Abstract
Aim: To assess the effectiveness of perioperative psychological interventions provided to patients with clinically severe obesity undergoing bariatric surgery regarding weight loss, BMI, quality of life, and psychosocial health using the Bayesian approach. Methods: We considered randomised trials that assessed the beneficial and harmful effects of perioperative psychological interventions in people with clinically severe obesity undergoing bariatric surgery. We searched four data sources from inception to 3 October 2021. The authors independently selected studies for inclusion, extracted data, and assessed the risk of bias. We conducted a meta-analysis using a Bayesian approach. PROSPERO: CRD42017077724. Results: Of 13,355 identified records, we included nine studies (published in 27 papers with 1060 participants (365 males; 693 females, 2 people with missing data)). Perioperative psychological interventions may provide little or no benefit for BMI (the last reported follow-up: MD [95% credible intervals] = −0.58 [−1.32, 0.15]; BF01 = 0.65; 7 studies; very low certainty of evidence) and weight loss (the last reported follow-up: MD = −0.50 [−2.21, 0.77]; BF01 = 1.24, 9 studies, very low certainty of evidence). Regarding psychosocial outcomes, the direction of the effect was mainly inconsistent, and the certainty of the evidence was low to very low. Conclusions: Evidence is anecdotal according to Bayesian factors and uncertain whether perioperative psychological interventions may affect weight-related and psychosocial outcomes in people with clinically severe obesity undergoing bariatric surgery. As the results are ambiguous, we suggest conducting more high-quality studies in the field to estimate the true effect, its direction, and improve confidence in the body of evidence.
1. Introduction
Recently, the prevalence of obesity has dramatically increased worldwide and has been estimated at 600 million people worldwide [1,2,3]. Nowadays, this problem affects not only high-income countries but low- and middle-income countries as well [4].
Obesity is a chronic condition and is classified in chapter E66 of the International Classification of Diseases [5]. It is characterized by excessive accumulation of adipose tissue, and according to the most common classification (World Health Organisation) is recognized when Body Mass Index (BMI) is ≥30 kg/m2 in adults [6].
Clinically severe obesity (CSO) is defined as a BMI of at least 40 kg/m2 or at least 35 kg/m2 with comorbid conditions such as type 2 diabetes, hypertension, dyslipidaemia, obstructive sleep apnoea, or stress urinary incontinence. People with CSO are more frequently affected by psychosomatic disorders and premature death compared to people without CSO because the condition influences disability, psychosocial well-being, and quality of life (QoL) [7,8,9].
Currently, bariatric surgery (BS) is the most effective treatment people with CSO [10,11,12]. However, it can be associated with adverse postsurgical outcomes, i.e., weight regain, occurrence of maladaptive eating behaviours, deterioration of the QoL, and others [13,14,15,16].
International guidelines and recommendations highlight a need for employing a multidisciplinary approach of care incorporating different types of support: psychological, dietary, or physical activity [17,18,19]. However, there are no recommendations on the type of perioperative psychological interventions (PPIs) and their optimal timing with respect to surgery [20]. These interventions could be helpful for patients who, due to low self-confidence, low self-efficacy, rigid patterns of behaviour, or cognitive schemas, find it difficult to comply with postoperative restrictions [21,22,23,24,25].
There is evidence showing the positive effect of behavioural interventions alone on weight loss (WL) in people with obesity and assessing their impact on improvement of comorbidities [26,27,28]. PPIs may play a significant role [29]; on the basis of cognitive-behavioural approach, these help to modify antecedent, behaviour, consequences, and thoughts, taking into account emotions, relationship, mindfulness, acceptance, values, goals, and meta-cognition, which are believed to maintain a positive energy balance [30,31,32,33]. According to Kulick et al. [34], PPIs seem to be beneficial as they provide different perspectives, coordinated expertise and skills, and sufficient patient engagement. Moreover, a greater focus on psychosocial functioning may also optimize post-operative weight outcomes [21,22,25,35,36]. According to Brennan et al. [37], the mechanism of action of PPIs for people with obesity is still unclear. So far, several systematic reviews have been published assessing the efficacy of perioperative interventions. However, several of them did not attempt to quantitatively summarise the data with meta-analysis [38,39,40,41,42] or they did not refer to the registered protocol [43,44,45], focused primarily on weight-related outcomes [20,46], did not take into account the outcomes we were interested in [47], or addressed a different population [48]. Thus far, there are no systematic reviews conducted according to the state-of-the-art methodological standards [49] in the discussed area.
The objective of this study was to assess the effectiveness of PPIs provided during the perioperative period to patients with CSO undergoing BS regarding WL, QoL, and psychosocial health.
2. Materials and Methods
We included randomized controlled trials (RCTs) that had to follow participants for a minimum of six months (time frame refers to the intervention itself or a combination of the intervention with a follow-up phase). This study was reported according to the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) statement [50]. The PRISMA checklist is included in the Supplementary Materials (Supplementary Materials, Table S1). A protocol has been registered in PROSPERO: CRD42017077724.
2.1. Eligibility Criteria
2.1.1. Population
We included studies on people of any age with CSO during the perioperative period (after qualification for BS or up to any time post-surgery).
2.1.2. Intervention
We defined PPIs as interventions aimed at changing habits, diet, or physical activity through cognitive (together with psychoeducation) and/or behavioural strategies [45,51,52]. They had to be provided in the form of structured interactions between participants and facilitators (psychologists, psychotherapists, therapists in training, or other trained professionals supervised by a clinical psychologist or therapist) [53]. We did not include interventions focusing solely on physical activity, social support, or dietary advice. Due to the lack of a strict definition of the perioperative period, we planned not to limit the starting timepoint of PPI.
2.1.3. Outcomes
All outcomes had to be measured at baseline (before the start of PPI) and at least six months post surgery, using a validated tool. Primary outcomes were: change in BMI, WL (kg, %WL), change in self-efficacy, and change in QoL. Secondary outcomes were: assessment of maladaptive eating behaviours (such as binge eating, grazing, or emotional overeating) [54], change in psychological symptoms (anxiety, depression), change in problems with relationships, change in cognitive function (memory improvement, executive function, attention), change in alcohol and other substances misuse, and change in suicidal behaviour.
2.2. Search Strategy
We searched the following electronic databases from inception to 3 October 2021: MEDLINE Ovid, Embase, Cochrane Central Register of Controlled Trials (CENTRAL), and ClinicalTrials.gov (search strategies are enclosed in Supplementary Materials, Table S2) without any language restrictions. We checked references of included studies for additional studies.
2.3. Study Selection
Pairs of authors (D.S., P.J., M.J.S., M.S. and K.W.J.) independently screened titles and abstracts and then full texts against eligibility criteria using Covidence software® and Rayyan QCRI. Any disagreement was resolved through discussion or consultation with another reviewer (MMB).
2.4. Data Extraction
Pairs of reviewers (D.S., P.J., M.J.S., M.S. and K.W.J.) independently extracted data on study settings, time frame, methods, details of population, intervention, and outcomes. We resolved any discrepancies by discussion. One review author (D.S.) additionally checked all extracted data again.
Attempts were made to contact corresponding authors in case of missing data or when any clarification was required.
2.5. Risk of Bias (ROB) Assessment
Pairs of reviewers (D.S., P.J., M.J.S., M.S. and K.W.J.) independently assessed ROB in every study using the Cochrane risk of bias assessment tool, according to the Cochrane Handbook [49,55]. Any disagreement was resolved by discussion or consultation with another reviewer (MMB).
2.6. Data Analysis and Synthesis
We expressed continuous data as mean differences with 95% credible intervals (CrI). We calculated pooled estimates using the random-effects model, as we believed there would inevitably be heterogeneity among the included trials [56]. The outcomes were estimated using Bayesian normal priors in JASP [57]. We provided Bayes factors (BF01) and used Lee and Wagenmakers’ thresholds for interpretations [58]. We used Markov Chain Monte Carlo sampler with four chains. Heterogeneity was assessed by analysing τ (group-level standard deviation). It was determined non-significant when ≤1 [59,60]. If performing meta-analysis was not possible, we presented results descriptively.
2.7. Assessment of Reporting Bias
For the investigation of small-study effect that could possibly explain publication bias, at least 10 studies should be included for a certain outcome to be able to produce a viable funnel plot or to run statistical tests for interpretation [61]. As we identified few studies for inclusion, we could not produce funnel plots for our comparisons.
2.8. Certainty of Evidence
We presented the overall certainty of evidence and justifications for each outcome separately as a “Summary of findings” table, in accordance with the GRADE approach. Two review authors (D.S., K.W.J.) independently rated the certainty of the evidence for each outcome.
2.9. Sensitivity Analysis
We performed a sensitivity analysis comparing studies with high, unclear, and low ROB for incomplete outcome data and selective outcome reporting. Additionally, we decided to analyse the PPI effect in subgroups according to the procedures, i.e., restrictive (e.g., LAGB, VBG, or SG), mixed (e.g., RYGB or GB), or both (post hoc analysis).
3. Results
We identified nine trials (published in 27 papers) out of 13,355 references, and 16 were labelled as ongoing (Supplementary Materials, Table S3). We presented details of the study flow on a PRISMA flow diagram (Figure 1) [56]. For detailed characteristics of included studies, see Table 1 and Supplementary Materials, Table S4.
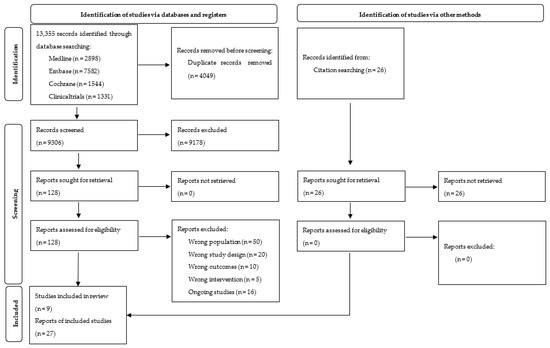
Figure 1.
PRISMA 2020 Flow Diagram.

Table 1.
Characteristics of included studies.
3.1. Included Studies
3.1.1. Participants
Overall, 1060 participants were included. In total, 533 were randomized to intervention and 527 to a comparator group (no information was provided about nine participants). The total sample size ranged from 36 [68] to 240 [62].
All trials reported mean BMI at baseline, which ranged from 35.4 kg/m2 to 51.4 kg/m2 (mean 45.2 ± 6.6) in the intervention group and from 36.5 kg/m2 to 50.90 kg/m2 (mean 44.5 ± 6) in the control group. The proportion of females ranged from 60.3% to 90.1% (mean 75.2%) in the intervention group and from 67% to 94.4% (mean 80%) in the control group. Mean age ranged from 43.5 to 51.0 years (mean 44.7 ± 9.9) in the intervention group, and from 39.3 to 53.9 years (mean 44.2 ± 10.3) in the control group (one study reported mean age of the whole sample to be 40.18 years [66]). Participants underwent different types of BS: Roux-en-Y-gastric by-pass (six studies), laparoscopic adjustable gastric banding (three studies), vertical banded gastroplasty (two studies), sleeve gastrectomy (one study), unspecified gastric bypass (two studies), laparoscopic sleeve gastrectomy (one study), and laparoscopic Roux-en-Y-gastric by-pass (one study).
3.1.2. Intervention
Two studies provided PPIs only before BS [63,70], four trials only after BS [66,67,68,69], and three both before and after surgery [62,64,65]. Three studies applied intervention in the form of group sessions, four individually, and two as both individual and group sessions. The mean number of sessions was 10 (from 3 to 27). Duration of intervention ranged from six weeks [69] to over two years [64], and the longest follow-up was 4 years and 4 weeks [63]. Single session duration ranged from 15 to 180 min. Two studies included interventions that were multidisciplinary [64,65], five trials focused on behavioural-related approaches such as cognitive-behavioural therapy and behavioural therapy [62,63,66,68,70], and two trials focused solely on education-related interventions [67,69].
3.1.3. Outcomes
The provided primary outcomes were: change in BMI, data on WL (kg or %), change in self-efficacy, change in QoL.
Among reported secondary outcomes were the occurrence of maladaptive eating behaviours and change in the severity of psychological symptoms. None of the research provided information about the change in problems with relationships, in cognitive function, in suicidal behaviour, or in alcohol and other substance misuse.
3.2. Excluded Studies
A list of excluded studies with reasons is provided in Supplementary Materials, Table S5.
3.3. ROB in Included Studies
Detailed ROB assessment is presented in Supplementary Materials, Table S4. For an overview of reviewers’ judgments on each ROB item for individual studies and across all research, see Supplementary Materials, Figures S1 and S2. We assessed two trials to be at high risk of bias on four domains [62,64], two studies on three domains [63,65], three studies on two domains [67,69,70], and two trials on one domain [66,68].
3.4. Effects of Interventions
See summary of findings table (Table 2) for the main comparison “PPI in patients with CSO undergoing BS”.

Table 2.
Summary of findings table.
3.4.1. Primary Outcomes
Each of the weight-related outcomes were analysed at three timepoints (6–12 months follow-up, 1–2 years follow-up, and the last reported follow-up of the study).
Weight-Related Outcomes
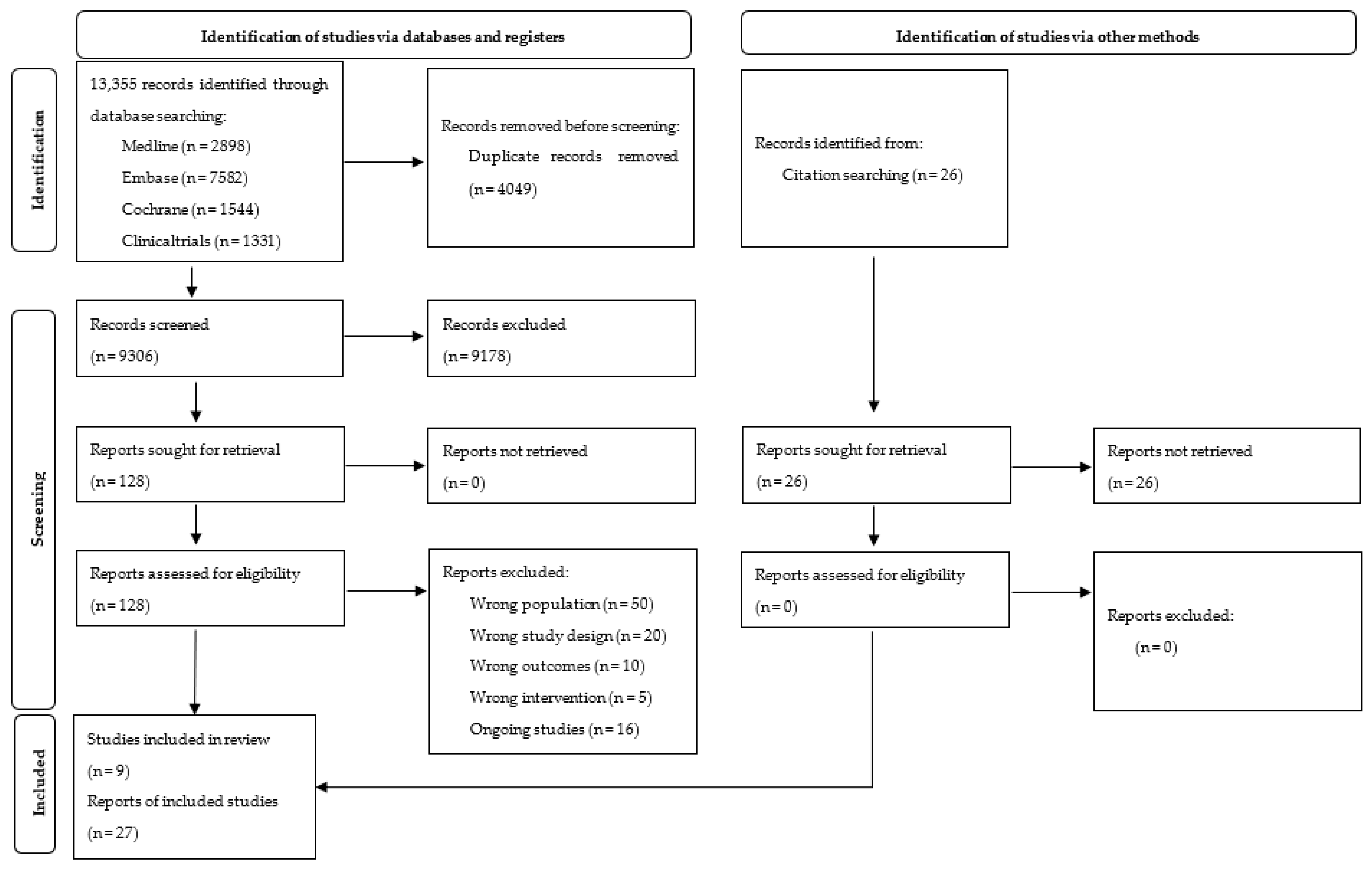
Pooling the studies in a random-effects meta-analysis demonstrated no differences between the intervention and control groups in BMI change from baseline at any of the investigated follow-ups: 6–12 months (−0.29 [−1.6, 0.83]; BF01 = 1.54), 1–2 year (−0.59 [−1.34, 0.12], BF01 = 0.59), and the last follow-up (−0.58 [−1.32, 0.15], BF01 = 0.65) (Figure 2, Supplementary Materials, Figures S3 and S4, Table S6).
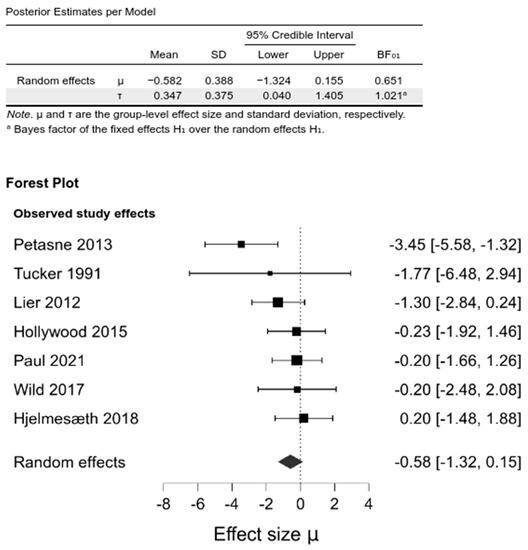
Figure 2.
Comparison between psychological intervention versus any control in the change of BMI at the last follow-up.
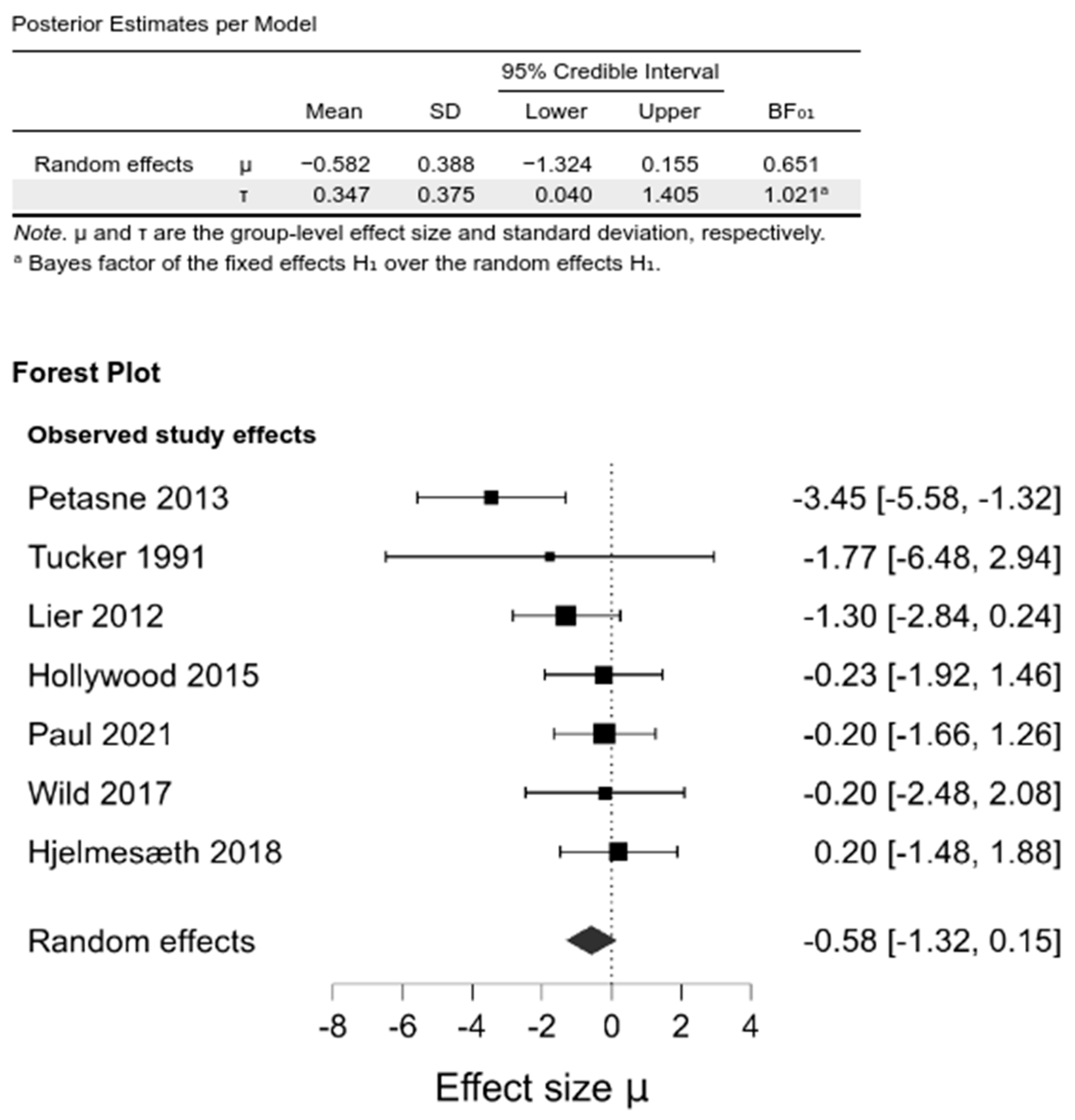
Upon pooling, we did not observe any differences between intervention and control groups in WL at any investigated follow-up (0.14 [−1.43, 1.99]; BF01 = 0.44, −0.56 [−2.2, 0.66]; BF01 = 1.18, −0.50 [−2.21, 0.77]; BF01 = 0.24 for 6–12 months, 1–2 year, and the last follow-up, respectively) (Figure 3, Supplementary Materials, Figures S5 and S6, Table S6).
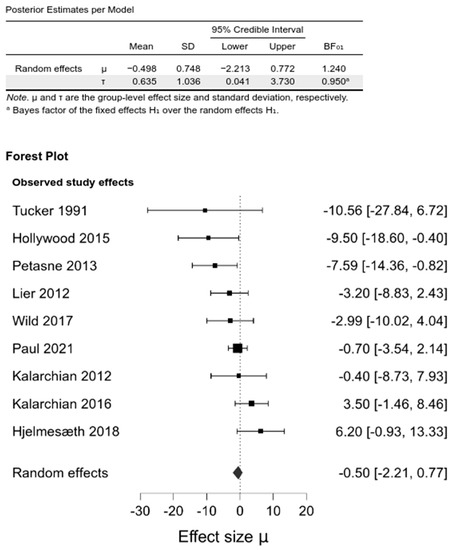
Figure 3.
Comparison between psychological intervention versus any control in the WL at the last follow-up.
Upon performing meta-analysis, we did not observe any differences in percentage WL between the intervention and control groups at any follow-up time-point (−1.60 [−4.68, 1.48], −0.54 [−2.79, 1.07]; BF01 = 1.17, −1.06 [−4.53, 0.92]; BF01 = 0.88, for 6–12 months, 1–2 year, and the last follow-up, respectively).
3.4.2. Secondary Outcomes
The trials described different eating behaviours within binge eating or episodes of bulimia. The direction of the effect was inconsistent (Supplementary Materials, Table S7). The certainty of evidence was very low in the 6–12-month and 1–2-year follow-ups and low for the last reported follow-up.
Two kinds of mood conditions were analysed in included studies: depressive and anxious. The utilised questionnaires included PHQ-9—Patient Health Questionnaire, HADS—Hospital Anxiety and Depression Scale, BDI—Beck Depression Inventory score, or POMS—Profile of Mood States. The direction of the effect was consistently in favour of intervention at 1–2 years’ follow-up. For the other follow-ups, it was inconsistent (Supplementary Materials, Table S7). The certainty of evidence was low for all the follow-ups.
3.4.3. Sensitivity Analysis
We did not observe differences between the subgroups analysed in terms of risk of bias. Additionally, there were two subgroups regarding the type of procedure: mixed and mixed/restrictive but without statistical significant differences between the subgroups (Supplementary Materials, Figure S7).
3.5. Certainty of the Evidence
We rated the certainty of evidence for the outcomes of interest as low or very low (Table 2).
4. Discussion
4.1. Summary of Main Results
This systematic review summarises eight RCTs examining the effect of PPI applied to people with CSO undergoing BS. Studies reported the effects of interventions with respect to BMI, WL, QoL, maladaptive eating behaviours, change in psychological symptoms, and self-efficacy, however inconsistently. Majority of trials did not report adequate information to assess the ROB, and six studies were assessed as high risk of bias on at least two domains.
There was heterogeneity in measurement tools, assessed outcomes, and unit of outcome measures. Based on the identified data, we could not demonstrate the benefit of PPIs with respect to BMI, WL, QoL, maladaptive eating behaviours, change in psychological symptoms, and self-efficacy. According to Bayes factors, efficacy of PPI on WL and BMI is based at most on anecdotal evidence. We found no data for problems with relationships, cognitive function, suicidal behaviour, or alcohol and other substances’ misuse.
4.2. Agreements and Disagreements with Other Studies or Reviews
In contrast to our results, there were several reviews where statistically significant reductions in post-operative weight and BMI were observed. In some of them, both RCTs and non-RCTs were included [45,46,48,71,72]. The effect of intervention presented in reviews where non-RCTs were considered should be analysed consciously because this study design is related to a higher RoB and the potential influence of confounding factors. David et al. noted significant benefits of psychosocial interventions for WL, but they did not synthesise the results [45]. In a study conducted by Świerz et al. [47], who defined perioperative period being 30 days before to 30 days after surgery, it was suggested that psychotherapy might have no effect on WL (the authors assessed the certainty of evidence as low). Stewart et al. [20], who considered any behavioural interventions, with the explicit aim of changing behaviour related to diet and/or physical activity, also found no significant difference in BMI change at 6- and 12-month follow-up after surgery, but regarding WL and percent excess weight change, they observed a greater change in the intervention groups after 2 years post-BS.
Among studies where the authors attempted to synthesize the psychological outcomes, Marshall et al. [48] demonstrated improvement in symptoms of depression, anxiety, and QoL after interventions provided by multidisciplinary team.
Bayesian methods are increasingly used in health care research, including meta-analyses. Nevertheless, to the best of our knowledge, this is the first systematic review in the field of PPI in BS, which utilises this methodology. The use of this approach allows accurate inference results despite the small sample size [73]. Compared to the classical one, it allows for more convenient determining which hypotheses (null or alternative) is more supported by the data (thresholds for Bayesian factors) [58,74].
Another significant strength of our study is including trials of any form of PPI. It also includes the effect of these interventions on other than weight-related outcomes, such as psychosocial outcomes (QoL, self-efficacy, maladaptive eating behaviours, etc.).
5. Conclusions
We explored the impact of different types of PPIs applied in people with CSO undergoing BS. Low to very-low certainty evidence suggests that PPIs might reduce weight regarding middle- and long-term post-surgery WL (follow-ups from 12 mo) and BMI. Additionally, the evidence is insufficient to conclude how PPI, and which specific type of PPI, affects psychosocial outcomes. We found no data for problems with relationships, cognitive function, suicidal behaviour, or alcohol, and other substances misuse.
Our results indicate no strong evidence basis to support the PPIs in people with CSO undergoing BS.
The results of this review should be interpreted with caution, as most of the evidence was rated as very low and low quality due to inconsistency, indirectness, or RoB for many of the outcomes measured.
High-quality trials with long-term follow-up are required to strengthen the body of evidence, as the current evidence is of low to very low methodological quality, and, at most, of anecdotal strength (Bayes factors). Furthermore, consistent measures of psychosocial outcomes using validated tools should be used in future research, and reports should provide adequate and transparent methodological details such as allocation concealment, blinding, attrition, selective reporting, and validity of tools.
6. Differences between the Protocol and Final Review
We added the analyses of the effect of PPIs in subgroups according to the procedures, i.e., restrictive (e.g., LAGB, VBG, or SG), mixed (e.g., RYGB or GB), or both (post-hoc analysis).
Supplementary Materials
The following supporting information can be downloaded at: https://www.mdpi.com/article/10.3390/nu14081592/s1, Table S1: The PRISMA checklist, Table S2: Search strategies, Table S3: List of ongoing studies, Table S4: Included studies, Table S5: List of excluded studies, Figure S1: Graphical presentation of review authors’ judgements about each risk of bias item for each included study, Figure S2: Review authors’ judgements about each risk of bias item presented as percentages across all included studies, Figure S3: Comparison between psychological intervention versus any control in the change of BMI at 6–12 months follow-up, Figure S4: Comparison between psychological intervention versus any control in the change of BMI at 1–2 years follow-up, Table S6: Results from random-effects meta-analyses, Figure S5: Comparison between psychological intervention versus any control in the WL at 6–12 months follow-up, Figure S6: Comparison between psychological intervention versus any control in the WL at 1–2 years follow-up, Table S7: Psychosocial outcomes, Figure S7: Subgroup analyses.
Author Contributions
Conceptualization, D.S., M.J.Ś. and M.M.B.; methodology, D.S., M.J.Ś., P.J. and M.M.B.; software, D.S. and P.J.; validation, D.S. and M.M.B.; formal analysis, D.S., M.J.Ś., P.J. and M.M.B.; investigation, D.S., M.J.Ś., M.S., K.W.J., P.J. and M.M.B.; data curation, D.S., M.J.Ś., M.S., K.W.J. and P.J.; writing—original draft preparation, D.S., M.J.Ś., P.J. and M.M.B.; writing—review and editing, D.S., M.J.Ś., M.S., K.W.J., P.J. and M.M.B.; visualization, D.S. and P.J.; supervision, D.S. and M.M.B.; project administration, D.S.; funding acquisition, P.J. All authors have read and agreed to the published version of the manuscript.
Funding
M.J.Ś.: D.S., K.W.J., M.S. and M.M.B. received support from the European Operational Programme Knowledge Education Development 2014–2020—grant number: MNiSW/2017/101/DIR/NN2. The grant was provided by the Ministry of Science and Higher Education and comprised presentation of the results of the project—‘Systematic Insight into Obesity’—which encompasses this paper. The funding body had no role in the design, in any stage of the review, or in writing the manuscript. Research project supported by program ‘Excellence initiative—research university’ for the University of Science and Technology.
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
The datasets used and/or analysed during the current study are available from the corresponding author on reasonable request.
Acknowledgments
We thank Magdalena Koperny for her help with the checking search strategies.
Conflicts of Interest
The authors declare no conflict of interest.
Abbreviations
| BDI | Beck Depression Inventory score |
| BF | Bayes factors |
| BMI | Body Mass Index |
| BS | Bariatric surgery |
| BT | Behavioural therapy |
| CAU | Care as usual |
| CBT | Cognitive-behavioural therapy |
| COI | Conflict of interest |
| CrI | Credible intervals |
| CSO | Clinically severe obesity |
| GB | Gastric Bypass |
| HADS | Hospital Anxiety and Depression Scale |
| IWQOL | Impact of Weight on Quality of Life |
| LAGB | Laparoscopic adjustable gastric banding |
| LRYGB | Laparoscopic Roux-En-Y Gastric Bypass |
| LSG | Laparoscopic sleeve gastrectomy |
| NR | Not reported |
| PHQ-9 | Patient Health Questionnaire |
| POMS | Profile of Mood States |
| PPI | Perioperative psychological interventions |
| PRISMA | Preferred Reporting Items for Systematic Reviews and Meta-Analyses |
| QoL | Quality of life |
| RCT | Randomized controlled trials |
| RYGB | Roux-En-Y Gastric Bypass |
| SEIQoL | Schedule for the Evaluation of Individual Quality of Life |
| SG | Sleeve Gastrectomy |
| VGB | Vertical Banded Gastroplasty |
| WL | Weight loss |
References
- Nguyen, D.M.; El-Serag, H.B. The Epidemiology of Obesity. Gastrointest. Endosc. Clin. N. Am. 2010, 39, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Flegal, K.M.; Kit, B.K.; Orpana, H.; Graubard, B.I. Association of all-cause mortality with overweight and obesity using standard body mass index categories a systematic review and meta-analysis. JAMA 2013, 309, 71–82. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- World Health Organization. Obesity and Overweight. Available online: http://www.who.int/news-room/fact-sheets/detail/obesity-and-overweight (accessed on 19 May 2021).
- Wang, Y.; Lim, H. The global childhood obesity epidemic and the association between socio-economic status and childhood obesity. Int. Rev. Psychiatry 2012, 24, 176–188. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- World Health Organization. International Statistical Classification of Diseases and Related Health Problems, 10th Revision (ICD-10), Fifth Version, 1st ed.; World Health Organization: Geneva, Switzerland, 2016. [Google Scholar]
- World Health Organization. The Challenge of Obesity in the WHO European Region. Fact Sheet EURO/13/05; World Health Organization: Copenhagen, Denmark; Bucharest, Romania, 2005; Volume 97. [Google Scholar]
- Griffiths, L.; Parsons, T.J.; Hill, A.J. Self-esteem and quality of life in obese children and adolescents: A systematic review. Int. J. Pediatr. Obes. 2010, 5, 282–304. [Google Scholar] [CrossRef] [PubMed]
- Olszanecka-Glinianowicz, M.; Ostrowska, L. Otyłość. In Choroby Wewnętrzne Kompendium Medycyny Praktycznej; Szczeklik, A., Gajewski, P., Eds.; Medycyna Praktyczna: Kraków, Poland, 2020; pp. 2694–2711. [Google Scholar]
- Wild, B.; Herzog, W.; Lechner, S.; Niehoff, D.; Brenner, H.; Müller, H.; Rothenbacher, D.; Stegmaier, C.; Raum, E. Gender specific temporal and cross-sectional associations between BMI-class and symptoms of depression in the elderly. J. Psychosom. Res. 2012, 72, 376–382. [Google Scholar] [CrossRef] [PubMed]
- Lau, D.C.W.; Douketis, J.D.; Morrison, K.M.; Hramiak, I.M.; Sharma, A.M.; Ur, E. 2006 Canadian clinical practice guidelines on the management and prevention of obesity in adults and children. CMAJ Can. Med. Assoc. J. 2007, 176, S1–S13. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Jakobsen, G.S.; Småstuen, M.C.; Sandbu, R.; Nordstrand, N.; Hofsø, D.; Lindberg, M.; Hertel, J.K.; Hjelmesæth, J. Association of bariatric surgery vs medicalobesity treatment with long-term medical complications and obesity-related comorbidities. JAMA 2018, 319, 291–301. [Google Scholar] [CrossRef] [Green Version]
- Colquitt, J.L.; Pickett, K.; Loveman, E.; Frampton, G.K. Surgery for weight loss in adults. Cochrane Database Syst. Rev. 2014, 8, CD003641. [Google Scholar] [CrossRef]
- Lauti, M.; Lemanu, D.; Zeng, I.S.L.; Su’a, B.; Hill, A.G.; MacCormick, A.D. Definition determines weight regain outcomes after sleeve gastrectomy. Surg. Obes. Relat. Dis. 2017, 13, 1123–1129. [Google Scholar] [CrossRef]
- Jirapinyo, P.; Dayyeh, B.K.A.; Thompson, C.C. Weight regain after Roux-en-Y gastric bypass has a large negative impact on the Bariatric Quality of Life Index. BMJ Open Gastroenterol. 2017, 4, e000153. [Google Scholar] [CrossRef]
- Maleckas, A.; Gudaitytė, R.; Petereit, R.; Venclauskas, L.; Veličkienė, D. Weight regain after gastric bypass: Etiology and treatment options. Gland Surg. 2016, 5, 617–624. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Robinson, A.H.; Adler, S.; Stevens, H.B.; Darcy, A.M.; Morton, J.M.; Safer, D.L. What variables are associated with successful weight loss outcomes for bariatric surgery after 1 year? Surg. Obes. Relat. Dis. 2014, 10, 697–704. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Mechanick, J.I.; Apovian, C.; Brethauer, S.; Garvey, W.T.; Joffe, A.M.; Kim, J.; Kushner, R.F.; Lindquist, R.; Pessah-Pollack, R.; Seger, J.; et al. Clinical practice guidelines for the perioperative nutrition, metabolic, and nonsurgical support of patients undergoing bariatric procedures—2019 update: Cosponsored by American Association of Clinical Endocrinologists/American College of Endcrinology. Surg. Obes. Relat. Dis. 2019, 16, 175–247. [Google Scholar] [CrossRef]
- Elkins, G.; Whitfield, P.; Marcus, J.; Symmonds, R.; Rodriguez, J.; Cook, T. Noncompliance with behavioral recommendations following bariatric surgery. Obes. Surg. 2005, 15, 546–551. [Google Scholar] [CrossRef] [PubMed]
- National Institute for Health and Care Excellence. Obesity: Identification, assessment and management. In Clinical Guideline 189; National Institute for Health and Care Excellence: Great Britain, UK, 2014. [Google Scholar]
- Stewart, F.; Avenell, A. Behavioural Interventions for Severe Obesity Before and/or After Bariatric Surgery: A Systematic Review and Meta-analysis. Obes. Surg. 2016, 26, 1203–1214. [Google Scholar] [CrossRef] [PubMed]
- Kalarchian, M.A.; Marcus, M.D. Psychosocial Concerns Following Bariatric Surgery: Current Status. Curr. Obes. Rep. 2019, 8, 1–9. [Google Scholar] [CrossRef]
- Coulman, K.D.; MacKichan, F.; Blazeby, J.M.; Owen-Smith, A. Patient experiences of outcomes of bariatric surgery: A systematic review and qualitative synthesis. Obes. Rev. 2017, 18, 547–559. [Google Scholar] [CrossRef] [Green Version]
- Velapati, S.R.; Shah, M.; Kuchkuntla, A.R.; Abu-Dayyeh, B.; Grothe, K.; Hurt, R.T.; Mundi, M.S. Weight Regain after Bariatric Surgery: Prevalence, Etiology, and Treatment. Curr. Nutr. Rep. 2018, 7, 329–334. [Google Scholar] [CrossRef]
- Kalarchian, M.A.; Marcus, M.D. Management of the bariatric surgery patient: Is there a role for the cognitive behavior therapist? Cogn. Behav. Pract. 2003, 10, 112–119. [Google Scholar] [CrossRef]
- Mitchell, J.E.; Christian, N.J.; Flum, D.R.; Pomp, A.; Pories, W.J.; Wolfe, B.M.; Courcoulas, A.P.; Belle, S.H. Postoperative Behavioral Variables and Weight Change 3 Years after Bariatric Surgery. JAMA Surg. 2016, 151, 752–757. [Google Scholar] [CrossRef]
- Jacob, A.; Moullec, G.; Lavoie, K.L.; Laurin, C.; Cowan, T.; Tisshaw, C.; Kazazian, C.; Raddatz, C.; Bacon, S.L. Impact of cognitive-behavioral interventions on weight loss and psychological outcomes: A meta-analysis. Health Psychol. 2018, 37, 417–432. [Google Scholar] [CrossRef] [PubMed]
- Goodpaster, B.H.; DeLany, J.P.; Otto, A.D.; Kuller, L.; Vockley, J.; South-Paul, J.E.; Thomas, S.B.; Brown, J.; McTigue, K.; Hames, K.C.; et al. Effects of diet and physical activity interventions on weight loss and cardiometabolic risk factors in severely obese adults: A randomized trial. JAMA 2010, 304, 1795–1802. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sogg, S.; Atwood, M.E.; Cassin, S.E.; Hawa, R.; Sockalingam, S. The Role of Psychosocial Interventions in Supporting Medical and Surgical Treatments for Severe Obesity. In Psychological Care in Severe Obesity; Cambridge University Press: Cambridge, UK, 2018. [Google Scholar]
- Kothandan, S.K. School based interventions versus family based interventions in the treatment of childhood obesity—A systematic review. Arch. Public Health 2014, 72, 3. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wadden, T.A.; Brownell, K.D.; Foster, G.D. Obesity: Responding to the global epidemic. J. Consult. Clin. Psychol. 2002, 70, 510. [Google Scholar] [CrossRef] [PubMed]
- Wing, R.R.; Hill, J.O. Successful Weight Loss Maintenance. Annu. Rev. Nutr. 2001, 21, 323–341. [Google Scholar] [CrossRef] [PubMed]
- Fabricatore, A.N. Behavior Therapy and Cognitive-Behavioral Therapy of Obesity: Is There a Difference? J. Am. Diet. Assoc. 2007, 107, 92–99. [Google Scholar] [CrossRef] [PubMed]
- Öst, L.-G. Efficacy of the third wave of behavioral therapies: A systematic review and meta-analysis. Behav. Res. Ther. 2008, 46, 296–321. [Google Scholar] [CrossRef]
- Kulick, D.; Hark, L.; Deen, D. The Bariatric Surgery Patient: A Growing Role for Registered Dietitians. J. Am. Diet. Assoc. 2010, 110, 593–599. [Google Scholar] [CrossRef]
- Jumbe, S.; Hamlet, C.; Meyrick, J. Psychological Aspects of Bariatric Surgery as a Treatment for Obesity. Curr. Obes. Rep. 2017, 6, 71–78. [Google Scholar] [CrossRef] [Green Version]
- Lindekilde, N.; Gladstone, B.P.; Lubeck, M.; Nielsen, J.; Clausen, L.; Vach, W.; Jones, A. The impact of bariatric surgery on quality of life: A systematic review and meta-analysis. Obes. Rev. 2015, 16, 639–651. [Google Scholar] [CrossRef]
- Brennan, L.; Murphy, K.D.; de la Piedad Garcia, X.; Ellis, M.E.; Metzendorf, M.I.; Mckenzie, J.E. Psychological interventions for adults who are overweight or obese. Cochrane Database Syst. Rev. 2018, 3, CD012114. [Google Scholar] [CrossRef]
- Liu, R.H. Do Behavioral Interventions Delivered before Bariatric Surgery Impact Weight Loss in Adults? A Systematic Scoping Review. Bariatr. Surg. Pract. Patient Care 2016, 11, 39–48. [Google Scholar] [CrossRef]
- Groller, K.D. Systematic review of patient education practices in weight loss surgery. Surg. Obes. Relat. Dis. 2017, 13, 1072–1085. [Google Scholar] [CrossRef] [PubMed]
- van Zyl, N.; Andrews, L.; Williamson, H.; Meyrick, J. The effectiveness of psychosocial interventions to support psychological well-being in post-operative bariatric patients: A systematic review of evidence. Obes. Res. Clin. Pract. 2020, 14, 404–420. [Google Scholar] [CrossRef]
- Cheroutre, C.; Guerrien, A.; Rousseau, A. Contributing of Cognitive-Behavioral Therapy in the Context of Bariatric Surgery: A Review of the Literature. Obes. Surg. 2020, 30, 3154–3166. [Google Scholar] [CrossRef]
- Shukla, A.P.; He, D.; Saunders, K.H.; Andrew, C.; Aronne, L.J. Current concepts in management of weight regain following bariatric surgery. Expert Rev. Endocrinol. Metab. 2018, 13, 67–76. [Google Scholar] [CrossRef]
- Andromalos, L.; Crowley, N.; Brown, J.; Craggs-Dino, L.; Handu, D.; Isom, K.; Lynch, A.; DellaValle, D. Nutrition Care in Bariatric Surgery: An Academy Evidence Analysis Center Systematic Review. J. Acad. Nutr. Diet. 2019, 119, 678–686. [Google Scholar] [CrossRef]
- Cassin, S.E.; Atwood, M. Cognitive Behavioural Therapy for Severe Obesity. In Psychiatric Care in Severe Obesity; Springer International Publishing: New York City, NY, USA, 2017. [Google Scholar]
- David, L.A.; Sijercic, I.; Cassin, S.E. Preoperative and post-operative psychosocial interventions for bariatric surgery patients: A systematic review. Obes. Rev. 2020, 21, e12926. [Google Scholar] [CrossRef]
- Julien, C.A.; Lavoie, K.L.; Ribeiro, P.A.B.; Dragomir, A.I.; Mercier, L.A.; Garneau, P.Y.; Pescarus, R.; Bacon, S.L. Behavioral weight management interventions in metabolic and bariatric surgery: A systematic review and meta-analysis investigating optimal delivery timing. Obes. Rev. 2021, 22, e13168. [Google Scholar] [CrossRef]
- Swierz, M.J.; Storman, D.; Jasinska, K.W.; Storman, M.; Staskiewicz, W.; Gorecka, M.; Skuza, A.; Tobola, P.; Bala, M.M. Systematic review and meta-analysis of perioperative behavioral lifestyle and nutritional interventions in bariatric surgery: A call for better research and reporting. Surg. Obes. Relat. Dis. 2020, 16, 2088–2104. [Google Scholar] [CrossRef]
- Marshall, S.; Mackay, H.; Matthews, C.; Maimone, I.R.; Isenring, E. Does intensive multidisciplinary intervention for adults who elect bariatric surgery improve post-operative weight loss, co-morbidities, and quality of life? A systematic review and meta-analysis. Obes. Rev. 2020, 21, e13012. [Google Scholar] [CrossRef] [PubMed]
- Cumpston, M.; Li, T.; Page, M.J.; Chandler, J.; Welch, V.A.; Higgins, J.P.T.; Thomas, J. Cochrane Handbook for Systematic Reviews of Interventions; John Wiley & Sons: Hoboken, NJ, USA, 2019. [Google Scholar]
- Page, M.J.; McKenzie, J.E.; Bossuyt, P.M.; Boutron, I.; Hoffmann, T.C.; Mulrow, C.D.; Shamseer, L.; Tetzlaff, J.M.; Akl, E.A.; Brennan, S.E.; et al. The PRISMA 2020 statement: An updated guideline for reporting systematic reviews. BMJ 2021, 372, 105906. [Google Scholar] [CrossRef]
- Cash, T.F.; Hrabosky, J.I. The Effects of Psychoeducation and Self-Monitoring in a Cognitive-Behavioral Program for Body-Image Improvement. Eat. Disord. 2003, 11, 255–270. [Google Scholar] [CrossRef] [PubMed]
- Lv, N.; Azar, K.M.; Rosas, L.G.; Wulfovich, S.; Xiao, L.; Ma, J. Behavioral lifestyle interventions for moderate and severe obesity: A systematic review. Prev. Med. 2017, 100, 180–193. [Google Scholar] [CrossRef]
- Goldbeck, L.; Fidika, A.; Herle, M.; Quittner, A.L. Psychological interventions for individuals with cystic fibrosis and their families. Cochrane Database Syst. Rev. 2014, 2014, CD003148. [Google Scholar]
- Rusch, M.D.; Andris, D. Maladaptive eating patterns after weight-loss surgery. Nutr. Clin. Pr. 2007, 22, 41–49. [Google Scholar] [CrossRef]
- Higgins, J.P.T.; Altman, D.G.; Gøtzsche, P.C.; Jüni, P.; Moher, D.; Oxman, A.D.; Savović, J.; Schulz, K.F.; Weeks, L.; Sterne, J.A.C.; et al. The Cochrane Collaboration’s tool for assessing risk of bias in randomised trials. BMJ 2011, 343, d5928. [Google Scholar] [CrossRef] [Green Version]
- Wood, L.; Egger, M.; Gluud, L.L.; Schulz, K.F.; Jüni, P.; Altman, D.G.; Gluud, C.; Martin, R.; Wood, A.J.G.; Sterne, J.A.C. Empirical evidence of bias in treatment effect estimates in controlled trials with different interventions and outcomes: Meta-epidemiological study. BMJ 2008, 336, 601–605. [Google Scholar] [CrossRef] [Green Version]
- van Doorn, J.; van den Bergh, D.; Böhm, U.; Dablander, F.; Derks, K.; Draws, T.; Etz, A.; Evans, N.J.; Gronau, Q.F.; Haaf, J.M.; et al. The JASP guidelines for conducting and reporting a Bayesian analysis. Psychon. Bull. Rev. 2020, 28, 813–826. [Google Scholar] [CrossRef]
- Lee, M.D.; Wagenmakers, E.J. Bayesian cognitive modeling: A practical course. In Bayesian Cognitive Modeling: A Practical Course; Cambridge University Press: Cambridge, UK, 2013. [Google Scholar]
- Spiegelhalter, D.J.; Abrams, K.R.; Myles, J.P. Bayesian Approaches to Clinical Trials and Health-Care Evaluation; Wiley: New York, NY, USA, 2004. [Google Scholar] [CrossRef]
- Sterne, J.A.; Gavaghan, D.; Egger, M. Publication and Related Bias in Meta-Analysis. J. Clin. Epidemiol. 2000, 53, 1119–1129. Available online: https://linkinghub.elsevier.com/retrieve/pii/S0895435600002420 (accessed on 8 October 2021). [CrossRef]
- Sterne, J.A.C.; Sutton, A.J.; Ioannidis, J.P.A.; Terrin, N.; Jones, D.R.; Lau, J.; Carpenter, J.; Rücker, G.; Harbord, R.M.; Schmid, C.H.; et al. Recommendations for examining and interpreting funnel plot asymmetry in meta-analyses of randomised controlled trials. BMJ 2011, 343, d4002. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kalarchian, M.A.; Marcus, M.D.; Courcoulas, A.P.; Cheng, Y.; Levine, M.D. Preoperative lifestyle intervention in bariatric surgery: A randomized clinical trial. Surg. Obes. Relat. Dis. 2016, 12, 180–187. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hjelmesæth, J.; Rosenvinge, J.H.; Gade, H.; Friborg, O. Effects of Cognitive Behavioral Therapy on Eating Behaviors, Affective Symptoms, and Weight Loss After Bariatric Surgery: A Randomized Clinical Trial. Obes. Surg. 2019, 29, 61–69. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lier, H.Ø.; Biringer, E.; Stubhaug, B.; Tangen, T. The impact of preoperative counseling on postoperative treatment adherence in bariatric surgery patients: A randomized controlled trial. Patient Educ. Couns. 2012, 87, 336–342. [Google Scholar] [CrossRef]
- Hollywood, A.; Ogden, J.; Pring, C. The Impact of Psychological Support on Psychological Outcomes and Patients’ Experiences of the Bariatric Service 1 and 2 Years after Bariatric Surgery. J. Obes. Bariatr. 2015, 2, 1–7. [Google Scholar]
- Tucker, J.A.; Samo, J.A.; Rand, C.S.W.; Woodward, E.R. Behavioral interventions to promote adaptive eating behavior and lifestyle changes following surgery for obesity: Results of a two-year outcome evaluation. Int. J. Eat. Disord. 1991, 10, 689–698. [Google Scholar] [CrossRef]
- Wild, B.; Hünnemeyer, K.; Sauer, H.; Hain, B.; Mack, I.; Schellberg, D.; Müller-Stich, B.P.; Weiner, R.; Meile, T.; Rudofsky, G.; et al. A 1-year videoconferencing-based psychoeducational group intervention following bariatric surgery: Results of a randomized controlled study. Surg. Obes. Relat. Dis. 2015, 11, 1349–1360. [Google Scholar] [CrossRef]
- Kalarchian, M.A.; Marcus, M.D.; Courcoulas, A.P.; Cheng, Y.; Levine, M.D.; Josbeno, D. Optimizing long-term weight control after bariatric surgery: A pilot study. Surg. Obes. Relat. Dis. 2012, 8, 710–715. [Google Scholar] [CrossRef] [Green Version]
- Nijamkin, M.P.; Campa, A.; Nijamkin, S.S.; Sosa, J. Comprehensive Behavioral-Motivational Nutrition Education Improves Depressive Symptoms Following Bariatric Surgery: A Randomized, Controlled Trial of Obese Hispanic Americans. J. Nutr. Educ. Behav. 2013, 45, 620–626. [Google Scholar] [CrossRef]
- Paul, L.; van der Heiden, C.; van Hoeken, D.; Deen, M.; Vlijm, A.; Klaassen, R.A.; Biter, L.U.; Hoek, H.W. Cognitive Behavioral Therapy Versus Usual Care before Bariatric Surgery: One-Year Follow-Up Results of a Randomized Controlled Trial. Obes. Surg. 2021, 31, 970–979. Available online: https://www.cochranelibrary.com/central/doi/10.1002/central/CN-02204803/full (accessed on 8 October 2021). [CrossRef]
- Beck, N.N.; Johannsen, M.; Støving, R.K.; Mehlsen, M.; Zachariae, R. Do postoperative psychotherapeutic interventions and support groups influence weight loss following bariatric surgery? A systematic review and meta-analysis of randomized and nonrandomized trials. Obes. Surg. 2012, 22, 1790–1797. [Google Scholar] [CrossRef] [PubMed]
- Rudolph, A.; Hilbert, A. Post-operative behavioural management in bariatric surgery: A systematic review and meta-analysis of randomized controlled trials. Obes. Rev. 2013, 14, 292–302. [Google Scholar] [CrossRef] [PubMed]
- Sutton, A.J.; Abrams, K.R. Bayesian methods in meta-analysis and evidence synthesis. Stat. Methods Med. Res. 2001, 10, 277–303. [Google Scholar] [CrossRef] [PubMed]
- Harold, J. Theory of Probability. In Philosophy of Science; Cambridge University Press: Cambridge, UK, 1940. [Google Scholar]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).