Abstract
Diabetes mellitus is a worldwide public health issue with numerous complications. Several risk factors are associated with diabetes, mainly due to patients following an unhealthy lifestyle. Copper is a crucial trace element, with various physiological actions. Different intake levels of copper might contribute to diabetes development due to its dual action as both an anti- and pro-oxidant. Aim: Due to the inconclusive findings regarding the relationship between copper consumption and the management of diabetes, we decided to conduct this extensive systematic review. Up to this date, no similar study has been available in the literature. In this review, we used the preferred reporting items for systematic reviews and meta-analysis (PRISMA) guidelines. Relevant articles were identified by searching the electronic databases CINAHL, EMBASE and Medline from their respective index dates to September 2022 using keywords such as “Copper Intake” and “Type 2 Diabetes”. Any paper that has investigated copper exposure through supplementation or any other method that indicates copper intake in human subjects with type 2 diabetes and measures at least one of the outcomes of interest related to diabetes was included in this review. This review is comprised of 4 cross-sectional studies, 3 cohort studies, 2 RCTs, and 2 interventional studies. Two cohort studies found positive associations between copper intake and the risk of developing T2DM, while no significant association was found in the third study. Regarding diabetes outcomes in the four cross-sectional studies, two found inverse associations, one found a positive relationship, and one found no significant association. In interventional studies, all studies found a protective effect of copper, including the RCT, while one found no significant association. The results are inconsistent concerning the association between copper consumption and the likelihood of developing diabetes are inconsistent. Individuals should receive an adequate dietary amount of copper that is within the RDA levels (900 µg/day) to avoid copper deficiency or toxicity. Further studies, especially RCTs, are strongly needed to enable researchers to elucidate more robust conclusions regarding this association.
1. Introduction
Diabetes mellitus (DM) is a chronic disease that arises from either partial or complete insulin deficiency [1]. Diabetes is becoming a worldwide public health issue due to its high prevalence [2]. In 2014, the World Health Organization (WHO) reported that diabetes affected 8.5% of all adults aged 18 and above, as per their own statistics [3]. Moreover, in 2019, approximately 1.5 million of total deaths were directly related to diabetes. Prevalence studies revealed that, in contrast to high-income countries, low- and middle-income countries exhibited a higher percentage [4].
Diabetes is a global health burden, with many health impacts. There are several complications that diabetic patients suffer from which adversely affect their quality of life, such as cardiovascular complications, peripheral neuropathy, nephropathy, retinopathy, mental diseases, hearing difficulties and oral conditions [5]. As per the Centers for Diseases Control and Prevention (CDC), diabetes symptoms are characterized by increased feelings of thirst (polydipsia), increased urination (nocturia), fatigue, blurry vision, skin dryness, delay in wound healing, concurrent infections and tingling limbs [6].
Diabetes can occur due to several reasons. The primary etiology for type 1 diabetes is auto-immune conditions, which leads to the destruction of the beta cells responsible for producing insulin in the pancreas [7]. However, for type 2 diabetes, the factors are mainly related to lifestyle behaviors and genetics [8]. Crucial modifiable factors that can lead to the onset of diabetes include excess body fat, physically inactivity and following an unhealthy eating pattern. Genetic mutations, hormonal diseases, pancreatic conditions, like pancreatitis, pancreatic cancer or trauma, as well as some medications, might also contribute to the development of diabetes development [9]. According to the CDC, choosing healthy food choices, being physically active and managing stress are scientifically proven guidelines for diabetes prevention and these are recommended through their national diabetes prevention program [10].
Over the past few decades, several studies were carried out to study the association between copper and diabetes. Copper is a crucial trace mineral needed for the efficient metabolism of many physiological reactions [11]. Dietary sources high in copper include organ meats and nuts [12]. Fruits and cereals may also be a good source of copper but will vary depending on the soil concentration of copper, which varies according to regions, pesticide use, and industrial copper emissions [12]. Zinc shares many of the dietary sources of copper, but it is also present at high concentrations in oysters, whole-grain cereals, and red meats [12]. Copper is an important cofactor needed for redox reactions [11]. The condition of copper overload and deficiency can both lead to adverse health outcomes. The effect of copper on oxidation can vary depending on the level of intake, as it can function as both an anti-oxidant and a pro-oxidant [13]. The anti-oxidant properties of copper are demonstrated through its being an essential component of copper/zinc superoxide dismutase (SOD) [14]. SOD enables free radical clearance in all body cells, hence defending against oxidative stress [15]. However, copper toxicity might induce insulin resistance by acting as a pro-oxidant [16]. Excessive copper promotes the generation of reactive oxygen species (ROS), which increases the oxidative stress and eventually leads to diabetes [17].
Findings concerning the relationship between copper intake and diabetes are inconclusive and scarce, as the available studies show inconsistent results. Thus, we aimed to conduct this comprehensive systematic review to elucidate the studies which examines the association between the copper intake and diabetes as up to this date, no similar study has been made available in the literature. Therefore, this systematic review will be the first to compile and review the available evidence regarding the association between dietary copper intake and diabetes so that clinicians can have access to guidance about the importance of copper supplementation in the amelioration and/or prevention of diabetes. This systematic review will support researchers who are aiming to find evidence regarding this association and will facilitate conducting research in this field. In addition, it will identify gaps in the literature that need to be tackled to establish a better understanding of the association between dietary copper intake and diabetes.
2. Methods
This systematic review adheres to the guidelines of preferred reporting items for systematic reviews and meta-analysis (PRISMA) (Table S1).
2.1. Literature Search
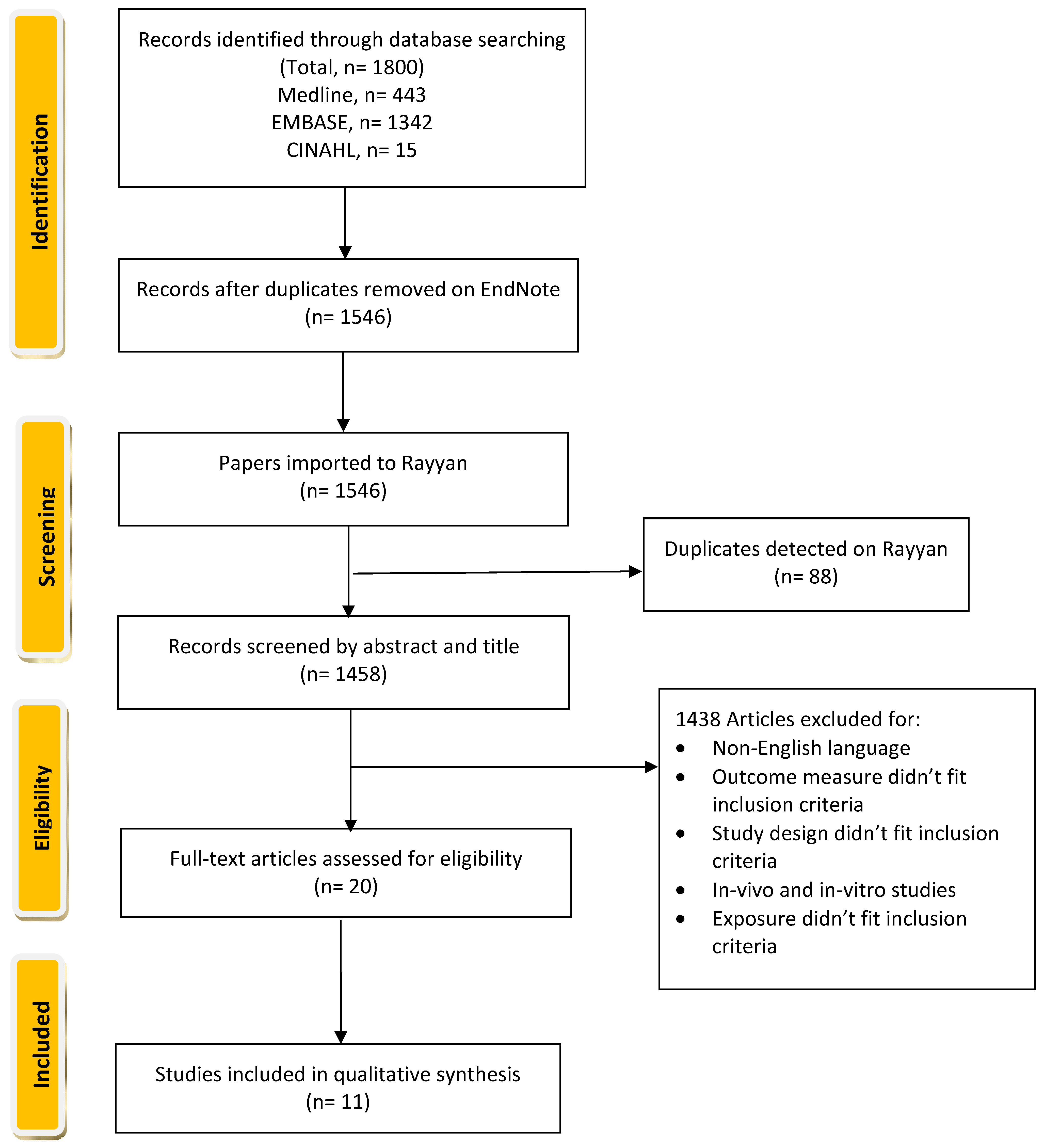
Articles of relevance were obtained by searching electronic databases Medline, EMBASE, and CINAHL from their respective index dates to September 2022. An additional free hand-search was conducted to ensure sure that this review includes almost all relevant papers and to reduce the possibility of missing any paper that could be included. The search strategy incorporated Medical Subject Headings, Emtree, and CINAHL subject heading (Table 1) for all three databases with search/key terms related to “Copper” and “Diabetes”. Articles from the three databases were imported into Endnote, where duplicates and articles composed in non-English languages were identified and removed. Articles were then imported into Rayyan Web for screening and assessment (Figure 1).

Table 1.
Search Terms used in Medline, Embase, and CINAHL.
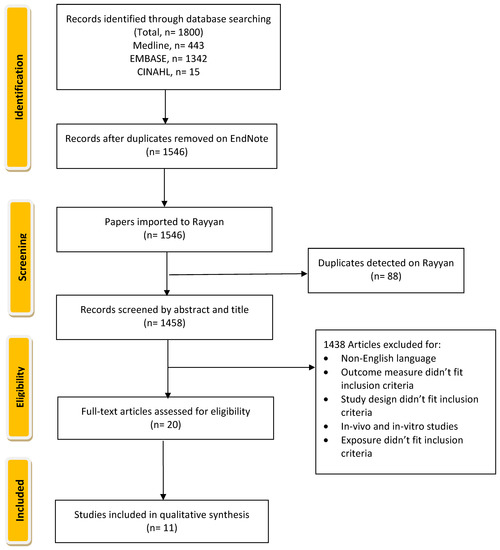
Figure 1.
Flow chart of The Systematic Review of Copper Intake and Type 2 diabetes.
2.2. Study Selection
In our systematic review, studies were assessed for potential inclusion based on specific criteria which include any paper that has investigated copper exposure through supplementation or any other method that indicates copper intake in human subjects with type 2 diabetes and measures at least one of the outcomes of interest (glycated hemoglobin A1c (HbA1c), fasting plasma glucose (FPG), 2 h plasma glucose (2-H PG), oral glucose tolerance test (OGTT), blood lipids (high-density lipoprotein (HDL), low-density lipoprotein (LDL), triglycerides (TG), any diabetes complications and total cholesterol levels (total Chol)), as well as any diabetes-related complication. Articles that were not based on original research (commentaries, conference proceedings, letters, and reviews) were excluded, as were in vitro and in vivo studies. Five reviewers were involved to independently screen all the articles by publication titles and abstracts and then evaluate the full-text publications for inclusion in this review. Disagreements were resolved by the authors performing a second review through a consensus-based discussion.
2.3. Data Extraction
The authors extracted data using a standardized approach. For each article identified, we extracted information on the study characteristics including the first author’s family name, publication year, country, study design, settings, sample size, population, and sample characteristics (sex, age, baseline weight/health status); we additionally noted the copper administration, outcome assessment, and main findings of each study. Table 2 provides a summary of all articles included in this review.

Table 2.
Characteristics of included studies.
2.4. Risk-of-Bias Assessment
We used the Newcastle–Ottawa scale (NOS) to conduct a quality assessment of the included studies [29]. This tool has three domains which assess selection, comparability, and outcome. For each study, a grade of a maximum of one point can be given for each item except comparability, which can be given a grade of up to 2 points. The evaluation of the studies’ quality is determined by their overall score on the Newcastle–Ottawa scale, which has a maximum score of 9. A rating of 0–2 suggests inadequate quality, 3–5 indicates moderate quality, while 6–9 implies good or high quality. To determine the quality of randomized controlled trials and other interventional studies, we employed the Cochrane risk-of-bias tool [30]. This tool evaluates the study across six categories: sequence generation, allocation concealment, blinding, incomplete outcome data, selective outcome reporting, and other potential threats to validity. Each domain is assessed with a ‘yes’, ‘no’, or ‘unclear’ response, indicating a low, high, or unclear risk of bias, respectively. If the first three questions receive a ‘yes’ response and no significant concerns are found in the last three domains, the study is classified as having a low risk of bias. If two domains receive an ‘unclear’ or ‘no’ response, the study is classified as having a moderate risk of bias. Finally, if three or more domains receive an ‘unclear’ or ‘no’ response, the study is classified as having a high risk of bias.
3. Results
3.1. Overall Quality Assessment
Descriptions of the main characteristics of the studies are presented in Table 2. A quality assessment was performed for all studies included in this systematic review. Based on the quality assessment of cross-sectional studies using the Newcastle–Ottawa scale [29], and as presented in Table 3, out of the four studies, only one study received a total quality score of 6, which indicates good quality. The other three studies received a total quality score of 5, which indicates studies to be of fair quality. This was mainly due to either issues with comparability or outcome assessments. Three cohort studies were included as well, and all were considered to be good or high-quality studies. To assess the RCT and other interventional trials involved in this review, we used the Cochrane risk-of-bias tool [30]. As presented in Table 4, out of the four trials included, only one had a low risk of bias. Two had an overall high risk of bias, and one had a serious overall risk of bias. The latter scenario was mostly due to being an old trial with a great deal of missing information.

Table 3.
Summary of the quality assessment of studies using Newcastle–Ottawa Scale.

Table 4.
Summary of the quality assessment of studies using Cochrane risk-of-bias tool for RCT’s.
3.2. Cross-Sectional Studies
Four cross-sectional studies were included in this systematic review. Zhang et al. used the NHANES database to conduct a cross-sectional study about the association between copper intake and the risk of diabetes nephropathy [20]. There were no significant differences between either group in term of hypertension or lipid profile. The study showed that as the intake of copper increases, the risk for diabetes nephropathy decreases, with a pooled odds ratio of 0.67 (95% CI: 0.54–0.84) [20]. A second cross-sectional study by Tan et al. into 128 overweight and obese Malaysian adults tested the association between dietary copper intake and insulin resistance [21]. All subjects had no diabetes, cardiovascular diseases, stroke, renal or endocrinal diseases. The study found that there is a noticeable and beneficial relationship between dietary copper and HOMA-IR, but only if the intake of copper is equal to or greater than 13.4 μg/kg/day, with an odds ratio of 0.276 (95%CI = 0.025–0.527, p value for trend = 0.033). Interestingly, the study found that the insulin-resistant group had significantly higher total cholesterol/HDL cholesterol and triglyceride and lower HDL cholesterol compared to the non-insulin-resistant group [21]. Another study by Norbitt et al. used the data from the ANDROMEDA project to assess dietary intake [22]. Subjects included in the study were diagnosed with AP between 2015–2019 with the most updated international diagnosis guideline and either did or did not use anti-diabetic medication or insulin use. The study excluded individuals with diabetes type 1 or gestational diabetes, chronic pancreatitis, intraoperative diagnosis of pancreatitis or post-endoscopic retrograde cholangiopancreatography pancreatitis. The results revealed a significant difference in the mean intake of copper between the 3 groups, as well as a significant negative association between the fasting plasma glucose and copper intake in the NAP group [22].
Sobhani et al. studied the association between the intake of fast food and multiple outcomes including anthropometric measurement, serum lipid profile and glucose metabolism in patients with diabetes nephropathy (DN) [23]. Participants were at stage 1 or 2 of diabetes, with a fasting blood glucose level of 126 mg/dL, the consumption hypoglycemic medication or insulin, and a proteinuria level of 300 mg/dL [23]. The results showed that a higher intake of fast food was associated with a higher intake of copper, among other nutrients [23]. In addition, it was found that copper intake was higher in those with higher intakes of fast food. This was related to higher levels of blood pressure and total cholesterol, rather than to any significant diabetic outcomes [23].
3.3. Cohort Studies
Three cohort studies were found to assess the impact of copper intake on diabetes parameters. The first one was performed by Eshak et al. aimed to assess the diet of the Japanese population [24]. After distributing food frequency questionnaires to their 16,160 subjects who have no risk of diabetes, cardiovascular diseases or cancer and following up with them for 5 years, 396 subjects developed T2DM (2.5%) [24]. This study showed that those who had higher dietary intakes of copper and iron had a higher risk of T2DM, with the risk being more pronounced among those who were older, overweight, smokers, and who had a family history of diabetes [24]. In the second cohort study, Cui et al. assessed the diet of 14,711 adults free of diabetes, with or without hypertension, and without any cardiovascular diseases in the China Health and Nutrition Survey and assessed the risk of those newly diagnosed with T2DM between 2000 and 2015 [25]. They found that the association between dietary copper and T2DM risk in healthy Chinese adults was not independent, but rather that copper was positively associated with a higher risk of developing T2DM, especially when selenium intake was lower than the medium (p interaction = 0.0292) [25]. The final cohort study by Laouali et al. monitored 70,991 women with or without hypertension or hypercholesteremia for 20 years, examining the association between T2DM incidence and dietary copper-to-zinc ratio [26]. After 20 years, they found that a copper-to-zinc ratio of less than 0.55 was associated with a lower risk of T2DM [26]. They also found that women with zinc intakes of less than 8 mg/day, a higher copper-to-zinc ration was associated with a higher T2DM risk, especially among obese females [26]. Higher copper-to-zinc ratios were associated with more frequent hypercholesterolemia and higher age and BMI at baseline [26].
3.4. Interventional Studies
Two RCTs and two interventional studies were performed to assess the impact of copper on diabetes outcomes. The randomized control trial by Gunasekara et al. aimed to evaluate the effect of a multimineral vitamin including copper, administered with or without zinc, on blood glucose levels in adult diabetics with or without essential hypertension and dyslipidemia. They excluded subjects with renal and liver disorders, those on hormonal therapy, those with a history of recent surgery, pregnant and lactating women and those who are undergoing insulin preparation therapy [18]. They that found that a combination of zinc and a multivitamin supplement (which had 2 mg of copper as part of its composition) reduced FBG by a mean of 0.33 mmol/L and reduced postprandial glucose levels by 1.55 mmol/L [18]. Therefore, they concluded that a multivitamin in addition to zinc could have beneficial effects in the management of diabetes in adults [18]. Alfawaz et al. also conducted a randomized control trial to assess dietary pattern changes in adults with prediabetes with no other chronic disease [19]. They observed copper deficiency at baseline for both groups but observed improved intakes of copper in the guidance group compared to the GA group (37.3% vs. 13.3%) as well as improvements in the fasting glucose levels and insulin resistance, which were clinically significant [19]. Therefore, they concluded that the fortification of micronutrients, especially copper, in the Saudi diet is recommended for diabetes management [19].
A non-randomized controlled trial was performed by Armstrong et al. to assess the effect of dietary treatment for newly diagnosed adults on the complications of diabetes [28]. The twenty participants that were in the intervention group had no cardiovascular diseases and only two had a background of diabetic retinopathy compared to the twenty healthy individuals [28]. No difference was difference in copper intakes [28]. They also found that, in the cases group, fasting blood glucose levels dropped from 13.6 at the beginning of the study to 9.7 mmol/L after adhering to the diet (p < 0.01). HbA1c also reduced from 7.44 ± 0.67% to 5.91 ± 0.57% (p < 0.01) [28]. Another clinical trial performed by Rostami et al. assessed the impact of spirulina supplementation on diabetes outcomes. Each 100 g of spirulina powder contains 0.7 mg of copper. Spirulina supplementation was able to significantly decrease the total cholesterol, LDL cholesterol, triglycerides and malondialdehyde serum level significantly [27]. This indicates that spirulina may help people with diabetes to control their lipid profile [27]. However, baseline triglyceride levels were higher in the spirulina-treated diabetic patients compared to the diabetic control group [27].
4. Discussion
This systematic review was conducted to elucidate the studies examining the association between copper intake and the development and management of diabetes. The final review included 3 cohort studies, 4 cross-sectional studies, 2 RCT, and 2 interventional studies. The bioavailability of copper is estimated to be between 30–40% of the ingested copper amount [30]. To begin the absorption process for copper, it first needs to be reduced by several reductase enzymes from dietary cupric (Cu2+) to cuprous copper (Cu1+) [31]. Then, it is taken up across the apical membrane by copper transporter 1 (CTR1) [31]. Once copper is inside the epithelial intestinal cell, it is transferred to the protein ATOX1, and then, in order to be exported to the portal vein, it is delivered to the protein ATP7A. Any excess copper is bound to metallothionein, which is a homeostatic strategy to the existence of avoid copper toxicity inside the cytosol of the epithelial intestinal cells [31]. After copper is absorbed, 95% of it binds to ceruloplasmin in the blood [32].
Several studies have found that serum copper concentrations are higher in diabetic patients compared to healthy subjects [33,34]. In gestational diabetes, studies have found relatively high levels in pregnant women, which may be explained by the elevated copper demand as it has a vital role in embryonic and fetal growth and development [35]. Copper is one of the most important trace minerals amongst those that work as an anti-oxidant involved in redox reactions [36]. Copper is an essential element of the enzyme copper/zinc superoxide dismutase (Cu/Zn SOD) which aids in the clearance of free radicals that accumulate in cells as a result of metabolic stress [36]. Metabolic stress in an important factor that can lead to the development of comorbid conditions such as diabetes. Moreover, one mechanism explaining the association between copper and diabetes is its involvement in the reactions of glutamic acid decarboxylase (GAD), a major beta cell anti-oxidant that is altered by ROS [36]. This explanation was provided through a study investigating the association between copper and diabetes which found that the presence of anti-GAD antibodies contribute to the pathogenesis of diabetes [36]. Copper also plays an essential role in regulating ROS production through its role in the electron transfer chain, which is highly reactive in redox reactions [35]. The negative impacts of metals are frequently attributed to the generation of free radicals, including copper. Studies have suggested that copper, especially at toxic levels, may have pro-oxidant activity. Therefore, a high intake of copper elevates oxidative stress, which may contribute to an increased body inflammation [36]. Copper can cause the production of reactive oxygen species (ROS) through the Fenton reaction, which can hinder various physiological processes, including those associated with insulin resistance development and abnormal glucose metabolism [35,37]. A study conducted by Galhardi et al. showed that high copper intake promoted oxidative stress development, as well as kidney dysfunction, among rats with diabetes [16]. Another study conducted on rats found that copper deprivation, achieved through feeding them copper-deficient diet containing copper chelating agent, contributed to alpha and beta pancreatic cell neogenesis [38]. In addition, a meta-analysis conducted in 2017 reported that the risk of diabetes was positively associated with serum copper intake [39]. Moreover, a review by Kaur et al. reported that copper level abnormalities, which include both toxic and deficient levels, may induce the pathogenesis of diabetes through causing oxidative damage [40]. Previous research showed that ROS may impair insulin-dependent glucose transport by regulating the activity of several signaling molecules such as MP-activated protein kinase (AMPK), p38 mitogen-activated protein kinases (p38MAPK), and Jun N-terminal kinase/stress-activated protein kinase (JNK/SAPK) [36]. In addition, the activation of these signaling molecules can hinder insulin-dependent glucose transportation, resulting in insulin resistance [36]. Another mechanism involved is the potential adverse effect of copper copper on pancreatic islets [35]. It has been suggested that copper ions trigger amylin peptide aggregation to amyloid fibril, which may be related to the decreased β-cell mass along with the progressive failure of islet β-cells in human body [35].
5. Strengths and Limitations of the Review
This systematic review has many strengths. Up to our knowledge, this is the first systematic review to evaluate the association between copper intake and type 2 diabetes and summarize almost all the available evidence regarding this association. In addition, this review was developed based on PRISMA, and the Rayyan application was used to perform initial screening, which enabled the research team to conduct blind review that would enhance the credibility of the findings. On the other hand, there are some limitations to this systematic review. Firstly, the majority of the studies included in this review are observational, with only two randomized controlled trials, making it difficult to draw a robust conclusion from the available papers presented in this review. Furthermore, we only included papers written in English, while all other foreign languages were excluded, limiting evidence provided by this review to English literature only. Additionally, some of the studies had high risk of bias and poor overall quality, making it difficult to draw firm conclusions from them. Finally, the dietary assessment methods used in the included studies are heterogenous and have variable validity and reliability, affecting their risk of bias and the strength of the evidence they provide. It is also important to note that dietary assessment methods have limited strength compared to biochemical indicators of copper intake/status.
6. Conclusions
To conclude, the physiological role of copper is dependent on its level. It has a dual action, as it can work as an anti-oxidant at adequate levels and as a pro-oxidant in excessive level. Based on this review, the findings regarding the association between copper intake and the risk of diabetes are inconsistent. However, when it comes to dietary recommendations regarding copper, it is important to consider individual differences in the intake. Individuals should receive an adequate dietary amount of copper that is within the RDA levels (900 µg/day) in order to avoid copper deficiency or toxicity. It is crucial to highlight the possible toxicity of copper, which may mainly be from supplements. Supplements can be recommended among severely deficient individuals who are unable to achieve adequate copper intake from diet. Further studies, especially RCTs, are strongly needed enable researchers to elucidate the association and present a more robust conclusion.
Supplementary Materials
The following supporting information can be downloaded at: https://www.mdpi.com/article/10.3390/nu15071655/s1, Table S1: PRISMA 2020 Checklist. Reference [40] is cited in the Supplementary Materials.
Funding
This article was supported by Qatar university Internal Grant No. QUST-1-CHS-2023-880. The findings achieved herein are solely the responsibility of the author[s].
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
Not applicable.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Sapra, A.; Bhandari, P.; Hughes, A.W. Diabetes Mellitus (Nursing); StatPearls Publishing: Treasure Island, FL, USA, 2021. [Google Scholar]
- Al-Lawati, J.A. Diabetes mellitus: A local and global public health emergency! Oman Med. J. 2017, 32, 177. [Google Scholar] [CrossRef]
- Diabetes. Available online: https://www.who.int/news-room/fact-sheets/detail/diabetes (accessed on 5 December 2022).
- Lam, A.A.; Lepe, A.; Wild, S.H.; Jackson, C. Diabetes comorbidities in low-and middle-income countries: An umbrella review. J. Glob. Health 2021, 11, 04040. [Google Scholar] [CrossRef] [PubMed]
- Pham, T.B.; Nguyen, T.T.; Truong, H.T.; Trinh, C.H.; Du, H.N.T.; Ngo, T.T.; Nguyen, L.H. Effects of diabetic complications on health-related quality of life impairment in Vietnamese patients with type 2 diabetes. J. Diabetes Res. 2020, 2020, 4360804. [Google Scholar] [CrossRef]
- Diabetes Symptoms|CDC. Available online: https://www.cdc.gov/diabetes/basics/symptoms.html (accessed on 6 December 2022).
- Yoon, J.-W.; Jun, H.-S. Autoimmune destruction of pancreatic β cells. Am. J. Ther. 2005, 12, 580–591. [Google Scholar] [CrossRef] [PubMed]
- Murea, M.; Ma, L.; Freedman, B.I. Genetic and environmental factors associated with type 2 diabetes and diabetic vascular complications. Rev. Diabet. Stud. RDS 2012, 9, 6. [Google Scholar] [CrossRef] [PubMed]
- Symptoms & Causes of Diabetes|NIDDK. Available online: https://www.niddk.nih.gov/health-information/diabetes/overview/symptoms-causes (accessed on 6 December 2022).
- Diabetes|Division of Global Health Protection|Global Health|CDC. Available online: https://www.cdc.gov/globalhealth/healthprotection/ncd/diabetes.html (accessed on 6 December 2022).
- Mekhilef, S.; Saidur, R.; Kamalisarvestani, M. Effect of dust, humidity and air velocity on efficiency of photovoltaic cells. Renew. Sustain. Energy Rev. 2012, 16, 2920–2925. [Google Scholar] [CrossRef]
- Bost, M.; Houdart, S.; Oberli, M.; Kalonji, E.; Huneau, J.F.; Margaritis, I. Dietary copper and human health: Current evidence and unresolved issues. J. Trace Elem. Med. Biol. Organ Soc. Miner. Trace Elem. (GMS) 2016, 35, 107–115. [Google Scholar] [CrossRef] [PubMed]
- Yin, J.-J.; Fu, P.P.; Lutterodt, H.; Zhou, Y.-T.; Antholine, W.E.; Wamer, W. Dual role of selected antioxidants found in dietary supplements: Crossover between anti-and pro-oxidant activities in the presence of copper. J. Agric. Food Chem. 2012, 60, 2554–2561. [Google Scholar] [CrossRef]
- Maritim, A.C.; Sanders, R.A.; Watkins, J.B., 3rd. Diabetes, oxidative stress, and antioxidants: A review. J. Biochem. Mol. Toxicol. 2003, 17, 24–38. [Google Scholar] [CrossRef]
- Fujii, J.; Homma, T.; Osaki, T. Superoxide Radicals in the Execution of Cell Death. Antioxidants 2022, 11, 501. [Google Scholar] [CrossRef]
- Galhardi, C.M.; Diniz, Y.S.; Faine, L.A.; Rodrigues, H.G.; Burneiko, R.C.M.; Ribas, B.O.; Novelli, E.L. Toxicity of copper intake: Lipid profile, oxidative stress and susceptibility to renal dysfunction. Food Chem. Toxicol. 2004, 42, 2053–2060. [Google Scholar] [CrossRef]
- Jomova, K.; Valko, M. Advances in metal-induced oxidative stress and human disease. Toxicology 2011, 283, 65–87. [Google Scholar] [CrossRef] [PubMed]
- Hettiarachchi, M.; Gunasekara, P.; Liyanage, C.; Lekamwasam, S. Effects of zinc and multimineral vitamin supplementation on glycemic and lipid control in adult diabetes. Diabetes Metab. Syndr. Obes. Targets Ther. 2011, 4, 53–60. [Google Scholar] [CrossRef] [PubMed]
- Alfawaz, H.; Naeef, A.F.; Wani, K.; Khattak, M.N.K.; Sabico, S.; Alnaami, A.M.; Al-Daghri, N.M. Improvements in glycemic, micronutrient, and mineral indices in Arab adults with pre-diabetes post-lifestyle modification program. Nutrients 2019, 11, 2775. [Google Scholar] [CrossRef]
- Zhang, G.; Sun, X.; Yuan, T.; Guo, C.; Zhou, Z.; Wang, L.; Dou, G. Certain Dietary Nutrients Reduce the Risk of Eye Affliction/Retinopathy in Individuals with Diabetes: National Health and Nutrition Examination Survey, 2003–2018. Int. J. Environ. Res. Public Health 2022, 19, 12173. [Google Scholar] [CrossRef] [PubMed]
- Tan, P.Y.; Mitra, S.R. Dietary copper and selenium are associated with insulin resistance in overweight and obese Malaysian adults. Nutr. Res. 2021, 93, 38–49. [Google Scholar] [CrossRef]
- Norbitt, C.F.; Kimita, W.; Ko, J.; Bharmal, S.H.; Petrov, M.S. Associations of habitual mineral intake with new-onset prediabetes/diabetes after acute pancreatitis. Nutrients 2021, 13, 3978. [Google Scholar] [CrossRef]
- Sobhani, S.R.; Mortazavi, M.; Kazemifar, M.; Azadbakht, L. The association between fast-food consumption with cardiovascular diseases risk factors and kidney function in patients with diabetic nephropathy. J. Cardiovasc. Thorac. Res. 2021, 13, 241–249. [Google Scholar] [CrossRef]
- Eshak, E.S.; Iso, H.; Maruyama, K.; Muraki, I.; Tamakoshi, A. Associations between dietary intakes of iron, copper and zinc with risk of type 2 diabetes mellitus: A large population-based prospective cohort study. Clin. Nutr. 2018, 37, 667–674. [Google Scholar] [CrossRef]
- Cui, Z.; Zhou, H.; Liu, K.; Wu, M.; Li, S.; Meng, S.; Meng, H. Dietary Copper and Selenium Intakes and the Risk of Type 2 Diabetes Mellitus: Findings from the China Health and Nutrition Survey. Nutrients 2022, 14, 2055. [Google Scholar] [CrossRef]
- Laouali, N.; MacDonald, C.-J.; Shah, S.; El Fatouhi, D.; Mancini, F.; Fagherazzi, G.; Boutron-Ruault, M.-C. Dietary copper/zinc ratio and type 2 diabetes risk in women: The E3N cohort study. Nutrients 2021, 13, 2502. [Google Scholar] [CrossRef] [PubMed]
- Rostami, H.A.A.; Marjani, A.; Mojerloo, M.; Rahimi, B.; Marjani, M. Effect of Spirulina on Lipid Profile, Glucose and Malondialdehyde Levels in Type 2 Diabetic Patients. Braz. J. Pharm. Sci. 2022, 58, e191140. [Google Scholar] [CrossRef]
- Armstrong, A.M.; Chestnutt, J.E.; Gormley, M.J.; Young, I.S. The effect of dietary treatment on lipid peroxidation and antioxidant status in newly diagnosed noninsulin dependent diabetes. Free Radic. Biol. Med. 1996, 21, 719–726. [Google Scholar] [CrossRef] [PubMed]
- Newcastle-Ottawa Scale. Available online: https://www.sciencedirect.com/topics/nursing-and-health-professions/newcastle-ottawa-scale (accessed on 15 November 2022).
- Wapnir, R.A. Copper absorption and bioavailability. Am. J. Clin. Nutr. 1998, 67, 1054S–1060S. [Google Scholar]
- Nishito, Y.; Kambe, T. Absorption mechanisms of iron, copper, and zinc: An overview. J. Nutr. Sci. Vitaminol. 2018, 64, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Hellman, N.E.; Kono, S.; Mancini, G.M.; Hoogeboom, A.J.; de Jong, G.J.; Gitlin, J.D. Mechanisms of copper incorporation into human ceruloplasmin. J. Biol. Chem. 2002, 277, 46632–46638. [Google Scholar] [CrossRef] [PubMed]
- Lin, C.-C.; Huang, H.-H.; Hu, C.-W.; Chen, B.-H.; Chong, I.-W.; Chao, Y.-Y.; Huang, Y.-L. Trace elements, oxidative stress and glycemic control in young people with type 1 diabetes mellitus. J. Trace Elem. Med. Biol. 2014, 28, 18–22. [Google Scholar] [CrossRef]
- Naka, T.; Kaneto, H.; Katakami, N.; Matsuoka, T.-A.; Harada, A.; Yamasaki, Y.; Matsuhisa, M.; Shimomura, I. Association of serum copper levels and glycemic control in patients with type 2 diabetes. Endocr. J. 2013, 60, 393–396. [Google Scholar] [CrossRef]
- Lian, S.; Zhang, T.; Yu, Y.; Zhang, B. Relationship of circulating copper level with gestational diabetes mellitus: A meta-analysis and systemic review. Biol. Trace Elem. Res. 2021, 199, 4396–4409. [Google Scholar] [CrossRef]
- Miyazaki, T.; Takenaka, T.; Inoue, T.; Sato, M.; Miyajima, Y.; Nodera, M.; Hanyu, M.; Ohno, Y.; Shibazaki, S.; Suzuki, H. Lipopolysaccharide-induced overproduction of nitric oxide and overexpression of iNOS and interleukin-1β proteins in zinc-deficient rats. Biol. Trace Elem. Res. 2012, 145, 375–381. [Google Scholar] [CrossRef]
- Basu, A.; Alman, A.C.; Snell-Bergeon, J.K. Associations of Dietary Antioxidants with Glycated Hemoglobin and Insulin Sensitivity in Adults with and without Type 1 Diabetes. J. Diabetes Res. 2022, 2022, 4747573. [Google Scholar] [CrossRef] [PubMed]
- Al-Abdullah, I.H.; Ayala, G.; Panigrahi, D.; Kumar, A.M.S.; Kumar, M.S.A. Neogenesis of pancreatic endocrine cells in copper-deprived rat models. Pancreas 2000, 21, 63–68. [Google Scholar] [CrossRef] [PubMed]
- Qiu, Q.; Zhang, F.; Zhu, W.; Wu, J.; Liang, M. Copper in diabetes mellitus: A meta-analysis and systematic review of plasma and serum studies. Biol. Trace Elem. Res. 2017, 177, 53–63. [Google Scholar] [CrossRef] [PubMed]
- Page, M.J.; McKenzie, J.E.; Bossuyt, P.M.; Boutron, I.; Hoffmann, T.C.; Mulrow, C.D.; Shamseer, L.; Tetzlaff, J.M.; Akl, E.A.; Brennan, S.E.; et al. The PRISMA 2020 statement: An updated guideline for reporting systematic reviews. BMJ 2021, 372, n71. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).