Journal Description
Cancers
Cancers
is a peer-reviewed, open access journal of oncology published semimonthly online. The North-East German Society for Gynecological Oncology (NOGGO), Irish Association for Cancer Research (IACR), Spanish Association for Cancer Research (ASEICA), Biomedical Research Centre (CIBM), British Neuro-Oncology Society (BNOS) and more are affiliated with Cancers and their members receive a discount on the article processing charges.
- Open Access— free for readers, with article processing charges (APC) paid by authors or their institutions.
- High Visibility: indexed within Scopus, SCIE (Web of Science), PubMed, PMC, Embase, CAPlus / SciFinder, and other databases.
- Journal Rank: JCR - Q2 (Oncology) / CiteScore - Q1 (Oncology)
- Rapid Publication: manuscripts are peer-reviewed and a first decision is provided to authors approximately 19.1 days after submission; acceptance to publication is undertaken in 2.6 days (median values for papers published in this journal in the second half of 2025).
- Recognition of Reviewers: reviewers who provide timely, thorough peer-review reports receive vouchers entitling them to a discount on the APC of their next publication in any MDPI journal, in appreciation of the work done.
- Sections: published in 18 topical sections.
- Companion journals for Cancers include: Radiation and Onco.
- Journal Clusters of Oncology: Cancers, Current Oncology, Onco and Targets.
Impact Factor:
4.4 (2024);
5-Year Impact Factor:
4.8 (2024)
Latest Articles
The Spectrum of Venetoclax-Based Treatments in Acute Myeloid Leukemia
Cancers 2026, 18(8), 1201; https://doi.org/10.3390/cancers18081201 (registering DOI) - 9 Apr 2026
Abstract
Background/Objectives: In recent years there has been a consistent development of clinical studies surrounding the incorporation of the B-cell lymphoma 2 (BCL-2) inhibitor venetoclax (VEN) into the treatment of acute myeloid leukemia (AML) Methods: A search of the literature showed a
[...] Read more.
Background/Objectives: In recent years there has been a consistent development of clinical studies surrounding the incorporation of the B-cell lymphoma 2 (BCL-2) inhibitor venetoclax (VEN) into the treatment of acute myeloid leukemia (AML) Methods: A search of the literature showed a tremendous development of experimental and clinical studies evaluating the impact of VEN-based regimens in the treatment of AML patients. This review comprehensively analyzes the available scientific evidence—including prospective clinical trials, retrospective cohorts, and real-world studies—to summarize current knowledge on the efficacy and safety of venetoclax-based regimens in AML patients. Results: Recent studies have evaluated VEN-based regimens in newly diagnosed (ND) and refractory/relapsed (R/R) AML patients, showing the efficacy of these treatments. VEN with hypomethylating agents (HMAs) became the standard-of-care for elderly/unfit AML patients. Recent studies strongly support the effectiveness of VEN-based regimens in frontline treatment of adult AML patients eligible for intensive treatments. VEN-based therapies were also used in combination with targeted therapies, thus generating triplet therapeutic regimens that are under evaluation for the treatment of some AML subtypes. However, the response to VEN+HMAs is highly variable and in part depends on tumor genetics; some patients are resistant or relapse following VEN-based treatments and future studies will be required to develop therapeutic strategies able to circumvent resistance and to identify patients at high risk of relapse. Prospective randomized trials are required to establish the real efficacy of VEN in various clinical settings and to refine maintenance and discontinuation strategies, aiming to improve long-term outcomes and to make more safe treatments based on VEN.
Full article
(This article belongs to the Special Issue Precision Medicine in Acute Myeloid Leukemia)
Open AccessArticle
Total Neoadjuvant Therapy Outcomes and Watch-and-Wait Feasibility in Locally Advanced Rectal Cancer: A Single-Institution Retrospective Cohort Study
by
Manuel Ramanović, Franc Anderluh, Ana Jeromen Peressutti, Petar Korošec, Irena Oblak, Ajra Šečerov Ermenc and Vaneja Velenik
Cancers 2026, 18(8), 1200; https://doi.org/10.3390/cancers18081200 (registering DOI) - 9 Apr 2026
Abstract
Background/Objectives: Total neoadjuvant therapy (TNT), integrating systemic chemotherapy and radiotherapy before surgery or surveillance, has become a standard approach for locally advanced rectal cancer (LARC). However, optimal sequencing strategies and long-term outcomes of watch-and-wait (W&W) following sandwich TNT remain insufficiently characterized. We
[...] Read more.
Background/Objectives: Total neoadjuvant therapy (TNT), integrating systemic chemotherapy and radiotherapy before surgery or surveillance, has become a standard approach for locally advanced rectal cancer (LARC). However, optimal sequencing strategies and long-term outcomes of watch-and-wait (W&W) following sandwich TNT remain insufficiently characterized. We evaluated oncologic outcomes and treatment response in patients treated with an institutional sandwich TNT protocol. Methods: We conducted a retrospective cohort study of consecutive patients with LARC treated with sandwich TNT (induction chemotherapy followed by hypofractionated intensity-modulated radiotherapy with simultaneous integrated boost [IMRT-SIB] chemoradiotherapy and consolidation chemotherapy) at the Institute of Oncology Ljubljana between 2016 and 2023. The primary endpoint was an overall complete response (CR; pathological [pCR] and clinical [cCR]). Secondary endpoints included tumor regression grade (TRG), major pathological response (MPR), R0 resection rate, organ preservation, overall survival (OS), and disease-free survival (DFS). Results: Among 205 patients (median age 61 years), overall CR was 29.5% (pCR 19.3% and cCR 10.2%). Major pathological response (TRG 3–4) occurred in 37.6%. R0 resection was achieved in 94.5%. In the W&W cohort (n = 21), local regrowth occurred in 33.3% (95% CI, 14.6–57.0%) over a median follow-up of 4.96 years. Total mesorectal excision (TME)-free survival at 5 years was 73.1% (95% CI, 55.0–97.2%). Estimated 5-year OS was 81.1% (95% CI, 75.5–87.2%) and 5-year DFS was 75.2% (95% CI, 69.0–82.0). In multivariable analysis, non-R0 resection (HR 6.06, 95% CI, 1.99–18.42), MRI circumferential resection margin positivity (HR 3.11, 95% CI, 1.53–6.33), and MRI extramural vascular invasion positivity (HR 1.97, 95% CI, 1.05–3.91) remained independent predictors of DFS. Conclusions: Institutional sandwich TNT yields meaningful tumor response and durable survival in MRI-defined high-risk LARC. Structured W&W offers organ preservation with acceptable oncologic control under intensive surveillance.
Full article
(This article belongs to the Section Cancer Survivorship and Quality of Life)
►▼
Show Figures
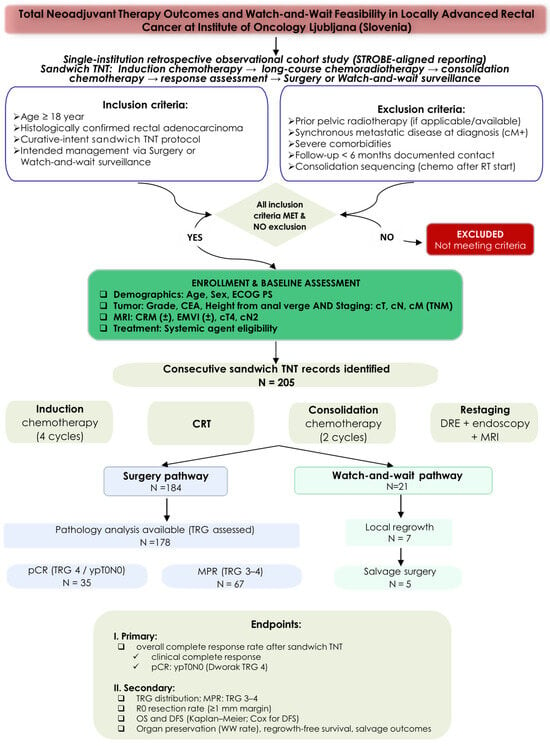
Figure 1
Open AccessArticle
Impact of Postoperative Liver Injury on the Oncological Short- and Long-Term Outcome After Liver Resection for Hepatocellular Carcinoma
by
Katharina Lang, Oliver Beetz, Iakovos Amygdalos, Clara A. Weigle, Bengt A. Wiemann, Julian Palzer, Sebastian Cammann, Georg Wiltberger, Thomas Vogel, Florian W. R. Vondran, Franziska A. Meister and Felix Oldhafer
Cancers 2026, 18(8), 1199; https://doi.org/10.3390/cancers18081199 - 9 Apr 2026
Abstract
Background: Postoperative liver injury remains a major challenge after curative-intended liver resection for hepatocellular carcinoma (HCC), impacting both early complications and long-term survival. The recently introduced SAAR (Sum of AST/ALT Ratios) score may serve as a novel marker for predicting postoperative outcomes. Methods:
[...] Read more.
Background: Postoperative liver injury remains a major challenge after curative-intended liver resection for hepatocellular carcinoma (HCC), impacting both early complications and long-term survival. The recently introduced SAAR (Sum of AST/ALT Ratios) score may serve as a novel marker for predicting postoperative outcomes. Methods: This retrospective single-center study included 213 patients undergoing liver resection for HCC between January 2007 and October 2024. The SAAR score was calculated using AST/ALT ratios on postoperative days 1 and 3 and correlated with post-hepatectomy liver failure (PHLF), disease-free survival (DFS), and overall survival (OS). The predictive performance of the SAAR score was compared with that of the combined APRI + ALBI score. Results: Patients with SAAR ≥ 2 showed significantly higher rates of PHLF (OR = 2.5, p = 0.019) and impaired long-term outcomes. Median DFS and OS were significantly reduced in the SAAR ≥ 2 group (7 vs. 24 months, p < 0.001; 12 vs. 32 months, p = 0.004, respectively). Multivariate Cox regression confirmed SAAR ≥ 2 as an independent predictor for DFS (HR = 2.1) and OS (HR = 1.9). The APRI + ALBI score also demonstrated strong preoperative predictive value for PHLF (AUC = 0.854) but not for long-term outcome.
Full article
(This article belongs to the Special Issue Advancements in Surgical and Multidisciplinary Management of Hepatopancreaticobiliary Cancers)
►▼
Show Figures
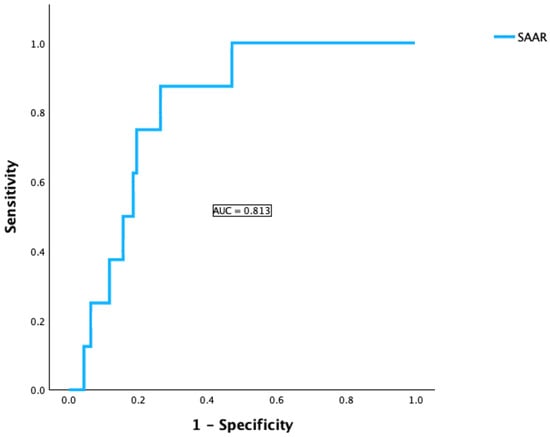
Figure 1
Open AccessArticle
Perioperative Blood Transfusion Impairs Overall Survival Following Radical Resection for Colorectal Cancer: A Propensity Score-Matched Analysis
by
Xiaoran Wang, Zesong Meng, Guangjun Wang, Guiying Wang and Lihua Liu
Cancers 2026, 18(8), 1198; https://doi.org/10.3390/cancers18081198 (registering DOI) - 9 Apr 2026
Abstract
Background/Objectives: Perioperative blood transfusion (BTF) remains controversial regarding its impact on oncological outcomes in colorectal cancer (CRC). This study aimed to evaluate the association between BTF, transfusion volume, and long-term prognosis in CRC patients undergoing radical resection. Methods: We conducted a retrospective cohort
[...] Read more.
Background/Objectives: Perioperative blood transfusion (BTF) remains controversial regarding its impact on oncological outcomes in colorectal cancer (CRC). This study aimed to evaluate the association between BTF, transfusion volume, and long-term prognosis in CRC patients undergoing radical resection. Methods: We conducted a retrospective cohort study of 1777 CRC patients who underwent radical surgery at the Fourth Hospital of Hebei Medical University between December 2007 and April 2015. Propensity score matching (PSM) was applied to minimize selection bias between BTF and non-BTF groups. Logistic regression identified factors associated with BTF receipt. Cox proportional hazards models assessed the association between BTF and 5-year overall survival (OS). X-tile analysis determined optimal cut-off values for transfusion volume stratification. Results: Among 1777 patients, 729 (41.02%) received BTF. After PSM, 524 well-matched pairs showed balanced baseline characteristics. Intestinal obstruction was independently associated with BTF requirement (p < 0.001). The BTF group demonstrated significantly inferior 1-, 3-, and 5-year OS compared with non-BTF groups in both overall and PSM cohorts (all p < 0.01). Multivariate Cox analysis identified BTF as an independent adverse prognostic factor (HR = 1.44, 95% CI 1.09–1.89, p = 0.01). X-tile analysis stratified patients into non-BTF, small-volume (≤4 units), and massive-volume (>4 units) groups. Massive-volume transfusion showed the poorest survival outcomes (p < 0.0001) and was independently associated with worse OS (HR = 1.61, 95% CI 1.18–2.20, p = 0.003). Supplementary analyses indicated that no independent association was observed between survival outcomes and either preoperative inflammatory markers or the specific timing and type of transfusion. Conclusions: BTF, particularly when exceeding 4 units, independently predicts inferior long-term survival in CRC patients following radical resection. These findings support the implementation of restrictive transfusion strategies in perioperative CRC management. Given its retrospective observational design, this study shows an association but does not establish causality, and our findings should be interpreted in light of the aforementioned limitations.
Full article
(This article belongs to the Section Clinical Research of Cancer)
►▼
Show Figures
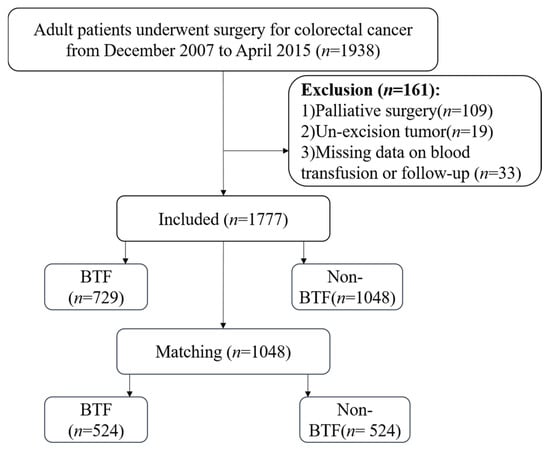
Figure 1
Open AccessArticle
A Multigene Signature for Prognostic Stratification of Nasopharyngeal Carcinoma
by
Yingying Liang, Zhiwen Mo and Muy-Teck Teh
Cancers 2026, 18(8), 1197; https://doi.org/10.3390/cancers18081197 - 9 Apr 2026
Abstract
Background: We previously pioneered a multigene mRNA test, qMIDSV2, validated through an international multicohort study with geographically and ethnically diverse oral squamous cell carcinoma (OSCC) patients from Europe and Asia. This study aimed to repurpose the qMIDSV2 test for
[...] Read more.
Background: We previously pioneered a multigene mRNA test, qMIDSV2, validated through an international multicohort study with geographically and ethnically diverse oral squamous cell carcinoma (OSCC) patients from Europe and Asia. This study aimed to repurpose the qMIDSV2 test for nasopharyngeal carcinoma (NPC). A molecular test independent of Epstein–Barr virus (EBV) status would be clinically useful for risk stratification in NPC patients with undetectable or low levels of EBV. Methods: This study investigated a Chinese cohort of 62 participants (18 donated normal nasopharyngeal mucosa (NPM) and 44 donated NPC tissue samples). Messenger RNA levels of 16 genes in each sample were quantified using the qPCR method, and an algorithm computed a malignancy index for cancer risk stratification. Results: We identified a unique 10-gene panel (containing eight target genes, namely NEK2, INHBA, FOXM1, TOP2A, BIRC5, CXCL8, NR3C1, and IVL, relative to two reference genes, YAP1 and POLR2A, collectively named qMIDSNPC) that demonstrated the best overall diagnostic performance in segregating NPM from NPC, with AUC = 0.909 and positive/negative predictive values of 91% PPV and 78% NPV, respectively. Furthermore, we demonstrated prognostic value of qMIDSNPC in segregating NPM from NPC stage III + IV, with AUC = 0.936, 92% PPV, and 84% NPV. Conclusions: Here, we present a simple qPCR-based 10-gene mRNA test, qMIDSNPC, with potential clinical utilities for rapid (1 h) prognostic stratification of NPC. Further studies involving geographically and ethnically independent NPC cohorts would be needed to validate the clinical use of qMIDSNPC in non-endemic NPC populations.
Full article
(This article belongs to the Section Cancer Biomarkers)
►▼
Show Figures
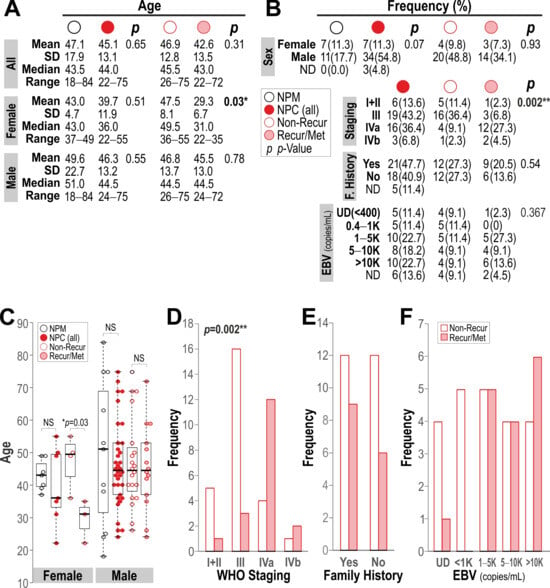
Figure 1
Open AccessReview
Triptolide: A Narrative Review of Its Traditional Use, Derivatives, Pharmacology, Antitumor Effect, and Clinical Applications
by
Yibo Geng, Bettina Kritzer and Javad Nazarian
Cancers 2026, 18(8), 1196; https://doi.org/10.3390/cancers18081196 - 9 Apr 2026
Abstract
Triptolide (TPL), a diterpenoid derived from the Chinese medicinal plant Tripterygium wilfordii, exhibits broad-spectrum biological and pharmacological activities, although its clinical translation is limited by systemic toxicity. Recent advances in the development of TPL derivatives have created new therapeutic opportunities. This review
[...] Read more.
Triptolide (TPL), a diterpenoid derived from the Chinese medicinal plant Tripterygium wilfordii, exhibits broad-spectrum biological and pharmacological activities, although its clinical translation is limited by systemic toxicity. Recent advances in the development of TPL derivatives have created new therapeutic opportunities. This review summarizes current knowledge of triptolide, with a focus on TPL’s toxicity profile, derivative strategies, and antitumor mechanisms across different tumor types, including glioma, pancreatic tumor, leukemia, lung cancer, gastric cancer and others. We also summarize the plant’s origin and traditional uses, TPL’s pharmacokinetics (PKs), and relevant clinical trials against tumors. The main mechanism of the TPL antitumor effect is to interfere with ATPase of XPB by covalently binding to it, as well as inducing the rapid depletion of RPB1 via hyperphosphorylation and ubiquitination. We also reviewed systemic toxicity including neuro-, cardio-, oto-, nephron-, hepato-, and hemato-toxicity, as well as digestive and reproductive toxicity. Finally, we searched clinical trial databases across three platforms for tumors and concluded that Minnelide has strong clinical potential for solid tumors. By critically evaluating TPL from multiple dimensions, specifically its traditional use, chemical derivatization, pharmacokinetics, antitumor mechanisms, toxicity, and clinical trials, this review aims to inform future strategies that maximize therapeutic efficacy while minimizing adverse effects.
Full article
(This article belongs to the Special Issue Preclinical and Clinical Research on the Efficacy of Anticancer Drugs)
►▼
Show Figures
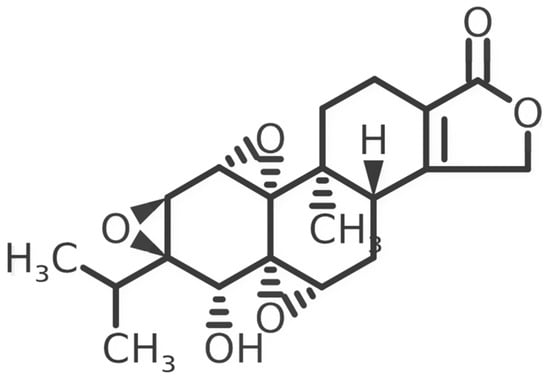
Figure 1
Open AccessArticle
Do All Stage IA Pancreatic Cancer Patients Need Adjuvant Chemotherapy?
by
John M. Lyons III, Mei-Chin Hsieh, Kenneth C. Avanzino, Mohammad Al Efishat and Quyen Chu
Cancers 2026, 18(8), 1195; https://doi.org/10.3390/cancers18081195 - 8 Apr 2026
Abstract
Background: National guidelines recommend adjuvant chemotherapy (AC) following resection for all stages of pancreatic cancer (PDAC), but the benefit of AC in Stage IA disease remains unclear. The objective of this study was to identify a subgroup of patients with Stage IA PDAC
[...] Read more.
Background: National guidelines recommend adjuvant chemotherapy (AC) following resection for all stages of pancreatic cancer (PDAC), but the benefit of AC in Stage IA disease remains unclear. The objective of this study was to identify a subgroup of patients with Stage IA PDAC that could possibly forego AC. Study Design: The National Cancer Database (NCDB) was queried to identify all patients with Stage IA PDAC diagnosed from 2010 to 2021. Patients who received AC were compared to those who did not. Multivariable analysis was conducted to identify risk factors associated with overall survival (OS). Results: There were 1421 patients eligible for analysis. On multivariable analysis, we found nine factors associated with worse overall OS: advanced age (p = 0.0414), lower median income (p = 0.0148), Medicare (p = 0.0180), higher-grade tumor histology (p = 0.0182), LVI (p = 0.0028), positive surgical margins (p = 0.0027), examination of fewer than 12 lymph nodes (p = 0.0395), and a length of stay greater than 7 days (p < 0.0001). OS was negatively correlated with an increased number of risk factors. Improved OS was observed following AC in patients with three (∆OS = +54 months; p = 0.0016) or four or more risk factors (∆OS = +11.4 months; p = 0.0250). However, patients with fewer than three risk factors did not experience improvement in OS following AC. Conclusions: AC does not appear to benefit Stage IA PDAC patients with fewer than three risk factors indicating that it may be safe to omit AC in these individuals.
Full article
(This article belongs to the Special Issue Novel Approaches to the Management of Patients with Gastrointestinal Tumors)
►▼
Show Figures
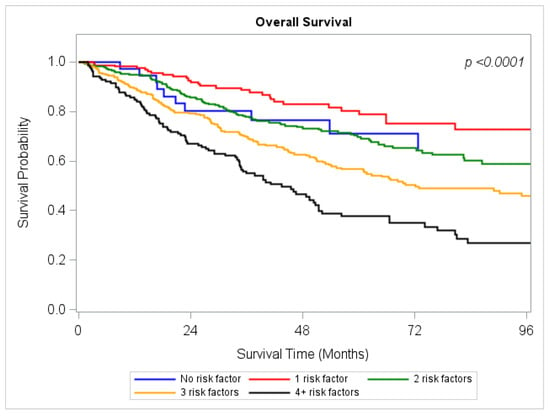
Figure 1
Open AccessArticle
Multimodal Deep Learning for Prediction of Progression-Free Survival in Patients with Neuroendocrine Tumors Undergoing 177Lu-Based Peptide Receptor Radionuclide Therapy
by
Simon Baur, Tristan Ruhwedel, Ekin Böke, Zuzanna Kobus, Gergana Lishkova, Christoph Wetz, Holger Amthauer, Christoph Roderburg, Frank Tacke, Julian M. Rogasch, Wojciech Samek, Henning Jann, Jackie Ma and Johannes Eschrich
Cancers 2026, 18(8), 1194; https://doi.org/10.3390/cancers18081194 - 8 Apr 2026
Abstract
Background/Objectives: Peptide receptor radionuclide therapy (PRRT) is an established treatment for metastatic neuroendocrine tumors (NETs), yet long-term disease control occurs only in a subset of patients. Predicting progression-free survival (PFS) could support individualized treatment planning. This study evaluates laboratory, imaging, and multimodal
[...] Read more.
Background/Objectives: Peptide receptor radionuclide therapy (PRRT) is an established treatment for metastatic neuroendocrine tumors (NETs), yet long-term disease control occurs only in a subset of patients. Predicting progression-free survival (PFS) could support individualized treatment planning. This study evaluates laboratory, imaging, and multimodal deep learning models for PFS prediction in PRRT-treated patients. Methods: In this retrospective, single-center study 116 patients with metastatic NETs undergoing [177Lu]Lu-DOTATOC were included. Clinical characteristics, laboratory values, and pretherapeutic somatostatin receptor positron emission tomography/computed tomographies (SR-PET/CTs) were collected. Seven models were trained to classify low- vs. high-PFS groups, including unimodal (laboratory, SR-PET, or CT) and multimodal fusion approaches. Performance was assessed via repeated 3-fold cross-validation with area under the receiver operating characteristic curve (AUROC) and area under the precision–recall curve (AUPRC). Explainability was evaluated by feature importance analysis and gradient based saliency maps. Results: Forty-two patients (36%) displayed short PFS (≤1 year) and 74 patients displayed long PFS (>1 year). Groups were similar in most characteristics, except for higher baseline chromogranin A (p = 0.003), elevated
(This article belongs to the Special Issue Advancing Therapeutic Strategies for Neuroendocrine Tumors: Towards Personalized and Multidisciplinary Care)
►▼
Show Figures

Figure 1
Open AccessReview
Tumor Microenvironment Acidosis and Alkalization-Oriented Interventions in Advanced Solid Tumors: A Narrative Review and Science-Based Medicine Perspective on Long-Tail Survival
by
Kazuyuki Suzuki, Shion Kachi and Hiromi Wada
Cancers 2026, 18(8), 1193; https://doi.org/10.3390/cancers18081193 - 8 Apr 2026
Abstract
Median overall survival remains a central endpoint in oncology, but it can obscure a clinically meaningful long tail of patients with advanced solid tumors who survive well beyond the median. One biological context in which this pattern may be relevant is tumor microenvironment
[...] Read more.
Median overall survival remains a central endpoint in oncology, but it can obscure a clinically meaningful long tail of patients with advanced solid tumors who survive well beyond the median. One biological context in which this pattern may be relevant is tumor microenvironment (TME) acidosis. Driven by aerobic glycolysis, hypoxia, impaired perfusion, and proton-export programs, acidic TME is increasingly implicated in invasion, therapeutic resistance, and immune suppression. This narrative review examines TME acidosis as the primary biological framework and considers long-tail survival as a clinical lens through which its implications may be interpreted. We summarize the biological basis and heterogeneity of acidic TME, review current approaches to clinical and translational assessment of tumor acidity, including acidoCEST magnetic resonance imaging (MRI) and positron emission tomography (PET)-based approaches, and discuss the potential and limitations of alkalization-oriented interventions such as buffering and diet-based strategies. Particular attention is given to the distinction between direct measurements of tumor acidity and clinically feasible but indirect markers such as urinary pH, which should not be interpreted as a direct surrogate for local tumor extracellular pH. From a science-based medicine perspective, long-tail survival is treated here as a hypothesis-generating clinical signal rather than proof of causality. Overall, alkalization-oriented interventions appear biologically plausible and clinically testable, but current clinical evidence remains limited and context-dependent. Future progress will require mechanistically informed biomarkers, careful safety evaluation, and trial designs capable of detecting delayed separation of survival curves and tail-oriented patterns of benefit.
Full article
(This article belongs to the Special Issue Modulating the Acidic Tumor Microenvironment to Enhance the Efficacy and Reduce the Toxicity of Cancer Treatment)
►▼
Show Figures

Figure 1
Open AccessArticle
Perioperative Outcomes of Neoadjuvant Immunochemotherapy for Locally Resectable Oesophageal Squamous Cell Carcinoma in Geriatric Patients Aged 70 Years or Older
by
Qi Li, Song Lu, Yi Wang, Guangyuan Liu and Zhenjun Liu
Cancers 2026, 18(8), 1192; https://doi.org/10.3390/cancers18081192 - 8 Apr 2026
Abstract
Background: Neoadjuvant chemoradiotherapy (nCRT) followed by surgery has become the standard treatment for oesophageal cancer. However, data on the outcomes of neoadjuvant immunochemotherapy (nICT) in geriatric patients (≥70 years) who face higher perioperative risks are limited. Objective: This study aimed to compare the
[...] Read more.
Background: Neoadjuvant chemoradiotherapy (nCRT) followed by surgery has become the standard treatment for oesophageal cancer. However, data on the outcomes of neoadjuvant immunochemotherapy (nICT) in geriatric patients (≥70 years) who face higher perioperative risks are limited. Objective: This study aimed to compare the perioperative outcomes of nICT versus nCRT in elderly patients with locally advanced oesophageal squamous cell carcinoma (ESCC). Method: This retrospective cohort study included 132 geriatric patients (median age: 72 years) treated with nICT (n = 51) or nCRT (n = 81) followed by esophagectomy at Sichuan Cancer Hospital (2021–2024). Intraoperative outcomes, postoperative pathologic stages, and complications, including pneumonia and anastomotic leakage, were assessed. Propensity score matching (PSM), overlap weighting (OW), and inverse probability of treatment weighting (IPTW) were used to adjust for baseline covariate imbalances in the sensitivity analysis. Results: Pathologic ypT0 stage tended to be higher in the nCRT group (p = 0.014), whereas ypN0 was higher in the nICT group (p = 0.035). No significant differences in intraoperative or postoperative outcomes between the two groups, except for pulmonary complications (p > 0.05). Compared with nCRT patients, nICT patients had significantly lower pulmonary complication rates (13.7% vs. 32.1%, p = 0.030), and multivariable analysis confirmed these findings (adjusted OR = 0.26; 95% CI: 0.08–0.85; p = 0.026). Sensitivity analyses showed consistent results. Conclusions: The safety of nICT is comparable to that of nCRT in geriatric ESCC patients, with significantly fewer pulmonary complications. These findings support nICT as a valuable alternative for elderly populations.
Full article
(This article belongs to the Section Cancer Therapy)
►▼
Show Figures

Figure 1
Open AccessArticle
Efficacy and Safety of Oral Progestogens (Megestrol Acetate and Medroxyprogesterone Acetate) in Heavily Pretreated Oestrogen Receptor-Positive Metastatic Breast Cancer: A 10-Year Multi-Site Study
by
Iseult M. Browne, Heng Chun Wong, Tazia Irfan, Chloe Chan, Stephen R. D. Johnston, Zoe Kemp, Emma Kipps, Marina Parton, Nicholas C. Turner and Alicia F. C. Okines
Cancers 2026, 18(8), 1191; https://doi.org/10.3390/cancers18081191 - 8 Apr 2026
Abstract
Background: Oral progestogens, including megestrol acetate (MA) and medroxyprogesterone acetate (MPA), have largely been superseded by aromatase inhibitors, tamoxifen, and selective oestrogen receptor degraders (SERDs) in oestrogen receptor-positive (ER-positive) metastatic breast cancer. However, they remain an option as late-line therapy after failure of
[...] Read more.
Background: Oral progestogens, including megestrol acetate (MA) and medroxyprogesterone acetate (MPA), have largely been superseded by aromatase inhibitors, tamoxifen, and selective oestrogen receptor degraders (SERDs) in oestrogen receptor-positive (ER-positive) metastatic breast cancer. However, they remain an option as late-line therapy after failure of standard treatments. Contemporary data are limited, particularly in patients previously treated with CDK4/6 inhibitors. Methods: We conducted a multi-site retrospective analysis of patients with ER-positive metastatic breast cancer treated with MA or MPA between 2014 and 2024 at four hospital sites across London, United Kingdom. Patients were identified using pharmacy dispensing records. Progression-free survival (PFS) and overall survival (OS) were estimated using the Kaplan–Meier method and Cox regression. Subgroup analyses included prior CDK4/6 inhibitor exposure, histology and liver metastases. Results: A total of 116 patients were included. Median PFS was 2.4 months (95% CI 2.2–2.9), and median OS was 3.3 months (95% CI 2.7–4.9). Prior CDK4/6 inhibitor exposure was associated with shorter PFS (1.9 vs. 2.8 months; HR 1.59; 95% CI 1.08–2.35, p = 0.019) and a trend toward shorter OS (3.1 vs. 3.6 months; HR 1.18, 95% CI 0.80–1.75, p = 0.41). Similarly, liver metastases were associated with shorter PFS (2.3 vs. 2.8 months; HR 1.78, 95% CI 1.12–2.85, p = 0.015), with a trend toward worse OS (3.1 vs. 4.9 months; HR 1.45, 95% CI 0.93–2.25, p = 0.103). A subset of patients derived prolonged benefit, with a 6-month PFS rate of 16%. Toxicity was manageable; thromboembolic events and oedema occurred in 9% and 11% of patients respectively. Appetite improvement was reported in 10%. Conclusions: MA and MPA demonstrated modest but clinically relevant late-line activity in heavily pretreated, endocrine-refractory ER-positive metastatic breast cancer. While prior exposure to CDK4/6 inhibitors was associated with shorter PFS, patients without liver metastases appeared to derive the greatest benefit. These findings support a role for oral progestogens in selected patients who have exhausted standard therapeutic options.
Full article
(This article belongs to the Special Issue Advances in Invasive Breast Cancer: Treatment and Prognosis (2nd Edition))
►▼
Show Figures

Figure 1
Open AccessArticle
Ensemble Machine Learning Predicts Platinum Resistance in Ovarian Cancer Using Laboratory Data
by
Xueting Peng, Yangyang Zhang, Chaoyu Zhu, Weijie Chen, Xiaohua Wu, Fan Zhong, Qinhao Guo and Lei Liu
Cancers 2026, 18(8), 1190; https://doi.org/10.3390/cancers18081190 - 8 Apr 2026
Abstract
Objectives: Platinum resistance remains a critical bottleneck in ovarian cancer management, yet reliable pre-treatment predictive tools are lacking. Existing markers like the platinum-free interval are retrospective, while genomic profiling is often cost-prohibitive. This study aimed to develop an accessible, machine learning-based dynamic weighted
[...] Read more.
Objectives: Platinum resistance remains a critical bottleneck in ovarian cancer management, yet reliable pre-treatment predictive tools are lacking. Existing markers like the platinum-free interval are retrospective, while genomic profiling is often cost-prohibitive. This study aimed to develop an accessible, machine learning-based dynamic weighted fusion (DWF) model using routine laboratory data to provide bidirectional risk stratification, particularly to reliably rule out platinum resistance before treatment initiation. Methods: In this retrospective study (2019–2023), seventy baseline clinical features were collected to differentiate platinum-resistant from platinum-sensitive ovarian cancer patients. We developed a DWF framework that dynamically integrates the top-performing classifiers from a library of 168 algorithms (combining 14 feature selection and 12 machine learning methods). Class imbalance was addressed via oversampling, and model efficacy was evaluated using area under the curve (AUC), accuracy, sensitivity, and specificity. Results: The DWF model achieved a robust AUC of 0.760 (95% CI: 0.683–0.837), outperforming all individual base classifiers. Subgroup analysis demonstrated highly consistent overall discrimination across initial treatment strategies (AUC of 0.755 for primary debulking surgery and 0.761 for neoadjuvant chemotherapy). Feature interpretation highlighted that resistance is driven by synergistic dysregulation of systemic inflammation and hypercoagulability, rather than single biomarkers. Conclusions: The proposed DWF model effectively leverages low-cost, standardized clinical data to serve as a robust bidirectional stratification tool. Its exceptional ability to rule out resistance provides clinicians with the evidence-based confidence to proceed with standard therapies, while its high-risk alerts identify candidates for early therapeutic adjustments and enhanced surveillance in ovarian cancer care.
Full article
(This article belongs to the Special Issue Novel Approaches to Machine Learning and Artificial Intelligence in Cancer Research and Care (2nd Edition))
►▼
Show Figures

Figure 1
Open AccessReview
Advances in Percutaneous and Endovascular Locoregional Therapies for Primary and Metastatic Lung Cancer
by
Maria Mihailescu, Adam G. Fish and David C. Madoff
Cancers 2026, 18(8), 1189; https://doi.org/10.3390/cancers18081189 - 8 Apr 2026
Abstract
Many patients with primary or metastatic lung cancer are not candidates for surgery, additional radiation, or further systemic therapy due to advanced age or comorbidities; this creates a need for minimally invasive locoregional options. Image-guided thermal ablation (IGTA) is being applied across a
[...] Read more.
Many patients with primary or metastatic lung cancer are not candidates for surgery, additional radiation, or further systemic therapy due to advanced age or comorbidities; this creates a need for minimally invasive locoregional options. Image-guided thermal ablation (IGTA) is being applied across a broader spectrum of lesions, while bronchial artery chemoembolization (BACE) is emerging as a therapy option for treatment-refractory advanced disease. Recent studies in thermal ablation have focused on optimizing energy delivery and protocols, as well as improving ablation zone predictability and analysis. Advances in lesion targeting, including cone beam CT fusion, electromagnetic guidance, and robotic-assisted ablation, allow for treatment of subcentimeter and ground-glass lesions in anatomically challenging locations. Growing clinical experience supports IGTA for intrathoracic oligoprogression and as salvage therapy after recurrence. In the endovascular space, improved imaging, microcatheters, and drug-eluting microspheres have expanded the use of BACE for disease and symptom control in advanced lung cancer. Multimodal strategies combining minimally invasive locoregional treatments with systemic therapies and radiation are being explored, with early data showing improvements in survival without increased toxicity. This narrative review synthesizes emerging techniques, clinical data, and indications for percutaneous and endovascular lung cancer treatments and underscores the need for prospective and randomized trials to refine patient selection, treatment sequencing, and long-term outcomes.
Full article
(This article belongs to the Special Issue Feature Papers in Section “Methods and Technologies Development” in 2026)
►▼
Show Figures

Figure 1
Open AccessReview
Current and Future Perspectives of Adjuvant Therapy for Resected Colorectal Liver Metastases
by
Kozo Kataoka, Kei Kimura, Ayako Imada, Kazuma Ito, Zhenxin Rao, Yuko Fukumoto, Jihyung Song, Yuki Horio, Ryuichi Kuwahara, Motoi Uchino, Takayuki Yoshino, Eiji Oki, Yukihide Kanemitsu and Masataka Ikeda
Cancers 2026, 18(8), 1188; https://doi.org/10.3390/cancers18081188 - 8 Apr 2026
Abstract
The liver is the most common site of metastatic disease in patients with colorectal cancer. However, the multidisciplinary management of colorectal liver metastases (CLMs) remains suboptimal. Over the past several decades, numerous randomized trials have evaluated the efficacy of adjuvant chemotherapy following CLM
[...] Read more.
The liver is the most common site of metastatic disease in patients with colorectal cancer. However, the multidisciplinary management of colorectal liver metastases (CLMs) remains suboptimal. Over the past several decades, numerous randomized trials have evaluated the efficacy of adjuvant chemotherapy following CLM resection, revealing improvements in disease-free survival. Nevertheless, these studies have not consistently demonstrated benefits in overall survival, resulting in controversy with regard to the role of routine postoperative chemotherapy. Circulating tumor DNA (ctDNA) has recently emerged as a promising biomarker for detecting molecular residual disease after surgery. Multiple studies have consistently shown that postoperative ctDNA positivity is strongly associated with inferior recurrence-free survival and overall survival in patients with colorectal cancer. In addition to its prognostic value, ctDNA may also assist in guiding postoperative therapeutic decisions. In prospective observational studies of CLM, adjuvant chemotherapy provided potential clinical benefits primarily in patients with ctDNA-positive disease, whereas limited benefits were observed in ctDNA-negative patients. These findings suggest that ctDNA-based detection of molecular residual disease may aid in developing a framework for risk-adapted postoperative management after CLM resection. However, several challenges remain, including the identification of an optimal treatment regimen for ctDNA-positive patients and the improvement of ctDNA assay sensitivity. Ongoing biomarker-driven clinical trials may clarify whether ctDNA-guided strategies can improve patient selection and clinical outcomes following curative resection of CLM.
Full article
(This article belongs to the Special Issue Improving Outcomes in Resected Colorectal Liver Metastases: Risk-Stratified Chemotherapy and Aggressive Integration of Thermal Ablation)
Open AccessArticle
CHRNB4-Mediated Neuroactive Signaling Rewiring Drives Adaptive Resistance to BCL-2 Inhibition in Acute Myeloid Leukemia
by
Hiroaki Koyama, Sachiko Seo, William Tse, Sicheng Bian and Shujun Liu
Cancers 2026, 18(8), 1187; https://doi.org/10.3390/cancers18081187 - 8 Apr 2026
Abstract
Background: The clinical efficacy of the BCL-2 inhibitor venetoclax in acute myeloid leukemia (AML) is significantly undermined by the frequent emergence of drug resistance, which precipitates disease progression and poor patient outcomes. However, the molecular landscape of this resistance remains insufficiently understood. Methods:
[...] Read more.
Background: The clinical efficacy of the BCL-2 inhibitor venetoclax in acute myeloid leukemia (AML) is significantly undermined by the frequent emergence of drug resistance, which precipitates disease progression and poor patient outcomes. However, the molecular landscape of this resistance remains insufficiently understood. Methods: To address this, we developed venetoclax-resistant AML cell models and utilized transcriptomic profiling integrated with comprehensive in vitro and in vivo functional assays. Results: Resistant cells demonstrated sustained proliferation even under the suppression of BCL-2, MCL-1, and key intrinsic apoptotic markers, including cleaved PARP and caspase-9, indicating a bypass mechanism independent of classical BCL-2 signaling. Compared to their sensitive counterparts, resistant Kasumi-1 (VENK) and MV4-11 (VENM) cells exhibit aggressive growth phenotypes in vitro and in vivo, characterized by larger, more numerous spheroids and colonies, alongside heightened tumorigenicity in murine models. Transcriptomic profiling and KEGG analysis identified the neuroactive ligand–receptor interaction (NLRI) pathway as a significant signaling node shared between these resistant lines. While multiple NLRI-associated genes were altered, CHRNB4 was consistently and significantly downregulated in both VENK and VENM cells and tumors. Re-expression of CHRNB4 in resistant cells, a primary gain-of-function approach, significantly impaired colony formation, and tumor growth in vivo. Clinically, CHRNB4 downregulation correlates with shortened overall survival and diminished response to venetoclax. Conclusions: Our findings implicate the NLRI pathway in venetoclax resistance and identify CHRNB4 as a robust prognostic indicator and a promising therapeutic target for developing next-generation AML strategies.
Full article
(This article belongs to the Section Molecular Cancer Biology)
►▼
Show Figures

Figure 1
Open AccessArticle
Chemotherapy Enrichment of ID Family Expression Is Associated with IL-6 Signaling in Ovarian Cancer
by
Megan Anne Keene, Darren Lighter, Cassandra Brenner, Ixchel Urbano, Katelyn Shelby, Samuel F. Gilbert, Mikella Robinson and Carrie D. House
Cancers 2026, 18(8), 1186; https://doi.org/10.3390/cancers18081186 - 8 Apr 2026
Abstract
Background/Objectives: Ovarian cancer (OC) remains the most lethal gynecologic malignancy, largely due to late-stage diagnosis and high rates of recurrence following platinum-based chemotherapy. Growing evidence implicates cancer stem-like cells (CSCs) in OC relapse, as these cells exhibit enhanced chemoresistance, stemness, epithelial–mesenchymal transition
[...] Read more.
Background/Objectives: Ovarian cancer (OC) remains the most lethal gynecologic malignancy, largely due to late-stage diagnosis and high rates of recurrence following platinum-based chemotherapy. Growing evidence implicates cancer stem-like cells (CSCs) in OC relapse, as these cells exhibit enhanced chemoresistance, stemness, epithelial–mesenchymal transition (EMT), and the capacity to remodel the tumor microenvironment. Inhibitors of DNA-binding (ID) 1-4 proteins are transcription factors with known redundancy; however, their collective role in OC chemotherapy response remains poorly defined. Here, we examined how ID family signaling responds to chemotherapy and contributes to CSC-associated features and microenvironment remodeling. Methods: Publicly available patient data, OC cell lines, and a subcutaneous xenograft mouse model were used to correlate changes in ID1-4 expression with CSCs, EMT, and the tumor microenvironment (TME). OC cell lines were used for in vitro assays to evaluate CSC features and IL-6 production in the presence of carboplatin and/or a small molecule inhibitor of ID proteins, AGX51. Results: Analysis of clinical datasets, cell lines, and in vivo models revealed enrichment of ID1-4 following chemotherapy, with additive increases across treatment cycles. In vivo ID2 and ID4 expression was associated with IL-6 secretion and loss of anti-tumoral macrophages. Pan-ID inhibition demonstrated that cumulative ID activity minimally supports CSC maintenance during chemotherapy, while more strongly regulating IL-6 secretion. Conclusions: IL-6 production from cancer cells was at least partially dependent on ID proteins, linking collective ID signaling to microenvironment remodeling and relapse potential in ovarian cancer.
Full article
(This article belongs to the Special Issue Ovarian Cancer Stem Cells and Tumor Microenvironment)
►▼
Show Figures

Figure 1
Open AccessArticle
Integrating Machine Learning and Dynamic Bayesian Networks to Identify the Factors Associated with Subsequent Intrapulmonary Metastasis Classification After Initial Single Primary Lung Cancer
by
Wei Liu, Aliss T. C. Chang, Joyce W. Y. Chan, Junko C. S. Chan, Rainbow W. H. Lau, Tony S. K. Mok and Calvin S. H. Ng
Cancers 2026, 18(8), 1185; https://doi.org/10.3390/cancers18081185 - 8 Apr 2026
Abstract
Background/Objectives: Intrapulmonary metastasis (IPM) after an initial single primary lung cancer (SPLC) is an adverse follow-up pattern; however, when studying population-based longitudinal records, the determinants remain unclear. We aimed to identify factors associated with subsequent IPM after initial SPLC using artificial intelligence (AI)-driven
[...] Read more.
Background/Objectives: Intrapulmonary metastasis (IPM) after an initial single primary lung cancer (SPLC) is an adverse follow-up pattern; however, when studying population-based longitudinal records, the determinants remain unclear. We aimed to identify factors associated with subsequent IPM after initial SPLC using artificial intelligence (AI)-driven analytical approaches. Methods: We used Surveillance, Epidemiology, and End Results (SEER) lung cancer records from 2000 to 2019. Adults with at least two records were restricted to those with SPLC at the first record. Outcome at the second record was registry-classified IPM versus persistent SPLC. A machine learning framework based on random forest models was developed using baseline variables, first record characteristics, and the interval between records. Temporal validation was performed by training on cases from 2000 to 2013 and testing on cases from 2014 to 2019. A dynamic Bayesian network (DBN) supported simulated intervention (SI) analyses to estimate model-implied risk ratios (RRs) with 95% confidence intervals (CIs). Results: Among 3450 patients, 361 had registry-classified IPM at the second record. The random forest model achieved an area under the curve (AUC) of 0.852 in internal validation and 0.929 in temporal validation. Surgery and record timing were the leading predictors. The DBN retained surgery as the only direct parent and achieved an AUC of 0.779. SI analyses showed higher IPM probability for pleural invasion level (PL) 3 versus PL 0, RR 1.378 (95% CI, 1.080–1.657). Lobectomy with mediastinal lymph node dissection versus wedge resection lowered the IPM probability, RR 0.378 (95% CI, 0.219–0.636). Conclusions: AI-based time-sequence modeling integrating machine learning and a DBN allowed for the identification of surgery, pleural invasion, and record timing as key factors associated with subsequent IPM classification after initial SPLC. This framework demonstrates the potential of combining predictive and probabilistic dependency modeling to investigate registry-based disease classification patterns, and may support hypothesis generation for future prospective studies.
Full article
(This article belongs to the Special Issue Advances in Artificial Intelligence Methodologies and Applications in Cancer)
►▼
Show Figures

Figure 1
Open AccessReview
Integrating Artificial Intelligence into Breast Cancer Histopathology: Toward Improved Diagnosis and Prognosis
by
Gavino Faa, Eleonora Lai, Flaviana Cau, Ferdinando Coghe, Massimo Rugge, Jasjit S. Suri, Claudia Codipietro, Benedetta Congiu, Simona Graziano, Ekta Tiwari, Andrea Pretta, Pina Ziranu, Mario Scartozzi and Matteo Fraschini
Cancers 2026, 18(7), 1184; https://doi.org/10.3390/cancers18071184 - 7 Apr 2026
Abstract
Histopathological evaluation of tissue sections remains the gold standard for the diagnosis, classification, and grading of breast cancer (BC). The widespread adoption of whole-slide imaging (WSI) has enabled the digitization of histological slides and facilitated the development of artificial intelligence (AI) approaches for
[...] Read more.
Histopathological evaluation of tissue sections remains the gold standard for the diagnosis, classification, and grading of breast cancer (BC). The widespread adoption of whole-slide imaging (WSI) has enabled the digitization of histological slides and facilitated the development of artificial intelligence (AI) approaches for computational pathology. In recent years, machine learning and deep learning (DL) algorithms have been increasingly investigated for the analysis of hematoxylin and eosin (H&E)-stained images, with potential applications in tumor detection, histological classification, prognostic stratification, and prediction of treatment response. This narrative review summarizes recent developments in AI-driven models applied to BC histopathology and discusses their potential role in supporting diagnostic and prognostic assessment. Several studies have demonstrated the promising performance of DL algorithms in tasks such as the detection of lymph node metastases, assessment of residual tumor after neoadjuvant therapy, and prediction of clinical outcomes from histopathological images. Emerging research has also explored the possibility of inferring molecular and biomarker information from histology images, although these approaches currently identify statistical associations rather than direct molecular measurements. Despite the rapid expansion of this research field, significant barriers remain before routine clinical implementation can be achieved. Key challenges include dataset bias, variability in staining and image acquisition, limited external validation across institutions, and the need for transparent and reproducible model development. In addition, the translation of AI-based systems into clinical practice requires compliance with regulatory frameworks governing software used for medical purposes, such as those established by the U.S. Food and Drug Administration. Overall, AI represents a promising research direction in computational pathology and may contribute to decision-support tools capable of assisting pathologists in the analysis of digital slides. Continued efforts toward methodological rigor, large multicenter datasets, and prospective validation studies will be essential to determine the future role of AI in BC histopathology.
Full article
(This article belongs to the Collection Artificial Intelligence in Oncology)
►▼
Show Figures

Figure 1
Open AccessArticle
Long-Term Young Adult Cancer Survivors with Ovarian Cancer: Subgroup Analysis of the Study “Expression VI–Carolin Meets HANNA–Holistic Analysis of Long-Term Survival with Ovarian Cancer”: The International NOGGO, ENGOT, and GCIG Survey
by
Desislava Dimitrova, Eleftherios Pierre Samartzis, Dario Zocholl, Maria-Pilar Barretina-Ginesta, Katharina Leitner, Pavel Havelka, Patriciu Achimas-Cadariu, Cagatay Taskiran, Suzana Mittelstadt, Els Van Nieuwenhuysen, Gerd Bauerschmitz, Viola Heinzelmann-Schwarz, Ainhoa Madariaga, Uta Ringsdorf, Tibor Zwimpfer, Caterina Madroñal, Hans-Martin Enzinger, Sara Al Rubaish, Jalid Sehouli and Hannah Woopen
Cancers 2026, 18(7), 1183; https://doi.org/10.3390/cancers18071183 - 7 Apr 2026
Abstract
Background/Objectives: Ovarian cancer is typically diagnosed in postmenopausal women, so there are limited data available for young adult cancer survivors (YACS). The aim was to assess the patient perspective of YACS. Methods: In this international and multicenter cross-sectional survey study, patient history, long-term
[...] Read more.
Background/Objectives: Ovarian cancer is typically diagnosed in postmenopausal women, so there are limited data available for young adult cancer survivors (YACS). The aim was to assess the patient perspective of YACS. Methods: In this international and multicenter cross-sectional survey study, patient history, long-term side effects, and patient perspective were assessed. Long-term survival was defined as survival of at least five years after cancer diagnosis. Two groups were defined: (1) 18–40 years and (2) ≥41 years. Results: Altogether, 1833 long-term survivors (LTS) have been recruited, with 1771 patients ≥41 years and 62 patients 18–40 years at recruitment. FIGO stages were similar; among the patients, 99.0% had received primary surgery followed by chemotherapy in 90.3%. Almost 50% still experienced long-term side effects. Patients ≤ 40 years reported more frequently not only gastrointestinal symptoms such as nausea/vomiting (44.4%, p = 0.01), bloating (59.3%, p = 0.038), and constipation (60%, p = 0.015) but also depression (31.4%, p = 0.02), lymphedema (45.3%, p = 0.026), and concentration difficulties (30.6%, p = 0.002). Distress levels were also higher in YACS, especially concerning insurance/finances, work/school, child care, worries, and sadness. Polyneuropathy and secondary cancer were the only side effects that were more frequent in the elder cohort (polyneuropathy: 20.3% vs. 4.3%, p = 0.002, and secondary cancer: 8.4% vs. 0%, p = 0.014). YACS were more physically active (p = 0.003) and interested in studies about long-term cancer survivorship in 87.2%. Conclusions: Long-term side effects are equally common in YACS after ovarian cancer, but with a focus on practical problems, mental health, gastrointestinal problems, and sexuality. This knowledge should be incorporated into follow-up care of ovarian cancer patients in order to improve quality of life.
Full article
(This article belongs to the Special Issue Survivorship Following Childhood, Adolescent, and Young Adult Cancer)
►▼
Show Figures
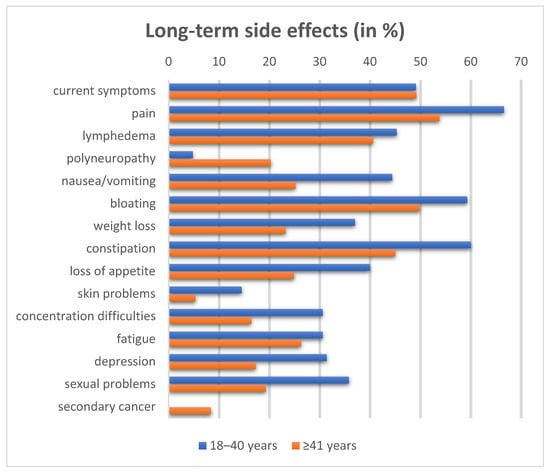
Figure 1
Open AccessSystematic Review
Supramaximal Resection in Glioblastoma: Expanding Surgical Boundaries in the Era of Precision Neuro-Oncology—A Systematic Review
by
Stuart D. Harper, Travis Perryman, Brandon Carlson-Clarke, Shivani Baisiwala, Brandon Rogowski, Amani Carson, Isha Sharma, Shail G. Patel, Eliana S. Oduro, Alondra Delgadillo, Nishvith Sudhakar, Mahmoud I. Youssef and Kunal S. Patel
Cancers 2026, 18(7), 1182; https://doi.org/10.3390/cancers18071182 - 7 Apr 2026
Abstract
Background: Glioblastoma remains the most aggressive and treatment-resistant primary brain tumor, with patient outcomes strongly associated with the extent of surgical resection. Tumor recurrence is largely driven by infiltrating glioma cells that extend beyond the contrast-enhancing margin, which has traditionally served as the
[...] Read more.
Background: Glioblastoma remains the most aggressive and treatment-resistant primary brain tumor, with patient outcomes strongly associated with the extent of surgical resection. Tumor recurrence is largely driven by infiltrating glioma cells that extend beyond the contrast-enhancing margin, which has traditionally served as the boundary for surgical resection. Advances in pre- and intraoperative imaging, functional mapping, and fluorescence guidance have challenged the conventional definition of “maximal safe resection” and given rise to the concept of supramaximal resection (SMR). This technique, where surgical resection extends beyond the contrast-enhancing border, has garnered significant interest in recent years and shown promising preliminary survival outcomes. However, the lack of standardized definitions and methodological consistency has limited reproducibility and clinical adoption. Methods: A systematic literature search of PubMed/MEDLINE, Embase, and Web of Science was performed from database inception through March 2026 in accordance with PRISMA guidelines. Studies investigating resection beyond the contrast-enhancing tumor margin in adult glioblastoma patients were evaluated for inclusion. Results: A total of 1045 records were identified, with 37 studies meeting inclusion criteria. Across studies, SMR was frequently associated with improved progression-free and overall survival in selected patients, particularly following complete contrast-enhancing tumor resection. However, substantial heterogeneity exists in SMR definitions, and the current body of evidence is largely retrospective and derived from high-volume centers. Conclusions: SMR represents a promising extension of maximal safe resection targeting infiltrative tumor beyond conventional imaging boundaries. While emerging evidence suggests survival benefits, variability in methodology and patient-specific factors require cautious interpretation. Future standardization and prospective validation are needed to better define the role of SMR within multimodal glioblastoma treatment.
Full article
(This article belongs to the Special Issue Modern Neurosurgical Management of Gliomas)
►▼
Show Figures
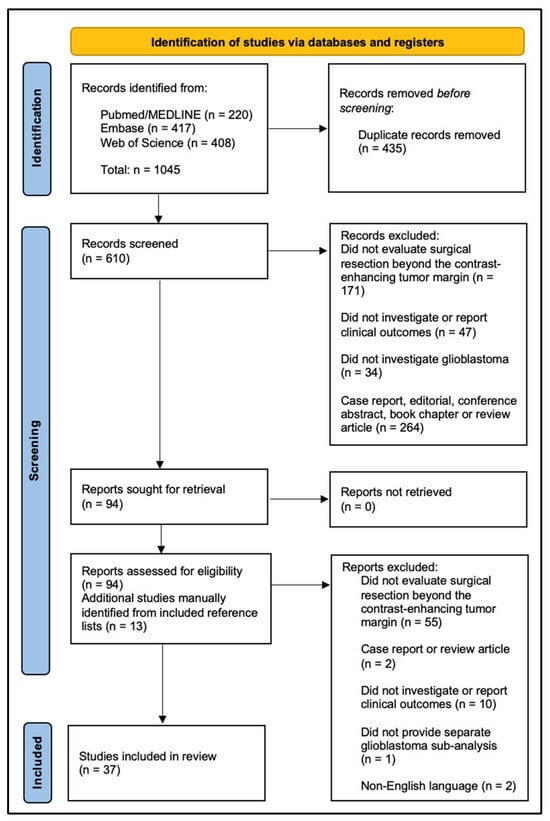
Figure 1

Journal Menu
► ▼ Journal Menu-
- Cancers Home
- Aims & Scope
- Editorial Board
- Reviewer Board
- Topical Advisory Panel
- Instructions for Authors
- Special Issues
- Topics
- Sections & Collections
- Article Processing Charge
- Indexing & Archiving
- Editor’s Choice Articles
- Most Cited & Viewed
- Journal Statistics
- Journal History
- Journal Awards
- Society Collaborations
- Conferences
- Editorial Office
Journal Browser
► ▼ Journal BrowserHighly Accessed Articles
Latest Books
E-Mail Alert
News
Topics
Topic in
Biomedicines, Cancers, JCM, Nanomaterials, Pharmaceutics, JNT
Application of Nanomaterials and Nanobiotechnology in Cancer
Topic Editors: Ayan Kumar Barui, Susheel Kumar NethiDeadline: 31 May 2026
Topic in
Cancers, Diagnostics, Medicina, Current Oncology
Prostate Cancer: Symptoms, Diagnosis & Treatment—3rd Edition
Topic Editors: Ana Faustino, Lúcio Lara Santos, Paula OliveiraDeadline: 30 June 2026
Topic in
Cancers, Current Oncology, JCM, Medicina, Onco
Cancer Biology and Radiation Therapy: 2nd Edition
Topic Editors: Chang Ming Charlie Ma, Ka Yu Tse, Ming-Yii Huang, Mukund SeshadriDeadline: 25 July 2026
Topic in
Biomedicines, Biomolecules, Cancers, Cells, Hematology Reports, IJMS
Advances in Molecular Pathogenesis and Targeted Therapies for Multiple Myeloma
Topic Editors: Chung Hoow Kok, Cindy H. S. Lee, Claudio CerchioneDeadline: 3 August 2026

Conferences
Special Issues
Special Issue in
Cancers
Neurosurgery Research on Brain Tumors
Guest Editor: Toshihide TanakaDeadline: 10 April 2026
Special Issue in
Cancers
Endothelial Cells in Inflammation, Tissue Repair, Ageing and Cancer: 2nd Edition
Guest Editor: Constantinos MikelisDeadline: 10 April 2026
Special Issue in
Cancers
Advances in Diagnostics and Treatments for Glioblastoma
Guest Editors: Sied Kebir, Manmeet Singh AhluwaliaDeadline: 10 April 2026
Special Issue in
Cancers
Radiation Therapy in Lung Cancer: From a Technical Revolution to a Combination with New Systemic Approaches (2nd Edition)
Guest Editor: Alessio BruniDeadline: 10 April 2026
Topical Collections
Topical Collection in
Cancers
Women’s Cancers Risk: Breast Cancer, Ovarian Cancer and Endometrial Cancer
Collection Editor: Ranjit Manchanda
Topical Collection in
Cancers
Molecular Signaling Pathways and Networks in Cancer
Collection Editors: Shihori Tanabe, You Song
Topical Collection in
Cancers
Combination Therapies in Cancers
Collection Editors: Patrycja Nowak-Sliwinska, Arjan W. Griffioen
Topical Collection in
Cancers
Emerging Therapeutics in Advanced Melanoma
Collection Editors: Jeremy S. Bordeaux, Luke D. Rothermel












