The SNP rs516946 Interacted in the Association of MetS with Dietary Iron among Chinese Males but Not Females
Abstract
1. Introduction
2. Materials and Methods
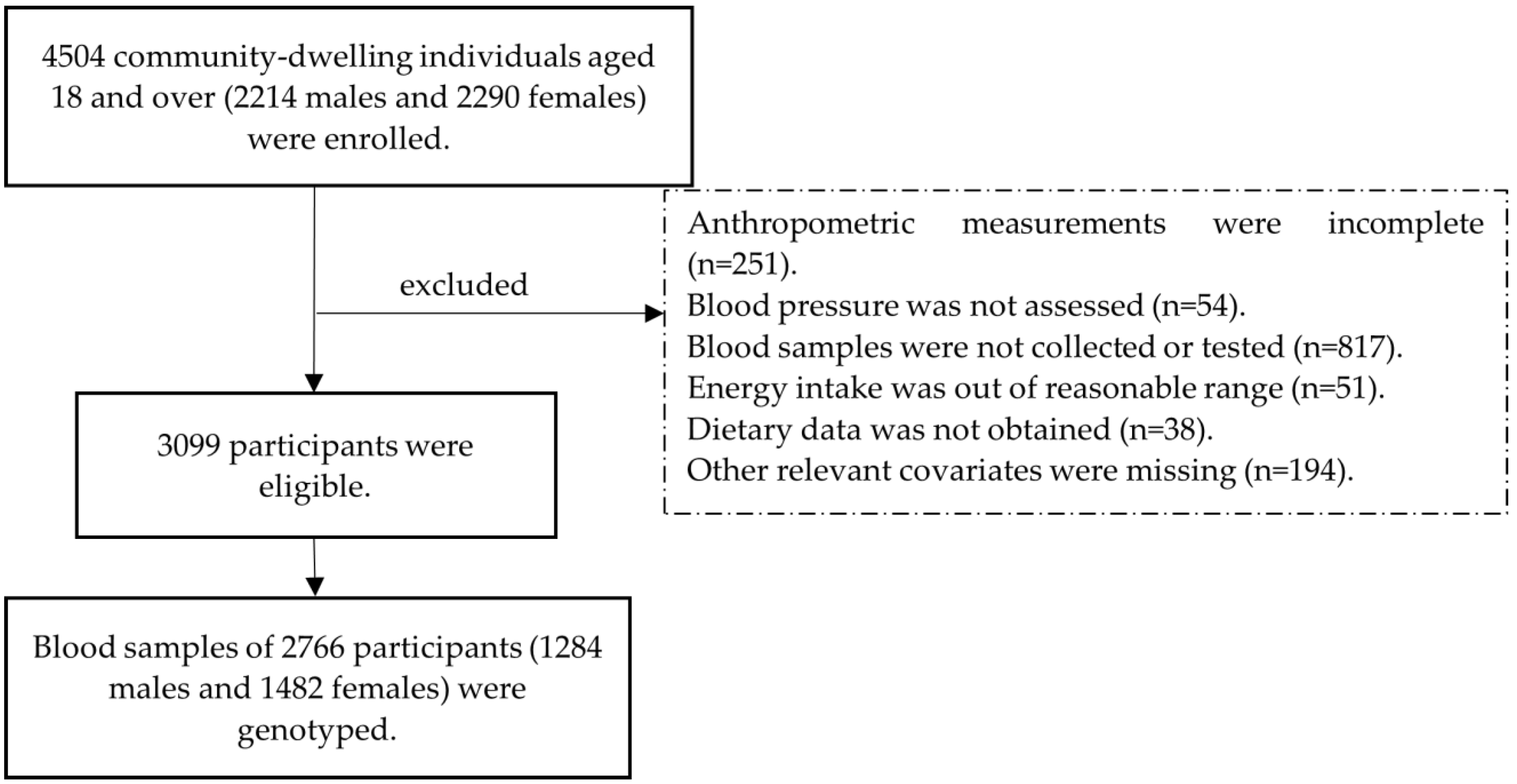
2.1. Study Population
2.2. Dietary Assessment
2.3. Potential Confounders
2.4. Anthropometric and Laboratory Measurements
2.5. Definition of Metabolic Syndrome
2.6. Statistical Analysis
3. Results
3.1. Characteristics of the Participants
3.2. The Associations of MetS Risk with Dietary Iron and the SNP rs516946
3.3. The Associations between Dietary Iron and MetS Risk Stratified by T Allele Presence of rs516946
3.4. The Correlation between Liver Metabolic Indicators and SNP rs516946
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
References
- Lee, L.; Sanders, R.A. Metabolic syndrome. Pediatr. Rev. 2012, 33, 459–468. [Google Scholar] [CrossRef] [PubMed]
- Huang, P.L. A comprehensive definition for metabolic syndrome. Dis. Model. Mech. 2009, 2, 231–237. [Google Scholar] [CrossRef] [PubMed]
- Gu, D.; Reynolds, K.; Wu, X.; Chen, J.; Duan, X.; Reynolds, R.F.; Whelton, P.K.; He, J. Prevalence of the metabolic syndrome and overweight among adults in China. Lancet 2005, 365, 1398–1405. [Google Scholar] [CrossRef]
- Li, Y.; Zhao, L.; Yu, D.; Wang, Z.; Ding, G. Metabolic syndrome prevalence and its risk factors among adults in China: A nationally representative cross-sectional study. PLoS ONE 2018, 13, e199293. [Google Scholar] [CrossRef] [PubMed]
- Zhu, Z.; He, Y.; Wu, F.; Zhao, L.; Wu, C.; Lu, Y.; Zang, J.; Wang, Z.; Sun, J.; Huang, J.; et al. The associations of dietary iron, zinc and magnesium with metabolic syndrome in china’s mega cities. Nutrients 2020, 12, 659. [Google Scholar] [CrossRef]
- Rajpathak, S.N.; Crandall, J.P.; Wylie-Rosett, J.; Kabat, G.C.; Rohan, T.E.; Hu, F.B. The role of iron in type 2 diabetes in humans. Biochim. Biophys. Acta 2009, 1790, 671–681. [Google Scholar] [CrossRef]
- Simcox, J.A.; McClain, D.A. Iron and diabetes risk. Cell Metab. 2013, 17, 329–341. [Google Scholar] [CrossRef]
- Gallagher, P.G.; Tse, W.T.; Scarpa, A.L.; Lux, S.E.; Forget, B.G. Structure and organization of the human ankyrin-1 gene. Basis for complexity of pre-mRNA processing. J. Biol. Chem. 1997, 272, 19220–19228. [Google Scholar] [CrossRef]
- Sun, L.; Zhang, X.; Wang, T.; Chen, M.; Qiao, H. Association of ANK1 variants with new-onset type 2 diabetes in a Han Chinese population from northeast China. Exp. Ther. Med. 2017, 14, 3184–3190. [Google Scholar] [CrossRef]
- Yuki, K.E.; Eva, M.M.; Richer, E.; Chung, D.; Paquet, M.; Cellier, M.; Canonne-Hergaux, F.; Vaulont, S.; Vidal, S.M.; Malo, D. Suppression of hepcidin expression and iron overload mediate Salmonella susceptibility in ankyrin 1 ENU-induced mutant. PLoS ONE 2013, 8, e55331. [Google Scholar] [CrossRef]
- Qin, L.; Nie, Y.; Zhang, H.; Chen, L.; Zhang, D.; Lin, Y.; Ru, K. Identification of new mutations in patients with hereditary spherocytosis by next-generation sequencing. J. Hum. Genet. 2020, 65, 427–434. [Google Scholar] [CrossRef] [PubMed]
- Wang, R.H.; Li, C.; Xu, X.; Zheng, Y.; Xiao, C.; Zerfas, P.; Cooperman, S.; Eckhaus, M.; Rouault, T.; Mishra, L.; et al. A role of SMAD4 in iron metabolism through the positive regulation of hepcidin expression. Cell Metab. 2005, 2, 399–409. [Google Scholar] [CrossRef] [PubMed]
- Nemeth, E.; Ganz, T. The role of hepcidin in iron metabolism. Acta Haematol. 2009, 122, 78–86. [Google Scholar] [CrossRef] [PubMed]
- Mendez-Sanchez, N.; Arrese, M.; Zamora-Valdes, D.; Uribe, M. Current concepts in the pathogenesis of nonalcoholic fatty liver disease. Liver Int. 2007, 27, 423–433. [Google Scholar] [CrossRef] [PubMed]
- Sachinidis, A.; Doumas, M.; Imprialos, K.; Stavropoulos, K.; Katsimardou, A.; Athyros, V.G. Dysmetabolic iron overload in metabolic syndrome. Curr. Pharm. Des. 2020, 26, 1019–1024. [Google Scholar] [CrossRef] [PubMed]
- Rochlani, Y.; Pothineni, N.V.; Kovelamudi, S.; Mehta, J.L. Metabolic syndrome: Pathophysiology, management, and modulation by natural compounds. Ther. Adv. Cardiovasc. Dis. 2017, 11, 215–225. [Google Scholar] [CrossRef]
- Yang, Y.; Wang, G.; Pan, X. Chinese Food Composition Book 1[M], 2nd ed.; Peking University Medical Press: Beijing, China, 2009. [Google Scholar]
- Yang, Y.; Wang, G.; Pan, X. Chinese Food Composition Book 2[M], 1st ed.; Peking University Medical Press: Beijing, China, 2005. [Google Scholar]
- Gong, W.; Liu, A.; Yao, Y.; Ma, Y.; Ding, C.; Song, C.; Yuan, F.; Zhang, Y.; Feng, G.; Chen, Z.; et al. Nutrient Supplement Use among the Chinese Population: A Cross-Sectional Study of the 2010(-)2012 China Nutrition and Health Surveillance. Nutrients 2018, 10, 1733. [Google Scholar] [CrossRef]
- Grundy, S.M.; Cleeman, J.I.; Daniels, S.R.; Donato, K.A.; Eckel, R.H.; Franklin, B.A.; Gordon, D.J.; Krauss, R.M.; Savage, P.J.; Smith, S.J.; et al. Diagnosis and management of the metabolic syndrome: An American Heart Association/National Heart, Lung, and Blood Institute Scientific Statement. Circulation 2005, 112, 2735–2752. [Google Scholar] [CrossRef]
- Knol, M.J.; van der Tweel, I.; Grobbee, D.E.; Numans, M.E.; Geerlings, M.I. Estimating interaction on an additive scale between continuous determinants in a logistic regression model. Int. J. Epidemiol. 2007, 36, 1111–1118. [Google Scholar] [CrossRef]
- Zhu, Z.; Wu, F.; Lu, Y.; Wu, C.; Wang, Z.; Zang, J.; Guo, C.; Jia, X.; Yao, J.; Peng, H.; et al. Total and nonheme dietary iron intake is associated with metabolic syndrome and its components in Chinese men and women. Nutrients 2018, 10, 1663. [Google Scholar] [CrossRef]
- Hansen, J.B.; Moen, I.W.; Mandrup-Poulsen, T. Iron: The hard player in diabetes pathophysiology. Acta Physiol. 2014, 210, 717–732. [Google Scholar] [CrossRef] [PubMed]
- Geissler, C.; Singh, M. Iron, meat and health. Nutrients 2011, 3, 283–316. [Google Scholar] [CrossRef] [PubMed]
- Fang, X.; Min, J.; Wang, F. A dose-response association between serum ferritin and metabolic syndrome? Atherosclerosis 2018, 279, 130–131. [Google Scholar] [CrossRef]
- Cho, G.J.; Shin, J.H.; Yi, K.W.; Park, H.T.; Kim, T.; Hur, J.Y.; Kim, S.H. Serum ferritin levels are associated with metabolic syndrome in postmenopausal women but not in premenopausal women. Menopause 2011, 18, 1120–1124. [Google Scholar] [CrossRef] [PubMed]
- Fernandez-Jimenez, M.C.; Moreno, G.; Wright, I.; Shih, P.C.; Vaquero, M.P.; Remacha, A.F. Iron deficiency in menstruating adult women: Much more than anemia. Women’s Health Rep. 2020, 1, 26–35. [Google Scholar] [CrossRef]
- Mehta, K.J.; Farnaud, S.J.; Sharp, P.A. Iron and liver fibrosis: Mechanistic and clinical aspects. World J. Gastroenterol. 2019, 25, 521–538. [Google Scholar] [CrossRef]

| All | Male | Female | p | |
|---|---|---|---|---|
| n (%) | 2766 (100.0) | 1284 (46.4) | 1482 (53.6) | |
| Age (%) | 0.513 | |||
| 18–44 years | 30.8 | 30.1 | 31.4 | |
| 45–59 years | 36.2 | 36.3 | 36.1 | |
| ≥60 years | 33.0 | 33.6 | 32.6 | |
| Years of Education, years (SD 1) | 9.5 (4.5) | 10.2 (4.0) | 8.9 (4.9) | <0.001 |
| Physical Activity Level (%) | <0.001 | |||
| Sedentary | 84.2 | 78.7 | 89.0 | |
| Moderate | 13.8 | 18.1 | 10.2 | |
| Vigorous | 2.0 | 3.3 | 0.8 | |
| Intentional Physical Exercise (%) | 24.8 | 25.2 | 24.5 | 0.94 |
| Smoking Status, % | ||||
| Never smoked | 72.0 | 40.8 | 98.8 | |
| Former smoker | 5.0 | 10.5 | 0.3 | |
| Current smoker | 23.0 | 48.8 | 1.0 | |
| Alcohol use (%) | <0.001 | |||
| Lifetime abstainers | 80.7 | 64.0 | 94.9 | |
| Nonheavy drinkers | 15.1 | 27.3 | 4.6 | |
| Infrequent heavy drinkers | 1.3 | 2.6 | 0.3 | |
| Frequent heavy drinkers | 3.0 | 6.1 | 0.3 | |
| Dietary Intake | ||||
| Energy, kcal/day (SD) | 1760.9 (842.5) | 1938.5 (884.5) | 1608.9 (773.4) | <0.001 |
| Total iron, mg/day (SD) | 19.7 (16.3) | 22.0 (20.4) | 17.7 (11.3) | <0.001 |
| Heme iron, mg/day (SD) | 1.6 (1.5) | 1.7 (1.6) | 1.5 (1.4) | <0.001 |
| Nonheme iron, mg/day (SD) | 18.4 (16.6) | 20.7 (21.0) | 16.3 (10.7) | <0.001 |
| Ferritin, ng/mL (SD) | 124.5 (122.5) | 165.8 (139.2) | 87.0 (89.8) | <0.001 |
| Metabolic Syndrome (%) | 23.9 | 21.8 | 25.7 | 0.011 |
| Metabolic Syndrome Components | ||||
| Elevated blood pressure (%) | 52.4 | 56.8 | 48.7 | <0.001 |
| Elevated waist circumference (%) | 33.6 | 25.5 | 40.5 | <0.001 |
| Elevated fasting glucose (%) | 23.1 | 25.4 | 21.1 | 0.007 |
| Elevated triglycerides (%) | 27.2 | 30.3 | 24.4 | <0.001 |
| Reduced HDL-C 2, % | 21.0 | 14.8 | 26.2 | <0.001 |
| T allele presence on rs516946 (%) | 23.7 | 21.9 | 25.4 | <0.001 |
| Genotypes of rs516946 (%) | 0.079 | |||
| CC 3 | 76.3 | 78.1 | 74.7 | |
| CT 4 | 22.4 | 20.8 | 23.9 | |
| TT 5 | 1.3 | 1.1 | 1.4 |
| β | p | |
|---|---|---|
| All | ||
| Dietary iron | 0.21 | <0.001 |
| rs516946 | 0.38 | 0.146 |
| Male | ||
| Dietary iron | 0.34 | <0.001 |
| rs516946 | 1.02 | 0.019 |
| Female | ||
| Dietary iron | 0.12 | 0.121 |
| rs516946 | 0.08 | 0.796 |
| β | p | |
|---|---|---|
| All | −0.21 | 0.033 |
| Male | −0.43 | 0.007 |
| Female | −0.11 | 0.381 |
| Gene Site | RERI 2 | AP 3 | S 4 |
|---|---|---|---|
| rs516946 | –0·81 (–2.23, –0.13) | –0.31 (–0.58, –0.08) | 0.70 (0.51, 0.86) |
| Q1 | Q2 | Q3 | Q4 | p-Value for Trend 2 | ||
|---|---|---|---|---|---|---|
| Total Iron Intake (mg/day) | <14.17 | (14.17–17.91) | (17.91–23.87) | ≥23.87 | ||
| n | 352 | 351 | 353 | 351 | ||
| Metabolic syndrome | ||||||
| Model 1 3 | T allele non-carriers | Reference | 1.36 (0.85, 2.16) | 2.06 (1.32, 3.22) | 2.14 (1.38, 3.34) | <0.001 |
| T allele carriers | 1.92 (0.99, 3.76) | 1.46 (0.63, 3.39) | 1.62 (0.76, 3.48) | 1.01 (0.41, 2.45) | 0.218 | |
| Model 2 4 | T allele non-carriers | Reference | 1.32 (0.81, 2.15) | 2.14 (1.31, 3.52) | 2.37 (1.36, 4.14) | <0.001 |
| T allele carriers | 1.78 (0.87, 3.63) | 1.48 (0.58, 3.78) | 1.97 (0.73, 5.32) | 1.81 (0.52, 6.32) | 0.854 | |
| Metabolic syndrome clinical indexes | ||||||
| Elevated blood pressure | ||||||
| Model 1 | T allele non-carriers | Reference | 1.24 (0.85, 1.83) | 1.17 (0.79, 1.72) | 1.32 (0.90, 1.95) | 0.217 |
| T allele carriers | 1.29 (0.67, 2.47) | 1.34 (0.62, 2.88) | 1.21 (0.59, 2.48) | 1.11 (0.51, 2.39) | 0.643 | |
| Model 2 | T allele non-carriers | Reference | 1.28 (0.85, 1.92) | 1.34 (0.87, 2.06) | 1.67 (1.02, 2.74) | 0.052 |
| T allele carriers | 1.12 (0.57, 2.21) | 1.33 (0.58, 3.02) | 1.23 (0.54, 2.81) | 1.37 (0.50, 3.81) | 0.757 | |
| Elevated waist circumference | ||||||
| Model 1 | T allele non-carriers | Reference | 1.52 (0.99, 2.34) | 1.86 (1.22, 2.84) | 2.07 (1.36, 3.15) | <0.001 |
| T allele carriers | 1.57 (0.82, 3.03) | 1.60 (0.72, 3.56) | 1.65 (0.79, 3.45) | 1.76 (0.80, 3.88) | 0.773 | |
| Model 2 | T allele non-carriers | Reference | 1.42 (0.91, 2.23) | 1.68 (1.07, 2.66) | 1.74 (1.04, 2.90) | 0.028 |
| T allele carriers | 1.45 (0.72, 2.92) | 1.66 (0.69, 3.97) | 2.27 (0.91, 5.63) | 3.03 (1.01, 9.14) | 0.166 | |
| Elevated fasting glucose | ||||||
| Model 1 | T allele non-carriers | Reference | 1.02 (0.67, 1.54) | 1.14 (0.76, 1.72) | 1.36 (0.91, 2.03) | 0.112 |
| T allele carriers | 0.66 (0.32, 1.35) | 1.12 (0.46, 2.70) | 1.19 (0.52, 2.69) | 1.11 (0.47, 2.66) | 0.244 | |
| Model 2 | T allele non-carriers | Reference | 1.13 (0.73, 1.75) | 1.34 (0.85, 2.13) | 1.75 (1.04, 2.93) | 0.032 |
| T allele carriers | 0.50 (0.22, 1.11) | 1.02 (0.39, 2.70) | 1.30 (0.48, 3.54) | 1.19 (0.36, 3.95) | 0.141 | |
| Elevated triglycerides | ||||||
| Model 1 | T allele non-carriers | Reference | 1.44 (0.98, 2.12) | 1.58 (1.07, 2.33) | 1.60 (1.09, 2.36) | 0.016 |
| T allele carriers | 1.48 (0.81, 2.71) | 0.94 (0.43, 2.08) | 1.41 (0.70, 2.84) | 1.21 (0.56, 2.59) | 0.890 | |
| Model 2 | T allele non-carriers | Reference | 1.42 (0.94, 2.13) | 1.53 (1.00, 2.36) | 1.63 (1.00, 2.64) | 0.049 |
| T allele carriers | 1.62 (0.85, 3.10) | 0.95 (0.40, 2.26) | 1.70 (0.72, 3.99) | 1.65 (0.57, 4.77) | 0.695 | |
| Reduced HDL-C 5 | ||||||
| Model 1 | T allele non-carriers | Reference | 0.95 (0.58, 1.56) | 1.34 (0.83, 2.14) | 1.12 (0.69, 1.82) | 0.372 |
| T allele carriers | 1.12 (0.52, 2.39) | 0.47 (0.15, 1.46) | 0.88 (0.35, 2.19) | 0.95 (0.36, 2.53) | 0.988 | |
| Model 2 | T allele non-carriers | Reference | 0.97 (0.58, 1.64) | 1.51 (0.88, 2.57) | 1.38 (0.74, 2.56) | 0.156 |
| T allele carriers | 1.16 (0.53, 2.57) | 0.59 (0.18, 1.97) | 1.25 (0.41, 3.83) | 1.81 (0.45, 7.26) | 0.441 | |
| n | r 4 | 95% CI | p | |
|---|---|---|---|---|
| AST and ferritin level 2 | 1404 | 0.258 | (0.21, 0.31) | <0.001 |
| AST and the SNP rs516946 3 | 1400 | 0.049 | (0.00, 0.10) | 0.068 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zhu, Z.; Wang, Z.; Zang, J.; Lu, Y.; Xiao, Z.; Zheng, G.; Wu, F. The SNP rs516946 Interacted in the Association of MetS with Dietary Iron among Chinese Males but Not Females. Nutrients 2022, 14, 2024. https://doi.org/10.3390/nu14102024
Zhu Z, Wang Z, Zang J, Lu Y, Xiao Z, Zheng G, Wu F. The SNP rs516946 Interacted in the Association of MetS with Dietary Iron among Chinese Males but Not Females. Nutrients. 2022; 14(10):2024. https://doi.org/10.3390/nu14102024
Chicago/Turabian StyleZhu, Zhenni, Zhengyuan Wang, Jiajie Zang, Ye Lu, Ziyi Xiao, Guangyong Zheng, and Fan Wu. 2022. "The SNP rs516946 Interacted in the Association of MetS with Dietary Iron among Chinese Males but Not Females" Nutrients 14, no. 10: 2024. https://doi.org/10.3390/nu14102024
APA StyleZhu, Z., Wang, Z., Zang, J., Lu, Y., Xiao, Z., Zheng, G., & Wu, F. (2022). The SNP rs516946 Interacted in the Association of MetS with Dietary Iron among Chinese Males but Not Females. Nutrients, 14(10), 2024. https://doi.org/10.3390/nu14102024







