Obesity, Leptin and Breast Cancer: Epidemiological Evidence and Proposed Mechanisms
Abstract
1. Introduction
2. The Epidemiological Association between Obesity and Breast Cancer
2.1. Increased BMI and Risk of Breast Cancer Occurrence
2.2. Increased BMI and Risk of Breast Cancer Mortality
2.3. Waist-To-Hip Ratio (WHR) and Breast Cancer
2.4. Obesity and Therapeutic Implications
2.5. Effects of Obesity on Quality of Life in Survivors
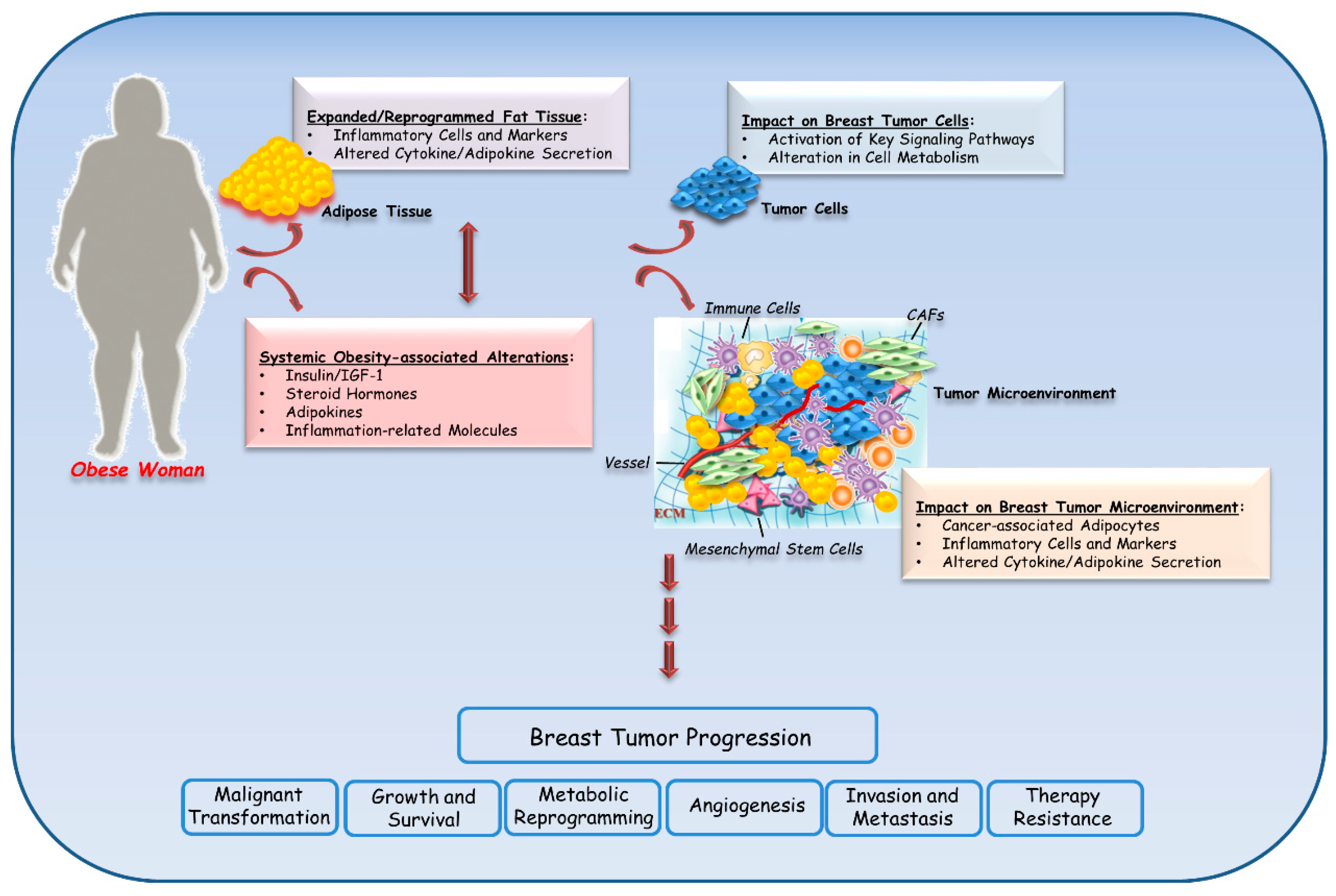
3. Biological Mechanisms Linking Obesity to Breast Cancer
3.1. Inflammation
3.2. Insulin-IGF-1 Axis
3.3. Estrogens
3.4. Adipokines
3.4.1. Adiponectin
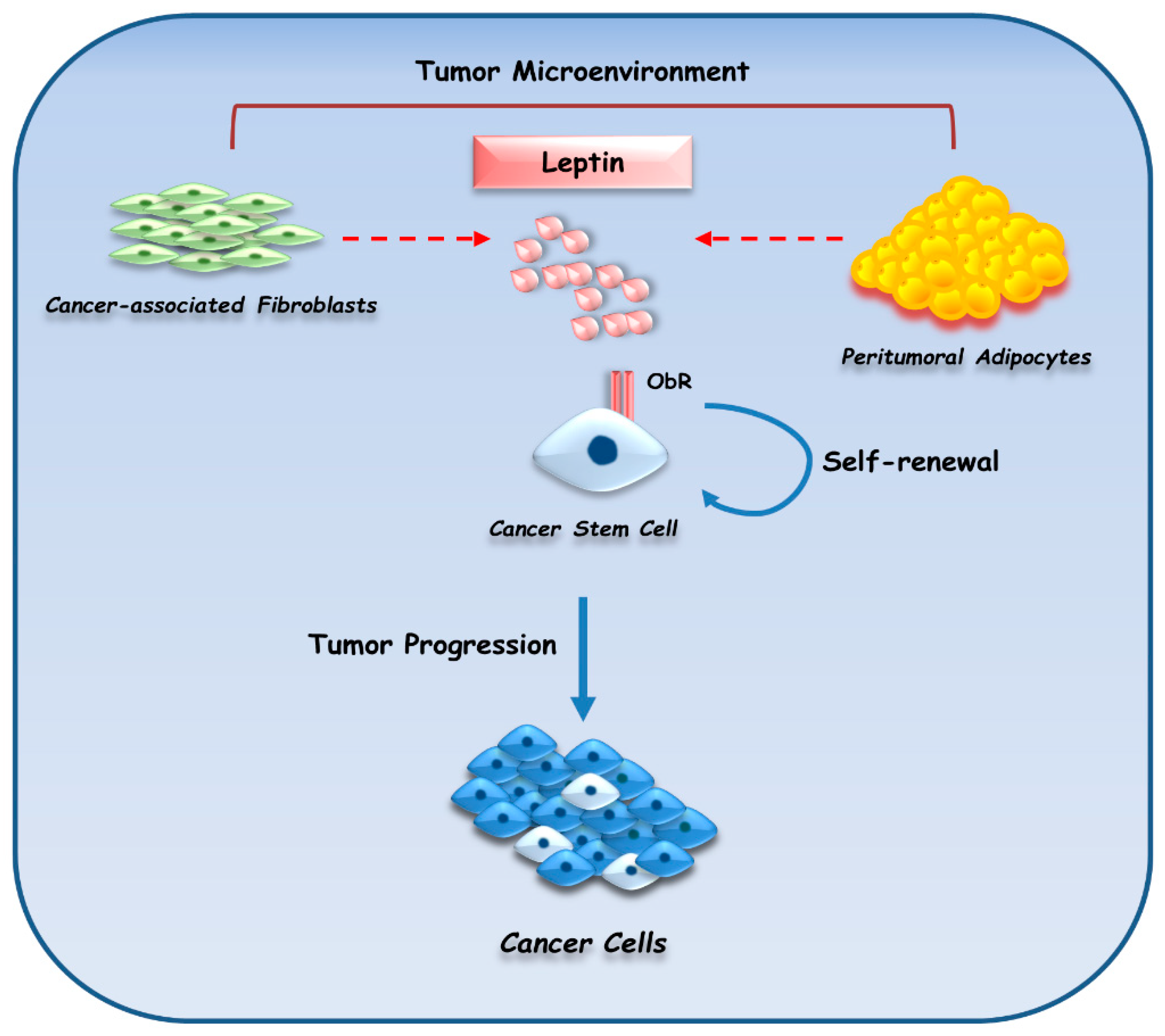
3.4.2. Leptin
Clinical Studies
In Vivo Studies
In Vitro Studies
4. Conclusions and Perspective
Author Contributions
Funding
Conflicts of Interest
References
- Ng, M.; Fleming, T.; Robinson, M.; Thomson, B.; Graetz, N.; Margono, C.; Mullany, E.C.; Biryukov, S.; Abbafati, C.; Abera, S.F.; et al. Global, regional, and national prevalence of overweight and obesity in children and adults during 1980-2013: A systematic analysis for the global burden of disease study 2013. Lancet 2014, 384, 766–781. [Google Scholar] [CrossRef]
- NCD Risk Factor Collaboration (NCD-RisC). Trends in adult body-mass index in 200 countries from 1975 to 2014: A pooled analysis of 1698 population-based measurement studies with 19.2 million participants. Lancet 2016, 387, 1377–1396. [Google Scholar] [CrossRef]
- Global Burden of Metabolic Risk Factors for Chronic Diseases Collaboration; Lu, Y.; Hajifathalian, K.; Ezzati, M.; Woodward, M.; Rimm, E.B.; Danaei, G. Metabolic mediators of the effects of body-mass index, overweight, and obesity on coronary heart disease and stroke: A pooled analysis of 97 prospective cohorts with 1.8 million participants. Lancet 2014, 383, 970–983. [Google Scholar] [PubMed]
- Global BMI Mortality Collaboration; Di Angelantonio, E.; Bhupathiraju, S.N.; Wormser, D.; Gao, P.; Kaptoge, S.; Berrington de Gonzalez, A.; Cairns, B.J.; Huxley, R.; Jackson, C.L.; et al. Body-mass index and all-cause mortality: Individual-participant-data meta-analysis of 239 prospective studies in four continents. Lancet 2016, 388, 776–786. [Google Scholar] [CrossRef]
- Renehan, A.G.; Tyson, M.; Egger, M.; Heller, R.F.; Zwahlen, M. Body-mass index and incidence of cancer: A systematic review and meta-analysis of prospective observational studies. Lancet 2008, 371, 569–578. [Google Scholar] [CrossRef]
- Basen-Engquist, K.; Chang, M. Obesity and cancer risk: Recent review and evidence. Curr. Oncol. Rep. 2011, 13, 71–76. [Google Scholar] [CrossRef] [PubMed]
- Goodwin, P.J.; Stambolic, V. Impact of the obesity epidemic on cancer. Annu. Rev. Med. 2015, 66, 281–296. [Google Scholar] [CrossRef]
- GBD 2015 Obesity Collaborators; Afshin, A.; Forouzanfar, M.H.; Reitsma, M.B.; Sur, P.; Estep, K.; Lee, A.; Marczak, L.; Mokdad, A.H.; Moradi-Lakeh, M.; et al. Health effects of overweight and obesity in 195 countries over 25 years. N. Engl. J. Med. 2017, 377, 13–27. [Google Scholar]
- Torre, L.A.; Islami, F.; Siegel, R.L.; Ward, E.M.; Jemal, A. Global cancer in women: Burden and trends. Cancer Epidemiol. Biomark. Prev. 2017, 26, 444–457. [Google Scholar] [CrossRef]
- Ferlay, J.; Soerjomataram, I.; Dikshit, R.; Eser, S.; Mathers, C.; Rebelo, M.; Parkin, D.M.; Forman, D.; Bray, F. Cancer incidence and mortality worldwide: Sources, methods and major patterns in globocan 2012. Int. J. Cancer 2015, 136, E359–E386. [Google Scholar] [CrossRef]
- Danaei, G.; Vander Hoorn, S.; Lopez, A.D.; Murray, C.J.; Ezzati, M.; Comparative Risk Assessment Collaborating Group. Causes of cancer in the world: Comparative risk assessment of nine behavioural and environmental risk factors. Lancet 2005, 366, 1784–1793. [Google Scholar] [CrossRef]
- McDonald, J.A.; Goyal, A.; Terry, M.B. Alcohol intake and breast cancer risk: Weighing the overall evidence. Curr. Breast Cancer Rep. 2013, 5, 208–221. [Google Scholar] [CrossRef] [PubMed]
- Lahart, I.M.; Metsios, G.S.; Nevill, A.M.; Carmichael, A.R. Physical activity, risk of death and recurrence in breast cancer survivors: A systematic review and meta-analysis of epidemiological studies. Acta Oncol. 2015, 54, 635–654. [Google Scholar] [CrossRef] [PubMed]
- Ordonez-Mena, J.M.; Schottker, B.; Mons, U.; Jenab, M.; Freisling, H.; Bueno-de-Mesquita, B.; O’Doherty, M.G.; Scott, A.; Kee, F.; Stricker, B.H.; et al. Quantification of the smoking-associated cancer risk with rate advancement periods: Meta-analysis of individual participant data from cohorts of the chances consortium. BMC Med. 2016, 14, 62. [Google Scholar] [CrossRef] [PubMed]
- Mullooly, M.; Khodr, Z.G.; Dallal, C.M.; Nyante, S.J.; Sherman, M.E.; Falk, R.; Liao, L.M.; Love, J.; Brinton, L.A.; Gierach, G.L. Epidemiologic risk factors for in situ and invasive breast cancers among postmenopausal women in the national institutes of health-aarp diet and health study. Am. J. Epidemiol. 2017, 186, 1329–1340. [Google Scholar] [CrossRef] [PubMed]
- O’Sullivan, C.C.; Loprinzi, C.L.; Haddad, T.C. Updates in the evaluation and management of breast cancer. Mayo Clin. Proc. 2018, 93, 794–807. [Google Scholar] [CrossRef] [PubMed]
- Duggan, C.; Tapsoba, J.D.; Wang, C.Y.; Campbell, K.L.; Foster-Schubert, K.; Gross, M.D.; McTiernan, A. Dietary weight loss, exercise, and oxidative stress in postmenopausal women: A randomized controlled trial. Cancer Prev. Res. 2016, 9, 835–843. [Google Scholar] [CrossRef]
- Habermann, N.; Makar, K.W.; Abbenhardt, C.; Xiao, L.; Wang, C.Y.; Utsugi, H.K.; Alfano, C.M.; Campbell, K.L.; Duggan, C.; Foster-Schubert, K.E.; et al. No effect of caloric restriction or exercise on radiation repair capacity. Med. Sci. Sports Exerc. 2015, 47, 896–904. [Google Scholar] [CrossRef]
- Imayama, I.; Alfano, C.M.; Mason, C.; Wang, C.; Duggan, C.; Campbell, K.L.; Kong, A.; Foster-Scubert, K.E.; Blackburn, G.L.; Wang, C.Y. Weight and metabolic effects of dietary weight loss and exercise interventions in postmenopausal antidepressant medication users and non-users: A randomized controlled trial. Prev. Med. 2013, 57, 525–532. [Google Scholar] [CrossRef]
- Mason, C.; Foster-Schubert, K.E.; Imayama, I.; Kong, A.; Xiao, L.; Bain, C.; Campbell, K.L.; Wang, C.Y.; Duggan, C.R.; Ulrich, C.M.; et al. Dietary weight loss and exercise effects on insulin resistance in postmenopausal women. Am. J. Prev. Med. 2011, 41, 366–375. [Google Scholar] [CrossRef]
- Mason, C.; Foster-Schubert, K.E.; Imayama, I.; Xiao, L.; Kong, A.; Campbell, K.L.; Duggan, C.R.; Wang, C.Y.; Alfano, C.M.; Ulrich, C.M.; et al. History of weight cycling does not impede future weight loss or metabolic improvements in postmenopausal women. Metabolism 2013, 62, 127–136. [Google Scholar] [CrossRef] [PubMed]
- Mason, C.; Xiao, L.; Imayama, I.; Duggan, C.R.; Bain, C.; Foster-Schubert, K.E.; Kong, A.; Campbell, K.L.; Wang, C.Y.; Neuhouser, M.L.; et al. Effects of weight loss on serum vitamin d in postmenopausal women. Am. J. Clin. Nutr. 2011, 94, 95–103. [Google Scholar] [CrossRef]
- Mason, C.; Xiao, L.; Imayama, I.; Duggan, C.R.; Foster-Schubert, K.E.; Kong, A.; Campbell, K.L.; Wang, C.Y.; Villasenor, A.; Neuhouser, M.L.; et al. Influence of diet, exercise, and serum vitamin d on sarcopenia in postmenopausal women. Med. Sci. Sports Exerc. 2013, 45, 607–614. [Google Scholar] [CrossRef] [PubMed]
- Rock, C.L.; Flatt, S.W.; Byers, T.E.; Colditz, G.A.; Demark-Wahnefried, W.; Ganz, P.A.; Wolin, K.Y.; Elias, A.; Krontiras, H.; Liu, J.; et al. Results of the exercise and nutrition to enhance recovery and good health for you (energy) trial: A behavioral weight loss intervention in overweight or obese breast cancer survivors. J. Clin. Oncol. 2015, 33, 3169–3176. [Google Scholar] [CrossRef] [PubMed]
- Giordano, L.; Gallo, F.; Petracci, E.; Chiorino, G.; Segnan, N.; Andromeda Working Group. The andromeda prospective cohort study: Predictive value of combined criteria to tailor breast cancer screening and new opportunities from circulating markers: Study protocol. BMC Cancer 2017, 17, 785. [Google Scholar] [CrossRef] [PubMed]
- Crawford, T.N.; Arikawa, A.Y.; Kurzer, M.S.; Schmitz, K.H.; Phipps, W.R. Cross-sectional study of factors influencing sex hormone-binding globulin concentrations in normally cycling premenopausal women. Fertil. Steril. 2015, 104, 1544–1551. [Google Scholar] [CrossRef] [PubMed]
- Arikawa, A.Y.; Jakits, H.E.; Flood, A.; Thomas, W.; Gross, M.; Schmitz, K.H.; Kurzer, M.S. Consumption of a high glycemic load but not a high glycemic index diet is marginally associated with oxidative stress in young women. Nutr. Res. 2015, 35, 7–13. [Google Scholar] [CrossRef]
- Smith, A.J.; Phipps, W.R.; Thomas, W.; Schmitz, K.H.; Kurzer, M.S. The effects of aerobic exercise on estrogen metabolism in healthy premenopausal women. Cancer Epidemiol. Biomark. Prev. 2013, 22, 756–764. [Google Scholar] [CrossRef]
- O’Dougherty, M.; Schmitz, K.H.; Hearst, M.O.; Covelli, M.; Kurzer, M.S. Dual conversations: Body talk among young women and their social contacts. Qual. Health Res. 2011, 21, 1191–1204. [Google Scholar] [CrossRef]
- Beasley, J.M.; Kwan, M.L.; Chen, W.Y.; Weltzien, E.K.; Kroenke, C.H.; Lu, W.; Nechuta, S.J.; Cadmus-Bertram, L.; Patterson, R.E.; Sternfeld, B.; et al. Meeting the physical activity guidelines and survival after breast cancer: Findings from the after breast cancer pooling project. Breast Cancer Res. Treat. 2012, 131, 637–643. [Google Scholar] [CrossRef]
- Beg, M.S.; Gupta, A.; Stewart, T.; Rethorst, C.D. Promise of wearable physical activity monitors in oncology practice. J. Oncol. Pract. 2017, 13, 82–89. [Google Scholar] [CrossRef]
- Cadmus-Bertram, L.A.; Marcus, B.H.; Patterson, R.E.; Parker, B.A.; Morey, B.L. Randomized trial of a fitbit-based physical activity intervention for women. Am. J. Prev. Med. 2015, 49, 414–418. [Google Scholar] [CrossRef] [PubMed]
- Ferguson, T.; Rowlands, A.V.; Olds, T.; Maher, C. The validity of consumer-level, activity monitors in healthy adults worn in free-living conditions: A cross-sectional study. Int. J. Behav. Nutr. Phys. Act. 2015, 12, 42. [Google Scholar] [CrossRef] [PubMed]
- Ibrahim, E.M.; Al-Homaidh, A. Physical activity and survival after breast cancer diagnosis: Meta-analysis of published studies. Med. Oncol. 2011, 28, 753–765. [Google Scholar] [CrossRef] [PubMed]
- Sullivan, A.N.; Lachman, M.E. Behavior change with fitness technology in sedentary adults: A review of the evidence for increasing physical activity. Front. Public Health 2016, 4, 289. [Google Scholar] [CrossRef] [PubMed]
- Thariat, J.; Creisson, A.; Chamignon, B.; Dejode, M.; Gastineau, M.; Hebert, C.; Boissin, F.; Topfer, C.; Gilbert, E.; Grondin, B. Integrating patient education in your oncology practice. Bull. Cancer 2016, 103, 674–690. [Google Scholar] [CrossRef]
- Tudor-Locke, C.; Hatano, Y.; Pangrazi, R.P.; Kang, M. Revisiting “how many steps are enough?”. Med. Sci. Sports Exerc. 2008, 40 (Suppl. 7), S537–S543. [Google Scholar] [CrossRef] [PubMed]
- Nilsson, H.; Angeras, U.; Bock, D.; Borjesson, M.; Onerup, A.; Fagevik Olsen, M.; Gellerstedt, M.; Haglind, E.; Angenete, E. Is preoperative physical activity related to post-surgery recovery? A cohort study of patients with breast cancer. BMJ Open 2016, 6, e007997. [Google Scholar] [CrossRef]
- Winkels, R.M.; Sturgeon, K.M.; Kallan, M.J.; Dean, L.T.; Zhang, Z.; Evangelisti, M.; Brown, J.C.; Sarwer, D.B.; Troxel, A.B.; Denlinger, C.; et al. The women in steady exercise research (wiser) survivor trial: The innovative transdisciplinary design of a randomized controlled trial of exercise and weight-loss interventions among breast cancer survivors with lymphedema. Contemp. Clin. Trials 2017, 61, 63–72. [Google Scholar] [CrossRef]
- Neuhouser, M.L.; Aragaki, A.K.; Prentice, R.L.; Manson, J.E.; Chlebowski, R.; Carty, C.L.; Ochs-Balcom, H.M.; Thomson, C.A.; Caan, B.J.; Tinker, L.F.; et al. Overweight, obesity, and postmenopausal invasive breast cancer risk: A secondary analysis of the women’s health initiative randomized clinical trials. JAMA Oncol. 2015, 1, 611–621. [Google Scholar] [CrossRef]
- Bhaskaran, K.; Douglas, I.; Forbes, H.; dos-Santos-Silva, I.; Leon, D.A.; Smeeth, L. Body-mass index and risk of 22 specific cancers: A population-based cohort study of 5.24 million uk adults. Lancet 2014, 384, 755–765. [Google Scholar] [CrossRef]
- Reeves, G.K.; Pirie, K.; Beral, V.; Green, J.; Spencer, E.; Bull, D.; Million Women Study Collaboration. Cancer incidence and mortality in relation to body mass index in the million women study: Cohort study. BMJ 2007, 335, 1134. [Google Scholar] [CrossRef]
- Sebastiani, F.; Cortesi, L.; Sant, M.; Lucarini, V.; Cirilli, C.; De Matteis, E.; Marchi, I.; Negri, R.; Gallo, E.; Federico, M. Increased incidence of breast cancer in postmenopausal women with high body mass index at the modena screening program. J. Breast Cancer 2016, 19, 283–291. [Google Scholar] [CrossRef] [PubMed]
- White, A.J.; Nichols, H.B.; Bradshaw, P.T.; Sandler, D.P. Overall and central adiposity and breast cancer risk in the sister study. Cancer 2015, 121, 3700–3708. [Google Scholar] [CrossRef] [PubMed]
- Munsell, M.F.; Sprague, B.L.; Berry, D.A.; Chisholm, G.; Trentham-Dietz, A. Body mass index and breast cancer risk according to postmenopausal estrogen-progestin use and hormone receptor status. Epidemiol. Rev. 2014, 36, 114–136. [Google Scholar] [CrossRef] [PubMed]
- Cotterchio, M.; Kreiger, N.; Theis, B.; Sloan, M.; Bahl, S. Hormonal factors and the risk of breast cancer according to estrogen- and progesterone-receptor subgroup. Cancer Epidemiol. Biomark. Prev. 2003, 12, 1053–1060. [Google Scholar] [PubMed]
- Bandera, E.V.; Chandran, U.; Hong, C.C.; Troester, M.A.; Bethea, T.N.; Adams-Campbell, L.L.; Haiman, C.A.; Park, S.Y.; Olshan, A.F.; Ambrosone, C.B.; et al. Obesity, body fat distribution, and risk of breast cancer subtypes in african american women participating in the amber consortium. Breast Cancer Res. Treat. 2015, 150, 655–666. [Google Scholar] [CrossRef] [PubMed]
- Nagrani, R.; Mhatre, S.; Rajaraman, P.; Soerjomataram, I.; Boffetta, P.; Gupta, S.; Parmar, V.; Badwe, R.; Dikshit, R. Central obesity increases risk of breast cancer irrespective of menopausal and hormonal receptor status in women of south asian ethnicity. Eur. J. Cancer 2016, 66, 153–161. [Google Scholar] [CrossRef]
- Gaudet, M.M.; Carter, B.D.; Patel, A.V.; Teras, L.R.; Jacobs, E.J.; Gapstur, S.M. Waist circumference, body mass index, and postmenopausal breast cancer incidence in the cancer prevention study-ii nutrition cohort. Cancer Causes Control 2014, 25, 737–745. [Google Scholar] [CrossRef]
- Nattenmuller, C.J.; Kriegsmann, M.; Sookthai, D.; Fortner, R.T.; Steffen, A.; Walter, B.; Johnson, T.; Kneisel, J.; Katzke, V.; Bergmann, M.; et al. Obesity as risk factor for subtypes of breast cancer: Results from a prospective cohort study. BMC Cancer 2018, 18, 616. [Google Scholar] [CrossRef]
- Laudisio, D.; Muscogiuri, G.; Barrea, L.; Savastano, S.; Colao, A. Obesity and breast cancer in premenopausal women: Current evidence and future perspectives. Eur. J. Obstet. Gynecol. Reprod. Biol. 2018, 230, 217–221. [Google Scholar] [CrossRef] [PubMed]
- Van den Brandt, P.A.; Spiegelman, D.; Yaun, S.S.; Adami, H.O.; Beeson, L.; Folsom, A.R.; Fraser, G.; Goldbohm, R.A.; Graham, S.; Kushi, L.; et al. Pooled analysis of prospective cohort studies on height, weight, and breast cancer risk. Am. J. Epidemiol. 2000, 152, 514–527. [Google Scholar] [CrossRef] [PubMed]
- Polotsky, A.J.; Hailpern, S.M.; Skurnick, J.H.; Lo, J.C.; Sternfeld, B.; Santoro, N. Association of adolescent obesity and lifetime nulliparity-the study of women’s health across the nation (SWAN). Fertil. Steril. 2010, 93, 2004–2011. [Google Scholar] [CrossRef] [PubMed]
- Jain, A.; Polotsky, A.J.; Rochester, D.; Berga, S.L.; Loucks, T.; Zeitlian, G.; Gibbs, K.; Polotsky, H.N.; Feng, S.; Isaac, B.; et al. Pulsatile luteinizing hormone amplitude and progesterone metabolite excretion are reduced in obese women. J. Clin. Endocrinol. MeTable 2007, 92, 2468–2473. [Google Scholar] [CrossRef] [PubMed]
- Lanari, C.; Molinolo, A.A. Progesterone receptors–animal models and cell signalling in breast cancer. Diverse activation pathways for the progesterone receptor: Possible implications for breast biology and cancer. Breast Cancer Res. 2002, 4, 240–243. [Google Scholar] [CrossRef] [PubMed]
- Campagnoli, C.; Abba, C.; Ambroggio, S.; Peris, C. Pregnancy, progesterone and progestins in relation to breast cancer risk. J. Steroid Biochem. Mol. Biol. 2005, 97, 441–450. [Google Scholar] [CrossRef] [PubMed]
- Lahmann, P.H.; Hoffmann, K.; Allen, N.; van Gils, C.H.; Khaw, K.T.; Tehard, B.; Berrino, F.; Tjonneland, A.; Bigaard, J.; Olsen, A.; et al. Body size and breast cancer risk: Findings from the european prospective investigation into cancer and nutrition (EPIC). Int. J. Cancer 2004, 111, 762–771. [Google Scholar] [CrossRef]
- Chen, Y.; Liu, L.; Zhou, Q.; Imam, M.U.; Cai, J.; Wang, Y.; Qi, M.; Sun, P.; Ping, Z.; Fu, X. Body mass index had different effects on premenopausal and postmenopausal breast cancer risks: A dose-response meta-analysis with 3,318,796 subjects from 31 cohort studies. BMC Public Health 2017, 17, 936. [Google Scholar] [CrossRef]
- Cecchini, R.S.; Costantino, J.P.; Cauley, J.A.; Cronin, W.M.; Wickerham, D.L.; Land, S.R.; Weissfeld, J.L.; Wolmark, N. Body mass index and the risk for developing invasive breast cancer among high-risk women in nsabp p-1 and star breast cancer prevention trials. Cancer Prev. Res. 2012, 5, 583–592. [Google Scholar] [CrossRef]
- Kawai, M.; Malone, K.E.; Tang, M.T.; Li, C.I. Height, body mass index (BMI), bmi change, and the risk of estrogen receptor-positive, HER2-positive, and triple-negative breast cancer among women ages 20 to 44 years. Cancer 2014, 120, 1548–1556. [Google Scholar] [CrossRef]
- John, E.M.; Sangaramoorthy, M.; Hines, L.M.; Stern, M.C.; Baumgartner, K.B.; Giuliano, A.R.; Wolff, R.K.; Slattery, M.L. Overall and abdominal adiposity and premenopausal breast cancer risk among hispanic women: The breast cancer health disparities study. Cancer Epidemiol. Biomark. Prev. 2015, 24, 138–147. [Google Scholar] [CrossRef] [PubMed]
- Ma, H.; Bernstein, L.; Ross, R.K.; Ursin, G. Hormone-related risk factors for breast cancer in women under age 50 years by estrogen and progesterone receptor status: Results from a case-control and a case-case comparison. Breast Cancer Res. 2006, 8, R39. [Google Scholar] [CrossRef] [PubMed]
- Chang, S.; Buzdar, A.U.; Hursting, S.D. Inflammatory breast cancer and body mass index. J. Clin. Oncol. 1998, 16, 3731–3735. [Google Scholar] [CrossRef] [PubMed]
- Schairer, C.; Li, Y.; Frawley, P.; Graubard, B.I.; Wellman, R.D.; Buist, D.S.; Kerlikowske, K.; Onega, T.L.; Anderson, W.F.; Miglioretti, D.L. Risk factors for inflammatory breast cancer and other invasive breast cancers. J. Natl. Cancer Inst. 2013, 105, 1373–1384. [Google Scholar] [CrossRef] [PubMed]
- Atkinson, R.L.; El-Zein, R.; Valero, V.; Lucci, A.; Bevers, T.B.; Fouad, T.; Liao, W.; Ueno, N.T.; Woodward, W.A.; Brewster, A.M. Epidemiological risk factors associated with inflammatory breast cancer subtypes. Cancer Causes Control 2016, 27, 359–366. [Google Scholar] [CrossRef] [PubMed]
- Druesne-Pecollo, N.; Touvier, M.; Barrandon, E.; Chan, D.S.; Norat, T.; Zelek, L.; Hercberg, S.; Latino-Martel, P. Excess body weight and second primary cancer risk after breast cancer: A systematic review and meta-analysis of prospective studies. Breast Cancer Res. Treat. 2012, 135, 647–654. [Google Scholar] [CrossRef] [PubMed]
- Protani, M.; Coory, M.; Martin, J.H. Effect of obesity on survival of women with breast cancer: Systematic review and meta-analysis. Breast Cancer Res. Treat. 2010, 123, 627–635. [Google Scholar] [CrossRef] [PubMed]
- Chan, D.S.; Vieira, A.R.; Aune, D.; Bandera, E.V.; Greenwood, D.C.; McTiernan, A.; Navarro Rosenblatt, D.; Thune, I.; Vieira, R.; Norat, T. Body mass index and survival in women with breast cancer-systematic literature review and meta-analysis of 82 follow-up studies. Ann. Oncol. 2014, 25, 1901–1914. [Google Scholar] [CrossRef] [PubMed]
- Playdon, M.C.; Bracken, M.B.; Sanft, T.B.; Ligibel, J.A.; Harrigan, M.; Irwin, M.L. Weight gain after breast cancer diagnosis and all-cause mortality: Systematic review and meta-analysis. J. Natl. Cancer Inst. 2015, 107, djv275. [Google Scholar] [CrossRef] [PubMed]
- Niraula, S.; Ocana, A.; Ennis, M.; Goodwin, P.J. Body size and breast cancer prognosis in relation to hormone receptor and menopausal status: A meta-analysis. Breast Cancer Res. Treat. 2012, 134, 769–781. [Google Scholar] [CrossRef]
- Phipps, A.I.; Chlebowski, R.T.; Prentice, R.; McTiernan, A.; Stefanick, M.L.; Wactawski-Wende, J.; Kuller, L.H.; Adams-Campbell, L.L.; Lane, D.; Vitolins, M.; et al. Body size, physical activity, and risk of triple-negative and estrogen receptor-positive breast cancer. Cancer Epidemiol. Biomark. Prev. 2011, 20, 454–463. [Google Scholar] [CrossRef] [PubMed]
- Fischer, J.P.; Cleveland, E.C.; Nelson, J.A.; Kovach, S.J.; Serletti, J.M.; Wu, L.C.; Kanchwala, S. Breast reconstruction in the morbidly obese patient: Assessment of 30-day complications using the 2005 to 2010 national surgical quality improvement program data sets. Plast. Reconstr. Surg. 2013, 132, 750–761. [Google Scholar] [CrossRef] [PubMed]
- Fischer, J.P.; Nelson, J.A.; Kovach, S.J.; Serletti, J.M.; Wu, L.C.; Kanchwala, S. Impact of obesity on outcomes in breast reconstruction: Analysis of 15,937 patients from the acs-nsqip datasets. J. Am. Coll. Surg. 2013, 217, 656–664. [Google Scholar] [CrossRef] [PubMed]
- Fischer, J.P.; Wes, A.M.; Tuggle, C.T.; Wu, L.C. Venous thromboembolism risk in mastectomy and immediate breast reconstruction: Analysis of the 2005 to 2011 American College of Surgeons National Surgical Quality Improvement Program data sets. Plast. Reconstr. Surg. 2014, 133, 263e–273e. [Google Scholar] [CrossRef] [PubMed]
- Jeevan, R.; Browne, J.P.; Pereira, J.; Caddy, C.M.; Sheppard, C.; van der Meulen, J.H.; Cromwell, D.A. Socioeconomic deprivation and inpatient complication rates following mastectomy and breast reconstruction surgery. Br. J. Surg. 2015, 102, 1064–1070. [Google Scholar] [CrossRef] [PubMed]
- DiSipio, T.; Rye, S.; Newman, B.; Hayes, S. Incidence of unilateral arm lymphoedema after breast cancer: A systematic review and meta-analysis. Lancet Oncol. 2013, 14, 500–515. [Google Scholar] [CrossRef]
- Goldsmith, C.; Haviland, J.; Tsang, Y.; Sydenham, M.; Yarnold, J.; Group, F.T. Large breast size as a risk factor for late adverse effects of breast radiotherapy: Is residual dose inhomogeneity, despite 3D treatment planning and delivery, the main explanation? Radiother. Oncol. 2011, 100, 236–240. [Google Scholar] [CrossRef]
- Dore, M.; Hennequin, C. Late sequelae and cosmetic outcome after radiotherapy in breast conserving therapy. Cancer Radiother. 2012, 16, 462–469. [Google Scholar]
- Verbelen, H.; Gebruers, N.; Beyers, T.; De Monie, A.C.; Tjalma, W. Breast edema in breast cancer patients following breast-conserving surgery and radiotherapy: A systematic review. Breast Cancer Res. Treat. 2014, 147, 463–471. [Google Scholar] [CrossRef]
- Ahn, J.; Ambrosone, C.B.; Kanetsky, P.A.; Tian, C.; Lehman, T.A.; Kropp, S.; Helmbold, I.; von Fournier, D.; Haase, W.; Sautter-Bihl, M.L.; et al. Polymorphisms in genes related to oxidative stress (CAT, MnSOD, MPO, and eNOS) and acute toxicities from radiation therapy following lumpectomy for breast cancer. Clin. Cancer Res. 2006, 12, 7063–7070. [Google Scholar] [CrossRef]
- Bergom, C.; Kelly, T.; Morrow, N.; Wilson, J.F.; Walker, A.; Xiang, Q.; Ahn, K.W.; White, J. Prone whole-breast irradiation using three-dimensional conformal radiotherapy in women undergoing breast conservation for early disease yields high rates of excellent to good cosmetic outcomes in patients with large and/or pendulous breasts. Int. J. Radiat. Oncol. Biol. Phys. 2012, 83, 821–828. [Google Scholar] [CrossRef] [PubMed]
- Griggs, J.J.; Sorbero, M.E.; Lyman, G.H. Undertreatment of obese women receiving breast cancer chemotherapy. Arch. Intern. Med. 2005, 165, 1267–1273. [Google Scholar] [CrossRef] [PubMed]
- Argolo, D.F.; Hudis, C.A.; Iyengar, N.M. The impact of obesity on breast cancer. Curr. Oncol. Rep. 2018, 20, 47. [Google Scholar] [CrossRef] [PubMed]
- Colleoni, M.; Li, S.; Gelber, R.D.; Price, K.N.; Coates, A.S.; Castiglione-Gertsch, M.; Goldhirsch, A. International Breast Cancer Study Group. Relation between chemotherapy dose, oestrogen receptor expression, and body-mass index. Lancet 2005, 366, 1108–1110. [Google Scholar] [CrossRef]
- Griggs, J.J.; Mangu, P.B.; Anderson, H.; Balaban, E.P.; Dignam, J.J.; Hryniuk, W.M.; Morrison, V.A.; Pini, T.M.; Runowicz, C.D.; Rosner, G.L.; et al. Appropriate chemotherapy dosing for obese adult patients with cancer: American Society of Clinical Oncology clinical practice guideline. J. Clin. Oncol. 2012, 30, 1553–1561. [Google Scholar] [CrossRef]
- Lyman, G.H.; Sparreboom, A. Chemotherapy dosing in overweight and obese patients with cancer. Nat. Rev. Clin. Oncol. 2013, 10, 451–459. [Google Scholar] [CrossRef]
- Rosner, G.L.; Hargis, J.B.; Hollis, D.R.; Budman, D.R.; Weiss, R.B.; Henderson, I.C.; Schilsky, R.L. Relationship between toxicity and obesity in women receiving adjuvant chemotherapy for breast cancer: Results from cancer and leukemia group b study 8541. J. Clin. Oncol. 1996, 14, 3000–3008. [Google Scholar] [CrossRef]
- Furlanetto, J.; Eiermann, W.; Marme, F.; Reimer, T.; Reinisch, M.; Schmatloch, S.; Stickeler, E.; Thomssen, C.; Untch, M.; Denkert, C.; et al. Higher rate of severe toxicities in obese patients receiving dose-dense (dd) chemotherapy according to unadjusted body surface area: Results of the prospectively randomized GAIN study. Ann. Oncol. 2016, 27, 2053–2059. [Google Scholar] [CrossRef]
- Irwin, M.L.; McTiernan, A.; Baumgartner, R.N.; Baumgartner, K.B.; Bernstein, L.; Gilliland, F.D.; Ballard-Barbash, R. Changes in body fat and weight after a breast cancer diagnosis: Influence of demographic, prognostic, and lifestyle factors. J. Clin. Oncol. 2005, 23, 774–782. [Google Scholar] [CrossRef]
- Lauby-Secretan, B.; Scoccianti, C.; Loomis, D.; Grosse, Y.; Bianchini, F.; Straif, K. International Agency for Research on Cancer Handbook Working Group. Body fatness and cancer—Viewpoint of the IARC working group. N. Engl. J. Med. 2016, 375, 794–798. [Google Scholar] [CrossRef]
- Sestak, I.; Distler, W.; Forbes, J.F.; Dowsett, M.; Howell, A.; Cuzick, J. Effect of body mass index on recurrences in tamoxifen and anastrozole treated women: An exploratory analysis from the ATAC trial. J. Clin. Oncol. 2010, 28, 3411–3415. [Google Scholar] [CrossRef] [PubMed]
- Pfeiler, G.; Konigsberg, R.; Fesl, C.; Mlineritsch, B.; Stoeger, H.; Singer, C.F.; Postlberger, S.; Steger, G.G.; Seifert, M.; Dubsky, P.; et al. Impact of body mass index on the efficacy of endocrine therapy in premenopausal patients with breast cancer: An analysis of the prospective ABCSG-12 trial. J. Clin. Oncol. 2011, 29, 2653–2659. [Google Scholar] [CrossRef]
- Ewertz, M.; Gray, K.P.; Regan, M.M.; Ejlertsen, B.; Price, K.N.; Thurlimann, B.; Bonnefoi, H.; Forbes, J.F.; Paridaens, R.J.; Rabaglio, M.; et al. Obesity and risk of recurrence or death after adjuvant endocrine therapy with letrozole or tamoxifen in the breast international group 1-98 trial. J. Clin. Oncol. 2012, 30, 3967–3975. [Google Scholar] [CrossRef]
- Dignam, J.J.; Wieand, K.; Johnson, K.A.; Fisher, B.; Xu, L.; Mamounas, E.P. Obesity, tamoxifen use, and outcomes in women with estrogen receptor-positive early-stage breast cancer. J. Natl. Cancer Inst. 2003, 95, 1467–1476. [Google Scholar] [CrossRef] [PubMed]
- Lippman, M.E.; Krueger, K.A.; Eckert, S.; Sashegyi, A.; Walls, E.L.; Jamal, S.; Cauley, J.A.; Cummings, S.R. Indicators of lifetime estrogen exposure: Effect on breast cancer incidence and interaction with raloxifene therapy in the multiple outcomes of raloxifene evaluation study participants. J. Clin. Oncol. 2001, 19, 3111–3116. [Google Scholar] [CrossRef] [PubMed]
- Sheng, J.Y.; Sharma, D.; Jerome, G.; Santa-Maria, C.A. Obese breast cancer patients and survivors: Management considerations. Oncology 2018, 32, 410–417. [Google Scholar] [PubMed]
- Elme, A.; Utriainen, M.; Kellokumpu-Lehtinen, P.; Palva, T.; Luoto, R.; Nikander, R.; Huovinen, R.; Kautiainen, H.; Järvenpää, S.; Penttinen, H.M.; et al. Obesity and physical inactivity are related to impaired physical health of breast cancer survivors. Anticancer Res. 2013, 33, 1595–1602. [Google Scholar] [PubMed]
- Connor, A.E.; Baumgartner, R.N.; Pinkston, C.M.; Boone, S.D.; Baumgartner, K.B. Obesity, ethnicity, and quality of life among breast cancer survivors and women without breast cancer: The long-term quality of life follow-up study. Cancer Causes Control 2016, 27, 115–124. [Google Scholar] [CrossRef] [PubMed]
- Pakiz, B.; Ganz, P.A.; Sedjo, R.L.; Flatt, S.W.; Demark-Wahnefried, W.; Liu, J.; Wolin, K.Y.; Rock, C.L. Correlates of quality of life in overweight or obese breast cancer survivors at enrollment into a weight loss trial. Psychooncology 2016, 25, 142–149. [Google Scholar] [CrossRef] [PubMed]
- Sun, Y.; Bao, W.; Liu, B.; Caan, B.J.; Lane, D.S.; Millen, A.E.; Simon, M.S.; Thomson, C.A.; Tinker, L.F.; Van Horn, L.V.; et al. Changes in overall diet quality in relation to survival in postmenopausal women with breast cancer: Results from the women’s health initiative. J. Acad. Nutr. Diet. 2018, 118, 1855–1863. [Google Scholar] [CrossRef] [PubMed]
- Orchard, T.S.; Andridge, R.R.; Yee, L.D.; Lustberg, M.B. Diet quality, inflammation, and quality of life in breast cancer survivors: A cross-sectional analysis of pilot study data. J. Acad. Nutr. Diet. 2018, 118, 578–588. [Google Scholar] [CrossRef] [PubMed]
- Kang, D.W.; Lee, J.; Suh, S.H.; Ligibel, J.; Courneya, K.S.; Jeon, J.Y. Effects of exercise on insulin, IGF axis, adipocytokines, and inflammatory markers in breast cancer survivors: A systematic review and meta-analysis. Cancer Epidemiol. Biomark. Prev. 2017, 26, 355–365. [Google Scholar] [CrossRef] [PubMed]
- Dossus, L.; Jimenez-Corona, A.; Romieu, I.; Boutron-Ruault, M.C.; Boutten, A.; Dupre, T.; Fagherazzi, G.; Clavel-Chapelon, F.; Mesrine, S. C-reactive protein and postmenopausal breast cancer risk: Results from the E3N cohort study. Cancer Causes Control 2014, 25, 533–539. [Google Scholar] [CrossRef] [PubMed]
- Pierce, B.L.; Ballard-Barbash, R.; Bernstein, L.; Baumgartner, R.N.; Neuhouser, M.L.; Wener, M.H.; Baumgartner, K.B.; Gilliland, F.D.; Sorensen, B.E.; McTiernan, A.; et al. Elevated biomarkers of inflammation are associated with reduced survival among breast cancer patients. J. Clin. Oncol. 2009, 27, 3437–3444. [Google Scholar] [CrossRef] [PubMed]
- Fuentes-Mattei, E.; Velazquez-Torres, G.; Phan, L.; Zhang, F.; Chou, P.C.; Shin, J.H.; Choi, H.H.; Chen, J.S.; Zhao, R.; Chen, J.; et al. Effects of obesity on transcriptomic changes and cancer hallmarks in estrogen receptor-positive breast cancer. J. Natl. Cancer Inst. 2014, 106, dju158. [Google Scholar] [CrossRef] [PubMed]
- Weisberg, S.P.; McCann, D.; Desai, M.; Rosenbaum, M.; Leibel, R.L.; Ferrante, A.W., Jr. Obesity is associated with macrophage accumulation in adipose tissue. J. Clin. Investig. 2003, 112, 1796–1808. [Google Scholar] [CrossRef] [PubMed]
- Lumeng, C.N.; Bodzin, J.L.; Saltiel, A.R. Obesity induces a phenotypic switch in adipose tissue macrophage polarization. J. Clin. Investig. 2007, 117, 175–184. [Google Scholar] [CrossRef]
- Gilbert, C.A.; Slingerland, J.M. Cytokines, obesity, and cancer: New insights on mechanisms linking obesity to cancer risk and progression. Annu. Rev. Med. 2013, 64, 45–57. [Google Scholar] [CrossRef]
- Cinti, S.; Mitchell, G.; Barbatelli, G.; Murano, I.; Ceresi, E.; Faloia, E.; Wang, S.; Fortier, M.; Greenberg, A.S.; Obin, M.S. Adipocyte death defines macrophage localization and function in adipose tissue of obese mice and humans. J. Lipid Res. 2005, 46, 2347–2355. [Google Scholar] [CrossRef]
- Howe, L.R.; Subbaramaiah, K.; Hudis, C.A.; Dannenberg, A.J. Molecular pathways: Adipose inflammation as a mediator of obesity-associated cancer. Clin. Cancer Res. 2013, 19, 6074–6083. [Google Scholar] [CrossRef]
- Iyengar, N.M.; Morris, P.G.; Zhou, X.K.; Gucalp, A.; Giri, D.; Harbus, M.D.; Falcone, D.J.; Krasne, M.D.; Vahdat, L.T.; Subbaramaiah, K.; et al. Menopause is a determinant of breast adipose inflammation. Cancer Prev. Res. 2015, 8, 349–358. [Google Scholar] [CrossRef] [PubMed]
- Iyengar, N.M.; Zhou, X.K.; Gucalp, A.; Morris, P.G.; Howe, L.R.; Giri, D.D.; Morrow, M.; Wang, H.; Pollak, M.; Jones, L.W.; et al. Systemic correlates of white adipose tissue inflammation in early-stage breast cancer. Clin. Cancer Res. 2016, 22, 2283–2289. [Google Scholar] [CrossRef] [PubMed]
- Koru-Sengul, T.; Santander, A.M.; Miao, F.; Sanchez, L.G.; Jorda, M.; Gluck, S.; Ince, T.A.; Nadji, M.; Chen, Z.; Penichet, M.L.; et al. Breast cancers from black women exhibit higher numbers of immunosuppressive macrophages with proliferative activity and of crown-like structures associated with lower survival compared to non-black latinas and caucasians. Breast Cancer Res. Treat. 2016, 158, 113–126. [Google Scholar] [CrossRef] [PubMed]
- Lamkanfi, M.; Dixit, V.M. Mechanisms and functions of inflammasomes. Cell 2014, 157, 1013–1022. [Google Scholar] [CrossRef] [PubMed]
- Yin, Z.; Deng, T.; Peterson, L.E.; Yu, R.; Lin, J.; Hamilton, D.J.; Reardon, P.R.; Sherman, V.; Winnier, G.E.; Zhan, M.; et al. Transcriptome analysis of human adipocytes implicates the NOD-like receptor pathway in obesity-induced adipose inflammation. Mol. Cell. Endocrinol. 2014, 394, 80–87. [Google Scholar] [CrossRef] [PubMed]
- Guo, B.; Fu, S.; Zhang, J.; Liu, B.; Li, Z. Targeting inflammasome/IL-1 pathways for cancer immunotherapy. Sci. Rep. 2016, 6, 36107. [Google Scholar] [CrossRef] [PubMed]
- Kolb, R.; Phan, L.; Borcherding, N.; Liu, Y.; Yuan, F.; Janowski, A.M.; Xie, Q.; Markan, K.R.; Li, W.; Potthoff, M.J.; et al. Obesity-associated NLRC4 inflammasome activation drives breast cancer progression. Nat. Commun. 2016, 7, 13007. [Google Scholar] [CrossRef] [PubMed]
- Gunter, M.J.; Hoover, D.R.; Yu, H.; Wassertheil-Smoller, S.; Rohan, T.E.; Manson, J.E.; Li, J.; Ho, G.Y.; Xue, X.; Anderson, G.L.; et al. Insulin, insulin-like growth factor-I, and risk of breast cancer in postmenopausal women. J. Natl. Cancer Inst. 2009, 101, 48–60. [Google Scholar] [CrossRef] [PubMed]
- Verheus, M.; Peeters, P.H.; Rinaldi, S.; Dossus, L.; Biessy, C.; Olsen, A.; Tjonneland, A.; Overvad, K.; Jeppesen, M.; Clavel-Chapelon, F.; et al. Serum c-peptide levels and breast cancer risk: Results from the european prospective investigation into cancer and nutrition (EPIC). Int. J. Cancer 2006, 119, 659–667. [Google Scholar] [CrossRef] [PubMed]
- Autier, P.; Koechlin, A.; Boniol, M.; Mullie, P.; Bolli, G.; Rosenstock, J.; Boyle, P. Serum insulin and c-peptide concentration and breast cancer: A meta-analysis. Cancer Causes Control 2013, 24, 873–883. [Google Scholar] [CrossRef] [PubMed]
- Endogenous Hormones and Breast Cancer Collaborative Group; Key, T.J.; Appleby, P.N.; Reeves, G.K.; Roddam, A.W. Insulin-like growth factor 1 (IGF1), IGF binding protein 3 (IGFBP3), and breast cancer risk: Pooled individual data analysis of 17 prospective studies. Lancet Oncol. 2010, 11, 530–542. [Google Scholar] [PubMed]
- Goodwin, P.J.; Ennis, M.; Pritchard, K.I.; Trudeau, M.E.; Koo, J.; Madarnas, Y.; Hartwick, W.; Hoffman, B.; Hood, N. Fasting insulin and outcome in early-stage breast cancer: Results of a prospective cohort study. J. Clin. Oncol. 2002, 20, 42–51. [Google Scholar] [CrossRef] [PubMed]
- Calori, G.; Lattuada, G.; Piemonti, L.; Garancini, M.P.; Ragogna, F.; Villa, M.; Mannino, S.; Crosignani, P.; Bosi, E.; Luzi, L. Prevalence, metabolic features, and prognosis of metabolically healthy obese italian individuals: The Cremona study. Diabetes Care 2011, 34, 210–215. [Google Scholar] [CrossRef] [PubMed]
- Duggan, C.; Irwin, M.L.; Xiao, L.; Henderson, K.D.; Smith, A.W.; Baumgartner, R.N.; Baumgartner, K.B.; Bernstein, L.; Ballad-Barbash, R.; McTiernan, A. Associations of insulin resistance and adiponectin with mortality in women with breast cancer. J. Clin. Oncol. 2011, 29, 32–39. [Google Scholar] [CrossRef] [PubMed]
- Law, J.H.; Habibi, G.; Hu, K.; Masoudi, H.; Wang, M.Y.; Stratford, A.L.; Park, E.; Gee, J.M.; Finlay, P.; Jones, H.E.; et al. Phosphorylated insulin-like growth factor-i/insulin receptor is present in all breast cancer subtypes and is related to poor survival. Cancer Res. 2008, 68, 10238–10246. [Google Scholar] [CrossRef] [PubMed]
- Christopoulos, P.F.; Msaouel, P.; Koutsilieris, M. The role of the insulin-like growth factor-1 system in breast cancer. Mol. Cancer 2015, 14, 43. [Google Scholar] [CrossRef] [PubMed]
- Ekyalongo, R.C.; Yee, D. Revisiting the IGF-1R as a breast cancer target. NPJ Precis. Oncol. 2017, 1, 14. [Google Scholar] [CrossRef] [PubMed]
- Christopoulos, P.F.; Corthay, A.; Koutsilieris, M. Aiming for the insulin-like growth factor-1 system in breast cancer therapeutics. Cancer Treat. Rev. 2018, 63, 79–95. [Google Scholar] [CrossRef] [PubMed]
- Endogenous Hormones and Breast Cancer Collaborative Group; Key, T.J.; Appleby, P.N.; Reeves, G.K.; Travis, R.C.; Alberg, A.J.; Barricarte, A.; Berrino, F.; Krogh, V.; Sieri, S.; et al. Sex hormones and risk of breast cancer in premenopausal women: A collaborative reanalysis of individual participant data from seven prospective studies. Lancet Oncol. 2013, 14, 1009–1019. [Google Scholar] [PubMed]
- Kaaks, R.; Tikk, K.; Sookthai, D.; Schock, H.; Johnson, T.; Tjonneland, A.; Olsen, A.; Overvad, K.; Clavel-Chapelon, F.; Dossus, L.; et al. Premenopausal serum sex hormone levels in relation to breast cancer risk, overall and by hormone receptor status—Results from the EPIC cohort. Int. J. Cancer 2014, 134, 1947–1957. [Google Scholar] [CrossRef] [PubMed]
- Key, T.J.; Appleby, P.N.; Reeves, G.K.; Roddam, A.; Dorgan, J.F.; Longcope, C.; Stancyzyk, F.Z.; Stephenson, H.E.; Falk, R.T.; Miller, R.; et al. Body mass index, serum sex hormones, and breast cancer risk in postmenopausal women. J. Natl. Cancer Inst. 2003, 95, 1218–1226. [Google Scholar] [PubMed]
- Suzuki, R.; Orsini, N.; Saji, S.; Key, T.J.; Wolk, A. Body weight and incidence of breast cancer defined by estrogen and progesterone receptor status—A meta-analysis. Int. J. Cancer 2009, 124, 698–712. [Google Scholar] [CrossRef] [PubMed]
- Vrieling, A.; Buck, K.; Kaaks, R.; Chang-Claude, J. Adult weight gain in relation to breast cancer risk by estrogen and progesterone receptor status: A meta-analysis. Breast Cancer Res. Treat. 2010, 123, 641–649. [Google Scholar] [CrossRef] [PubMed]
- Bezemer, I.D.; Rinaldi, S.; Dossus, L.; Gils, C.H.; Peeters, P.H.; Noord, P.A.; Bueno-de-Mesquita, H.B.; Johnsen, S.P.; Overvad, K.; Olsen, A.; et al. C-peptide, IGF-I, sex-steroid hormones and adiposity: A cross-sectional study in healthy women within the european prospective investigation into cancer and nutrition (EPIC). Cancer Causes Control 2005, 16, 561–572. [Google Scholar] [CrossRef] [PubMed]
- Boyapati, S.M.; Shu, X.O.; Gao, Y.T.; Dai, Q.; Yu, H.; Cheng, J.R.; Jin, F.; Zheng, W. Correlation of blood sex steroid hormones with body size, body fat distribution, and other known risk factors for breast cancer in post-menopausal chinese women. Cancer Causes Control 2004, 15, 305–311. [Google Scholar] [CrossRef] [PubMed]
- Lukanova, A.; Lundin, E.; Zeleniuch-Jacquotte, A.; Muti, P.; Mure, A.; Rinaldi, S.; Dossus, L.; Micheli, A.; Arslan, A.; Lenner, P.; et al. Body mass index, circulating levels of sex-steroid hormones, IGF-I and IGF-binding protein-3: A cross-sectional study in healthy women. Eur. J. Endocrinol. 2004, 150, 161–171. [Google Scholar] [CrossRef] [PubMed]
- McTiernan, A.; Wu, L.; Chen, C.; Chlebowski, R.; Mossavar-Rahmani, Y.; Modugno, F.; Perri, M.G.; Stanczyk, F.Z.; Van Horn, L.; Wang, C.Y.; et al. Relation of BMI and physical activity to sex hormones in postmenopausal women. Obesity 2006, 14, 1662–1677. [Google Scholar] [CrossRef] [PubMed]
- Riedt, C.S.; Brolin, R.E.; Sherrell, R.M.; Field, M.P.; Shapses, S.A. True fractional calcium absorption is decreased after Roux-en-Y gastric bypass surgery. Obesity 2006, 14, 1940–1948. [Google Scholar] [CrossRef] [PubMed]
- Bulun, S.E.; Chen, D.; Moy, I.; Brooks, D.C.; Zhao, H. Aromatase, breast cancer and obesity: A complex interaction. Trends Endocrinol. MeTable 2012, 23, 83–89. [Google Scholar] [CrossRef]
- Bulun, S.E.; Lin, Z.; Imir, G.; Amin, S.; Demura, M.; Yilmaz, B.; Martin, R.; Utsunomiya, H.; Thung, S.; Gurates, B.; et al. Regulation of aromatase expression in estrogen-responsive breast and uterine disease: From bench to treatment. Pharmacol. Rev. 2005, 57, 359–383. [Google Scholar] [CrossRef]
- Subbaramaiah, K.; Morris, P.G.; Zhou, X.K.; Morrow, M.; Du, B.; Giri, D.; Kopelovich, L.; Hudis, C.A.; Dannenberg, A.J. Increased levels of COX-2 and prostaglandin E2 contribute to elevated aromatase expression in inflamed breast tissue of obese women. Cancer Discov. 2012, 2, 356–365. [Google Scholar] [CrossRef] [PubMed]
- Hotamisligil, G.S.; Arner, P.; Caro, J.F.; Atkinson, R.L.; Spiegelman, B.M. Increased adipose tissue expression of tumor necrosis factor-alpha in human obesity and insulin resistance. J. Clin. Investig. 1995, 95, 2409–2415. [Google Scholar] [CrossRef] [PubMed]
- Catalano, S.; Marsico, S.; Giordano, C.; Mauro, L.; Rizza, P.; Panno, M.L.; Andò, S. Leptin enhances, via AP-1, expression of aromatase in the MCF-7 cell line. J. Biol. Chem. 2003, 278, 28668–28676. [Google Scholar] [CrossRef] [PubMed]
- Barone, I.; Giordano, C.; Malivindi, R.; Lanzino, M.; Rizza, P.; Casaburi, I.; Bonofiglio, D.; Catalano, S.; Andò, S. Estrogens and PTP1B function in a novel pathway to regulate aromatase enzymatic activity in breast cancer cells. Endocrinology 2012, 153, 5157–5166. [Google Scholar] [CrossRef] [PubMed]
- Chen, D.; Reierstad, S.; Lin, Z.; Lu, M.; Brooks, C.; Li, N.; Innes, J.; Bulun, S.E. Prostaglandin E(2) induces breast cancer related aromatase promoters via activation of p38 and c-Jun NH(2)-terminal kinase in adipose fibroblasts. Cancer Res. 2007, 67, 8914–8922. [Google Scholar] [CrossRef] [PubMed]
- Catalano, S.; Barone, I.; Giordano, C.; Rizza, P.; Qi, H.; Gu, G.; Malivindi, R.; Bonofiglio, D.; Andò, S. Rapid estradiol/ERalpha signaling enhances aromatase enzymatic activity in breast cancer cells. Mol. Endocrinol. 2009, 23, 1634–1645. [Google Scholar] [CrossRef] [PubMed]
- Barone, I.; Brusco, L.; Fuqua, S.A. Estrogen receptor mutations and changes in downstream gene expression and signaling. Clin. Cancer Res. 2010, 16, 2702–2708. [Google Scholar] [CrossRef] [PubMed]
- Matafome, P.; Santos-Silva, D.; Sena, C.M.; Seica, R. Common mechanisms of dysfunctional adipose tissue and obesity-related cancers. Diabetes Metab. Res. Rev. 2013, 29, 285–295. [Google Scholar] [CrossRef] [PubMed]
- Choi, J.; Cha, Y.J.; Koo, J.S. Adipocyte biology in breast cancer: From silent bystander to active facilitator. Prog. Lipid Res. 2018, 69, 11–20. [Google Scholar] [CrossRef] [PubMed]
- Dalamaga, M.; Diakopoulos, K.N.; Mantzoros, C.S. The role of adiponectin in cancer: A review of current evidence. Endocr. Rev. 2012, 33, 547–594. [Google Scholar] [CrossRef] [PubMed]
- Boudeau, J.; Baas, A.F.; Deak, M.; Morrice, N.A.; Kieloch, A.; Schutkowski, M.; Prescott, A.R.; Clevers, H.C.; Alessi, D.R. MO25α/β interact with STRADα/β enhancing their ability to bind, activate and localize LKB1 in the cytoplasm. EMBO J. 2003, 22, 5102–5114. [Google Scholar] [CrossRef] [PubMed]
- Alessi, D.R.; Sakamoto, K.; Bayascas, J.R. LKB1-dependent signaling pathways. Annu. Rev. Biochem. 2006, 75, 137–163. [Google Scholar] [CrossRef] [PubMed]
- Miyoshi, Y.; Funahashi, T.; Kihara, S.; Taguchi, T.; Tamaki, Y.; Matsuzawa, Y.; Noguchi, S. Association of serum adiponectin levels with breast cancer risk. Clin. Cancer Res. 2003, 9, 5699–5704. [Google Scholar] [PubMed]
- Mantzoros, C.; Petridou, E.; Dessypris, N.; Chavelas, C.; Dalamaga, M.; Alexe, D.M.; Papadiamantis, Y.; Markopoulos, C.; Spanos, E.; Chrousos, G. Adiponectin and breast cancer risk. J. Clin. Endocrinol. Metab. 2004, 89, 1102–1107. [Google Scholar] [CrossRef] [PubMed]
- Tworoger, S.S.; Eliassen, A.H.; Kelesidis, T.; Colditz, G.A.; Willett, W.C.; Mantzoros, C.S.; Hankinson, S.E. Plasma adiponectin concentrations and risk of incident breast cancer. J. Clin. Endocrinol. Metab. 2007, 92, 1510–1516. [Google Scholar] [CrossRef] [PubMed]
- Macis, D.; Guerrieri-Gonzaga, A.; Gandini, S. Circulating adiponectin and breast cancer risk: A systematic review and meta-analysis. Int. J. Epidemiol. 2014, 43, 1226–1236. [Google Scholar] [CrossRef] [PubMed]
- Oh, S.W.; Park, C.Y.; Lee, E.S.; Yoon, Y.S.; Lee, E.S.; Park, S.S.; Kim, Y.; Sung, N.J.; Yun, Y.H.; Lee, K.S.; et al. Adipokines, insulin resistance, metabolic syndrome, and breast cancer recurrence: A cohort study. Breast Cancer Res. 2011, 13, R34. [Google Scholar] [CrossRef] [PubMed]
- Jeong, Y.J.; Bong, J.G.; Park, S.H.; Choi, J.H.; Oh, H.K. Expression of leptin, leptin receptor, adiponectin, and adiponectin receptor in ductal carcinoma in situ and invasive breast cancer. J. Breast Cancer 2011, 14, 96–103. [Google Scholar] [CrossRef] [PubMed]
- Kang, J.H.; Lee, Y.Y.; Yu, B.Y.; Yang, B.S.; Cho, K.H.; Yoon, D.K.; Roh, Y.K. Adiponectin induces growth arrest and apoptosis of MDA-MB-231 breast cancer cell. Arch. Pharm. Res. 2005, 28, 1263–1269. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Lam, J.B.; Lam, K.S.; Liu, J.; Lam, M.C.; Hoo, R.L.; Wu, D.; Cooper, G.J.; Xu, A. Adiponectin modulates the glycogen synthase kinase-3β/β-catenin signaling pathway and attenuates mammary tumorigenesis of MDA-MB-231 cells in nude mice. Cancer Res. 2006, 66, 11462–11470. [Google Scholar] [CrossRef] [PubMed]
- Dos Santos, E.; Benaitreau, D.; Dieudonne, M.N.; Leneveu, M.C.; Serazin, V.; Giudicelli, Y.; Pecquery, R. Adiponectin mediates an antiproliferative response in human MDA-MB 231 breast cancer cells. Oncol. Rep. 2008, 20, 971–977. [Google Scholar] [PubMed]
- Grossmann, M.E.; Nkhata, K.J.; Mizuno, N.K.; Ray, A.; Cleary, M.P. Effects of adiponectin on breast cancer cell growth and signaling. Br. J. Cancer 2008, 98, 370–379. [Google Scholar] [CrossRef] [PubMed]
- Nakayama, S.; Miyoshi, Y.; Ishihara, H.; Noguchi, S. Growth-inhibitory effect of adiponectin via adiponectin receptor 1 on human breast cancer cells through inhibition of s-phase entry without inducing apoptosis. Breast Cancer Res. Treat. 2008, 112, 405–410. [Google Scholar] [CrossRef] [PubMed]
- Panno, M.L.; Naimo, G.D.; Spina, E.; Andò, S.; Mauro, L. Different molecular signaling sustaining adiponectin action in breast cancer. Curr. Opin. Pharmacol. 2016, 31, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Mauro, L.; Pellegrino, M.; De Amicis, F.; Ricchio, E.; Giordano, F.; Rizza, P.; Catalano, S.; Bonofiglio, D.; Sisci, D.; Panno, M.L.; et al. Evidences that estrogen receptor alpha interferes with adiponectin effects on breast cancer cell growth. Cell Cycle 2014, 13, 553–564. [Google Scholar] [CrossRef] [PubMed]
- Mauro, L.; Pellegrino, M.; Giordano, F.; Ricchio, E.; Rizza, P.; De Amicis, F.; Catalano, S.; Bonofiglio, D.; Panno, M.L.; Andò, S. Estrogen receptor-alpha drives adiponectin effects on cyclin D1 expression in breast cancer cells. FASEB J. 2015, 29, 2150–2160. [Google Scholar] [CrossRef] [PubMed]
- Mauro, L.; Naimo, G.D.; Gelsomino, L.; Malivindi, R.; Bruno, L.; Pellegrino, M.; Tarallo, R.; Memoli, D.; Weisz, A.; Panno, M.L.; et al. Uncoupling effects of estrogen receptor alpha on LKB1/AMPK interaction upon adiponectin exposure in breast cancer. FASEB J. 2018, 32, 4343–4355. [Google Scholar] [CrossRef] [PubMed]
- Andò, S.; Catalano, S. The multifactorial role of leptin in driving the breast cancer microenvironment. Nat. Rev. Endocrinol. 2011, 8, 263–275. [Google Scholar] [CrossRef] [PubMed]
- Andò, S.; Barone, I.; Giordano, C.; Bonofiglio, D.; Catalano, S. The multifaceted mechanism of leptin signaling within tumor microenvironment in driving breast cancer growth and progression. Front. Oncol. 2014, 4, 340. [Google Scholar] [PubMed]
- Barone, I.; Giordano, C.; Bonofiglio, D.; Andò, S.; Catalano, S. Leptin, obesity and breast cancer: Progress to understanding the molecular connections. Curr. Opin. Pharmacol. 2016, 31, 83–89. [Google Scholar] [CrossRef] [PubMed]
- Giordano, C.; Barone, I.; Vircillo, V.; Panza, S.; Malivindi, R.; Gelsomino, L.; Pellegrino, M.; Rago, V.; Mauro, L.; Lanzino, M.; et al. Activated FXR inhibits leptin signaling and counteracts tumor-promoting activities of cancer-associated fibroblasts in breast malignancy. Sci. Rep. 2016, 6, 21782. [Google Scholar] [CrossRef] [PubMed]
- Barone, I.; Catalano, S.; Gelsomino, L.; Marsico, S.; Giordano, C.; Panza, S.; Bonofiglio, D.; Bossi, G.; Covington, K.R.; Fuqua, S.A.; et al. Leptin mediates tumor-stromal interactions that promote the invasive growth of breast cancer cells. Cancer Res. 2012, 72, 1416–1427. [Google Scholar] [CrossRef] [PubMed]
- Giordano, C.; Chemi, F.; Panza, S.; Barone, I.; Bonofiglio, D.; Lanzino, M.; Cordella, A.; Campana, A.; Hashim, A.; Rizza, P.; et al. Leptin as a mediator of tumor-stromal interactions promotes breast cancer stem cell activity. Oncotarget 2016, 7, 1262–1275. [Google Scholar] [CrossRef] [PubMed]
- Niu, J.; Jiang, L.; Guo, W.; Shao, L.; Liu, Y.; Wang, L. The association between leptin level and breast cancer: A meta-analysis. PLoS ONE 2013, 8, e67349. [Google Scholar] [CrossRef] [PubMed]
- Pan, H.; Deng, L.L.; Cui, J.Q.; Shi, L.; Yang, Y.C.; Luo, J.H.; Qin, D.; Wang, L. Association between serum leptin levels and breast cancer risk: An updated systematic review and meta-analysis. Medicine 2018, 97, e11345. [Google Scholar] [CrossRef] [PubMed]
- Gui, Y.; Pan, Q.; Chen, X.; Xu, S.; Luo, X.; Chen, L. The association between obesity related adipokines and risk of breast cancer: A meta-analysis. Oncotarget 2017, 8, 75389–75399. [Google Scholar] [CrossRef] [PubMed]
- Ishikawa, M.; Kitayama, J.; Nagawa, H. Enhanced expression of leptin and leptin receptor (OB-R) in human breast cancer. Clin. Cancer Res. 2004, 10, 4325–4331. [Google Scholar] [CrossRef] [PubMed]
- Garofalo, C.; Koda, M.; Cascio, S.; Sulkowska, M.; Kanczuga-Koda, L.; Golaszewska, J.; Russo, A.; Sulkowski, S.; Surmacz, E. Increased expression of leptin and the leptin receptor as a marker of breast cancer progression: Possible role of obesity-related stimuli. Clin. Cancer Res. 2006, 12, 1447–1453. [Google Scholar] [CrossRef] [PubMed]
- Jarde, T.; Caldefie-Chezet, F.; Damez, M.; Mishellany, F.; Penault-Llorca, F.; Guillot, J.; Vasson, M.P. Leptin and leptin receptor involvement in cancer development: A study on human primary breast carcinoma. Oncol. Rep. 2008, 19, 905–911. [Google Scholar] [CrossRef] [PubMed]
- Miyoshi, Y.; Funahashi, T.; Tanaka, S.; Taguchi, T.; Tamaki, Y.; Shimomura, I.; Noguchi, S. High expression of leptin receptor mRNA in breast cancer tissue predicts poor prognosis for patients with high, but not low, serum leptin levels. Int. J. Cancer 2006, 118, 1414–1419. [Google Scholar] [CrossRef] [PubMed]
- Lee, W.M.; Lu, S.; Medline, A.; Archer, M.C. Susceptibility of lean and obese Zucker rats to tumorigenesis induced by N-methyl-N-nitrosourea. Cancer Lett. 2001, 162, 155–160. [Google Scholar] [CrossRef]
- Cleary, M.P.; Phillips, F.C.; Getzin, S.C.; Jacobson, T.L.; Jacobson, M.K.; Christensen, T.A.; Juneja, S.C.; Grande, J.P.; Maihle, N.J. Genetically obese MMTV-TGF-alpha/Lep(ob)Lep(ob) female mice do not develop mammary tumors. Breast Cancer Res. Treat. 2003, 77, 205–215. [Google Scholar] [CrossRef] [PubMed]
- Cleary, M.P.; Juneja, S.C.; Phillips, F.C.; Hu, X.; Grande, J.P.; Maihle, N.J. Leptin receptor-deficient MMTV-TGF-alpha/Lepr(db)Lepr(db) female mice do not develop oncogene-induced mammary tumors. Exp. Biol. Med. 2004, 229, 182–193. [Google Scholar] [CrossRef]
- Park, J.; Kusminski, C.M.; Chua, S.C.; Scherer, P.E. Leptin receptor signaling supports cancer cell metabolism through suppression of mitochondrial respiration in vivo. Am. J. Pathol. 2010, 177, 3133–3144. [Google Scholar] [CrossRef] [PubMed]
- Zheng, Q.; Dunlap, S.M.; Zhu, J.; Downs-Kelly, E.; Rich, J.; Hursting, S.D.; Berger, N.A.; Reizes, O. Leptin deficiency suppresses MMTV-Wnt-1 mammary tumor growth in obese mice and abrogates tumor initiating cell survival. Endocr. Relat. Cancer 2011, 18, 491–503. [Google Scholar] [CrossRef] [PubMed]
- Chang, C.C.; Wu, M.J.; Yang, J.Y.; Camarillo, I.G.; Chang, C.J. Leptin-STAT3-G9a signaling promotes obesity-mediated breast cancer progression. Cancer Res. 2015, 75, 2375–2386. [Google Scholar] [CrossRef] [PubMed]
- Strong, A.L.; Ohlstein, J.F.; Biagas, B.A.; Rhodes, L.V.; Pei, D.T.; Tucker, H.A.; LIamas, C.; Bowles, A.C.; Dutreil, M.F.; Zhang, S.; et al. Leptin produced by obese adipose stromal/stem cells enhances proliferation and metastasis of estrogen receptor positive breast cancers. Breast Cancer Res. 2015, 17, 112. [Google Scholar] [CrossRef] [PubMed]
- Catalano, S.; Mauro, L.; Marsico, S.; Giordano, C.; Rizza, P.; Rago, V.; Montanaro, D.; Maggiolini, M.; Panno, M.L.; Andò, S. Leptin induces, via ERK1/ERK2 signal, functional activation of estrogen receptor alpha in MCF-7 cells. J. Biol. Chem. 2004, 279, 19908–19915. [Google Scholar] [CrossRef] [PubMed]
- Mauro, L.; Catalano, S.; Bossi, G.; Pellegrino, M.; Barone, I.; Morales, S.; Giordano, C.; Bartella, V.; Casaburi, I.; Andò, S. Evidences that leptin up-regulates E-cadherin expression in breast cancer: Effects on tumor growth and progression. Cancer Res. 2007, 67, 3412–3421. [Google Scholar] [CrossRef] [PubMed]
- Garofalo, C.; Sisci, D.; Surmacz, E. Leptin interferes with the effects of the antiestrogen ICI 182,780 in MCF-7 breast cancer cells. Clin. Cancer Res. 2004, 10, 6466–6475. [Google Scholar] [CrossRef] [PubMed]
- Fiorio, E.; Mercanti, A.; Terrasi, M.; Micciolo, R.; Remo, A.; Auriemma, A.; Molino, A.; Parolin, V.; Di Stefano, B.; Bonetti, F.; et al. Leptin/Her2 crosstalk in breast cancer: In vitro study and preliminary in vivo analysis. BMC Cancer 2008, 8, 305. [Google Scholar] [CrossRef] [PubMed]
- Saxena, N.K.; Taliaferro-Smith, L.; Knight, B.B.; Merlin, D.; Anania, F.A.; O’Regan, R.M.; Sharma, D. Bidirectional crosstalk between leptin and insulin-like growth factor-I signaling promotes invasion and migration of breast cancer cells via transactivation of epidermal growth factor receptor. Cancer Res. 2008, 68, 9712–9722. [Google Scholar] [CrossRef] [PubMed]
- Soma, D.; Kitayama, J.; Yamashita, H.; Miyato, H.; Ishikawa, M.; Nagawa, H. Leptin augments proliferation of breast cancer cells via transactivation of Her2. J. Surg. Res. 2008, 149, 9–14. [Google Scholar] [CrossRef] [PubMed]
- Giordano, C.; Vizza, D.; Panza, S.; Barone, I.; Bonofiglio, D.; Lanzino, M.; Sisci, D.; De Amicis, F.; Fuqua, S.A.; Catalano, S.; et al. Leptin increases HER2 protein levels through a STAT3-mediated up-regulation of Hsp90 in breast cancer cells. Mol. Oncol. 2013, 7, 379–391. [Google Scholar] [CrossRef] [PubMed]
- Guo, S.; Gonzalez-Perez, R.R. Notch, IL-1 and Leptin crosstalk outcome (NILCO) is critical for leptin-induced proliferation, migration and VEGF/VEGFR-2 expression in breast cancer. PLoS ONE 2011, 6, e21467. [Google Scholar] [CrossRef] [PubMed]
- Lipsey, C.C.; Harbuzariu, A.; Daley-Brown, D.; Gonzalez-Perez, R.R. Oncogenic role of leptin and Notch interleukin-1 leptin crosstalk outcome in cancer. World J. Methodol. 2016, 6, 43–55. [Google Scholar] [CrossRef] [PubMed]
- Feldman, D.E.; Chen, C.; Punj, V.; Tsukamoto, H.; Machida, K. Pluripotency factor-mediated expression of the leptin receptor (OB-R) links obesity to oncogenesis through tumor-initiating stem cells. Proc. Natl. Acad. Sci. USA 2012, 109, 829–834. [Google Scholar] [CrossRef] [PubMed]
- Zhou, J.; Wulfkuhle, J.; Zhang, H.; Gu, P.; Yang, Y.; Deng, J.; Margolick, J.B.; Liotta, L.A.; Petricoin, E.; Zhang, Y. Activation of the PTEN/mTOR/STAT3 pathway in breast cancer stem-like cells is required for viability and maintenance. Proc. Natl. Acad. Sci. USA 2007, 104, 16158–16163. [Google Scholar] [CrossRef] [PubMed]
- Pratt, M.A.; Tibbo, E.; Robertson, S.J.; Jansson, D.; Hurst, K.; Perez-Iratxeta, C.; Lau, R.; Niu, M.Y. The canonical NF-KappaB pathway is required for formation of luminal mammary neoplasias and is activated in the mammary progenitor population. Oncogene 2009, 28, 2710–2722. [Google Scholar] [CrossRef] [PubMed]
- Knight, B.B.; Oprea-Ilies, G.M.; Nagalingam, A.; Yang, L.; Cohen, C.; Saxena, N.K.; Sharma, D. Survivin upregulation, dependent on leptin-EGFR-Notch1 axis, is essential for leptin-induced migration of breast carcinoma cells. Endocr. Relat. Cancer 2011, 18, 413–428. [Google Scholar] [CrossRef] [PubMed]
- Guo, S.; Liu, M.; Wang, G.; Torroella-Kouri, M.; Gonzalez-Perez, R.R. Oncogenic role and therapeutic target of leptin signaling in breast cancer and cancer stem cells. Biochim. Biophys. Acta 2012, 1825, 207–222. [Google Scholar] [CrossRef] [PubMed]
- Wang, T.; Fahrmann, J.F.; Lee, H.; Li, Y.J.; Tripathi, S.C.; Yue, C.; Zhang, C.; Lifshitz, V.; Song, J.; Yuan, Y.; et al. JAK/STAT3-regulated fatty acid β-oxidation is critical for breast cancer stem cell self-renewal and chemoresistance. Cell MeTable 2018, 27, 1357. [Google Scholar] [CrossRef] [PubMed]
- Catalano, S.; Leggio, A.; Barone, I.; De Marco, R.; Gelsomino, L.; Campana, A.; Malivindi, R.; Panza, S.; Giordano, C.; Liguori, A.; et al. A novel leptin antagonist peptide inhibits breast cancer growth in vitro and in vivo. J. Cell. Mol. Med. 2015, 19, 1122–1132. [Google Scholar] [CrossRef] [PubMed]

| Clinicaltrials.gov | Interventions | Status | Eligible Criteria, Primary Outcome and Purpose | Results |
|---|---|---|---|---|
| NCT01758146 | Drug: Tamoxifen Drug: Letrozole | Phase 3 | Eligibility criteria: Postmenopausal patients (45–80 years) with hormone receptor positive breast cancer (tumor stage IB, IC, or II irrespective of nodal stage (<10 positive nodes)). Primary outcome: Disease free survival (time frame: from date of random assignment to the first event over 5 years). Event in the form of locoregional recurrence, distant metastasis, cancer in the contralateral breast, second primary cancer, or death from any cause. Purpose: To see the impact of obesity on the efficacy of adjuvant endocrine therapy with aromatase inhibitors in postmenopausal patients with early breast cancer in terms of: (1) Locoregional recurrence; (2) Distant metastases; (3) Disease-free survival; (4) Overall survival. | Not applicable |
| NCT00470119 | Behavioral: Behavioral dietary intervention Behavioral: Exercise intervention | Not applicable | Eligibility criteria: Postmenopausal patients (50–75 years) with no history of invasive or “in situ” breast cancer and at increased risk for developing breast cancer due to several lifestyle risk factors (e.g., lack of physical activity, excess weight, obesity, etc.). Primary outcome: Serum estrone concentrations as measured by radioimmunoassay (time frame: at baseline and 12 months) Purpose: To compare the effects of a 1-year exercise intervention, reduced-calorie diet intervention, or a combined exercise and reduced-calorie diet intervention vs. no intervention on serum estrone concentrations in overweight or obese postmenopausal women. | [17,18,19,20,21,22,23] |
| NCT01172886 | Not applicable | Unknown | Eligibility criteria: Healthy women and women operated for breast cancer (35–80 years). Primary outcome: Biospecimen retention: Samples without DNA. Blood samples. Purpose: To evaluate if the adjustments in lifestyle with physical activity intensification and healthy diet may represent modifiable factors on which sporadic breast cancer primary prevention may work. | Not applicable |
| NCT02750826 | Other: Health education program Other: Weight loss intervention | Phase 3 | Eligibility criteria: Subjects (≥18 years) with histologically confirmed invasive breast cancer and with registration within 12 months after the first histologic diagnosis of the disease.Primary outcome: Invasive disease-free survival (time frame: up to 10 years).Purpose: To compare the effect of a supervised weight loss intervention plus health education materials versus health education materials alone on invasive disease-free survival (IDFS) in overweight (body mass index, BMI 27–29.9 kg/m2) and obese (BMI ≥30 kg/m2) women diagnosed with HER-2 (Human epidermal growth factor receptor 2) negative, stage II, and III breast cancer. | Not applicable |
| NCT01112839 | Behavioral: Less intensive Behavioral: Intensive group | Completed | Eligibility criteria: Patients (≥21 years) with breast cancer (stages I (≥1 cm), stage II, or stage IIIA, B, C excluding distant metastasis) diagnosed between 6 months and 5 years earlier; after initial therapies were completed. BMI between 25 and 45 kg/m2. Primary outcome: Weight loss (time frame: 2 years). Purpose: To explore whether two different programs that are focused on weight management, through increased exercise and a healthy diet, are feasible, and have an impact on body weight, quality of life and fatigue. | [24] |
| NCT02618538 | Not applicable | Enrolling by invitation | Eligibility criteria: All women (46–67 years) undergoing a screening mammography in the participating centers (Turin, Vercelli, and Biella). Primary outcome: Positive predictive value (PPV) of breast cancer risk factors (time frame: 2 years). Purpose: To estimate in a large cohort of women attending breast cancer screening, the predictive positive values of different breast cancer risk factors (alone or in combination) in order to identify appropriate risk-based stratifications for personalized screening. | [25] |
| NCT00393172 | Behavioral: exercise | Phase 2 Phase 3 | Eligibility criteria: Premenopausal healthy women (18–30 years). Primary outcome: Changes in urine levels of F2-isoprostanes (time frame: before and after study). Purpose: To compare the changes in urine levels of a stable marker of oxidative stress (F_2-isoprostanes) in healthy young women who undergo exercise training during 4 menstrual cycles vs. no exercise. | [26,27,28,29] |
| NCT00509626 | Behavioral: exercise intervention Other: questionnaire administration Procedure: CAM (Complementary and Alternative Medicine) exercise therapy Procedure: management of therapy complications Procedure: psychosocial assessment and care Procedure: quality-of-life assessment | Phase 1 Phase 2 | Eligibility criteria: Premenopausal and postmenopausal patients (≥ 18 years) with diagnosis of primary breast cancer and BMI ≥20 kg/m2 and <39 kg/m2. Primary outcome: Accrual (phase I); Retention (phase I); Weight change after 6 months (phase II). Purpose: To determine whether: (1) conducting a clinical trial that uses a 6-month physical activity intervention initiated within 45 days after surgery for early-stage breast cancer and prior to initiation of adjuvant chemotherapy, hormonal therapy, and/or radiotherapy is feasible (phase I); (2) the participation in a 6-month physical activity intervention initiated within 45 days after surgery for early-stage breast cancer decreases weight gain in patients treated with adjuvant chemotherapy and differentially affects weight change in women who are premenopausal compared to those who are postmenopausal at diagnosis (phase II). | Not applicable |
| NCT03529383 | Device: Connected device Behavioral: Therapeutic education | Not applicable | Eligibility criteria: Patients (18–75 years) operated for a first primary non-metastatic invasive breast carcinoma, with the prescription of an adjuvant treatment (chemotherapy, hormonotherapy, radiotherapy). Primary outcome: Number of patients achieving the internationally recommended level of physical activity of at least 150 min per week of moderate-to-vigorous physical activity [intensity ≥3 METs (Metabolic equivalent of task)] (time frame: 6 months). Assessment by the RPAQ self-administered questionnaire. Purpose: To identify best modalities to implement exercise during and after breast cancer treatment. In a public health perspective, the challenge is to reduce geographical, social, and organizational inequalities among patients to practice regular physical activity and to promote the systematic integration of physical activity in routine cancer care. | [10,13,30,31,32,33,34,35,36,37] |
| NCT01707121 | Not applicable | Completed | Eligibility criteria: Patients (≥18 years) scheduled for cholecystectomy, breast cancer surgery, colorectal cancer surgery. Primary outcome: Length of sick-leave/time to work (time frame: 6 weeks) for breast cancer and gallbladder surgery. Length of hospital stay (time frame: 6 weeks] for colorectal surgery. Purpose: To investigate whether a higher physical activity prior to a surgical procedure reduces hospital stay, sick leave, and the complication rate. | [38] |
| NCT02273206 | Behavioral: Preventive care management for depression Behavioral: Preventive care management for cancer screening | Not applicable | Eligibility criteria: Patients (50–64 years) from the Bronx with no cancer diagnosis within the past six months and overdue for breast, cervical, or colorectal cancer screening. Primary outcome: Changes from baseline in number of participants with colorectal, breast, and/or cervical cancer screening (time frame: 12 months).Chart Review: Review of electronic medical records at each of the three community health centers.Self-Report: We will ask participants about their participation (yes/no) in specific screening methods: Pap testing (past 3 years), mammography (past 2 years), and colorectal screening (FOBT (fecal occult blood test)/FIT (fecal immunochemical test), past year; flexible sigmoidoscopy, the past 5 years; and colonoscopy, past 10 years). Change from baseline in Patient Health Questionnaire (PHQ)-9 at 6 months (time frame: 6 months). The PHQ-9 is the 9-item depression module from the full PHQ, and is a well-validated Diagnostic and Statistical Manual of Mental Disorders 4th Edition (DSM-IV) criterion-based measure for screening and diagnosing depressive episode, assessing severity, and monitoring treatment response, either in person or over the phone.Change from baseline in Patient Health Questionnaire (PHQ)-9 at 12 months (time frame: 12 months). The PHQ-9 is the 9-item depression module from the full PHQ, and is a well-validated Diagnostic and Statistical Manual of Mental Disorders 4th Edition (DSM-IV) criterion-based measure for screening and diagnosing depressive episodes, assessing severity, and monitoring treatment response, either in person or over the phone.Change from baseline in The Hopkins Symptom Checklist (SCL-20) at 6 months (time frame: 6 months). The SCL-20 consists of 20 depression items on a 4-point scale from the SCL-90, and has been shown to be a valid and reliable measure of depression in diverse outpatient and community populations.Change from baseline in The Hopkins Symptom Checklist (SCL-20) at 12 months (time frame: 12 months). The SCL-20 consists of the 20 depression items on a 4-point scale from the SCL-90, and has been shown to be a valid and reliable measure of depression in diverse outpatient and community populations. Purpose: To increase the investigators understanding of how to enhance primary care systems’ ability to improve a range of outcomes related to cancer screening and depression among low-income minority women, and how to best support this population in making cancer-screening decisions. | Not applicable |
| NCT01515124 | Behavioral: Exercise intervention Behavioral: Weight loss intervention | Not applicable | Eligibility criteria: Overweight or obese child, adult, or older adult female breast cancer survivor, at least 2 months after treatment (e.g., surgery, chemotherapy, or radiotherapy), currently free of cancer. Primary outcome: Clinical lymphedema exacerbation rate (time frame: Baseline, 6, and 12 months). Lymphedema outcomes include: incident events requiring medical care for lymphedema (e.g., flare-ups or cellulitic infections), arm swelling in the affected limb (interlimb volume differences), and pain and lymphedema symptoms (number and severity). Purpose: To test the effects of these interventions on lymphedema outcomes, breast cancer recurrence, and quality of life. | [39] |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Andò, S.; Gelsomino, L.; Panza, S.; Giordano, C.; Bonofiglio, D.; Barone, I.; Catalano, S. Obesity, Leptin and Breast Cancer: Epidemiological Evidence and Proposed Mechanisms. Cancers 2019, 11, 62. https://doi.org/10.3390/cancers11010062
Andò S, Gelsomino L, Panza S, Giordano C, Bonofiglio D, Barone I, Catalano S. Obesity, Leptin and Breast Cancer: Epidemiological Evidence and Proposed Mechanisms. Cancers. 2019; 11(1):62. https://doi.org/10.3390/cancers11010062
Chicago/Turabian StyleAndò, Sebastiano, Luca Gelsomino, Salvatore Panza, Cinzia Giordano, Daniela Bonofiglio, Ines Barone, and Stefania Catalano. 2019. "Obesity, Leptin and Breast Cancer: Epidemiological Evidence and Proposed Mechanisms" Cancers 11, no. 1: 62. https://doi.org/10.3390/cancers11010062
APA StyleAndò, S., Gelsomino, L., Panza, S., Giordano, C., Bonofiglio, D., Barone, I., & Catalano, S. (2019). Obesity, Leptin and Breast Cancer: Epidemiological Evidence and Proposed Mechanisms. Cancers, 11(1), 62. https://doi.org/10.3390/cancers11010062









