Functional Foods, a Hope to Delay Muscle Dystrophy Progression: A Potential Role for Omega Fatty Acids
Abstract
1. Muscular Dystrophies
2. Common Features of MD
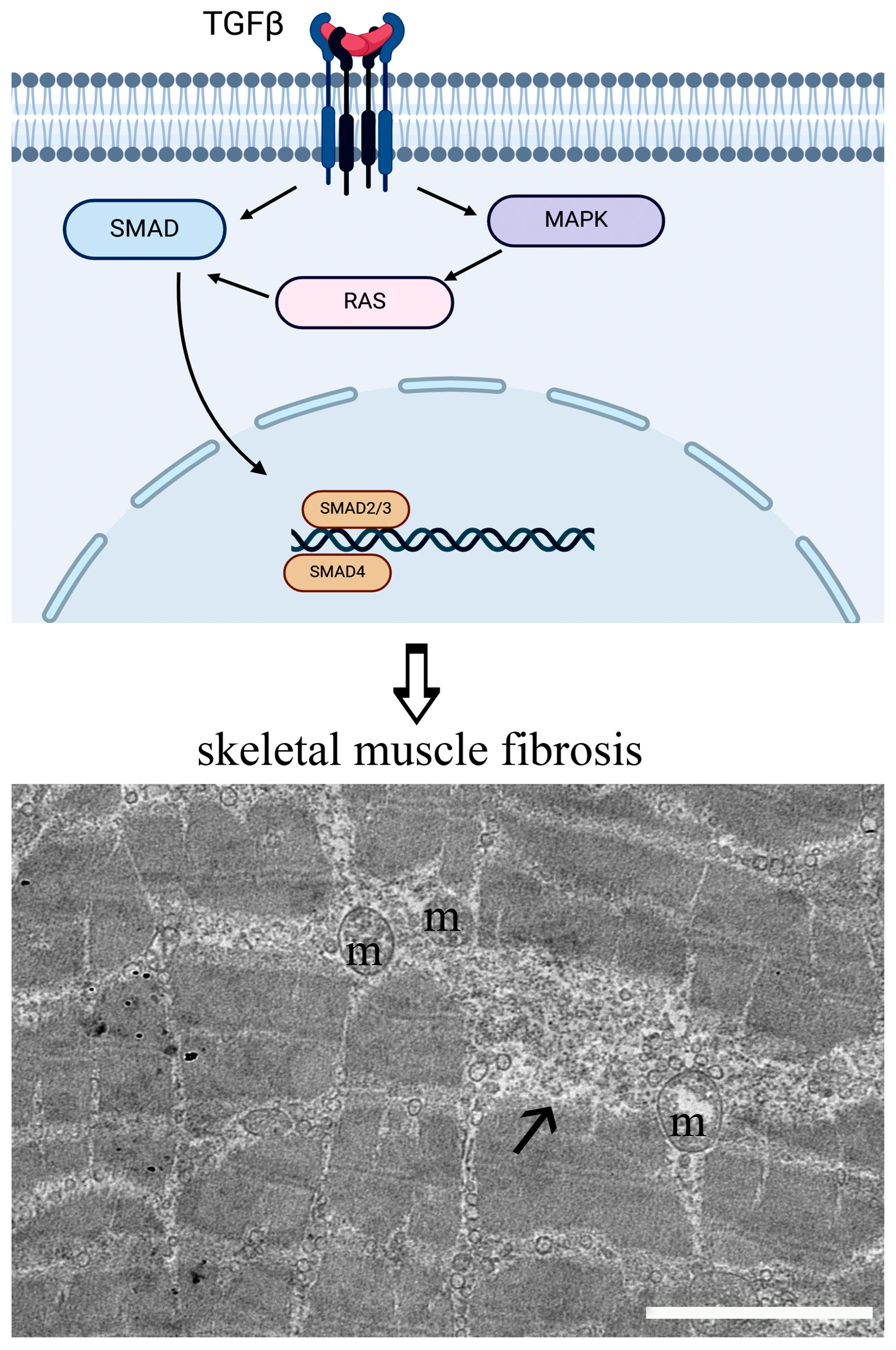
2.1. Fibrosis
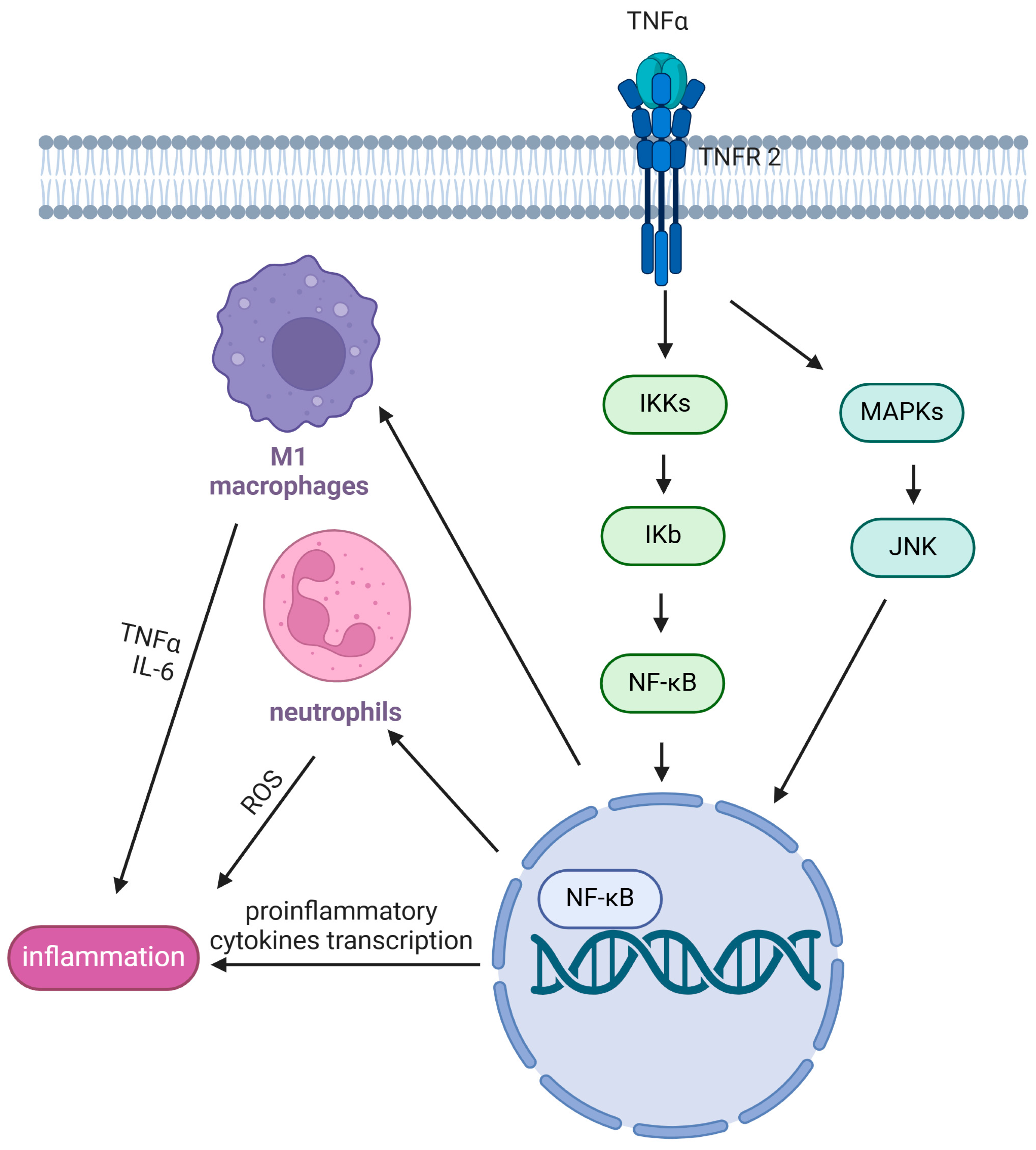
2.2. Inflammation and Oxidative Stress
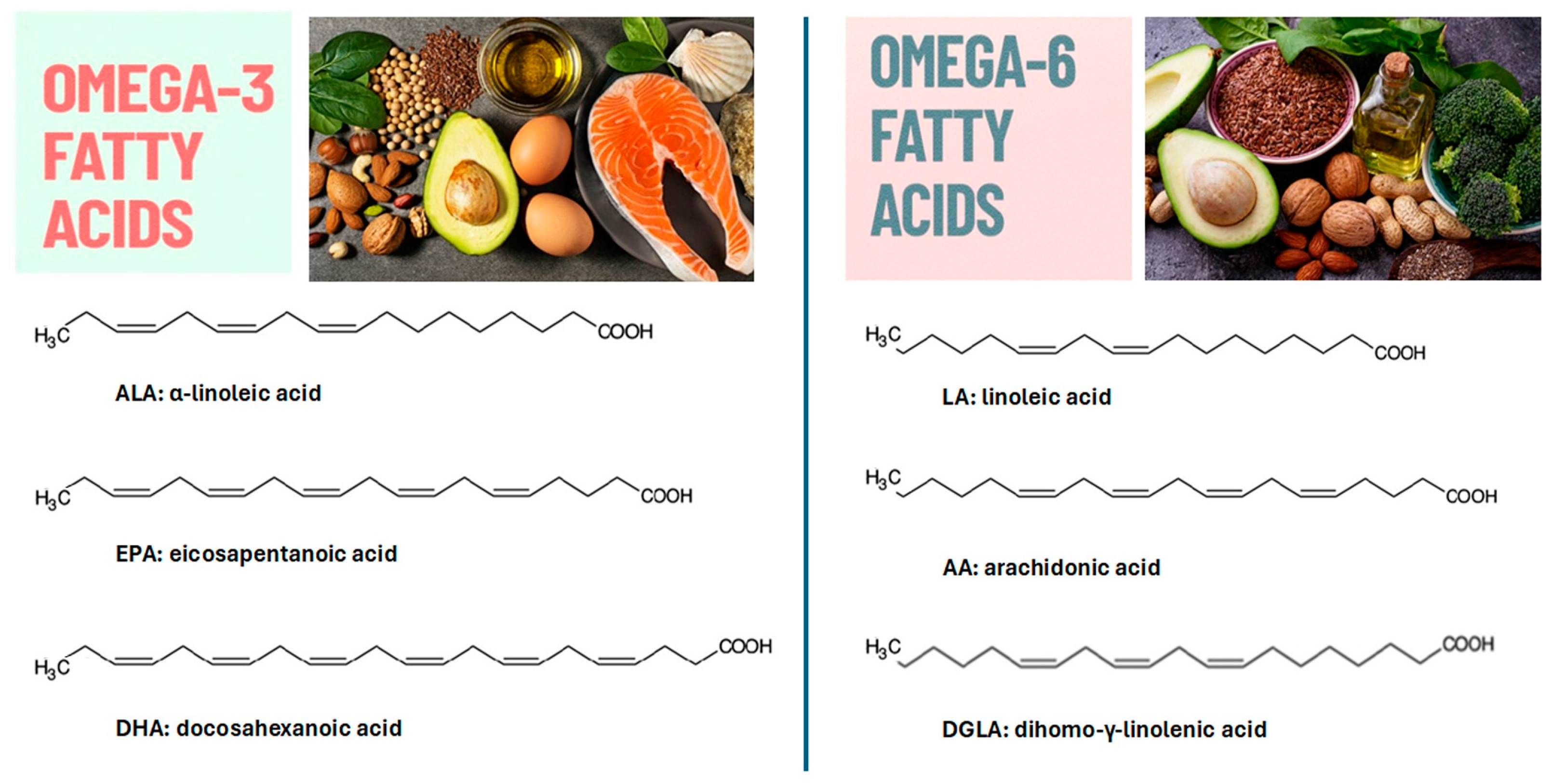
3. Omega Fatty Acids
4. Omega Fatty Acids and Dystrophies
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- LaPelusa, A.; Asuncion, R.M.D.; Kentris, M. Muscular Dystrophy. In StatPearls [Internet]; StatPearls Publishing: Treasure Island, FL, USA, 2025. Available online: https://www.ncbi.nlm.nih.gov/sites/books/NBK560582/ (accessed on 26 February 2024).
- Colapicchioni, V.; Millozzi, F.; Parolini, O.; Palacios, D. Nanomedicine, a valuable tool for skeletal muscle disorders: Challenges, promises, and limitations. Wiley Interdiscip. Rev. Nanomed. Nanobiotechnol. 2022, 14, e1777. [Google Scholar] [CrossRef] [PubMed]
- Bushby, K.M.; Collins, J.; Hicks, D. Collagen type VI myopathies. Adv. Exp. Med. Biol. 2014, 802, 185–199. [Google Scholar]
- Waldrop, M.A.; Flanigan, K.M. Update in Duchenne and Becker muscular dystrophy. Curr. Opin. Neurol. 2019, 32, 722–727. [Google Scholar] [CrossRef] [PubMed]
- Brisson, J.D.; Gagnon, C.; Brais, B.; Côté, I.; Mathieu, J. A study of impairments in oculopharyngeal muscular dystrophy. Muscle Nerve 2020, 62, 201–207. [Google Scholar] [CrossRef] [PubMed]
- Felice, K.J. Distal Myopathies. Neurol. Clin. 2020, 38, 637–659. [Google Scholar] [CrossRef]
- Ghasemi, M.; Emerson, C.P., Jr.; Hayward, L.J. Outcome Measures in Facioscapulohumeral Muscular Dystrophy Clinical Trials. Cells 2022, 11, 687. [Google Scholar] [CrossRef]
- Johnson, N.E.; Statland, J.M. The Limb-Girdle Muscular Dystrophies. Continuum 2022, 28, 1698–1714. [Google Scholar]
- Takamizawa, K.; Kim, K.S.; Ueda, H. Emery-Dreifuss muscular dystrophy with dilated cardiomyopathy preceding skeletal muscle symptoms. Cardiol. Young 2022, 32, 1175–1177. [Google Scholar] [CrossRef]
- Timchenko, L. Myotonic Dystrophy: From Molecular Pathogenesis to Therapeutics. Int. J. Mol. Sci. 2022, 23, 11954. [Google Scholar] [CrossRef]
- Angelliaume, A.; Harper, L.; Lalioui, A.; Delgove, A.; Lefèvre, Y. Tailor-made management of thoracic scoliosis with cervical hyperextension in muscular dystrophy. Eur. Spine J. 2018, 27, 264–269. [Google Scholar] [CrossRef]
- Erkut, E.; Yokota, T. Therapeutics for Duchenne Muscular Dystrophy. Int. J. Mol. Sci. 2022, 23, 1832. [Google Scholar] [CrossRef]
- Soltanzadeh, P. Myotonic Dystrophies: A Genetic Overview. Genes 2022, 13, 367. [Google Scholar] [CrossRef]
- Mercuri, E.; Bonnemann, C.G.; Muntoni, F. Muscular dystrophies. Lancet 2019, 394, 2025–2038. [Google Scholar] [CrossRef] [PubMed]
- Duan, D.; Goemans, N.; Takeda, S.; Mercuri, E.; Aartsma-Rus, A. Duchenne muscular dystrophy. Nat. Rev. Dis. Primers 2021, 7, 13. [Google Scholar] [CrossRef]
- Elangkovan, N.; Dickson, G. Gene Therapy for Duchenne Muscular Dystrophy. J. Neuromuscul. Dis. 2021, 8, S303–S316. [Google Scholar] [CrossRef] [PubMed]
- Faenza, I.; Blalock, W.; Bavelloni, A.; Schoser, B.; Fiume, R.; Pacella, S.; Piazzi, M.; D’Angelo, A.; Cocco, L. A role for PLCβ1 in myotonic dystrophies type 1 and 2. FASEB J. 2012, 26, 3042–3048. [Google Scholar] [CrossRef]
- Liao, Q.; Zhang, Y.; He, J.; Huang, K. Global Prevalence of Myotonic Dystrophy: An Updated Systematic Review and Meta-Analysis. Neuroepidemiology 2022, 56, 163–173. [Google Scholar] [CrossRef] [PubMed]
- Pandey, S.N.; Kesari, A.; Yokota, T.; Pandey, G.S. Muscular Dystrophy: Disease Mechanisms and Therapies. Biomed. Res. Int. 2015, 2015, 456348. [Google Scholar] [CrossRef]
- Nikolić-Kokić, A.; Marinković, D.; Perić, S.; Stević, Z.; Spasić, M.B.; Blagojević, D.; Rakocˇević-Stojanović, V. Redox imbalance in peripheral blood of type 1 myotonic dystrophy patients. Redox Rep. 2016, 21, 232–237. [Google Scholar] [CrossRef]
- Koc, F.; Atli, G.; Menziletoglu, S.Y.; Kose, S. Antioxidant imbalance in the erythrocytes of Myotonic dystrophy Type 1 patients. Arch. Biochem. Biophys. 2020, 680, 108230. [Google Scholar] [CrossRef]
- Markati, T.; Oskoui, M.; Farrar, M.A.; Duong, T.; Goemans, N.; Servais, L. Emerging therapies for Duchenne muscular dystrophy. Lancet Neurol. 2022, 21, 814–829. [Google Scholar] [CrossRef] [PubMed]
- Quattrocelli, M.; Zelikovich, A.S.; Salamone, I.M.; Fischer, J.A.; McNally, E.M. Mechanisms and Clinical Applications of Glucocorticoid Steroids in Muscular Dystrophy. J. Neuromuscul. Dis. 2021, 8, 39–52. [Google Scholar] [CrossRef]
- Aronson, J.K. Defining ‘nutraceuticals’, neither nutritious nor pharmaceutical. Br. J. Clin. Pharmacol. 2017, 83, 8–19. [Google Scholar] [CrossRef] [PubMed]
- Salucci, S.; Burattini, S.; Giordano, F.M.; Lucarini, S.; Diamantini, G.; Falcieri, E. Further Highlighting on the Prevention of Oxidative Damage by Polyphenol-Rich Wine Extracts. J. Med. Food 2017, 20, 410–419. [Google Scholar] [CrossRef]
- Salucci, S.; Bartoletti-Stella, A.; Bavelloni, A.; Aramini, B.; Blalock, W.L.; Fabbri, F.; Vannini, I.; Sambri, V.; Stella, F.; Faenza, I. Extra Virgin Olive Oil (EVOO), a Mediterranean Diet Component, in the Management of Muscle Mass and Function Preservation. Nutrients 2022, 14, 3567. [Google Scholar] [CrossRef]
- González-Jamett, A.; Vásquez, W.; Cifuentes-Riveros, G.; Martínez-Pando, R.; Sáez, J.C.; Cárdenas, A.M. Oxidative Stress, Inflammation and Connexin Hemichannels in Muscular Dystrophies. Biomedicines 2022, 10, 507. [Google Scholar] [CrossRef]
- Mahdy, M.A.A. Skeletal muscle fibrosis: An overview. Cell Tissue Res. 2019, 375, 575–588. [Google Scholar] [CrossRef]
- Dridi, H.; Wu, W.; Reiken, S.R.; Ofer, R.M.; Liu, Y.; Yuan, Q.; Sittenfeld, L.; Kushner, J.; Muchir, A.; Worman, H.J.; et al. Ryanodine receptor remodeling in cardiomyopathy and muscular dystrophy caused by lamin A/C gene mutation. Hum. Mol. Genet. 2021, 29, 3919–3934. [Google Scholar] [CrossRef] [PubMed]
- Deconinck, N.; Richard, P.; Allamand, V.; Behin, A.; Laforêt, P.; Ferreiro, A.; De Becdelievre, A.; Ledeuil, C.; Gartioux, C.; Nelson, I.; et al. Bethlem myopathy: Long-term follow-up identifies COL6 mutations predicting severe clinical evolution. J. Neurol. Neurosurg. Psychiatry 2015, 86, 1337–1346. [Google Scholar] [CrossRef]
- Fan, Y.; Liu, A.; Wei, C.; Yang, H.; Chang, X.; Wang, S.; Yuan, Y.; Bonnemann, C.; Wu, Q.; Wu, X.; et al. Genetic and clinical findings in a Chinese cohort of patients with collagen VI-related myopathies. Clin. Genet. 2018, 93, 1159–1171. [Google Scholar] [CrossRef]
- Inoue, M.; Saito, Y.; Yonekawa, T.; Ogawa, M.; Iida, A.; Nishino, I.; Noguchi, S. Causative variant profile of collagen VI-related dystrophy in Japan Orphanet. J. Rare Dis. 2021, 16, 284. [Google Scholar]
- Smith, L.R.; Barton, E.R. Regulation of fibrosis in muscular dystrophy. Matrix Biol. 2018, 68–69, 602–615. [Google Scholar] [CrossRef] [PubMed]
- Harish, P.; Forrest, L.; Herath, S.; Dickson, G.; Malerba, A.; Popplewell, L. Inhibition of Myostatin Reduces Collagen Deposition in a Mouse Model of Oculopharyngeal Muscular Dystrophy (OPMD) With Established Disease. Front. Physiol. 2020, 11, 184. [Google Scholar] [CrossRef]
- Parker, E.; Hamrick, M.W. Role of fibro-adipogenic progenitor cells in muscle atrophy and musculoskeletal diseases. Curr. Opin. Pharmacol. 2021, 58, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Perandini, L.A.; Chimin, P.; da Lutkemeyer, D.S.; Câmara, N.O.S. Chronic inflammation in skeletal muscle impairs satellite cells function during regeneration: Can physical exercise restore the satellite cell niche? FEBS J. 2018, 285, 1973–1984. [Google Scholar] [CrossRef]
- Phelps, M.; Stuelsatz, P.; Yablonka-Reuveni, Z. Expression profile and overexpression outcome indicate a role for βKlotho in skeletal muscle fibro/adipogenesis. FEBS J. 2016, 283, 1653–1668. [Google Scholar] [CrossRef] [PubMed]
- Fernández-Simón, E.; Suárez-Calvet, X.; Carrasco-Rozas, A.; Piñol-Jurado, P.; López-Fernández, S.; Pons, G.; Bech Serra, J.J.; de la Torre, C.; de Luna, N.; Gallardo, E.; et al. RhoA/ROCK2 signalling is enhanced by PDGF-AA in fibro-adipogenic progenitor cells: Implications for Duchenne muscular dystrophy. Cachexia Sarcopenia Muscle 2022, 3, 1373–1384. [Google Scholar] [CrossRef]
- Lemos, D.R.; Babaeijandaghi, F.; Low, M.; Chang, C.K.; Lee, S.T.; Fiore, D.; Zhang, R.H.; Natarajan, A.; Nedospasov, S.A.; Rossi, F.M.V. Nilotinib reduces muscle fibrosis in chronic muscle injury by promoting TNF-mediated apoptosis of fibro/adipogenic progenitors. Nat. Med. 2015, 21, 786–794. [Google Scholar] [CrossRef]
- Ieronimakis, N.; Hays, A.; Prasad, A.; Janebodin, K.; Duffield, J.S.; Reyes, M. PDGFRα signalling promotes fibrogenic responses in collagen-producing cells in Duchenne muscular dystrophy. J. Pathol. 2016, 240, 410–424. [Google Scholar] [CrossRef]
- Piñol-Jurado, P.; Suárez-Calvet, X.; Fernández-Simón, E.; Gallardo, E.; De La Oliva, N.; Martínez-Muriana, A.; Gómez-Gálvez, P.; Escudero, L.M.; Pérez-Peiró, M.; Wollin, L.; et al. Nintedanib decreases muscle fibrosis and improves muscle function in a murine model of dystrophinopathy. Cell Death Dis. 2018, 9, 776. [Google Scholar] [CrossRef]
- Beaudry, K.M.; Binet, E.R.; Collao, N.; De Lisio, M. Nutritional Regulation of Muscle Stem Cells in Exercise and Disease: The Role of Protein and Amino Acid Dietary Supplementation. Front. Physiol. 2022, 13, 915390. [Google Scholar] [CrossRef] [PubMed]
- Villalta, S.A.; Rosenberg, A.S.; Bluestone, J.A. The immune system in Duchenne muscular dystrophy: Friend or foe. Rare Dis. 2015, 3, e1010966. [Google Scholar] [CrossRef]
- Tidball, J.G.; Welc, S.S.; Wehling-Henricks, M. Immunobiology of inherited muscular dystrophies. Compr. Physiol. 2018, 8, 1313–1356. [Google Scholar] [PubMed]
- Herbelet, S.; Rodenbach, A.; Paepe, B.D.; De Bleecker, J.L. Anti-Inflammatory and general glucocorticoid physiology in skeletal muscles affected by Duchenne muscular dystrophy: Exploration of steroid-sparing agents. Int. J. Mol. Sci. 2020, 21, 4596. [Google Scholar] [CrossRef]
- Yin, X.; Tang, Y.; Li, J.; Dzuricky, A.T.; Pu, C.; Fu, F.; Wang, B. Genetic ablation of P65 subunit of NF-κB in mdx mice to improve muscle physiological function. Muscle Nerve 2017, 56, 759–767. [Google Scholar] [CrossRef] [PubMed]
- Hightower, R.M.; Reid, A.L.; Gibbs, D.E.; Wang, Y.; Widrick, J.J.; Kunkel, L.M.; Kastenschmidt, J.M.; Villalta, S.A.; van Groen, T.; Chang, H. The SINE Compound KPT-350 Blocks Dystrophic Pathologies in DMD Zebrafish and Mice. Mol. Ther. 2020, 28, 189–201. [Google Scholar] [CrossRef]
- Ballmann, C.; Denney, T.S.; Beyers, R.J.; Quindry, T.; Romero, M.; Amin, R.; Selsby, J.T.; Quindry, J.C. Lifelong quercetin enrichment and cardioprotection in Mdx/Utrn+/- mice. Am. J. Physiol. Heart Circ. Physiol. 2017, 312, H128–H140. [Google Scholar] [CrossRef]
- Suntar, I.; Sureda, A.; Belwal, T.; Sanches Silva, A.; Vacca, R.A.; Tewari, D.; Sobarzo-Sánchez, E.; Nabavi, S.F.; Shirooie, S.; Dehpour, A.R.; et al. Natural products, PGC-1 α, and Duchenne muscular dystrophy. Acta Pharm. Sin. B 2020, 10, 734–745. [Google Scholar] [CrossRef]
- Rowe, G.C.; Raghuram, S.; Jang, C.; Nagy, J.; Patten, I.S.; Goyal, A.; Chun Chan, M.; Liu, L.X.; Jiang, A.; Spokes, K.C.; et al. PGC-1α induces SPP1 to activate macrophages and orchestrate functional angiogenesis in skeletal muscle. Circ. Res. 2014, 115, 504–517. [Google Scholar] [CrossRef]
- Dort, J.; Fabre, P.; Molina, T.; Dumont, N.A. Macrophages are key regulators of stem cells during skeletal muscle regeneration and diseases. Stem Cells Int. 2019, 2019, 4761427. [Google Scholar] [CrossRef]
- Ribeiro, A.F.; Souza, L.S.; Almeida, C.F.; Ishiba, R.; Fernandes, S.A.; Guerrieri, D.A.; Santos, A.L.F.; Onofre-Oliveira, P.C.G.; Vainzof, M. Muscle satellite cells and impaired late stage regeneration in different murine models for muscular dystrophies. Sci. Rep. 2019, 9, 11842. [Google Scholar] [CrossRef] [PubMed]
- Mancio, R.D.; Hermes, T.A.; Macedo, A.B.; Mizobuti, D.S.; Rupcic, I.F.; Minatel, E. Dystrophic phenotype improvement in the diaphragm muscle of mdx mice by diacerhein. PLoS ONE 2017, 12, e0182449. [Google Scholar] [CrossRef] [PubMed]
- Tulangekar, A.; Sztal, T.E. Inflammation in Duchenne Muscular Dystrophy-Exploring the Role of Neutrophils in Muscle Damage and Regeneration. Biomedicines 2021, 9, 1366. [Google Scholar] [CrossRef] [PubMed]
- Terrill, J.R.; Duong, M.N.; Turner, R.; Le Guiner, C.; Boyatzis, A.; Kettle, A.J.; Grounds, M.D.; Arthur, P.G. Levels of inflammation and oxidative stress, and a role for taurine in dystropathology of the Golden Retriever Muscular Dystrophy dog model for Duchenne muscular dystrophy. Redox Biol. 2016, 9, 276–286. [Google Scholar] [CrossRef]
- Grounds, M.D.; Terrill, J.R.; Al-Mshhdani, B.A.; Duong, M.N.; Radley-Crabb, H.G.; Arthur, P.G. Biomarkers for Duchenne muscular dystrophy: Myonecrosis, inflammation and oxidative stress. Dis. Model. Mech. 2020, 13, dmm043638. [Google Scholar] [CrossRef]
- Budzinska, M.; Zimna, A.; Kurpisz, M. The role of mitochondria in Duchenne muscular dystrophy. J. Physiol. Pharmacol. 2021, 72, 157–166. [Google Scholar]
- Salucci, S.; Baldassarri, V.; Canonico, B.; Burattini, S.; Battistelli, M.; Guescini, M.; Papa, S.; Stocchi, V.; Falcieri, E. Melatonin behavior in restoring chemical damaged C2C12 myoblasts. Microsc. Res. Tech. 2016, 79, 532–540. [Google Scholar] [CrossRef]
- Le Moal, E.; Pialoux, V.; Juban, G.; Groussard, C.; Zouhal, H.; Chazaud, B.; Mounier, R. Redox Control of Skeletal Muscle Regeneration. Antioxid. Redox Signal. 2017, 27, 276–310. [Google Scholar] [CrossRef]
- Mosca, N.; Petrillo, S.; Bortolani, S.; Monforte, M.; Ricci, E.; Piemonte, F.; Tasca, G. Redox Homeostasis in Muscular Dystrophies. Cells 2021, 10, 1364. [Google Scholar] [CrossRef]
- Kourakis, S.; Timpani, C.A.; de Haan, J.B.; Gueven, N.; Fischer, D.; Rybalka, E. Targeting Nrf2 for the Treatment of Duchenne Muscular Dystrophy. Redox Biol. 2021, 8, 101803. [Google Scholar] [CrossRef]
- Kovac, S.; Angelova, P.R.; Holmström, K.M.; Zhang, Y.; Dinkova-Kostova, A.T.; Abramov, A.Y. Nrf2 Regulates ROS Production by Mitochondria and NADPH Oxidase. Biochim. Biophys. Acta 2015, 1850, 794–801. [Google Scholar] [CrossRef]
- Zhang, M.; Xu, Y.; Qiu, Z.; Jiang, L. Sulforaphane Attenuates Angiotensin II-Induced Vascular Smooth Muscle Cell Migration via Suppression of NOX4/ROS/Nrf2 Signaling. Int. J. Biol. Sci. 2019, 15, 148–157. [Google Scholar] [CrossRef] [PubMed]
- da Silva, H.N.M.; Covatti, C.; da Rocha, G.L.; Mizobuti, D.S.; Mâncio, R.D.; Hermes, T.A.; Kido, L.A.; Cagnon, V.H.A.; Pereira, E.C.L.; Minatel, E. Oxidative Stress, Inflammation, and Activators of Mitochondrial Biogenesis: Tempol Targets in the Diaphragm Muscle of Exercise Trained-mdx Mice. Front. Physiol. 2021, 12, 649793. [Google Scholar] [CrossRef]
- Calder, P.C. n-3 PUFA and inflammation: From membrane to nucleus and from bench to bedside. Proc. Nutr. Soc. 2020, 79, 404–416. [Google Scholar] [CrossRef]
- Calder, P.C. Functional roles of fatty acids and their effects on human health. J. Parenter. Enter. Nutr. 2015, 39, 18S–32S. [Google Scholar] [CrossRef]
- Farag, M.A.; Gad, M.Z. Omega-9 fatty acids: Potential roles in inflammation and cancer management. J. Genet. Eng. Biotechnol. 2022, 20, 48. [Google Scholar] [CrossRef] [PubMed]
- Dunbar, B.S.; Bosire, R.V.; Deckelbaum, R.J. Omega 3 and omega 6 fatty acids in human and animal health: An African perspective. Mol. Cell. Endocrinol. 2014, 398, 69–77. [Google Scholar] [CrossRef] [PubMed]
- Calvo, M.J.; Martínez, M.S.; Torres, W.; Chávez-Castillo, M.; Luzardo, E.; Villasmil, N.; Salazar, J.; Velasco, M.; Bermúdez, M. Omega-3 polyunsaturated fatty acids and cardiovascular health: A molecular view into structure and function. Vessel Plus 2017, 1, 116–128. [Google Scholar] [CrossRef][Green Version]
- Oppedisano, F.; Macrì, R.; Gliozzi, M.; Musolino, V.; Carresi, C.; Maiuolo, J.; Bosco, F.; Nucera, S.; Caterina Zito, M.; Guarnieri, L. The anti-inflammatory and antioxidant properties of n-3 PUFAs: Their role in cardiovascular protection. Biomedicines 2020, 8, 306. [Google Scholar] [CrossRef]
- Shahidi, F.; Ambigaipalan, P. Omega-3 Polyunsaturated Fatty Acids and Their Health Benefits. Annu. Rev. Food Sci. Technol. 2018, 9, 345–381. [Google Scholar] [CrossRef]
- Kimura, I.; Ichimura, A.; Ohue-Kitano, R.; Igarash, M. Free fatty acid receptors in health and disease. Physiol. Rev. 2020, 100, 171–210. [Google Scholar] [CrossRef] [PubMed]
- Kapoor, B.; Kapoor, D.; Gautam, S.; Singh, R.; Bhardwaj, S. Dietary Polyunsaturated Fatty Acids (PUFAs): Uses and Potential Health Benefits. Curr. Nutr. Rep. 2021, 10, 232–242. [Google Scholar] [CrossRef] [PubMed]
- Djuricic, I.; Calder, P.C. Beneficial Outcomes of Omega-6 and Omega-3 Polyunsaturated Fatty Acids on Human Health: An Update for 2021. Nutrients 2021, 13, 2421. [Google Scholar] [CrossRef] [PubMed]
- Gibson, R.A.; Muhlhausler, B.; Makrides, M. Conversion of linoleic acid and alpha-linolenic acid to long-chain polyunsaturated fatty acids (LCPUFAs), with a focus on pregnancy, lactation and the first 2 years of life. Matern. Child. Nutr. 2011, 7, 17–26. [Google Scholar] [CrossRef]
- Vedtofte, M.S.; Jakobsen, M.U.; Lauritzen, L.; Heitmann, B.L. Dietary α-linolenic acid, linoleic acid, and n–3 long-chain PUFA and risk of ischemic heart disease. Am. J. Clin. Nutr. 2011, 94, 1097–1103. [Google Scholar] [CrossRef]
- Liput, K.P.; Lepczyński, A.; Ogłuszka, M.; Nawrocka, A.; Poławska, E.; Grzesiak, A.; Ślaska, B.; Pareek, C.S.; Czarnik, U.; Pierzchała, M. Effects of Dietary n–3 and n–6 Polyunsaturated Fatty Acids in Inflammation and Cancerogenesis. Int. J. Mol. Sci. 2021, 22, 6965. [Google Scholar] [CrossRef]
- Caligiuri, S.P.B.; Parikh, M.; Stamenkovic, A.; Pierce, G.N.; Aukema, H.M. Dietary modulation of oxylipins in cardiovascular disease and aging. Am. J. Physiol. Heart Circ. Physiol. 2017, 313, H903–H918. [Google Scholar] [CrossRef]
- DiNicolantonio, J.; Okeefe, J. Importance of maintaining a low omega-6/omega-3 ratio for reducing platelet aggregation, coagulation and thrombosis. Open Heart 2019, 6, e001011. [Google Scholar] [CrossRef]
- Doenyas-Barak, K.; Berman, S.; Abu-Hamad, R.; Golik, A.; Rahimi-Levene, N.; Efrati, S. N-3 fatty acid supplementation to routine statin treatment inhibits platelet function, decreases patients’ daytime blood pressure, and improves inflammatory status. Eur. J. Clin. Pharmacol. 2012, 68, 1139–1146. [Google Scholar] [CrossRef]
- Kander, T.; Lindblom, E.; Schott, U. Dose-response effects of omega-3 on platelet aggregation: An observational study. J. Int. Med. Res. 2018, 46, 5074–5082. [Google Scholar] [CrossRef]
- Lauritzen, L.; Brambilla, P.; Mazzocchi, A.; Harsløf, L.B.S.; Ciappolino, V.; Agostoni, C. DHA Effects in Brain Development and Function. Nutrients 2016, 8, 6. [Google Scholar] [CrossRef] [PubMed]
- Innes, J.K.; Calder, P.C. Omega-6 fatty acids and inflammation. Prostaglandins Leukot. Essent. Fatty Acids 2018, 132, 41–48. [Google Scholar] [CrossRef] [PubMed]
- Calder, P.C. Omega-3 fatty acids and inflammatory processes: From molecules to man Biochem. Soc. Trans. 2017, 45, 1105–1115. [Google Scholar] [CrossRef]
- Serhan, C.N.; Back, M.; Chiurchiù, V.; Hersberger, M.; Mittendorfer, B.; Calder, P.C.; Waitzberg, D.L.; Stoppe, C.; Klek, S.; Martindale, R.G. Expert consensus report on lipid mediators: Role in resolution of inflammation and muscle preservation. FASEB J. 2024, 38, e23699. [Google Scholar] [CrossRef]
- Bushby, K.; Finkel, R.; Birnkrant, D.J.; Case, L.E.; Clemens, P.R.; Cripe, L.; Kaul, A.; Kinnett, K.; McDonald, C.; Pandya, S.; et al. For the DMD care considerations working group, diagnosis, and management of Duchenne muscular dystrophy: Part 1: Diagnosis, and pharmacological and psychosocial management. Lancet Neurol. 2010, 9, 77–93. [Google Scholar] [CrossRef] [PubMed]
- Krishna, L.; Prashant, A.; Kumar, Y.H.; Paneyala, S.; Patil, S.J.; Ramachandra, S.C.; Vishwanath, P. Molecular and Biochemical Therapeutic Strategies for Duchenne Muscular Dystrophy. Neurol. Int. 2024, 16, 731–760. [Google Scholar] [CrossRef]
- Fogagnolo, M.A.; Minatel, E.; Santo Neto, H.; Marques, M.J. Effects of fish oil containing eicosapentaenoic acid and docosahexaenoic acid on dystrophic mdx mice. Clin. Nutr. 2013, 32, 636–642. [Google Scholar] [CrossRef]
- Villaldama-Soriano, M.A.; Rodríguez-Cruz, M.; Hernández-De la Cruz, S.Y.; Almeida-Becerril, T.; Cárdenas-Conejo, A.; Wong-Baeza, C. Pro-inflammatory monocytes are increased in Duchenne muscular dystrophy and suppressed with omega-3 fatty acids: A double-blind, randomized, placebo-controlled pilot study. Eur. J. Neurol. 2022, 29, 855–864. [Google Scholar] [CrossRef]
- Harwood, J.L. Polyunsaturated Fatty Acids: Conversion to Lipid Mediators, Roles in Inflammatory Diseases and Dietary Sources. Int. J. Mol. Sci. 2023, 24, 8838. [Google Scholar] [CrossRef]
- Apolinário, L.M.; De Carvalho, S.C.; Santo Neto, H.; Marques, M.G. Long-term therapy with omega-3 ameliorates myonecrosis and benefits skeletal muscle regeneration in mdx mice. Anat. Rec. 2015, 298, 1589–1596. [Google Scholar] [CrossRef]
- Fogagnolo Maurício, A.; Pereira, J.A.; Santo Neto, H.; Marques, M.J. Effects of fish oil containing eicosapentaenoic acid and docosahexaenoic acid on dystrophic mdx mice hearts at later stages of dystrophy. Nutrition 2016, 32, 855–862. [Google Scholar] [CrossRef] [PubMed]
- de Carvalho, S.C.; Matsumura, C.Y.; Santo Neto, H.; Marques, M.J. Identification of plasma interleukins as biomarkers for deflazacort and omega-3 based Duchenne muscular dystrophy therapy. Cytokine 2018, 102, 55–61. [Google Scholar] [CrossRef]
- de Carvalho, S.C.; Apolinário, L.M.; Matheus, S.M.; Santo Neto, H.; Marques, M.J. EPA protects against muscle damage in the mdx mouse model of Duchenne muscular dystrophy by promoting a shift from the M1 to M2 macrophage phenotype. J. Neuroimmunol. 2013, 264, 41–47. [Google Scholar] [CrossRef]
- de Carvalho, S.C.; Hindi, S.M.; Kumar, A.; Marques, M.J. Effects of omega-3 on matrix metalloproteinase-9, myoblast transplantation and satellite cell activation in dystrophin-deficient muscle fibers. Cell Tissue Res. 2017, 369, 591–602. [Google Scholar] [CrossRef]
- Tripodi, L.; Molinaro, D.; Farini, A.; Cadiao, G.; Villa, C.; Torrente, Y. Flavonoids and Omega3 Prevent Muscle and Cardiac Damage in Duchenne Muscular Dystrophy Animal Model. Cells 2021, 10, 2917. [Google Scholar] [CrossRef] [PubMed]
- Carotenuto, F.; Minieri, M.; Monego, G.; Fiaccavento, R.; Bertoni, A.; Sinigaglia, F.; Vecchini, A.; Carosella, L.; Di Nardo, P. A diet supplemented with ALA-rich flaxseed prevents cardiomyocyte apoptosis by regulating caveolin-3 expression. Cardiovasc. Res. 2013, 100, 422–431. [Google Scholar] [CrossRef] [PubMed]
- Caligiuri, S.P.; Aukema, H.M.; Ravandi, A.; Guzman, R.; Dibrov, E.; Pierce, G.N. Flaxseed consumption reduces blood pressure in patients with hypertension by altering circulating oxylipins via an alpha-linolenic acid-induced inhibition of soluble epoxide hydrolase. Hypertension 2014, 64, 53–59. [Google Scholar] [CrossRef]
- Dahiya, S.; Bhatnagar, S.; Hindi, S.M.; Jiang, C.; Paul, P.K.; Kuang, S.; Kumar, A. Elevated levels of active matrix metalloproteinase-9 cause hypertrophy in skeletal muscle of normal and dystrophin-deficient mdx mice. Hum. Mol. Genet. 2011, 20, 4345–4359. [Google Scholar] [CrossRef]
- Fogagnolo Maurício, A.; Camaçari de Carvalho, S.; Santo Neto, U.; Marques, M.J. Effects of dietary omega-3 on dystrophic cardiac and diaphragm muscles as evaluated by 1H magnetic resonance spectroscopy: Metabolic profile and calcium-related proteins. Clin. Nutr. ESPEN 2017, 20, 60–67. [Google Scholar] [CrossRef]
- Matsumura, C.Y.; Taniguti, A.P.; Pertille, A.; Santo Neto, H.; Marques, M.J. Stretch-activated calcium channel protein TRPC1 is correlated with the different degrees of the dystrophic phenotype in mdx mice. Am. J. Physiol. Cell Physiol. 2011, 301, C1344–C1350. [Google Scholar] [CrossRef]
- Rodríguez-Cruz, M.; Cruz-Guzmán, O.D.R.; Almeida-Becerril, T.; Solís-Serna, A.D.; Atilano-Miguel, S.; Sánchez-González, J.R.; Barbosa-Cortés, L.; Ruíz-Cruz, E.D.; Huicochea, J.C.; Cárdenas-Conejo, A.; et al. Potential therapeutic impact of omega-3 long chain-polyunsaturated fatty acids on inflammation markers in Duchenne muscular dystrophy: A double-blind, controlled randomized trial. Clin. Nutr. 2018, 37, 1840–1851. [Google Scholar] [CrossRef]
- Sitzia, C.; Meregalli, M.; Belicchi, M.; Farini, A.; Arosio, M.; Bestetti, D.; Villa, C.; Valenti, L.; Brambilla, P.; Torrente, Y. Preliminary Evidences of Safety and Efficacy of Flavonoids- and Omega 3-Based Compound for Muscular Dystrophies Treatment: A Randomized Double-Blind Placebo Controlled Pilot Clinical Trial. Front. Neurol. 2019, 10, 755. [Google Scholar] [CrossRef] [PubMed]
- Ren, S.; Yao, C.; Liu, Y.; Feng, G.; Dong, X.; Gao, B.; Qian, S. Antioxidants for Treatment of Duchenne Muscular Dystrophy: A Systematic Review and Meta-Analysis. Eur. Neurol. 2022, 85, 377–388. [Google Scholar] [CrossRef] [PubMed]
- Burstein, S.H.; McQuain, C.A.; Ross, A.H.; Salmonsen, R.A.; Zurier, R.E. Resolution of inflammation by N-arachidonoylglycine. J. Cell Biochem. 2011, 112, 3227–3233. [Google Scholar] [CrossRef]
- Dort, J.; Orfi, Z.; Fabre, P.; Molina, T.; Conte, T.C.; Greffard, K.; Pellerito, O.; Bilodeau, J.F.; Dumont, N.A. Resolvin-D2 targets myogenic cells and improves muscle regeneration in Duchenne muscular dystrophy. Nat. Commun. 2021, 12, 6264. [Google Scholar] [CrossRef]
- Honkisz-Orzechowska, E.; Łażewska, D.; Baran, G.; Kieć-Kononowicz, K. Uncovering the Power of GPR18 Signalling: How RvD2 and Other Ligands Could Have the Potential to Modulate and Resolve Inflammation in Various Health Disorders. Molecules 2024, 29, 1258. [Google Scholar] [CrossRef]
- Giannakis, N.; Sansbury, B.E.; Patsalos, A.; Hays, T.T.; Riley, C.O.; Han, X.; Spite, M.; Nagy, L. Dynamic changes to lipid mediators support transitions among macrophage subtypes during muscle regeneration. Nat. Immunol. 2019, 20, 626–636. [Google Scholar] [CrossRef] [PubMed]
- Dort, J.; Orfi, Z.; Fiscaletti, M.; Campeau, P.M.; Dumont, N.A. Gpr18 agonist dampens inflammation, enhances myogenesis, and restores muscle function in models of Duchenne muscular dystrophy. Front. Cell Dev. Biol. 2023, 11, 1187253. [Google Scholar] [CrossRef]
- Rodríguez-Cruz, M.; Atilano-Miguel, S.; Barbosa-Cortes, L.; Bernabé-García, M.; Cárdenas-Conejo, A.; Del Rocío Cruz-Guzmán, O.; Maldonado-Hernández, J. Evidence of muscle loss delay and improvement of hyperinsulinemia and insulin resistance in Duchenne muscular dystrophy supplemented with omega-3 fatty acids: A randomized study. Clin. Nutr. 2019, 38, 2087–2097. [Google Scholar] [CrossRef]
- Kopecky, J.; Rossmeisl, M.; Flachs, P.; Kuda, O.; Brauner, P.; Jilkova, Z.; Stankova, B.; Tvrzicka, E.; Bryhn, M. n-3 PUFA: Bioavailability and modulation of adipose tissue function. Proc. Nutr. Soc. 2009, 68, 361–369. [Google Scholar] [CrossRef]
- Rodríguez-Cruz, M.; Sánchez, R.; Escobar, R.E.; Cruz-Guzmán, O.R.; López-Alarcón, M.; Bernabé-García, M.; Coral-Vázquez, R.; Matute, G.; Velázquez Wong, A.C. Evidence of insulin resistance and other metabolic alterations in boys with Duchenne or Becker muscular dystrophy. Int. J. Endocrinol. 2015, 2015, 867273. [Google Scholar] [CrossRef] [PubMed]
- Saure, C.; Caminiti, C.; Weglinski, J.; Pérez, F.D.; Monges, S. Energy expenditure, body composition, and prevalence of metabolic disorders in patients with Duchenne muscular dystrophy. Diabetes Metab. Syndr. Clin. Res. Rev. 2018, 12, 81–85. [Google Scholar] [CrossRef] [PubMed]
- Korotkova, M.; Lundberg, I.E. The skeletal muscle arachidonic acid cascade in health and inflammatory disease. Nat. Rev. Rheumatol. 2014, 10, 295–303. [Google Scholar] [CrossRef] [PubMed]
- Hoxha, M. Duchenne muscular dystrophy: Focus on arachidonic acid metabolites. Biomed. Pharmacother. 2019, 110, 796–802. [Google Scholar] [CrossRef]
- Lim, J.H.; Gerhart-Hines, Z.; Dominy, J.E.; Lee, Y.; Kim, S.; Tabata, M.; Xiang, Y.K.; Puigserver, P.J. Oleic acid stimulates complete oxidation of fatty acids through protein kinase A-dependent activation of SIRT1-PGC1alpha complex. Biol. Chem. 2013, 288, 7117–7126. [Google Scholar] [CrossRef]
| MD Disease | Main Pathological Features | Age of Onset (Range) | Gender |
|---|---|---|---|
| DMD | Severe muscle weakness with frequent falls, difficulty walking, and wheelchair dependence | Childhood | Men |
| Beker dystrophy | progressive weakness and wasting | Slow progression 11–25 years | Men |
| Myotonic muscular dystrophy | Muscle loss, weakness, myotonia | Adults | Men and women |
| Limb-Girdle Muscular Dystrophy | muscle weakness of the upper arms, upper legs, shoulders, and hips | 2–40 years | Men and women |
| Fascioscapulohumeral Muscular Dystrophy | Facial weakness, scapular winging, foot drop | 15–30 years | Men and women |
| Emery–Dreifuss Muscular Dystrophy | Joint contractures, muscle weakness | adolescent | Men and Women |
| Distal Muscular Dystrophy | Distal muscle weakness | 40–60 years | Men and women |
| Oculopharyngeal Muscular Dystrophy | Muscle weakness of eyelids, face, and throat | 40–50 years | Men and women |
| Collagen Type VI-Related Disorders | Low muscle tone, with joint laxity, contractures of the arms or legs, and decreased flexibility of the spine. | Childhood, sometimes in adolescence and adulthood. | Men |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Versari, I.; Bavelloni, A.; Traversari, M.; Burattini, S.; Battistelli, M.; Gobbi, P.; Faenza, I.; Salucci, S. Functional Foods, a Hope to Delay Muscle Dystrophy Progression: A Potential Role for Omega Fatty Acids. Nutrients 2025, 17, 1039. https://doi.org/10.3390/nu17061039
Versari I, Bavelloni A, Traversari M, Burattini S, Battistelli M, Gobbi P, Faenza I, Salucci S. Functional Foods, a Hope to Delay Muscle Dystrophy Progression: A Potential Role for Omega Fatty Acids. Nutrients. 2025; 17(6):1039. https://doi.org/10.3390/nu17061039
Chicago/Turabian StyleVersari, Ilaria, Alberto Bavelloni, Mirko Traversari, Sabrina Burattini, Michela Battistelli, Pietro Gobbi, Irene Faenza, and Sara Salucci. 2025. "Functional Foods, a Hope to Delay Muscle Dystrophy Progression: A Potential Role for Omega Fatty Acids" Nutrients 17, no. 6: 1039. https://doi.org/10.3390/nu17061039
APA StyleVersari, I., Bavelloni, A., Traversari, M., Burattini, S., Battistelli, M., Gobbi, P., Faenza, I., & Salucci, S. (2025). Functional Foods, a Hope to Delay Muscle Dystrophy Progression: A Potential Role for Omega Fatty Acids. Nutrients, 17(6), 1039. https://doi.org/10.3390/nu17061039









