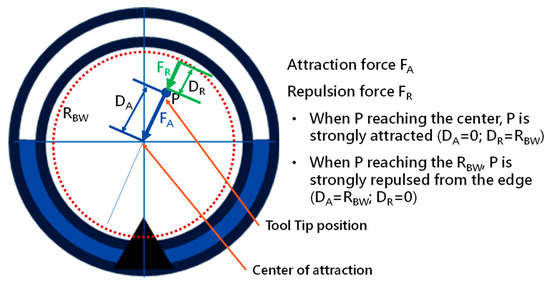
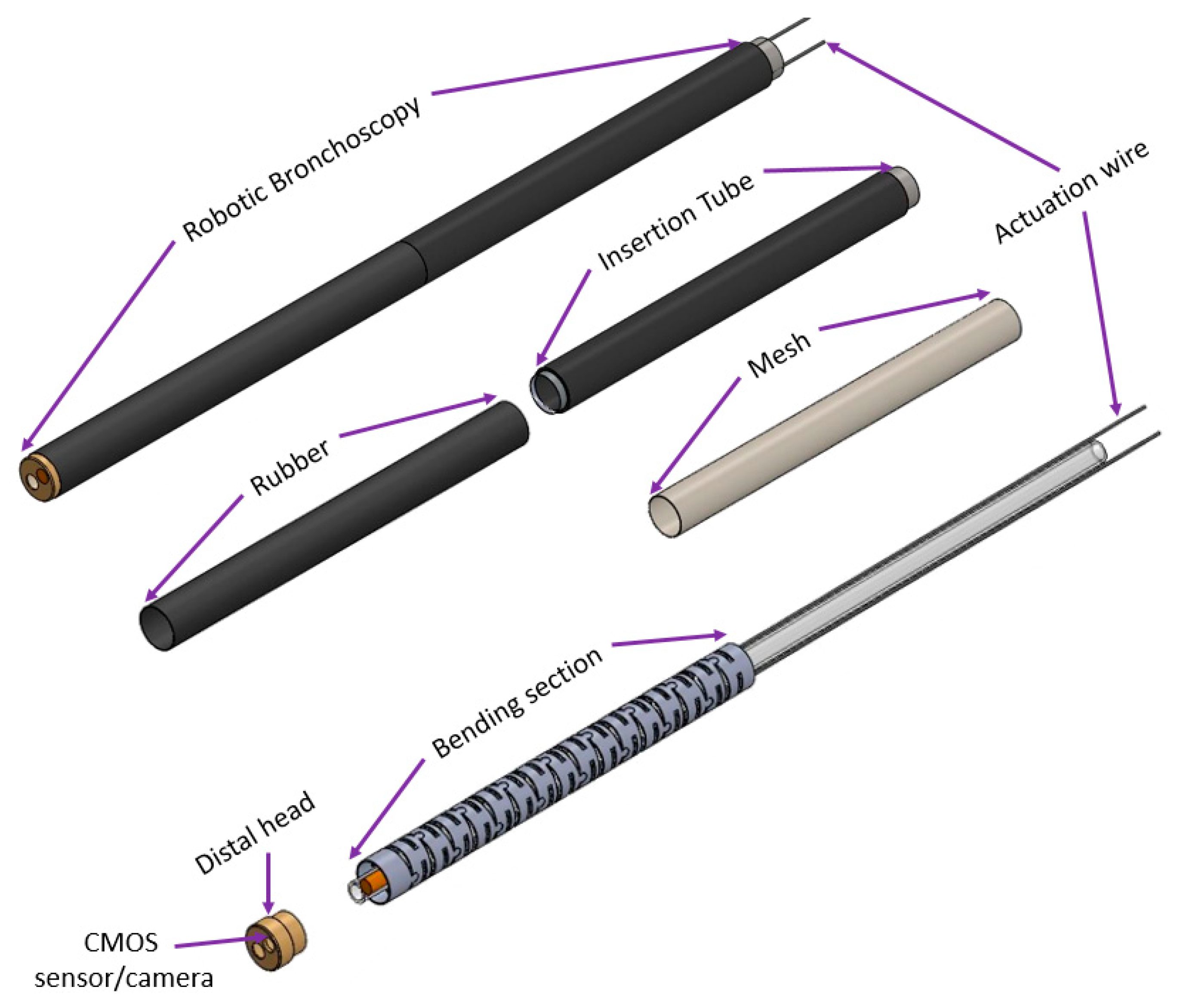
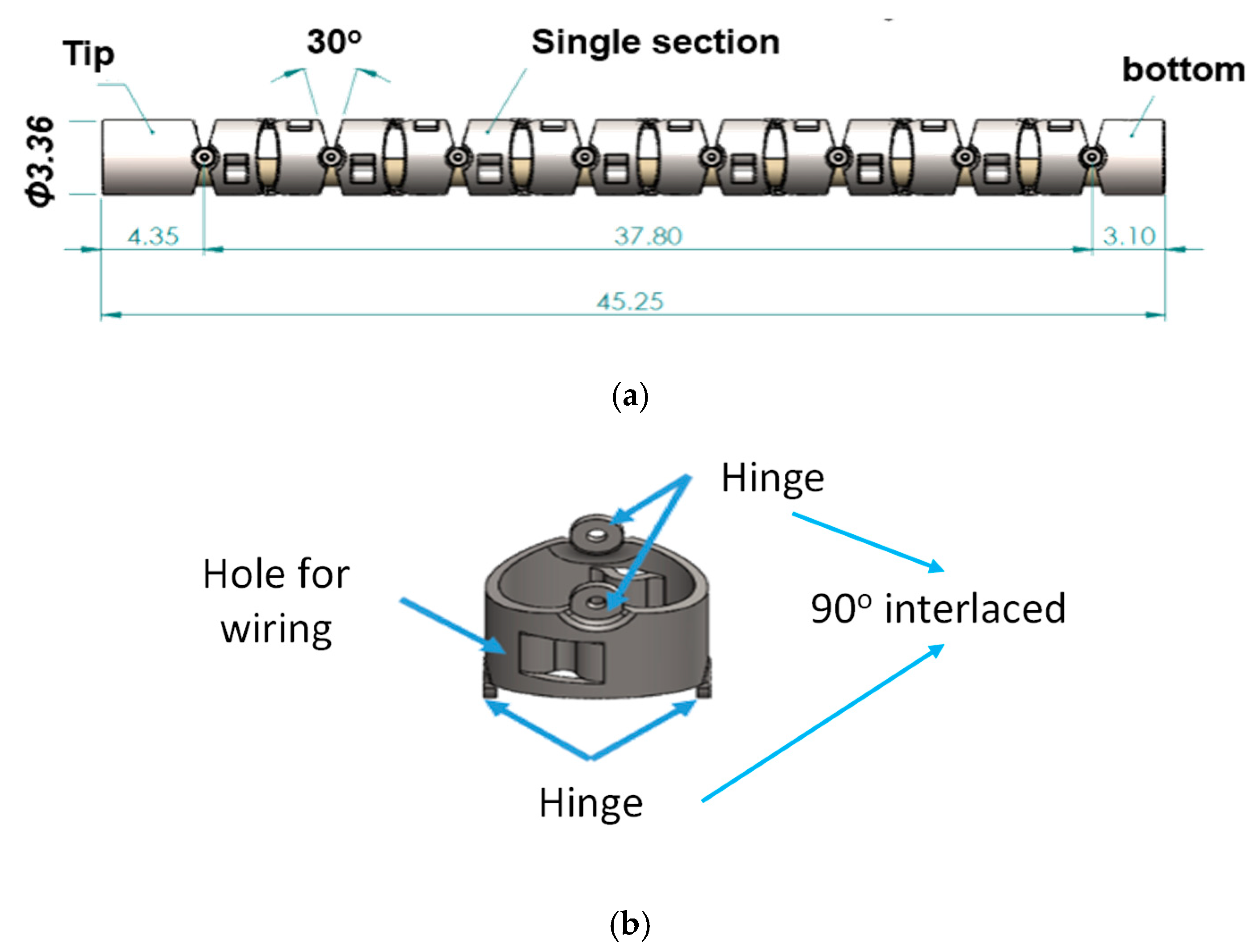
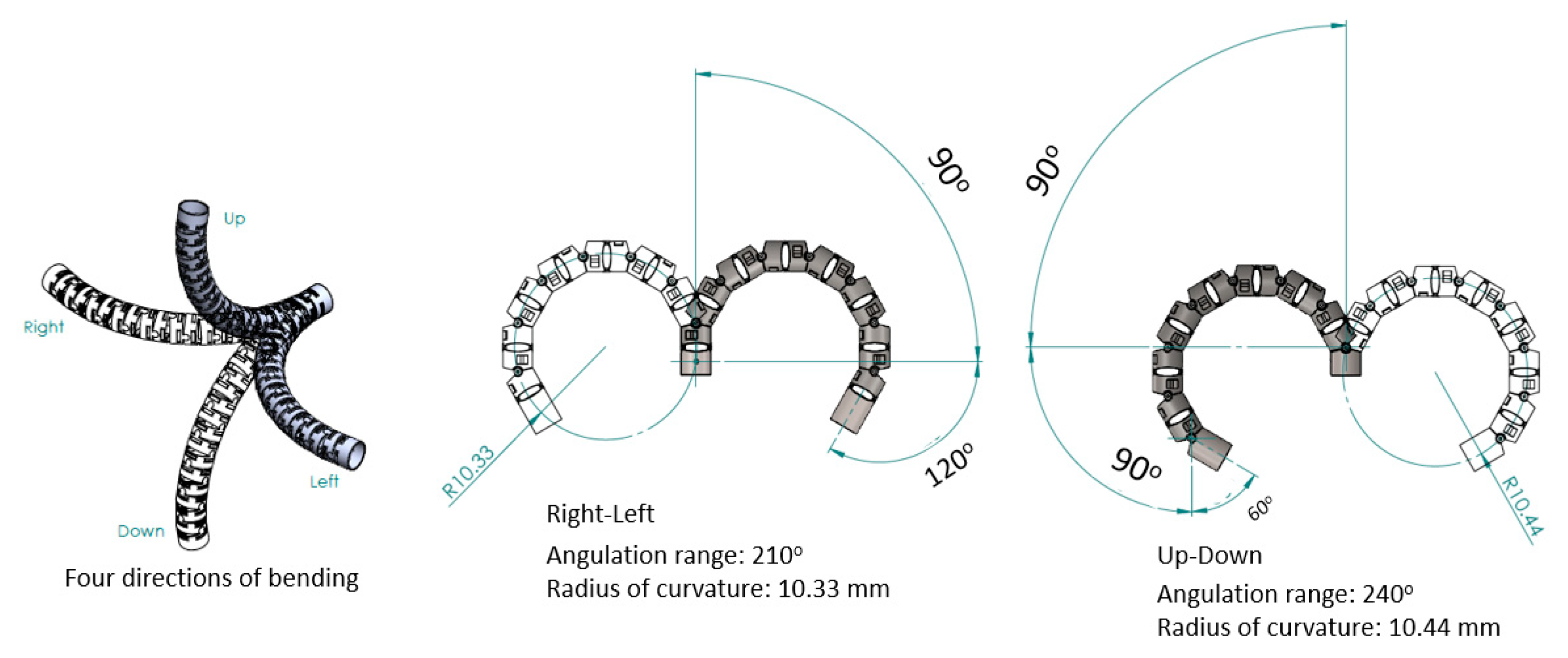
Abstract
The miniature serpentine robot can be applied to NOTES (Natural Orifice Transluminal Endoscopic Surgery). In this paper, a bronchoscopy application is addressed. This paper describes the basic mechanical design and control scheme of this miniature serpentine robotic bronchoscopy. In addition, off-line backward path planning and real-time and in situ forward navigation in this miniature serpentine robot are discussed. The proposed backward-path-planning algorithm utilizes the 3D model of a bronchial tree constructed from the synthetization of medical images such as images from CT (Computed Tomography), MRI (Magnetic Resonance Imaging), or X-ray, to define a series of nodes/events backward from the destination, for example, the lesion, to the original starting point, for example, the oral cavity. Accordingly, forward navigation is designed to make sure this series of nodes/events shall be passed/occur from the origin to the destination. This combination of backward-path planning and forward navigation does not require accurate positioning information of the tip of the miniature serpentine robot, which is where the CMOS bronchoscope is located. Collaboratively, a virtual force is introduced to maintain the tip of the miniature serpentine robot at the center of the bronchi. Results show that this method of path planning and navigation of the miniature serpentine robot for bronchoscopy applications works.
1. Introduction
In the years since 2020, the world has been experiencing the impact of the COVID-19 pandemic. According to global statistics from the World Health Organization, more than 750 million people have been confirmed infected, and this number of infected continues to increase. Although the symptoms caused by the COVID-19 infection can be reduced via vaccinations, there still exists uncured injuries located deep within the lungs, such as pulmonary fibrosis. In addition, COPD (Chronic Obstructive Pulmonary Disease) causes 1 death every 10 s globally. COPD is caused by long-term inflammation in the respiratory tract, resulting in the airflow via the respiratory tract being strongly obstructed (obstructive ventilatory dysfunction). Essentially, COPD includes Chronic Bronchitis and Emphysema, of which the symptoms are irreversible and cannot be cured.
As described above, the symptoms (pulmonary fibrosis and obstructive ventilatory dysfunction) caused by the COVID-19 infection and COPD cannot be cured because of the irreversible injuries located deep within the lungs and bronchi. Researchers have tried to utilize Mesenchymal Stem Cells, which have the abilities of self-regeneration and multilineage differentiation, to cure irreversible diseases [1,2,3,4]. Golchin, A. et al. [1] showed the effectiveness of MSC therapy on COVID-19 based on MSCs’ immunomodulatory and regenerative properties and concluded that cost-effective utilization of MSC therapy is a critical issue. Kim, K. et al. [2] demonstrated that an intravenous injection of MSC is safe and acceptable. Mallis, P. et al. [3] concluded that MSCs can successfully be activated by a COVID-19 patient’s serum and secrete anti-inflammatory cytokines and growth factors. P.M. George et al. [4] also showed the potential of MSC therapy for COVID-19.
On the other hand, Zhang, Y. et al. [5] adopted allogeneic adipose-derived mesenchymal stem cells (AMSCs) for COPD mice via intratracheal and intravenous administration. Their results [5] showed that MSC therapy for COPD is effective. In addition, ref. [5] showed that intratracheal administration of AMSCs is more effective compared to intravenous administration of AMSCs. Nejaddehbashi, F. et al. [6] also concluded that the anti-inflammatory effects of intratracheal AMSCs treatment are more potent than systemic administration for the treatment of emphysema.
Based on previous studies [1,2,3,4,5,6], adopting MSC therapy for the treatment of COVID-19 and COPD may be a potential treatment solution, while a critical issue will be how to effectively deliver MSCs directly to the lesion. Therefore, research on MSC delivery is emerging. Go, G. et al. [7] designed microrobots with an overall scaffold size of 357.55 ± 18.57 μm and pore sizes of 43.85 ± 13.39 μm, which can contain MSCs. These microrobots containing MSCs can be injected via a catheter needle into the knee for cartilage regeneration in vivo and be navigated by an Electro-Magnetic Actuation (EMA) system [7]. This mechanism cannot be adopted in the lungs for delivering MSCs to the lesion because penetration via the skin by a catheter needle to the lungs has a very high probability of causing risky pneumothorax.
Another resolution for delivering MSCs into the deep lungs (bronchi) could be a bronchoscopy via the human oral cavity. This kind of method depends heavily on the bronchoscopy operation experiences of the surgeons. Thus, studies and developments on robotic bronchoscopy have been conducted. Chen, A.C. et al. [8] showed the capability of a robotic bronchoscopy via the support of real-time images from a bronchoscope, electromagnetic navigation, and CT views. However, it still requires surgeons to control the movement of this robot-assisted bronchoscopy. This kind of serpentine robot [9,10,11,12] has been discussed as a feasible solution for moving inside the bronchi of the lungs.
Beyond the research on robotic bronchoscopy, path planning and navigation for this robotic bronchoscopy is a critical issue. Refs. [13,14] discussed several schemes to fulfill the functionalities of path planning and navigation. In [13], Ho, E. et al. reviewed several research studies related to path planning for a robotic bronchoscopy; for example, a bronchoscopist begins by reviewing the CT scan of the chest carefully and then writing down the plans to take—starting from the distal trachea to the main bronchus, lobar bronchus, segmental airway, and subsegmental airways, and finally the target. Another planning scheme mentioned in [13] is a computer-generated pathway where the bronchoscopist identifies and marks the target lesion and then the computer accordingly generates a pathway from the central airway to the target lesion. Regarding the navigation phase—guiding the robotics bronchoscopy while moving inside the bronchi—ref. [13] indicated that a synchronization procedure (Registration) between the position of the robotic bronchoscopy and the planned virtual pathway is required in advance. Without this synchronization, the robotic bronchoscopy could be navigated along the wrong path. Furthermore, ref. [13] described the significance of a live bronchoscopy view in order to correct the real-time movement of the robotic bronchoscopy. Duan, X., et al. [14] adopted an electromagnetic (EM) localization system integrated into the robotic bronchoscopy for further navigation.
After reviewing the research described above, we can draw the following conclusions: 1. MSC therapy for healing COVID-19 and COPD could be effective but requires a direct way to deliver the MSCs to the target in vivo; 2. adopting a miniature serpentine robot as a container for MSCs is an effective method of delivering MSCs directly to the target bronchi; accordingly, 3. path-planning and navigation inside the lungs become critical issues when an automatic robotic bronchoscopy is applied. Therefore, in this paper, a miniature serpentine robotic bronchoscopy is proposed. Section 2 of this paper will describe the basic mechanical design and control scheme of this proposed miniature serpentine robotic bronchoscopy. In addition, off-line backward path planning and real-time and in situ forward navigation of this miniature serpentine robot is discussed in Section 2. The implementation and results are described in Section 3. Then, Section 4 contains the discussion and conclusions.
3. System Implementation and Results
3.1. Implementation
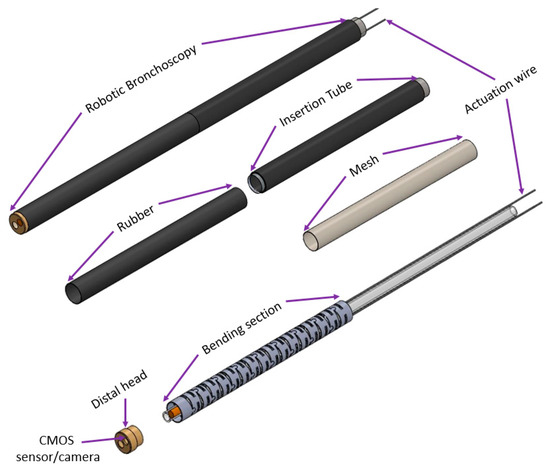
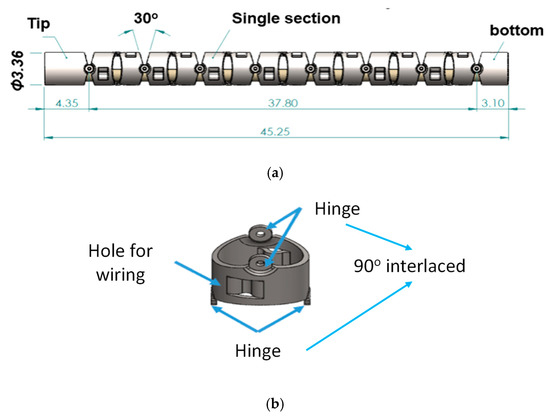
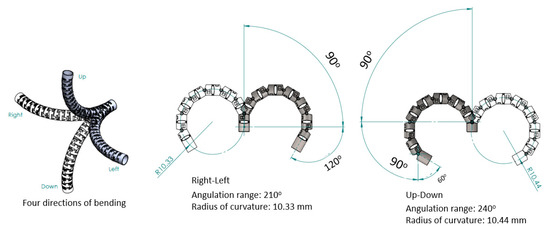
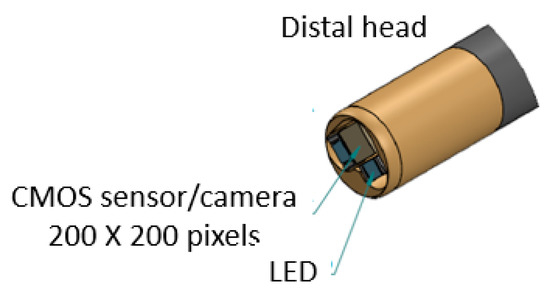
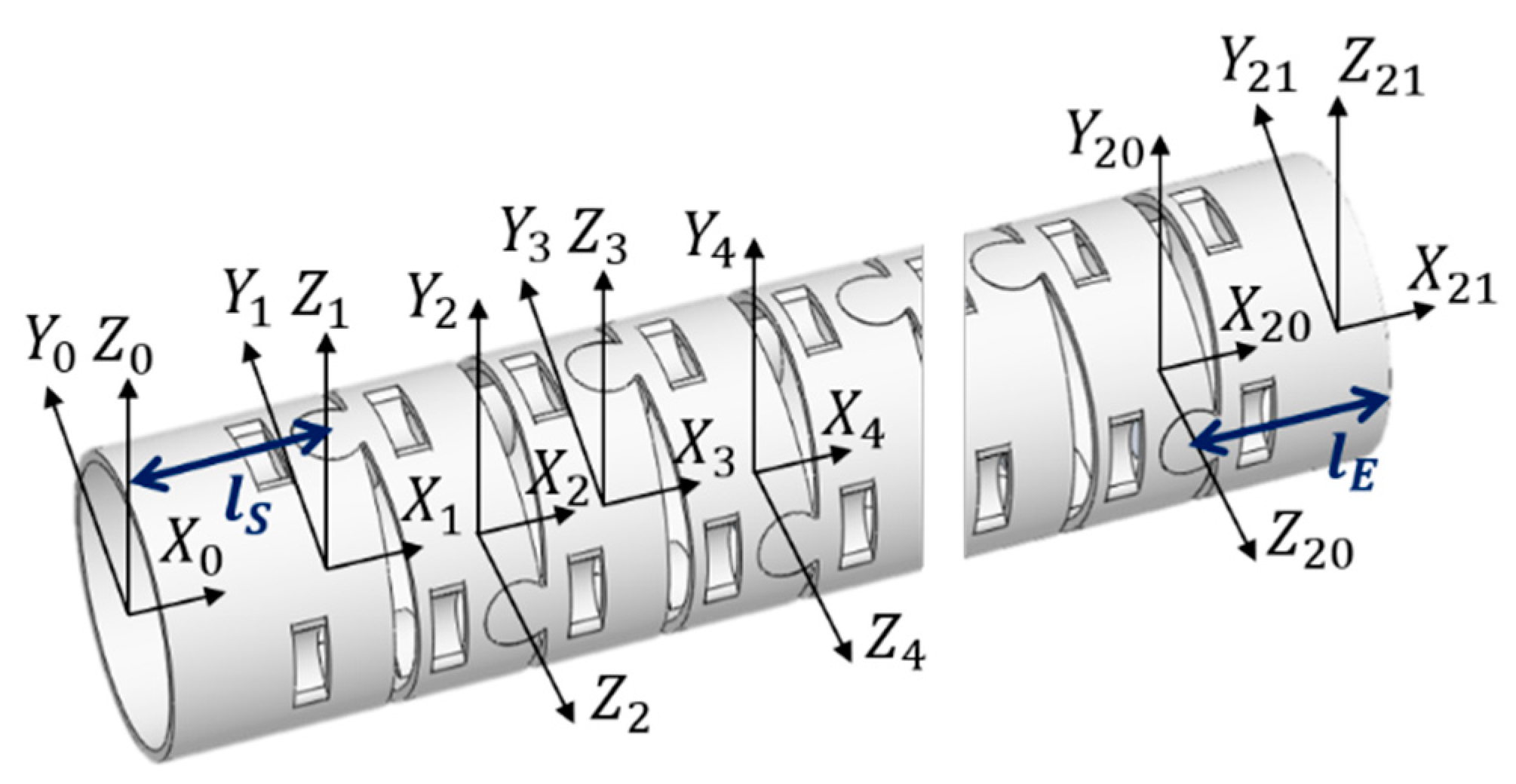
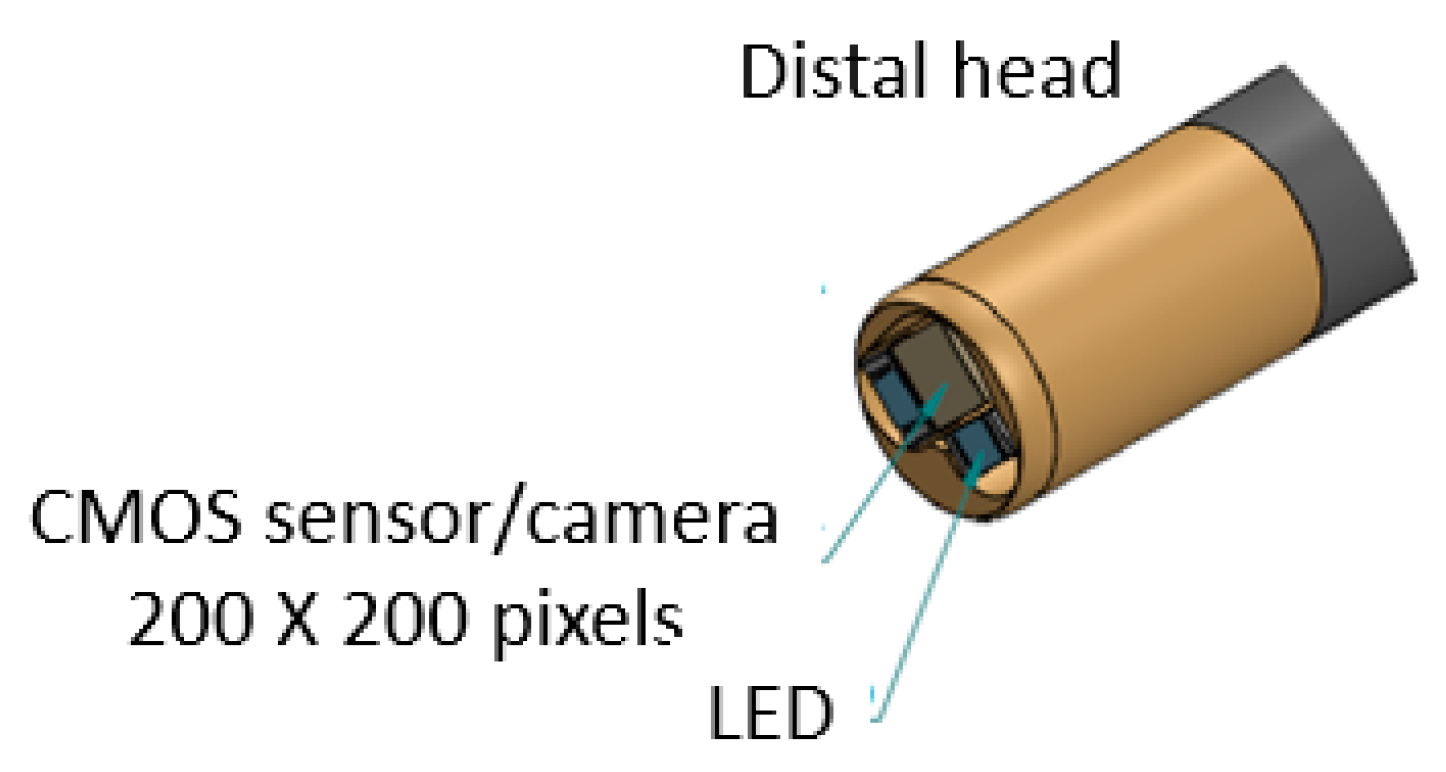
Based on the mechanical design described in Section 2.1, consecutive sub-sections of the bending section of the robotic bronchoscopy are manufactured by precise laser engraving of one piece of stainless-steel tube, of which the diameter can be selected to satisfy the size of the CMOS sensor/camera adopted. Four metal wires pass through the holes of each sub-section to actuate bending with any desired direction and angle. Figure 14 shows pictures of the bending sections with outer diameters of 3.36 mm and 4.0 mm, respectively, as well as the 3D orientation of this bending section. A CMOS sensor/camera (OV6948 with an optical size of 1/36″, 200 × 200 pixels, and 30 fps) is integrated into the distal head at the tip of the bending section. With reference to Figure 1, it displays the assembly of this robotic bronchoscopy. It shall be noted that SUS304 and SUS316L are applied as the materials of mechanical components to satisfy the FDA requirements for materials. Moreover, the overall manufacturing and assembly process will be GMP-certified. In addition, a gravitational indicator is added to our bronchoscopy implementation to indicate the horizontal level for determining the right or left side.

Figure 14.
Pictures of the bending section of the robotic bronchoscopy.
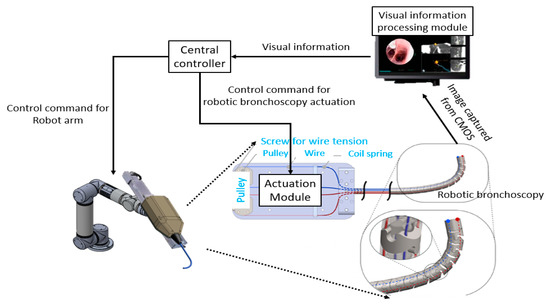
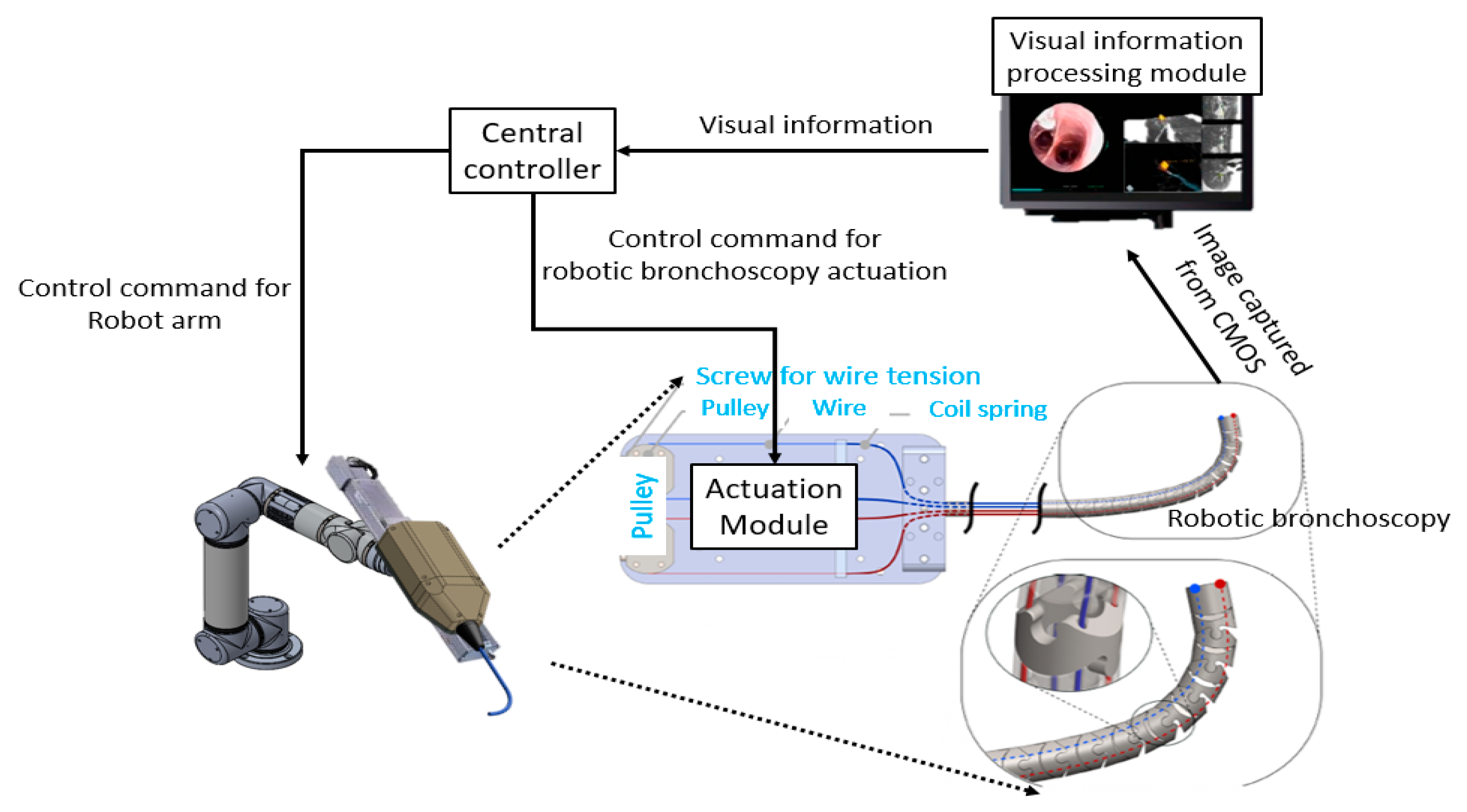
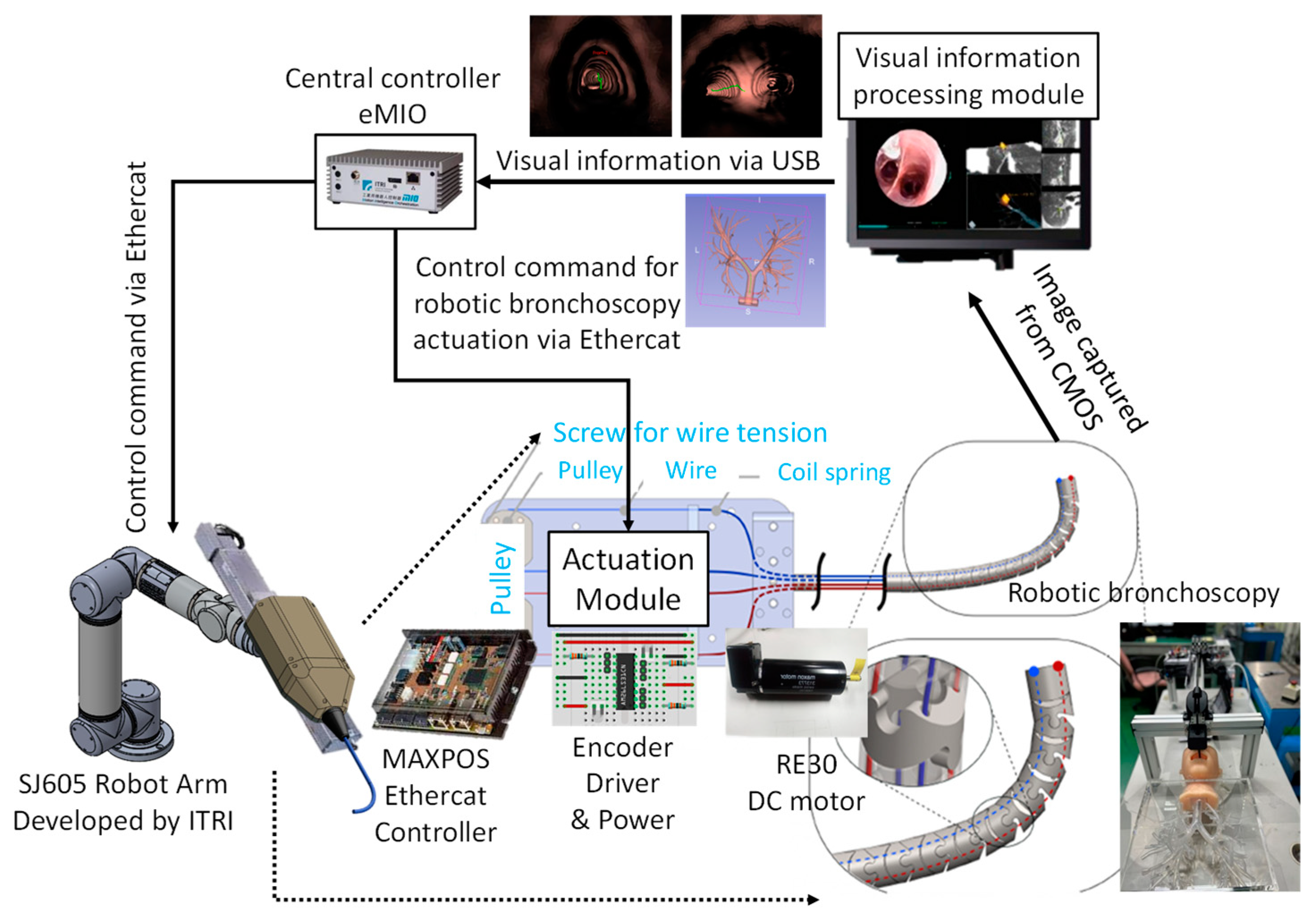
As per the implementation of the control scheme, the system diagram is shown in Figure 15. An eMIO controller is adopted as a central control coordinator to coordinate the movements of the robot arm and the proposed robotic bronchoscopy, primarily based on the image information from the CMOS sensor/camera and our proposed path planning and navigation algorithms. An SJ605 Collaborative Robot developed by ITRI is adopted in this system. Two RE30 DC motors and correlated drivers and Ethercat controllers are utilized to actuate the wires to control the bending section of the robotic bronchoscopy. Visual servo functionality is implemented to guide the tip of the bending section to aim at the desired position. In Section 3.2, simulated results of the proposed backward path planning and forward navigation are described.
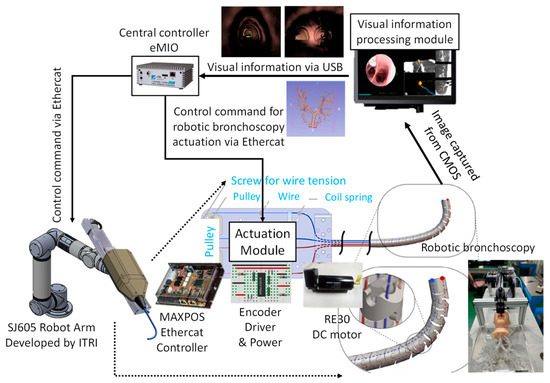
Figure 15.
System implementation diagram of the proposed robotic bronchoscopy.
3.2. Results
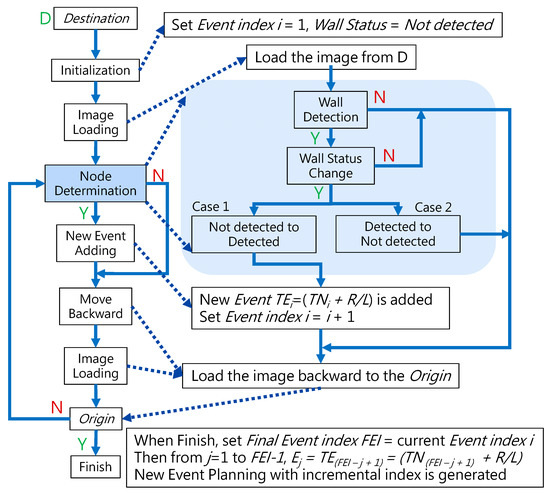
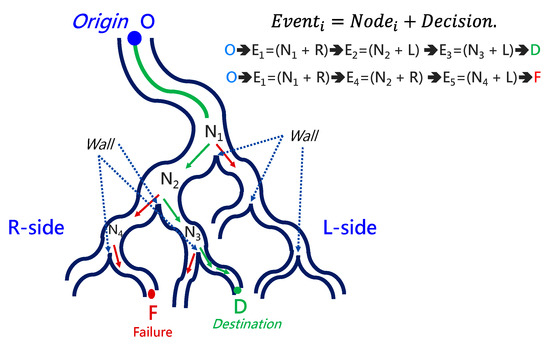
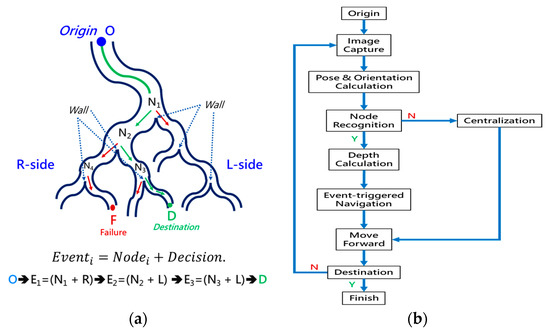
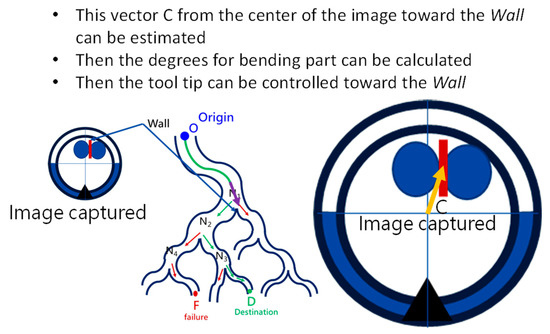
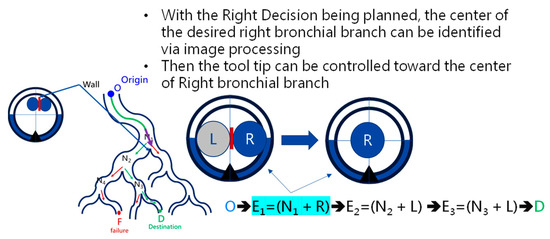
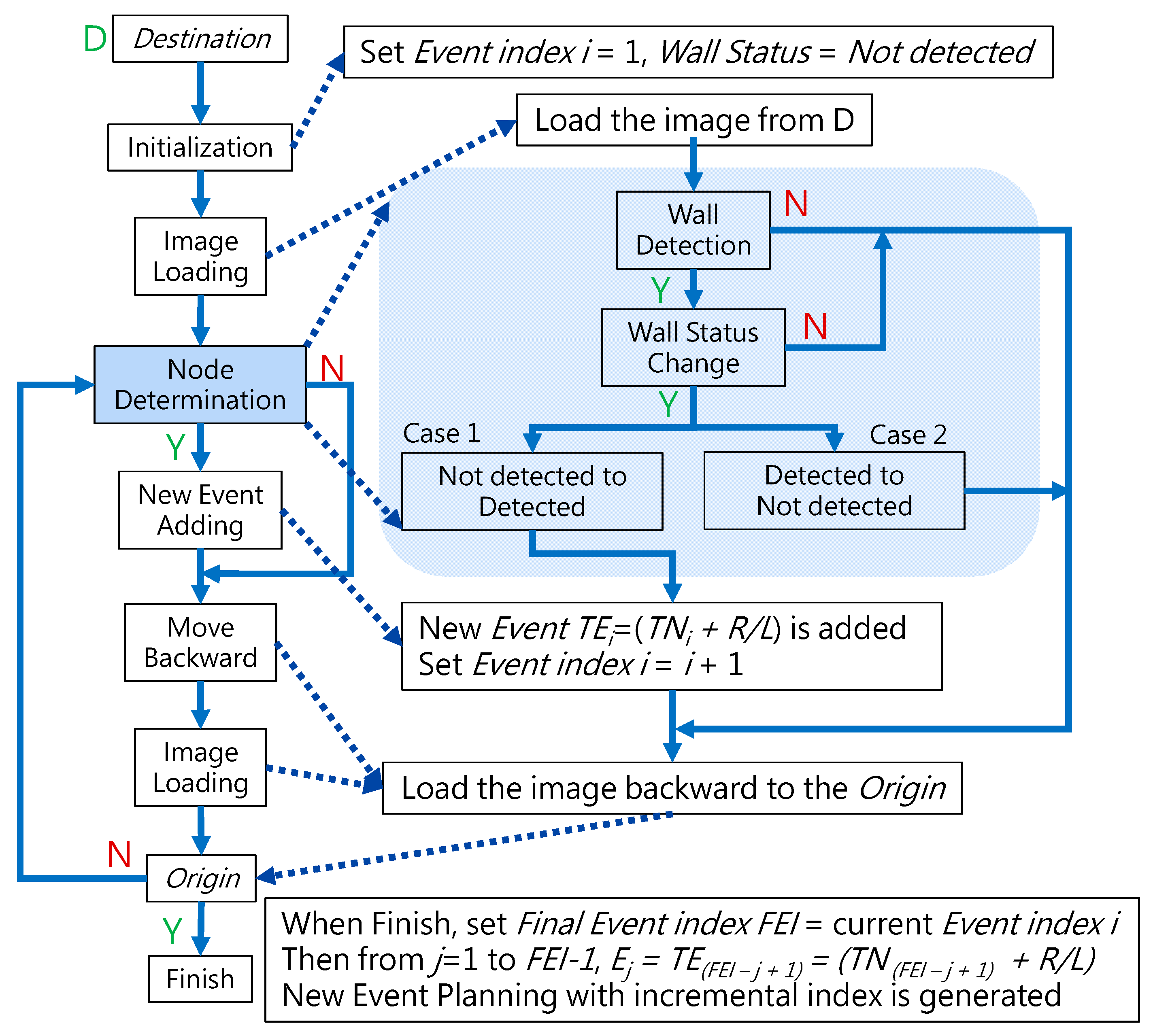
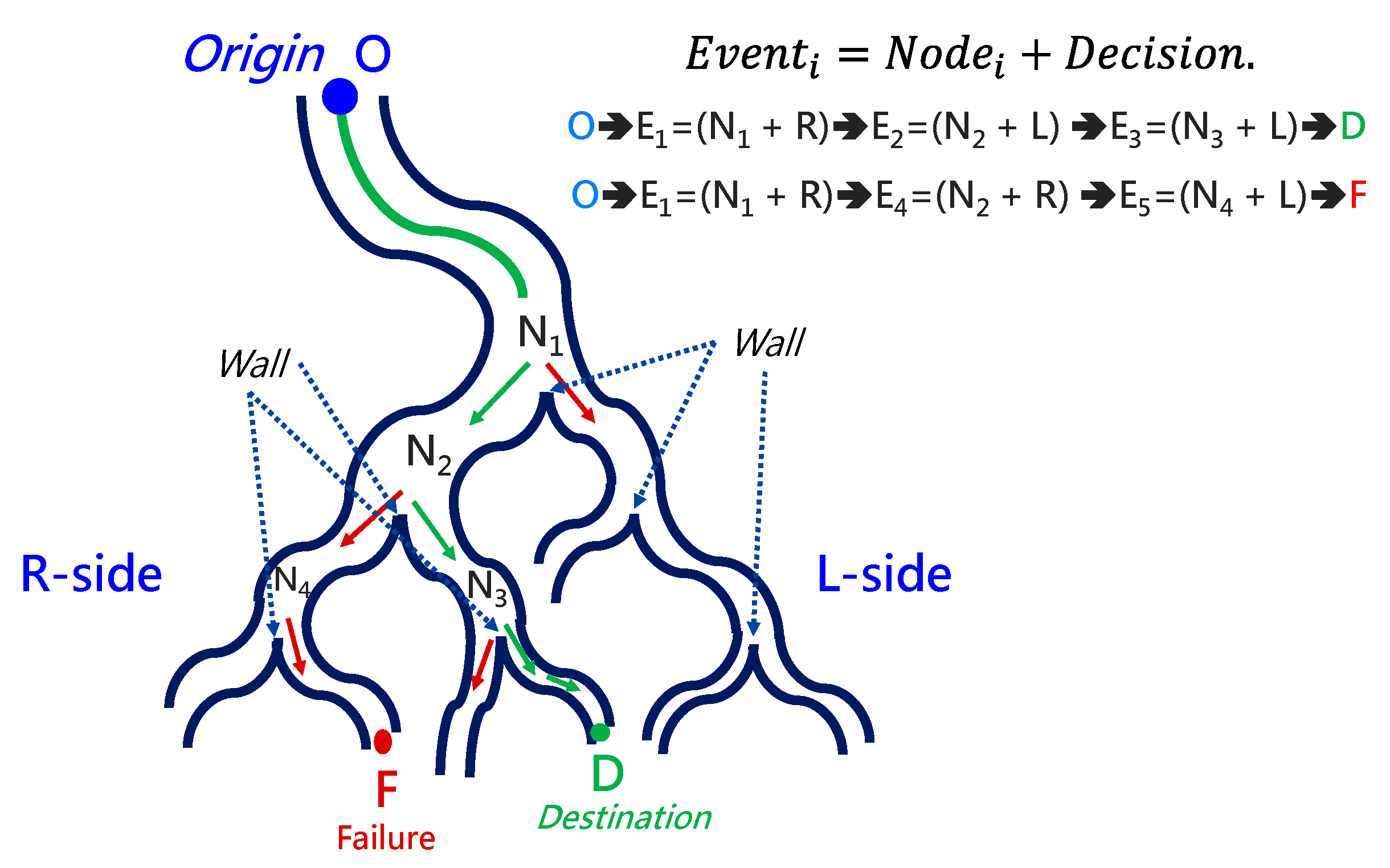
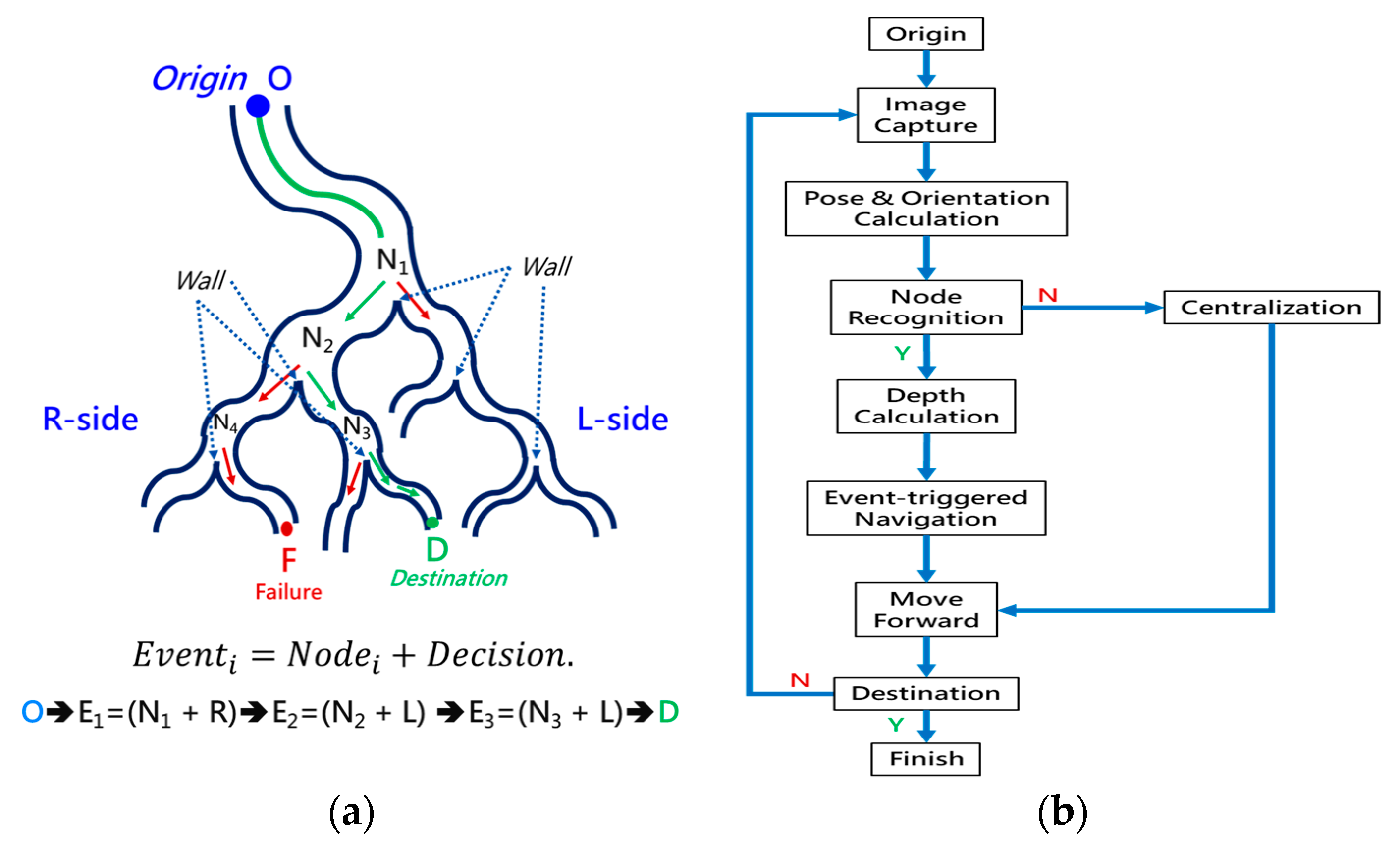
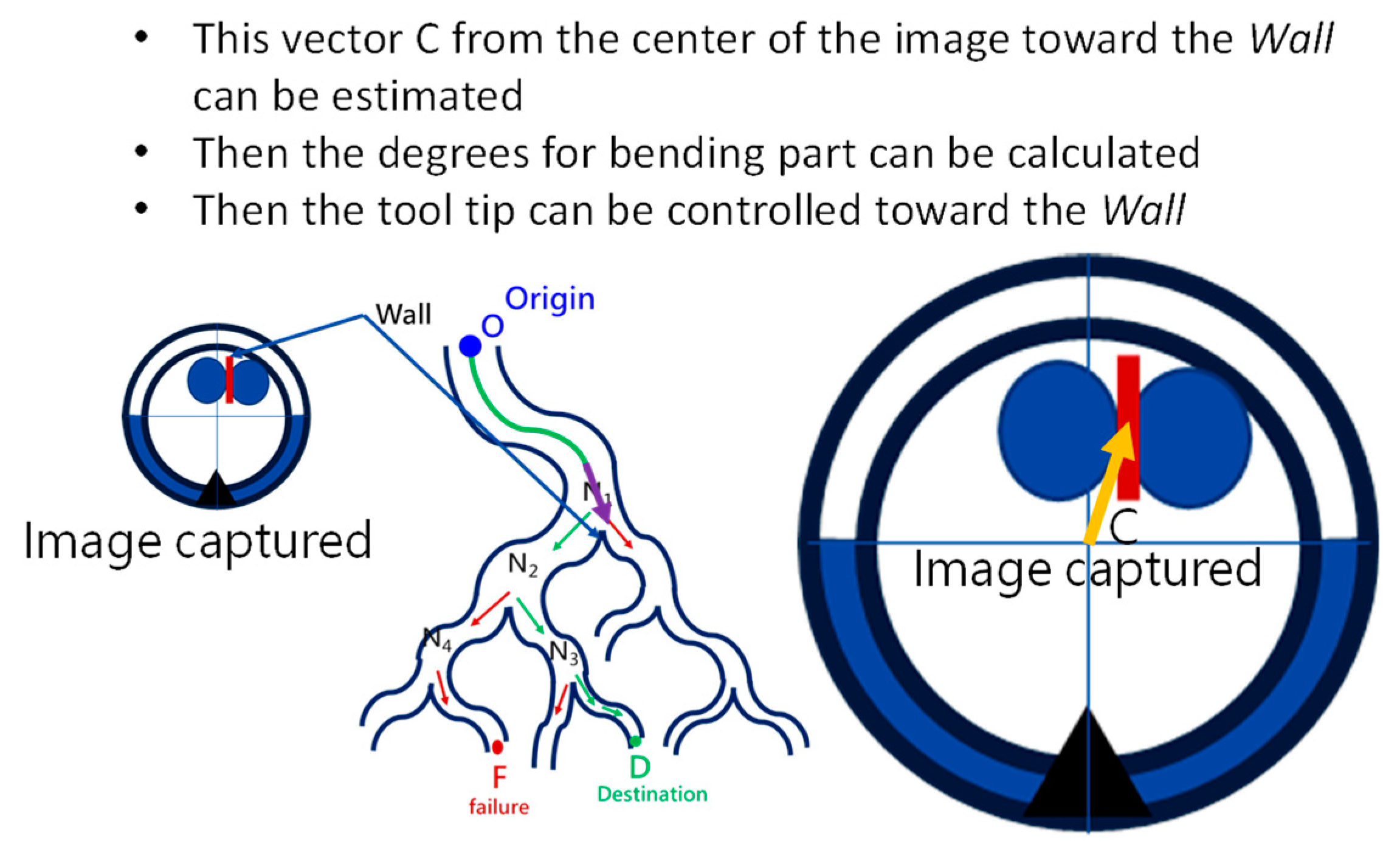
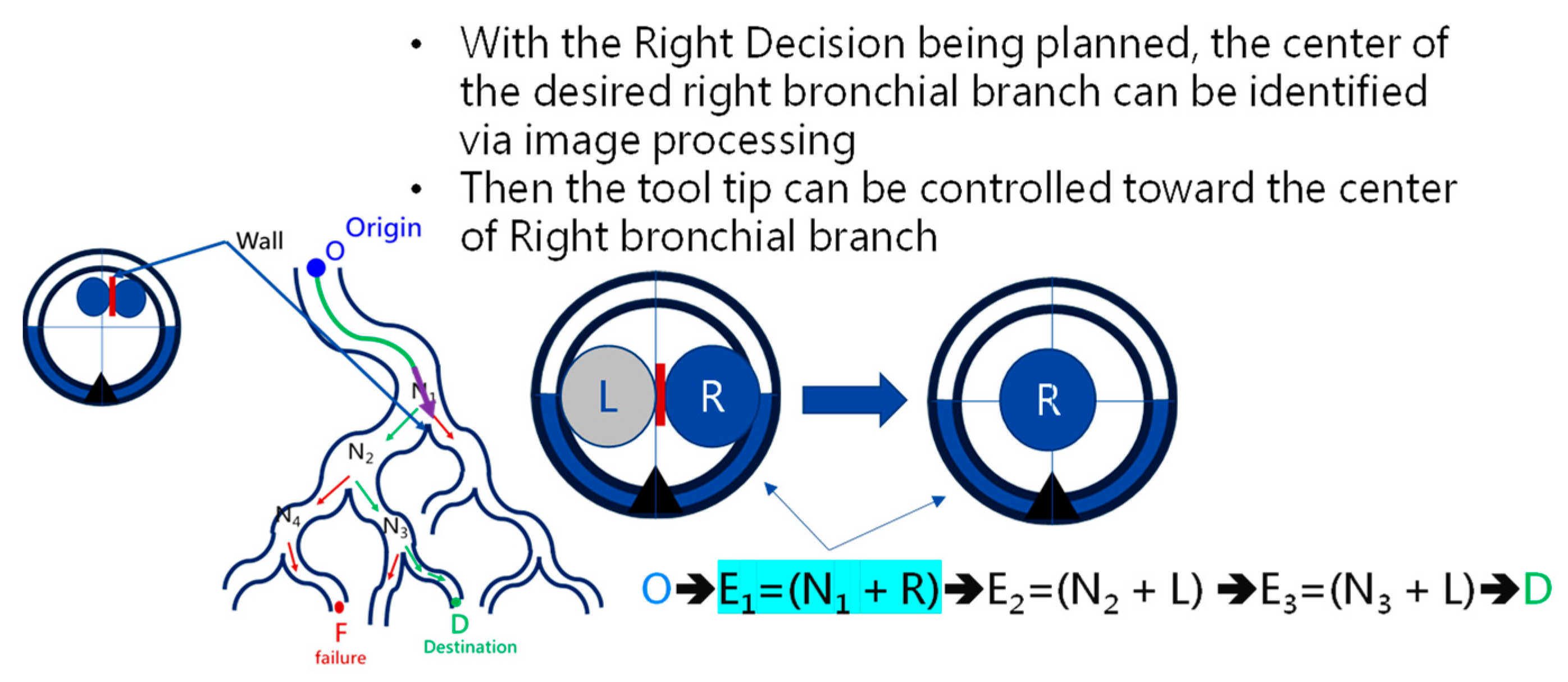
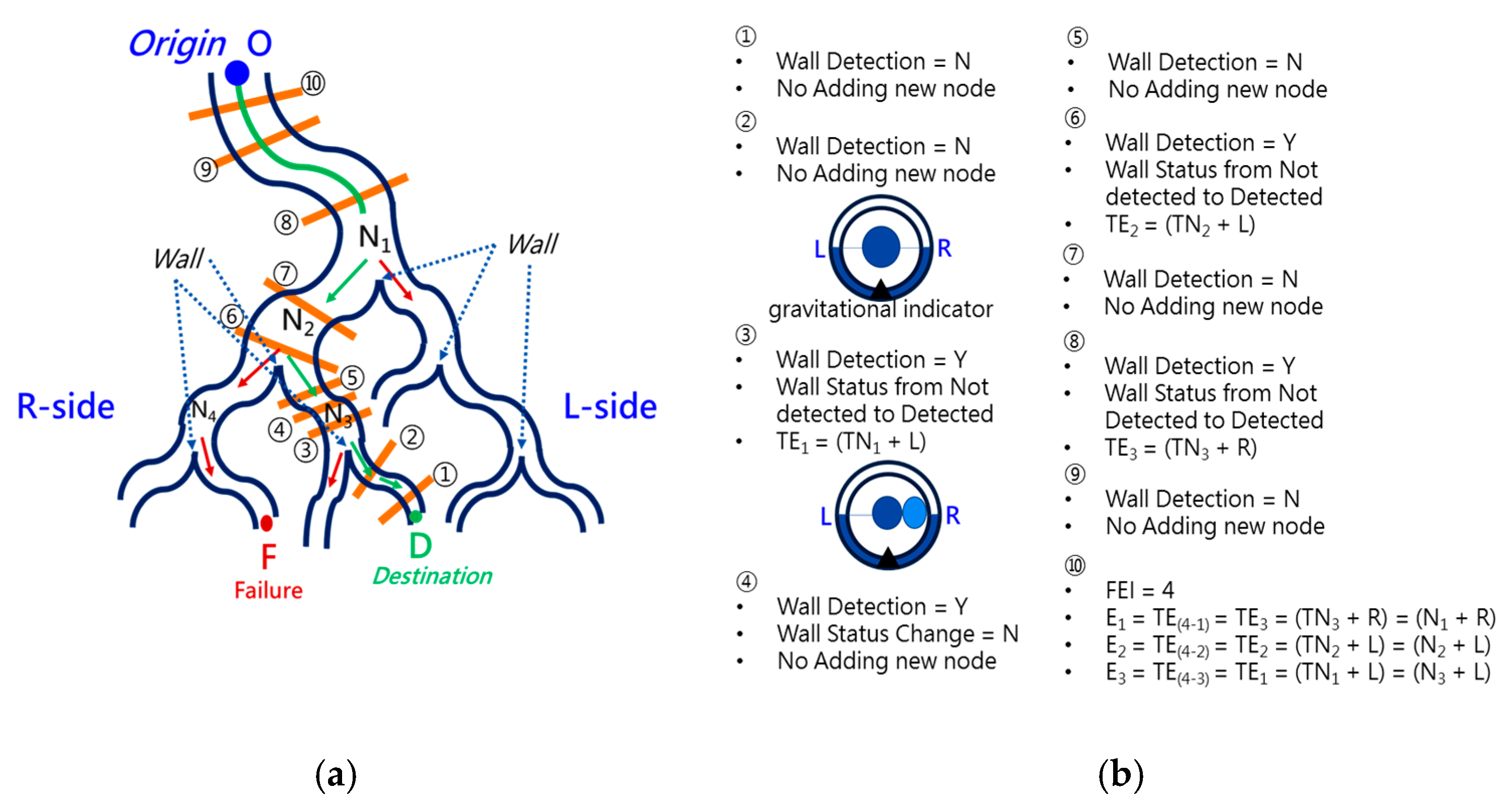
To navigate the robotic bronchoscopy when moving inside the bronchial tree by following the pre-determined path planning with planned and ordered Events, an algorithm of backward path planning was described in Section 2.3. Referring to Figure 7, which shows the proposed backward path planning, and Figure 8, which shows the simplified bronchial tree, as shown in Figure 16, an initialization image at Destination D is loaded, and at point ① according to the Node Determination, Wall Detection is NO. This leads to No New Node Added as shown in Figure 16. Based on the proposed backward path planning, the virtual tip of the robotic bronchoscopy is controlled to move backward from the Destination to the Origin while the corresponding medical images are loaded along with the backward moving path. Thus, at point ②, the same status is concluded: No New Node Added. At point ③, Wall Detection is YES and Wall Status is changed from Not detected to Detected. In addition, from the image history and 3D model of the bronchial tree reconstructed from medical images, a Right-Hole will appear on the right side once the tip has moved backward slightly from point ② to point ③, then a New Event with New Node and Decision () is added, as shown in Figure 16. In the following steps, the Decision corresponding to an Event is specified by the same scheme. At point ④, the Wall is still detected but Wall Status is not changed, and the planning result is No New Node Added. Meanwhile, at point ⑤, there is no Wall detected since the tip of the bronchoscopy is located far from the previous Wall. The planning result is No New Node Added. Meanwhile, at point ⑥, Wall Detection is YES and Wall Status is changed from Not detected to Detected, and then a New Event with a New Node and Decision ( is added. At point ⑦, the status is the same as at point ⑤, and No New Node is added. At point ⑧, accordingly, Wall Detection is YES and Wall Status is changed from Not detected to Detected, and then a New Event with a New Node and Decision ( is added. At point ⑨, the planning result is No New Node Added. Finally, at point ①, the Origin, the Final Event Index FEI is 4, and by applying Equation (5), a series of planned and ordered Events are listed, as shown in Table 3.
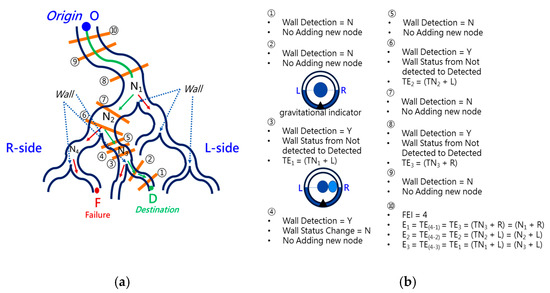
Figure 16.
Simulated backward path planning: (a) Simplified bronchial tree; (b) corresponding path planning at points ① to ⑩.

Table 3.
The simulated results of backward path planning based on Figure 16.
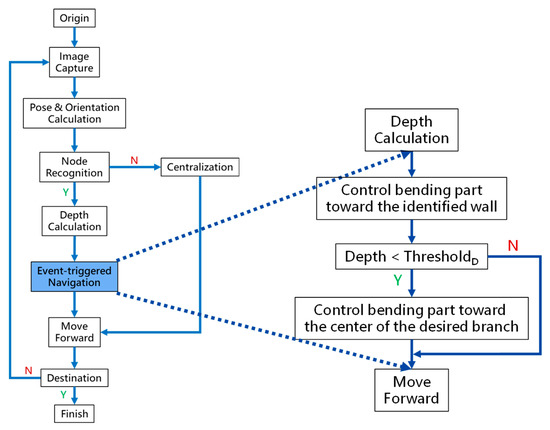
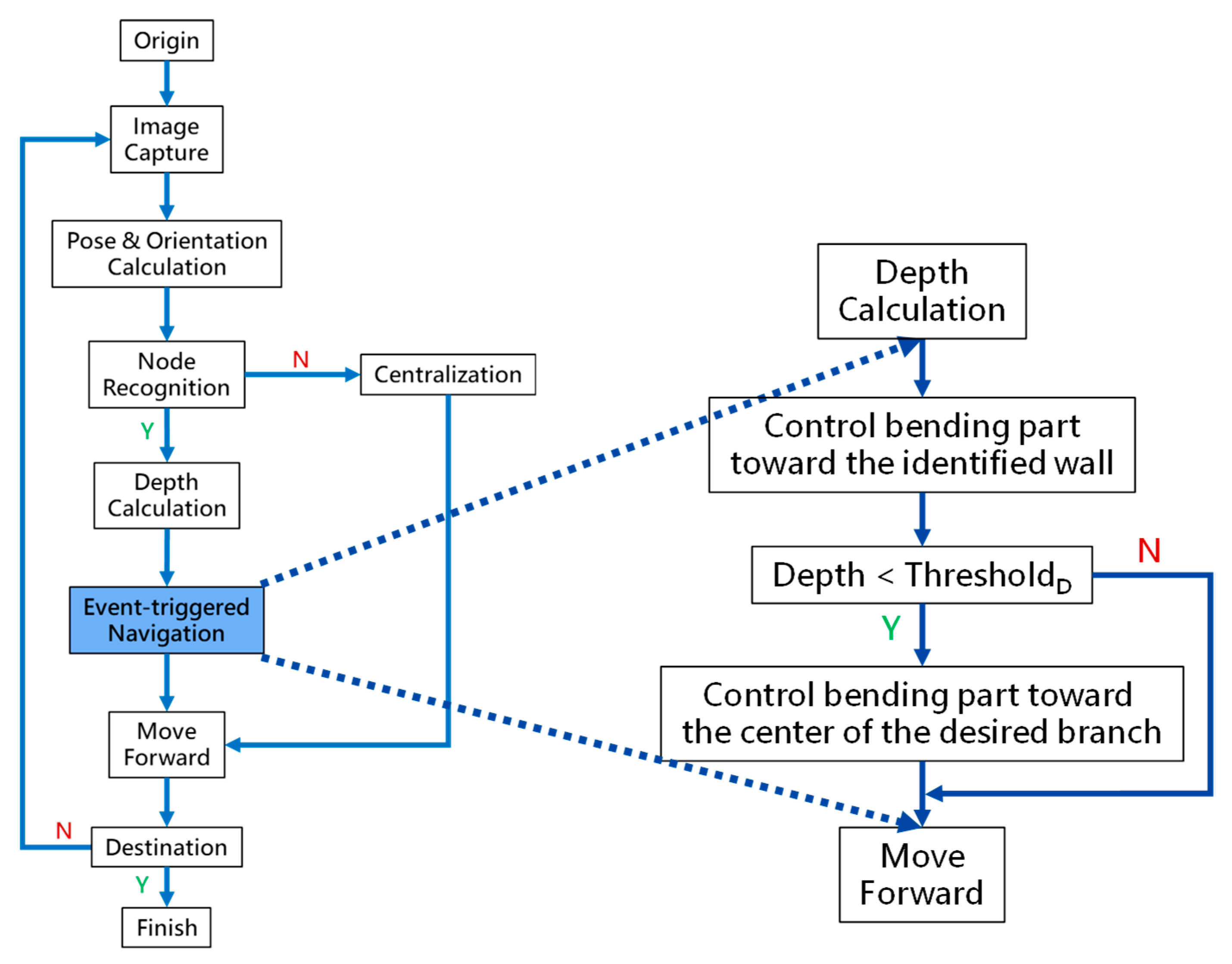
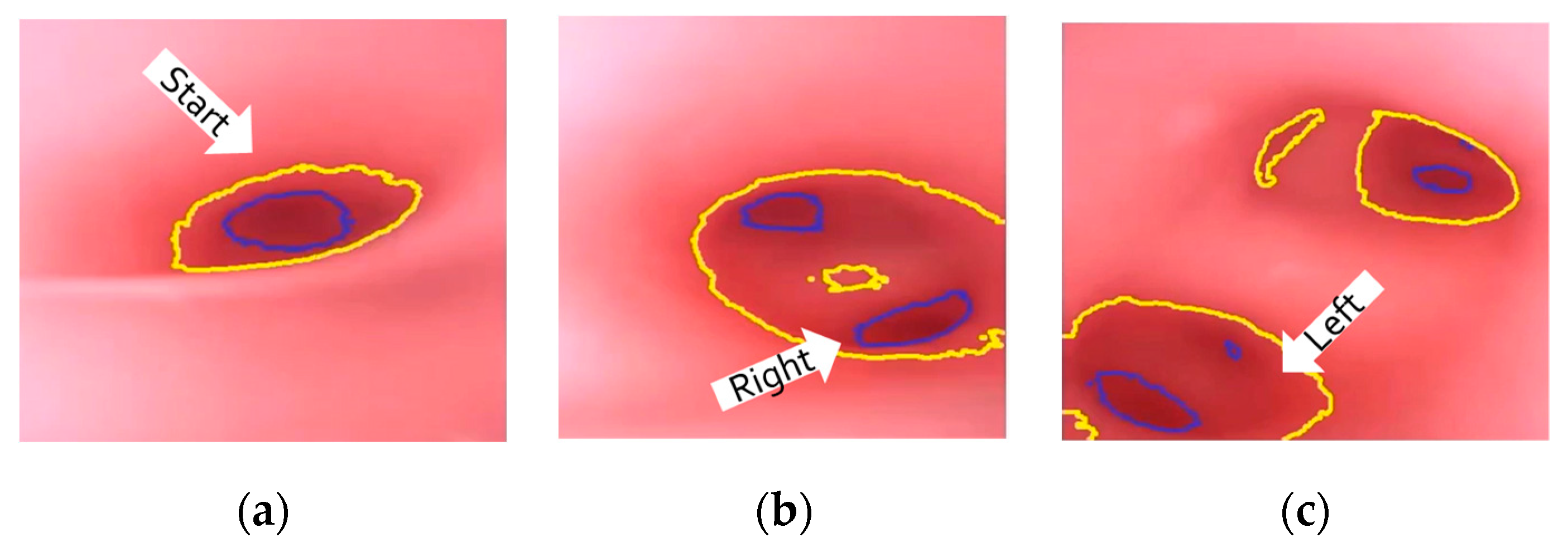
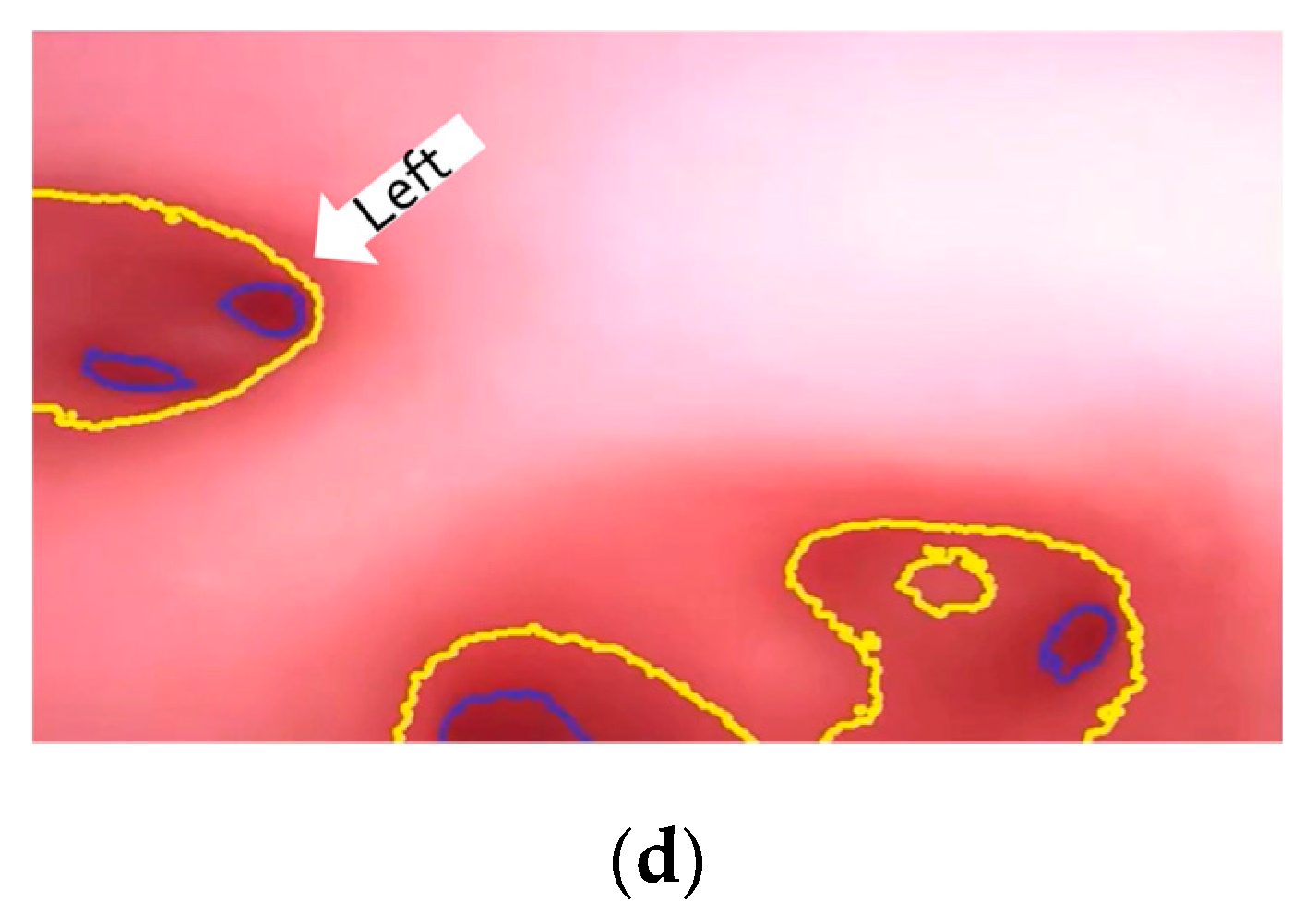
At the navigation stage, following the navigation algorithm described in Section 2.4 and as shown in Figure 9, by adopting image recognition to identify the Node, making a Decision at this Node, marking and recording that this Event has been triggered, and moving forward until the next Node is identified, the Destination will be reached. In Figure 17, the results of an experiment in the bronchial tree phantom with our proposed algorithms are illustrated. The correctness of image recognition can navigate the robotics bronchoscopy to move along the correct path. In Figure 17a, image recognition is illustrated as yellow and blue circles. During the insertion of the robotic bronchoscopy along the bronchi, a bifurcation is then detected, illustrated as two blue circles in Figure 17b. According to the first Event definition, Right Decision is selected as indicated in Figure 17b. Meanwhile, in Figure 17c,d, the Decisions of the second and third Events are Left and Left, respectively. This experiment demonstrates the feasibility of the proposed algorithms.
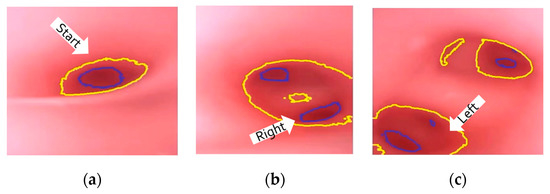
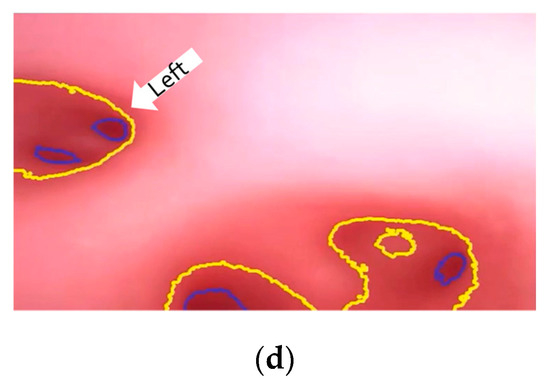
Figure 17.
Experiment results in bronchial tree phantom with the proposed algorithms: (a) yellow and blue circles shows the results of image recognition at the start; (b) Two blue circles show the bifurcation and Right Decision is indicated; (c) Left Decision at Event2 is indicated; (d) Left Decision at Event3 is indicated.
4. Discussion and Conclusions
In this paper, a review of MSC therapy has been addressed. Researchers have preliminarily demonstrated the potential effectiveness. Therefore, a miniature serpentine robotic bronchoscopy is proposed to deliver MSCs as close to the target as possible. Furthermore, a corresponding mechanical design and control scheme has been also discussed. In addition, a backward path planning and forward navigation algorithm is described to efficiently perform path planning from the oral cavity to the deep bronchi and navigate the robotic bronchoscopy to move correctly to the planned destination.
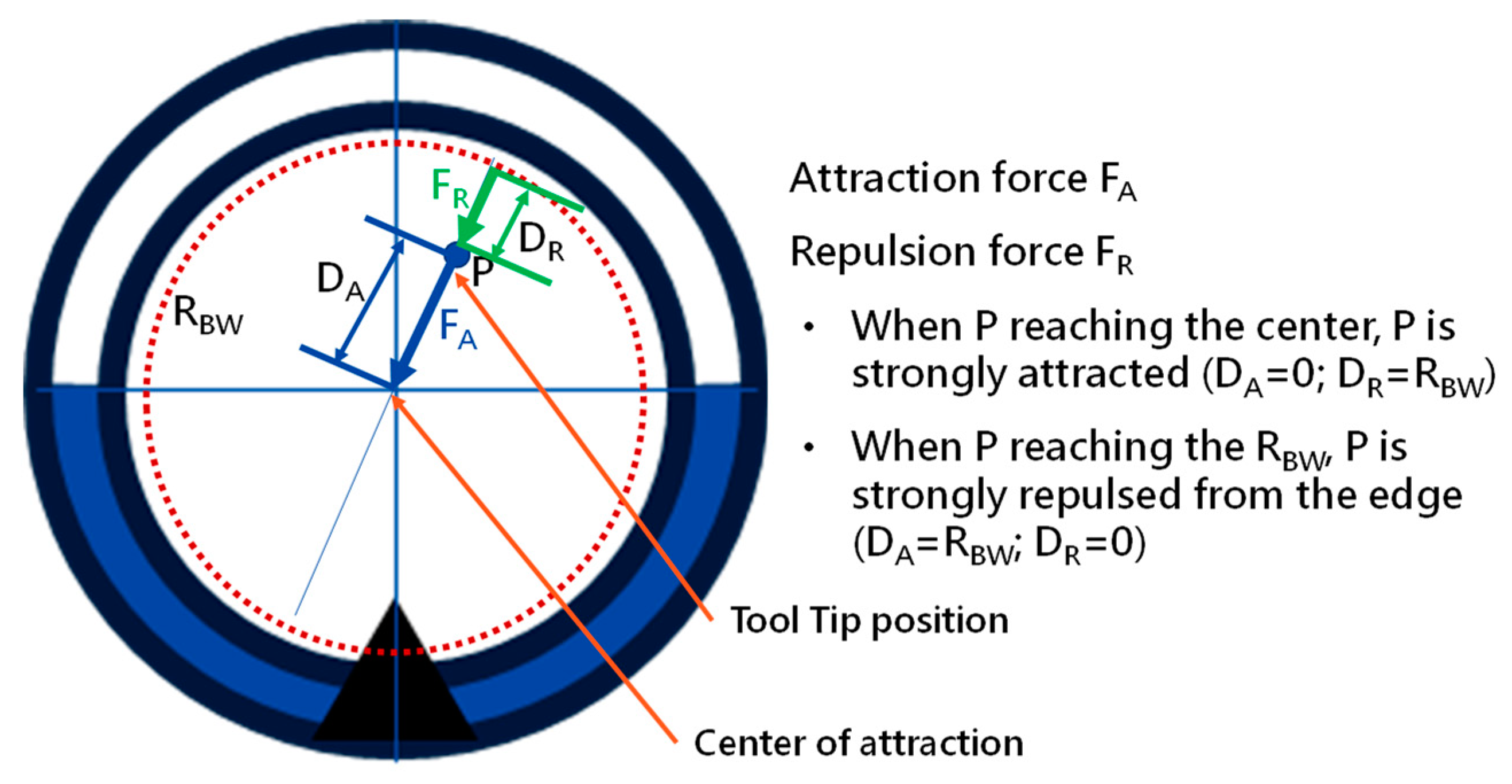
In the proposed path planning and navigation algorithms, the correctness of image processing to identify a Wall and map the identified image to the planned Event is significant. Furthermore, a major point of difference compared to other navigation algorithms or solutions is that the accurate position of the tip of the robotic bronchoscopy is not required during the entire navigation in the proposed navigation scheme in this paper. The only information required is a correct map and record of the planned Events that are triggered. In addition, making the related appropriate Decision at the correct Node is also significant.
The implementation of this robotic bronchoscopy with path planning and navigation has demonstrated the feasibility of this solution. More simulations and experiments will be conducted to further verify the efficiency of the proposed path planning and navigation mechanism. In addition, a force sensor is under design and development, which will be attached to this proposed robotic bronchoscopy to provide extra sensory information such as haptic/force feedback. The provision of the haptic/force information supports the system in reducing the probability of tissue damage due to any applied force. With this haptic/force information, this robotic bronchoscopy can perform more sophisticated operations within limited space inside the human body. Moreover, an MSC cell sprayer is under development to spray MSCs uniformly onto the target with the working channel provided via the proposed miniature serpentine robotic bronchoscopy. This proposed robotic bronchoscopy can be a platform for carrying various tools with their own specific purposes. Moreover, with multiple miniature serpentine robotic endoscopes, more complicated and dexterous operation procedures could be performed.
5. Patents
USPTO Patent Application No. 18/071,450.
Author Contributions
Conceptualization, C.-P.K. and S.H.; formal analysis, H.-Y.W.; funding acquisition, C.-P.K., S.H. and C.-Y.W.; methodology, C.-P.K.; project administration, S.H.; resources, C.-Y.W.; software, A.-P.W.; validation, A.-P.W.; writing—original draft, C.-P.K.; writing—review and editing, C.-P.K. and S.H. All authors have read and agreed to the published version of the manuscript.
Funding
This research was funded by the Industrial Technology Research Institute with the project grant N301AR5341 “Research on Integration of RFA & Cell Spray Coating within Miniature Serpentine Robotic Bronchoscopy”.
Data Availability Statement
Data sharing is not applicable to this article.
Acknowledgments
The authors acknowledge funding support by the research project N301AR5341 “Research on Integration of RFA & Cell Spray Coating within Miniature Serpentine Robotic Bronchoscopy” of Industrial Technology Research Institute.
Conflicts of Interest
The authors declare no conflict of interest.
Abbreviations
The following abbreviations are used in this manuscript:
| AMSC | Adipose-derived Mesenchymal Stem Cells |
| CMOS | Complementary Metal-Oxide-Semiconductor |
| COPD | Chronic Obstructive Pulmonary Disease |
| COVID-19 | Coronavirus Disease 2019 |
| CT | Computed Tomography |
| EMA | Electro-Magnetic Actuation |
| MRI | Magnetic Resonance Imaging |
| MSC | Mesenchymal Stem Cells |
| NOTES | Natural Orifice Transluminal Endoscopic Surgery |
References
- Golchin, A.; Seyedjafari, E.; Ardeshirylajimi, A. Mesenchymal Stem Cell Therapy for COVID-19: Present or Future. Stem Cell Rev. Rep. 2020, 16, 427–433. [Google Scholar] [CrossRef] [PubMed]
- Kim, K.; Bae, K.S.; Kim, H.S.; Lee, W.-Y. Effectiveness of Mesenchymal Stem Cell Therapy for COVID-19-Induced ARDS Patients: A Case Report. Medicina 2022, 58, 1698. [Google Scholar] [CrossRef] [PubMed]
- Mallis, P.; Chatzistamatiou, T.; Dimou, Z.; Sarri, E.F.; Georgiou, E.; Salagianni, M.; Triantafyllia, V.; Andreakos, E.; Stavropoulos-Giokas, C.; Michalopoulos, E. Mesenchymal Stromal Cell Delivery as a Potential Therapeutic Strategy against COVID-19: Promising Evidence from in vitro Results. World J. Biol. Chem. 2022, 13, 47–65. [Google Scholar] [CrossRef] [PubMed]
- George, P.M.; Wells, A.U.; Jenkins, R.G. Pulmonary Fibrosis and COVID-19: The Potential Role for Antifibrotic Therapy. Lancet Respir. Med. 2020, 8, 807–815. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Fei, X.; Zhang, X.; Bao, W.; Han, L.; Xue, Y.; Hao, H.; Zhou, X.; Zhang, M. Adipose-derived Mesenchymal Stem Cells Suppress Ozone-mediated Airway Inflammation in a Mouse Model of Chronic Obstructive Pulmonary Disease. Mol. Immunol. 2022, 151, 95–102. [Google Scholar] [CrossRef] [PubMed]
- Nejaddehbashi, F.; Radan, M.; Bayati, V.; Dianat, M.; Ali Mard, S.; Mansouri, Z. Adipose-derived Mesenchymal Stem Cells in Emphysema: Comparison of Inflammatory Markers Changes in Response to Intratracheal and Systemic Delivery Method. Tissue Cell 2023, 80, 102011. [Google Scholar] [CrossRef] [PubMed]
- Go, G.; Jeong, S.G.; Yoo, A.; Han, J.; Kang, B.; Kim, S.J.; Nguyen, T.; Jin, Z.; Kim, C.S.; Seo, Y.; et al. Human Adipose–derived Mesenchymal Stem Cell–based Medical Microrobot System for Knee Cartilage Regeneration in vivo. Sci. Robot. 2020, 5, eaay6626. [Google Scholar] [CrossRef] [PubMed]
- Chen, A.C.; Pastis, N.J.; Mahajan, A.K.; Khandhar, S.J.; Simoff, M.J.; Machuzak, M.S.; Cicenia, J.; Gildea, T.R.; Silvestri, G.A. Robotic Bronchoscopy for Peripheral Pulmonary Lesions: A Multicenter Pilot and Feasibility Study (BENEFIT). Chest 2021, 159, 845–852. [Google Scholar] [CrossRef] [PubMed]
- Donno, A.D.; Zorn, L.; Zanne, P.; Nageotte, F.; Mathelin, M.D. Introducing STRAS: A New Flexible Robotic System for Minimally Invasive Surgery. In Proceedings of the IEEE International Conference on Robotics and Automation, Karlsruhe, Germany, 6–10 May 2013; pp. 1213–1220. [Google Scholar]
- Webster, R.J.; Okamura, A.M.; Cowan, N.J. Toward Active Cannulas: Miniature Snake-like Surgical Robots. In Intelligent Robots and Systems; IEEE: New York, NY, USA, 2006; pp. 2857–2863. [Google Scholar]
- Simaan, N.; Xu, K.; Kapoor, A.; Wei, W.; Kazanzides, P.; Flint, P.; Taylor, R. Design and Integration of a Telerobotic System for Minimally Invasive Surgery of the Throat. Int. J. Robot. Res.-Spec. Issue Med. Robot. 2009, 28, 1134–1153. [Google Scholar] [CrossRef] [PubMed]
- Yeshmukhametov, A.; Koganezawa, K.; Yamamoto, Y. Design and Kinematics of Cable-Driven Continuum Robot Arm with Universal Joint Backbone. In Proceedings of the IEEE International Conference on Robotics and Biomimetics (ROBIO), Kuala Lumpur, Malaysia, 12–15 December 2018; pp. 2444–2449. [Google Scholar]
- Ho, E.; Hedstrom, G.; Septimiu, M. Robotic Bronchoscopy in Diagnosing Lung Cancer—The Evidence, Tips and Tricks: A Clinical Practice Review. Ann. Transl. Med. 2023. [Google Scholar] [CrossRef]
- Duan, X.; Xie, D.; Zhang, R.; Li, X.; Sun, J.; Qian, C.; Song, X.; Li, C. A Novel Robotic Bronchoscope System for Navigation and Biopsy of Pulmonary Lesions. Cyborg Bionic Syst. 2023, 4, 0013. [Google Scholar] [CrossRef] [PubMed]
- Chen, Z.Y.; Wu, H.Y.; Huang, S.; Chang, J.Y. Kinematic Analysis and Mechanism Design of Surgical Tendon-driven Serpentine Manipulators. In Proceedings of the International Conference on Advanced Mechanism and Machine Technology (CSMMT), Bengaluru, India, 9–10 July 2021. Paper Number 058. [Google Scholar]
- Wu, H.Y.; Huang, S.; Wu, C.Y.; Kuan, C.P.; Wang, A.P. The Design and Control Scheme of Miniature Serpentine Robot for In-Body Visual Servo Applications. In Proceedings of the World Congress on Micro and Nano Manufacturing (WCMNM 2022), Leuven, Belgium, 19–22 September 2022. [Google Scholar]
- Monarch Platform. Available online: https://www.jnjmedtech.com/en-US/product-family/monarch (accessed on 12 March 2023).
- Intuitive Da Vinci SP. Available online: https://www.intuitive.com/en-us/products-and-services/da-vinci/systems/sp (accessed on 12 March 2023).
- Marinho, M.M.; Ishida, H.; Harada, K.; Deie, K.; Mitsuishi, M. Virtual Fixture Assistance for Suturing in Robot-Aided Pediatric Endoscopic Surgery. IEEE Robot. Autom. Lett. 2019, 5, 524–531. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).