Abstract
Snakebite envenomation is considered a neglected tropical disease, affecting tens of thousands of people each year. The recommended treatment is the use of antivenom, which is composed of immunoglobulins or immunoglobulin fragments obtained from the plasma of animals hyperimmunized with one (monospecific) or several (polyspecific) venoms. In this review, the efforts made in the improvement of the already available antivenoms and the development of new antivenoms, focusing on snakes of medical importance from sub-Saharan Africa and Latin America, are described. Some antivenoms currently used are composed of whole IgGs, whereas others use F(ab’)2 fragments. The classic methods of attaining snake antivenoms are presented, in addition to new strategies to improve their effectiveness. Punctual changes in immunization protocols, in addition to the use of cross-reactivity between venoms from different snakes for the manufacture of more potent and widely used antivenoms, are presented. It is known that venoms are a complex mixture of components; however, advances in the field of antivenoms have shown that there are key toxins that, if effectively blocked, are capable of reversing the condition of in vivo envenomation. These studies provide an opportunity for the use of monoclonal antibodies in the development of new-generation antivenoms. Thus, monoclonal antibodies and their fragments are described as a possible alternative for the production of antivenoms, regardless of the venom. This review also highlights the challenges associated with their development.
Key Contribution:
This review aims to contribute to the understanding of the development of serum therapy in recent decades, showing its importance over the years and the need for its improvement. The advance in the quality of antivenoms and the search for alternatives for the treatment of snakebites are also covered.
1. Snakebites and Serum Therapy
The estimated number of snakebites in the world is about 400,000/year with approximately 20,000 deaths/year [1]. Another estimate shows that there are around 1.8–2.7 million snake envenomations annually, resulting in around 81,000–138,000 deaths, and that there may be as many as 400,000 people with permanent disabilities resulting from snakebite each year [2,3,4].
The greatest burden of snakebite envenomation occurs in Asia, sub-Saharan Africa, Latin America, and parts of Oceania [1]. In 2017, the World Health Organization considered ophidism a neglected tropical disease of the highest priority, and set a commitment with several countries to reduce snakebite accidents by 50% by 2030 [5].
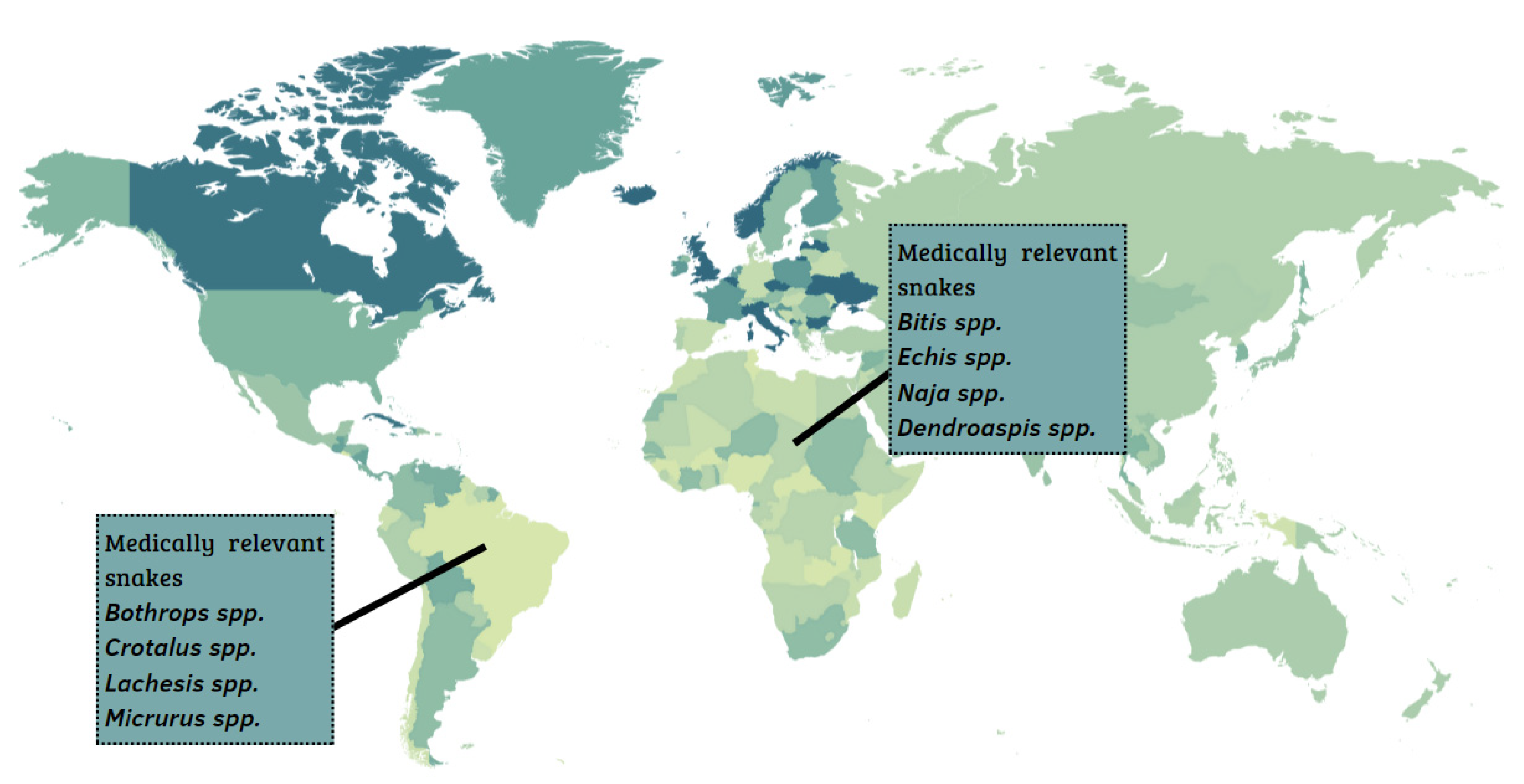
In the Middle East and North Africa, 17 snake species are found, and, in sub-Saharan Africa, encompassing the Central, East, South, and West regions of the African continent, 26 species are found. Among these, the most medically relevant snakes belong to the genera Echis spp., Naja spp., Dendroaspis spp., and Bitis spp. (Figure 1) [6]. In Latin America, the most important snakes belong to the genera Bothrops spp., Lachesis spp., Crotalus spp., and Micrurus spp. [7,8].
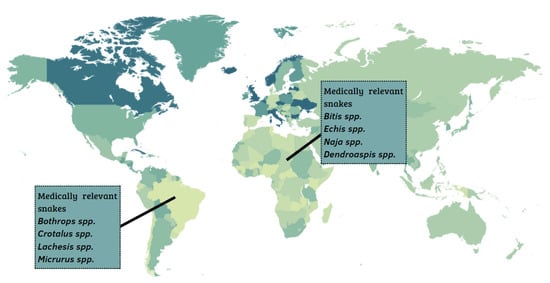
Figure 1.
Geographic distribution of the most medically important snake species in Africa and Latin America.
The Bitis genus includes six species responsible for a large number of bites: B. arietans, B. somalica, B. parviocula, B. gabonica, B. rhinoceros, and B. nasicornis [9,10].
B. arietans, a snake of great medical importance, is responsible for a large number of serious accidents, mainly in children and rural workers in Africa [10,11]. The Bitis arietans bite causes local damage, such as necrosis, and systemic symptoms, such as fever, neutrophilic leukocytosis, thrombocytopenia, hemolysis, and bleeding, which can result in anemia, reduced resistance to infections, diffuse hemorrhage, myocardial damage, coagulopathy, hypotension, and death [9,10].
Regarding the genus Dendroaspis, D. polylepis stands out and is popularly known as black mamba, whereas D. angusticeps and D. viridis are called green mambas. The envenomation may cause hypotension, tachycardia, paresthesia in superior and inferior limbs, and respiratory failure in their victims [12]. Proteomics studies reveled that the D. polylepis venom is mainly composed of Kunitz-type molecules, which include mamba dendrotoxins (63%), three-finger toxins (31%), and metalloproteases (3%) [13].
The genus Naja is composed of a large number of snakes, totaling 33 species [14], including N. naja, N. annulifera, N. melanoleuca, N. mossambica, and N. nigricollis. Most species have neurotoxic venom, acting on the nervous system, causing paralysis. In addition, many venoms from Naja spp. have cytotoxic characteristics that cause swelling and necrosis, in addition to cardiotoxic components [15].
According to reports, cases of human casualties resulting from E. ocellatus envenoming are higher than those of all other African snakes combined, accounting for 90% of bites and over 60% of mortalities, and several thousand permanent disabilities [16,17].
In Latin America, the prominent genera are Bothrops spp. and Crotalus spp., as they account for more than 95% of reported accidents. Bothrops species are abundant [18], with a wide geographic distribution, since they have successfully colonized most of the South American territory [19,20]. The Bothrops genus is responsible for the majority of accidents in Brazil (around 85%). The accidents gain attention because of the gravity of the symptoms resulting from the complex mechanism of action of many toxins, such as Snake Venom Metallo Proteases (SVMPs), Snake Venom Serine Proteases (SVSPs) and phospholipases A2 (PLA2s) [21,22,23]. Accidents caused by the Bothrops genus result in symptoms characterized by hemorrhage, inflammation, and disturbances in the coagulation cascade [24], with local and systemic clinical manifestations. The local manifestations are characterized by edema, ecchymosis, pain, and blisters with serous, hemorrhagic, or necrotic content that may occur. In systemic manifestations, gingivorrhagia, microscopic hematuria, purpura, bleeding in recent wounds, intense hemorrhage, shock, and renal failure are observed in the most severe cases [24,25,26,27].
Other snakes of medical importance in Brazil belong to the genus Crotalus (i.e., C. durissus terrificus, C. d. cascavela, C. d. collineatus, C. d. ruruima, C. vergandis, C. d. ruruima, C. d. marajoensis). Usually, their venoms are mainly composed of neurotoxins and myotoxins, such as crotoxin [27,28], crotamin [29,30], and gyroxin [28,30]. Responsible for both the neurotoxic and systemic myotoxic effects characteristic of this venom, crotoxin comprises two sub-units that are non-covalently linked: the non-catalytic A (CA), or crotapotin, and the catalytic unit, crotoxin B (CB) and phospholipase A2 (PLA2) [31]. Crotapotin is an acidic polypeptide with no detectable enzymatic activity. PLA2 catalyzes the hydrolysis of the sn-2-acyl chain of phospholipids resulting in fatty acids and lysophospholipids. Crotapotin, working as a chaperon, potentiates the toxicity of PLA2 by about 35-fold [25,32,33]. It is important to note that snakes of the Crotalus genus, commonly called rattlesnakes, are the cause of a large number of accidents in North America, especially in the United States. Fortunately, accidents rarely cause morbidity or fatalities [31].
Thus, snake venoms are a complex mixture, composed mainly of proteins (±90–95%), in addition to peptides, carbohydrates, segments derived from nucleic acids, metal ions, biogenic amines, lipids, and free amino acids, which have different biological activities [32]. Effective antivenoms are expected to be able to neutralize the major toxins in a venom.
The only treatment for snakebite is the use of antivenoms, or serum therapy, which, when injected into an envenomed human bitten by a snake, mainly neutralizes the toxins of the venom used in its production. Snake antivenoms are specific immunoglobulins produced by fractionation of plasma generally obtained from large domestic animals, mainly horses, hyperimmunized with the venoms of interest.
Specific neutralizing antibodies were first identified in the serum of experimental animals, i.e., rabbits or guinea pigs, immunized against diphtheria or tetanus toxins [34]. In 1894, the serum was successfully used for treating children suffering from severe diphtheria, and was manufactured by Burroughs Welcome, United Kingdom. In the same year, two groups, simultaneously but independently, described the antitoxic properties of the serum from rabbits and guinea pigs immunized against cobra and viper venoms [34,35]. In 1895, a similar procedure was repeated with horses immunized with snake venoms in India by Haffkine and in Vietnam by Lépnay [36]. The snake antivenom specificity—whose range includes the biting species—was demonstrated by Vital Brazil [37,38].
Antivenoms can be raised against the venom of a single species (monospecific) or against multiple species (polyspecific), and have already proven to be effective in preventing many of the lethal and damaging effects of envenomation [4]. Antivenom administration can reverse the major effects of envenomation, such as anti-hemostasis, neurotoxicity, and hypotension [4]. In contrast, venom-induced local damage is not well prevented by antivenoms unless it is administered soon after the bite [39]. In addition, despite being therapeutically efficient, some antivenoms currently used can induce adverse reactions.
The vast majority of antivenom manufacturers refine IgG extracted from animal plasma, producing F(ab’)2 fragments through enzymatic digestion with pepsin. Other industries use papain to produce even smaller Fab fragments, improving safety and tissue distribution. This IgG format, however, has the disadvantage of rapid renal clearance. Removal of the Fc fraction of the antigen-binding fragment reduces the risk of adverse reactions [40]. Nonetheless, some antivenoms are made by whole IgG molecules, which are purified from plasma by caprylic acid precipitation or affinity chromatography, which yield a safer product but at a higher cost [4].
2. Immunoglobulins for Therapeutic and Diagnostic Applications
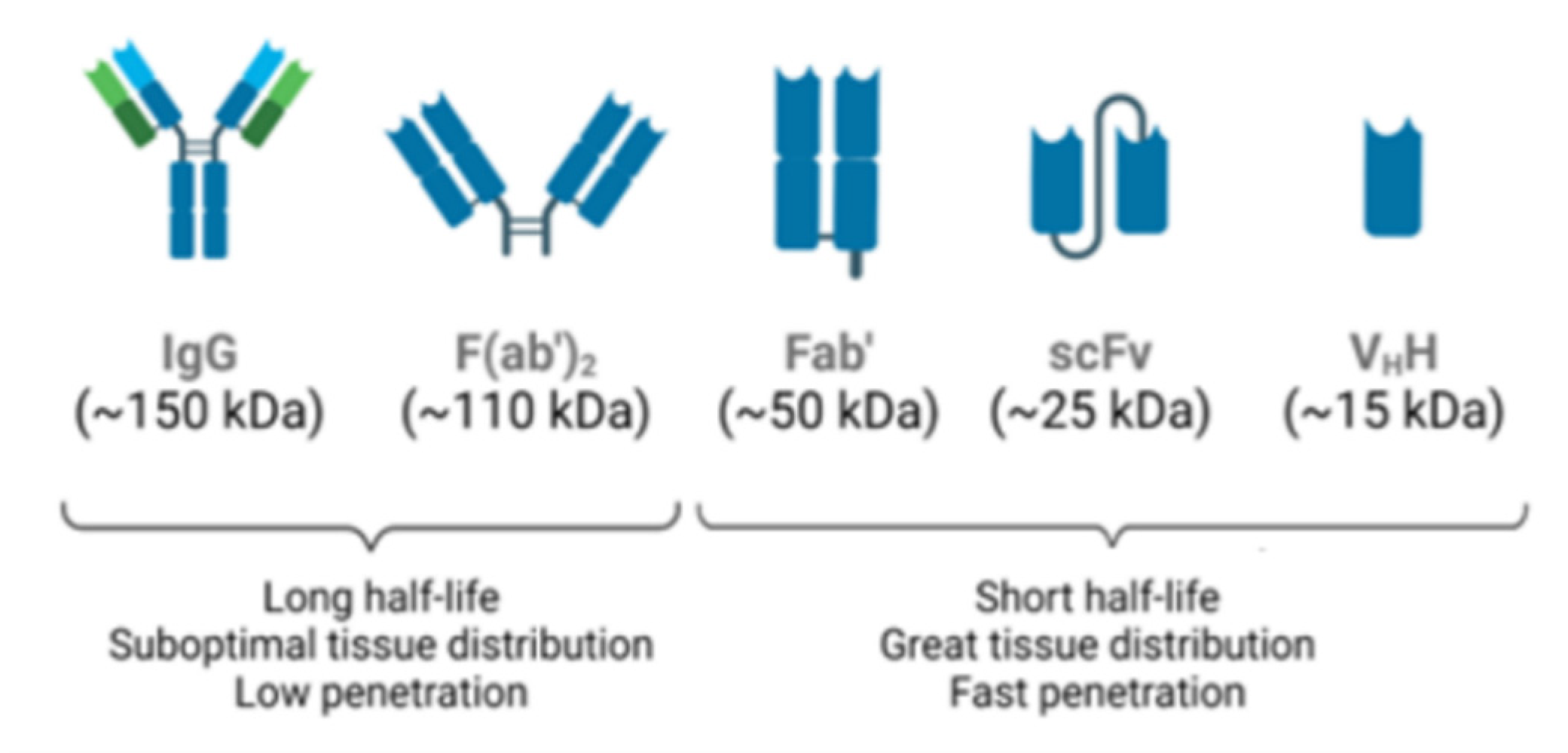
The most widely used immunoglobulin for therapeutic and diagnostic applications [41], human IgG (Figure 2), is a protein made up of two identical heavy chains and two identical light chains, κ or λ, that are interconnected by a series of disulfide bonds. Each heavy chain contains three constant domains (i.e., CH1, CH2, and CH3) and one variable domain (VH). The light chain contains one constant domain (CL) and one variable domain (VH). The variable regions, at the N-terminus of the antigen-binding fragment (Fab), determine the specificity, diversity, and affinity of antigen binding. Within each variable domain, there are three hypervariable regions called complementary determining regions (CDR1, CDR2, and CDR3), which are primarily responsible for antigen recognition and binding. The remainder of the VH and VL domains are the framework regions that act as a scaffold to support the CDR’s loops. The CDRs and framework regions in each of the variable domains contribute to antigen binding [41].
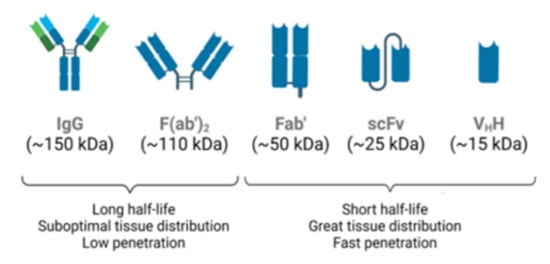
Figure 2.
Different antibody formats with biological activities. IgG: whole molecule. F(ab’)2: product of IgG digestion with pepsin. Fab: product of digestion of IgG with papain. Diabody: non-covalent dimers of scFv fragments. scFv: single-chain variable fragments. VhH: single domain antigen specific fragment. Schematic diagram developed on the biorender platform.
Regarding formats, IgGs are the most abundant class of monoclonal antibodies (mAbs) approved for therapeutic use, accounting for 82% of the total [42]. FDA-approved antibodies can be classified as humanized (46%), human (41%), chimeric (10%), or murine (3%) [42], and are powerful tools for use in therapy and diagnosis. mAbs are one of the fastest growing classes of therapeutic biomolecules, with a market valued at USD 168.70 billion in 2021, and projected to reach USD 188.18 billion in 2022, recording a growth rate of 11.5% [43].
The first mAb approved for human use by the FDA was muromonab-CD3 (Orthoclone OKT3) in 1986, a murine mAb that acts as an immunosuppressant for acute transplant rejection, targeting CD3 expressed by T cells [42]. Currently, more than 100 monoclonal antibodies are approved by the FDA, with 6–12 approvals per year [44]. These numbers are expected to increase as technological advances have made research and development of monoclonal antibodies cheaper, faster, and more efficient.
Antibody engineering has also focused its attention on the development of functional antibody fragments, which have more favorable features, such as a smaller molecular size and higher affinity, when compared with whole IgGs (Figure 2) [45]. Currently, antibody fragments represent 9% of the total number of antibodies approved for therapeutic or diagnostic purposes [44]. Several antibody fragments are already in clinical trials, with antigen-binding fragments and single-chain variable fragments (scFvs) representing the majority [46].
There has also been a growing interest in heavy-chain only antibodies (HcAbs), the smallest naturally occurring antigen-binding fragment formed by a single heavy chain with a variable domain found in camelids or sharks (Figure 2). As the smallest known functional antibody fragments, nanobodies (VhHs) are molecules of approximately 15 kDa. Their small size, high solubility, tissue permeability, and stability to changes in pH and temperature, allow interaction with sites inaccessible to conventional antibodies and having low immunogenicity. Thus, nanobodies are promising candidates for different applications in biomedical research, diagnosis, and therapy [47].
Unlike Fab or (Fab’)2, an scFv is not a fragment of an antibody, but rather a protein formed by the fusion of VH and VL variable regions, connected by a short linker (Figure 2). This linker is rich in glycine, to provide flexibility, in addition to serine or threonine, to increase solubility and to also allow the connection from the N-terminus of the VL to the C-terminus of the VH, or vice versa [48]. This fusion protein retains the specificity of the original IgGs despite the removal of constant regions and the linker introduction.
ScFvs have some advantages when compared to chimeric or humanized antibodies, since these molecules maintain binding specificity and, due to their size and absence of a constant region, have little immunogenicity, better tissue penetration, and do not activate the complement system. Furthermore, scFvs can be produced in a prokaryotic expression system, as these molecules do not require glycosylation. However, their binding affinities and half-lives can be reduced [48]. As of June 2022, there were 4 Fabs, 2 scFvs, and only one humanized nanobody approved by the FDA for use [49].
Essentially, there are two ways in which antibodies can neutralize toxins, either by directly or indirectly inhibiting them. In direct inhibition, the antibody binds to the toxin and competes for the site of interaction, whether of an enzymatic toxin or not [50]. Indirect inhibition can occur through three different mechanisms: (i) allosteric inhibition, where the antibody binding to the toxin induces a conformational change, losing activity [51]; (ii) through a steric hindrance effect where there is binding of the antibody to a region close to the active site; or (iii) by preventing the dissociation of toxin complexes, preventing the formation of active toxins [52]. Considering the remarkable progress in the mechanisms underlying the immune response, and the immunochemistry of immunoglobulins and the high number of snakebites, studies have been designed with the objective of improving the neutralizing properties of antibody-antitoxins.
3. Classic Methods for Improving Serum Therapy
Currently, about 31 antivenoms are available worldwide for the treatment of snakebites, as listed by Laustsen et al. [53]. Despite their importance for the current treatment envenomation, conventional antivenoms are partially ineffective in neutralizing some effects caused by snake venoms in vitro and in vivo [54,55], and they can also cause some side effects [56,57]. In in vivo studies, the neutralization potential of the anti-bothropic serum, either with the treatment of the animals before or immediately after the application of the bothropic venom, was studied by Battellino et al. (2003), where different application schedules of the anti-bothropic serum produced by the Butantan Institute were analyzed: before (15 min), at the same time, and after (15 min) the application of B. jararaca venom [58]. The authors concluded that the low neutralization did not occur due to the lack of specific antibodies, but due to the low interaction of the antibodies with the venom components, probably due to the difficulty in distributing the F(ab’)2 fragments in the tissues. It is important to emphasize that the results indicate the formation of hemorrhagic lesions even with the in vitro pre-incubation procedure, but with a proportion almost six times lower when compared to the control. Blind studies with humans envenomed by the snake Echis ocellatus in Ghana showed that a new antivenom was ineffective in combating lethality, demonstrating the need for pilot tests before its single and general distribution in a region is initiated [54].
For this reason, new strategies have been used to optimize the treatment of snakebites, whether designing experiments to obtain new high-quality antivenoms or improving the effectiveness of current commercial antivenoms.
Some strategies for developing antivenoms are based on cross-reactivity. The evaluation of the cross-reactivity of antivenoms, i.e., the ability of antibodies to neutralize toxins from different snake species, yields the possibility of attaining paraspecific antivenoms, which can be easier to produce and more profitable [59]. Some studies show that, although antivenoms are produced from a limited number of species, they show very interesting cross-reactivity with other species from the same region. Among several examples [60,61], the cross-reactivity between nine snake venoms of the genus Bothrops and a serum produced against the venom of B. jararaca stands out [62]. In fact, despite the diversity of this genus in Brazil, the victims are treated with the pentavalent serum produced by the Butantan Institute, which is attained using the venoms of B. jararaca (50%), B. jararacussu (12.5%), B. alternatus (12.5%), B. moojeni (12.5%), and B. neuwiedi (12.5%) [24,62] as antigens. Moreover, another study aiming at the development of antivenoms against eight snake species found in Mozambique resulted in high titers of Abs against Bitis arietans, B. nasicornis, and B. rhinoceros (5.18 × 106, 3.60 × 106, and 3.50 × 106 U-E/mL, respectively), and against Naja melanoleuca, N. mossambica, and N. annulifera (7.41 × 106, 3.07 × 106, and 2.60 × 106 U-E/mL, respectively), but lower titers against the Dendroaspis angusticeps and D. polylepis venoms (1.87 × 106 and 1.67 × 106 U-E/m, respectively) [63].
An SVMP with hemorrhagic activity present in the venom of B. arietans was recognized by antibodies present in a series of polyvalent antivenoms, composed of F(ab)’2 portions, attained by horse and chicken immunizations. In addition to anti-Bitis spp. serum, anti-Bothrops spp., anti-Lachesis muta, anti-Crotalus spp., and anti-Naja spp. recognized the purified SVMPs. The observed cross-reactivity indicates that metalloproteases induce an immunological signature, probably due to the presence of common epitopes among the different SVMPs present in various snake venoms [64].
The use of adjuvants can also improve antivenom antibody titers. Using the venom of Crotalus d. terrificus as an antigen and different immunization protocols, it was shown that venom emulsified with Freund’s adjuvant induced a more protective and sustained immune response compared to Al(OH)3 or liposome particles [65]. In addition to adjuvants, chicken hyperimmunization has also demonstrated the effectiveness of IgYs in recognizing, combining, and neutralizing the toxic and lethal components present in venoms from snakes of the Bothrops and Crotalus genera [66].
Another simple strategy that can improve the quality of an antivenom is changing the immunization protocol and the amount of immunogen injected. Increasing the interval between boosters and using a smaller amount of venom appear to result in antibodies with higher titers and affinity [67].
Several studies have indicated that, when purified toxins are used as antigens, instead of the total venom where they are present, they favor the production of antibodies with better titers and affinity [68]. In one study, the use of crude Crotalus venom, and purified crotoxin and PLA2 present in this venom, as immunogens in horses were compared. The results indicated that the serum obtained against the total venom showed both low titers and neutralizing capacity. By comparison, immunization with crotoxin resulted in a serum with greater specificity and, if used as an adjuvant, can prevent injuries and adverse reactions in serum-producing animals. Finally, immunization with PLA2 resulted in less neutralizing serum, especially when PLA2 epitopes are in their free form. In general, antibodies with high titers and the ability to cross-react were produced, but there was no increase in affinity [69]. Using purified toxins, a potential new antivenom was developed against SVSPs from Bothrops jararaca venom with enzymes identified as unblocked by the antivenom produced by the Butantan Institute. Isogenic C57BL/6 and BALB/c mice were immunized with a pool of four purified serine proteases (KN-BJ2, BjSP, HS112, and BPA), which were not inhibited by commercial antiserum. The results showed that the two sera obtained were able to block the SVSPs of five bothropic venoms, indicating that the use of purified toxins can further improve the quality of the sera [70,71].
4. Next-Generation Antivenom: Monoclonal Antibodies and Their Fragments
The discovery and development of monoclonal antibodies became cheaper, quicker, and more efficient in recent years, paving the way for the development of “next-generation antivenom”. This is the use of human monoclonal antibody mixtures that target the key toxins in snake venoms. Toxins from snake venoms feature a synergic effect, so their action provokes a greater toxification and/or lethality than the sum of their separate effects. As a consequence, neutralizing certain key toxins from the venom is enough to drastically reduce the effects of the whole envenomation [72,73].
Various different monoclonal antibodies have been discovered and developed against toxins from different venomous animals, such as snakes, scorpions, spiders, and bees, as reviewed by Lausten and colleagues [53].
The first stage in these developments occurred in 1982, when Boulain and colleagues developed the first homogeneous population of high-affinity monoclonal antibodies specifically for Naja nigricollis snake α-toxin [74]. The spleen cells were fused with myeloma to obtain a hybridoma, and the resulting toxin-binding antibodies were purified and tested. This antibody neutralizes the biological activity of the toxin under both in vivo and in vitro conditions [74]. In 1995, Meng and collaborators used, for the first time, the technique of phage display to find monoclonal antibody fragments against animal toxins. In this study, scFvs from the library of human semi-synthetic antibodies against crotoxin were identified [75], and two years later Lafaye and colleagues developed the first human scFv targeting venom toxins using phage display [52].
Another strategy gaining momentum is the use of humanized or human monoclonal antibodies. These antibodies, which are compatible with the human organism, present a lower risk of reducing the immune characteristics of the antivenom [76]. The first fully human IgG was developed against hemorrhagic metalloproteases from Protobothrops flavoviridis, and was attained from the fusion of myeloma cells SP2/0-Ag-14 and cells from KM mice spleens previously immunized with the toxin. In this study, 300 hybridoma cells were produced to attain IgGs for the toxin HR1a, and 80 reactive antibodies were identified [77].
In 2018, the first development of fully human monoclonal IgGs against snake venom using phage display was reported. The IgGs were selected from a naïve human library of scFvs, and showed specificity to dendrotoxins from Dendroaspis polylepis. Monoclonal antibodies were able to prevent lethality when pre-incubated with the toxin fraction and, in addition, showed that an oligoclonal mixture of toxin-binding antibodies was able to prevent lethality for the entire venom [53].
The firsts antibody formats for recombinant antivenom were monoclonal IgGs and single-chain variable fragments. In vivo studies have shown that monoclonal IgGs targeting snake toxins are able to neutralize myotoxic, hemorrhagic, and proteolytic effects. Monoclonal IgGs that neutralize these effects were developed for some medically relevant snakes from the genera Naja spp., Crotalus spp., Echis spp., Laticauda spp., and Bothrops spp. [50,74,78,79,80,81]. Monoclonal antibodies against a phospholipase A2, in addition to a metalloproteinase and a thrombin-like via hybridoma, were obtained and, when used together, prevented the in vivo lethality of Bothrops atrox venom [80]. Other studies have also been published on monoclonal IgGs targeting Bothrops spp. toxins, such as the development of a mAb that neutralizes the hemotoxic effects of Atroxlysin-I from B. atrox venom [82]. In 1988, Lomonte and Kahan developed the first mAb against B. asper, a murine IgG that neutralizes the myotoxic effects from the venom [83]. In 2010, mAbs capable of neutralizing B. asper BaP1 in the nanomolar range were developed, blocking its hemotoxic effects [84]. To our knowledge, no mAb-targeting Bitis spp. toxins have been developed.
Another antibody format widely used in snakebite immunotherapy is Fab, which is mainly used in conventional serum therapy, in a polyclonal mixture. Two Fabs targeting snake toxins, cardiotoxin from Naja nigricollis and b1-bungarotoxin from Bungarus multicinctuse, neutralize their in vitro and in vivo effects [85,86].
The single-chain variable fragment format has been extensively studied and developed for snakebite immunotherapy, and several studies have identified human scFvs capable of decreasing the effects on Bothrops jararacussu and Crotalus durissus toxins [75,87,88,89,90,91,92,93]. Due to their small size, although most IgGs are expressed in hybridoma cells or mammalian cells, different expression systems can be used to produce scFvs, with microbial systems being the most used, and even plants have been used [94,95].
The use of recombinant nanobodies that are able to recognize medically relevant snake venom toxins has also gained attention [96,97,98]. Using a naive VHH library, clones against α-cobrotoxin present in Naja kaouthia venom were identified [99]. Humanized-single domain antibodies for the phospholipase present in the N. kaouthia venom were able to inhibit this activity up to 50% [100]. Using a library of VHH immune genes, a clone with high affinity for α-cobratoxin was selected and fused to a human Fc fragment to create a VHH2-Fc antibody that, when pre-incubated with the toxin, neutralized its lethality [101]. An immune VHH library for phospholipases A2 from Bothrops jararacussu venom was constructed, and the selected clones neutralized in vivo myotoxic activity, and presented cross-reactivity with PLA2 from different Bothrops species [102]. Moreover, from an immune VHH library, clones against the hemorrhagic and myotoxic fractions of B. atrox venom were selected and were able to neutralize these effects; however, the nanobodies were not able to prevent lethality [98].
These studies show that monoclonal antibodies and/or antibody fragments, as an innovative antivenom for viperid and elapid species, are feasible, and may be the next step in snakebite therapy. Antibody fragments, due to their small size, diffuse rapidly through the body, reaching a higher tissue biodistribution when compared to conventional serum therapy [53].
Research regarding the improvement in heterologous antivenom is extremely important and, to this day, it is the only effective and available treatment for snakebite envenomation. In countries that suffer the highest rate of snakebites, the production of recombinant antivenom may seem unfeasible, since the research and development of monoclonal antibodies requires a high level of technology [42]. However, the high investment required can be offset by a cheaper final product compared to heterologous sera. Theoretically, an oligoclonal mixture of antibodies with 25% cross-reactivity can equal the costs of current treatment and, when taking into account different formats and even expression strategies, the treatment can be up to 10 times cheaper [103]. The technology needed for antibody research and development has become cheaper every year, with some estimates even surpassing Moore’s law [104].
Finally, the use of animals for research, development, and manufacturing has been a controversial topic for many years, and there is a public desire to reduce animal use, particularly where non-animal-derived alternatives are present [105]. Since the treatment of snakebites has historically used immunization of large mammals, the substitution by recombinant sera, made from a cocktail of monoclonal antibodies, may be a safer—and even cost-competitive—alternative for future therapy of envenomation [106,107].
Author Contributions
W.D.S., F.C.V.P., O.A.S. and S.A.A. conceived and designed the manuscript. W.D.S., F.C.V.P., P.J.S., S.A.A., D.A.S., Â.A.A.M. and F.C.M. wrote the manuscript; L.Y.S. supported the editing of the manuscript; L.Y.S., F.R.G., K.S.G. and F.C.M. assisted with writing and manuscript changes; W.D.S. and F.C.V.P. funding acquisition. F.C.V.P., P.J.S. and S.A.A. critically reviewed the manuscript. All authors have read and agreed to the published version of the manuscript.
Funding
São Paulo Research Foundation: 2013/07467-1; National Council for Scientific and Technological Development: PROÁFRICA, CNPq: 490048/2005-6; São Paulo Research Foundation: 2019/20832-7.
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Acknowledgments
The authors thank to Guilherme Hiromi Yoshikawa by supporting in the preparation the article figures. Elaine Rodrigues and Lia Aguiar Xavier dos Santos by supporting the integration between researchers and administrative services, and Severino Ramos da Silva, responsible for maintenance of animals under experimentation.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Kasturiratne, A.; Wickremasinghe, A.R.; de Silva, N.; Gunawardena, N.K.; Pathmeswaran, A.; Premaratna, R.; Savioli, L.; Lalloo, D.G.; de Silva, H.J. The Global Burden of Snakebite: A Literature Analysis and Modelling Based on Regional Estimates of Envenoming and Deaths. PLoS Med. 2008, 5, e218. [Google Scholar] [CrossRef]
- Chippaux, J.-P. Snakebite Envenomation Turns Again into a Neglected Tropical Disease! J. Venom. Anim. Toxins Incl. Trop. Dis. 2017, 23, 38. [Google Scholar] [CrossRef] [PubMed]
- Seifert, S.A.; Armitage, J.O.; Sanchez, E.E. Snake Envenomation. N. Engl. J. Med. 2022, 386, 68–78. [Google Scholar] [CrossRef] [PubMed]
- Gutiérrez, J.M.; Calvete, J.J.; Habib, A.G.; Harrison, R.A.; Williams, D.J.; Warrell, D.A. Snakebite Envenoming. Nat. Rev. Dis. Primers 2017, 3, 17063. [Google Scholar] [CrossRef] [PubMed]
- WHO Snakebite: WHO Targets 50% Reduction in Deaths and Disabilities. Available online: https://www.who.int/news/item/06-05-2019-snakebite-who-targets-50-reduction-in-deaths-and-disabilities (accessed on 23 August 2022).
- Mp, A.; Mamman, M.; Po, Y.; Mg, M.; Ada, G.; Io, A.; Pe, U. Overview of the Venomous Snakes in West Africa, Clinical Manifestation, Diagnosis and Treatment. J. Med. Biol. 2019, 1, 11–26. [Google Scholar]
- World Health Organization. Guidelines for the Production, Control and Regulation of Snake Antivenom Immunoglobulins; WHO Press: Geneva, Switzerland, 2016; Available online: https://extranet.who.int/pqweb/vaccines-production-control-and-regulation-snke-antivenom-immunoglobulins (accessed on 14 July 2022).
- Gutiérrez, J.M. Preclinical Assessment of the Neutralizing Efficacy of Snake Antivenoms in Latin America and the Caribbean: A Review. Toxicon 2018, 146, 138–150. [Google Scholar] [CrossRef] [PubMed]
- Currier, R.B.; Harrison, R.A.; Rowley, P.D.; Laing, G.D.; Wagstaff, S.C. Intra-Specific Variation in Venom of the African Puff Adder (Bitis arietans): Differential Expression and Activity of Snake Venom Metalloproteinases (SVMPs). Toxicon 2010, 55, 864–873. [Google Scholar] [CrossRef]
- Warrell, D.A.; Ormerod, L.D.; Davidson, N.M. Bites by Puff-Adder (Bitis arietans) in Nigeria, and Value of Antivenom. Br. Med. J. 1975, 4, 697–700. [Google Scholar] [CrossRef]
- Bey, T.A.; Boyer, L.V.; Walter, F.G.; McNally, J.; Desai, H. Exotic Snakebite: Envenomation by an African Puff Adder (Bitis arietans). J. Emerg. Med. 1997, 15, 827–831. [Google Scholar] [CrossRef]
- Závada, J.; Valenta, J.; Kopecký, O.; Stach, Z.; Leden, P. Black Mamba Dendroaspis Polylepis Bite: A Case Report. Prague Med. Rep. 2011, 112, 298–304. [Google Scholar] [PubMed]
- Laustsen, A.H.; Lomonte, B.; Lohse, B.; Fernández, J.; Gutiérrez, J.M. Unveiling the Nature of Black Mamba (Dendroaspis polylepis) Venom through Venomics and Antivenom Immunoprofiling: Identification of Key Toxin Targets for Antivenom Development. J. Proteom. 2015, 119, 126–142. [Google Scholar] [CrossRef]
- The Reptile Database. Available online: http://reptile-database.org/ (accessed on 23 August 2022).
- Amr, Z.S.; Abu Baker, M.A.; Warrell, D.A. Terrestrial Venomous Snakes and Snakebites in the Arab Countries of the Middle East. Toxicon 2020, 177, 1–15. [Google Scholar] [CrossRef] [PubMed]
- Stock, R.P.; Massougbodji, A.; Alagón, A.; Chippaux, J.-P. Bringing Antivenoms to Sub-Saharan Africa. Nat. Biotechnol. 2007, 25, 173–177. [Google Scholar] [CrossRef] [PubMed]
- Chippaux, J.-P. Estimate of the Burden of Snakebites in Sub-Saharan Africa: A Meta-Analytic Approach. Toxicon 2011, 57, 586–599. [Google Scholar] [CrossRef] [PubMed]
- Schoch, C.L.; Ciufo, S.; Domrachev, M.; Hotton, C.L.; Kannan, S.; Khovanskaya, R.; Leipe, D.; Mcveigh, R.; O’Neill, K.; Robbertse, B.; et al. NCBI Taxonomy: A Comprehensive Update on Curation, Resources and Tools. Database 2020, 1–21. [Google Scholar] [CrossRef]
- Barlow, A.; Pook, C.E.; Harrison, R.A.; Wüster, W. Coevolution of Diet and Prey-Specific Venom Activity Supports the Role of Selection in Snake Venom Evolution. Proc. R. Soc. B Biol. Sci. 2009, 276, 2443–2449. [Google Scholar] [CrossRef]
- Melgarejo, A.R.; Cardoso, J.L.C.; França, F.O.S.; Wen, F.H.; Málaque, C.M.S.; Haddad, V., Jr. Serpentes peçonhentas do Brasil. In Animais Peçonhentos no Brasil–Biologia, Clínica e Terapêutica Dos Acidentes, 2nd ed.; Sarvier: São Paulo, Brazil, 2008; pp. 42–70. [Google Scholar]
- Gren, E.C.K.; Kitano, E.S.; Andrade-Silva, D.; Iwai, L.K.; Reis, M.S.; Menezes, M.C.; Serrano, S.M.T. Comparative Analysis of the High Molecular Mass Subproteomes of Eight Bothrops Snake Venoms. Comp. Biochem. Physiol. Part D Genom. Proteom. 2019, 30, 113–121. [Google Scholar] [CrossRef]
- Gonçalves-Machado, L.; Pla, D.; Sanz, L.; Jorge, R.J.B.; Leitão-De-Araújo, M.; Alves, M.L.M.; Alvares, D.J.; de Miranda, J.; Nowatzki, J.; de Morais-Zani, K.; et al. Combined Venomics, Venom Gland Transcriptomics, Bioactivities, and Antivenomics of Two Bothrops jararaca Populations from Geographic Isolated Regions within the Brazilian Atlantic Rainforest. J. Proteom. 2016, 135, 73–89. [Google Scholar] [CrossRef]
- Nicolau, C.A.; Carvalho, P.C.; Junqueira-de-Azevedo, I.L.M.; Teixeira-Ferreira, A.; Junqueira, M.; Perales, J.; Neves-Ferreira, A.G.C.; Valente, R.H. An In-Depth Snake Venom Proteopeptidome Characterization: Benchmarking Bothrops jararaca. J. Proteom. 2017, 151, 214–231. [Google Scholar] [CrossRef]
- Cardoso, J.L.C.; França, F.O.S.; Wen, F.H.; Málaque, C.M.S.; Haddad Jr., V. Venomous Animals in Brazil: Biology, Clinic and Therapeutics of Envenomations. Rev. Inst. Med. Trop. Sao Paulo 2003, 45, 338. [Google Scholar] [CrossRef]
- Sousa, L.F.; Portes-Junior, J.A.; Nicolau, C.A.; Bernardoni, J.L.; Nishiyama-Jr, M.Y.; Amazonas, D.R.; Freitas-de-Sousa, L.A.; Mourão, R.H.; Chalkidis, H.M.; Valente, R.H.; et al. Functional Proteomic Analyses of Bothrops atrox Venom Reveals Phenotypes Associated with Habitat Variation in the Amazon. J. Proteom. 2017, 159, 32–46. [Google Scholar] [CrossRef]
- Azevedo-Marques, M.M.; Cupo, P.; Coimbra, T.M.; Hering, S.E.; Rossi, M.A.; Laure, C.J. Myonecrosis, Myoglobinuria and Acute Renal Failure Induced by South American Rattlesnake (Crotalus durissus Terrificus) Envenomation in Brazil. Toxicon 1985, 23, 631–636. [Google Scholar] [CrossRef]
- Radis-Baptista, G.; Kerkis, I. Crotamine, a Small Basic Polypeptide Myotoxin from Rattlesnake Venom with Cell-Penetrating Properties. Curr. Pharm. Des. 2011, 17, 4351–4361. [Google Scholar] [CrossRef]
- Barrabin, H.; Martiarena, J.L.; Vidal, J.C.; Barrio, A. Isolation and characterization of gyroxin from Crotalus durissus Terrificus venom. In Toxins Animal, Plant and Microbial; Rosemberg, P., Ed.; Pergamon Press: Oxford, UK, 1978; pp. 113–133. [Google Scholar]
- Barrio, A. Gyrotoxin, a New Neurotoxin of Crotales durissus terrificus Venom. In Acta Physiologica Latinoamericana; Assn Latinoamer Cienc Fisiol: Buenos Aires, Argentina, 1961; Volume 11. [Google Scholar]
- Baudou, F.G.; Rodriguez, J.P.; Fusco, L.; de Roodt, A.R.; de Marzi, M.C.; Leiva, L. South American Snake Venoms with Abundant Neurotoxic Components. Composition and Toxicological Properties. A Literature Review. Acta Trop. 2021, 224, 106119. [Google Scholar] [CrossRef]
- Walter, F.G.; Stolz, U.; Shirazi, F.; McNally, J. Epidemiology of Severe and Fatal Rattlesnake Bites Published in the American Association of Poison Control Centers’ Annual Reports. Clin. Toxicol 2009, 47, 663–669. [Google Scholar] [CrossRef]
- Tasoulis, T.; Pukala, T.L.; Isbister, G.K. Investigating Toxin Diversity and Abundance in Snake Venom Proteomes. Front Pharmacol. 2022, 12, 768015. [Google Scholar] [CrossRef]
- Behring, E.V.; Kitasato, S. Ueber das Zustandekommen der Diphtherie-Immunität und der Tetanus-Immunität bei Thieren. Mol. Immunol. 2013, 28, 1319–1320. [Google Scholar]
- Hawgood, B.J. Albert Calmette 1863–1933 Fondateur de La Sérothérapie Antivenimeuse. In Annales de l’Institut Pasteur/Actualites; Elsevier Masson: Paris, France, 1999; Volume 10, pp. 139–146. [Google Scholar] [CrossRef]
- Bochner, R.; Goyffon, M. L’oeuvre Scientifique De Césaire Phisalix (1852–1906) Découvreur Du Sérum Antivenimeux. Bulletin De La Société Herpétologique De France 2007, 123, 32. [Google Scholar]
- Lahariya, C. A Brief History of Vaccines & Vaccination in India. Indian J. Med. Res. 2014, 139, 491–511. [Google Scholar]
- Brazil, V. Memória Histórica do Instituto Butantan; Elvino Pocai: São Paulo, Brazil, 1941; pp. 105–112. [Google Scholar]
- Brazil, V.; Maibon., J. La Défense Contre L’ophidisme, 2nd ed.; Saint-Paul, Impr. Pocai-Weiss: São Paulo, Brazil, 1914. [Google Scholar]
- Cardoso, J.L.; Fan, H.W.; França, F.O.; Jorge, M.T.; Leite, R.P.; Nishioka, S.A.; Avila, A.; Sano-Martins, I.S.; Tomy, S.C.; Santoro, M.L.; et al. Randomized comparative trial of three antivenoms in the treatment of envenoming by lance-headed vipers (Bothrops jararaca) in São Paulo, Brazil. QJM Int. J. Med. 1993, 86, 315–325. [Google Scholar]
- Kang, T.H.; Jung, S.T. Boosting therapeutic potency of antibodies by taming Fc domain functions. Exp. Mol. Med. 2019, 51, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Charles, A.; Janeway, J.; Travers, P.; Walport, M.; Shlomchik, M.J. Immunobiology: The Immune System in Health and Disease, 5th ed.; Routledge: Oxfordshire, UK, 2001. [Google Scholar]
- Lu, R.-M.; Hwang, Y.-C.; Liu, I.-J.; Lee, C.-C.; Tsai, H.-Z.; Li, H.-J.; Wu, H.-C. Development of Therapeutic Antibodies for the Treatment of Diseases. J. Biomed. Sci. 2020, 27, 1. [Google Scholar] [CrossRef]
- Monoclonal Antibodies (MAbS) Market Analysis, Size and Trends Global Forecast to 2022–2030. Available online: https://www.grandviewresearch.com/industry-analysis/monoclonal-antibodies-market (accessed on 14 July 2022).
- Kaplon, H.; Chenoweth, A.; Crescioli, S.; Reichert, J.M. Antibodies to Watch in 2022. In MAbs; Taylor & Francis: Oxfordshire, UK, 2022; Volume 14. [Google Scholar] [CrossRef]
- Xenaki, K.T.; Oliveira, S.; van Bergen en Henegouwen, P.M.P. Antibody or Antibody Fragments: Implications for Molecular Imaging and Targeted Therapy of Solid Tumors. Front. Immunol. 2017, 8, 1287. [Google Scholar] [CrossRef] [Green Version]
- Nelson, A.L.; Reichert, J.M. Development Trends for Therapeutic Antibody Fragments. Nat. Biotechnol. 2009, 27, 331–337. [Google Scholar] [CrossRef]
- Steeland, S.; Vandenbroucke, R.E.; Libert, C. Nanobodies as Therapeutics: Big Opportunities for Small Antibodies. Drug Discov. Today 2016, 21, 1076–1113. [Google Scholar] [CrossRef]
- Yusakul, G.; Sakamoto, S.; Pongkitwitoon, B.; Tanaka, H.; Morimoto, S. Effect of Linker Length between Variable Domains of Single Chain Variable Fragment Antibody against Daidzin on Its Reactivity. Biosci. Biotechnol. Biochem. 2016, 80, 1306–1312. [Google Scholar] [CrossRef]
- The Antibody Society Antibody Therapeutics Approved or in Regulatory Review in the EU or US. Available online: https://www.antibodysociety.org/resources/approved-antibodies/ (accessed on 23 August 2022).
- Charpentier, I.; Pillet, L.; Karlsson, E.; Couderc, J.; Ménez, A. Recognition of the Acetylcholine Receptor Binding Site of a Long-Chain Neurotoxin by Toxin-Specific Monoclonal Antibodies. J. Mol. Recognit. 1990, 3, 74–81. [Google Scholar] [CrossRef]
- Engmark, M.; Lomonte, B.; Gutiérrez, J.M.; Laustsen, A.H.; de Masi, F.; Andersen, M.R.; Lund, O. Cross-Recognition of a Pit Viper (Crotalinae) Polyspecific Antivenom Explored through High-Density Peptide Microarray Epitope Mapping. PLoS Negl. Trop. Dis. 2017, 11, e0005768. [Google Scholar] [CrossRef]
- Lafaye, P.; Choumet, V.; Demangel, C.; Bon, C.; Mazié, J.-C. Biologically Active Human Anti-Crotoxin ScFv Isolated from a Semi-Synthetic Phage Library. Immunotechnology 1997, 3, 117–125. [Google Scholar] [CrossRef]
- Laustsen, A.H.; María Gutiérrez, J.; Knudsen, C.; Johansen, K.H.; Bermúdez-Méndez, E.; Cerni, F.A.; Jürgensen, J.A.; Ledsgaard, L.; Martos-Esteban, A.; Øhlenschlæger, M.; et al. Pros and Cons of Different Therapeutic Antibody Formats for Recombinant Antivenom Development. Toxicon 2018, 146, 151–175. [Google Scholar] [CrossRef]
- Visser, L.E.; Kyei-Faried, S.; Belcher, D.W.; Geelhoed, D.W.; van Leeuwen, J.S.; van Roosmalen, J. Failure of a New Antivenom to Treat Echis ocellatus Snake Bite in Rural Ghana: The Importance of Quality Surveillance. Trans. R. Soc. Trop. Med. Hyg. 2008, 102, 445–450. [Google Scholar] [CrossRef]
- Habib, A.G.; Onayade, A.A.; Yakubu, A.; Nasidi, A.; Theakston, R.D.G.; Smith, D.C.; Daudu, I.J.; Warrell, D.A.; Meyer, W.P. First Clinical Experiences with a New Ovine Fab Echis ocellatus Snake Bite Antivenom in Nigeria: Randomized Comparative Trial with Institute Pasteur Serum (Ipser) Africa Antivenom. Am. J. Trop. Med. Hyg. 1997, 56, 291–300. [Google Scholar] [CrossRef]
- León, G.; Herrera, M.; Segura, Á.; Villalta, M.; Vargas, M.; Gutiérrez, J.M. Pathogenic Mechanisms Underlying Adverse Reactions Induced by Intravenous Administration of Snake Antivenoms. Toxicon 2013, 76, 63–76. [Google Scholar] [CrossRef]
- Squaiella-Baptistão, C.C.; Gutiérrez, J.M.; Tambourgi, D.V.; Marcelino, J.R.; Ribeiro da Cunha, L.E. Anticomplementary Activity of Horse IgG and F(Ab’)2 Antivenoms. Am. J. Trop. Med. Hyg. 2014, 90, 574–584. [Google Scholar] [CrossRef] [PubMed]
- Battellino, C.; Piazza, R.; da Silva, A.M.M.; Cury, Y.; Farsky, S.H.P. Assessment of Efficacy of Bothropic Antivenom Therapy on Microcirculatory Effects Induced by Bothrops jararaca Snake Venom. Toxicon 2003, 41, 583–593. [Google Scholar] [CrossRef]
- Williams, D.J.; Gutiérrez, J.-M.; Calvete, J.J.; Wüster, W.; Ratanabanangkoon, K.; Paiva, O.; Brown, N.I.; Casewell, N.R.; Harrison, R.A.; Rowley, P.D.; et al. Ending the Drought: New Strategies for Improving the Flow of Affordable, Effective Antivenoms in Asia and Africa. J. Proteom. 2011, 74, 1735–1767. [Google Scholar] [CrossRef]
- Fasoli, E.; Sanz, L.; Wagstaff, S.; Harrison, R.A.; Righetti, P.G.; Calvete, J.J. Exploring the Venom Proteome of the African Puff Adder, Bitis arietans, Using a Combinatorial Peptide Ligand Library Approach at Different PHs. J. Proteom. 2010, 73, 932–942. [Google Scholar] [CrossRef]
- Ferreira, L.A.F.; Henriques, O.B.; Andreoni, A.A.S.; Vital, G.R.F.; Campos, M.M.C.; Habermehl, G.G.; de Moraes, V.L.G. Antivenom and Biological Effects of Ar-Turmerone Isolated from Curcuma Longa (Zingiberaceae). Toxicon 1992, 30, 1211–1218. [Google Scholar] [CrossRef]
- Silva, W.D.D.; Guidolin, R.; Raw, I.; Higashi, H.G.; Caricati, C.P.; Morais, J.F.; Lima, M.L.S.; Yamaguchi, I.K.; Nishikawa, A.K.; Stephano, M.A.; et al. Cross-reactivity of horse monovalent antivenoms to venoms of ten Bothrops species. Mem. Inst. Butantan 1989, 51, 153–168. [Google Scholar]
- Sousa, L.F.; Nicolau, C.A.; Peixoto, P.S.; Bernardoni, J.L.; Oliveira, S.S.; Portes-Junior, J.A.; Mourão, R.H.V.; Lima-dos-Santos, I.; Sano-Martins, I.S.; Chalkidis, H.M.; et al. Comparison of Phylogeny, Venom Composition and Neutralization by Antivenom in Diverse Species of Bothrops Complex. PLoS Negl. Trop. Dis. 2013, 7, e2442. [Google Scholar] [CrossRef]
- Guidolin, F.R.; Caricati, C.P.; Marcelino, J.R.; da Silva, W.D. Development of Equine IgG Antivenoms against Major Snake Groups in Mozambique. PLoS Negl. Trop. Dis. 2016, 10, e0004325. [Google Scholar] [CrossRef] [PubMed]
- Tahis, L.D.S.; Magnóli, F.C.; Dias da Silva, W. Characterization of a Hemorrhage-Inducing Component Present in Bitis arietans Venom. Afr. J. Biotechnol. 2015, 14, 999–1008. [Google Scholar] [CrossRef]
- Freitas, T.V.; Fortes-Dias, C.L.; Diniz, C.R.; Velarde, D.T.; Freitas, C.F. Immunization of Horses with Crotalus durissus Terrificus (South American Rattlesnake) Venom. A Comparison of Four Different Procedures. Braz. J. Med. Biol. Res. 1991, 24, 281–290. [Google Scholar] [PubMed]
- Almeida, C.M.C.; Kanashiro, M.M.; Rangel Filho, F.B.; Mata, M.F.R.; Kipnis, T.L.; Dias da Silva, W. Development of Snake Antivenom Antibodies in Chickens and Their Purification from Yolk. Vet. Rec. 1998, 143, 579–584. [Google Scholar] [CrossRef]
- Guidlolin, R.G.; Marcelino, R.M.; Gondo, H.H.; Morais, J.F.; Ferreira, R.A.; Silva, C.L.; Kipnis, T.L.; Silva, J.A.; Fafetine, J.; da Silva, W.D. Polyvalent horse F(Ab’)2 snake antivenom: Development of process to produce polyvalent horse F(Ab’)2 antibodies anti-african snake venom. Afr. J. Biotechnol. 2010, 9, 2446–2455. [Google Scholar]
- Alvarenga, L.; Zahid, M.; Tommaso, A.; Juste, M.; Aubrey, N.; Billiald, P.; Muzard, J. Engineering Venom’s Toxin-Neutralizing Antibody Fragments and Its Therapeutic Potential. Toxins 2014, 6, 2541–2567. [Google Scholar] [CrossRef]
- Guidolin, F.R.; Tambourgi, D.V.; Guidolin, R.; Marcelino, J.R.; Okamoto, C.K.; Magnoli, F.C.; Queiroz, G.P.; Dias da Silva, W. Characterization of Anti-Crotalic Antibodies. Toxicon 2013, 66, 7–17. [Google Scholar] [CrossRef]
- Kuniyoshi, A.K.; Kodama, R.T.; Cajado-Carvalho, D.; Iwai, L.K.; Kitano, E.; da Silva, C.C.F.; Duzzi, B.; Dias da Silva, W.; Portaro, F.C. Experimental Antivenom against Serine Proteases from the Bothrops jararaca Venom Obtained in Mice, and Its Comparison with the Antibothropic Serum from the Butantan Institute. Toxicon 2019, 169, 59–67. [Google Scholar] [CrossRef]
- Laustsen, A.H. Toxin Synergism in Snake Venoms. Toxin Rev. 2016, 35, 165–170. [Google Scholar] [CrossRef]
- Calvete, J.J. Venomics: Integrative Venom Proteomics and Beyond. Biochem. J. 2017, 474, 611–634. [Google Scholar] [CrossRef]
- Boulain, J.C.; Menez, A.; Couderc, J.; Faure, G.; Liacopoulos, P.; Fromageot, P. Neutralizing Monoclonal Antibody Specific for Naja nigricollis Toxin.Alpha.: Preparation, Characterization and Localization of the Antigenic Binding Site. Biochemistry 1982, 21, 2910–2915. [Google Scholar] [CrossRef]
- Jianxin, M.; John, T.R.; Kaiser, I.I. Specificity and Binding Affinity of an Anti-Crotoxin Combinatorial Antibody Selected from a Phage-Displayed Library. Biochem. Pharm. 1995, 50, 1969–1977. [Google Scholar] [CrossRef]
- Hansel, T.T.; Kropshofer, H.; Singer, T.; Mitchell, J.A.; George, A.J.T. The Safety and Side Effects of Monoclonal Antibodies. Nat. Rev. Drug Discov. 2010, 9, 325–338. [Google Scholar] [CrossRef] [PubMed]
- Morine, N.; Matsuda, S.; Terada, K.; Eto, A.; Ishida, I.; Oku, H. Neutralization of Hemorrhagic Snake Venom Metalloproteinase HR1a from Protobothrops flavoviridis by Human Monoclonal Antibody. Toxicon 2008, 51, 345–352. [Google Scholar] [CrossRef]
- Iddon, D.; Hommel, M.; Theakston, R.D.G. Characterisation of a Monoclonal Antibody Capable of Neutralising the Haemorrhagic Activity of West African Echis carinatus (Carpet Viper) Venom. Toxicon 1988, 26, 167–179. [Google Scholar] [CrossRef]
- Perez, J.C.; Garcia, V.E.; Huang, S.Y. Production of a Monoclonal Antibody against Hemorrhagic Activity of Crotalus atrox (Western Diamondback Rattlesnake) Venom. Toxicon 1984, 22, 967–973. [Google Scholar] [CrossRef]
- Ledsgaard, L.; Kilstrup, M.; Karatt-Vellatt, A.; McCafferty, J.; Laustsen, A. Basics of Antibody Phage Display Technology. Toxins 2018, 10, 236. [Google Scholar] [CrossRef]
- Frauches, T.S.; Petretski, J.H.; Arnholdt, A.C.V.; Lasunskaia, E.B.; de Carvalho, E.C.Q.; Kipnis, T.L.; da Silva, W.D.; Kanashiro, M.M. Bothropic Antivenom Based on Monoclonal Antibodies, Is It Possible? Toxicon 2013, 71, 49–56. [Google Scholar] [CrossRef] [PubMed]
- Schneider, F.S.; Nguyen, D.L.; Castro, K.L.; Cobo, S.; Machado de Avila, R.A.; Ferreira, N. de A.; Sanchez, E.F.; Nguyen, C.; Granier, C.; Galéa, P.; et al. Use of a Synthetic Biosensor for Neutralizing Activity-Biased Selection of Monoclonal Antibodies against Atroxlysin-I, an Hemorrhagic Metalloproteinase from Bothrops atrox Snake Venom. PLoS Negl. Trop. Dis. 2014, 8, e2826. [Google Scholar] [CrossRef]
- Lomonte, B.; Kahan, L. Production and Partial Characterization of Monoclonal Antibodies to Bothrops asper (Terciopelo) Myotoxin. Toxicon 1988, 26, 675–689. [Google Scholar] [CrossRef]
- Fernandes, I.; Assumpção, G.G.; Silveira, C.R.F.; Faquim-Mauro, E.L.; Tanjoni, I.; Carmona, A.K.; Alves, M.F.M.; Takehara, H.A.; Rucavado, A.; Ramos, O.H.P.; et al. Immunochemical and Biological Characterization of Monoclonal Antibodies against BaP1, a Metalloproteinase from Bothrops asper Snake Venom. Toxicon 2010, 56, 1059–1065. [Google Scholar] [CrossRef] [PubMed]
- Guillon, V.; Alzari, P.M.; Grognet, J.-M. Preliminary Crystallographic Study of the Fab Fragment of a Monoclonal Antibody Directed against a Cobra Cardiotoxin. J. Mol. Biol. 1986, 189, 723–724. [Google Scholar] [CrossRef]
- Yang, C.C.; Chan, H.L. Preparation and Characterization of Β1-Bungarotoxin Bispecific Monoclonal Antibody. IUBMB Life 1999, 47, 1039–1048. [Google Scholar] [CrossRef] [PubMed]
- Cardoso, N.; England, F.; Vaughan, M.; Lafaye, P. Neutralizing Human Anti Crotoxin ScFv Isolated from a Nonimmunized Phage Library. Scand. J. Immunol. 2000, 51, 337–344. [Google Scholar] [CrossRef]
- Castro, J.M.A.; Oliveira, T.S.; Silveira, C.R.F.; Caporrino, M.C.; Rodriguez, D.; Moura-da-Silva, A.M.; Ramos, O.H.P.; Rucavado, A.; Gutiérrez, J.M.; Magalhães, G.S.; et al. A Neutralizing Recombinant Single Chain Antibody, ScFv, against BaP1, A P-I Hemorrhagic Metalloproteinase from Bothrops asper Snake Venom. Toxicon 2014, 87, 81–91. [Google Scholar] [CrossRef]
- Kulkeaw, K.; Sakolvaree, Y.; Srimanote, P.; Tongtawe, P.; Maneewatch, S.; Sookrung, N.; Tungtrongchitr, A.; Tapchaisri, P.; Kurazono, H.; Chaicumpa, W. Human Monoclonal ScFv Neutralize Lethal Thai Cobra, Naja kaouthia, Neurotoxin. J. Proteom. 2009, 72, 270–282. [Google Scholar] [CrossRef]
- Lee, C.-H.; Lee, Y.-C.; Liang, M.-H.; Leu, S.-J.; Lin, L.-T.; Chiang, J.-R.; Yang, Y.-Y. Antibodies against Venom of the Snake Deinagkistrodon acutus. Appl. Environ. Microbiol. 2016, 82, 71–80. [Google Scholar] [CrossRef]
- Oliveira, J.G.; Soares, S.G.; Soares, A.M.; Giglio, J.R.; Teixeira, J.E.; Barbosa, J.E. Expression of Human Recombinant Antibody Fragments Capable of Partially Inhibiting the Phospholypase Activity of Crotalus Durissus Terrificus Venom. Basic Clin. Pharm. Toxicol. 2009, 105, 84–91. [Google Scholar] [CrossRef]
- Roncolato, E.C.; Pucca, M.B.; Funayama, J.C.; Bertolini, T.B.; Campos, L.B.; Barbosa, J.E. Human Antibody Fragments Specific for Bothrops Jararacussu Venom Reduce the Toxicity of Other Bothrops sp. Venoms. J. Immunotoxicol 2013, 10, 160–168. [Google Scholar] [CrossRef]
- Silva, L.C.; Pucca, M.B.; Pessenda, G.; Campos, L.B.; Martinez, E.Z.; Cerni, F.A.; Barbosa, J.E. Discovery of Human ScFvs That Cross-Neutralize the Toxic Effects of B. jararacussu and C. d. Terrificus Venoms. Acta Trop. 2018, 177, 66–73. [Google Scholar] [CrossRef]
- Gomes, M.; Alvarez, M.A.; Quellis, L.R.; Becher, M.L.; Castro, J.M.D.A.; Gameiro, J.; Caporrino, M.C.; Moura-da-Silva, A.M.; de Oliveira Santos, M. Expression of an ScFv Antibody Fragment in Nicotiana Benthamiana and in Vitro Assessment of Its Neutralizing Potential against the Snake Venom Metalloproteinase BaP1 from Bothrops asper. Toxicon 2019, 160, 38–46. [Google Scholar] [CrossRef] [PubMed]
- Souza, J.B.; Cardoso, R.; Almeida-Souza, H.O.; Carvalho, C.P.; Correia, L.I.V.; Faria, P.C.B.; Araujo, G.R.; Mendes, M.M.; Rodrigues, R.S.; Rodrigues, V.M.; et al. Generation and In-Planta Expression of a Recombinant Single Chain Antibody with Broad Neutralization Activity on Bothrops pauloensis Snake Venom. Int. J. Biol. Macromol. 2020, 149, 1241–1251. [Google Scholar] [CrossRef] [PubMed]
- Julve Parreño, J.M.; Huet, E.; Fernández-del-Carmen, A.; Segura, A.; Venturi, M.; Gandía, A.; Pan, W.; Albaladejo, I.; Forment, J.; Pla, D.; et al. A Synthetic Biology Approach for Consistent Production of Plant-Made Recombinant Polyclonal Antibodies against Snake Venom Toxins. Plant Biotechnol. J. 2018, 16, 727–736. [Google Scholar] [CrossRef] [PubMed]
- Luiz, M.; Pereira, S.; Gonçalves, N.; Prado, N.; Dill, L.; Soares, A.; Zanchi, F.; Zuliani, J.; Stábeli, R.; Fernandes, C. Inhibition of the Myotoxicity Induced by Crotoxin B, from Crotalus durissus Terrificus Venom, by Camelid Nanobodies. In Proceedings of the Anais do III Simpósio Internacional de Imunobiológicos; Instituto de Tecnologia em Imunobiológicos; Bio-Manguinhos: Rio de Janeiro, Brazil, 2016; pp. 80–81. [Google Scholar]
- Bailon Calderon, H.; Yaniro Coronel, V.O.; Cáceres Rey, O.A.; Colque Alave, E.G.; Leiva Duran, W.J.; Padilla Rojas, C.; Montejo Arevalo, H.; García Neyra, D.; Galarza Pérez, M.; Bonilla, C.; et al. Development of Nanobodies Against Hemorrhagic and Myotoxic Components of Bothrops atrox Snake Venom. Front. Immunol. 2020, 11, 655. [Google Scholar] [CrossRef] [PubMed]
- Stewart, C.S.; MacKenzie, C.R.; Christopher Hall, J. Isolation, Characterization and Pentamerization of α-Cobrotoxin Specific Single-Domain Antibodies from a Naïve Phage Display Library: Preliminary Findings for Antivenom Development. Toxicon 2007, 49, 699–709. [Google Scholar] [CrossRef]
- Chavanayarn, C.; Thanongsaksrikul, J.; Thueng-in, K.; Bangphoomi, K.; Sookrung, N.; Chaicumpa, W. Humanized-Single Domain Antibodies (VH/VHH) That Bound Specifically to Naja kaouthia Phospholipase A2 and Neutralized the Enzymatic Activity. Toxins 2012, 4, 554–567. [Google Scholar] [CrossRef]
- Richard, G.; Meyers, A.J.; McLean, M.D.; Arbabi-Ghahroudi, M.; MacKenzie, R.; Hall, J.C. In Vivo Neutralization of α-Cobratoxin with High-Affinity Llama Single-Domain Antibodies (VHHs) and a VHH-Fc Antibody. PLoS ONE 2013, 8, e69495. [Google Scholar] [CrossRef]
- Prado, N.D.R.; Pereira, S.S.; da Silva, M.P.; Morais, M.S.S.; Kayano, A.M.; Moreira-Dill, L.S.; Luiz, M.B.; Zanchi, F.B.; Fuly, A.L.; EF Huacca, M.; et al. Inhibition of the Myotoxicity Induced by Bothrops jararacussu Venom and Isolated Phospholipases A2 by Specific Camelid Single-Domain Antibody Fragments. PLoS ONE 2016, 11, e0151363. [Google Scholar] [CrossRef]
- Jenkins, T.P.; Laustsen, A.H. Cost of Manufacturing for Recombinant Snakebite Antivenoms. Front. Bioeng. Biotechnol. 2020, 8, 703. [Google Scholar] [CrossRef]
- Kris, A. Wetterstrand DNA Sequencing Costs: Data. Available online: https://www.genome.gov/about-genomics/fact-sheets/DNA-Sequencing-Costs-Data (accessed on 23 August 2022).
- Gray, A.C.; Sidhu, S.S.; Chandrasekera, P.C.; Hendriksen, C.F.M.; Borrebaeck, C.A.K. Animal-Friendly Affinity Reagents: Replacing the Needless in the Haystack. Trends Biotechnol. 2016, 34, 960–969. [Google Scholar] [CrossRef]
- Laustsen, A.H.; Greiff, V.; Karatt-Vellatt, A.; Muyldermans, S.; Jenkins, T.P. Animal Immunization, in Vitro Display Technologies, and Machine Learning for Antibody Discovery. Trends Biotechnol. 2021, 39, 1263–1273. [Google Scholar] [CrossRef] [PubMed]
- Laustsen, A.H.; Ainsworth, S.; Lomonte, B.; Kini, R.M.; Chávez-Olórtegui, C. Editorial: Novel Immunotherapies Against Envenomings by Snakes and Other Venomous Animals. Front. Immunol. 2020, 11, 1004. [Google Scholar] [CrossRef] [PubMed]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).