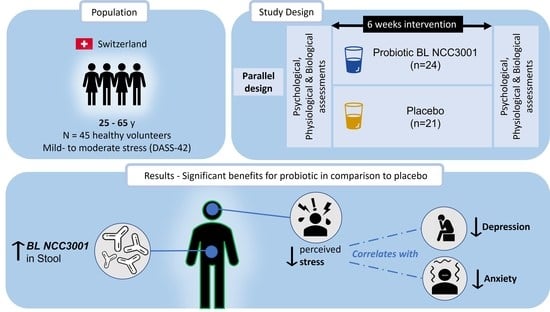
Bifidobacterium longum subsp. longum Reduces Perceived Psychological Stress in Healthy Adults: An Exploratory Clinical Trial
Abstract
:1. Introduction
2. Materials and Methods
2.1. Study Design and Population
2.2. Intervention
2.3. Procedure
2.4. Salivary Cortisol
2.5. Autonomic Parameters
2.6. Fecal BL NCC3001 Abundance Analysis
2.7. Statistical Analysis
3. Results
3.1. Increased Abundance of BL NCC3001 in Stool Shows That the Probiotic Was Successfully Delivered
3.2. Probiotic Intervention Reduced Perceived Stress Score
3.3. Impact of the Probiotic Intervention on Subjective Reports Independent of Acute Stress
3.4. Impact of the Probiotic Intervention on Stress and Autonomic Nervous System Parameters Linked to Acute Stress Response
3.5. Multivariate Analysis Revealed Positive Correlation between Stress Reduction and Reductions in Anxiety and Depression
4. Discussion
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Acknowledgments
Conflicts of Interest
References
- Duchaine, C.S.; Aube, K.; Gilbert-Ouimet, M.; Vezina, M.; Ndjaboue, R.; Massamba, V.; Talbot, D.; Lavigne-Robichaud, M.; Trudel, X.; Pena-Gralle, A.B.; et al. Psychosocial Stressors at Work and the Risk of Sickness Absence Due to a Diagnosed Mental Disorder: A Systematic Review and Meta-analysis. JAMA Psychiatry 2020, 77, 842–851. [Google Scholar] [CrossRef]
- Cryan, J.F.; O’Riordan, K.J.; Cowan, C.S.M.; Sandhu, K.V.; Bastiaanssen, T.F.S.; Boehme, M.; Codagnone, M.G.; Cussotto, S.; Fulling, C.; Golubeva, A.V.; et al. The Microbiota-Gut-Brain Axis. Physiol. Rev. 2019, 99, 1877–2013. [Google Scholar] [CrossRef]
- Zahar, S.; Schneider, N.; Makwana, A.; Chapman, S.; Corthesy, J.; Amico, M.; Hudry, J. Dietary tryptophan-rich protein hydrolysate can acutely impact physiological and psychological measures of mood and stress in healthy adults. Nutr. Neurosci. 2022, 26, 303–312. [Google Scholar] [CrossRef] [PubMed]
- Berding, K.; Bastiaanssen, T.F.S.; Moloney, G.M.; Boscaini, S.; Strain, C.R.; Anesi, A.; Long-Smith, C.; Mattivi, F.; Stanton, C.; Clarke, G.; et al. Feed your microbes to deal with stress: A psychobiotic diet impacts microbial stability and perceived stress in a healthy adult population. Mol. Psychiatry 2023, 28, 601–610. [Google Scholar] [CrossRef]
- Hill, C.; Guarner, F.; Reid, G.; Gibson, G.R.; Merenstein, D.J.; Pot, B.; Morelli, L.; Canani, R.B.; Flint, H.J.; Salminen, S.; et al. Expert consensus document. The International Scientific Association for Probiotics and Prebiotics consensus statement on the scope and appropriate use of the term probiotic. Nat. Rev. Gastroenterol. Hepatol. 2014, 11, 506–514. [Google Scholar] [CrossRef]
- Zhang, N.; Zhang, Y.; Li, M.; Wang, W.; Liu, Z.; Xi, C.; Huang, X.; Liu, J.; Huang, J.; Tian, D.; et al. Efficacy of probiotics on stress in healthy volunteers: A systematic review and meta-analysis based on randomized controlled trials. Brain Behav. 2020, 10, e01699. [Google Scholar] [CrossRef]
- Pinto-Sanchez, M.I.; Hall, G.B.; Ghajar, K.; Nardelli, A.; Bolino, C.; Lau, J.T.; Martin, F.P.; Cominetti, O.; Welsh, C.; Rieder, A.; et al. Probiotic Bifidobacterium longum NCC3001 Reduces Depression Scores and Alters Brain Activity: A Pilot Study in Patients With Irritable Bowel Syndrome. Gastroenterology 2017, 153, 448–459.e8. [Google Scholar] [CrossRef]
- McGowan, R.T.S.; Barnett, H.R.; Czarnecki-Maulden Gail, L.; Si, X.; Perez-Camargo, G.; Martin, F. Tapping into those ‘gut feelings’: Impact of BL999 (Bifidobacterium longum) on anxiety in dogs. In Proceedings of the Veterinary Behavior Symposium, Denver, CO, USA, 12 July 2018; pp. 8–9. [Google Scholar]
- Bercik, P.; Park, A.J.; Sinclair, D.; Khoshdel, A.; Lu, J.; Huang, X.; Deng, Y.; Blennerhassett, P.A.; Fahnestock, M.; Moine, D.; et al. The anxiolytic effect of Bifidobacterium longum NCC3001 involves vagal pathways for gut-brain communication. Neurogastroenterol. Motil. 2011, 23, 1132–1139. [Google Scholar] [CrossRef]
- Bercik, P.; Verdu, E.F.; Foster, J.A.; Macri, J.; Potter, M.; Huang, X.; Malinowski, P.; Jackson, W.; Blennerhassett, P.; Neufeld, K.A.; et al. Chronic gastrointestinal inflammation induces anxiety-like behavior and alters central nervous system biochemistry in mice. Gastroenterology 2010, 139, 2102–2112.e1. [Google Scholar] [CrossRef]
- Fond, G.; Loundou, A.; Hamdani, N.; Boukouaci, W.; Dargel, A.; Oliveira, J.; Roger, M.; Tamouza, R.; Leboyer, M.; Boyer, L. Anxiety and depression comorbidities in irritable bowel syndrome (IBS): A systematic review and meta-analysis. Eur. Arch. Psychiatry Clin. Neurosci. 2014, 264, 651–660. [Google Scholar] [CrossRef] [PubMed]
- Dalile, B.; Vervliet, B.; Bergonzelli, G.; Verbeke, K.; Van Oudenhove, L. Colon-delivered short-chain fatty acids attenuate the cortisol response to psychosocial stress in healthy men: A randomized, placebo-controlled trial. Neuropsychopharmacology 2020, 45, 2257–2266. [Google Scholar] [CrossRef]
- Cohen, S.; Kamarck, T.; Mermelstein, R. A Global Measure of Perceived Stress. J. Health Soc. Behav. 1983, 24, 385–396. [Google Scholar] [CrossRef] [PubMed]
- Buysse, D.J.; Reynolds, C.F., 3rd; Monk, T.H.; Berman, S.R.; Kupfer, D.J. The Pittsburgh Sleep Quality Index: A new instrument for psychiatric practice and research. Psychiatry Res. 1989, 28, 193–213. [Google Scholar] [CrossRef] [PubMed]
- Zigmond, A.S.; Snaith, R.P. The hospital anxiety and depression scale. Acta Psychiatr. Scand. 1983, 67, 361–370. [Google Scholar] [CrossRef] [PubMed]
- Svedlund, J.; Sjodin, I.; Dotevall, G. GSRS—A clinical rating scale for gastrointestinal symptoms in patients with irritable bowel syndrome and peptic ulcer disease. Dig. Dis. Sci. 1988, 33, 129–134. [Google Scholar] [CrossRef]
- Lee, E.-H. Review of the Psychometric Evidence of the Perceived Stress Scale. Asian Nurs. Res. 2012, 6, 121–127. [Google Scholar] [CrossRef]
- Smeets, T.; Cornelisse, S.; Quaedflieg, C.W.; Meyer, T.; Jelicic, M.; Merckelbach, H. Introducing the Maastricht Acute Stress Test (MAST): A quick and non-invasive approach to elicit robust autonomic and glucocorticoid stress responses. Psychoneuroendocrinology 2012, 37, 1998–2008. [Google Scholar] [CrossRef]
- Watson, D.; Clark, L.A.; Tellegen, A. Development and validation of brief measures of positive and negative affect: The PANAS scales. J. Personal. Soc. Psychol. 1988, 54, 1063–1070. [Google Scholar] [CrossRef]
- Marteau, T.M.; Bekker, H. The development of a six-item short-form of the state scale of the Spielberger State-Trait Anxiety Inventory (STAI). Br. J. Clin. Psychol. 1992, 31, 301–306. [Google Scholar] [CrossRef]
- Dalile, B.; La Torre, D.; Kalc, P.; Zoppas, F.; Roye, C.; Loret, C.; Lamothe, L.; Bergonzelli, G.; Courtin, C.M.; Vervliet, B.; et al. Extruded Wheat Bran Consumption Increases Serum Short-Chain Fatty Acids but Does Not Modulate Psychobiological Functions in Healthy Men: A Randomized, Placebo-Controlled Trial. Front. Nutr. 2022, 9, 896154. [Google Scholar] [CrossRef]
- Pruessner, J.C.; Gaab, J.; Hellhammer, D.H.; Lintz, D.; Schommer, N.; Kirschbaum, C. Increasing correlations between personality traits and cortisol stress responses obtained by data aggregation. Psychoneuroendocrinology 1997, 22, 615–625. [Google Scholar] [CrossRef] [PubMed]
- Schmidt, K.; Cowen, P.J.; Harmer, C.J.; Tzortzis, G.; Errington, S.; Burnet, P.W. Prebiotic intake reduces the waking cortisol response and alters emotional bias in healthy volunteers. Psychopharmacology 2015, 232, 1793–1801. [Google Scholar] [CrossRef] [PubMed]
- Liu, C.M.; Aziz, M.; Kachur, S.; Hsueh, P.R.; Huang, Y.T.; Keim, P.; Price, L.B. BactQuant: An enhanced broad-coverage bacterial quantitative real-time PCR assay. BMC Microbiol. 2012, 12, 56. [Google Scholar] [CrossRef]
- Messaoudi, M.; Violle, N.; Bisson, J.F.; Desor, D.; Javelot, H.; Rougeot, C. Beneficial psychological effects of a probiotic formulation (Lactobacillus helveticus R0052 and Bifidobacterium longum R0175) in healthy human volunteers. Gut Microbes 2011, 2, 256–261. [Google Scholar] [CrossRef] [PubMed]
- Patterson, E.; Griffin, S.M.; Ibarra, A.; Ellsiepen, E.; Hellhammer, J. Lacticaseibacillus paracasei Lpc-37(R) improves psychological and physiological markers of stress and anxiety in healthy adults: A randomized, double-blind, placebo-controlled and parallel clinical trial (the Sisu study). Neurobiol. Stress 2020, 13, 100277. [Google Scholar] [CrossRef]
- Siegel, M.P.; Conklin, S.M. Acute intake of B. longum probiotic does not reduce stress, anxiety, or depression in young adults: A pilot study. Brain Behav. Immun Health 2020, 2, 100029. [Google Scholar] [CrossRef]
- Moloney, G.M.; Long-Smith, C.M.; Murphy, A.; Dorland, D.; Hojabri, S.F.; Ramirez, L.O.; Marin, D.C.; Bastiaanssen, T.F.S.; Cusack, A.M.; Berding, K.; et al. Improvements in sleep indices during exam stress due to consumption of a Bifidobacterium longum. Brain Behav. Immun Health 2021, 10, 100174. [Google Scholar] [CrossRef]
- Kelly, J.R.; Allen, A.P.; Temko, A.; Hutch, W.; Kennedy, P.J.; Farid, N.; Murphy, E.; Boylan, G.; Bienenstock, J.; Cryan, J.F.; et al. Lost in translation? The potential psychobiotic Lactobacillus rhamnosus (JB-1) fails to modulate stress or cognitive performance in healthy male subjects. Brain Behav. Immun 2017, 61, 50–59. [Google Scholar] [CrossRef]
- Lew, L.C.; Hor, Y.Y.; Yusoff, N.A.A.; Choi, S.B.; Yusoff, M.S.B.; Roslan, N.S.; Ahmad, A.; Mohammad, J.A.M.; Abdullah, M.; Zakaria, N.; et al. Probiotic Lactobacillus plantarum P8 alleviated stress and anxiety while enhancing memory and cognition in stressed adults: A randomised, double-blind, placebo-controlled study. Clin. Nutr. 2019, 38, 2053–2064. [Google Scholar] [CrossRef]
- Dalile, B.; La Torre, D.; Verbeke, K.; Van Oudenhove, L.; Vervliet, B. When the mind says one thing, but the HPA axis says another: Lack of coherence between subjective and neuroendocrine stress response trajectories in healthy men. Psychoneuroendocrinology 2022, 139, 105692. [Google Scholar] [CrossRef]
- Moffat, S.D.; An, Y.; Resnick, S.M.; Diamond, M.P.; Ferrucci, L. Longitudinal Change in Cortisol Levels Across the Adult Life Span. J. Gerontol. A Biol. Sci. Med. Sci. 2020, 75, 394–400. [Google Scholar] [CrossRef]
- Rincon-Cortes, M.; Herman, J.P.; Lupien, S.; Maguire, J.; Shansky, R.M. Stress: Influence of sex, reproductive status and gender. Neurobiol. Stress 2019, 10, 100155. [Google Scholar] [CrossRef]
- Carvalho, K.M.B.; Ronca, D.B.; Michels, N.; Huybrechts, I.; Cuenca-Garcia, M.; Marcos, A.; Molnar, D.; Dallongeville, J.; Manios, Y.; Schaan, B.D.; et al. Does the Mediterranean Diet Protect against Stress-Induced Inflammatory Activation in European Adolescents? The HELENA Study. Nutrients 2018, 10, 1770. [Google Scholar] [CrossRef]
- Hill, E.E.; Zack, E.; Battaglini, C.; Viru, M.; Viru, A.; Hackney, A.C. Exercise and circulating cortisol levels: The intensity threshold effect. J. Endocrinol. Invest. 2008, 31, 587–591. [Google Scholar] [CrossRef]
- Fekedulegn, D.B.; Andrew, M.E.; Burchfiel, C.M.; Violanti, J.M.; Hartley, T.A.; Charles, L.E.; Miller, D.B. Area under the curve and other summary indicators of repeated waking cortisol measurements. Psychosom. Med. 2007, 69, 651–659. [Google Scholar] [CrossRef]
- Mikolajczak, M.; Roy, E.; Luminet, O.; de Timary, P. Resilience and hypothalamic-pituitary-adrenal axis reactivity under acute stress in young men. Stress 2008, 11, 477–482. [Google Scholar] [CrossRef]
- Quaedflieg, C.; Meyer, T.; van Ruitenbeek, P.; Smeets, T. Examining habituation and sensitization across repetitive laboratory stress inductions using the MAST. Psychoneuroendocrinology 2017, 77, 175–181. [Google Scholar] [CrossRef]
- Liu, Y.; Yu, H.; Shi, Y.; Ma, C. The effect of perceived stress on depression in college students: The role of emotion regulation and positive psychological capital. Front. Psychol. 2023, 14, 1110798. [Google Scholar] [CrossRef]
- Boehme, M.; Guzzetta, K.E.; Wasén, C.; Cox, L.M. The gut microbiota is an emerging target for improving brain health during ageing. Gut Microbiome 2022, 4, e2. [Google Scholar] [CrossRef]
- Ma, T.; Jin, H.; Kwok, L.Y.; Sun, Z.; Liong, M.T.; Zhang, H. Probiotic consumption relieved human stress and anxiety symptoms possibly via modulating the neuroactive potential of the gut microbiota. Neurobiol. Stress 2021, 14, 100294. [Google Scholar] [CrossRef]
- Lach, G.; Schellekens, H.; Dinan, T.G.; Cryan, J.F. Anxiety, Depression, and the Microbiome: A Role for Gut Peptides. Neurotherapeutics 2018, 15, 36–59. [Google Scholar] [CrossRef]
- Breit, S.; Kupferberg, A.; Rogler, G.; Hasler, G. Vagus Nerve as Modulator of the Brain-Gut Axis in Psychiatric and Inflammatory Disorders. Front. Psychiatry 2018, 9, 44. [Google Scholar] [CrossRef]
- Dalile, B.; Van Oudenhove, L.; Vervliet, B.; Verbeke, K. The role of short-chain fatty acids in microbiota–gut–brain communication. Nat. Rev. Gastroenterol. Hepatol. 2019, 16, 461–478. [Google Scholar] [CrossRef]
- La Torre, D.; Verbeke, K.; Dalile, B. Dietary fibre and the gut–brain axis: Microbiota-dependent and independent mechanisms of action. Gut Microbiome 2021, 2, e3. [Google Scholar] [CrossRef]
- Miller, G.E.; Rohleder, N.; Cole, S.W. Chronic interpersonal stress predicts activation of pro- and anti-inflammatory signaling pathways 6 months later. Psychosom. Med. 2009, 71, 57–62. [Google Scholar] [CrossRef]
- Kelly, J.R.; Kennedy, P.J.; Cryan, J.F.; Dinan, T.G.; Clarke, G.; Hyland, N.P. Breaking down the barriers: The gut microbiome, intestinal permeability and stress-related psychiatric disorders. Front. Cell. Neurosci. 2015, 9, 392. [Google Scholar] [CrossRef]
- Doney, E.; Cadoret, A.; Dion-Albert, L.; Lebel, M.; Menard, C. Inflammation-driven brain and gut barrier dysfunction in stress and mood disorders. Eur. J. Neurosci. 2022, 55, 2851–2894. [Google Scholar] [CrossRef]
- Firth, J.; Solmi, M.; Wootton, R.E.; Vancampfort, D.; Schuch, F.B.; Hoare, E.; Gilbody, S.; Torous, J.; Teasdale, S.B.; Jackson, S.E.; et al. A meta-review of “lifestyle psychiatry”: The role of exercise, smoking, diet and sleep in the prevention and treatment of mental disorders. World Psychiatry 2020, 19, 360–380. [Google Scholar] [CrossRef]
- Wauters, L.; Van Oudenhove, L.; Accarie, A.; Geboers, K.; Geysen, H.; Toth, J.; Luypaerts, A.; Verbeke, K.; Smokvina, T.; Raes, J.; et al. Lactobacillus rhamnosus CNCM I-3690 decreases subjective academic stress in healthy adults: A randomized placebo-controlled trial. Gut Microbes 2022, 14, 2031695. [Google Scholar] [CrossRef]
- Lee, H.J.; Hong, J.K.; Kim, J.K.; Kim, D.H.; Jang, S.W.; Han, S.W.; Yoon, I.Y. Effects of Probiotic NVP-1704 on Mental Health and Sleep in Healthy Adults: An 8-Week Randomized, Double-Blind, Placebo-Controlled Trial. Nutrients 2021, 13, 2660. [Google Scholar] [CrossRef]




| Parameter | Probiotic | Placebo | Total |
|---|---|---|---|
| N Age (years) | 24 37.5 ± 10 | 21 40.7 ± 9.0 | 45 39.0 ± 9.6 |
| BMI (kg/m2) | 23.2 ± 2.5 | 22.8 ± 2.5 | 23.0 ± 2.5 |
| Gender | 13/11 (54/46%) | 13/8 (62/38%) | 26/19 (58/42%) |
| DASS-42 Score | 19.1 ± 2.7 | 18.8 ± 2.9 | 19.0 ± 2.8 |
| DASS-42 Stress Level (mild-to-moderate) | 44/56% | 46/54% | 45/55% |
| Baseline | Post-Intervention, 6 w | 6 w: Probiotic vs. Placebo Comparison | |||||
|---|---|---|---|---|---|---|---|
| Endpoint | Placebo | Probiotic | Placebo | Probiotic | Estimate | 95% CI | p Value |
| PSS-14 | 24.5 ± 6.7 | 26.4 ± 6.1 | 22.0 ± 7.9 | 20.2 ± 6.0 | −3.28 | (−5.93, −0.62) | 0.017 * |
| HADS-A | 7.6 ± 3.9 | 7.0 ± 3.2 | 6.3 ± 3.5 | 5.2 ± 2.6 | −0.65 | (−2.05, 0.76) | 0.358 |
| HADS-D | 4.6 ± 3.1 | 4.6 ± 3.2 | 4.2 ± 2.5 | 3.3 ± 2.7 | −0.83 | (−2.03, 0.38) | 0.172 |
| GSRS Reflux | 1.3 ± 0.6 | 1.5 ± 0.9 | 1.3 ± 0.5 | 1.5 ± 0.9 | 0 | (0, 0) | 0.853 1 |
| GSRS Abdominal Pain | 2.1 ± 1.1 | 1.9 ± 0.9 | 1.7 ± 0.7 | 1.6 ± 0.6 | −0.04 | (−0.24, 0.16) | 0.676 2 |
| GSRS Indigestion | 2.6 ± 1.2 | 2.4 ± 1.0 | 2.2 ± 0.9 | 2.0 ± 0.8 | −0.04 | (−0.20, 0.12) | 0.577 2 |
| GSRS Diarrhea | 1.6 ± 1.0 | 1.6 ± 0.9 | 1.6 ± 0.6 | 1.4 ± 0.4 | −0.06 | (−0.26, 0.13) | 0.504 2 |
| GSRS Constipation | 2.0 ± 1.2 | 1.8 ± 0.9 | 1.7 ± 0.9 | 1.4 ± 0.5 | −0.12 | (−0.32, 0.08) | 0.238 2 |
| PSQI Subjective Sleep Quality | 1.2 ± 0.4 | 1.1 ± 0.6 | 1.1 ± 0.3 | 0.8 ± 0.5 | 0 | 0 | 0.037 1* |
| PSQI Sleep Latency | 1.0 ± 0.7 | 1.0 ± 0.6 | 0.8 ± 0.6 | 0.8 ± 0.6 | 0 | 0 | 0.923 1 |
| PSQI Sleep Duration | 0.2 ± 0.4 | 0.2 ± 0.5 | 0.3 ± 0.5 | 0.2 ± 0.4 | 0 | 0 | 0.391 1 |
| PSQI Habitual Sleep Efficiency | 0.4 ± 0.7 | 0.3 ± 0.6 | 0.3 ± 0.6 | 0.2 ± 0.4 | 0 | 0 | 0.358 1 |
| PSQI Sleep Disturbances | 1.2 ± 0.4 | 1.0 ± 0.4 | 1.0 ± 0.4 | 1.0 ± 0.0 | 0 | 0 | 0.554 1 |
| PSQI Use of Sleeping Medication | 0.0 ± 0.2 | 0.1 ± 0.3 | 0.0 ± 0.0 | 0.1 ± 0.3 | 0 | 0 | 0.182 1 |
| PSQI Daytime Dysfunction | 1.1 ± 0.6 | 1.1 ± 0.5 | 0.7 ± 0.6 | 0.7 ± 0.6 | 0 | 0 | 0.862 1 |
| PSQI Total | 5.2 ± 1.8 | 4.8 ± 2.2 | 4.3 ± 1.7 | 3.8 ± 1.4 | −0.38 | (−1.13, 0.37) | 0.308 1 |
| Baseline | Post-Intervention, 6 w | 6 w: Probiotic vs. Placebo Comparison | |||||
|---|---|---|---|---|---|---|---|
| Endpoint | Placebo | Probiotic | Placebo | Probiotic | Estimate | 95% CI | p Value |
| Acute Stress AUCg | 326.4 ± 217.6 | 296.3 ± 211.2 | 219.7 ± 151.1 | 276.7 ± 185.0 | 0.39 | (0.09, 0.69) | 0.013 1* |
| Acute Stress AUCi | 251.2 ± 199.7 | 201.9 ± 189.6 | 139.5 ± 125.5 | 165.1 ± 174.7 | 1.58 | (−1.29, 4.45) | 0.271 1 |
| Acute Stress Cmax | 10.6 ± 7.6 | 9.4 ± 7.2 | 6.9 ± 5.3 | 8.2 ± 6.3 | 0.35 | (−0.02, 0.71) | 0.06 1 |
| Acute Stress ICmax | 9.3 ± 7.3 | 7.8 ± 6.9 | 5.5 ± 4.9 | 6.1 ± 6.3 | 0.21 | (−0.32, 0.75) | 0.422 1 |
| CAR AUCg | 315.9 ± 117.1 | 294.6 ± 126.3 | 337.7 ± 156.1 | 337.8 ± 166.7 | 0.31 | (−2.34, 2.97) | 0.812 2 |
| CAR AUCi | 130.9 ± 100.1 | 111.4 ± 110.1 | 154.1 ± 126.2 | 130.4 ± 121.4 | −0.84 | (−4.18, 2.49) | 0.611 2 |
| CAR Cmax | 7.4 ± 2.7 | 7.1 ± 3.1 | 7.9 ± 4.1 | 7.8 ± 3.9 | −0.11 | (−2.60, 2.37) | 0.927 |
| CAR ICmax | 4.1 ± 2.6 | 3.5 ± 3.1 | 4.8 ± 3.9 | 4.0 ± 3.3 | 0.07 | (−0.48, 0.62) | 0.795 1 |
| Baseline | Post-Intervention, 6 w | 6 w: Probiotic vs. Placebo Comparison | ||||||
|---|---|---|---|---|---|---|---|---|
| Endpoint | Period | Placebo | Probiotic | Placebo | Probiotic | Estimate | 95% CI | p Value |
| Mean HR (1/min) | Before | 73.9 ± 9.5 | 73.9 ± 8.6 | 73.6 ± 11.6 | 74.7 ± 8.2 | 0.94 | (−3.20, 5.08) | 0.649 |
| During 1 | 77.5 ± 10.1 | 76.6 ± 9.5 | 78.1 ± 11.7 | 77.2 ± 8.0 | −1.85 | (−5.25, 1.55) | 0.284 1 | |
| After 8 | 69.8 ± 7.5 | 66.4 ± 8.4 | 67.6 ± 9.2 | 68.8 ± 6.6 | 0.53 | (−2.89, 3.95) | 0.760 1 | |
| RMSSD (ms) | Before | 42.6 ± 32.6 | 46.3 ± 33.8 | 33.2 ± 11.6 | 64.0 ± 101.3 | 0.17 | (−0.22, 0.56) | 0.382 2 |
| During 1 | 59.2 ± 50.5 | 77.4 ± 82.0 | 37.7 ± 22.6 | 52.9 ± 39.2 | 2.63 | (−2.31, 7.72) | 0.225 1,3 | |
| After 8 | 48.1 ± 18.3 | 89.6 ± 93.7 | 63.9 ± 70.3 | 60.4 ± 35.1 | 3.38 | (−13.96, 14.32) | 0.610 1,3 | |
| Sympathetic-Vagal Balance = LF/HF | Before | 2.9 ± 2.0 | 3.3 ± 3.4 | 3.6 ± 2.4 | 2.8 ± 2.4 | −0.26 | (−0.74, 0.23) | 0.29 2 |
| During 1 | 3.2 ± 1.9 | 3.4 ± 1.99 | 3.2 ± 2.0 | 3.8 ± 2.9 | 0.22 | (−0.03, 0.47) | 0.08 1,4 | |
| After 8 | 4.6 ± 4.3 | 3.5 ± 3.96 | 3.2 ± 2.3 | 2.9 ± 2.7 | 0.07 | (−0.18, 0.32) | 0.592 1,4 | |
| LF (Hz) | Before | 0.1 ± 0.03 | 0.1 ± 0.02 | 0.1 ± 0.02 | 0.1 ± 0.02 | −0.003 | (−0.02, 0.01) | 0.644 |
| During 1 | 0.1 ± 0.03 | 0.1 ± 0.02 | 0.1 ± 0.03 | 0.1 ± 0.02 | −0.005 | (−0.02, 0.01) | 0.603 1 | |
| After 8 | 0.1 ± 0.02 | 0.1 ± 0.02 | 0.1 ± 0.03 | 0.1 ± 0.02 | −0.007 | (−0.03, 0.01) | 0.471 1 | |
| HF (Hz) | Before | 0.2 ± 0.07 | 0.3 ± 0.06 | 0.2 ± 0.06 | 0.2 ± 0.07 | 0.02 | (−0.03, 0.05) | 0.453 |
| During 1 | 0.2 ± 0.07 | 0.2 ± 0.08 | 0.2 ± 0.06 | 0.2 ± 0.08 | 0.01 | (−0.03, 0.06) | 0.529 1 | |
| After 8 | 0.2 ± 0.05 | 0.2 ± 0.05 | 0.2 ± 0.06 | 0.2 ± 0.06 | 0.01 | (−0.03, 0.05) | 0.661 1 | |
| Skin Conductance (microS) | Before | 1.5 ± 2.1 | 1.5 ± 1.8 | 1.7 ± 2.3 | 1.9 ± 3.2 | −0.11 | (−0.90, 0.67) | 0.77 2 |
| During 1 | 3.4 ± 3.5 | 3.5 ± 2.6 | 3.5 ± 3.9 | 3.8 ± 4.9 | −0.03 | (−0.21, 0.16) | 0.786 1,5 | |
| After 8 | 3.5 ± 4.5 | 2.1 ± 2.4 | 2.6 ± 3.0 | 3.0 ± 4.2 | −0.01 | (−0.2, 0.18) | 0.911 1,5 | |
| Baseline | Post-Intervention, 6 w | 6 w: Probiotic vs. Placebo Comparison | |||||
|---|---|---|---|---|---|---|---|
| Endpoint | Placebo | Probiotic | Placebo | Probiotic | Estimate | 95% CI | p Value |
| Positive PANAS | −1.6 ± 7.1 | −2 ± 5.2 | 3.7 ± 3.7 | 0.8 ± 3.1 | −2.87 | (−5.01, −0.73) | 0.009 ** |
| Negative PANAS | 3.1 ± 6.4 | 0.4 ± 4.2 | 0.6 ± 4.5 | 1.1 ± 4.3 | 1.18 | (−1.51, 3.86) | 0.38 |
| STAI-6 State | −0.6 ± 1.3 | −0.9 ± 1.5 | 0 ± 1.4 | −0.5 ± 1.4 | −0.43 | (−1.27, 0.40) | 0.301 |
| STAI-6 Trait | −0.2 ± 1.2 | −0.4 ± 0.8 | −0.6 ± 1.3 | −1 ± 1.4 | −0.43 | (−1.23, 0.37) | 0.286 |
| VAS Stressful | 26.1 ± 26.1 | 16.4 ± 21 | 22.7 ± 26.5 | 14.6 ± 24.6 | −3.42 | (−17.98, 11.15) | 0.638 |
| VAS Painful | 63 ± 29.9 | 58.1 ± 21.8 | 69.1 ± 21.8 | 50.2 ± 26.8 | −16.5 | (−30.26, −2.74) | 0.02 * |
| VAS Uncomfortable | 52.4 ± 26.2 | 43.4 ± 23.5 | 50.9 ± 29.1 | 39.9 ± 28.0 | −10.04 | (−28.69, 8.62) | 0.282 |
| VAS Total | 47.1 ± 22.4 | 39.3 ± 17.5 | 47.6 ± 20.7 | 34.9 ± 21.1 | −8.83 | (−19.21, 1.56) | 0.093 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Boehme, M.; Rémond-Derbez, N.; Lerond, C.; Lavalle, L.; Keddani, S.; Steinmann, M.; Rytz, A.; Dalile, B.; Verbeke, K.; Van Oudenhove, L.; et al. Bifidobacterium longum subsp. longum Reduces Perceived Psychological Stress in Healthy Adults: An Exploratory Clinical Trial. Nutrients 2023, 15, 3122. https://doi.org/10.3390/nu15143122
Boehme M, Rémond-Derbez N, Lerond C, Lavalle L, Keddani S, Steinmann M, Rytz A, Dalile B, Verbeke K, Van Oudenhove L, et al. Bifidobacterium longum subsp. longum Reduces Perceived Psychological Stress in Healthy Adults: An Exploratory Clinical Trial. Nutrients. 2023; 15(14):3122. https://doi.org/10.3390/nu15143122
Chicago/Turabian StyleBoehme, Marcus, Noëla Rémond-Derbez, Clara Lerond, Luca Lavalle, Sonia Keddani, Myriam Steinmann, Andreas Rytz, Boushra Dalile, Kristin Verbeke, Lukas Van Oudenhove, and et al. 2023. "Bifidobacterium longum subsp. longum Reduces Perceived Psychological Stress in Healthy Adults: An Exploratory Clinical Trial" Nutrients 15, no. 14: 3122. https://doi.org/10.3390/nu15143122
APA StyleBoehme, M., Rémond-Derbez, N., Lerond, C., Lavalle, L., Keddani, S., Steinmann, M., Rytz, A., Dalile, B., Verbeke, K., Van Oudenhove, L., Steiner, P., Berger, B., Vicario, M., Bergonzelli, G., Colombo Mottaz, S., & Hudry, J. (2023). Bifidobacterium longum subsp. longum Reduces Perceived Psychological Stress in Healthy Adults: An Exploratory Clinical Trial. Nutrients, 15(14), 3122. https://doi.org/10.3390/nu15143122