birth: A randomized controlled trial. Obstet. Gynecol. 2010, 115, 234–242. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Makrides, M.; Gibson, R.A.; McPhee, A.J.; Yelland, L.; Quinlivan, J.; Ryan, P. DOMInO Investigative Team, and the Effect of DHA Supplementation during Pregnancy on Maternal Depression and Neurodevelopment of Young Children. JAMA 2010, 304, 1675. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Olsen, S.F.; Halldorsson, T.I.; Li, M.; Strøm, M.; Mao, Y.; Che, Y.; Wang, Y.; Duan, F.; Olsen, J.; Zhou, W. Examining the Effect of Fish Oil Supplementation in Chinese Pregnant Women on Gestation Duration and Risk of Preterm Delivery. J. Nutr. 2019, 149, 1942–1951. [Google Scholar] [CrossRef] [PubMed]
- Mosaad, E.; Peiris, H.N.; Holland, O.; Morean Garcia, I.; Mitchell, M.D. The Role(s) of Eicosanoids and Exosomes in Human Parturition. Front. Physiol. 2020, 11, 594313. [Google Scholar] [CrossRef]
- Ishihara, T.; Yoshida, M.; Arita, M. Omega-3 fatty acid-derived mediators that control inflammation and tissue homeostasis. Int. Immunol. 2019, 31, 559–567. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Yamashita, A.; Kawana, K.; Tomio, K.; Taguchi, A.; Isobe, Y.; Iwamoto, R.; Masuda, K.; Furuya, H.; Nagamatsu, T.; Nagasaka, K.; et al. Increased tissue levels of omega-3 polyunsaturated fatty acids prevents pathological preterm birth. Sci. Rep. 2013, 3, 1–7. [Google Scholar] [CrossRef]
- Olsen, S.F.; Secher, N.J.; Björnsson, S.; Weber, T.; Atke, A. The potential benefits of using fish oil in relation to preterm labor: The case for a randomized controlled trial? Acta Obstet. Gynecol. Scand. 2003, 82, 978–982. [Google Scholar] [CrossRef]
- Higgins, J.P.T.; Altman, D.G.; Gotzsche, P.C.; Juni, P.; Moher, D.; Oxman, A.D.; Savovic, J.; Schulz, K.F.; Weeks, L.; Sterne, J.A.C. The Cochrane Collaboration’s tool for assessing risk of bias in randomised trials. BMJ 2011, 343, d5928. [Google Scholar] [CrossRef] [Green Version]
- Linsell, L.; Johnson, S.; Wolke, D.; O’Reilly, H.; Morris, J.K.; Kurinczuk, J.J.; Marlow, N. Cognitive trajectories from infancy to early adulthood following birth before 26 weeks of gestation: A prospective, population-based cohort study. Arch. Dis. Child. 2018, 103, 363–370. [Google Scholar] [CrossRef] [Green Version]
- Liu, L.; Johnson, H.L.; Cousens, S.; Perin, J.; Scott, S.; Lawn, J.E.; Rudan, I.; Campbell, H.; Cibulskis, R.; Li, M.; et al. Global, regional, and national causes of child mortality: An updated systematic analysis for 2010 with time trends since 2000. Lancet 2012, 379, 2151–2161. [Google Scholar] [CrossRef]
- Godhamgaonkar, A.A.; Wadhwani, N.S.; Joshi, S.R. Exploring the role of LC-PUFA metabolism in pregnancy complications. Prostaglandins Leukot. Essent. Fat. Acids 2020, 163, 102203. [Google Scholar] [CrossRef]
- Lalooha, F. Evaluation of the effect of omega-3 supplements in the prevention of preeclampsia among high risk women. Afr. J. Pharm. Pharmacol. 2012, 6, 2580–2583. [Google Scholar] [CrossRef] [Green Version]
- Ostadrahimi, A.; Salehi-Pourmehr, H.; Mohammad-Alizadeh-Charandabi, S.; Heidarabady, S.; Farshbaf-Khalili, A. The effect of perinatal fish oil supplementation on neurodevelopment and growth of infants: A randomized controlled trial. Eur. J. Nutr. 2017, 57, 2387–2397. [Google Scholar] [CrossRef] [PubMed]
- Smuts, C.M.; Huang, M.; Mundy, D.; Plasse, T.; Major, S.; Carlson, S.E. A randomized trial of docosahexaenoic acid supplementation during the third trimester of pregnancy. Obstet. Gynecol. 2003, 101, 469–479. [Google Scholar] [CrossRef]
- Makrides, M.; Best, K.; Yelland, L.; McPhee, A.; Zhou, S.; Quinlivan, J.; Dodd, J.; Atkinson, E.; Safa, H.; van Dam, J.; et al. A Randomized Trial of Prenatal n−3 Fatty Acid Supplementation and Preterm Delivery. N. Engl. J. Med. 2019, 381, 1035–1045. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Saccone, G.; Berghella, V. Omega-3 supplementation to prevent recurrent preterm birth: A systematic review and metaanalysis of randomized controlled trials. Am. J. Obstet. Gynecol. 2015, 213, 135–140. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kar, S.; Wong, M.; Rogozinska, E.; Thangaratinam, S. Effects of omega-3 fatty acids in prevention of early preterm delivery: A systematic review and meta-analysis of randomized studies. Eur. J. Obstet. Gynecol. Reprod. Biol. 2016, 198, 40–46. [Google Scholar] [CrossRef] [PubMed]
- Harris, M.A.; Reece, M.S.; McGregor, J.A.; Wilson, J.W.; Burke, S.M.; Wheeler, M.; Anderson, J.E.; Auld, G.W.; French, J.I.; Allen, K.G.D. The Effect of Omega-3 Docosahexaenoic Acid Supplementation on Gestational Length: Randomized Trial of Supplementation Compared to Nutrition Education for Increasing n-3 Intake from Foods. Biomed Res. Int. 2015, 2015, 1–8. [Google Scholar] [CrossRef] [Green Version]
- Cholewski, M.; Tomczykowa, M.; Tomczyk, M. A Comprehensive Review of Chemistry, Sources and Bioavailability of Omega-3 Fatty Acids. Nutrients 2018, 10, 1662. [Google Scholar] [CrossRef] [Green Version]
- von Schacky, C. Omega-3 Fatty Acids in Pregnancy—The Case for a Target Omega-3 Index. Nutrients 2020, 12, 898. [Google Scholar] [CrossRef] [Green Version]
- Jamilian, M.; Samimi, M.; Mirhosseini, N.; Ebrahimi, F.A.; Aghadavod, E.; Taghizadeh, M.; Asemi, Z. A randomized double-blinded, placebo-controlled trial investigating the effect of fish oil supplementation on gene expression related to insulin action, blood lipids, and inflammation in gestational diabetes mellitus-fish oil supplementation and gestation. Nutrients 2018, 10, 163. [Google Scholar] [CrossRef] [Green Version]
- Bisgaard, H.; Stokholm, J.; Chawes, B.L.; Vissing, N.H.; Bjarnadóttir, E.; Schoos, A.-M.M.; Wolsk, H.M.; Pedersen, T.M.; Vinding, R.K.; Thorsteinsdóttir, S.; et al. Fish Oil–Derived Fatty Acids in Pregnancy and Wheeze and Asthma in Offspring. N. Engl. J. Med. 2016, 375, 2530–2539. [Google Scholar] [CrossRef] [PubMed]
- Bulstra-Ramakers, M.T.E.W.; Huisjes, H.J.; Visser, G.H.A. The effects of 3g eicosapentaenoic acid daily on recurrence of intrauterine growth retardation and pregnancy induced hypertension. BJOG Int. J. Obstet. Gynaecol. 1994, 102, 123–126. [Google Scholar] [CrossRef] [PubMed]
- D’Almeida, A.; Carter, J.P.; Anatol, A.; Prost, C. Effects of a Combination of Evening Primrose Oil (Gamma Linolenic Acid) and Fish Oil (Eicosapentaenoic + Docahexaenoic Acid) versus Magnesium, and versus Placebo in Preventing Pre-Eclampsia. Women Health 1992, 19, 117–131. [Google Scholar] [CrossRef] [PubMed]
- De Groot, R.H.M.; Hornstra, G.; Van Houwelingen, A.C.; Roumen, F. Effect of α-linolenic acid supplementation during pregnancy on maternal and neonatal polyunsaturated fatty acid status and pregnancy outcome. Am. J. Clin. Nutr. 2004, 79, 251–260. [Google Scholar] [CrossRef] [Green Version]
- Dilli, D.; Doğan, N.N.; İpek, M.Ş.; Çavuş, Y.; Ceylaner, S.; Doğan, H.; Dursun, A.; Küçüközkan, T.; Zenciroğlu, A. MaFOS-GDM trial: Maternal fish oil supplementation in women with gestational diabetes and cord blood DNA methylation at insulin like growth factor-1 (IGF-1) gene. Clin. Nutr. ESPEN 2017, 23, 73–78. [Google Scholar] [CrossRef]
- Haghiac, M.; Yang, X.H.; Presley, L.; Smith, S.; Dettelback, S.; Minium, J.; Belury, M.A.; Catalano, P.M.; Hauguel-De Mouzon, S. Dietary omega-3 fatty acid supplementation reduces inflammation in obese pregnant women: A randomized double-blind controlled clinical trial. PLoS ONE 2015, 10, 1–14. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hauner, H.; Much, D.; Vollhardt, C.; Brunner, S.; Schmid, D.; Sedlmeier, E.M.; Heimberg, E.; Schuster, T.; Zimmermann, A.; Schneider, K.T.M.; et al. Effect of reducing the n-6:n-3 long-chain PUFA ratio during pregnancy and lactation on infant adipose tissue growth within the first year of life: An open-label randomized controlled trial. Am. J. Clin. Nutr. 2012, 95, 383–394. [Google Scholar] [CrossRef] [Green Version]
- Helland, I.B.; Saugstad, O.D.; Smith, L.; Saarem, K.; Solvoll, K.; Ganes, T.; Drevon, C.A. Similar effects on infants of n-3 and n-6 fatty acids supplementation to pregnant and lactating women. Pediatrics 2001, 108. [Google Scholar] [CrossRef] [Green Version]
- Horvaticek, M.; Djelmis, J.; Ivanisevic, M.; Oreskovic, S.; Herman, M. Effect of eicosapentaenoic acid and docosahexaenoic acid supplementation on C-peptide preservation in pregnant women with type-1 diabetes: Randomized placebo controlled clinical trial. Eur. J. Clin. Nutr. 2017, 71, 968–972. [Google Scholar] [CrossRef]
- Taghizadeh, M.; Jamilian, M.; Mazloomi, M.; Sanami, M.; Asemi, Z. A randomized-controlled clinical trial investigating the effect of omega-3 fatty acids and Vitamin E co-supplementation on markers of insulin metabolism and lipid profiles in gestational diabetes. J. Clin. Lipidol. 2016, 10, 386–393. [Google Scholar] [CrossRef]
- Farshbaf-Khalili, A.; Mohammad-Alizadeh, S.; Farshbaf-Khalili, A.; Mohammadi, F.; Ostadrahimi, A. Fish-Oil Supplementation and Maternal Mental Health: A Triple-Blind, Randomized Controlled Trial. Iran. Red Crescent Med. J. 2016, 19. [Google Scholar] [CrossRef]
- Malcolm, C.A.; Hamilton, R.; McCulloch, D.L.; Montgomery, C.; Weaver, L.T. Scotopic Electroretinogram in Term Infants Born of Mothers Supplemented with Docosahexaenoic Acid during Pregnancy. Investig. Opthalmology Vis. Sci. 2003, 44, 3685. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Mardones, F.; Urrutia, M.-T.; Villarroel, L.; Rioseco, A.; Castillo, O.; Rozowski, J.; Tapia, J.-L.; Bastias, G.; Bacallao, J.; Rojas, I. Effects of a dairy product fortified with multiple micronutrients and omega-3 fatty acids on birth weight and gestation duration in pregnant Chilean women. Public Health Nutr. 2007, 11, 30–40. [Google Scholar] [CrossRef]
- Miller, S.; Harris, M.A.; Baker, S.; Davalos, D.; Clark, A.; Mc Girr, K. Intake of Total Omega-3 Docosahexaenoic Acid Associated with Increased Gestational Length and Improved Cognitive Performance at 1 Year of Age. J. Nutr. Health Food Eng. 2016, 5, 642–651. [Google Scholar] [CrossRef]
- Min, Y.; Djahanbakhch, O.; Hutchinson, J.; Bhullar, A.S.; Raveendran, M.; Hallot, A.; Eram, S.; Namugere, I.; Nateghian, S.; Ghebremeskel, K. Effect of docosahexaenoic acid-enriched fish oil supplementation in pregnant women with type 2 diabetes on membrane fatty acids and fetal body composition-double-blinded randomized placebo-controlled trial. Diabet. Med. 2014, 31, 1331–1340. [Google Scholar] [CrossRef] [PubMed]
- Min, Y.; Djahanbakhch, O.; Hutchinson, J.; Eram, S.; Bhullar, A.S.; Namugere, I.; Ghebremeskel, K. Efficacy of docosahexaenoic acid-enriched formula to enhance maternal and fetal blood docosahexaenoic acid levels: Randomized double-blinded placebo-controlled trial of pregnant women with gestational diabetes mellitus. Clin. Nutr. 2016, 35, 608–614. [Google Scholar] [CrossRef] [Green Version]
- Mozurkewich, E.; Clinton, C.; Chilimigras, J.; Hamilton, S.; Allbaugh, L.; Berman, D.; Marcus, S.; Vazquez, D.; Romero, V.; Treadwell, M.; et al. The Mothers, Omega-3 & Mental Health Study: A double-blind, randomized controlled trial. Am. J. Obstet. Gynecol. 2013, 208, S19–S20. [Google Scholar] [CrossRef]
- Olsen, S.F.; Sørensen, J.D.; Secher, N.J.; Hedegaard, M.; Brink Henriksen, T.; Hansen, H.S.; Grant, A. Randomised controlled trial of effect of fish-oil supplementation on pregnancy duration. Lancet 1992, 339, 1003–1007. [Google Scholar] [CrossRef]
- Onwude, J.L.; Lilford, R.J.; Hjartardottir, H.; Staines, A.; Tuffnell, D. A randomised double blind placebo controlled trial of fish oil in high risk pregnancy. BJOG Int. J. Obstet. Gynaecol. 1995, 102, 95–100. [Google Scholar] [CrossRef] [PubMed]
- Pellonperä, O.; Mokkala, K.; Houttu, N.; Vahlberg, T.; Koivuniemi, E.; Tertti, K.; Rönnemaa, T.; Laitinen, K. Efficacy of fish oil and/or probiotic intervention on the incidence of gestational diabetes mellitus in an at-risk group of overweight and obese women: A randomized, placebo-controlled, double-blind clinical trial. Diabetes Care 2019, 42, 1009–1017. [Google Scholar] [CrossRef]
- Ramakrishnan, U.; Stein, A.D.; Parra-Cabrera, S.; Wang, M.; Imhoff-Kunsch, B.; Juárez-Márquez, S.; Rivera, J.; Martorell, R. Effects of docosahexaenoic acid supplementation during pregnancy on gestational age and size at birth: Randomized, double-blind, placebo-controlled trial in Mexico. Food Nutr. Bull. 2010, 31, 108–116. [Google Scholar] [CrossRef]
- Razavi, M.; Jamilian, M.; Samimi, M.; Afshar Ebrahimi, F.; Taghizadeh, M.; Bekhradi, R.; Seyed Hosseini, E.; Haddad Kashani, H.; Karamali, M.; Asemi, Z. The effects of Vitamin D and omega-3 fatty acids co-supplementation on biomarkers of inflammation, oxidative stress and pregnancy outcomes in patients with gestational diabetes. Nutr. Metab. 2017, 14, 1–9. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Smuts, C.M.; Borod, E.; Peeples, J.M.; Carlson, S.E. High-DHA eggs: Feasibility as a means to enhance circulating DHA in mother and infant. Lipids 2003, 38, 407–414. [Google Scholar] [CrossRef] [PubMed]
- Soldo, D.; Mikulić-Kajić, M.; Spalldi Barišić, L.; Penava, N.; Orlović, M.; Soldo, N.; Kajić, M. Effect of n-3 long-chain polyunsaturated fatty acids supplementation in healthy mothers on DHA and EPA profiles in maternal and umbilical blood: A randomized controlled trial. J. Perinat. Med. 2018, 47, 200–206. [Google Scholar] [CrossRef]
- Tofail, F.; Kabir, I.; Hamadani, J.D.; Chowdhury, F.; Yesmin, S.; Mehreen, F.; Huda, S.N. Supplement of fish-oil and soy-oil during pregnancy and psychomotor development of infants. J. Health Popul. Nutr. 2006, 24, 48–56. [Google Scholar] [CrossRef] [PubMed]
- Van Goor, S.A.; Dijck-Brouwer, D.A.J.; Hadders-Algra, M.; Doornbos, B.; Erwich, J.J.H.M.; Schaafsma, A.; Muskiet, F.A.J. Human milk arachidonic acid and docosahexaenoic acid contents increase following supplementation during pregnancy and lactation. Prostaglandins Leukot. Essent. Fat. Acids 2009, 80, 65–69. [Google Scholar] [CrossRef]





Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Serra, R.; Peñailillo, R.; Monteiro, L.J.; Monckeberg, M.; Peña, M.; Moyano, L.; Brunner, C.; Vega, G.; Choolani, M.; Illanes, S.E. Supplementation of Omega 3 during Pregnancy and the Risk of Preterm Birth: A Systematic Review and Meta-Analysis. Nutrients 2021, 13, 1704. https://doi.org/10.3390/nu13051704
Serra R, Peñailillo R, Monteiro LJ, Monckeberg M, Peña M, Moyano L, Brunner C, Vega G, Choolani M, Illanes SE. Supplementation of Omega 3 during Pregnancy and the Risk of Preterm Birth: A Systematic Review and Meta-Analysis. Nutrients. 2021; 13(5):1704. https://doi.org/10.3390/nu13051704
Chicago/Turabian StyleSerra, Ramón, Reyna Peñailillo, Lara J. Monteiro, Max Monckeberg, Macarena Peña, Lía Moyano, Camila Brunner, Georgina Vega, Mahesh Choolani, and Sebastián E. Illanes. 2021. "Supplementation of Omega 3 during Pregnancy and the Risk of Preterm Birth: A Systematic Review and Meta-Analysis" Nutrients 13, no. 5: 1704. https://doi.org/10.3390/nu13051704
APA StyleSerra, R., Peñailillo, R., Monteiro, L. J., Monckeberg, M., Peña, M., Moyano, L., Brunner, C., Vega, G., Choolani, M., & Illanes, S. E. (2021). Supplementation of Omega 3 during Pregnancy and the Risk of Preterm Birth: A Systematic Review and Meta-Analysis. Nutrients, 13(5), 1704. https://doi.org/10.3390/nu13051704





