Enhanced Effect of β-Lactoglobulin Immunization in Mice with Mild Intestinal Deterioration Caused by Low-Dose Dextran Sulphate Sodium: A New Experimental Approach to Allergy Studies
Abstract
1. Introduction
2. Materials and Methods
2.1. Animals
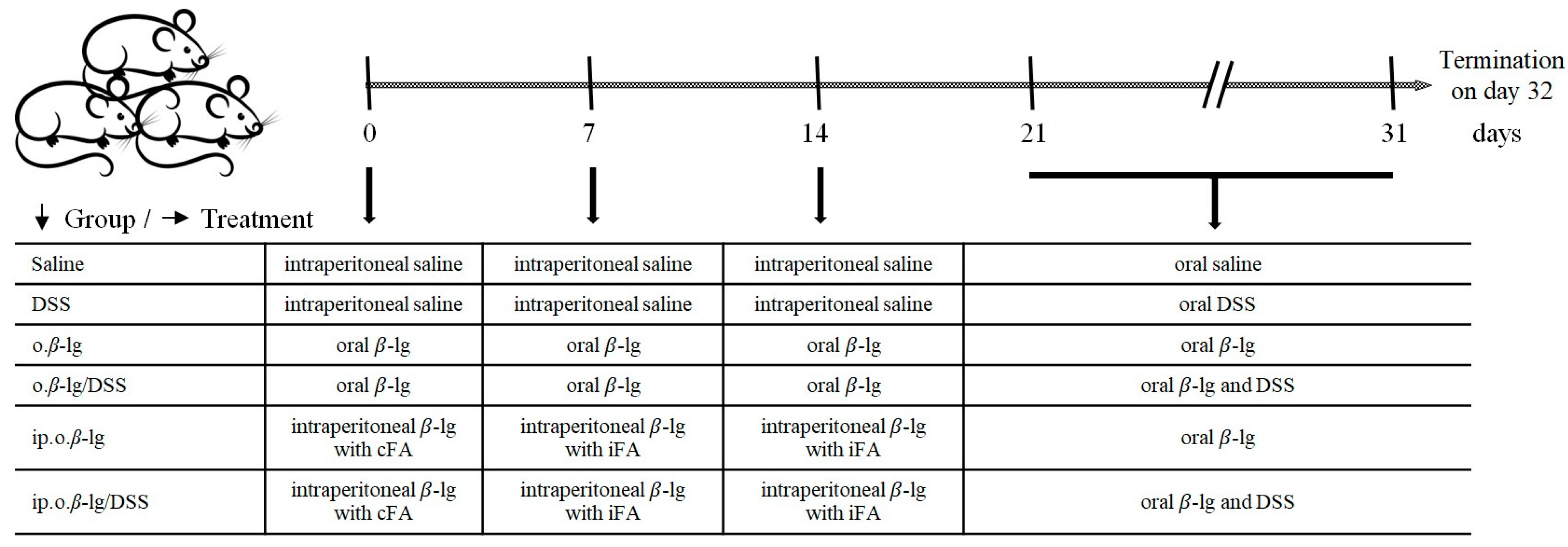
2.2. Experimental Design
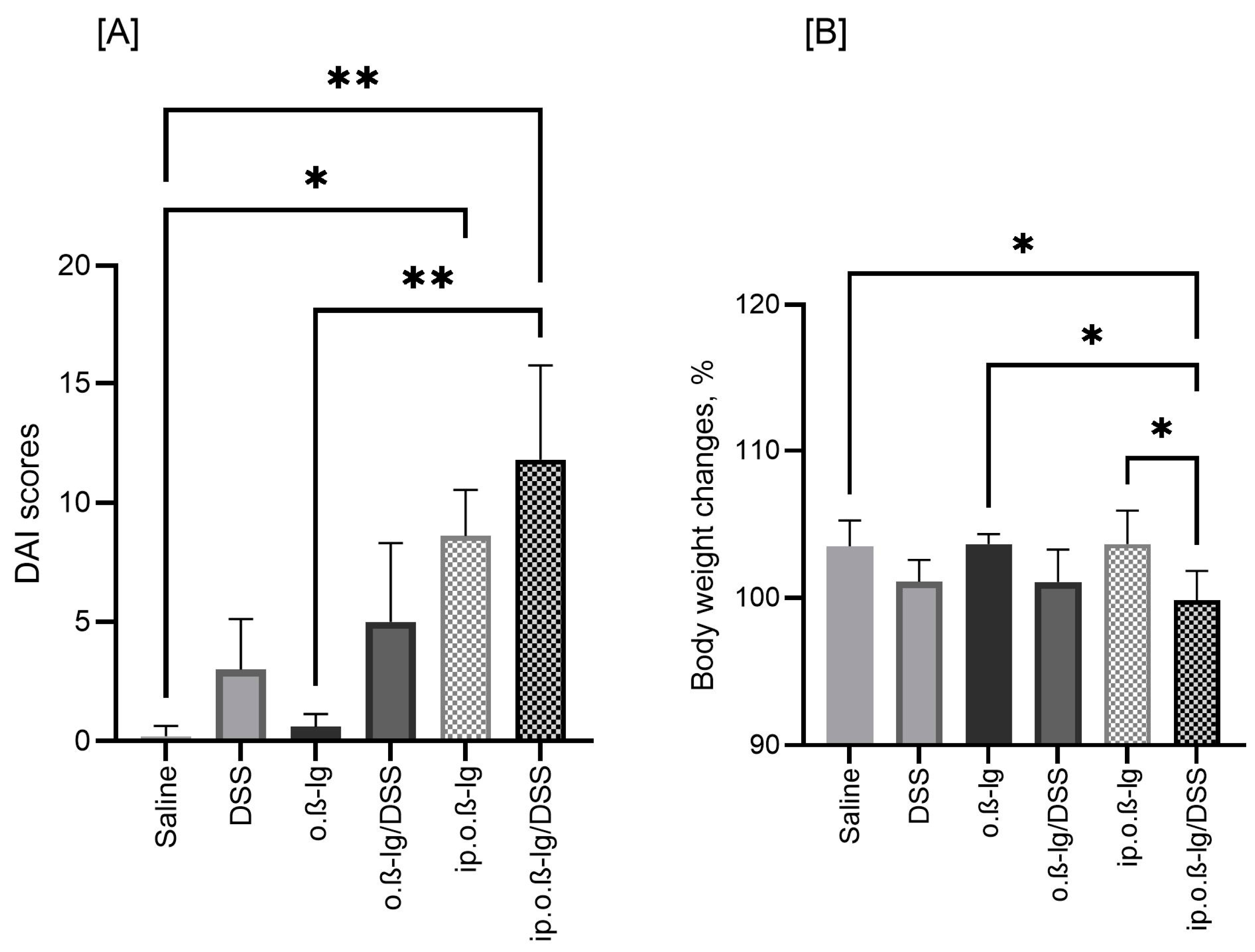
2.3. DAI and Body Weight
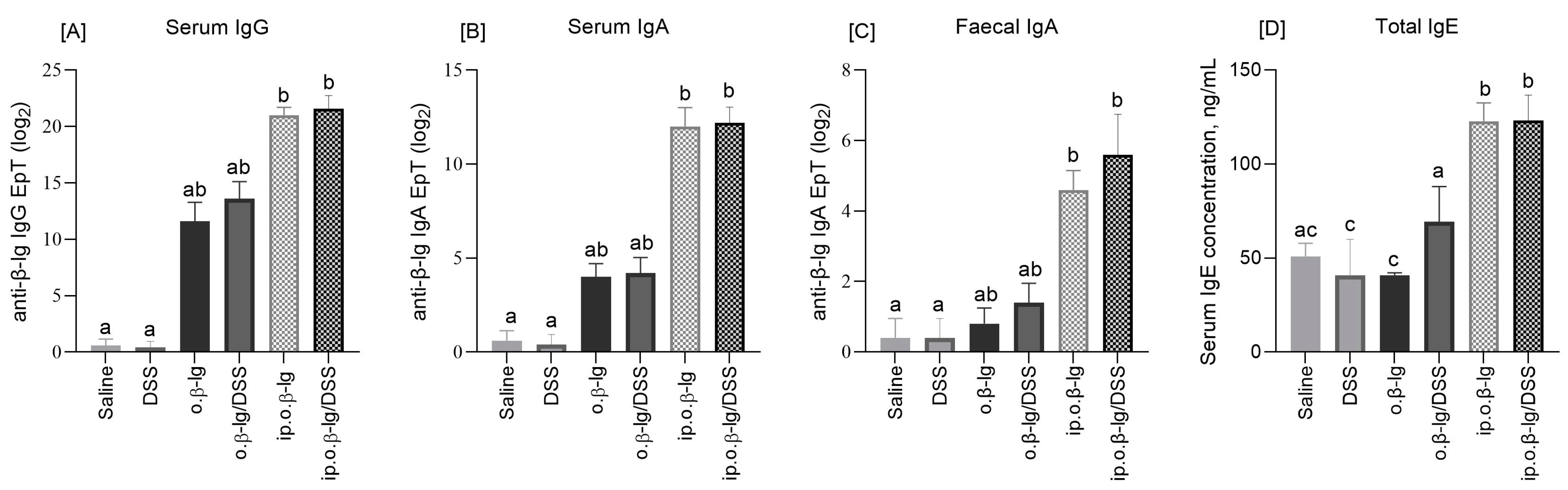
2.4. Antibody Measurements
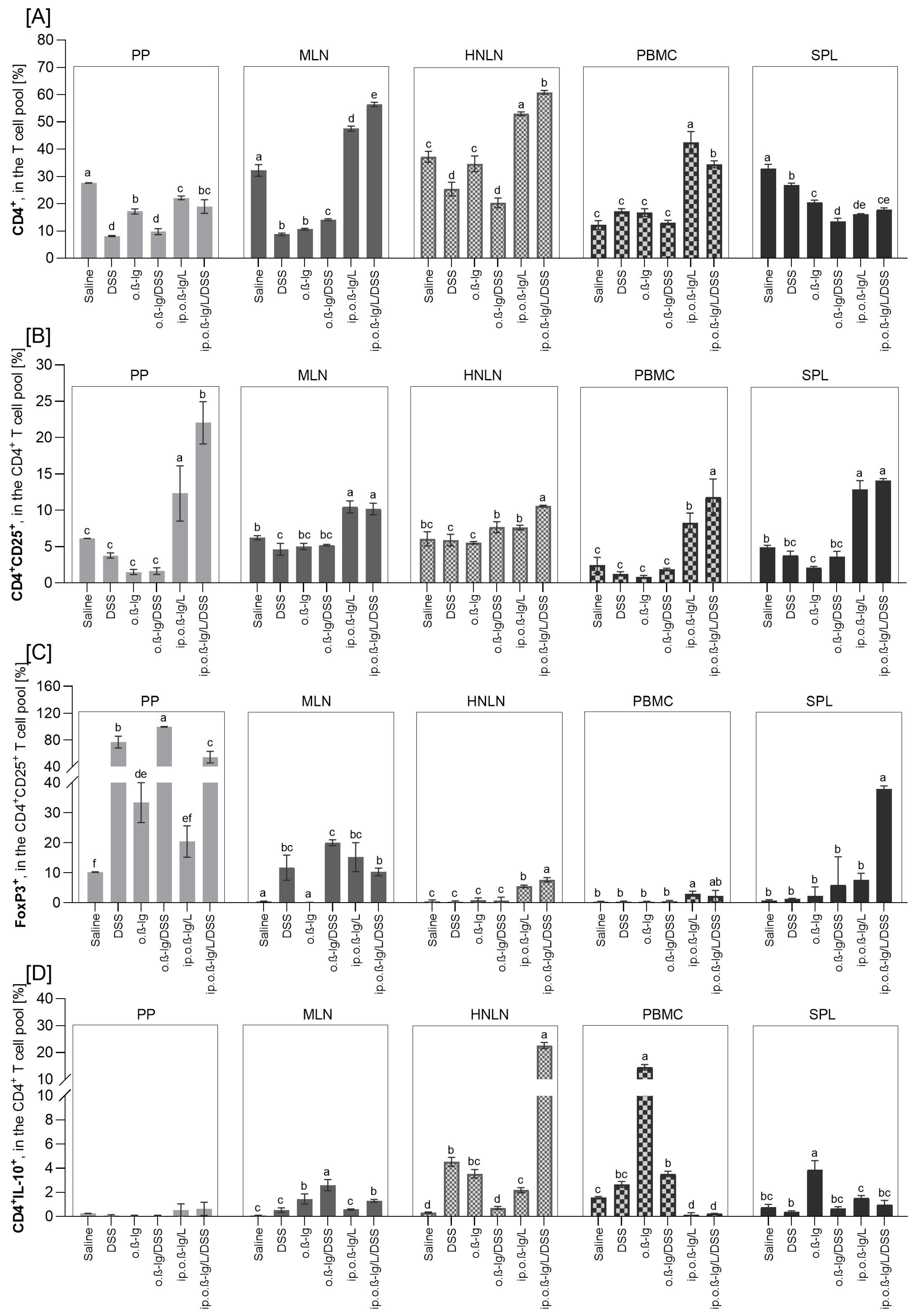
2.5. Lymphocyte Isolation and Phenotyping
2.6. Bacterial DNA Extraction and Cecal Microbiota Enumeration
2.7. Short-Chain Fatty Acid (SCFA) Determination
2.8. Total RNA Extraction, Reverse Transcription, and Gene Expression Analysis
| Target | Forward | Reverse | References |
|---|---|---|---|
| Bacterial primers: | |||
| Eubacteria | gtgstgcayggyygtcgtca | acgtcrtccmcnccttcctc | [14] |
| Bifidobacterium | tcgcgtc(c/t)ggtgtgaaag | ccacatccagc(a/g)tccac | [15] |
| Lactobacillus | agcagtagggaatcttcca | caccgctacacatggag | [16,17] |
| Mouse primers: | |||
| Interleukin 1β | ttgacggaccccaaaagatg | agaaggtgctcatgtcctcat | [18] |
| Interleukin 4 | gcctgggtcaagctgactac | atgtacgatgtcgccactcc | [12] |
| Interleukin 6 | gacaaagccagagtccttcagagag | ctaggtttgccgagtagatctc | [19] |
| Interleukin 8 | cacctcaagaacatccagagct | caagcagaactgaactaccatcg | [20] |
| Interferon gamma (IFN-γ) | cgctacacactgcatcttgg | tccttttgccagttcctcca | [21] |
| Tumor necrosis factor alpha (TNF-α) | ttcctgcaccctctgtctttc | cagttctatggcccagaccc | [13] |
| Transforming growth factor beta (TGF-β) | agacggaatacagggctttcgattca | cttgggcttgcgacccacgtagta | [22] |
| Toll-like receptor 2 (TLR-2) | gctagcctgccttgtttctc | ggctttttgttgccaaggct | [13] |
| Toll-like receptor 4 (TLR-4) | gacgctcatgtgagtgagtgta | agagatcacggaccaaggga | [13] |
| Claudin 2 (CLA2) | ggctgttaggcacatccat | tggcaccaacataggaactc | [23] |
| Claudin 12 (CLA12) | gtcctctcctttctggcaac | atgtcgatttcaatggcaga | [23] |
| Occludin (OCC) | gctgtgatgtgtgtgagctg | gacggtctacctggaggaac | [23] |
| Zonula occludens (ZO-1) | aggacaccaaagcatgtgag | ggcattcctgctggttaca | [23] |
| β-actin (ACT) | ggactcctatgtgggtgacgagg | gggagagcatagccctcgtagat | [24] |
2.9. Protein Extraction and Western Blotting
2.10. Measurements of Cytokines Through Flow Cytometry
2.11. Statistical Analysis
3. Results and Discussion
3.1. Sensitized β-lg Mice Respond to Low-Dose DSS with Reduced BW and Increased DAI Scores
3.2. No Expressed Effect of Low-Dose DSS on the Humoral Response of Mice
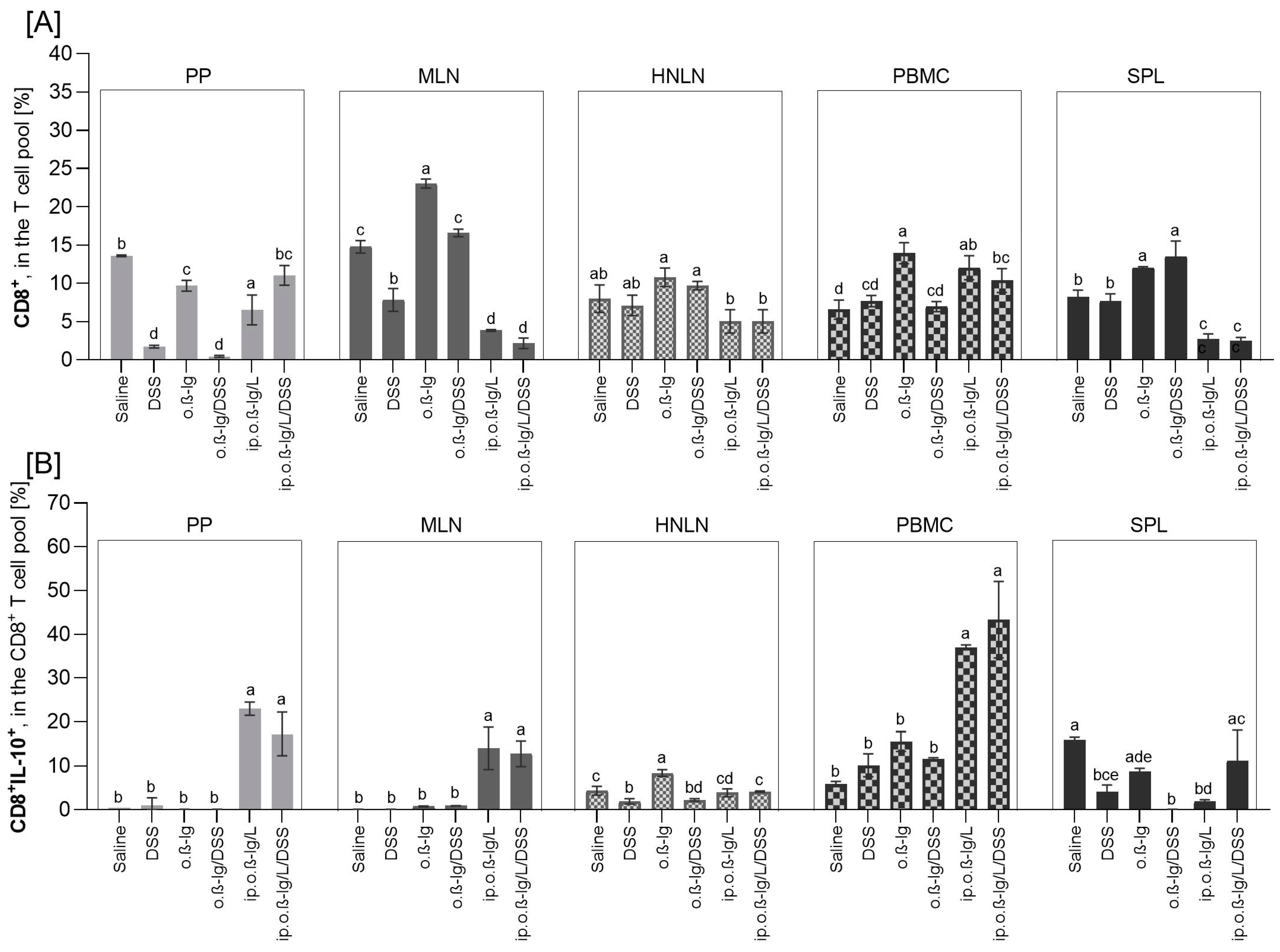
3.3. β-lg, Immunization Route and Low-Dose DSS affect T Cell Distribution in Inductive and Peripheral Tissues
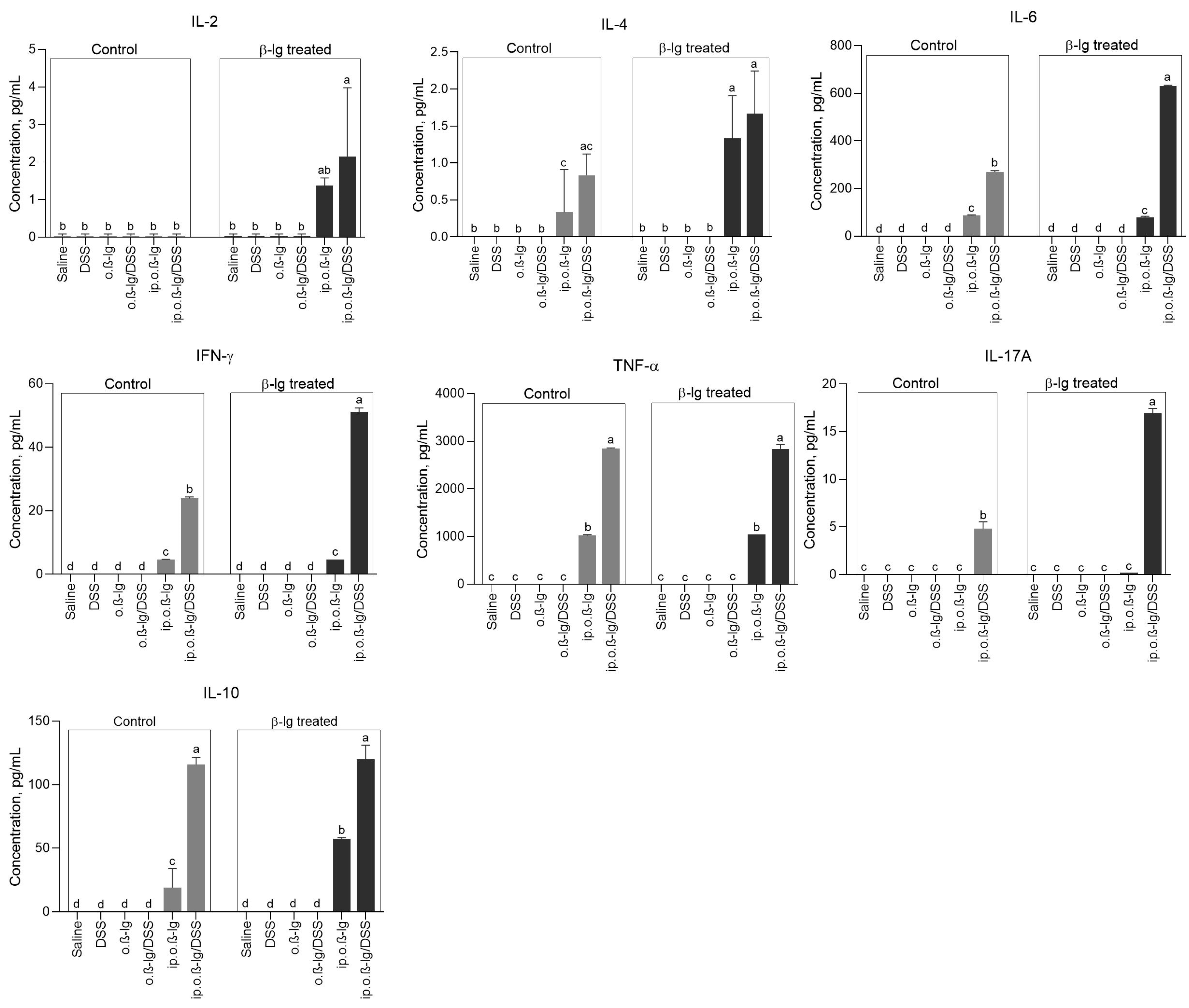
3.4. Sensitized β-lg Mice Given Low-Dose DSS Produced More Pro-Inflammatory Cytokines
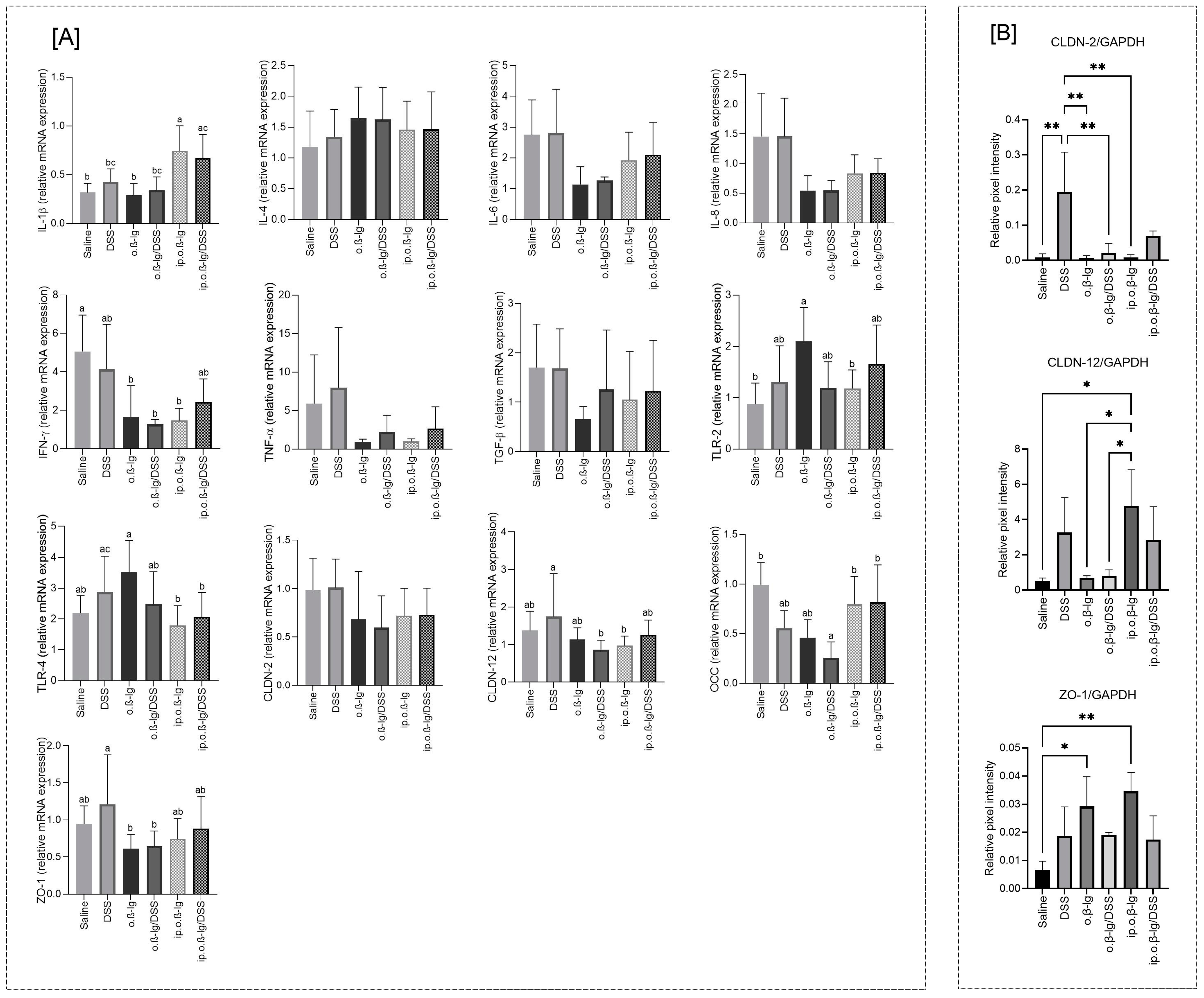
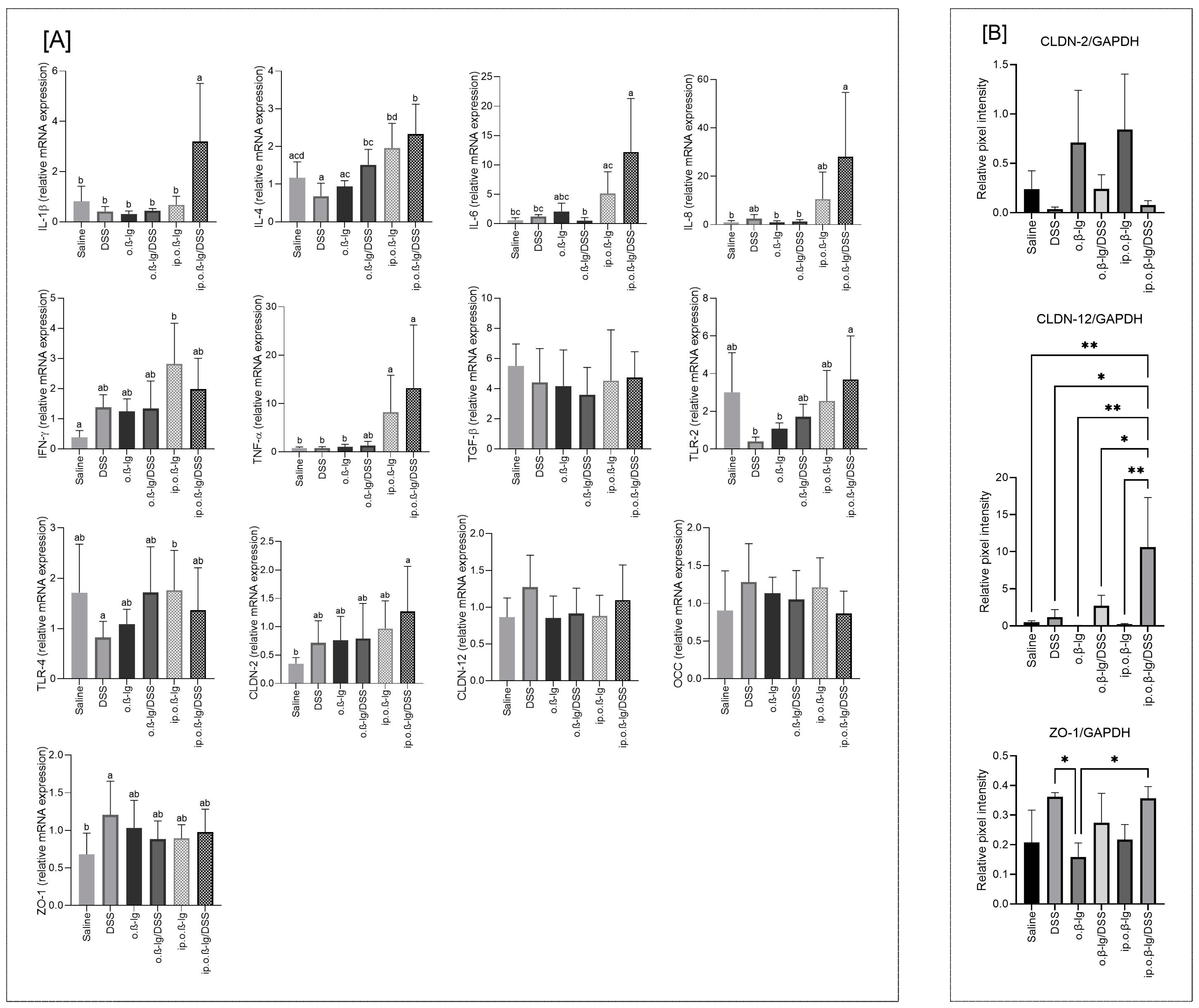
3.5. Gut-Tissue-Dependent Effect of β-lg and Low-Dose DSS on Gene Expression of Regulatory Molecules
3.6. Sensitizing Mice with β-lg in the Presence of Low-Dose DSS Does Not Cause Major Shifts in Total Bacterial Counts, but It Does Affect the Microbiota Balance and the Profile of Volatile Fatty Acids
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Kanagaratham, C.; Sallis, B.F.; Fiebiger, E. Experimental models for studying food allergy. Cell. Mol. Gastroenterol. Hepatol. 2018, 6, 356–369. [Google Scholar] [CrossRef] [PubMed]
- Schülke, S.; Albrecht, M. Mouse models for food allergies: Where do we stand? Cells 2019, 8, 546. [Google Scholar] [CrossRef]
- Cianferoni, A. Non-IgE-mediated anaphylaxis. J. Allergy Clin. Immunol. 2021, 147, 1123–1131. [Google Scholar] [CrossRef]
- Jarvinen, K.M.; Konstantinou, G.N.; Pilapil, M.; Arrieta, M.-C.; Noone, S.; Sampson, H.A.; Meddings, J.; Nowak-Węgrzyn, A. Intestinal permeability in children with food allergy on specific elimination diets. Pediatr. Allergy Immunol. 2013, 24, 589–595. [Google Scholar] [CrossRef] [PubMed]
- Stephen-Victor, E.; Crestani, E.; Chatila, T.A. Dietary and microbial determinants in food allergy. Immunity 2020, 53, 277–289. [Google Scholar] [CrossRef]
- Bøgh, K.L.; van Bilsen, J.; Głogowski, R.; López-Expósito, I.; Bouchaud, G.; Blanchard, C.; Bodinier, M.; Smit, J.; Pieters, R.; Bastiaan-Net, S.; et al. Current challenges facing the assessment of the allergenic capacity of food allergens in animal models. Clin. Transl. Allergy 2016, 6, 21. [Google Scholar] [CrossRef]
- Andreassen, M.; Rudi, K.; Angell, I.L.; Dirven, H.; Nygaard, U.C. Allergen immunization induces major changes in microbiota composition and short-chain fatty acid production in different gut segments in a mouse model of lupine food allergy. Int. Arch. Allergy Immunol. 2018, 177, 311–323. [Google Scholar] [CrossRef]
- Feehley, T.; Plunkett, C.H.; Bao, R.; Hong, S.M.C.; Culleen, E.; Belda-Ferre, P.; Campbell, E.; Aitoro, R.; Nocerino, R.; Paparo, L.; et al. Healthy infants harbor intestinal bacteria that protect against food allergy. Nat. Med. 2019, 25, 448–453. [Google Scholar] [CrossRef]
- Feuille, E.; Ceballos, C.; Benkov, K.; Nowak-Wegrzyn, A.H. Inflammatory bowel disease and food allergies. J. Allergy Clin. Immunol. 2015, 135, AB251. [Google Scholar] [CrossRef]
- Imanzadeh, F.; Nasri, P.; Sadeghi, S.; Sayyari, A.; Dara, N.; Abdollah, H.; Nilipoor, Y.; Mansuri, M.; Khatami, K.; Rouhani, P.; et al. Food allergy among Iranian children with inflammatory bowel disease: A preliminary report. J. Res. Med. Sci. 2015, 20, 855–859. [Google Scholar] [CrossRef]
- Chen, B.; Wu, Y.; Wu, H.; Meng, X.; Chen, H. Establishment of food allergy model in dextran sulfate sodium induced colitis mice. Foods 2023, 12, 1007. [Google Scholar] [CrossRef] [PubMed]
- Fotschki, J.; Szyc, A.M.; Laparra, J.M.; Markiewicz, L.H.; Wróblewska, B. Immune-modulating properties of horse milk administered to mice sensitised to cow milk. J. Dairy Sci. 2016, 99, 9395–9404. [Google Scholar] [CrossRef] [PubMed]
- Świątecka, D.; Złotkowska, D.; Markiewicz, L.H.; Szyc, A.M.; Wróblewska, B. Impact of whey proteins on the systemic and local intestinal level of mice with diet induced obesity. Food Funct. 2017, 8, 1708–1717. [Google Scholar] [CrossRef]
- Fuller, Z.; Louis, P.; Mihajlovski, A.; Rungapamestry, V.; Ratcliffe, B.; Duncan, A.J. Influence of cabbage processing methods and prebiotic manipulation of colonic microflora on glucosinolate breakdown in man. Br. J. Nutr. 2007, 98, 364–372. [Google Scholar] [CrossRef]
- Rinttila, T.; Kassinen, A.; Malinen, E.; Krogius, L.; Palva, A. Development of an extensive set of 16S rDNA-targeted primers for quantification of pathogenic and indigenous bacteria in faecal samples by real-time PCR. J. Appl. Microbiol. 2004, 97, 1166–1177. [Google Scholar] [CrossRef] [PubMed]
- Walter, J.; Hertel, C.; Tannock, G.W.; Lis, C.M.; Munro, K.; Hammes, W.P. Detection of Lactobacillus, Pediococcus, Leuconostoc, and Weissella species in human feces by using group-specific PCR primers and denaturing gradient gel electrophoresis. Appl. Environ. Microbiol. 2001, 67, 2578–2585. [Google Scholar] [CrossRef]
- Heilig, H.G.J.; Zoetendal, E.G.; Vaughan, E.E.; Marteau, P.; Akkermans, A.D.L.; de Vos, W.M. Molecular diversity of Lactobacillus spp. and other lactic acid bacteria in the human intestine as determined by specific amplification of 16S ribosomal DNA. Appl. Environ. Microbiol. 2002, 68, 114–123. [Google Scholar] [CrossRef]
- Carvalho, F.A.; Nalbantoglu, I.; Aitken, J.D.; Uchiyama, R.; Su, Y.; Doho, G.H.; Vijay-Kumar, M.; Gewirtz, A.T. Cytosolic flagellin receptor NLRC4 protects mice against mucosal and systemic challenges. Mucosal Immunol. 2012, 5, 288–298. [Google Scholar] [CrossRef]
- Rodriguez, T.E.; Falkowski, N.R.; Harkema, J.R.; Huffnagle, G.B. Role of Neutrophils in Preventing and Resolving Acute Fungal Sinusitis. Infect. Immun. 2007, 75, 5663–5668. [Google Scholar] [CrossRef][Green Version]
- Yasuda, Y.; Matsumura, Y.; Kasahara, K.; Ouji, N.; Sugiura, S.; Mikasa, K.; Kita, E. Microbial exposure early in life regulates airway inflammation in mice after infection with Streptococcus pneumoniae with enhancement of local resistance. Am. J. Physiol.-Lung Cell. Mol. Physiol. 2010, 298, L67–L78. [Google Scholar] [CrossRef]
- Liu, X.; Yan, C.; Yang, A.; Yu, E.; Yu, J.; Zhou, C.; Wang, Y.; Wang, K.; Sun, Y.; Cheng, Y. Efficacy of anti-programmed cell death protein 1 monoclonal antibody combined with bevacizumab and/or Pseudomonas aeruginosa injection in transplanted tumor of mouse forestomach carcinoma cell gastric cancer in mice and its mechanism in regulating tumor immune microenvironment. Clin. Exp. Immunol. 2023, 213, 328–338. [Google Scholar] [CrossRef] [PubMed]
- Jarnicki, A.G.; Lysaght, J.; Todryk, S.; Mills, K.H.G. Suppression of antitumor immunity by IL-10 and TGF-β-producing t cells infiltrating the growing tumor: Influence of tumor environment on the induction of CD4+ and CD8+ regulatory T cells. J. Immunol. 2006, 177, 896–904. [Google Scholar] [CrossRef] [PubMed]
- Bergmann, K.R.; Liu, S.X.L.; Tian, R.; Kushnir, A.; Turner, J.R.; Li, H.-L.; Chou, P.M.; Weber, C.R.; De Plaen, I.G. Bifidobacteria stabilize claudins at tight junctions and prevent intestinal barrier dysfunction in mouse necrotizing enterocolitis. Am. J. Pathol. 2013, 182, 1595–1606. [Google Scholar] [CrossRef] [PubMed]
- Maroof, A.; Penny, M.; Kingston, R.; Murray, C.; Islam, S.; Bedford, P.A.; Knight, S.C. Interleukin-4 can induce interleukin-4 production in dendritic cells. Immunology 2006, 117, 271–279. [Google Scholar] [CrossRef]
- Markiewicz, L.H.; Szymkiewicz, A.; Szyc, A.; Wróblewska, B. A simultaneous two-colour detection method of human IgG- and IgE-reactive proteins from lactic acid bacteria. J. Microbiol. Methods 2016, 126, 72–75. [Google Scholar] [CrossRef]
- Markiewicz, L.H.; Ogrodowczyk, A.M.; Wiczkowski, W.; Wróblewska, B. Phytate and butyrate differently influence the proliferation, apoptosis and survival pathways in human cancer and healthy colonocytes. Nutrients 2021, 13, 1887. [Google Scholar] [CrossRef]
- Yu, L.C.-H. Intestinal Epithelial Barrier Dysfunction in Food Hypersensitivity. J. Allergy 2012, 2012, 596081. [Google Scholar] [CrossRef]
- Samadi, N.; Klems, M.; Untersmayr, E. The role of gastrointestinal permeability in food allergy. Ann. Allergy Asthma Immunol. 2018, 121, 168–173. [Google Scholar] [CrossRef]
- Niewiem, M.; Grzybowska-Chlebowczyk, U. Intestinal barrier permeability in allergic diseases. Nutrients 2022, 14, 1893. [Google Scholar] [CrossRef] [PubMed]
- Wasilewska, E.; Zlotkowska, D.; Wroblewska, B. Yogurt starter cultures of Streptococcus thermophilus and Lactobacillus bulgaricus ameliorate symptoms and modulate the immune response on mouse model of dextran sulfate sodium-induced colitis. J. Dairy Sci. 2019, 102, 37–53. [Google Scholar] [CrossRef]
- Bouchikhi, I.; Grar, H.; Guendouz, M.; Brahimi, C.E.; Kheroua, O.; Saidi, D.; Kaddouri, H. Intestinal inflammation enhances the development of egg white-induced anaphylaxis in Balb/c mice. Rev. Française Allergol. 2019, 59, 369–379. [Google Scholar] [CrossRef]
- Martino, D.; Neeland, M.; Dang, T.; Cobb, J.; Ellis, J.; Barnett, A.; Tang, M.; Vuillermin, P.; Allen, K.; Saffery, R. Epigenetic dysregulation of naive CD4+ T-cell activation genes in childhood food allergy. Nat. Commun. 2018, 9, 3308. [Google Scholar] [CrossRef] [PubMed]
- McGee, H.S.; Agrawal, D.K. Naturally occurring and inducible T-regulatory cells modulating immune response in allergic asthma. Am. J. Respir. Crit. Care Med. 2009, 180, 211–225. [Google Scholar] [CrossRef] [PubMed]
- Xin, L.; Gao, J.; Ge, X.; Tian, C.; Ma, W.; Tianc, Z.; Zheng, X.; Hou, J. Increased pro-inflammatory cytokine-secreting regulatory T cells are correlated with the plasticity of T helper cell differentiation and reflect disease status in asthma. Respir. Med. 2018, 143, 129–138. [Google Scholar] [CrossRef]
- Chen, T.; Hou, X.; Ni, Y.; Du, W.; Han, H.; Yu, Y.; Shi, G. The imbalance of FOXP3/GATA3 in regulatory T cells from the peripheral blood of asthmatic patients. J. Immunol. Res. 2018, 2018, 3096183. [Google Scholar] [CrossRef] [PubMed]
- Lan, F.; Zhang, N.; Bachert, C.; Zhang, L. Stability of regulatory T cells in T helper 2–biased allergic airway diseases. Allergy 2020, 75, 1918–1926. [Google Scholar] [CrossRef]
- Wang, Y.; Su, M.A.; Wan, Y.Y. An essential role of the transcription factor GATA-3 for the function of regulatory T cells. Immunity 2011, 35, 337–348. [Google Scholar] [CrossRef]
- Maul, J.; Loddenkemper, C.; Mundt, P.; Berg, E.; Giese, T.; Stallmach, A.; Zeitz, M’.; Duchmann, R. Peripheral and intestinal regulatory CD4+CD25high T cells in inflammatory bowel disease. Gastroenterology 2005, 128, 1868–1878. [Google Scholar] [CrossRef]
- Nedelkopoulou, N.; Dhawanc, A.; Xiniasd, I.; Gidarise, D.; Farmakia, E. Interleukin 10: The critical role of a pleiotropic cytokine in food allergy. Allergol. Immunopathol. 2020, 48, 401–408. [Google Scholar] [CrossRef]
- Yogev, N.; Bedke, T.; Kobayashi, Y.; Brockmann, L.; Lukas, D.; Regen, T.; Croxford, A.L.; Nikolav, A.; Hovelmeyer, N.; von Stebut, E.; et al. CD4+ T-cell-derived IL-10 promotes CNS inflammation in mice by sustaining effector T cell survival. Cell Rep. 2022, 38, 110565. [Google Scholar] [CrossRef]
- Brockmann, L.; Soukou, S.; Steglich, B.; Czarnewski, P.; Zhao, L.; Wende, S.; Bedke, T.; Ergen, C.; Manthey, C.; Agalioti, T.; et al. Molecular and functional heterogeneity of IL-10-producing CD4+ T cells. Nat. Commun. 2018, 9, 5457. [Google Scholar] [CrossRef] [PubMed]
- Churlaud, G.; Pitoiset, F.; Jebbawi, F.; Lorenzon, R.; Bellier, B.; Rosenzwajg, M.; Klatzmann, D. Human and mouse CD8+CD25+FOXP3+ regulatory T cells at steady state and during interleukin-2 therapy. Front. Immunol. 2015, 6, 171. [Google Scholar] [CrossRef] [PubMed]
- Lin, L.; Dai, F.; Wei, J.; Chen, Z. CD8+ Tregs ameliorate inflammatory reactions in a murine model of allergic rhinitis. Allergy Asthma Clin. Immunol. 2021, 17, 74. [Google Scholar] [CrossRef]
- Kaushik, A.; Dunham, D.; Han, X.; Do, E.; Andorf, S.; Gupta, S.; Fernandes, A.; Kost, L.E.; Sindher, S.B.; Yu, W.; et al. CD8+ T cell differentiation status correlates with the feasibility of sustained unresponsiveness following oral immunotherapy. Nat. Commun. 2022, 13, 6646. [Google Scholar] [CrossRef]
- Tenorio, I.A.; Sabra, A.; Sabra, S. Decreased subsets of T lymphocytes CD8 in patients with IgE food allergy. World Allergy Organ. J. 2015, 8, A272. [Google Scholar] [CrossRef]
- de Vos, G.; Milush, J.; Aaron, J.; Pichardo, Y.; Nazari, R.; Chin, M.; Serebrisky, D.; Rosenstreich, D.; Nixon, D.; Wiznia, A.; et al. A decrease of CD3+CD8+ T-cells is significantly associated with atopy, wheezing and increased IgE levels in young children. J. Allergy Clin. Immunol. 2010, 127, AB259. [Google Scholar] [CrossRef]
- Smith, L.K.; Boukhaled, G.M.; Condotta, S.A.; Mazouz, S.; Guthmiller, J.J.; Vijay, R.; Butler, N.S.; Bruneau, J.; Shoukry, N.H.; Krawczyk, C.M.; et al. Interleukin-10 directly inhibits CD8+ T cell function by enhancing N-glycan branching to decrease antigen sensitivity. Immunity 2018, 48, 299–312.e5. [Google Scholar] [CrossRef]
- Xu, L.; Kitani, A.; Fuss, I.; Strober, W. Cutting edge: Regulatory T cells induce CD4+CD25-Foxp3- T cells or are self-induced to become Th17 cells in the absence of exogenous TGF-beta. J. Immunol. 2007, 178, 6725–6729. [Google Scholar] [CrossRef] [PubMed]
- Lohr, J.; Knoechel, B.; Wang, J.J.; Villarino, A.V.; Abbas, A.K. Role of IL-17 and regulatory T lymphocytes in a systemic autoimmune disease. J. Exp. Med. 2006, 203, 2785–2791. [Google Scholar] [CrossRef]
- Mangan, P.R.; Harrington, L.E.; O’Quinn, D.B.; Helms, W.S.; Bullard, D.C.; Elson, C.O.; Hatton, R.D.; Wahl, S.M.; Schoeb, T.R.; Weaver, C.T. Transforming growth factor-b induces development of the TH17 lineage. Nature 2006, 441, 231–234. [Google Scholar] [CrossRef]
- Bettelli, E.; Carrier, Y.; Gao, W.; Korn, T.; Strom, T.B.; Oukka, M.; Weiner, H.L.; Kuchroo, V.K. Reciprocal developmental pathways for the generation of pathogenic effector TH17 and regulatory T cells. Nature 2006, 441, 235–238. [Google Scholar] [CrossRef] [PubMed]
- Kuwabara, T.; Ishikawa, F.; Kondo, M.; Kakiuchi, T. The role of IL-17 and related cytokines in inflammatory autoimmune diseases. Mediat. Inflamm. 2017, 2017, 3908061. [Google Scholar] [CrossRef] [PubMed]
- Kinashi, Y.; Hase, K. Partners in leaky gut syndrome: Intestinal dysbiosis and autoimmunity. Front. Immunol. 2021, 12, 673708. [Google Scholar] [CrossRef] [PubMed]
- Sutton, C.E.; Lalor, S.J.; Sweeney, C.M.; Brereton, C.F.; Lavelle, E.C.; Mills, K.H. Interleukin-1 and IL-23 induce innate IL-17 production from γδ T cells, amplifying Th17 responses and autoimmunity. Immunity 2009, 31, 331–341. [Google Scholar] [CrossRef] [PubMed]
- Rawat, M.; Nighot, M.; Al-Sadi, R.; Gupta, Y.; Viszwapriya, D.; Yochum, G.; Koltun, W.; Ma, T.Y. IL1B increases intestinal tight junction permeability by upregulation of MIR200C-3p, which degrades occludin mRNA. Gastroenterology 2020, 159, 1375–1389. [Google Scholar] [CrossRef]
- Kaminsky, L.W.; Al-Sadi, R.; Ma, T.Y. IL-1β and the intestinal epithelial tight junction barrier. Front. Immunol. 2021, 12, 767456. [Google Scholar] [CrossRef]
- Złotkowska, D.; Stachurska, E.; Fuc, E.; Wróblewska, B.; Mikołajczyk, A.; Wasilewska, E. Differences in regulatory mechanisms induced by β-lactoglobulin and κ-casein in cow’s milk allergy mouse model—In Vivo and Ex Vivo studies. Nutrients 2021, 13, 349. [Google Scholar] [CrossRef]
- Złotkowska, D.; Kuczyński, M.; Fuc, E.; Fotschki, J.; Wróblewska, B. Glycation of whey proteins increases the ex vivo immune response of lymphocytes sensitised to β-lactoglobulin. Nutrients 2023, 15, 3110. [Google Scholar] [CrossRef]
- Fuc, E.; Złotkowska, D.; Wróblewska, B. Milk and meat allergens from Bos taurus β-lactoglobulin, α-casein, and bovine serum albumin: An in-vivo study of the immune response in mice. Nutrients 2019, 11, 2095. [Google Scholar] [CrossRef]
- Lu, Z.; Ding, L.; Lu, Q.; Chen, Y.-H. Claudins in intestines. Distribution and functional significance in health and diseases. Tissue Barriers 2013, 1, e24978. [Google Scholar] [CrossRef]
- Kim, S.; Kim, G.-H. Roles of claudin-2, ZO-1 and occludin in leaky HK-2 cells. PLoS ONE 2017, 12, e0189221. [Google Scholar] [CrossRef] [PubMed]
- Mu, Q.; Kirby, J.; Reilly, C.M.; Luo, X.M. Leaky gut as a danger signal for autoimmune diseases. Front. Immunol. 2017, 8, 598. [Google Scholar] [CrossRef] [PubMed]
- Lameris, A.L.; Huybers, S.; Kaukinen, K.; Mäkelä, T.H.; Bindels, R.J.; Hoenderop, J.G.; Nevalainen, P.I. Expression profiling of claudins in the human gastrointestinal tract in health and during inflammatory bowel disease. Scand. J. Gastroenterol. 2013, 48, 58–69. [Google Scholar] [CrossRef]
- Sturgeon, C.; Fasano, A. Zonulin, a regulator of epithelial and endothelial barrier functions, and its involvement in chronic inflammatory diseases. Tissue Barriers 2016, 4, e1251384. [Google Scholar] [CrossRef] [PubMed]
- Fasano, A. All disease begins in the (leaky) gut: Role of zonulin-mediated gut permeability in the pathogenesis of some chronic inflammatory diseases. F1000Research 2020, 9, 69. [Google Scholar] [CrossRef]
- Kuo, W.-T.; Zuo, L.; Odenwald, M.A.; Madha, S.; Singh, G.; Gurniak, C.B.; Abraham, C.; Turner, J.R. The tight junction protein zo-1 is dispensable for barrier function but critical for effective mucosal repair. Gastroenterology 2021, 161, 1924–1939. [Google Scholar] [CrossRef]
- Kalliomäki, M.; Kirjavainen, P.; Eerola, E.; Kero, P.; Salminen, S.; Isolauri, E. Distinct patterns of neonatal gut microflora in infants in whom atopy was and was not developing. J. Allergy Clin. Immunol. 2001, 107, 129–134. [Google Scholar] [CrossRef]
- Bisgaard, H.; Li, N.; Bonnelykke, K.; Chawes, B.L.; Skov, T.; Paludan-Müller, G.; Stokholm, J.; Smith, B.; Krogfelt, K.A. Reduced diversity of the intestinal microbiota during infancy is associated with increased risk of allergic disease at school age. J. Allergy Clin. Immunol. 2011, 128, 646–652. [Google Scholar] [CrossRef]
| Saline | DSS | o.β-lg | o.β-lg/DSS | ip.o.β-lg | ip.o.β-lg/DSS | |
|---|---|---|---|---|---|---|
| TNB 3 | 10.44 ± 0.23 | 10.34 ± 0.12 | 10.63 ± 0.20 | 10.26 ± 0.25 | 10.24 ± 0.04 | 10.35 ± 0.04 |
| Lactobacillus spp. | 7.78 ± 0.19 b | 7.42 ± 0.03 cd | 7.06 ± 0.12 a | 7.62 ± 0.08 bd | 7.64 ± 0.02 bd | 7.11 ± 0.13 ac |
| Bifidobacterium spp. | 6.08 ± 0.49 | 6.08 ± 0.13 | 5.93 ± 0.06 | 6.04 ± 0.18 | 5.76 ± 0.32 | 6.00 ± 0.12 |
| Saline | DSS | o.β-lg | o.β-lg/DSS | ip.o.β-lg | ip.o.β-lg/DSS | |
|---|---|---|---|---|---|---|
| SCFA, µmol/g of digesta | ||||||
| Acetic acid | 60.20 ± 10.21 bd | 64.17 ± 7.16 be | 54.26 ± 8.55 cde | 47.32 ± 1.30 ad | 35.72 ± 1.93 a | 71.22 ± 8.34 b |
| Propionic acid | 10.71 ± 5.53 ab | 13.19 ± 4.84 ac | 7.54 ± 1.82 bc | 9.20 ± 1.18 ab | 5.27 ± 1.87 b | 15.79 ± 3.04 a |
| Butyric acid | 21.27 ± 8.72 | 15.89 ± 3.24 | 23.78 ± 2.44 | 13.41 ± 0.55 | 12.70 ± 2.48 | 23.27 ± 8.65 |
| Valeric acid | 0.57 ± 0.04 cd | 0.53 ± 0.04 cd | 0.68 ± 0.11 ac | 0.49 ± 0.06 bd | 0.35 ± 0.02 b | 0.77 ± 0.10 a |
| Isovaleric acid | 0.14 ± 0.03 ab | 0.14 ± 0.05 ab | 0.22 ± 0.03 b | 0.23 ± 0.06 b | 0.12 ± 0.01 a | 0.20 ± 0.06 ab |
| Isobutyric acid | 0.47 ± 0.04 ab | 0.57 ± 0.10 ab | 0.43 ± 0.16 ab | 0.51 ± 0.09 ab | 0.35 ± 0.07 a | 0.62 ± 0.20 b |
| Total PSCFA 3 | 1.17 ± 0.06 c | 1.24 ± 0.18 c | 1.33 ± 0.19 bcd | 1.22 ± 0.18 c | 0.81 ± 0.08 a | 1.59 ± 0.07 bd |
| Total SCFA | 93.36 ± 9.86 bd | 94.48 ± 8.50 bd | 86.91 ± 12.86 cd | 71.15 ± 1.88 ac | 54.50 ± 4.52 a | 111.87 ± 8.04 b |
| Profile, % of total SCFA | ||||||
| Acetic acid | 64.22 ± 5.02 | 67.83 ± 1.63 | 62.36 ± 0.83 | 66.52 ± 1.72 | 65.67 ± 2.20 | 63.60 ± 5.31 |
| Propionic acid | 11.30 ± 4.94 | 13.71 ± 4.26 | 8.58 ± 0.93 | 12.91 ± 1.32 | 9.61 ± 2.94 | 14.27 ± 3.39 |
| Butyric acid | 23.22 ± 9.78 | 17.14 ± 5.09 | 27.52 ± 1.48 | 18.85 ± 0.75 | 23.22 ± 3.48 | 20.70 ± 7.27 |
| Total PSCFA | 1.26 ± 0.12 ab | 1.32 ± 0.22 a | 1.54 ± 0.21 b | 1.73 ± 0.29 ab | 1.50 ± 0.11 ab | 1.43 ± 0.12 ab |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Złotkowska, D.; Markiewicz, L.H.; Ogrodowczyk, A.M.; Wróblewska, B.; Wasilewska, E. Enhanced Effect of β-Lactoglobulin Immunization in Mice with Mild Intestinal Deterioration Caused by Low-Dose Dextran Sulphate Sodium: A New Experimental Approach to Allergy Studies. Nutrients 2024, 16, 3430. https://doi.org/10.3390/nu16203430
Złotkowska D, Markiewicz LH, Ogrodowczyk AM, Wróblewska B, Wasilewska E. Enhanced Effect of β-Lactoglobulin Immunization in Mice with Mild Intestinal Deterioration Caused by Low-Dose Dextran Sulphate Sodium: A New Experimental Approach to Allergy Studies. Nutrients. 2024; 16(20):3430. https://doi.org/10.3390/nu16203430
Chicago/Turabian StyleZłotkowska, Dagmara, Lidia Hanna Markiewicz, Anna Maria Ogrodowczyk, Barbara Wróblewska, and Ewa Wasilewska. 2024. "Enhanced Effect of β-Lactoglobulin Immunization in Mice with Mild Intestinal Deterioration Caused by Low-Dose Dextran Sulphate Sodium: A New Experimental Approach to Allergy Studies" Nutrients 16, no. 20: 3430. https://doi.org/10.3390/nu16203430
APA StyleZłotkowska, D., Markiewicz, L. H., Ogrodowczyk, A. M., Wróblewska, B., & Wasilewska, E. (2024). Enhanced Effect of β-Lactoglobulin Immunization in Mice with Mild Intestinal Deterioration Caused by Low-Dose Dextran Sulphate Sodium: A New Experimental Approach to Allergy Studies. Nutrients, 16(20), 3430. https://doi.org/10.3390/nu16203430







