Abstract
The naturally occurring compounds ecdysterone and turkesterone, which are present in plants, including Rhaponticum carthamoides Willd. (Iljin), Spinacia oleracea L., Chenopodium quinoa Willd., and Ajuga turkestanica (Regel) Briq, are widely recognized due to their possible advantages for both general health and athletic performance. The current review investigates the beneficial biological effects of ecdysterone and turkesterone in nutrition, highlighting their roles not only in enhancing athletic performance but also in the management of various health problems. Plant-based diets, associated with various health benefits and environmental sustainability, often include sources rich in phytoecdysteroids. However, the therapeutic potential of phytoecdysteroid-rich extracts extends beyond sports nutrition, with promising applications in treating chronic fatigue, cardiovascular diseases, and neurodegenerative disorders.
1. Introduction
Ecdysteroids are a class of invertebrate steroid hormones, first found in insects, in which they regulate activities such as molting, development, and reproduction, including the critical metamorphic phases in arthropods [1,2]. The first ecdysteroid, ecdysone, was isolated from silkworm pupae by Butenandt and Karlson in 1954, and its structure was presented in 1965 by Huber and Hoppe [3,4]. Nowadays, over 550 ecdysteroids are known [5]. They possess a tetrahydroxylated four-ring structure, a cyclopentanoperhydrophenanthrene skeleton consisting of 27–30 carbon atoms with a β-side chain at C17, originating from cholesterol or alternative sterols [1,6].
Ecdysteroids are classified into three main groups based on their natural origin, including phytoecdysteroids (PEs), zooecdysteroids, and mycoecdysteroids [1]. Phytoecdysteroids are a class of bioactive molecules produced by plants as a defense against herbivorous insects [7]. They are widely distributed through the plant kingdom, and research results suggest that only around 6% of plant species contain detectable levels of PEs [6,8,9,10]. Phytoecdysteroids were first reported in the mid-1960s and discovered in diverse plant sources, including the leaves of Podocarpus nakaii, the pinnae of Pteridium aquilinum, the bark of Podocarpus elatus, the roots of Achyranthes fauriei, etc. [11]. Some ecdysteroids, including ecdysone, 20-hydroxyecdysone (20HE), makisterone A, and ajugasterone C, are found in both botanical and zoological environments [6,8]. Phytoecdysteroids occur in a variety of plant families, including Asteraceae, Amaranthaceae, Commelinaceae, Liliaceae, Lamiaceae, Magnoliaceae, Podocarpaceae, Ranunculaceae, etc. Representative species of plant families include Ajuga turkestanica (Regel) Briq. (A. turkestanica), Rhaponticum carthamoides Willd. (Iljin) (R. carthamoides), Pfaffia glomerata (Spreng.) Pedersen, Cyanotis arachnoidea C.B. Clarke (C. arachnoidea), etc. [1,5,12]. These plants have been used as adaptogens, antioxidants, tonifying agents, and to promote muscle growth and strength since ancient times [13,14,15]. The most common and isolated PEs from these plants are 20HE, ajugasterone C, turkesterone, polypodine B, ponasterones A, B, and C, etc. [16,17]. Some of the essential factors for ecdysteroid levels in plants are the developmental stage, the growing conditions, and the ecotype [18]. It is considered that 20HE does not bind to the androgen receptor, suggesting that ecdysteroids may exhibit anabolic effects via a mechanism other than androgens. It has been proposed that they activate the PI3K pathway [19]. Dietary supplements (DSs), as well as dietary regimes and plant-based diets, have grown in popularity in recent years for a variety of reasons, including popular objectives, such as extending lifespan, improving health quality, facilitating weight loss efforts, and improving athletic performance [20,21,22,23,24,25]. In general, DSs are an important part of athletes’ dietary plans. These products not only enrich the diet but also have an essential role in performance, adaptation, endurance, and recovery [26,27]. With regard to the DSs intended to increase physical strength, some contain ecdysteroids due to their anabolic effects, adaptogenic potential, and ergogenic properties [23]. In general, the ecdysteroids usually found in DSs are 20HE and turkesterone [28]. Moreover, utilizing naturally derived steroids for enhancing muscle performance is preferred over using anabolic steroids [29,30]. Notably, 20HE is widely available in large amounts at competitive market costs and can be easily isolated and purified from selected plant species recognized for high accumulation rates [31]. Furthermore, the inclusion of 20HE in World Anti-Doping Agency (WADA) monitoring programs since 2020 demonstrates the increased regulatory attention given to these substances [15,32,33]. The inclusion of 20HE in the WADA monitoring program could be regarded as controversial because the compound also presents in some common foods, such as spinach (Spinacia oleracea L., Chenopodiaceae) and quinoa (Chenopodium quinoa Willd., Chenopodiaceae). These plant species are considered functional foods with good nutrition profiles [34,35,36,37] and are included in the nutritional regimens of many professional athletes, plant-based diets, and other healthy regimens.
Plant-based diets not only gained popularity in the last several years but are also associated with a reduction in the risk of chronic diseases, including diabetes, heart disease, and certain cancers. These diets are typically higher in fiber, vitamins, minerals, and antioxidants [38]. Additionally, it is considered that plant-based diets have a smaller environmental footprint compared to diets high in animal products. Moreover, they often include specific foods, such as spinach quinoa [39]. The most common sources of ecdysterone and turkesterone in DSs are plants such as C. arachnoides, A. turkestanica, and R. carthamoides [28,34].
The purpose of this review is to assess the evidence for the beneficial role of ecdysteroids in human nutrition and indicate prospects for future research.
2. Materials and Methods
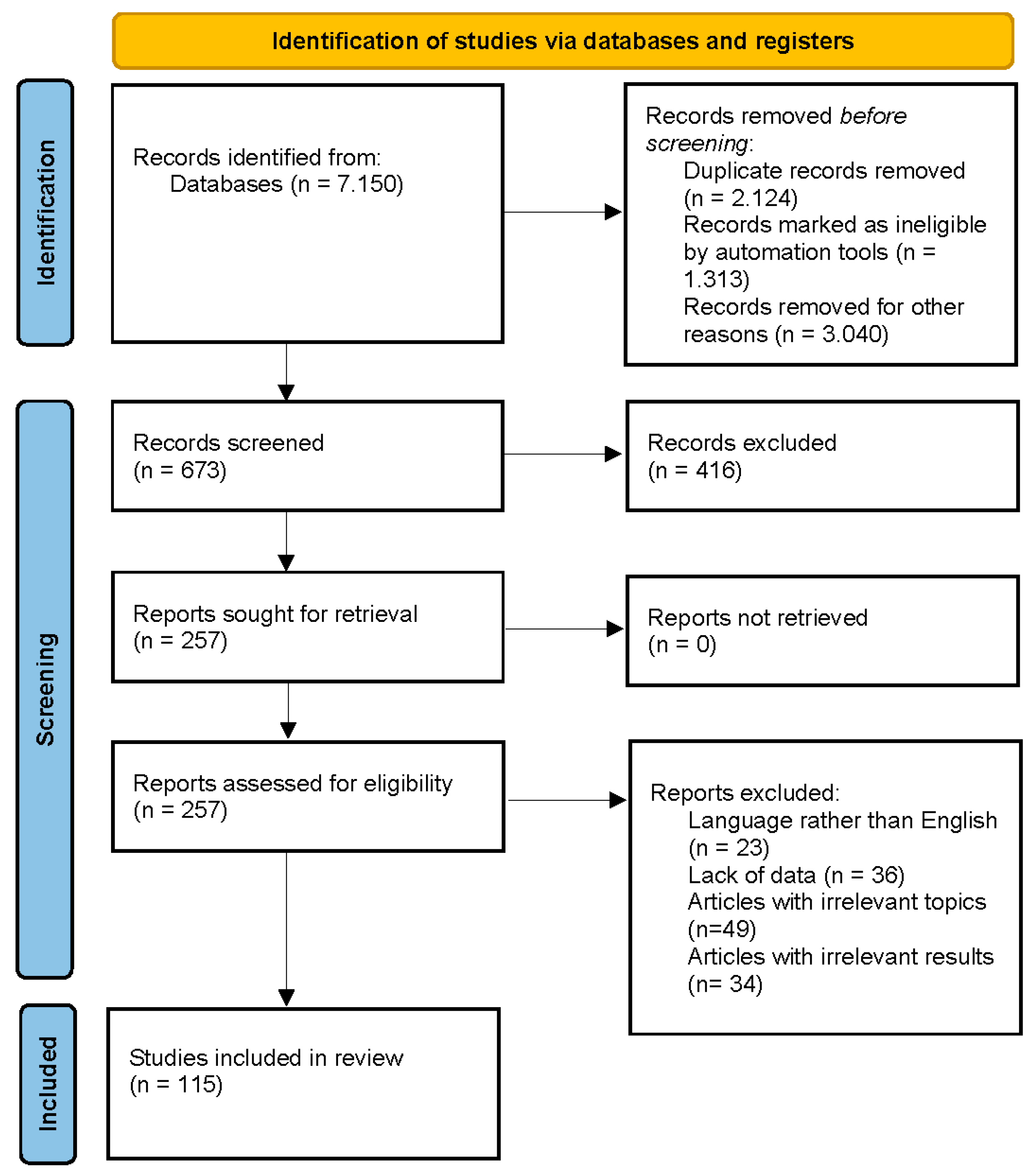
The search strategy was to investigate the biological activities of ecdysterone, turkesterone, R. carthamoides, S. oleracea, C. quinoa, and A. turkestanica. We screened original published research papers in databases such as Scopus, PubMed, Science Direct, and Google Scholar. We used a specific set of keywords and combinations to search the databases for research papers. These keywords and combinations were “ecdysterone”, “turkesterone”, “R. carthamoides”, “spinach”, “Spinacia oleacea”, “quinoa”, “Chenopodium quinoa”, “Ajuga turkestanica”, “biological activity”, and “biological effects”. We did not select a time frame for our search. The final step involved reading and identifying the selected articles. A total of 227 papers, of which 115 are about the biological activities of ecdysterone, turkesterone, and plants containing these PEs, were selected and included in the current review. This was carried out in accordance with the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) criteria shown in Figure 1.
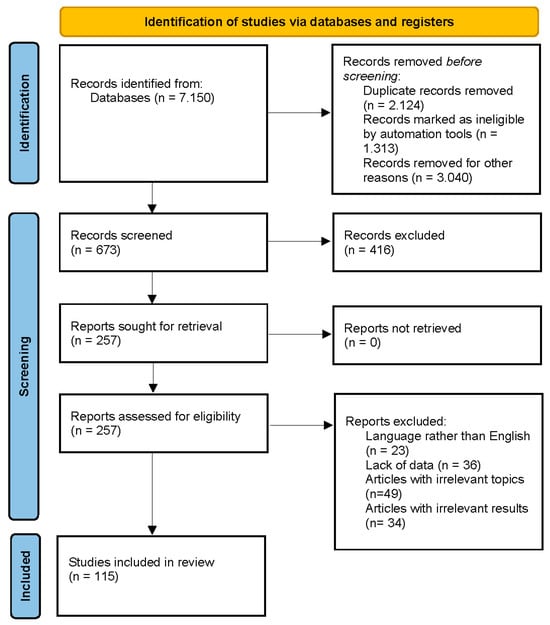
Figure 1.
PRISMA 2020 flow diagram [40].
3. Results and Discussion
3.1. 20-Hydroxyecdysterone
Among more than 520 known ecdysteroids, one of the most common is 20HE, with various biological activities and commercially available substances. Ecdysterone is a structurally characteristic ecdysteroid—2β,3β,14α,20β,22α,25β-Hexahydroxycholest-7-en-6-one (ecdysterone, β-ecdysone, 20-hydroxyecdysone) [5,41]. Important for the biological activity of ecdysteroids are the double bond at C-7, the keto group at C-6, and the hydroxyl groups at positions C-2, C-3, C-14, and C-22, and the hydroxyl group at C-20 correlates with anabolic activity [28].
The role of 20-HE as a defensive chemical in plants has been well-established [18]. Although R. carthamoides is considered as one of the main sources of the compound, many other plant species that are rich sources of 20HE are C. arachnoidea, Pfaffia (P. glomerata, P. iresinoides), and Serratula (S. centauroides, S. coronata, etc.) [31]. It has been reported that Serratula coronata L. juice and the roots of R. carthamoides contain roughly 1.5% 20HE. The C. arachnoidea roots contained up to 4–5% 20HE. Ecdysteroids are also found in substantial amounts in agricultural products, such as spinach, sugar beets, and saltbush seeds. Ecdysterone is used as an ingredient in dietary supplements in the field of sports nutrition [12,17,26,34].
The precise mechanism of action of 20HE remains unclear, but it is hypothesized that it exerts its effects by enhancing protein synthesis in skeletal muscles and the heart [42], boosting ATP synthesis in muscles [43], reducing hyperglycemia in diabetic animals [44], lowering plasma cholesterol levels [45], promoting the production of red blood cells, and decreasing the activity of triglyceride lipase [31].
In recent decades, significant research has been performed on the potential performance-enhancing benefits and therapeutic applications of ecdysterone [15,23,28]. The World Anti-Doping Agency (WADA) [33] agreed to add ecdysterone to their monitoring program in 2020, and the inquiry has continued ever since. Moreover, ecdysterone is widely thought to be nontoxic to mammals. According to Ogawa et al., the LD50 values for orally administrated 20HE in mice exceeded 9 g/kg, whereas intraperitoneally injected 20HE had an LD50 value of 7.8 g/kg [46]. Seidlova-Wuttke et al. conducted a study in which ovariectomized rats were fed dosages of up to 500 mg/kg daily for three months with no obvious adverse responses [47]. In general, ecdysteroids are considered safe for mammals [46]. Nowadays, numerous DSs containing ecdysteroids are available on the market. The focus of most of these products is sporting individuals [48].
Faster adaptation, the stimulation of protein synthesis [49], stress and anxiety reduction [50], antioxidant defense, the protection of joint cartilage [51], and neuroprotection [52] are some of the most important benefits that foods and plants rich in 20-hydroxyecdysterone might provide. Table 1 presents in vitro studies investigating the biological effects of 20HE.

Table 1.
20-hydroxyecdysone in vitro studies.
Table 1 provides a summary of studies on ecdysterone and various cellular processes and disorders. The data suggest that ecdysterone’s biological effects are associated with histamine release inhibition [54] and neuroprotective effects [55] and show promising therapeutic potential in Alzheimer's disease [55]. Ecdysterone possesses an anti-adipogenic effect, which may be important for future studies on ecdysterone in obesity treatment [69]. Moreover, the effects on cell lines are associated with endothelial dysfunction prevention, osteoporosis prevention, glucose regulation, cytotoxic effects, antibacterial properties, immunomodulatory effects, and anti-inflammatory activities [52,53,54,55,56,57,58,60,61,62,63,64,65,66,67,68,69,70,71,72,73]. Within these studies, no serious effects were reported [52,53,54,55,56,57,58,60,61,62,63,64,65,66,67,68,69,70,71,72,73]. 20-hydroxyecdysone has various pharmacological effects, demonstrating its potential as a therapeutic agent in numerous states, including allergy responses, neurological illnesses, cancer, and inflammatory disorders. However, in vitro studies are insufficient, and more in vivo studies are needed to determine the specific mechanisms of action and investigate their therapeutic uses. Table 2 presents in vivo studies and trials including 20HE.

Table 2.
20-hydroxyecdysterone in vivo studies and trials.
The in vivo studies presented in Table 2, suggest that ecdysterone possesses effects on osteoblast differentiation and bone regeneration, joint morphology and osteoporosis, Alzheimer’s disease, and lipid metabolism, as well as having anti-obesity, anti-diabetic, and neuroprotective effects [47,49,50,51,74,75,76,78,79,80,81,82,83,84,85,86,87,88,89,90,91,92,93,94,95,96,97,98,99,100,101]. Some of the in vivo studies confirm the in vitro studies about anti-obesity, anti-diabetic, neuroprotective, and cytotoxic effects, as well as the prevention of Alzheimer’s disease. A controlled randomized study investigated the utilization of 20HE in metabolic syndrome [99]. Ecdysterone has great potential for use in medications intended to cure a variety of illnesses. Furthermore, ecdysterone does not appear to possess any severe side effects [19,47,49,50,51,74,75,76,77,78,79,80,81,82,83,84,85,86,87,88,89,90,91,92,93,94,95,96,97,98,99,100,101]. These suggest that ecdysterone supplementation is safe. In a study involving 20-hydroxyecdysone in the dietary supplement “Peak Ecdysone”, conducted on 46 men over a 10-week period, it was discovered that 20HE supplementation resulted in increases in body weight and muscle mass, as well as improvements in power and strength performance, without adverse effects or changes to the steroid profile [26]. It is considered that lower doses showed no significant effects but reported that amounts more than 5 μg/kg body weight were considered effective [102]. This shows that ecdysterone administration may be beneficial for improving athletic performance without compromising health.
We found multicenter randomized double-blind studies on ecdysterone and ecdysterone-rich extracts. Further research is needed to understand the mechanisms of action and possible long-term consequences of these supplements. To fully assess the benefits and possible future applications of ecdysterone, multicenter randomized double-blind trials are required.
3.2. Rhaponticum carthamoides
Rhaponticum carthamoides (Willd.) Iljin is a perennial herb of the Asteraceae family that is also known as maral root or Russian leuzea. It grows in the harsh conditions of South Siberia’s Altai and Saian mountains. It is a semi-rosulate plant that may grow to be 150 cm tall [13,103,104]. The use of R. carthamoides for medicinal purposes dates back to ancient times, and traditional Siberian medicine has long praised the plant for its ability to treat weariness and debility after sickness [13,103]. In the history of Russian scientific investigation, R. carthamoides has received a lot of attention in the domain of physical performance improvement. Research over the last century has shown its muscle- and strength-building capabilities, resulting in widespread use among elite athletes in Soviet and Russian sports [13]. In 1969, Brekhman and Dardymov classified R. carthamoides as an adaptogen, now widely used in herbal medicine to promote resistance to stress, such as trauma, anxiety, and fatigue [13,103,105].
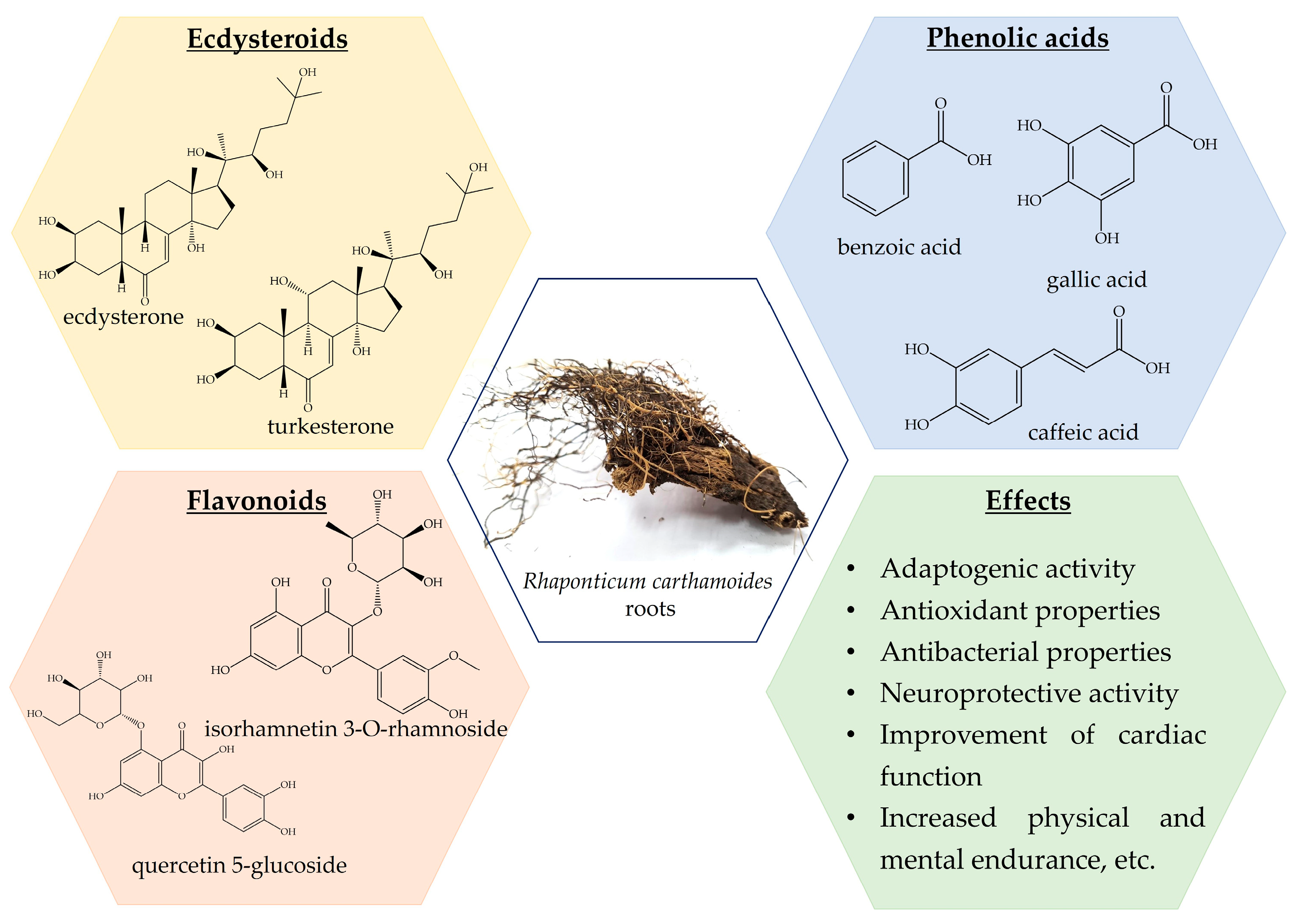
Previously, a wide range of chemical classes was found in R. carthamoides roots, with steroids, particularly ecdysteroids, flavonoids, lignans, and phenolic compounds (Figure 2) [13,106,107,108,109,110,111,112,113]. Moreover, R. carthamoides roots are a source of essential oil, which is characterized by antimicrobial, antioxidant, and anti-inflammatory effects [13,107,114,115,116,117]. Fifty different ecdysteroid chemicals have been found in the plant’s roots, aerial parts, and seeds [13,113]. An examination into the PE composition of R. carthamoides revealed the extraction of 20HE, inokosterone, leuzeasterone, polypodine B, rhapisterone, makisterone, carthamoleusterone, turkesteron, and their derivates from its underground parts [13,115,118]. The concentration of 20HE (β-ecdysone, ecdysterone, and polypodine A) varies between 0.049% and 1.74% [107,112,119].
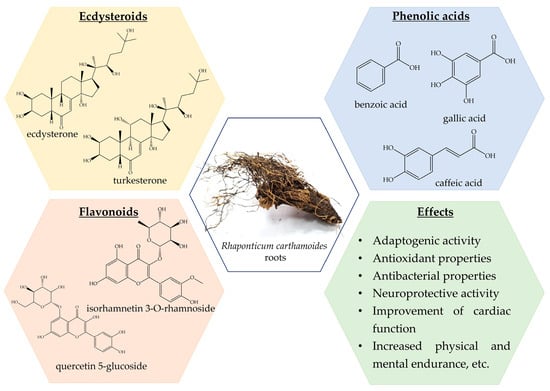
Figure 2.
Rhaponticum carthamoides phytochemical composition and effects.
Extracts from its roots and rhizomes are currently employed in a wide range of DSs and nutraceutical formulations. They are used to increase muscle growth, alleviate impotence, and reduce physical and mental fatigue [13]. Furthermore, R. carthamoides and its derivatives are used in cosmetics and herbal teas [13,106]. Rhaponticum carthamoides gained serious popularity in the last decade, especially after the introduction of numerous DSs containing leuzea extracts [120].
One of the most important benefits of leuzea supplementation is the potential to increase the working capacity of the skeletal muscles [42]. However, for the athletes, the adaptogenic activity of leuzea seems to be of the greatest importance [103]. Table 3 summarizes a wide range of in vivo investigations on the pharmacological activities of R. carthamoides.

Table 3.
Rhaponticum carthamoides in vitro studies.
Extracts from R. carthamoides show a variety of activities, including antibacterial, cytotoxic, anti-adipogenic, and antioxidant [69,121,122,123,124,125,126,127,128,129,130,131,132]. The investigations into R. carthamoides revealed that its extracts are low in toxicity [69,121,122,123,124,125,126,127,128,129,130,131,132]. In vitro research on cell cultures may not give comprehensive answers, but they serve as a basis for future in vivo investigations. Table 4 summarizes the findings from in vivo research and trials with R. carthamoides extracts.

Table 4.
Rhaponticum carthamoides in vivo studies and trials.
The diverse range of bioactivities of R. carthamoides are presented in Table 4. These investigations include a wide range of activities, such as cognitive function effects, metabolic regulation, stress management, and cytotoxicity [44,133,134,135,136,137,138,139,140,141,142]. Mosharrof et al. investigated the effect of R. carthamoides extract on learning and memory processes in rats [133]. Petkov et al. found that an aqueous-alcoholic extract of R. carthamoides exhibited significant stimulating effects on the central nervous system, including improved learning and memory ability in rats [134]. Dushkin et al. investigated the therapeutic potential of R. carthamoides extract, as well as extracts from Glycyrrhiza glabra and Punica granatum. They observed that treatment with R. carthamoides extract significantly improved glucose and lipid metabolism compared to the other extracts [135]. Furthermore, studies on the combination administration of R. carthamoides and Rhodiola rosea on resistance exercise, the combination of these extracts boosted muscle protein synthesis and average power performance in rats [136]. These findings provide valuable insights into R. carthamoides’ therapeutic potential for enhancing overall health and well-being. There are no observable indications or symptoms of toxicity, indicating a wide margin of safety [44,133,134,135,136,137,138,139,140,141,142]. Contrary to the randomized trial, which reported improved physical performance after the intake of 20HE, the investigation into the effects of R. carthamoides and R. rosea extracts suggests no potential effects on physical performance and fatigue [26,142]. These made R. carthamoides a valuable resource in sports nutrition. Further research is needed to understand the mechanisms of action and possible long-term consequences of these supplements. Moreover, the multicenter randomized controlled trials are limited.
3.3. Spinacia oleracea
Spinach (Spinacia oleracea L.) is an annual plant belonging to the family Chenopodiaceae; it can be divided into two subspecies, ssp. glabra and ssp. spinosa [143,144]. It originated in central Asia, specifically Persia, and its use dates back to ancient times [145,146].
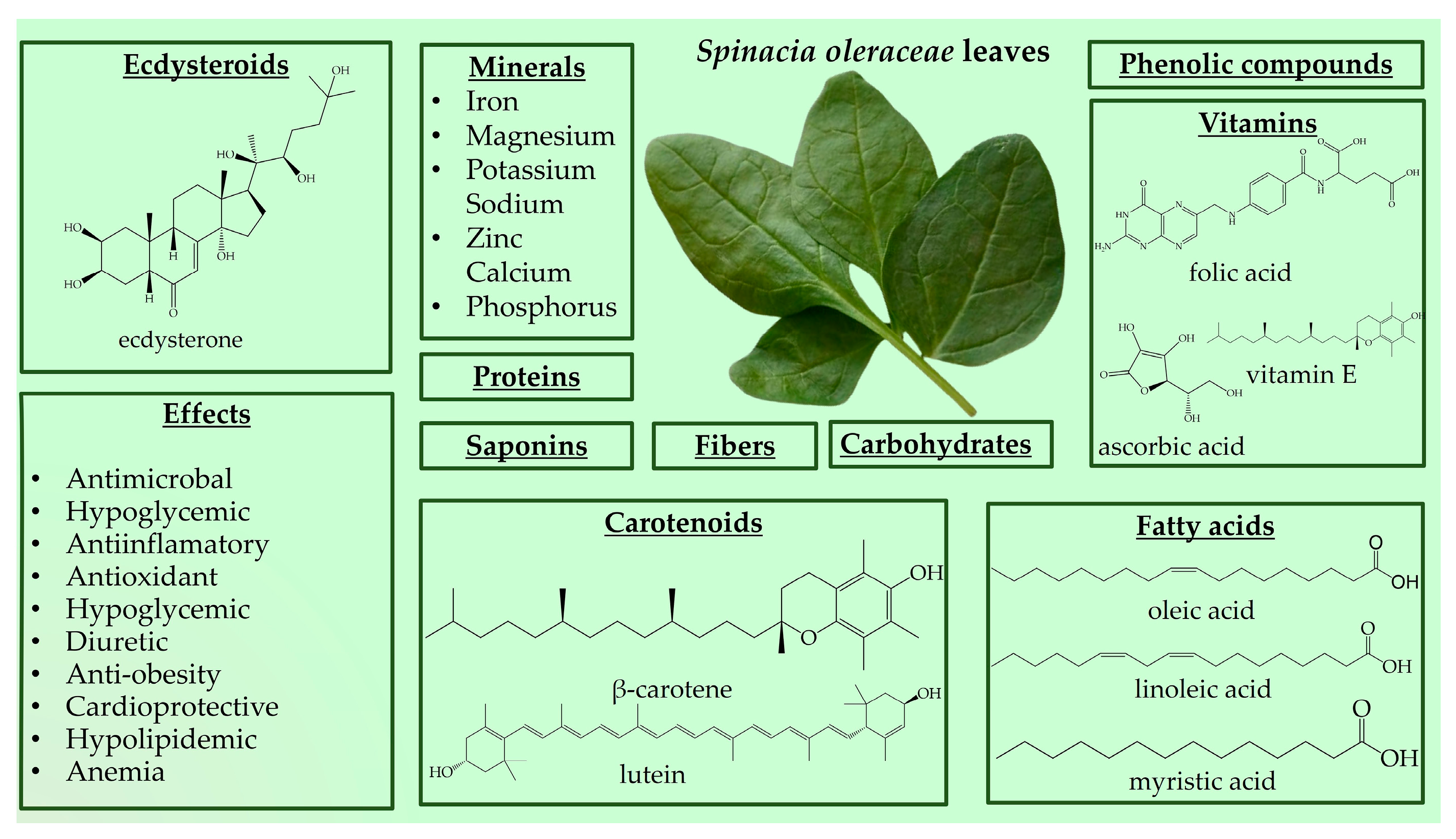
Spinach is a great source of nutrition and phytochemical constituents [143,144,146,147]. The nutrient composition can be divided into six major components, including carbohydrates (approximately 50%), proteins (approximately 14%), fats (approximately 23%), fiber, minerals, and vitamins (Figure 3) [146,148,149,150,151,152]. The high iron content makes it a valuable food for anemia [143]. A disadvantage of spinach is its high nitrate content, which may cause methemoglobinemia [143].
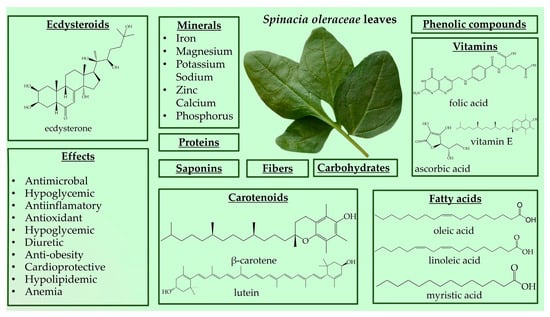
Figure 3.
Spinacia oleracea nutrient profile and biological effects.
Spinach is source of beneficial phytonutrient constituents, such as phenolic compounds (flavonoids, phenolic acids, stilbenes, and lignans) and carotenoids (lutein, zeaxanthin, and β-carotene) [146,149,153]. Spinach also contains steroids, terpenes, tannins, and cardenolides [154]. Moreover, Spinacia oleracea produces large amounts of PEs, with the major component being 20HE [155,156]. Spinach has stimulated ecdysteroid accumulation in response to mechanical or insect injury, and PEs are metabolically stable in this plant species [11,157]. The reported content of 20HE in spinach leaves ranges from 17.1 to 885 µg/g [158,159]. However, variations exist regarding the levels of 20HE in spinach, with some reporting lower amounts, such as 50 μg/g dry mass, 10.3 and 16.8 μg/g in different spinach accessions, and 0.44 mg% dry weight [8,156,160]. Grucza et al. reported even lower levels of 20HE content in fresh spinach leaves at 10 µg/100 g [161]. These discrepancies in 20HE and phytochemical content may be attributed to different processing methods applied to spinach leaves [149].
Spinach is a versatile plant that may be consumed fresh in salads and smoothies or cooked in recipes such as steamed vegetables, casseroles, and soups [143,144,146,147,149]. Due to the wide variety of bioactive and phytochemical compounds, S. oleracea has a wide variety of potential functionalities, mainly antioxidant, antimicrobial, anticancer, anti-obesity, hypoglycemic, and hypolipidemic [146,149,151]. Spinach leaves are utilized for their, emollient, wholesome, antipyretic, diuretic, laxative, and anthelmintic substances, as well as their anti-inflammatory effects and joint pain relief (Figure 3) [151]. Spinach, as a significant antioxidant and nitric oxide donor, is a valuable vegetable in athletes’ diets [162]. In recent years, the consumption of spinach has increased due to consumers’ concerns about healthy eating [144]. Spinach’s traditional claim to increase muscle strength may have scientific support due to its ecdysteroid concentration. Because of its anabolic qualities, ecdysterone has gained popularity as a natural sports performance booster. As a result, ecdysterone-containing dietary supplements made from spinach and other plant extracts have become increasingly popular. Athletes are advised to take up to 1000 mg of ecdysterone daily; however, even high daily consumption of spinach (1 kg) rarely surpasses 100 mg. Studies on humans that have been given supplements containing ecdysterone have, over time, demonstrated increases in physical strength and muscular mass [34,163]. In Table 5 are presents in vivo studies on the biological activities of S. oleracea.

Table 5.
Spinacia oleracea in vivo studies.
Table 5 summarizes investigations into potential health benefits connected with S. oleracea. Gorelick-Feldman et al. investigated the effects of PEs and extracts from A. turkestanica and S. oleracea on protein synthesis and physical performance [19]. Panda et. al. investigated whether spinach extract may reduce hyperlipidemia by reducing pancreatic lipase activity. This suggests the importance of consuming spinach in managing lipid levels and obesity [164]. The spinach extract demonstrated promising appetite suppression effect which make it potential nutrient in appetite regulation [165].
These results show that spinach is associated with numerous health advantages, such as antioxidant characteristics, lipid-lowering effects, appetite regulation, tissue regeneration, and protection against oxidative stress-induced damage [156,162,164,165,166,167,168,169,170]. These highlight spinach’s potential as a useful nutrient. The data from in vivo experiments with animals are insufficient. Table 6 presents controlled trials involving S. oleracea.

Table 6.
Spinacia oleracea randomized controlled trials.
The table presents randomized controlled trials investigating the effects of spinach and its derivatives. In a study on daily supplementation with S. oleracea extract, combined with moderate-intensity exercise, in over 50-year-old individuals, Perez-Pinero et. al. reported that spinach extract increases muscle-related factors and muscle quality [171]. Due to these results, spinach can be included in the nutritional diets of athletes, combined with physical exercises. Bohlooli et. al. reported that in well-trained men, daily oral supplementation with spinach reduced indicators of oxidative stress and muscle injury after training [162]. In a randomized control trial, Maruyama et. al. reported that the consumption of spinach in obese and insulin-resistant patients supplies antioxidants and improves lipid profiles in patients [172]. Also, a potential therapeutic application of spinach-derived thylakoid supplementation might be obesity management [174]. Moreover, the consumption of spinach extract significantly reduced hunger and increased postprandial plasma glucose concentrations, indicating a potential role in appetite regulation [175]. The findings from these studies collectively underscore the diverse health-promoting effects of spinach and its derivatives, ranging from muscle health and oxidative stress reduction to metabolic regulation [162,171,172,173,174,175,176,177,178]. Furthermore, the consumption of spinach is not associated with adverse effects [162,171,172,173,174,175,176,177,178]. These effects and the safety profile of spinach could correspond with its use in sports nutrition. Further double-blind multicenter randomized controlled trials are required to confirm the good therapeutic profile of spinach.
3.4. Chenopodium quinoa
Quinoa (Chenopodium quinoa Willd., Chenopodiaceae) is an annual herbaceous plant that is classified as a gluten-free pseudo-cereal [179,180]. Quinoa originated in the Andes of South America, specifically Peru, Bolivia, Ecuador, Colombia, and Chile [179,181]. The consumption of quinoa dates back 5000 years [182]. It grows to a height of 3–7 feet, the woody stem can be branched or unbranched, and it comes in different colors (green, red, and purple) [179,181]. Quinoa seeds are small, spherical, and flat, with colors that range from white to grey, black, yellow, and red [182,183].
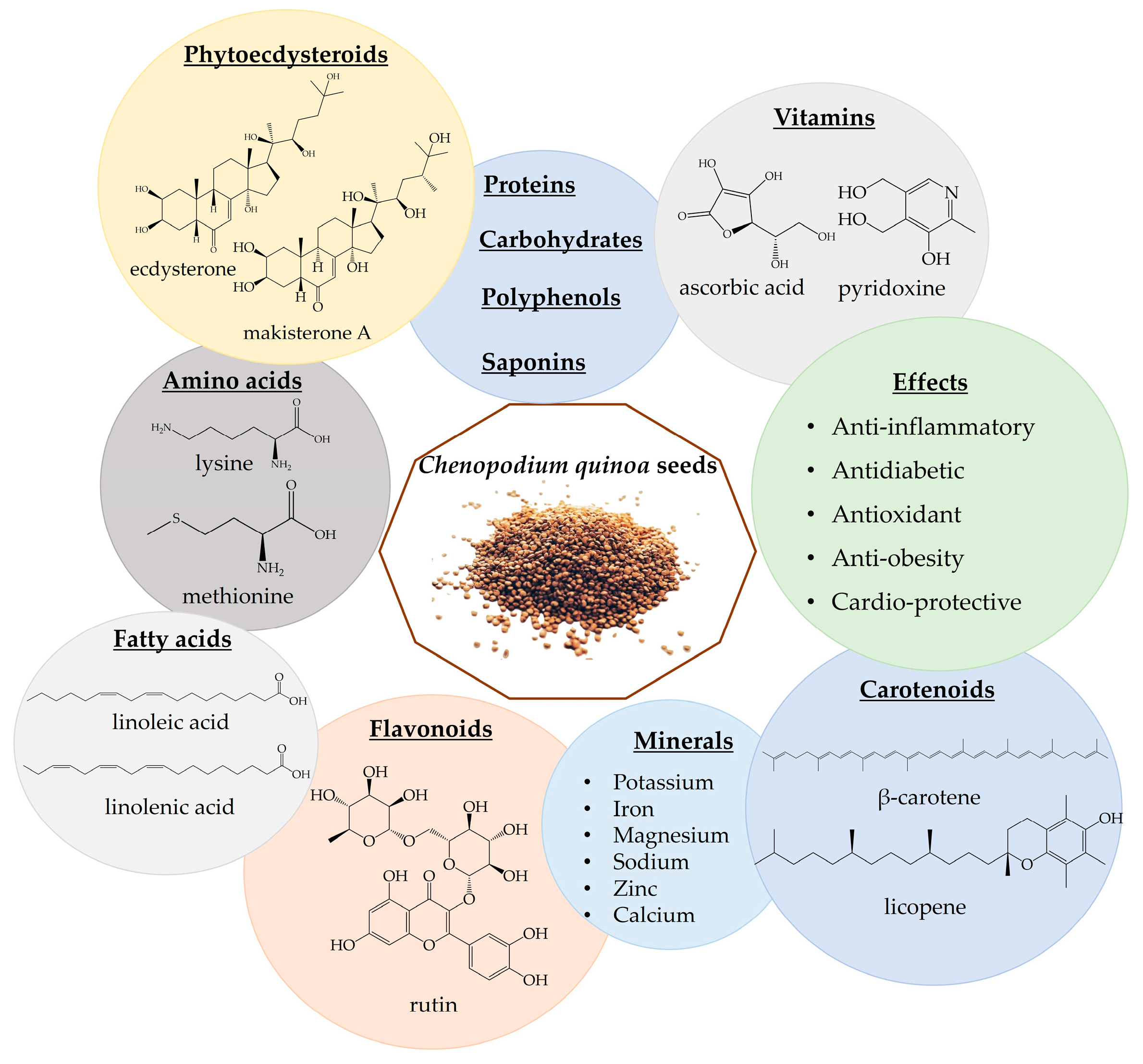
The quinoa grain is characterized by high protein (13–15%), fiber (3–4%), carbohydrates (59–61%), fatty acids, vitamins, amino acids, and minerals [35,184,185,186,187,188]. Quinoa also contains a broad spectrum of polyphenols, carotenoids, phytoecdysteroids, phytosterols, saponins, and tannins [185,186]. There are about 36 different types of hytoecdysteroids detected in quinoa seeds, with high amounts of 20-hydroxyecdysone (up to 90% of PE content) and lower levels of makisterone A, 24(28)-dehydromakisterone A, 24-epi-makisterone A and polypodine B (Figure 4). It also contains PE derivates, such as 25,27-dehydroinokosterone, 24,25-dehydroinokosterone, and 5β-hydroxy-24(28)-dehydromakisterone A [12,35,189,190,191,192,193,194]. It is considered that both quinoa and spinach are poor sources of ecdysterone [36], compared to R. carthamooides and A. turkestanica [5,12,13]. However, quinoa seeds contain 4–12 times more 20HE by dry weight than spinach leaves [19,189,195].
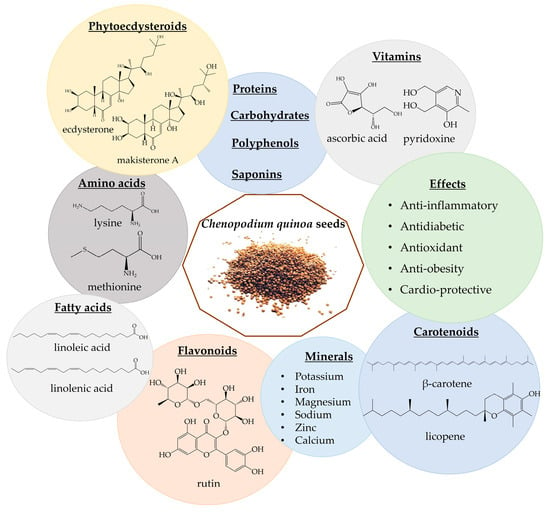
Figure 4.
Chenopodium quinoa phytochemicals and biological effects.
Quinoa seeds have been utilized as flour, added to soups, and incorporated into bread recipes. The rise of new food products featuring ancient grains, including quinoa, is observed globally, offering new opportunities for these nutritious grains in the market [196,197]. Quinoa seeds are considered a “functional food”; it is considered that the consumption of 50 g of quinoa for 6 weeks is safe [198,199]. Apart from being rich in nutrients, quinoa also exhibits health-promoting properties, including anti-inflammatory, antidiabetic, antioxidant, anti-obesity, and cardio-beneficial effects (Figure 4) [35,191,192,193]. Moreover, not only the presence of phytoecdysteroids but also the presence of amino acids could increase muscle performance and lead to an increase in lean mass [200]. Table 7 presents in vitro studies involving quinoa.

Table 7.
Chenopodium quinoa in vitro studies.
The in vitro studies presented in Table 7 reported quinoa’s effects on gut microbiota and cancer cells, antioxidant and antidiabetic properties, platelet activity, and modulating collagenase activity [201,202,203,204,205,206,207]. Quinoa’s polysaccharides enhanced the synthesis of short-chain fatty acids and the composition of the microbiota. They might function as prebiotics [201]. Gawlik-Dziki et al. reported that quinoa possesses possible chemopreventive and anticarcinogenic qualities [202]. Furthermore, quinoa’s potential as a rich source of antioxidants is highlighted by the study by Alvarez-Jubete et. al. examining the polyphenol content and antioxidant qualities of methanolic extracts from the grain [203]. Overall, these studies highlight quinoa’s broad bioactive qualities and promise as a functional food. More studies, including in vivo, are required to explain the mechanism of action and the potential use of quinoa in DSs. Table 8 presents in vivo studies involving quinoa.

Table 8.
Chenopodium quinoa in vivo studies.
The studies presented in the table correspond with the potential effects of quinoa extract. Foucault et. al. conducted a study on the ability of quinoa extract enriched in 20HE to prevent obesity, and it was observed that quinoa and 20HE demonstrate anti-obesity activity in rats [208]. Moreover, Foucault et. al. reported that quinoa extract and 20HE regulated glucose and lipid metabolism [211]. Sidorova et. al. revealed that quinoa effectively normalized oxidative stress markers and improved cognitive function [209]. Meneguetti et al. reported that hydrolyzed quinoa decreased body weight, blood triacylglycerol level, and fat deposition [200]. Supplementation with quinoa and chia seed extracts may regulate metabolic indicators and showed hepatoprotective, anti-inflammatory, and antioxidant activities in rats [212]. Quinoa extracts showed anti-obesity, antioxidant, hypoglycemic, immunoregulatory, and collagenase-modulating effects in in vivo experiments with rats [208,209,210,211,212,213,214]. No serious adverse effects were reported within the experiments, which correlated with the safety profile of quinoa extracts and their safe nutrition in sports. No double-blind multicenter randomized controlled trials were found, so further trials are recommended to be conducted.
3.5. Turkesterone and Ajuga turkestanica
Turkesterone is a PE, with 27 carbon atoms and seven OH-groups; it is considered that the OH groups at C-20 and C-11 are responsible for the anabolic effects [28,215]. Turkesterone is considered to possess an anabolic effect, and it is used as DS by athletes, instead of anabolic steroids [28,30]. It is found in some endemic plants, such as A. turkestanica, R. carthamoides, Triticum aestivum L., Vitex scabra, etc. [13,69,216,217,218]. The highest content of turkesterone is considered to be in Ajuga turkestanica [19]. Moreover, in DSs, turkesterone is derived from A. turkestanica [28].
Ecdysteroid-enriched preparations of A. turkestanica (or pure turkesterone) exhibit anabolic effects [216,219,220].
Ajuga turkestanica, a plant species of the Lamiaceae family, grows wild in the Boysun mountainous region of Central Asia, Uzbekistan. It is a perennial plant, that normally grows on clay, petrous, and rubbly slopes and rocks, with a height of 40–60 cm [14]. Both the aerial parts and root sections are utilized in folk medicine to prevent obesity, hair loss, and gastrointestinal diseases [14,216].
The phytochemical profile of A. turkestanica has indicated the existence of several biologically active secondary metabolites, such as carbohydrates, iridoids, diterpenes, phytoecdysteroids, flavonoids, sterol glycosides, and phenylethanoid glycosides [14,53,216]. The A. turkestanica extract contained 2.1% (w/w) turkesterone and 0.9% (w/w) 20HE [19]. Other isolated Pes are cyasterone, ajugasterone B, α-ecdysone, ecdysone 2,3-monoacetonide, and 22-acetylcyasterone [59,221,222].
Extracts from A. turkestanica are associated with wound-healing effects [223], as well as antiproliferative, anti-stress and immunostimulating, antimicrobial, and antioxidant effects [12,59]. Most of these effects are likely due to the presence of Pes [224]. Table 9 presents studies about the biological effects of turkesterone. Ajuga turkestanica and turkesterone products are commercially available not only online but also in bodybuilding centers [12]. Table 9 presents studies on the biological effects of turkesterone, while Table 10 presents studies on A. turkestanica.

Table 9.
Turkesterone studies.

Table 10.
Ajuga turkestanica studies.
The studies presented in the tables reported the various effects of turkesterone and A. turkestanica. Turkesterone is associated with a reduction in lipid accumulation in human adipocytes [69]. In a stress-induced mouse model, ecdysterone and turkesterone were found to prevent stress-related consequences and restore immunological activity [138]. Additionally, turkesterone exhibited beneficial effects on the endocrine and exocrine function in alloxan-induced diabetic rats, suggesting its potential therapeutic application in diabetes management [225]. Furthermore, PEs from A. turkestanica demonstrate hypoglycemic activity in models of hyperglycemia and diabetes, suggesting their potential therapeutic use in managing blood glucose levels [227]. Turkesterone presents a safer option to conventional anabolic steroids, displaying significant anabolic effects without inducing androgenic side effects in in vivo animal studies [19,219]. Moreover, A. turkestanica also enhances muscle regeneration and maintenance [29,226]. These results suggest that turkesterone extracted from A. turkestanica possesses diverse physiological effects, including potential performance enhancement and antiadipogenic, immunomodulatory, and hypoglycemic activities, making it a promising candidate for sports nutrition. Further research is needed to fully understand the mechanisms of action and therapeutic potential of turkesterone and A. turkestanica [19,29,59,69,77,138,219,220,225,226,227].
4. Conclusions
Phytoecdysteroids, widely distributed across various plant species, have been valued for their adaptogenic properties and potential to enhance muscle strength and performance. 20-hydroxyecdysone and turkesterone are two of the most noteworthy PEs, which are found in plants such as R. carthamoides, S. oleracea, C. quinoa, and A. turkestanica. Nowadays, these plants are used not just in plant-based diets for their high nutritional value but also as major components of DSs. Since 2020, ecdysterone has garnered attention and has been included in the WADA’s monitoring program due to its ability to enhance physical endurance. Studies on the benefits of 20HE, turkesterone, and extracts from these plants are limited. The preparations from these plants show promise not only as nutritional supplements but also as therapeutic agents for treating medicinal disorders, such as chronic tiredness, heart disorders, and neurodegenerative diseases. Future double-blind randomized multicenter trials are critical for fully assessing the effectiveness and safety of these preparations for cardiovascular disease, chronic tiredness, obesity, and osteoporosis. These studies will provide valuable insights into the therapeutic potential of phytoecdysteroid-rich extracts.
Author Contributions
Conceptualization, V.T., S.I. and K.I.; methodology, V.T. and S.I.; software, V.T.; validation, V.T., S.I. and K.I.; formal analysis, V.T.; investigation, V.T., S.I. and K.K.; resources, V.T. and S.I.; data curation, V.T. and S.I.; writing—original draft preparation, V.T., S.I. and K.K.; writing—review and editing, S.I., D.C. and K.I.; visualization, V.T., S.I. and K.I.; supervision, S.I., D.C. and K.I. All authors have read and agreed to the published version of the manuscript.
Funding
This research received no external funding.
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
Not applicable.
Conflicts of Interest
The authors declare no conflicts of interest.
References
- Arif, Y.; Singh, P.; Bajguz, A.; Hayat, S. Phytoecdysteroids: Distribution, Structural Diversity, Biosynthesis, Activity, and Crosstalk with Phytohormones. Int. J. Mol. Sci. 2022, 23, 8664. [Google Scholar] [CrossRef]
- Lafont, R.; Balducci, C.; Dinan, L. Ecdysteroids. Encyclopedia 2021, 1, 1267–1302. [Google Scholar] [CrossRef]
- Huber, R.; Hoppe, W. Zur Chemie des Ecdysons, VII: Die Kristall-und Molekülstrukturanalyse des Insektenverpuppungshormons Ecdyson mit der automatisierten Faltmolekülmethode. Chem. Berichte 1965, 98, 2403–2424. [Google Scholar] [CrossRef]
- Butenandt, A.; Karlson, P. Über die Isolierung eines Metamorphose-Hormons der Insekten in kristallisierter Form. Z. Für Naturforschung 1954, 9, 389–391. [Google Scholar] [CrossRef]
- Ecdybase. The Ecdysone Handbook—A Free Online Ecdysteroids Database. Available online: https://ecdybase.org/ (accessed on 3 May 2023).
- Das, N.; Mishra, S.K.; Bishayee, A.; Ali, E.S.; Bishayee, A. The Phytochemical, Biological, and Medicinal Attributes of Phytoecdysteroids: An Updated Review. Acta Pharm. Sin. B 2021, 11, 1740–1766. [Google Scholar] [CrossRef]
- Nakanishi, K. The Ecdysones. Pure Appl. Chem. 1971, 25, 167–196. [Google Scholar] [CrossRef]
- Dinan, L. Phytoecdysteroids: Biological Aspects. Phytochemistry 2001, 57, 325–339. [Google Scholar] [CrossRef]
- Kubo, I.; Klocke, J.A.; Asano, S. Effects of Ingested Phytoecdysteroids on the Growth and Development of Two Lepidopterous Larvae. J. Insect Physiol. 1983, 29, 307–316. [Google Scholar] [CrossRef]
- Robbins, W.E.; Kaplanis, J.N.; Thompson, M.J.; Shortino, T.J.; Joyner, S.C. Ecdysones and Synthetic Analogs: Molting Hormone Activity and Inhibitive Effects on Insect Growth, Metamorphosis and Reproduction. Steroids 1970, 16, 105–125. [Google Scholar] [CrossRef]
- Dinan, L. Ecdysteroid Structure-Activity Relationships. In Studies in Natural Products Chemistry; Atta-ur-Rahman, Ed.; Bioactive Natural Products (Part J); Elsevier: Amsterdam, The Netherlands, 2003; Volume 29, pp. 3–71. [Google Scholar]
- Savchenko, R.G.; Veskina, N.A.; Odinokov, V.N.; Benkovskaya, G.V.; Parfenova, L.V. Ecdysteroids: Isolation, Chemical Transformations, and Biological Activity. Phytochem. Rev. 2022, 21, 1445–1486. [Google Scholar] [CrossRef]
- Kokoska, L.; Janovska, D. Chemistry and Pharmacology of Rhaponticum carthamoides: A Review. Phytochemistry 2009, 70, 842–855. [Google Scholar] [CrossRef] [PubMed]
- Mamarasulov, B.; Davranov, K.; Jabborova, D. Phytochemical, Pharmacological and Biological Properties of Ajuga turkestanica (Rgl.) Brig (Lamiaceae). Ann. Phytomed. Int. J. 2020, 9, 44–57. [Google Scholar] [CrossRef]
- Todorova, V.; Ivanov, K.; Delattre, C.; Nalbantova, V.; Karcheva-Bahchevanska, D.; Ivanova, S. Plant Adaptogens—History and Future Perspectives. Nutrients 2021, 13, 2861. [Google Scholar] [CrossRef]
- Bajguz, A.; Bąkała, I.; Talarek, M. Chapter 5—Ecdysteroids in Plants and Their Pharmacological Effects in Vertebrates and Humans. In Studies in Natural Products Chemistry; Atta-ur-Rahman, Ed.; Elsevier: Amsterdam, The Netherlands, 2015; Volume 45, pp. 121–145. [Google Scholar]
- Odinokov, V.N.; Galyautdinov, I.V.; Nedopekin, D.V.; Khalilov, L.M.; Shashkov, A.S.; Kachala, V.V.; Dinan, L.; Lafont, R. Phytoecdysteroids from the Juice of Serratula coronata L. (Asteraceae). Insect Biochem. Mol. Biol. 2002, 32, 161–165. [Google Scholar] [CrossRef] [PubMed]
- Dinan, L.; Lafont, R. Chapter Two—Ecdysteroids as Defensive Chemicals. In Advances in Insect Physiology; Jurenka, R., Ed.; Academic Press: Cambridge, MA, USA, 2022; Volume 63, pp. 107–154. [Google Scholar]
- Gorelick-Feldman, J.; MacLean, D.; Ilic, N.; Poulev, A.; Lila, M.A.; Cheng, D.; Raskin, I. Phytoecdysteroids Increase Protein Synthesis in Skeletal Muscle Cells. J. Agric. Food Chem. 2008, 56, 3532–3537. [Google Scholar] [CrossRef]
- Todorova, M.N.; Savova, M.S.; Mihaylova, L.V.; Georgiev, M.I. Icariin Improves Stress Resistance and Extends Lifespan in Caenorhabditis Elegans through Hsf-1 and Daf-2-Driven Hormesis. Int. J. Mol. Sci. 2023, 25, 352. [Google Scholar] [CrossRef] [PubMed]
- Savova, M.S.; Todorova, M.N.; Apostolov, A.G.; Yahubyan, G.T.; Georgiev, M.I. Betulinic Acid Counteracts the Lipid Accumulation in Caenorhabditis Elegans by Modulation of Nhr-49 Expression. Biomed. Pharmacother. 2022, 156, 113862. [Google Scholar] [CrossRef]
- Ivanova, S.; Delattre, C.; Karcheva-Bahchevanska, D.; Benbasat, N.; Nalbantova, V.; Ivanov, K. Plant-Based Diet as a Strategy for Weight Control. Foods 2021, 10, 3052. [Google Scholar] [CrossRef]
- Kraiem, S.; Al-Jaber, M.Y.; Al-Mohammed, H.; Al-Menhali, A.S.; Al-Thani, N.; Helaleh, M.; Samsam, W.; Touil, S.; Beotra, A.; Georgakopoulas, C.; et al. Analytical Strategy for the Detection of Ecdysterone and Its Metabolites in vivo in uPA(+/+)-SCID Mice with Humanized Liver, Human Urine Samples, and Estimation of Prevalence of Its Use in Anti-doping Samples. Drug Test. Anal. 2021, 13, 1341–1353. [Google Scholar] [CrossRef]
- Dwyer, J.T.; Allison, D.B.; Coates, P.M. Dietary Supplements in Weight Reduction. J. Am. Diet. Assoc. 2005, 105, 80–86. [Google Scholar] [CrossRef]
- Staynova, R.; Yanachkova, V. Weight Management Strategies and Food Supplement Intake among Bulgarian Adults: Results of a National Survey. Pharmacia 2023, 70, 1119–1126. [Google Scholar] [CrossRef]
- Isenmann, E.; Ambrosio, G.; Joseph, J.F.; Mazzarino, M.; de la Torre, X.; Zimmer, P.; Kazlauskas, R.; Goebel, C.; Botrè, F.; Diel, P.; et al. Ecdysteroids as Non-Conventional Anabolic Agent: Performance Enhancement by Ecdysterone Supplementation in Humans. Arch. Toxicol. 2019, 93, 1807–1816. [Google Scholar] [CrossRef]
- Ambrosio, G.; Wirth, D.; Joseph, J.F.; Mazzarino, M.; de la Torre, X.; Botrè, F.; Parr, M.K. How Reliable Is Dietary Supplement Labelling? Experiences from the Analysis of Ecdysterone Supplements. J. Pharm. Biomed. Anal. 2020, 177, 112877. [Google Scholar] [CrossRef]
- Todorova, V.; Ivanov, K.; Karcheva-Bahchevanska, D.; Ivanova, S. Development and Validation of High-Performance Liquid Chromatography for Identification and Quantification of Phytoecdysteroids Ecdysterone and Turkesterone in Dietary Supplements. Processes 2023, 11, 1786. [Google Scholar] [CrossRef]
- Zubeldia, J.M.; Hernández-Santana, A.; Jiménez-del-Rio, M.; Pérez-López, V.; Pérez-Machín, R.; García-Castellano, J.M. In Vitro Characterization of the Efficacy and Safety Profile of a Proprietary Ajuga turkestanica Extract. Chin. Med. 2012, 2012, 26159. [Google Scholar] [CrossRef]
- Lee, J.H.; Han, J.H.; Ham, H.J.; Kim, H.; Lee, J.; Baek, S.Y. Development of a Method for Simultaneous Screening of Four Natural-Derived Steroids and Their Analogues Used as Dietary Supplements via Liquid Chromatography-Quadrupole-Time of Flight Mass Spectrometry and Liquid Chromatography-Tandem Mass Spectrometry. Food Addit. Contam. Part A 2022, 39, 829–837. [Google Scholar] [CrossRef]
- Dinan, L.; Dioh, W.; Veillet, S.; Lafont, R. 20-Hydroxyecdysone, from Plant Extracts to Clinical Use: Therapeutic Potential for the Treatment of Neuromuscular, Cardio-Metabolic and Respiratory Diseases. Biomedicines 2021, 9, 492. [Google Scholar] [CrossRef]
- Pálinkás, Z.; Békési, D.; Utczás, M. Quantitation of Ecdysterone and Targeted Analysis of WADA-Prohibited Anabolic Androgen Steroids, Hormones, and Metabolic Modulators in Ecdysterone-Containing Dietary Supplements. Separations 2023, 10, 242. [Google Scholar] [CrossRef]
- The WADA 2020 Monitoring Program. Available online: https://www.wada-ama.org/sites/default/files/wada_2020_english_monitoring_program_.pdf (accessed on 3 May 2023).
- Hunyadi, A.; Herke, I.; Lengyel, K.; Báthori, M.; Kele, Z.; Simon, A.; Tóth, G.; Szendrei, K. Ecdysteroid-Containing Food Supplements from Cyanotis Arachnoidea on the European Market: Evidence for Spinach Product Counterfeiting. Sci. Rep. 2016, 6, 37322. [Google Scholar] [CrossRef]
- Navruz-Varli, S.; Sanlier, N. Nutritional and Health Benefits of Quinoa (Chenopodium quinoa Willd.). J. Cereal Sci. 2016, 69, 371–376. [Google Scholar] [CrossRef]
- Isenmann, E.; Yuliandra, T.; Touvleliou, K.; Broekmann, M.; de la Torre, X.; Botrè, F.; Diel, P.; Parr, M.K. Quinoa as Functional Food? Urinary Elimination of Ecdysterone after Consumption of Quinoa Alone and in Combination with spinach. bioRxiv 2023. [Google Scholar] [CrossRef]
- Schmidt, D.; Verruma-Bernardi, M.R.; Forti, V.A.; Borges, M.T.M.R. Quinoa and Amaranth as Functional Foods: A Review. Food Rev. Int. 2023, 39, 2277–2296. [Google Scholar] [CrossRef]
- Fehér, A.; Gazdecki, M.; Véha, M.; Szakály, M.; Szakály, Z. A Comprehensive Review of the Benefits of and the Barriers to the Switch to a Plant-Based Diet. Sustainability 2020, 12, 4136. [Google Scholar] [CrossRef]
- Sidorova, Y.S.; Petrov, N.A.; Shipelin, V.A.; Mazo, V.K. Spinach and quinoa—Prospective food sources of biologically active substances. Vopr. Pitan. 2020, 89, 100–106. [Google Scholar] [CrossRef] [PubMed]
- Page, M.J.; McKenzie, J.E.; Bossuyt, P.M.; Boutron, I.; Hoffmann, T.C.; Mulrow, C.D.; Shamseer, L.; Tetzlaff, J.M.; Akl, E.A.; Brennan, S.E.; et al. The PRISMA 2020 Statement: An Updated Guideline for Reporting Systematic Reviews. BMJ 2021, 372, n71. [Google Scholar] [CrossRef] [PubMed]
- Cahlíková, L.; Macáková, K.; Chlebek, J.; Hošt’álková, A.; Kulhánková, A.; Opletal, L. Ecdysterone and Its Activity on Some Degenerative Diseases. Nat. Prod. Commun. 2011, 6, 1934578X1100600527. [Google Scholar] [CrossRef]
- Chermnykh, N.S.; Shimanovskiĭ, N.L.; Shutko, G.V.; Syrov, V.N. The action of methandrostenolone and ecdysterone on the physical endurance of animals and on protein metabolism in the skeletal muscles. Farmakol. Toksikol. 1988, 51, 57–60. [Google Scholar]
- Kholodova, I.D.; Tugaĭ, V.A.; Zimina, V.P. Effect of vitamin D3 and 20-hydroxyecdysone on the content of ATP, creatine phosphate, carnosine and Ca2+ in skeletal muscles. Ukr. Biokhimicheskii Zhurnal (1978) 1997, 69, 3–9. [Google Scholar]
- Yoshida, T.; Otaka, T.; Uchiyama, M.; Ogawa, S. Effect of Ecdysterone on Hyperglycemia in Experimental Animals. Biochem. Pharmacol. 1971, 20, 3263–3268. [Google Scholar] [CrossRef]
- Lupien, P.J.; Hinse, C.; Chaudhary, K.D. Ecdysone as A Hypocholesterolemic Agent. Arch. Int. Physiol. Biochim. 1969, 77, 206–212. [Google Scholar] [CrossRef]
- Ogawa, S.; Nishimoto, N.; Matsuda, H. Pharmacology of Ecdysones in Vertebrates. In Invertebrate Endocrinology and Hormonal Heterophylly; Burdette, W.J., Ed.; Springer: Berlin/Heidelberg, Germany, 1974; pp. 341–344. ISBN 978-3-642-65769-6. [Google Scholar]
- Seidlova-Wuttke, D.; Ehrhardt, C.; Wuttke, W. Metabolic Effects of 20-OH-Ecdysone in Ovariectomized Rats. J. Steroid Biochem. Mol. Biol. 2010, 119, 121–126. [Google Scholar] [CrossRef]
- Lafont, R.; Dinan, L. Practical Uses for Ecdysteroids in Mammals Including Humans: And Update. J. Insect Sci. 2003, 3, 7. [Google Scholar] [CrossRef]
- Syrov, V.N. Comparative Experimental Investigation of the Anabolic Activity of Phytoecdysteroids and Steranabols. Pharm. Chem. J. 2000, 34, 193–197. [Google Scholar] [CrossRef]
- Franco, R.R.; de Almeida Takata, L.; Chagas, K.; Justino, A.B.; Saraiva, A.L.; Goulart, L.R.; de Melo Rodrigues Ávila, V.; Otoni, W.C.; Espindola, F.S.; da Silva, C.R. A 20-Hydroxyecdysone-Enriched Fraction from Pfaffia glomerata (Spreng.) Pedersen Roots Alleviates Stress, Anxiety, and Depression in Mice. J. Ethnopharmacol. 2021, 267, 113599. [Google Scholar] [CrossRef]
- Kapur, P.; Wuttke, W.; Jarry, H.; Seidlova-Wuttke, D. Beneficial Effects of β-Ecdysone on the Joint, Epiphyseal Cartilage Tissue and Trabecular Bone in Ovariectomized Rats. Phytomedicine 2010, 17, 350–355. [Google Scholar] [CrossRef]
- Zou, Y.; Wang, R.; Guo, H.; Dong, M. Phytoestrogen β-Ecdysterone Protects PC12 Cells Against MPP+-Induced Neurotoxicity In Vitro: Involvement of PI3K-Nrf2-Regulated Pathway. Toxicol. Sci. Off. J. Soc. Toxicol. 2015, 147, 28–38. [Google Scholar] [CrossRef]
- Mamadalieva, N.; Egamberdieva, D.; Tiezzi, A. In Vitro Biological Activities of the Components from Silene wallichiana. Med. Aromat. Plant Sci. Biotechnol. 2013, 7, 1–6. [Google Scholar]
- Takei, M.; Endo, K.; Nishimoto, N.; Shiobara, Y.; Inoue, S.; Matsuo, S. Effect of Ecdysterone on Histamine Release from Rat Peritoneal Mast Cells. J. Pharm. Sci. 1991, 80, 309–310. [Google Scholar] [CrossRef]
- Xu, T.; Niu, C.; Zhang, X.; Dong, M. β-Ecdysterone Protects SH-SY5Y Cells against β-Amyloid-Induced Apoptosis via c-Jun N-Terminal Kinase- and Akt-Associated Complementary Pathways. Lab. Invest. 2018, 98, 489–499. [Google Scholar] [CrossRef]
- Hung, T.-J.; Chen, W.-M.; Liu, S.-F.; Liao, T.-N.; Lee, T.-C.; Chuang, L.-Y.; Guh, J.-Y.; Hung, C.-Y.; Hung, Y.-J.; Chen, P.; et al. 20-Hydroxyecdysone Attenuates TGF-Β1-Induced Renal Cellular Fibrosis in Proximal Tubule Cells. J. Diabetes Complicat. 2012, 26, 463–469. [Google Scholar] [CrossRef]
- Jin, Z.; Wang, B.; Ren, L.; Yang, J.; Zheng, Z.; Yao, F.; Ding, R.; Wang, J.; He, J.; Wang, W.; et al. 20-Hydroxyecdysone Inhibits Inflammation via SIRT6-Mediated NF-κB Signaling in Endothelial Cells. Biochim. Et Biophys. Acta (BBA)-Mol. Cell Res. 2023, 1870, 119460. [Google Scholar] [CrossRef]
- Tang, Y.-H.; Yue, Z.-S.; Li, G.-S.; Zeng, L.-R.; Xin, D.-W.; Hu, Z.-Q.; Xu, C.-D. Effect of Β-ecdysterone on Glucocorticoid-induced Apoptosis and Autophagy in Osteoblasts. Mol. Med. Rep. 2018, 17, 158–164. [Google Scholar] [CrossRef]
- Mamadalieva, N.; El-Readi, M.Z.; Ovidi, E.; Ashour, M.L.; Hamoud, R.; Sagdullaev, S.S.; Azimova, S.S.; Tiezzi, A.; Wink, M. Antiproliferative, Antimicrobial and Antioxidant Activities of the Chemical Constituents of Ajuga turkestanica. Phytopharmacology 2013, 4, 1–18. [Google Scholar]
- Chen, Q.; Xia, Y.; Qiu, Z. Effect of Ecdysterone on Glucose Metabolism in vitro. Life Sci. 2006, 78, 1108–1113. [Google Scholar] [CrossRef]
- Shuvalov, O.; Fedorova, O.; Tananykina, E.; Gnennaya, Y.; Daks, A.; Petukhov, A.; Barlev, N.A. An Arthropod Hormone, Ecdysterone, Inhibits the Growth of Breast Cancer Cells via Different Mechanisms. Front. Pharmacol. 2020, 11, 561537. [Google Scholar] [CrossRef]
- Smirnova, G.; Bezmaternykh, K.; Oktyabrsky, O.N. The Effect of 20-hydroxyecdysone on the Susceptibility of Escherichia coli to Different Antibiotics. J. Appl. Microbiol. 2016, 121, 1511–1518. [Google Scholar] [CrossRef]
- Martins, A.; Sipos, P.; Dér, K.; Csábi, J.; Miklos, W.; Berger, W.; Zalatnai, A.; Amaral, L.; Molnár, J.; Szabó-Révész, P.; et al. Ecdysteroids Sensitize MDR and Non-MDR Cancer Cell Lines to Doxorubicin, Paclitaxel, and Vincristine but Tend to Protect Them from Cisplatin. BioMed Res. Int. 2015, 2015, e895360. [Google Scholar] [CrossRef]
- Trenin, D.S.; Volodin, V.V. 20-Hydroxyecdysone as a Human Lymphocyte and Neutrophil Modulator: In Vitro Evaluation. Arch. Insect Biochem. Physiol. 1999, 41, 156–161. [Google Scholar] [CrossRef]
- Mamadalieva, N.Z.; El-Readi, M.Z.; Janibekov, A.A.; Tahrani, A.; Wink, M. Phytoecdysteroids of Silene Guntensis and Their in Vitro Cytotoxic and Antioxidant Activity. Z. Für Naturforschung C 2011, 66, 215–224. [Google Scholar] [CrossRef]
- Romaniuk-Drapała, A.; Lisiak, N.; Totoń, E.; Matysiak, A.; Nawrot, J.; Nowak, G.; Kaczmarek, M.; Rybczyńska, M.; Rubiś, B. Proapoptotic and Proautophagic Activity of 20-Hydroxyecdysone in Breast Cancer Cells in vitro. Chem. Biol. Interact. 2021, 342, 109479. [Google Scholar] [CrossRef]
- Omanakuttan, A.; Bose, C.; Pandurangan, N.; Kumar, G.B.; Banerji, A.; Nair, B.G. Nitric Oxide and ERK Mediates Regulation of Cellular Processes by Ecdysterone. Exp. Cell Res. 2016, 346, 167–175. [Google Scholar] [CrossRef]
- Martucciello, S.; Paolella, G.; Muzashvili, T.; Skhirtladze, A.; Pizza, C.; Caputo, I.; Piacente, S. Steroids from Helleborus Caucasicus Reduce Cancer Cell Viability Inducing Apoptosis and GRP78 Down-Regulation. Chem. Biol. Interact. 2018, 279, 43–50. [Google Scholar] [CrossRef]
- Todorova, V.; Savova, M.S.; Ivanova, S.; Ivanov, K.; Georgiev, M.I. Anti-Adipogenic Activity of Rhaponticum carthamoides and Its Secondary Metabolites. Nutrients 2023, 15, 3061. [Google Scholar] [CrossRef]
- Zhang, X.; Xu, X.; Xu, T.; Qin, S. β-Ecdysterone Suppresses Interleukin-1β-Induced Apoptosis and Inflammation in Rat Chondrocytes via Inhibition of NF-κB Signaling Pathway. Drug Dev. Res. 2014, 75, 195–201. [Google Scholar] [CrossRef]
- Catalan, R.E.; Aragones, M.D.; Godoy, J.E.; Martinez, A.M. Ecdysterone Induces Acetylcholinesterase in Mammalian Brain. Comp. Biochem. Physiol. Part C Comp. Pharmacol. 1984, 78, 193–195. [Google Scholar] [CrossRef]
- Dai, W.-W.; Wang, L.-B.; Jin, G.-Q.; Wu, H.-J.; Zhang, J.; Wang, C.-L.; Wei, Y.-J.; Lee, J.-H.; Lay, Y.-A.E.; Yao, W. Beta-Ecdysone Protects Mouse Osteoblasts from Glucocorticoid-Induced Apoptosis in vitro. Planta Med. 2017, 83, 888–894. [Google Scholar] [CrossRef]
- Fang, L.; Li, J.; Zhou, J.; Wang, X.; Guo, L. Isolation and Purification of Three Ecdysteroids from the Stems of Diploclisia Glaucescens by High-Speed Countercurrent Chromatography and Their Anti-Inflammatory Activities in vitro. Molecules 2017, 22, 1310. [Google Scholar] [CrossRef]
- Yan, C.-P.; Wang, X.-K.; Jiang, K.; Yin, C.; Xiang, C.; Wang, Y.; Pu, C.; Chen, L.; Li, Y.-L. β-Ecdysterone Enhanced Bone Regeneration through the BMP-2/SMAD/RUNX2/Osterix Signaling Pathway. Front. Cell Dev. Biol. 2022, 10, 883228. [Google Scholar] [CrossRef]
- Catalán, R.E.; Martinez, A.M.; Aragones, M.D.; Miguel, B.G.; Robles, A.; Godoy, J.E. Alterations in Rat Lipid Metabolism Following Ecdysterone Treatment. Comp. Biochem. Physiol. Part B Comp. Biochem. 1985, 81, 771–775. [Google Scholar] [CrossRef]
- Gholipour, P.; Komaki, A.; Ramezani, M.; Parsa, H. Effects of the Combination of High-Intensity Interval Training and Ecdysterone on Learning and Memory Abilities, Antioxidant Enzyme Activities, and Neuronal Population in an Amyloid-Beta-Induced Rat Model of Alzheimer’s Disease. Physiol. Behav. 2022, 251, 113817. [Google Scholar] [CrossRef] [PubMed]
- Lawrence, M.M.; Zwetsloot, K.A.; Arthur, S.T.; Sherman, C.A.; Huot, J.R.; Badmaev, V.; Grace, M.; Lila, M.A.; Nieman, D.C.; Shanely, R.A. Phytoecdysteroids Do Not Have Anabolic Effects in Skeletal Muscle in Sedentary Aging Mice. Int. J. Environ. Res. Public. Health 2021, 18, 370. [Google Scholar] [CrossRef]
- Kizelsztein, P.; Govorko, D.; Komarnytsky, S.; Evans, A.; Wang, Z.; Cefalu, W.T.; Raskin, I. 20-Hydroxyecdysone Decreases Weight and Hyperglycemia in a Diet-Induced Obesity Mice Model. Am. J. Physiol.-Endocrinol. Metab. 2009, 296, E433–E439. [Google Scholar] [CrossRef]
- Dai, W.; Jiang, L.; Lay, Y.-A.E.; Chen, H.; Jin, G.; Zhang, H.; Kot, A.; Ritchie, R.O.; Lane, N.E.; Yao, W. Prevention of Glucocorticoid Induced Bone Changes with Beta-Ecdysone. Bone 2015, 74, 48–57. [Google Scholar] [CrossRef]
- Wu, J.; Gao, L.; Shang, L.; Wang, G.; Wei, N.; Chu, T.; Chen, S.; Zhang, Y.; Huang, J.; Wang, J.; et al. Ecdysterones from Rhaponticum carthamoides (Willd.) Iljin Reduce Hippocampal Excitotoxic Cell Loss and Upregulate mTOR Signaling in Rats. Fitoterapia 2017, 119, 158–167. [Google Scholar] [CrossRef]
- Catalan, R.E.; Martinez, A.M.; Aragones, M.D.; Miguel, B.G.; Robles, A.; Godoy, J.E. Effect of Ecdysterone Treatment on the Cyclic AMP-Protein Kinase System in Adipose Tissue. J. Steroid Biochem. 1982, 16, 573–576. [Google Scholar] [CrossRef]
- Xia, X.; Zhang, Q.; Liu, R.; Wang, Z.; Tang, N.; Liu, F.; Huang, G.; Jiang, X.; Gui, G.; Wang, L.; et al. Effects of 20-Hydroxyecdysone on Improving Memory Deficits in Streptozotocin-Induced Type 1 Diabetes Mellitus in Rat. Eur. J. Pharmacol. 2014, 740, 45–52. [Google Scholar] [CrossRef]
- You, W.-L.; Xu, Z.-L. β-Ecdysone Promotes Osteogenic Differentiation of Bone Marrow Mesenchymal Stem Cells. J. Gene Med. 2020, 22, e3207. [Google Scholar] [CrossRef]
- Catalan, R.E.; Aragonés, M.D.; Martínez, A.M. Effect of Ecdysterone on Cyclic AMP and Cyclic GMP in Mouse Plasma. Biochem. Biophys. Res. Commun. 1979, 87, 1018–1023. [Google Scholar] [CrossRef]
- Catalán, R.E.; Aragonés, M.D.; Martínez, A.M. Effect of Ecdysterone on the Cyclic AMP-Protein Kinase System in Mouse Liver. Biochem. Biophys. Res. Commun. 1979, 89, 44–49. [Google Scholar] [CrossRef]
- Parr, M.K.; Zhao, P.; Haupt, O.; Ngueu, S.T.; Hengevoss, J.; Fritzemeier, K.H.; Piechotta, M.; Schlörer, N.; Muhn, P.; Zheng, W.-Y.; et al. Estrogen Receptor Beta Is Involved in Skeletal Muscle Hypertrophy Induced by the Phytoecdysteroid Ecdysterone. Mol. Nutr. Food Res. 2014, 58, 1861–1872. [Google Scholar] [CrossRef]
- Marschall, M.J.M.; Ringseis, R.; Gessner, D.K.; Grundmann, S.M.; Most, E.; Wen, G.; Maheshwari, G.; Zorn, H.; Eder, K. Effect of Ecdysterone on the Hepatic Transcriptome and Lipid Metabolism in Lean and Obese Zucker Rats. Int. J. Mol. Sci. 2021, 22, 5241. [Google Scholar] [CrossRef]
- Lim, H.-S.; Yoon, K.; Lee, D.H.; Lee, Y.-S.; Chung, J.H.; Park, G. Effects of 20-Hydroxyecdysone on UVB-Induced Photoaging in Hairless Mice. Biomed. Pharmacother. Biomed. Pharmacother. 2023, 164, 114899. [Google Scholar] [CrossRef]
- Gao, L.; Cai, G.; Shi, X. Beta-Ecdysterone Induces Osteogenic Differentiation in Mouse Mesenchymal Stem Cells and Relieves Osteoporosis. Biol. Pharm. Bull. 2008, 31, 2245–2249. [Google Scholar] [CrossRef]
- Ehrhardt, C.; Wessels, J.T.; Wuttke, W.; Seidlová-Wuttke, D. The Effects of 20-Hydroxyecdysone and 17β-Estradiol on the Skin of Ovariectomized Rats. Menopause 2011, 18, 323–327. [Google Scholar] [CrossRef]
- Sun, Y.; Zhao, D.-L.; Liu, Z.-X.; Sun, X.-H.; Li, Y. Beneficial Effect of 20-hydroxyecdysone Exerted by Modulating Antioxidants and Inflammatory Cytokine Levels in Collagen-induced Arthritis: A Model for Rheumatoid Arthritis. Mol. Med. Rep. 2017, 16, 6162–6169. [Google Scholar] [CrossRef]
- Anthony, T.G.; Mirek, E.T.; Bargoud, A.R.; Phillipson-Weiner, L.; DeOliveira, C.M.; Wetstein, B.; Graf, B.L.; Kuhn, P.E.; Raskin, I. Evaluating the Effect of 20-Hydroxyecdysone (20HE) on Mechanistic Target of Rapamycin Complex 1 (mTORC1) Signaling in the Skeletal Muscle and Liver of Rats. Appl. Physiol. Nutr. Metab. Physiol. Appl. Nutr. Metab. 2015, 40, 1324–1328. [Google Scholar] [CrossRef]
- Konovalova, N.P.; Mitrokhin, Y.I.; Volkova, L.M.; Sidorenko, L.I.; Todorov, I.N. Ecdysterone Modulates Antitumor Activity of Cytostatics and Biosynthesis of Macromolecules in Tumor-Bearing Mice. Biol. Bull. Russ. Acad. Sci. 2002, 29, 530–536. [Google Scholar] [CrossRef]
- Fenner, R.; Zimmer, A.R.; Neves, G.; Kliemann, M.; Gosmann, G.; Rates, S.M.K. Hypnotic Effect of Ecdysterone Isolated from Pfaffia glomerata (Spreng.) Pedersen. Rev. Bras. Farmacogn. 2008, 18, 170–176. [Google Scholar] [CrossRef]
- Fedorov, V.N.; Punegova, N.V.; Zainullin, V.G.; Punegov, V.V.; Sychev, R.L. Extraction of Ecdysterone-80 from Serratula coronata L. and Evaluation of Its Pharmacological Actions. II. Cardioprotective Properties. Effects on Hormone-Transmitter Balance in Chronic Cardiac Failure. Pharm. Chem. J. 2009, 43, 36–40. [Google Scholar] [CrossRef]
- Ramazanov, N.S.; Bobayev, I.D.; Yusupova, U.Y.; Aliyeva, N.K.; Egamova, F.R.; Yuldasheva, N.K.; Syrov, V.N. Phytoecdysteroids-Containing Extract from Stachys Hissarica Plant and Its Wound-Healing Activity. Nat. Prod. Res. 2017, 31, 593–597. [Google Scholar] [CrossRef]
- Yang, L.; Pan, J. Therapeutic Effect of Ecdysterone Combine Paeonol Oral Cavity Direct Administered on Radiation-Induced Oral Mucositis in Rats. Int. J. Mol. Sci. 2019, 20, 3800. [Google Scholar] [CrossRef]
- Tóth, N.; Szabó, A.; Kacsala, P.; Héger, J.; Zádor, E. 20-Hydroxyecdysone Increases Fiber Size in a Muscle-Specific Fashion in Rat. Phytomed. Int. J. Phytother. Phytopharm. 2008, 15, 691–698. [Google Scholar] [CrossRef]
- Seidlova-Wuttke, D.; Wuttke, W. In a Placebo-Controlled Study ß-Ecdysone (ECD) Prevented the Development of the Metabolic Syndrome. Planta Med. 2012, 78, CL37. [Google Scholar] [CrossRef]
- Dioh, W.; Tourette, C.; Del Signore, S.; Daudigny, L.; Dupont, P.; Balducci, C.; Dilda, P.J.; Lafont, R.; Veillet, S. A Phase 1 Study for Safety and Pharmacokinetics of BIO101 (20-Hydroxyecdysone) in Healthy Young and Older Adults. J. Cachexia Sarcopenia Muscle 2023, 14, 1259–1273. [Google Scholar] [CrossRef]
- Wilborn, C.D.; Taylor, L.W.; Campbell, B.I.; Kerksick, C.; Rasmussen, C.J.; Greenwood, M.; Kreider, R.B. Effects of Methoxyisoflavone, Ecdysterone, and Sulfo-Polysaccharide Supplementation on Training Adaptations in Resistance-Trained Males. J. Int. Soc. Sports Nutr. 2006, 3, 19–27. [Google Scholar] [CrossRef]
- Bathori, M.; Toth, N.; Hunyadi, A.; Marki, A.; Zador, E. Phytoecdysteroids and Anabolic-Androgenic Steroids—Structure and Effects on Humans. Curr. Med. Chem. 2008, 15, 75–91. [Google Scholar] [CrossRef]
- Łotocka, B.; Geszprych, A. Anatomy of the Vegetative Organs and Secretory Structures of Rhaponticum carthamoides (Asteraceae). Bot. J. Linn. Soc. 2004, 144, 207–233. [Google Scholar] [CrossRef]
- Timofeev, N.P. Leuzea carthamoides DC.: Application Prospects as Pharmpreparations and Biologically Active Components. In Functional Foods for Chronic Diseases; D&A Incorporated: Richardson, TX, USA, 2006. [Google Scholar]
- Brekhman, I.I.; Dardymov, I.V. New Substances of Plant Origin Which Increase Nonspecific Resistance. Annu. Rev. Pharmacol. 1969, 9, 419–430. [Google Scholar] [CrossRef]
- Opletal, L.; Sovová, M.; Dittrich, M.; Solich, P.; Dvorák, J.; Krátký, F.; Cerovský, J.; Hofbauer, J. Phytotherapeutic aspects of diseases of the circulatory system. 6. Leuzea carthamoides (Willd.) DC: The status of research and possible use of the taxon. Ceska Slov. Farm. 1997, 46, 247–255. [Google Scholar] [PubMed]
- Todorova, V.; Ivanov, K.; Ivanova, S. Comparison between the Biological Active Compounds in Plants with Adaptogenic Properties (Rhaponticum carthamoides, Lepidium meyenii, Eleutherococcus senticosus and Panax ginseng). Plants 2022, 11, 64. [Google Scholar] [CrossRef]
- Sharaf, M.; Skiba, A.; Weglarz, Z.; El-Ansari, M.A. Two Flavonol 5-O-Glycosides from the Roots of Leuzea carthamoides. Fitoterapia 2001, 72, 940–942. [Google Scholar] [CrossRef]
- Skiba, A.; Weglarz, Z. Phenolic acids of Rhaponticum carthamoides. Acta Hortic. 2003, 597, 119–124. [Google Scholar] [CrossRef]
- Szendrei, K.; Varga, E.; Hajdú, Z.; Herke, I.; Lafont, R.; Girault, J.P. Ajugasterone C and 5-Deoxykaladasterone, an Ecdysteroid Artifact, from Leuzea Carthamoides. J. Nat. Prod. 1988, 51, 993–995. [Google Scholar] [CrossRef]
- Píš, J.; Buděšínskў, M.; Vokáč, K.; Laudová, V.; Harmatha, J. Ecdysteroids from the Roots of Leuzea carthamoides. Phytochemistry 1994, 37, 707–711. [Google Scholar] [CrossRef]
- Girault, J.-P.; Lafont, R.; Varga, E.; Hajdu, Z.; Herke, I.; Szendrei, K. Ecdysteroids from Leuzea Carthamoides. Phytochemistry 1988, 27, 737–741. [Google Scholar] [CrossRef]
- Bastaev, U.A.; Abubakirov, N.K. Phytoecdysteroids of Rhaponticum carthamoides. Chem. Nat. Compd. 1987, 23, 565–568. [Google Scholar] [CrossRef]
- Skała, E.; Rijo, P.; Garcia, C.; Sitarek, P.; Kalemba, D.; Toma, M.; Szemraj, J.; Pytel, D.; Wysokińska, H.; Śliwiński, T. The Essential Oils of Rhaponticum carthamoides Hairy Roots and Roots of Soil-Grown Plants: Chemical Composition and Antimicrobial, Anti-Inflammatory, and Antioxidant Activities. Oxid. Med. Cell. Longev. 2016, 2016, 8505384. [Google Scholar] [CrossRef] [PubMed]
- Todorova, V.; Ivanova, S.; Georgieva, Y.; Nalbantova, V.; Karcheva-Bahchevanska, D.; Benbassat, N.; Savova, M.S.; Georgiev, M.I.; Ivanov, K. Chemical Composition and Histochemical Localization of Essential Oil from Wild and Cultivated Rhaponticum carthamoides Roots and Rhizomes. Plants 2022, 11, 2061. [Google Scholar] [CrossRef]
- Havlik, J.; Budesinsky, M.; Kloucek, P.; Kokoska, L.; Valterova, I.; Vasickova, S.; Zeleny, V. Norsesquiterpene Hydrocarbon, Chemical Composition and Antimicrobial Activity of Rhaponticum carthamoides Root Essential Oil. Phytochemistry 2009, 70, 414–418. [Google Scholar] [CrossRef]
- Geszprych, A.; Weglarz, Z. Composition of Essential Oil from Underground and Aboveground Organs of Rhaponticum carthamoides [Willd.] Iljin. Herba Pol. 2002, 48, 188–192. [Google Scholar]
- Krasnov, E.A.; Saratikov, A.S.; Yakunina, G.D. Inokosterone and Ecdysterone from Rhaponticum carthamoides. Chem. Nat. Compd. 1976, 12, 494–495. [Google Scholar] [CrossRef]
- Ramazanov, N.S.; Makshimov, E.S.; Saatov, Z.; Mamatkhanov, A.U.; Abdullaev, N.D. Phytoecdysteroids of Plants of the genus Rhaponticum I. Carthamosterone a from Rh. Carthamoides. Chem. Nat. Compd. 1997, 33, 301–302. [Google Scholar] [CrossRef]
- Martirosyan, D.M. Functional Foods for Chronic Diseases; D&A Inc.: Richardson, TX, USA, 2006; ISBN 978-0-9767535-2-0. [Google Scholar]
- Skała, E.; Kowalczyk, T.; Toma, M.; Szemraj, J.; Radek, M.; Pytel, D.; Wieczfinska, J.; Wysokińska, H.; Śliwiński, T.; Sitarek, P. Induction of Apoptosis in Human Glioma Cell Lines of Various Grades through the ROS-Mediated Mitochondrial Pathway and Caspase Activation by Rhaponticum carthamoides Transformed Root Extract. Mol. Cell. Biochem. 2018, 445, 89–97. [Google Scholar] [CrossRef] [PubMed]
- Skała, E.; Sitarek, P.; Toma, M.; Szemraj, J.; Radek, M.; Nieborowska-Skorska, M.; Skorski, T.; Wysokińska, H.; Śliwiński, T. Inhibition of Human Glioma Cell Proliferation by Altered Bax/Bcl-2-P53 Expression and Apoptosis Induction by Rhaponticum carthamoides Extracts from Transformed and Normal Roots. J. Pharm. Pharmacol. 2016, 68, 1454–1464. [Google Scholar] [CrossRef]
- Zheng, Z.; Xian, Y.; Jin, Z.; Yao, F.; Liu, Y.; Deng, Y.; Wang, B.; Chen, D.; Yang, J.; Ren, L.; et al. Rhaponticum carthamoides Improved Energy Metabolism and Oxidative Stress through the SIRT6/Nrf2 Pathway to Ameliorate Myocardial Injury. Phytomedicine 2022, 105, 154197. [Google Scholar] [CrossRef]
- Jurkštienė, V.; Pavilonis, A.; Garšvienė, D.; Juozulynas, A.; Samsonienė, L.; Daukšienė, D.; Jankauskienė, K.; Šimonienė-Kazlauskienė, G.; Stankevičius, E. Investigation of the Antimicrobial Activity of Rhaponticum (Rhaponticum carthamoides D.C. Iljin) and Shrubby Cinquefoil (Potentilla fruticosa L.). Medicina 2011, 47, 24. [Google Scholar] [CrossRef]
- Peschel, W.; Kump, A.; Prieto, J.M. Effects of 20-Hydroxyecdysone, Leuzea carthamoides Extracts, Dexamethasone and Their Combinations on the NF-κB Activation in HeLa Cells. J. Pharm. Pharmacol. 2011, 63, 1483–1495. [Google Scholar] [CrossRef]
- Gaube, F.; Wölfl, S.; Pusch, L.; Werner, U.; Kroll, T.C.; Schrenk, D.; Hartmann, R.W.; Hamburger, M. Effects of Leuzea carthamoides on Human Breast Adenocarcinoma MCF-7 Cells Determined by Gene Expression Profiling and Functional Assays. Planta Med. 2008, 74, 1701–1708. [Google Scholar] [CrossRef]
- Skała, E.; Sitarek, P.; Różalski, M.; Krajewska, U.; Szemraj, J.; Wysokińska, H.; Śliwiński, T. Antioxidant and DNA Repair Stimulating Effect of Extracts from Transformed and Normal Roots of Rhaponticum carthamoides against Induced Oxidative Stress and DNA Damage in CHO Cells. Oxid. Med. Cell. Longev. 2016, 2016, 5753139. [Google Scholar] [CrossRef] [PubMed]
- Publication—Composition and Biological Activity of Rhaponticum carthamoides Extracts Obtained from Plants Collected in Poland and Russia—University of Gdańsk. Available online: https://repozytorium.bg.ug.edu.pl/info/article/UOGab0a3e66748d484882bc3dd7288a480a/ (accessed on 22 February 2024).
- Šelepcová, L.; Jalč, D.; Javorský, P.; Baran, M. Influence of Rhaponticum carthamoides Wild on the Growth of Ruminal Bacteria in Vitro and on Fermentation in an Artificial rumen (Rusitec). Arch. Für Tierernaehrung 1993, 43, 147–156. [Google Scholar] [CrossRef] [PubMed]
- Kokoska, L.; Janovska, D.; Rada, V.; Nepovim, A.; Vanek, T. In Vitro. Antibacterial Activity of Four Leuzea. Species. Pharm. Biol. 2005, 43, 8–11. [Google Scholar] [CrossRef]
- Kokoska, L.; Polesny, Z.; Rada, V.; Nepovim, A.; Vanek, T. Screening of Some Siberian Medicinal Plants for Antimicrobial Activity. J. Ethnopharmacol. 2002, 82, 51–53. [Google Scholar] [CrossRef] [PubMed]
- Mácsai, L.; Datki, Z.L.; Csupor, D.; Horváth, A.; Zomborszki, Z.P. Biological Activities of Four Adaptogenic Plant Extracts and Their Active Substances on a Rotifer Model. Evid.-Based Complement. Altern. Med. 2018, 2018, 3690683. [Google Scholar] [CrossRef]
- Mosharrof, A.H. Effects of Extract from Rhapontcum carthamoides (Willd) Iljin (Leuzea) on Learning and Memory in Rats. Acta Physiol. Pharmacol. Bulg. 1987, 13, 37–42. [Google Scholar] [PubMed]
- Petkov, V.; Roussinov, K.; Todorov, S.; Lazarova, M.; Yonkov, D.; Draganova, S. Pharmacological Investigations on Rhaponticum carthamoides. Planta Med. 1984, 50, 205–209. [Google Scholar] [CrossRef] [PubMed]
- Dushkin, M.; Khrapova, M.; Kovshik, G.; Chasovskikh, M.; Menshchikova, E.; Trufakin, V.; Shurlygina, A.; Vereschagin, E. Effects of Rhaponticum carthamoides versus Glycyrrhiza glabra and Punica granatum Extracts on Metabolic Syndrome Signs in Rats. BMC Complement. Altern. Med. 2014, 14, 33. [Google Scholar] [CrossRef]
- Roumanille, R.; Vernus, B.; Brioche, T.; Descossy, V.; Van Ba, C.T.; Campredon, S.; Philippe, A.G.; Delobel, P.; Bertrand-Gaday, C.; Chopard, A.; et al. Acute and Chronic Effects of Rhaponticum carthamoides and Rhodiola Rosea Extracts Supplementation Coupled to Resistance Exercise on Muscle Protein Synthesis and Mechanical Power in Rats. J. Int. Soc. Sports Nutr. 2020, 17, 58. [Google Scholar] [CrossRef]
- Baev, A.Y.; Charyshnikova, O.S.; Khasanov, F.A.; Nebesnaya, K.S.; Makhmudov, A.R.; Rahmedova, M.T.; Khushbaktova, Z.A.; Syrov, V.N.; Levitskaya, Y.V. Ecdysterone Prevents Negative Effect of Acute Immobilization Stress on Energy Metabolism of Rat Liver Mitochondria. J. Steroid Biochem. Mol. Biol. 2022, 219, 106066. [Google Scholar] [CrossRef]
- Shakhmurova, G.A.; Syrov, V.N.; Khushbaktova, Z.A. Immunomodulating and Antistress Activity of Ecdysterone and Turkesterone under Immobilization-Induced Stress Conditions in Mice. Pharm. Chem. J. 2010, 44, 7–9. [Google Scholar] [CrossRef]
- Neves, C.S.; Gomes, S.S.L.; Dos Santos, T.R.; De Almeida, M.M.; De Souza, Y.O.; Garcia, R.M.G.; Otoni, W.C.; Chedier, L.M.; Viccini, L.F.; De Campos, J.M.S. The Phytoecdysteroid β-Ecdysone Is Genotoxic in Rodent Bone Marrow Micronuclei and Allium cepa L. Assays. J. Ethnopharmacol. 2016, 177, 81–84. [Google Scholar] [CrossRef]
- Plotnikov, M.B.; Aliev, O.I.; Vasil’ev, A.S.; Andreeva, V.Y.; Krasnov, E.A.; Kalinkina, G.I. Effect of Rhaponticum carthamoides Extract on Structural and Metabolic Parameters of Erythrocytes in Rats with Cerebral Ischemia. Bull. Exp. Biol. Med. 2008, 146, 45–48. [Google Scholar] [CrossRef] [PubMed]
- Selepcova, L.; Sommer, A.; Vargova, M. Effect of Feeding on a Diet Containing Varying Amounts of Rhaponticum carthamoides Hay Meal on Selected Morphological Parameters in Rats. EJE 2013, 92, 391–397. [Google Scholar]
- Ryan, E.D.; Gerstner, G.R.; Mota, J.A.; Trexler, E.T.; Giuliani, H.K.; Blue, M.N.M.; Hirsch, K.R.; Smith-Ryan, A.E. The Acute Effects of a Multi-Ingredient Herbal Supplement on Performance Fatigability: A Double-Blind, Randomized, and Placebo-Controlled Trial. J. Diet. Suppl. 2021, 18, 507–516. [Google Scholar] [CrossRef] [PubMed]
- Nešković, M.; Ćulafić, L. Spinach (Spinacia oleracea L.). In Crops II; Bajaj, Y.P.S., Ed.; Biotechnology in Agriculture and Forestry; Springer: Berlin/Heidelberg, Germany, 1988; pp. 370–385. ISBN 978-3-642-73520-2. [Google Scholar]
- Murcia, M.A.; Jiménez-Monreal, A.M.; Gonzalez, J.; Martínez-Tomé, M. Chapter 11—Spinach. In Nutritional Composition and Antioxidant Properties of Fruits and Vegetables; Jaiswal, A.K., Ed.; Academic Press: Cambridge, MA, USA, 2020; pp. 181–195. ISBN 978-0-12-812780-3. [Google Scholar]
- Pandey, S.C.; Kalloo, G. 23—Spinach: Spinacia oleracea L. In Genetic Improvement of Vegetable Crops; Kalloo, G., Bergh, B.O., Eds.; Pergamon: Amsterdam, The Netherlands, 1993; pp. 325–336. ISBN 978-0-08-040826-2. [Google Scholar]
- Huda-Faujan, N.; Zubairi, S.I.; Baker, A.A.A. Nutritional and Bioactive Constituents of Antioxidant and Antimicrobial Properties in Spinacia oleracea: A Review. Sains Malays. 2023, 52, 2571–2585. [Google Scholar] [CrossRef]
- Babu, N.R.; Divakar, J.; Krishna, U.L.; Vigneshwaran, C. Study of Antimicrobial, Antioxidant, Anti-Inflammatory Activities and Phytochemical Analysis of Cooked and Uncooked Different Spinach Leaves. J. Pharmacogn. Phytochem. 2018, 7, 1798–1803. [Google Scholar]
- Simko, I.; Hayes, R.J.; Mou, B.; McCreight, J.D. Lettuce and Spinach. In Yield Gains in Major U.S. Field Crops; John Wiley & Sons, Ltd.: Hoboken, NJ, USA, 2014; pp. 53–85. ISBN 978-0-89118-620-5. [Google Scholar]
- Roberts, J.L.; Moreau, R. Functional Properties of Spinach (Spinacia oleracea L.) Phytochemicals and Bioactives. Food Funct. 2016, 7, 3337–3353. [Google Scholar] [CrossRef] [PubMed]
- Morelock, T.E.; Correll, J.C. Spinach. In Vegetables I: Asteraceae, Brassicaceae, Chenopodicaceae, and Cucurbitaceae; Prohens, J., Nuez, F., Eds.; Handbook of Plant Breeding; Springer: New York, NY, USA, 2008; pp. 189–218. ISBN 978-0-387-30443-4. [Google Scholar]
- Salehi, B.; Tumer, T.B.; Ozleyen, A.; Peron, G.; Dall’Acqua, S.; Rajkovic, J.; Naz, R.; Nosheen, A.; Mudau, F.N.; Labanca, F.; et al. Plants of the genus Spinacia: From Bioactive Molecules to Food and Phytopharmacological Applications. Trends Food Sci. Technol. 2019, 88, 260–273. [Google Scholar] [CrossRef]
- Ambo, A.I.; Patience, O.; Ayakeme, E.B. Evaluation of the proximate composition and metal content of spinach (Spinacia oleracea) from selected towns in nasarawa state, Nigeria. Sci. World J. 2023, 18, 26–30. [Google Scholar]
- Lomnitski, L.; Bergman, M.; Nyska, A.; Ben-Shaul, V.; Grossman, S. Composition, Efficacy, and Safety of Spinach Extracts. Nutr. Cancer 2003, 46, 222–231. [Google Scholar] [CrossRef]
- Olasupo, A.; Aborisade, A.; Olagoke, O. Phytochemical Analysis and Antibacterial Activities of Spinach Leaf. Am. J. Phytomed. Clin. Ther. 2018, 6, 8. [Google Scholar]
- Bakrim, A.; Maria, A.; Sayah, F.; Lafont, R.; Takvorian, N. Ecdysteroids in Spinach (Spinacia oleracea L.): Biosynthesis, Transport and Regulation of Levels. Plant Physiol. Biochem. 2008, 46, 844–854. [Google Scholar] [CrossRef] [PubMed]
- Gorelick, J.; Iraqi, R.H.; Bernstein, N. Ecdysteroid Content and Therapeutic Activity in Elicited Spinach Accessions. Plants 2020, 9, 727. [Google Scholar] [CrossRef] [PubMed]
- Schmelz, E.A.; Grebenok, R.J.; Galbraith, D.W.; Bowers, W.S. Insect-Induced Synthesis of Phytoecdysteroids in Spinach, Spinacia oleracea. J. Chem. Ecol. 1999, 25, 1739–1757. [Google Scholar] [CrossRef]
- Bajkacz, S.; Rusin, K.; Wolny, A.; Adamek, J.; Erfurt, K.; Chrobok, A. Highly Efficient Extraction Procedures Based on Natural Deep Eutectic Solvents or Ionic Liquids for Determination of 20-Hydroxyecdysone in Spinach. Molecules 2020, 25, 4736. [Google Scholar] [CrossRef]
- Fang, X.; Szołtysik, R.; Tang, J.; Bajkacz, S. Efficient Extraction and Sensitive HPLC-MS/MS Quantification of Selected Ecdysteroids in Plants. J. Food Compos. Anal. 2022, 110, 104580. [Google Scholar] [CrossRef]
- Saeng-ngam, S.; Juntawong, N.; Vajarothai, S.; Visetson, S. Comparative study of moulting hormone content in different plant species. In Proceedings of the 42nd Kasetsart University Annual Conference, Kasetsart, Thailand, 3–6 February 2004; pp. 284–290. [Google Scholar]
- Grucza, K.; Wicka, M.; Drapała, A.; Kwiatkowska, D. Determination of Ecdysterone in Dietary Supplements and Spinach by Ultra-High-Performance Liquid Chromatography-Tandem Mass Spectrometry. Separations 2022, 9, 8. [Google Scholar] [CrossRef]
- Bohlooli, S.; Barmaki, S.; Khoshkhahesh, F.; Nakhostin-Roohi, B. The Effect of Spinach Supplementation on Exercise-Induced Oxidative Stress. J. Sports Med. Phys. Fit. 2015, 55, 609–614. [Google Scholar]
- Yuliandra, T.; Touvleliou, K.; de la Torre, X.; Botrè, F.; Loke, S.; Isenmann, E.; Valder, S.; Diel, P.; Parr, M.K. Urinary Excretion of Ecdysterone and Its Metabolites Following Spinach Consumption. Mol. Nutr. Food Res. 2023, 67, 2200518. [Google Scholar] [CrossRef]
- Panda, V.; Shinde, P.; Dande, P. Consumption of Spinacia oleracea (Spinach) and Aerobic Exercise Controls Obesity in Rats by an Inhibitory Action on Pancreatic Lipase. Arch. Physiol. Biochem. 2020, 126, 187–195. [Google Scholar] [CrossRef]
- Panda, V.; Shinde, P. Appetite Suppressing Effect of Spinacia oleracea in Rats: Involvement of the Short Term Satiety Signal Cholecystokinin. Appetite 2017, 113, 224–230. [Google Scholar] [CrossRef]
- Bhatia, A.L.; Jain, M. Spinacia oleracea L. Protects against Gamma Radiations: A Study on Glutathione and Lipid Peroxidation in Mouse Liver. Phytomed. Int. J. Phytother. Phytopharm. 2004, 11, 607–615. [Google Scholar] [CrossRef]
- Rahati, S.; Kamalinezhad, M.; Ebrahimi, A.; Eshraghian, M.; Pishva, H. Accelerated Wound Healing Induced by Spinach Extract in Experimental Model Diabetic Rats with Streptozotocin. Sci. Rep. 2023, 13, 14933. [Google Scholar] [CrossRef]
- Heo, J.-C.; Park, C.-H.; Lee, H.-J.; Kim, S.-O.; Kim, T.-H.; Lee, S.-H. Amelioration of Asthmatic Inflammation by an Aqueous Extract of Spinacia oleracea Linn. Int. J. Mol. Med. 2010, 25, 409–414. [Google Scholar] [CrossRef]
- Ko, S.-H.; Park, J.-H.; Kim, S.-Y.; Lee, S.W.; Chun, S.-S.; Park, E. Antioxidant Effects of Spinach (Spinacia oleracea L.) Supplementation in Hyperlipidemic Rats. Prev. Nutr. Food Sci. 2014, 19, 19–26. [Google Scholar] [CrossRef]
- Breitbart, E.; Lomnitski, L.; Nyska, A.; Malik, Z.; Bergman, M.; Sofer, Y.; Haseman, J.K.; Grossman, S. Effects of Water-Soluble Antioxidant from Spinach, NAO, on Doxorubicin-Induced Heart Injury. Hum. Exp. Toxicol. 2001, 20, 337–345. [Google Scholar] [CrossRef]
- Pérez-Piñero, S.; Ávila-Gandía, V.; Rubio Arias, J.A.; Muñoz-Carrillo, J.C.; Losada-Zafrilla, P.; López-Román, F.J. A 12-Week Randomized Double-Blind Placebo-Controlled Clinical Trial, Evaluating the Effect of Supplementation with a Spinach Extract on Skeletal Muscle Fitness in Adults Older than 50 Years of Age. Nutrients 2021, 13, 4373. [Google Scholar] [CrossRef]
- Maruyama, C.; Kikuchi, N.; Masuya, Y.; Hirota, S.; Araki, R.; Maruyama, T. Effects of Green-Leafy Vegetable Intake on Postprandial Glycemic and Lipidemic Responses and α-Tocopherol Concentration in Normal Weight and Obese Men. J. Nutr. Sci. Vitaminol. 2013, 59, 264–271. [Google Scholar] [CrossRef] [PubMed]
- Nikrad, N.; Farhangi, M.A.; Fard Tabrizi, F.P.; Vaezi, M.; Mahmoudpour, A.; Mesgari-Abbasi, M. The Effect of Calorie-Restriction along with Thylakoid Membranes of Spinach on the Gut-Brain Axis Pathway and Oxidative Stress Biomarkers in Obese Women with Polycystic Ovary Syndrome: A Randomized, Double-Blind, Placebo-Controlled Clinical Trial. J. Ovarian Res. 2023, 16, 216. [Google Scholar] [CrossRef]
- Saeidi, A.; Saei, M.A.; Mohammadi, B.; Zarei, H.R.A.; Vafaei, M.; Mohammadi, A.S.; Barati, M.; Montazer, M.; Razi, O.; Kiyumi, M.H.A.; et al. Supplementation with Spinach-Derived Thylakoid Augments the Benefits of High Intensity Training on Adipokines, Insulin Resistance and Lipid Profiles in Males with Obesity. Front. Endocrinol. 2023, 14, 1141796. [Google Scholar] [CrossRef] [PubMed]
- Rebello, C.J.; Chu, J.; Beyl, R.; Edwall, D.; Erlanson-Albertsson, C.; Greenway, F.L. Acute Effects of a Spinach Extract Rich in Thylakoids on Satiety: A Randomized Controlled Crossover Trial. J. Am. Coll. Nutr. 2015, 34, 470–477. [Google Scholar] [CrossRef]
- Schirrmacher, G.; Skurk, T.; Hauner, H.; Grassmann, J. Effect of Spinacia oleraceae L. and Perilla frutescens L. on Antioxidants and Lipid Peroxidation in an Intervention Study in Healthy Individuals. Plant Foods Hum. Nutr. 2010, 65, 71–76. [Google Scholar] [CrossRef]
- Corte-Real, J.; Guignard, C.; Gantenbein, M.; Weber, B.; Burgard, K.; Hoffmann, L.; Richling, E.; Bohn, T. No Influence of Supplemental Dietary Calcium Intake on the Bioavailability of Spinach Carotenoids in Humans. Br. J. Nutr. 2017, 117, 1560–1569. [Google Scholar] [CrossRef] [PubMed]
- Kopsell, D.A.; Lefsrud, M.G.; Kopsell, D.E.; Wenzel, A.J.; Gerweck, C.; Curran-Celentano, J. Spinach Cultigen Variation for Tissue Carotenoid Concentrations Influences Human Serum Carotenoid Levels and Macular Pigment Optical Density Following a 12-Week Dietary Intervention. J. Agric. Food Chem. 2006, 54, 7998–8005. [Google Scholar] [CrossRef]
- Hussain, M.I.; Farooq, M.; Syed, Q.A.; Ishaq, A.; Al-Ghamdi, A.A.; Hatamleh, A.A. Botany, Nutritional Value, Phytochemical Composition and Biological Activities of Quinoa. Plants 2021, 10, 2258. [Google Scholar] [CrossRef]
- Chen, X.; Zhang, Y.; Cao, B.; Wei, X.; Shen, Z.; Su, N. Assessment and Comparison of Nutritional Qualities of Thirty Quinoa (Chenopodium quinoa Willd.) Seed Varieties. Food Chem. X 2023, 19, 100808. [Google Scholar] [CrossRef] [PubMed]
- National Research Council; Policy, Global Affairs, Board on Science; Technology for International Development and Ad Hoc Panel of the Advisory Committee on Technology Innovation. Lost Crops of the Incas: Little-Known Plants of the Andes with Promise for Worldwide Cultivation; National Academies Press: Washington, DC, USA, 1989; ISBN 978-0-309-04264-2. [Google Scholar]
- Nowak, V.; Du, J.; Charrondière, U.R. Assessment of the Nutritional Composition of Quinoa (Chenopodium quinoa Willd.). Food Chem. 2016, 193, 47–54. [Google Scholar] [CrossRef]
- Repo-Carrasco, R.; Espinoza, C.; Jacobsen, S.-E. Nutritional Value and Use of the Andean Crops Quinoa (Chenopodium quinoa) and Kañiwa (Chenopodium pallidicaule). Food Rev. Int. 2003, 19, 179–189. [Google Scholar] [CrossRef]
- Bielecka, J.; Markiewicz-Żukowska, R.; Puścion-Jakubik, A.; Grabia, M.; Nowakowski, P.; Soroczyńska, J.; Socha, K. Gluten-Free Cereals and Pseudocereals as a Potential Source of Exposure to Toxic Elements among Polish Residents. Nutrients 2022, 14, 2342. [Google Scholar] [CrossRef]
- Pathan, S.; Siddiqui, R.A. Nutritional Composition and Bioactive Components in Quinoa (Chenopodium quinoa Willd.) Greens: A Review. Nutrients 2022, 14, 558. [Google Scholar] [CrossRef]
- Kozioł, M.J. Chemical Composition and Nutritional Evaluation of Quinoa (Chenopodium quinoa Willd.). J. Food Compos. Anal. 1992, 5, 35–68. [Google Scholar] [CrossRef]
- Abugoch James, L.E. Chapter 1 Quinoa (Chenopodium quinoa Willd.). In Advances in Food and Nutrition Research; Academic Press: Cambridge, MA, USA, 2009; Volume 58, pp. 1–31. [Google Scholar]
- Pereira, E.; Encina-Zelada, C.; Barros, L.; Gonzales-Barron, U.; Cadavez, V.; Ferreira, I.C. Chemical and Nutritional Characterization of Chenopodium quinoa Willd (Quinoa) Grains: A Good Alternative to Nutritious Food. Food Chem. 2019, 280, 110–114. [Google Scholar] [CrossRef]
- Kumpun, S.; Maria, A.; Crouzet, S.; Evrard-Todeschi, N.; Girault, J.-P.; Lafont, R. Ecdysteroids from Chenopodium quinoa Willd., an Ancient Andean Crop of High Nutritional Value. Food Chem. 2011, 125, 1226–1234. [Google Scholar] [CrossRef]
- Zhu, N.; Kikuzaki, H.; Vastano, B.C.; Nakatani, N.; Karwe, M.V.; Rosen, R.T.; Ho, C.-T. Ecdysteroids of Quinoa Seeds (Chenopodium quinoa Willd.). J. Agric. Food Chem. 2001, 49, 2576–2578. [Google Scholar] [CrossRef] [PubMed]
- Chaudhary, N.; Walia, S.; Kumar, R. Functional Composition, Physiological Effect and Agronomy of Future Food Quinoa (Chenopodium quinoa Willd.): A Review. J. Food Compos. Anal. 2023, 118, 105192. [Google Scholar] [CrossRef]
- Ren, G.; Teng, C.; Fan, X.; Guo, S.; Zhao, G.; Zhang, L.; Liang, Z.; Qin, P. Nutrient Composition, Functional Activity and Industrial Applications of Quinoa (Chenopodium quinoa Willd.). Food Chem. 2023, 410, 135290. [Google Scholar] [CrossRef]
- Vilcacundo, R.; Hernández-Ledesma, B. Nutritional and Biological Value of Quinoa (Chenopodium quinoa Willd.). Curr. Opin. Food Sci. 2017, 14, 1–6. [Google Scholar] [CrossRef]
- Repo-Carrasco-Valencia, R.; Vidaurre-Ruiz, J.M. Bioactive Compounds in Quinoa (Chenopodium quinoa) and Kañiwa (Chenopodium pallidicaule). In Biology and Biotechnology of Quinoa: Super Grain for Food Security; Varma, A., Ed.; Springer: Singapore, 2021; pp. 243–264. ISBN 9789811638329. [Google Scholar]
- Graf, B.L.; Kamat, S.; Cheong, K.Y.; Komarnytsky, S.; Driscoll, M.; Di, R. Phytoecdysteroid-Enriched Quinoa Seed Leachate Enhances Healthspan and Mitochondrial Metabolism in Caenorhabditis elegans. J. Funct. Foods 2017, 37, 1–7. [Google Scholar] [CrossRef]
- Angeli, V.; Miguel Silva, P.; Crispim Massuela, D.; Khan, M.W.; Hamar, A.; Khajehei, F.; Graeff-Hönninger, S.; Piatti, C. Quinoa (Chenopodium quinoa Willd.): An Overview of the Potentials of the “Golden Grain” and Socio-Economic and Environmental Aspects of Its Cultivation and Marketization. Foods 2020, 9, 216. [Google Scholar] [CrossRef] [PubMed]
- Bastidas, E.G.; Roura, R.; Rizzolo, D.a.D.; Massanés, T.; Gomis, R. Quinoa (Chenopodium quinoa Willd), from Nutritional Value to Potential Health Benefits: An Integrative Review. J. Nutr. Food Sci. 2016, 6, 3. [Google Scholar]
- Zevallos, V.F.; Herencia, I.L.; Chang, F.; Donnelly, S.; Ellis, J.H.; Ciclitira, P.J. Gastrointestinal Effects of Eating Quinoa (Chenopodium quinoa Willd.) in Celiac Patients. Off. J. Am. Coll. Gastroenterol. ACG 2014, 109, 270–278. [Google Scholar] [CrossRef]
- Vega-Gálvez, A.; Miranda, M.; Vergara, J.; Uribe, E.; Puente, L.; Martínez, E.A. Nutrition Facts and Functional Potential of Quinoa (Chenopodium quinoa Willd.), an Ancient Andean Grain: A Review. J. Sci. Food Agric. 2010, 90, 2541–2547. [Google Scholar] [CrossRef] [PubMed]
- Meneguetti, Q.A.; Brenzan, M.A.; Batista, M.R.; Bazotte, R.B.; Silva, D.R.; Garcia Cortez, D.A. Biological Effects of Hydrolyzed Quinoa Extract from Seeds of Chenopodium quinoa Willd. J. Med. Food 2011, 14, 653–657. [Google Scholar] [CrossRef] [PubMed]
- Zeyneb, H.; Pei, H.; Cao, X.; Wang, Y.; Win, Y.; Gong, L. In Vitro Study of the Effect of Quinoa and Quinoa Polysaccharides on Human Gut Microbiota. Food Sci. Nutr. 2021, 9, 5735–5745. [Google Scholar] [CrossRef] [PubMed]
- Gawlik-Dziki, U.; Świeca, M.; Sułkowski, M.; Dziki, D.; Baraniak, B.; Czyż, J. Antioxidant and Anticancer Activities of Chenopodium Quinoa Leaves Extracts—In Vitro Study. Food Chem. Toxicol. 2013, 57, 154–160. [Google Scholar] [CrossRef] [PubMed]
- Alvarez-Jubete, L.; Wijngaard, H.; Arendt, E.K.; Gallagher, E. Polyphenol Composition and in Vitro Antioxidant Activity of Amaranth, Quinoa Buckwheat and Wheat as Affected by Sprouting and Baking. Food Chem. 2010, 119, 770–778. [Google Scholar] [CrossRef]
- Ren, G.; Zhu, Y.; Shi, Z.; Li, J. Detection of Lunasin in Quinoa (Chenopodium quinoa Willd.) and the in vitro Evaluation of Its Antioxidant and Anti-Inflammatory Activities. J. Sci. Food Agric. 2017, 97, 4110–4116. [Google Scholar] [CrossRef] [PubMed]
- Tan, M.; Zhao, Q.; Zhao, B. Physicochemical Properties, Structural Characterization and Biological Activities of Polysaccharides from Quinoa (Chenopodium quinoa Willd.) Seeds. Int. J. Biol. Macromol. 2021, 193, 1635–1644. [Google Scholar] [CrossRef]
- Nsimba, R.Y.; Kikuzaki, H.; Konishi, Y. Ecdysteroids Act as Inhibitors of Calf Skin Collagenase and Oxidative Stress. J. Biochem. Mol. Toxicol. 2008, 22, 240–250. [Google Scholar] [CrossRef] [PubMed]
- Alarcón, M.; Bustos, M.; Mendez, D.; Fuentes, E.; Palomo, I.; Lutz, M. In Vitro Assay of Quinoa (Chenopodium quinoa Willd.) and Lupin (Lupinus spp.) Extracts on Human Platelet Aggregation. Plant Foods Hum. Nutr. 2020, 75, 215–222. [Google Scholar] [CrossRef]
- Foucault, A.-S.; Mathe, V.; Lafont, R.; Even, P.; Dioh, W.; Veillet, S.; Tome, D.; Huneau, J.-F.; Hermier, D.; Quignard-Boulange, A. Quinoa Extract Enriched in 20-Hydroxyecdysone Protects Mice From Diet-Induced Obesity and Modulates Adipokines Expression. Obesity 2012, 20, 270–277. [Google Scholar] [CrossRef]
- Sidorova, Y.S.; Shipelin, V.A.; Petrov, N.A.; Zorin, S.N.; Mazo, V.K. Anxiolytic and Antioxidant Effect of Phytoecdysteroids and Polyphenols from Chenopodium quinoa on an in vivo Restraint Stress Model. Molecules 2022, 27, 9003. [Google Scholar] [CrossRef]
- Graf, B.L.; Poulev, A.; Kuhn, P.; Grace, M.H.; Lila, M.A.; Raskin, I. Quinoa Seeds Leach Phytoecdysteroids and Other Compounds with Anti-Diabetic Properties. Food Chem. 2014, 163, 178–185. [Google Scholar] [CrossRef]
- Foucault, A.-S.; Even, P.; Lafont, R.; Dioh, W.; Veillet, S.; Tomé, D.; Huneau, J.-F.; Hermier, D.; Quignard-Boulangé, A. Quinoa Extract Enriched in 20-Hydroxyecdysone Affects Energy Homeostasis and Intestinal Fat Absorption in Mice Fed a High-Fat Diet. Physiol. Behav. 2014, 128, 226–231. [Google Scholar] [CrossRef]
- Omran, N.H.; El-Bahy, A.A.Z.; Hosny, H.T.A.; Handoussa, H. Quinoa and Chia Modulate AMPK/PPAR-ɣ Signaling in High-Fat Diet–Induced Obesity Rat Model. Rev. Bras. Farmacogn. 2023, 33, 583–594. [Google Scholar] [CrossRef]
- Ballegaard, A.-S.R.; Larsen, J.M.; Rasmussen, P.H.; Untersmayr, E.; Pilegaard, K.; Bøgh, K.L. Quinoa (Chenopodium quinoa Willd.) Seeds Increase Intestinal Protein Uptake. Mol. Nutr. Food Res. 2021, 65, e2100102. [Google Scholar] [CrossRef] [PubMed]
- Al-Qabba, M.M.; El-Mowafy, M.A.; Althwab, S.A.; Alfheeaid, H.A.; Aljutaily, T.; Barakat, H. Phenolic Profile, Antioxidant Activity, and Ameliorating Efficacy of Chenopodium quinoa Sprouts against CCl4-Induced Oxidative Stress in Rats. Nutrients 2020, 12, 2904. [Google Scholar] [CrossRef]
- Sláma, K.; Abubakirov, N.K.; Gorovits, M.B.; Baltaev, U.A.; Saatov, Z. Hormonal Activity of Ecdysteroids from Certain Asiatic Plants. Insect Biochem. Mol. Biol. 1993, 23, 181–185. [Google Scholar] [CrossRef]
- Guibout, L.; Mamadalieva, N.; Balducci, C.; Girault, J.-P.; Lafont, R. The Minor Ecdysteroids from Ajuga Turkestanica. Phytochem. Anal. 2015, 26, 293–300. [Google Scholar] [CrossRef]
- Janeczko, A.; Oklestkova, J.; Tarkowská, D.; Drygaś, B. Naturally Occurring Ecdysteroids in Triticum aestivum L. and Evaluation of Fenarimol as a Potential Inhibitor of Their Biosynthesis in Plants. Int. J. Mol. Sci. 2021, 22, 2855. [Google Scholar] [CrossRef]
- Suksamrarn, A.; Kumpun, S.; Yingyongnarongkul, B.-E. Ecdysteroids of Vitex Scabra Stem Bark. J. Nat. Prod. 2002, 65, 1690–1692. [Google Scholar] [CrossRef]
- Mamatkhanov, A.U.; Yakubova, M.R.; Syrov, V.N. Isolation of Turkesterone from the Epigeal Part of Ajuga Turkestanica and Its Anabolic Activity. Chem. Nat. Compd. 1998, 34, 150–154. [Google Scholar] [CrossRef]
- Syrov, V.N.; Saatov, Z.; Sagdullaev, S.S.; Mamatkhanov, A.U. Study of the Structure—Anabolic Activity Relationship for Phytoecdysteroids Extracted from Some Plants of Central Asia. Pharm. Chem. J. 2001, 35, 667–671. [Google Scholar] [CrossRef]
- Abdukadirov, I.T.; Yakubova, M.R.; Nuriddinov, K.R.; Mamatkhanov, A.U.; Turakhozhaev, M.T. Ecdysterone and Turkesterone in Ajuga Turkestanica Determined by HPLC. Chem. Nat. Compd. 2005, 41, 475–476. [Google Scholar] [CrossRef]
- Abdukadirov, I.T.; Khodzhaeva, M.A.; Turakhozhaev, M.T.; Mamatkhanov, A.U. Carbohydrates from Ajuga Turkestanica. Chem. Nat. Compd. 2004, 40, 85–86. [Google Scholar] [CrossRef]
- Syrov, V.N.; Khushbaktova, Z.A.; Tolibaev, I.; Eletskaya, N.V.; Mamatkhanov, A.U. Effect of a Lipid Concentrate from the Aboveground Portion of Ajuga Turkestanica on the Metabolic Processes and Dynamics of Healing Skin Wounds Experimentally. Pharm. Chem. J. 1994, 28, 837–840. [Google Scholar] [CrossRef]
- Dinan, L.; Harmatha, J.; Volodin, V.; Lafont, R. Phytoecdysteroids: Diversity, Biosynthesis and Distribution. In Ecdysone: Structures and Functions; Smagghe, G., Ed.; Springer: Dordrecht, The Netherlands, 2009; pp. 3–45. ISBN 978-1-4020-9112-4. [Google Scholar]
- Kuchkarova, L.S.; Rokhimova, S.O.; Syrov, V.N. Effect of Turkesterone on the Pancreas Histology and Function in Diabetic Rats. Int. J. Curr. Res. Rev. 2020, 12, 2–5. [Google Scholar] [CrossRef]
- Arthur, S.T.; Zwetsloot, K.A.; Lawrence, M.M.; Nieman, D.C.; Lila, M.A.; Grace, M.H.; Howden, R.; Cooley, I.D.; Tkach, J.F.; Keith, M.D.; et al. Ajuga Turkestanica Increases Notch and Wnt Signaling in Aged Skeletal Muscle. Eur. Rev. Med. Pharmacol. Sci. 2014, 18, 2584–2592. [Google Scholar]
- Kutepova, T.A.; Syrov, V.N.; Khushbaktova, Z.A.; Saatov, Z. Hypoglycemic Activity of the Total Ecdysteroid Extract from Ajuga turkestanica. Pharm. Chem. J. 2001, 35, 608–609. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).