1. Introduction
Terpenoids belong to the largest class of structurally diverse secondary metabolites present in plants, exerting a variety of diverse pharmacological effects [
1]. A widely used representative of this class of natural products, especially for its intense sweetness (about 300-times sweeter than regular sugar), is stevioside, a diterpenoid glycoside derived from
Stevia rebaudiana Bertoni. Steviol glycosides are composed of a diterpene
ent-kaurene core (steviol), linked to one or more glucose units. Stevia leaves have been utilized by Paraguayan Indians for centuries to sweeten Mate tea. Both stevia extract and stevioside have been known for their healthy properties by local people in South America and used in traditional medicine for centuries. Numerous studies have confirmed various therapeutic benefits of rebaudioside A (a related compound) and stevioside and its metabolic components, such as steviol and isosteviol [
2,
3]. These medical properties include antitumor, antidiarrheal, antihyperglycemic, antihypertensive, anti-inflammatory, diuretic and immunomodulatory activities. The aforementioned effects have driven many researchers to synthetize diverse derivatives of both stevioside and its aglycon steviol [
2,
4]. Stevioside has been authorized as a food additive in many countries, for instance, Japan, Korea, Brazil, the United States and the European Union [
2,
5]. It should be emphasized that stevioside and the products of its hydrolysis, i.e., steviol and isosteviol (ISV), are non-toxic, especially at low doses [
6].
Acidic hydrolysis of stevioside affords a structural isomer of steviol, a tetracyclic diterpenoid isosteviol (ISV). Isosteviol-related compounds, possessing an
ent-beyerane skeleton, have aroused interest because of their numerous pharmacological effects, including antibacterial, anticancer, anti-inflammatory, glucocorticoid agonist and cardioprotective properties [
1,
6]. Regarding examples of anticancer activity, Mizushina et al. [
7] reported that ISV strongly repressed mammalian DNA polymerases and human DNA topoisomerase II. Moreover, in this study, ISV precluded the growth of human cancer cells, with LD
50 values of 84–167 µM. In addition to this, 500 µg of ISV caused a noticeable decrease in 12-O-tetradecanoylphorbol-13-acetate (TPA)-induced inflammation (a repressive effect of 53.0%). Accordingly, Takasaki and coworkers reported that isosteviol displayed significant inhibitory activity in a two-step carcinogenesis assay, where TPA and 7,12-dimethylbenz[α]anthracene (DMBA)-induced mouse skin was used [
8]. Interestingly, Al-Dhabi et al. [
9] investigated in vitro antibacterial, antibiofilm, anticancer, antifungal and antioxidant properties of ISV. The studied compound showed efficacy against bacteria, such as
Staphylococcus aureus,
Staphylococcus epidermidis and
Klebsiella pneumoniae, as well as against
Aspergillus niger,
Candida albicans and
Trichophyton mentagrophytes. ISV also displayed reasonably better antibiofilm activity against
Escherichia coli,
Salmonella typhi and
Pseudomonas aeruginosa. What is more, it showed substantial antioxidant properties and anticancer activity against Vero and MCF7 cell lines. The cardiovascular actions of isosteviol include, among other things, vasodilatation, decreasing cellular reactive oxygen species (ROS) generation and subsequent repressive effects on angiotensin-II-induced cell proliferation and endothelin-1 secretion, protective effect against the development of cardiac hypertrophy through the regulation of transient outward potassium and L-type calcium channels, improvement in H9c2 cardiomyocyte viability, restoring mitochondrial membrane potential and inhibition of cell apoptosis [
6]. In addition, a study by Nordenoft et al. [
10] proved antidiabetic actions of isosteviol—ISV improved glucose and insulin sensitivity in genetically obese diabetic KKay mice. What is more, it improved the lipid profile and upregulated the gene expression of key beta-cell genes, inter-alia insulin regulatory transcription factors. Thus, ISV could be considered in the prevention of obesity complications.
Obesity is a multifactorial metabolic disease characterized by complexity and chronicity (ICD-10 code E66). In recent decades, its prevalence has reached epidemic proportions, affecting almost 20% of the population worldwide [
11]. This entity is mainly classified with body mass index (BMI, kg/m
2) and adults with a BMI of 30 kg/m
2 and above are considered to suffer from obesity. Unfortunately, obesity is viewed through the prism of the associated pathologies rather than an individual entity and, for that reason, it is often underdiagnosed or inadequately treated. The role of obesity in the development of other noncommunicable chronic diseases, such as dyslipidemia, type 2 diabetes mellitus, hypertension, cardiovascular diseases and cancer, has been well established [
12]. It should be noticed that obesity is the second-most-common preventable cause of cancer. At the same time, it possibly constitutes the most common preventable cause of cancer in non-smokers [
13].
The abovementioned non-caloric sweeteners used as alternatives to sucrose have attracted huge attention, not only due to a high incidence of obesity, but also diabetes and dental caries. There is constant demand for low-calorie drinks and food. They are widely used in diets for diabetics and phenylketonuria patients and diets aimed at weight loss in obese individuals. What is more, a multitude of possible biological actions of the analogues in the structure of stevioside may potentially enable the use of these compounds as drugs or food additives, playing an important role not only in weight loss, but also in reducing infectious diseases, enhancing cardiovascular protection and slowing the progress of various oxidative-stress-related diseases, diabetes and cancer. In other words, steviol-related compounds may be beneficial for individuals suffering from obesity and comorbidities.
In the present contribution, a series of isosteviol derivatives synthetized and evaluated for activated coagulation factor X (FXa) inhibitory activity by Shi et al. [
14] was subjected to molecular modelling studies. In their study, Hayashi et al. [
15] revealed a previously unknown role of activated coagulation factor Xa- protease-activated receptor (FXa-PAR) signaling in developing brown adipose tissue (BAT) dysfunction and systemic metabolic disorder in a murine dietary obese model. However, when an FXa inhibitor was administered, it alleviated BAT whitening, enhanced thermogenic response and systemic glucose intolerance upon dietary obesity. Moreover, ROS levels were reduced in BAT. In this light, the suppression of FXa-PAR1 signaling could become a new therapeutic target for the pharmacotherapy of obesity and diabetes. For this reason, this pilot study was conducted on twenty isosteviol derivatives bearing thiourea fragments to establish a mathematical model that may be used for the prediction of FXa inhibitory activity of new potential isosteviol derivatives beneficial for obese patients.
4. Discussion
Currently, obesity is a serious problem for many societies and its prevalence is increasing worldwide. It is related to a pro-coagulant state, which results in the development of numerous comorbidities, for instance, atherosclerotic disease. Coagulation factor X (FX) is involved in the coagulation cascade, which has become the main target of anticoagulant therapy. Activated FX (FXa) exerts pleiotropic biological activities, mediated through protease-activated receptor (PAR) signaling. Hayashi et al. [
15] showed that coagulation factors and protease-activated receptor 1 (PAR1) are upregulated in BAT under metabolic stress. PAR1 is a prevalent form in BAT and coagulation factor-PAR1-mediated signaling contributes to a functional decline in this tissue by excessive mitochondrial production of ROS, resulting in systemic glucose intolerance in a mouse model of diet-induced obesity. According to this study, inhibition of coagulation factor-PAR1 signaling in BAT alleviates metabolic dysfunction [
15]. Kaur et al. [
22] investigated the involvement of the intestinal FXa-PAR2 axis in the regulation of diet-induced obesity in a murine model. Their results suggested that FXa-PAR2 signaling in the intestinal epithelium is an important factor in the regulation of postprandial glucose-dependent insulinotropic polypeptide (GIP) and early onset obesity. In view of the above observations, new therapies for the treatment of obesity and obesity-related disorders are urgently needed and new FXa inhibitors have enormous potential to be used as drugs or possible food additives.
The application of the multivariate adaptive regression splines procedure for model building in the present molecular modelling study led to the establishment of a portfolio of eight QSAR submodels (see
Table 6). It should be noted that five out of eight submodels meet the initial validation requirements and may be used for the prediction of FXa inhibitory activity of isosteviol derivatives bearing thiourea fragments. The submodel that best describes the quantitative structure–activity relationship was selected for predictive purposes. Its precision, accuracy and predictability were additionally confirmed through an extended validation protocol (see
Table 7). However, analysis of
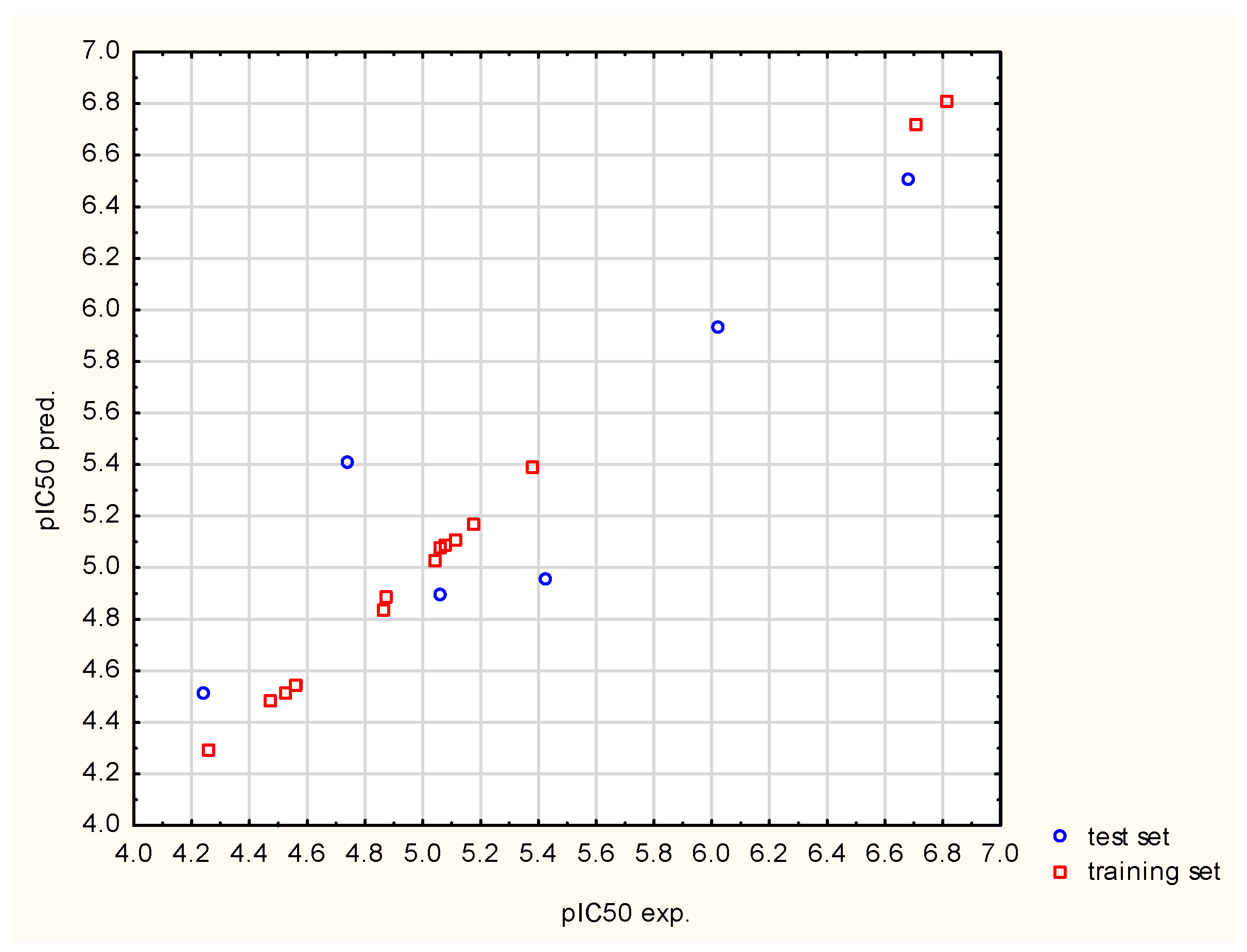
Figure 2 reveals that there is a greater scatter in the experimental data with respect to those determined from the model within the test than in the case of the training set. This is normal and is due to the fact that the test data were not used during the model training phase. Observed differences between experimental and calculated values are acceptable because validation parameters are within the limits described in the literature (see
Table 6). Additionally, it can be noticed that the experimental and calculated values that are oscillating around the straight line represent the complete correlation. This indirectly proves that the residuals in the model values oscillate around the experimental values, which is an expected phenomenon. What attracts great attention is the simplicity of the resulting model, since it incorporates only six basis functions and first-degree interactions. This fact is strongly connected with the algorithm’s operation principle because the relationship is modeled solely on the basis of data, which, in this case, are a set of only twenty compounds [
18]. Moreover, an equal contribution of variables to the model, denoted as the number of appearances in a basis function, is observed (see
Table 4). In order to explore which molecular properties affect the studied activity the most, it is necessary to analyze the nature and number of individual descriptors forming the QSAR model. The following classes of descriptors may be distinguished: 2D Atom Pairs, Weighted Holistic Invariant Molecular (WHIM) descriptors, 3D-MoRSE (Molecular Representation of Structures based on Electron diffraction) descriptors, Radial Distribution Function (RDF) descriptors and GETAWAY (Geometry, Topology, and Atom-Weights Assembly) descriptors. Interestingly, five out of six predictive descriptors encode the molecule’s 3D geometrical properties (two WHIM descriptors, 3D-MoRSE descriptor, RDF descriptor and GETAWAY descriptor) and the last descriptor, i.e., a representative of 2D Atom Pairs, describes the way a property is distributed along the topological structure.
The representative of 2D Atom Pairs, i.e., B01[C-Cl], is the first to appear in the established QSAR model. This class of substructure descriptors, applicable to any pair of atoms and bond types between them, is founded on a two-dimensional representation of a molecule. Those descriptors frequently are Boolean variables encoding the presence or absence of a particular atom pair in each molecule [
23]. The B01[C-Cl] descriptor included in the model is based on counting a chlorine atom in an individual compound and has a positive impact on FXa inhibitory activity. It should be noted that the reported phenomenon is consistent with the results of a study conducted by Shi et al. [
14], in which the positive impact of the introduction of a chlorine atom on the inhibitory activity of studied compounds was also emphasized. The introduction of electron-donating groups into the phenyl ring had a negative influence on FXa inhibitory activity, but the introduction of the chlorine atom had the exact opposite effect.
Weighted holistic invariant molecular descriptors contain global and directional information and are estimated by a principal component analysis on Cartesian coordinates of the atoms weighted in different ways. They encode relevant molecular 3-dimensional information concerning molecular size, shape, symmetry, and atom distribution with respect to invariant reference frames [
21]. There are two types of WHIM descriptors (E2m and L3v) incorporated into the obtained model, with their largest cumulative contribution as about 33%. L3v reflects the size of the molecule, whereas E2m is a variable encoding atomic distribution.
3D-MoRSE (Molecular Representation of Structures based on Electron diffraction) descriptors were designed for encoding the 3D structure of a molecule by a fixed number of variables. Despite the fact that descriptors from this block comprise information pertaining to the whole molecule, they are defined mainly by short-distance atom pairs [
24]. Descriptor Mor06i is a signal 06/weighted by ionization potential, which, in the case of the studied compounds, may increase contributions of chlorine.
Radial distribution function descriptors with RDF070i, included in the elaborated model, are based on the distance distribution in the geometrical representation of a molecule and constitute a radial distribution function code, which exhibits similar characteristics as the 3D-MoRSE code. In addition to information about interatomic distances of a whole molecule, they contain information on bond distances, ring types, planar and nonplanar systems and atom types. RDF descriptors are unique, concerning the three-dimensional arrangement of the atoms, invariable against the translation and rotation of the entire molecule and independent of the number of atoms [
25,
26]. In this study, the presence of the RDF070i descriptor in the QSAR model suggests a certain dependence between FXa inhibitory activity of thiourea isosteviol analogues and the 3D distribution of ionization potential.
The last variable belongs to GETAWAY descriptors. This block of descriptors has been proposed in order to match 3D-molecular geometry, atom relatedness and chemical information with the use of various atomic weighting schemes. These descriptors are a source of local or/and distributed information on molecular structure [
27]. The HATS7s descriptor, which is included in the resulting model, is calculated from the leverage matrix, obtained by the centered atomic coordinates and related to intrinsic properties of an individual molecule.
To summarize, the accomplished model contains one 2D and five 3D descriptors, which suggests that the molecular 3D conformation is very important for the MARSplines modeling process and, as a consequence, for FXa inhibitory activity. In addition, what should be emphasized is the joint use of GETAWAY and WHIM descriptors, with a cumulative share in the model of 50%. This combination of variables provides more predictive models, especially in the case of biological activities, as suggested by Consonni et al. [
27]. The elaborated model has a very high application value, confirmed by an extensive validation protocol and, for that reason, it may be employed to predict FXa inhibitory activity of new isosteviol analogues bearing thiourea fragments. In light of this fact, the MARSplines procedure presented in this study may become either a part of a computer-aided drug design or a QSAR strategy for searching new health-beneficial food additives.