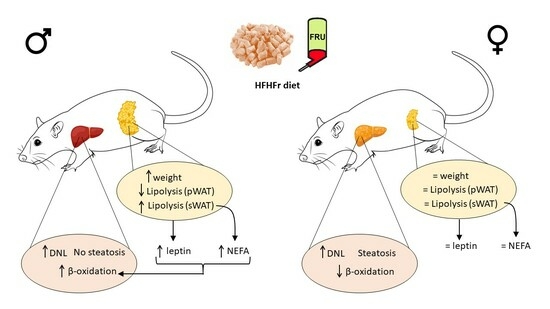
Adipose Tissue Protects against Hepatic Steatosis in Male Rats Fed a High-Fat Diet plus Liquid Fructose: Sex-Related Differences
Abstract
Share and Cite
Bentanachs, R.; Blanco, L.; Montesinos, M.; Sala-Vila, A.; Lázaro, I.; Rodríguez-Morató, J.; Sánchez, R.M.; Laguna, J.C.; Roglans, N.; Alegret, M. Adipose Tissue Protects against Hepatic Steatosis in Male Rats Fed a High-Fat Diet plus Liquid Fructose: Sex-Related Differences. Nutrients 2023, 15, 3909. https://doi.org/10.3390/nu15183909
Bentanachs R, Blanco L, Montesinos M, Sala-Vila A, Lázaro I, Rodríguez-Morató J, Sánchez RM, Laguna JC, Roglans N, Alegret M. Adipose Tissue Protects against Hepatic Steatosis in Male Rats Fed a High-Fat Diet plus Liquid Fructose: Sex-Related Differences. Nutrients. 2023; 15(18):3909. https://doi.org/10.3390/nu15183909
Chicago/Turabian StyleBentanachs, Roger, Laia Blanco, Maria Montesinos, Aleix Sala-Vila, Iolanda Lázaro, Jose Rodríguez-Morató, Rosa María Sánchez, Juan Carlos Laguna, Núria Roglans, and Marta Alegret. 2023. "Adipose Tissue Protects against Hepatic Steatosis in Male Rats Fed a High-Fat Diet plus Liquid Fructose: Sex-Related Differences" Nutrients 15, no. 18: 3909. https://doi.org/10.3390/nu15183909
APA StyleBentanachs, R., Blanco, L., Montesinos, M., Sala-Vila, A., Lázaro, I., Rodríguez-Morató, J., Sánchez, R. M., Laguna, J. C., Roglans, N., & Alegret, M. (2023). Adipose Tissue Protects against Hepatic Steatosis in Male Rats Fed a High-Fat Diet plus Liquid Fructose: Sex-Related Differences. Nutrients, 15(18), 3909. https://doi.org/10.3390/nu15183909