Theragnostic Use of Radiolabelled Dota-Peptides in Meningioma: From Clinical Demand to Future Applications
Abstract
:1. Introduction
2. Methods
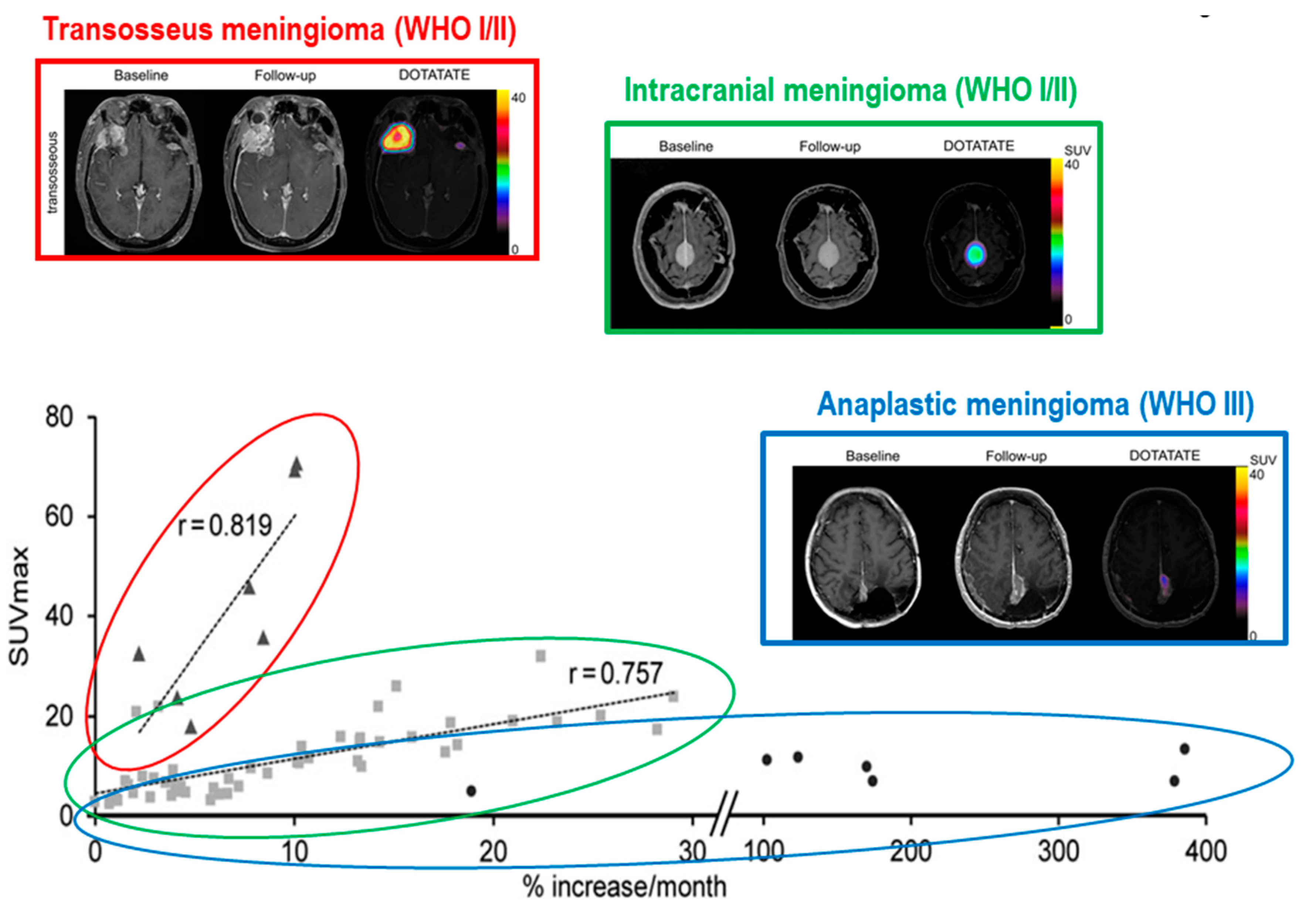
3. Diagnostic Challenges for MRI Imaging in Meningioma
4. Diagnostic Challenges for PET Imaging in Meningioma
5. SSTR-PET in the Definition of Tumour Growth and Tumour Extent for Surgery Planning
6. SSTR Radio-Guided Surgery
7. SSTR-PET in Radiotherapy Planning
8. SSTR-PET in Restaging of Treated Patients
9. Radionuclide Therapy in Meningioma
9.1. 90-Yttrium
9.2. 177-Lutetium
9.3. Miscellaneous
10. Conclusions and Future Perspectives
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Dolecek, T.A.; Propp, J.M.; Stroup, N.E.; Kruchko, C. CBTRUS statistical report: Primary brain and central nervous system tumors diagnosed in the United States in 2005–2009. Neuro-Oncology 2012, 14 (Suppl. 5), v1–v49. [Google Scholar] [CrossRef] [PubMed]
- Ostrom, Q.T.; Gittleman, H.; Fulop, J.; Liu, M.; Blanda, R.; Kromer, C.; Wolinsky, Y.; Kruchko, C.; Barnholtz-Sloan, J.S. CBTRUS Statistical Report: Primary Brain and Central Nervous System Tumors Diagnosed in the United States in 2008–2012. Neuro-Oncology 2015, 17 (Suppl. 4), iv1–iv62. [Google Scholar] [CrossRef] [PubMed]
- Vernooij, M.W.; Ikram, M.A.; Tanghe, H.L.; Vincent, A.J.; Hofman, A.; Krestin, G.P.; Niessen, W.J.; Breteler, M.M.; van der Lugt, A. Incidental findings on brain MRI in the general population. N. Engl. J. Med. 2007, 357, 1821–1828. [Google Scholar] [CrossRef] [PubMed]
- Louis, D.N.; Perry, A.; Reifenberger, G.; von Deimling, A.; Figarella-Branger, D.; Cavenee, W.K.; Ohgaki, H.; Wiestler, O.D.; Kleihues, P.; Ellison, D.W. The 2016 World Health Organization Classification of Tumors of the Central Nervous System: A summary. Acta Neuropathol. 2016, 131, 803–820. [Google Scholar] [CrossRef] [PubMed]
- Riemenschneider, M.J.; Perry, A.; Reifenberger, G. Histological classification and molecular genetics of meningiomas. Lancet Neurol. 2006, 5, 1045–1054. [Google Scholar] [CrossRef]
- Rogers, L.; Gilbert, M.; Vogelbaum, M.A. Intracranial meningiomas of atypical (WHO grade II) histology. J. Neurooncol. 2010, 99, 393–405. [Google Scholar] [CrossRef] [PubMed]
- Wu, A.; Jin, M.C.; Meola, A.; Wong, H.N.; Chang, S.D. Efficacy and toxicity of particle radiotherapy in WHO grade II and grade III meningiomas: A systematic review. Neurosurg. Focus 2019, 46, E12. [Google Scholar] [CrossRef] [PubMed]
- Stafford, S.L.; Perry, A.; Suman, V.J.; Meyer, F.B.; Scheithauer, B.W.; Lohse, C.M.; Shaw, E.G. Primarily resected meningiomas: Outcome and prognostic factors in 581 Mayo Clinic patients, 1978 through 1988. Mayo Clin. Proc. 1998, 73, 936–942. [Google Scholar] [CrossRef]
- Jääskeläinen, J.; Haltia, M.; Laasonen, E.; Wahlström, T.; Valtonen, S. The growth rate of intracranial meningiomas and its relation to histology. An analysis of 43 patients. Surg. Neurol. 1985, 24, 165–172. [Google Scholar] [CrossRef]
- Starke, R.M.; Przybylowski, C.J.; Sugoto, M.; Fezeu, F.; Awad, A.J.; Ding, D.; Nguyen, J.H.; Sheehan, J.P. Gamma Knife radiosurgery of large skull base meningiomas. J. Neurosurg. 2015, 122, 363–372. [Google Scholar] [CrossRef]
- Patibandla, M.R.; Lee, C.C.; Sheehan, J. Stereotactic Radiosurgery of Central Skull Base Meningiomas-Volumetric Evaluation and Long-Term Outcomes. World Neurosurg. 2017, 108, 176–184. [Google Scholar] [CrossRef] [PubMed]
- Rubinstein, A.B.; Shalit, M.N.; Cohen, M.L.; Zandbank, U.; Reichenthal, E. Radiation-induced cerebral meningioma: A recognizable entity. J. Neurosurg. 1984, 61, 966–971. [Google Scholar] [CrossRef] [PubMed]
- Yamanaka, R.; Hayano, A.; Kanayama, T. Radiation-Induced Meningiomas: An Exhaustive Review of the Literature. World Neurosurg. 2017, 97, 635–644. [Google Scholar] [CrossRef] [PubMed]
- Proctor, D.T.; Ramachandran, S.; Lama, S.; Sutherland, G.R. Towards Molecular Classification of Meningioma: Evolving Treatment and Diagnostic Paradigms. World Neurosurg. 2018, 119, 366–373. [Google Scholar] [CrossRef] [PubMed]
- Goldbrunner, R.; Minniti, G.; Preusser, M.; Jenkinson, M.D.; Sallabanda, K.; Houdart, E.; von Deimling, A.; Stavrinou, P.; Lefranc, F.; Lund-Johansen, M.; et al. EANO guidelines for the diagnosis and treatment of meningiomas. Lancet Oncol. 2016, 17, e383–e391. [Google Scholar] [CrossRef] [Green Version]
- Burford, B.J.; Welch, V.; Waters, E.; Tugwell, P.; Moher, D.; O’Neill, J.; Koehlmoos, T.; Petticrew, M. Testing the PRISMA-Equity 2012 reporting guideline: The perspectives of systematic review authors. PLoS ONE 2013, 8, e75122. [Google Scholar] [CrossRef] [PubMed]
- Buetow, M.P.; Buetow, P.C.; Smirniotopoulos, J.G. Typical, atypical, and misleading features in meningioma. Radiographics 1991, 11, 1087–1106. [Google Scholar] [CrossRef] [PubMed]
- Whittle, I.R.; Smith, C.; Navoo, P.; Collie, D. Meningiomas. Lancet 2004, 363, 1535–1543. [Google Scholar] [CrossRef]
- O’Leary, S.; Adams, W.M.; Parrish, R.W.; Mukonoweshuro, W. Atypical imaging appearances of intracranial meningiomas. Clin. Radiol. 2007, 62, 10–17. [Google Scholar] [CrossRef]
- Hakyemez, B.; Yildirim, N.; Erdoğan, C.; Kocaeli, H.; Korfali, E.; Parlak, M. Meningiomas with conventional MRI findings resembling intraaxial tumors: Can perfusion-weighted MRI be helpful in differentiation? Neuroradiology 2006, 48, 695–702. [Google Scholar] [CrossRef]
- Guermazi, A.; Lafitte, F.; Miaux, Y.; Adem, C.; Bonneville, J.F.; Chiras, J. The dural tail sign-beyond meningioma. Clin. Radiol. 2005, 60, 171–188. [Google Scholar] [CrossRef] [PubMed]
- Watts, J.; Box, G.; Galvin, A.; Brotchie, P.; Trost, N.; Sutherland, T. Magnetic resonance imaging of meningiomas: A pictorial review. Insights Imaging 2014, 5, 113–122. [Google Scholar] [CrossRef] [PubMed]
- Pieper, D.R.; Al-Mefty, O.; Hanada, Y.; Buechner, D. Hyperostosis associated with meningioma of the cranial base: Secondary changes or tumor invasion. Neurosurgery 1999, 44, 742–747. [Google Scholar] [CrossRef] [PubMed]
- Kotapka, M.J.; Kalia, K.K.; Martinez, A.J.; Sekhar, L.N. Infiltration of the carotid artery by cavernous sinus meningioma. J. Neurosurg. 1994, 81, 252–255. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Larson, J.J.; van Loveren, H.R.; Balko, M.G.; Tew, J.M., Jr. Evidence of meningioma infiltration into cranial nerves: Clinical implications for cavernous sinus meningiomas. J. Neurosurg. 1995, 83, 596–599. [Google Scholar] [CrossRef] [PubMed]
- Lang, F.F.; Macdonald, O.K.; Fuller, G.N.; DeMonte, F. Primary extradural meningiomas: A report on nine cases and review of the literature from the era of computerized tomography scanning. J. Neurosurg. 2000, 93, 940–950. [Google Scholar] [CrossRef] [PubMed]
- Kim, H.S.; Kim, S.H.; Kim, H.J.; Kang, S.W.; Kim, S.J.; Lee, J.H.; Hong, S.P.; Cho, Y.S.; Choi, J.Y. Primary Benign Intraosseous Meningioma on (18)F-FDG PET/CT Mimicking Malignancy. Nucl. Med. Mol. Imaging 2014, 48, 153–156. [Google Scholar] [CrossRef] [PubMed]
- Albano, D.; La Grutta, L.; Grassedonio, E.; Patti, C.; Lagalla, R.; Midiri, M.; Galia, M. Pitfalls in whole body MRI with diffusion weighted imaging performed on patients with lymphoma: What radiologists should know. Magn. Reson. Imaging 2016, 34, 922–931. [Google Scholar] [CrossRef]
- Krupa, K.; Bekiesińska-Figatowska, M. Artifacts in magnetic resonance imaging. Pol. J. Radiol. 2015, 80, 93–106. [Google Scholar]
- Bahrami, S.; Yim, C.M. Quality initiatives: Blind spots at brain imaging. Radiographics 2009, 29, 1877–1896. [Google Scholar] [CrossRef]
- Valotassiou, V.; Leondi, A.; Angelidis, G.; Psimadas, D.; Georgoulias, P. SPECT and PET imaging of meningiomas. Sci. World J. 2012, 2012, 412580. [Google Scholar] [CrossRef] [PubMed]
- Ericson, K.; Lilja, A.; Bergstrom, M.; Collins, V.P.; Eriksson, L.; Ehrin, E.; von Holst, H.; Lundqvist, H.; Långsrom, B.B.; Mosskin, M. Positron emission tomography with ([11C]methyl)-L-methionine, [11C] D-glucose and [68Ga]EDTA in supratentorial tumors. J. Comput. Assist. Tomogr. 1985, 9, 683–689. [Google Scholar] [CrossRef]
- Di Chiro, G.; Hatazawa, J.; David, A.K.; Rizzoli, H.V.; De Michele, D.J. Glucose utilization by intracranial meningiomas as an index of tumor aggressivity and probability of recurrence: A PET study. Radiology 1987, 164, 521–526. [Google Scholar] [CrossRef] [PubMed]
- Okuchi, S.; Okada, T.; Yamamoto, A.; Kanagaki, M.; Fushimi, Y.; Okada, T.; Yamauchi, M.; Kataoka, M.; Arakawa, Y.; Takahashi, J.C.; et al. Grading meningioma: A comparative study of thallium-SPECT and FDG-PET. Medicine (Baltim.) 2015, 94, e549. [Google Scholar] [CrossRef] [PubMed]
- Cremerius, U.; Bares, R.; Weis, J.; Sabri, O.; Mull, M.; Schröder, J.M.; Gilsbach, J.M.; Buell, U. Fasting improves discrimination of grade 1 and atypical or malignant meningioma in FDG-PET. J. Nucl. Med. 1997, 38, 26–30. [Google Scholar]
- Kado, H.; Ogawa, T.; Hatazawa, J.; Iwase, M.; Kanno, I.; Okudera, T.; Uemura, K. Radiation-induced meningioma evaluated with positron emission tomography with fludeoxyglucose F 18. Am. J. Neuroradiol. 1996, 17, 937–938. [Google Scholar] [PubMed]
- Lee, J.W.; Kang, K.W.; Park, S.H.; Lee, S.M.; Paeng, J.C.; Chung, J.K.; Lee, M.C.; Lee, D.S. 18F-FDG PET in the assessment of tumor grade and prediction of tumor recurrence in intracranial meningioma. Eur J. Nucl. Med. Mol. Imaging 2009, 36, 1574–1582. [Google Scholar] [CrossRef]
- Iuchi, T.; Iwasate, Y.; Namba, H.; Osato, K.; Saeki, N.; Yamaura, A.; Uchida, Y. Glucose and methionine uptake and proliferative activity in meningiomas. Neurol. Res. 1999, 21, 640–644. [Google Scholar]
- Park, Y.S.; Jeon, B.C.; Oh, H.S.; Lee, S.M.; Chun, B.K.; Chang, H.K. FDG PET/CT assessment of the biological behavior of meningiomas. J. Korean Neurosurg. Soc. 2006, 40, 428–433. [Google Scholar]
- Mitamura, K.; Yamamoto, Y.; Norikane, T.; Hatakeyama, T.; Okada, M.; Nishiyama, Y. Correlation of 18F-FDG and 11C-methionine uptake on PET/CT with Ki-67 immunohistochemistry in newly diagnosed intracranial meningiomas. Ann. Nucl. Med. 2018, 32, 627–633. [Google Scholar] [CrossRef]
- Tomura, N.; Saginoya, T.; Goto, H. 11C-Methionine Positron Emission Tomography/Computed Tomography Versus 18F-Fluorodeoxyglucose Positron Emission Tomography/Computed Tomography in Evaluation of Residual or Recurrent World Health Organization Grades II and III Meningioma After Treatment. J. Comput. Assist. Tomogr. 2018, 42, 517–521. [Google Scholar] [CrossRef] [PubMed]
- Astner, S.T.; Dobrei-Ciuchendea, M.; Essler, M.; Bundschuh, R.A.; Sai, H.; Schwaiger, M.; Molls, M.; Weber, W.A.; Grosu, A.L. Effect of 11C-methionine-positron emission tomography on gross tumor volume delineation in stereotactic radiotherapy of skull base meningiomas. Int J. Radiat. Oncol. Biol. Phys. 2008, 72, 1161–1167. [Google Scholar] [CrossRef] [PubMed]
- Ikeda, H.; Tsuyuguchi, N.; Kunihiro, N.; Ishibashi, K.; Goto, T.; Ohata, K. Analysis of progression and recurrence of meningioma using (11)C-methionine PET. Ann. Nucl. Med. 2013, 27, 772–780. [Google Scholar] [CrossRef] [PubMed]
- Ryttefors, M.; Danfors, T.; Lantini, F.; Montelius, A.; Blomquist, E.; Gudjonsson, O. Long-term evaluation of the effect of hypofractionated high-energy proton treatment of benign meningiomas by means of (11)C-L-methionine positron emission tomography. Eur. J. Nucl. Med. Imaging 2016, 43, 1432–1443. [Google Scholar] [CrossRef] [PubMed]
- Arita, H.; Kinoshita, M.; Okita, Y.; Hirayama, R.; Watabe, T.; Ishohashi, K.; Kijima, N.; Kagawa, N.; Fujimoto, Y.; Kishima, H.; et al. Clinical characteristics of meningiomas assessed by 11C-methionine and 18F-fluorodeoxyglucose positron-emission tomography. J. Neurooncol. 2012, 107, 379–386. [Google Scholar] [CrossRef] [PubMed]
- Dittmar, J.O.; Kratochwil, C.; Dittmar, A.; Welzel, T.; Habermehl, D.; Rieken, S.; Giesel, F.L.; Haberkorn, U.; Debus, J.; Combs, S.E. First intraindividual comparison of contrast-enhanced MRI, FET- and DOTATOC-PET in patients with intracranial meningiomas. Radiat. Oncol. 2017, 12, 169. [Google Scholar] [CrossRef] [PubMed]
- Cornelius, J.F.; Stoffels, G.; Filß, C.; Galldiks, N.; Slotty, P.; Kamp, M.; el Khatib, M.; Hänggi, D.; Sabel, M.; Felsberg, J.; et al. Uptake and tracer kinetics of O-(2-(18)F-fluoroethyl)-L-tyrosine in meningiomas: Preliminary results. Eur. J. Nucl. Med. Mol. Imaging 2015, 42, 459–467. [Google Scholar] [CrossRef]
- Cornelius, J.F.; Langen, K.J.; Stoffels, G.; Hänggi, D.; Sabel, M.; Jakob Steiger, H. Positron emission tomography imaging of meningioma in clinical practice: Review of literature and future directions. Neurosurgery 2012, 70, 1033–1041. [Google Scholar] [CrossRef]
- Tateishi, U.; Tateishi, K.; Hino-Shishikura, A.; Torii, I.; Inoue, T.; Kawahara, N. Multimodal approach to detect osseous involvement in meningioma: Additional value of (18)F-fluoride PET/CT for conventional imaging. Radiology 2014, 273, 521–528. [Google Scholar] [CrossRef]
- Giovacchini, G.; Fallanca, F.; Landoni, C.; Gianolli, L.; Picozzi, P.; Attuati, L.; Terreni, M.; Picchio, M.; Messa, C.; Fazio, F. C-11 choline versus F-18fluorodeoxyglucose for imaging meningiomas: An initial experience. Clin. Nucl. Med. 2009, 34, 7–10. [Google Scholar] [CrossRef]
- Tateishi, U.; Tateishi, K.; Shizukuishi, K.; Shishikura, A.; Murata, H.; Inoue, T.; Kawahara, N. 18F-fluoride PET/CT allows detection of hyperostosis and osseous involvement in meningioma: Initial experience. Clin. Nucl. Med. 2013, 38, e125–e131. [Google Scholar] [CrossRef] [PubMed]
- Liu, R.S.; Chang, C.P.; Guo, W.Y.; Pan, D.H.; Ho, D.M.; Chang, C.W.; Yang, B.H.; Wu, L.C.; Yeh, S.H. 11C-acetate versus 18F-FDG PET in detection of meningioma and monitoring the effect of gamma-knife radiosurgery. J. Nucl. Med. 2010, 51, 883–891. [Google Scholar] [CrossRef] [PubMed]
- Nakamura, M.; Roser, F.; Michel, J.; Jacobs, C.; Samii, M. Volumetric analysis of the growth rate of incompletely resected intracranial meningiomas. Zent. Neurochir. 2005, 66, 17–23. [Google Scholar] [CrossRef] [PubMed]
- Fountain, D.M.; Soon, W.C.; Matys, T.; Guilfoyle, M.R.; Kirollos, R.; Santarius, T. Volumetric growth rates of meningioma and its correlation with histological diagnosis and clinical outcome: A systematic review. Acta Neurochir. (Wien) 2017, 159, 435–445. [Google Scholar] [CrossRef] [PubMed]
- Nakasu, S.; Fukami, T.; Nakajima, M.; Watanabe, K.; Ichikawa, M.; Matsuda, M. Growth pattern changes of meningiomas: Long-term analysis. Neurosurgery 2005, 56, 946–955. [Google Scholar] [PubMed]
- Nakasu, S.; Nakasu, Y.; Fukami, T.; Jito, J.; Nozaki, K. Growth curve analysis of asymptomatic and symptomatic meningiomas. J. Neurooncol. 2011, 102, 303–310. [Google Scholar] [CrossRef]
- Lee, E.J.; Kim, J.H.; Park, E.S.; Kim, Y.H.; Lee, J.K.; Hong, S.H.; Cho, Y.H.; Kim, C.J. A novel weighted scoring system for estimating the risk of rapid growth in untreated intracranial meningiomas. J. Neurosurg. 2017, 127, 971–980. [Google Scholar] [CrossRef] [Green Version]
- Oya, S.; Kim, S.H.; Sade, B.; Lee, J.H. The natural history of intracranial meningiomas. J. Neurosurg. 2011, 114, 1250–1256. [Google Scholar] [CrossRef] [Green Version]
- Sommerauer, M.; Burkhardt, J.K.; Frontzek, K.; Rushing, E.; Buck, A.; Krayenbuehl, N.; Weller, M.; Schaefer, N.; Kuhn, F.P. 68Gallium-DOTATATE PET in meningioma: A reliable predictor of tumor growth rate? Neuro. Oncol. 2016, 18, 1021–1027. [Google Scholar] [CrossRef]
- Kunz, W.G.; Jungblut, L.M.; Kazmierczak, P.M.; Vettermann, F.J.; Bollenbacher, A.; Tonn, J.C.; Schichor, C.; Rominger, A.; Albert, N.L.; Bartenstein, P.; et al. Improved Detection of Transosseous Meningiomas Using 68Ga-DOTATATE PET/CT Compared with Contrast-Enhanced MRI. J. Nucl. Med. 2017, 58, 1580–1587. [Google Scholar] [CrossRef]
- Afshar-Oromieh, A.; Giesel, F.L.; Linhart, H.G.; Haberkorn, U.; Haufe, S.; Combs, S.E.; Podlesek, D.; Eisenhut, M.; Kratochwil, C. Detection of cranial meningiomas: Comparison of ⁶⁸Ga-DOTATOC PET/CT and contrast-enhanced MRI. Eur. J. Nucl. Med. Mol. Imaging 2012, 39, 1409–1415. [Google Scholar] [CrossRef] [PubMed]
- Rachinger, W.; Stoecklein, V.M.; Terpolilli, N.A.; Haug, A.R.; Ertl, L.; Pöschl, J.; Schüller, U.; Schichor, C.; Thon, N.; Tonn, J.C. Increased 68Ga-DOTATATE uptake in PET imaging discriminates meningioma and tumor-free tissue. J. Nucl. Med. 2015, 56, 347–353. [Google Scholar] [CrossRef] [PubMed]
- Henze, M.; Schuhmacher, J.; Hipp, P.; Kowalski, J.; Becker, D.W.; Doll, J.; Mäcke, H.R.; Hofmann, M.; Debus, J.; Haberkorn, U. PET imaging of somatostatin receptors using [68GA]DOTA-D-Phe1-Tyr3-octreotide: First results in patients with meningiomas. J. Nucl. Med. 2001, 42, 1053–1056. [Google Scholar] [PubMed]
- Dammers, R.; Hsu, S.P.; Krisht, A.F. Radioguided improved resection of a cranial base meningioma. Neurosurgery 2009, 64, onse84–onse85. [Google Scholar] [CrossRef] [PubMed]
- Gay, E.; Vuillez, J.P.; Palombi, O.; Brard, P.Y.; Bessou, P.; Passagia, J.G. Intraoperative and postoperative gamma detection of somatostatin receptors in bone-invasive en plaque meningiomas. Neurosurgery 2005, 57, 107–113. [Google Scholar] [CrossRef] [PubMed]
- Solfaroli Camillocci, E.; Schiariti, M.; Bocci, V.; Carollo, A.; Chiodi, G.; Colandrea, M.; Collamati, F.; Cremonesi, M.; Donnarumma, R.; Ferrari, M.E.; et al. First ex vivo validation of a radioguided surgery technique with β-radiation. Phys. Med. 2016, 32, 1139–1144. [Google Scholar] [CrossRef] [PubMed]
- Collamati, F.; Pepe, A.; Bellini, F.; Bocci, V.; Chiodi, G.; Cremonesi, M.; De Lucia, E.; Ferrari, M.E.; Frallicciardi, P.M.; Grana, C.M.; et al. Toward radioguided surgery with β-decays: Uptake of a somatostatin analogue, DOTATOC, in meningioma and high-grade glioma. J. Nucl. Med. 2015, 56, 3–8. [Google Scholar] [CrossRef]
- Milker-Zabel, S.; Zabel-du Bois, A.; Henze, M.; Huber, P.; Schulz-Ertner, D.; Hoess, A.; Haberkorn, U.; Debus, J. Improved target volume definition for fractionated stereotactic radiotherapy in patients with intracranial meningiomas by correlation of CT, MRI, and [68Ga]-DOTATOC-PET. Int J. Radiat. Oncol. Biol. Phys. 2006, 65, 222–227. [Google Scholar] [CrossRef]
- Gehler, B.; Paulsen, F.; Oksüz, M.O.; Hauser, T.K.; Eschmann, S.M.; Bares, R.; Pfannenberg, C.; Bamberg, M.; Bartenstein, P.; Belka, C.; et al. [68Ga]-DOTATOC-PET/CT for meningioma IMRT treatment planning. Radiat. Oncol. 2009, 4, 56. [Google Scholar] [CrossRef]
- Nyuyki, F.; Plotkin, M.; Graf, R.; Michel, R.; Steffen, I.; Denecke, T.; Geworski, L.; Fahdt, D.; Brenner, W.; Wurm, R. Potential impact of 68Ga-DOTATOC PET/CT on stereotactic radiotherapy planning of meningiomas. Eur. J. Nucl. Med. Mol. Imaging 2010, 37, 310–318. [Google Scholar] [CrossRef]
- Graf, R.; Nyuyki, F.; Steffen, I.G.; Michel, R.; Fahdt, D.; Wust, P.; Brenner, W.; Budach, V.; Wurm, R.; Plotkin, M. Contribution of 68Ga-DOTATOC PET/CT to target volume delineation of skull base meningiomas treated with stereotactic radiation therapy. Int J. Radiat. Oncol. Biol. Phys. 2013, 85, 68–73. [Google Scholar] [CrossRef] [PubMed]
- Combs, S.E.; Welzel, T.; Habermehl, D.; Rieken, S.; Dittmar, J.O.; Kessel, K.; Jäkel, O.; Haberkorn, U.; Debus, J. Prospective evaluation of early treatment outcome in patients with meningiomas treated with particle therapy based on target volume definition with MRI and 68Ga-DOTATOC-PET. Acta Oncol. 2013, 52, 514–520. [Google Scholar] [CrossRef] [PubMed]
- Madani, I.; Lomax, A.J.; Albertini, F.; Trnková, P.; Weber, D.C. Dose-painting intensity-modulated proton therapy for intermediate- and high-risk meningioma. Radiat. Oncol. 2015, 10, 72. [Google Scholar] [CrossRef] [PubMed]
- Stade, F.; Dittmar, J.O.; Jäkel, O.; Kratochwil, C.; Haberkorn, U.; Debus, J.; Combs, S.E. Influence of 68Ga-DOTATOC on sparing of normal tissue for radiation therapy of skull base meningioma: Differential impact of photon and proton radiotherapy. Radiat. Oncol. 2018, 13, 58. [Google Scholar] [CrossRef] [PubMed]
- Maclean, J.; Fersht, N.; Sullivan, K.; Kayani, I.; Bomanji, J.; Dickson, J.; O’Meara, C.; Short, S. Simultaneous 68Ga DOTATATE Positron Emission Tomography/Magnetic Resonance Imaging in Meningioma Target Contouring: Feasibility and Impact Upon Interobserver Variability Versus Positron Emission Tomography/Computed Tomography and Computed Tomography/Magnetic Resonance Imaging. Clin. Oncol. (R. Coll. Radiol.) 2017, 29, 448–458. [Google Scholar] [PubMed]
- Acker, G.; Kluge, A.; Lukas, M.; Conti, A.; Pasemann, D.; Meinert, F.; Anh Nguyen, P.T.; Jelgersma, C.; Loebel, F.; Budach, V.; et al. Impact of 68Ga-DOTATOC PET/MRI on robotic radiosurgery treatment planning in meningioma patients: First experiences in a single institution. Neurosurg. Focus 2019, 46, E9. [Google Scholar] [CrossRef]
- Jaeckle, K.A. Neuroimaging for central nervous system tumors. Semin. Oncol. 1991, 18, 150–157. [Google Scholar] [PubMed]
- Galldiks, N.; Albert, N.L.; Sommerauer, M.; Grosu, A.L.; Ganswindt, U.; Law, I.; Preusser, M.; Le Rhun, E.; Vogelbaum, M.A.; Zadeh, G.; et al. PET imaging in patients with meningioma-report of the RANO/PET Group. Neuro-Oncology 2017, 19, 1576–1587. [Google Scholar] [CrossRef] [PubMed]
- Ivanidze, J.; Roytman, M.; Lin, E.; Magge, R.S.; Pisapia, D.J.; Liechty, B.; Karakatsanis, N.; Ramakrishna, R.; Knisely, J.; Schwartz, T.H.; et al. Gallium-68 DOTATATE PET in the Evaluation of Intracranial Meningiomas. J. Neuroimaging 2019, in press. [Google Scholar] [CrossRef]
- Minutoli, F.; Amato, E.; Sindoni, A.; Cardile, D.; Conti, A.; Herberg, A.; Baldari, S. Peptide receptor radionuclide therapy in patients with inoperable meningiomas: Our experience and review of the literature. Cancer Biother. Radiopharm. 2014, 29, 193–199. [Google Scholar] [CrossRef]
- Kwekkeboom, D.J.; de Herder, W.W.; Kam, B.L.; van Eijck, C.H.; van Essen, M.; Kooij, P.P.; Feelders, R.A.; van Aken, M.O.; Krenning, E.P. Treatment with the radiolabeled somatostatin analog [177 Lu-DOTA 0,Tyr3]octreotate: Toxicity, efficacy, and survival. J. Clin. Oncol. 2008, 26, 2124–2130. [Google Scholar] [CrossRef] [PubMed]
- Krenning, E.P.; Kooij, P.P.; Bakker, W.H.; Breeman, W.A.; Postema, P.T.; Kwekkeboom, D.J.; Oei, H.Y.; de Jong, M.; Visser, T.J.; Reijs, A.E.; et al. Radiotherapy with a radiolabeled somatostatin analogue, [111In-DTPA-D-Phe1]-octreotide. A case history. Ann. N. Y. Acad. Sci. 1994, 733, 496–506. [Google Scholar] [CrossRef] [PubMed]
- Otte, A.; Jermann, E.; Behe, M.; Goetze, M.; Bucher, H.C.; Roser, H.W.; Heppeler, A.; Mueller-Brand, J.; Maecke, H.R. DOTATOC: A powerful new tool for receptor-mediated radionuclide therapy. Eur. J. Nucl. Med. 1997, 24, 792–795. [Google Scholar] [PubMed] [Green Version]
- Albert, R.; Smith-Jones, P.; Stolz, B.; Simeon, C.; Knecht, H.; Bruns, C.; Pless, J. Direct synthesis of [DOTA-DPhe1]-octreotide and [DOTA-DPhe1,Tyr3]-octreotide (SMT487): Two conjugates for systemic delivery of radiotherapeutical nuclides to somatostatin receptor positive tumors in man. Bioorg. Med. Chem. Lett. 1998, 8, 1207–1210. [Google Scholar] [CrossRef]
- Bodei, L.; Kidd, M.; Paganelli, G.; Grana, C.M.; Drozdov, I.; Cremonesi, M.; Lepensky, C.; Kwekkebom, D.J.; Baum, R.P.; Krenning, E.P.; et al. Long-term tolerability of PRRT in 807 patients with neuroendocrine tumours: The value and limitations of clinical factors. Eur. J. Nucl. Med. Mol. Imaging 2015, 42, 5–19. [Google Scholar] [CrossRef] [PubMed]
- Rolleman, E.J.; Valkema, R.; de Jong, M.; Kooij, P.P.; Krenning, E.P. Safe and effective inhibition of renal uptake of radiolabeled octreotide by a combination of lysine and arginine. Eur. J. Nucl. Med. Mol. Imaging 2003, 30, 9–15. [Google Scholar] [CrossRef]
- Bushnell, D.; Menda, Y.; O’Dorisio, T.; Madsen, M.; Miller, S.; Carlisle, T.; Squires, S.; Kahn, D.; Walkner, W.; Connolly, M.; et al. Effects of intravenous amino acid administration with Y-90 DOTA-Phe1-Tyr3-Octreotide (SMT487[OctreoTher) treatment. Cancer Biother. Radiopharm. 2004, 19, 35–41. [Google Scholar] [CrossRef] [PubMed]
- Del Prete, M.; Buteau, F.A.; Beauregard, J.M. Personalized 177Lu-octreotate peptide receptor radionuclide therapy of neuroendocrine tumours: A simulation study. Eur. J. Nucl. Med. Mol. Imaging 2017, 44, 1490–1500. [Google Scholar] [CrossRef]
- Sundlöv, A.; Sjögreen-Gleisner, K.; Svensson, J.; Ljungberg, M.; Olsson, T.; Bernhardt, P.; Tennvall, J. Individualised 177Lu-DOTATATE treatment of neuroendocrine tumours based on kidney dosimetry. Eur. J. Nucl. Med. Mol. Imaging 2017, 44, 1480–1489. [Google Scholar] [CrossRef] [Green Version]
- FDA Approves Lutathera for GEP NET Therapy. J. Nucl. Med. 2018, 59, 9N.
- Kratochwil, C.; Fendler, W.P.; Eiber, M.; Baum, R.; Bozkurt, M.F.; Czernin, J.; Delgado Bolton, R.C.; Ezzidin, S.; Forrer, F.; Hicks, R.J.; et al. EANM procedure guidelines for radionuclide therapy with (177)Lu-labelled PSMA ligands ((177Lu)Lu-PSMA-RLT). Eur. J. Nucl. Med. Mol. Imaging 2019, in press. [Google Scholar] [CrossRef] [PubMed]
- Durand, A.; Champier, J.; Jouvet, A.; Labrousse, F.; Honnorat, J.; Guyotat, J.; Fèvre-Montange, M. Expression of c-Myc, neurofibromatosis Type 2, somatostatin receptor 2 and erb-B2 in human meningiomas: Relation to grades or histotypes. Clin. Neuropathol. 2008, 27, 334–345. [Google Scholar] [CrossRef] [PubMed]
- Bartolomei, M.; Bodei, L.; De Cicco, C.; Grana, C.M.; Cremonesi, M.; Botteri, E.; Baio, S.M.; Aricò, D.; Sansovini, M.; Paganelli, G. Peptide receptor radionuclide therapy with (90)Y-DOTATOC in recurrent meningioma. Eur. J. Nucl. Med. Mol. Imaging 2009, 36, 1407–1416. [Google Scholar] [CrossRef] [PubMed]
- Gerster-Gilliéron, K.; Forrer, F.; Maecke, H.; Mueller-Brand, J.; Merlo, A.; Cordier, D. 90Y-DOTATOC as a Therapeutic Option for Complex Recurrent or Progressive Meningiomas. J. Nucl. Med. 2015, 56, 1748–1751. [Google Scholar] [CrossRef] [PubMed]
- Marincek, N.; Radojewski, P.; Dumont, R.A.; Brunner, P.; Müller-Brand, J.; Maecke, H.R.; Briel, M.; Walter, M.A. Somatostatin receptor-targeted radiopeptide therapy with 90Y-DOTATOC and 177Lu-DOTATOC in progressive meningioma: Long-term results of a phase II clinical trial. J. Nucl. Med. 2015, 56, 171–176. [Google Scholar] [CrossRef] [PubMed]
- Seystahl, K.; Stoecklein, V.; Schüller, U.; Rushing, E.; Nicolas, G.; Schäfer, N.; Ilhan, H.; Pangalu, A.; Weller, M.; Tonn, J.C.; et al. Somatostatin receptor-targeted radionuclide therapy for progressive meningioma: Benefit linked to 68Ga-DOTATATE/-TOC uptake. Neuro-Oncology 2016, 18, 1538–1547. [Google Scholar] [CrossRef] [PubMed]
- Parghane, R.V.; Talole, S.; Basu, S. Prevalence of hitherto unknown brain meningioma detected on 68Ga-DOTATATE positron-emission tomography/computed tomography in patients with metastatic neuroendocrine tumor and exploring potential of 177Lu-DOTATATE peptide receptor radionuclide therapy as single-shot treatment approach targeting both tumors. World J. Nucl. Med. 2019, 18, 160–170. [Google Scholar]
- Bodei, L.; Cremonesi, M.; Grana, C.M.; Fazio, N.; Iodice, S.; Baio, S.M.; Bartolomei, M.; Lombardo, D.; Ferrari, M.E.; Sansovini, M.; et al. Peptide receptor radionuclide therapy with ¹⁷⁷Lu-DOTATATE: The IEO phase I-II study. Eur. J. Nucl. Med. Mol. Imaging 2011, 38, 2125–2135. [Google Scholar] [CrossRef]
- van Essen, M.; Krenning, E.P.; Kooij, P.P.; Bakker, W.H.; Feelders, R.A.; de Herder, W.W.; Wolbers, J.G.; Kwekkeboom, D.J. Effects of therapy with [177Lu-DOTA0, Tyr3] octreotate in patients with paraganglioma, meningioma, small cell lung carcinoma, and melanoma. J. Nucl. Med. 2006, 47, 1599–1606. [Google Scholar]
- Sabet, A.; Ahmadzadehfar, H.; Herrlinger, U.; Wilinek, W.; Biersack, H.J.; Ezziddin, S. Successful radiopeptide targeting of metastatic anaplastic meningioma: Case report. Radiat. Oncol. 2011, 6, 94. [Google Scholar] [CrossRef]
- Kreissl, M.C.; Hänscheid, H.; Löhr, M.; Verburg, F.A.; Schiller, M.; Lassmann, M.; Reiners, C.; Samnick, S.S.; Buck, A.K.; Flentje, M.; et al. Combination of peptide receptor radionuclide therapy with fractionated external beam radiotherapy for treatment of advanced symptomatic meningioma. Radiat. Oncol. 2012, 7, 99. [Google Scholar] [CrossRef] [PubMed]
- Paganelli, G.; Zoboli, S.; Cremonesi, M.; Bodei, L.; Ferrari, M.; Grana, C.; Bartolomei, M.; Orsi, F.; De Cicco, C.; Mäcke, H.R.; et al. Receptor-mediated radiotherapy with 90Y-DOTA-D-Phe1-Tyr3-octreotide. Eur. J. Nucl. Med. 2001, 28, 426–434. [Google Scholar] [CrossRef] [PubMed]
- Otte, A.; Herrmann, R.; Heppeler, A.; Behe, M.; Jermann, E.; Powell, P.; Maecke, H.R.; Muller, J. Yttrium-90 DOTATOC: First clinical results. Eur. J. Nucl. Med. 1999, 26, 1439–1447. [Google Scholar] [CrossRef] [PubMed]
- Afshar-Oromieh, A.; Wolf, M.B.; Kratochwil, C.; Giesel, F.L.; Combs, S.E.; Dimitrakopoulou-Strauss, A.; Gnirs, R.; Roethke, M.C.; Schlemmer, H.P.; Haberkorn, U. Comparison of ⁶⁸Ga-DOTATOC-PET/CT and PET/MRI hybrid systems in patients with cranial meningioma: Initial results. Neuro-Oncology 2015, 17, 312–319. [Google Scholar] [CrossRef] [PubMed]


| Radionuclide | Physical Half-Life | Emission Peaks Energy | Main Applications | Decay (Abundance) | Penetration Range | Source of Production | Specific Activity |
|---|---|---|---|---|---|---|---|
| 111-Indium (111In) | 67.9 h | γ (173 keV) γ (247 keV) | Imaging | Electron Capture (100%) | 0.002–0.5 mm | Cyclotron | Medium |
| 90-Yttrium (90Y) | 64 h | β− (2288 keV) | Therapy | β− (100%) | 4–8 mm | Generator or reactor | High |
| 177-Lutetium (177Lu) | 6.7 days | β− (500 keV) γ (208 keV) γ (113 keV) | Only in therapy | β− (100%) γ (27%) | 1–2 mm | Reactor | Medium/high |
| Author [Ref] | Meningioma (Total Cohort) | Year | Therapy | Cycles | Total Activity (Gbq) | Type of Response | PFS after PRRT (Months) | Previous Treatment | FU (Months) | Other Main Results (If Present) |
|---|---|---|---|---|---|---|---|---|---|---|
| Bartolomei et al. [93] | 29 (29) | 2009 | 90Y-DOTATOC | 2–6 (range) | 5–15 (range) | SD 19 PD 10 | 61 (median for G1) 13 (median for G2-G3) | Surgery 26 RT 18 CT 1 CT + RT 1 | 3 (for response assessment) 4–77 (range) | Median OS was 40 months. Stabilization of neurological symptoms in 41% until 1 year from last PRRT. |
| Gester–Gillierson et al. [94] | 15 (15) | 2015 | 90Y-DOTATOC | 2–4 (range) | 13 (median) 1.35–14.8 (range) | SD 13 PD 2 | 24 (median) 0–137 (range) | Surgery 6 NA or naive 5 Surgery + RT 3 Surgery + RT + CT 1 | 49.7 (mean) 12–137 (range) | Hematologic, neurologic, and renal toxicities were transient and moderate. |
| Marincek et al. [95] | 34 (34) | 2015 | 90Y-DOTATOC 177Lu-DOTATOC | 1–4 (range) | 1.5–18.3 (range for Y) 7.4–22.2 (range for Lu) | SD 23 PD 11 | NA | Surgery 25 CT 11 RT 1 | 21.8 (median) 1–137.4 (range) | PRRT may improve the quality and longevity of life with no significant complication. |
| Seystahl et al. [96] | 20 (20) | 2016 | 177Lu-DOTATATE90Y-DOTATOC | 1–4 (range) 3 (median) | 13.7–27.6 (range) 20.2 (median) | SD 10 PD 10 | 5.4 (median) | RT 18 ** AE 8 ** Surgery 7 ** CT 6 ** | 20 (median) | PFS at 6 months in 42%. OS at 12 months in 79%. |
| Parghane et al. [97] | 5 (500) | 2019 | 177Lu-DOTATATE | 2–6 (range) | 19.86 (mean) 13.28–29.97 (range) | SD 5 §§ PR 3 § PD 2 § | 26.25 (mean) 16.65–35.84 (range) | CT 2 ** SSA 2 ** Surgery 1 ** | 19.4 (mean) 8–36 (range) | Regard to neurological symptomatic response: CR in 2/5, PD in 2/5, and PR in 1/5 |
| Bodei et al. [98] | 1 (51) | 2011 | 177Lu-DOTATATE | 1–6 * (range) | 3.7–29.2 (range) * | SD 1 | 36 (median) * | SSA 43 * Surgery 35 * Surgery + SSA 30 * CT 11 * | 29 (median) * 4–66 (range) * | OS in 68% at 36 months. * |
| Minutoli et al. [80] | 8 (8) | 2014 | 111In-Pentetreotide 90Y-DOTATOC 177Lu-DOTATATE | 2–4 (range) | 4.8–29 (range) | SD 5 PR 2 PD 1 | NA | Surgery 4 NA or naive 2 Surgery + RT 1 Surgery + PRRT 1 | 4–50 (range) | Significant improvement of clinical condition in 4/8 patients. 111In might be used in cases with a high risk of renal toxicity |
| Van Essen et al. [99] | 5 (22) | 2006 | 177Lu-octreotate | 2–4 (range) | 14.8–29.6 (range) | PD 3 SD 2 | NA | RT + Surgery 3 RT + CT 2 | 3 (at least) | PRRT could be used if the disease is slowly progressive. |
| Sabet et al. [100] | 1 (1) | 2011 | 177Lu-DOTATE | 3 | 18.7 | SD | NA | NA | 3 | Pain reduction and improved life-quality. |
| Kreissl et al. [101] | 10 (10) | 2012 | RT + 177Lu-DOTATATE or 177Lu-DOTATOC | 1 | 7.4 ± 0.3 | SD 8 PR 1 CR 1 | NA | Surgery 9 Surgery + RT 1 | 13.4 (median) 1.1–17.0 (range) | Increased uptake of 68Ga-DOTA in meningioma after the combined therapy |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Laudicella, R.; Albano, D.; Annunziata, S.; Calabrò, D.; Argiroffi, G.; Abenavoli, E.; Linguanti, F.; Albano, D.; Vento, A.; Bruno, A.; et al. Theragnostic Use of Radiolabelled Dota-Peptides in Meningioma: From Clinical Demand to Future Applications. Cancers 2019, 11, 1412. https://doi.org/10.3390/cancers11101412
Laudicella R, Albano D, Annunziata S, Calabrò D, Argiroffi G, Abenavoli E, Linguanti F, Albano D, Vento A, Bruno A, et al. Theragnostic Use of Radiolabelled Dota-Peptides in Meningioma: From Clinical Demand to Future Applications. Cancers. 2019; 11(10):1412. https://doi.org/10.3390/cancers11101412
Chicago/Turabian StyleLaudicella, Riccardo, Domenico Albano, Salvatore Annunziata, Diletta Calabrò, Giovanni Argiroffi, Elisabetta Abenavoli, Flavia Linguanti, Domenico Albano, Antonio Vento, Antonio Bruno, and et al. 2019. "Theragnostic Use of Radiolabelled Dota-Peptides in Meningioma: From Clinical Demand to Future Applications" Cancers 11, no. 10: 1412. https://doi.org/10.3390/cancers11101412








