Impact of Extrinsic and Intrinsic Hypoxia on Catecholamine Biosynthesis in Absence or Presence of Hif2α in Pheochromocytoma Cells
Abstract
:1. Introduction
2. Results
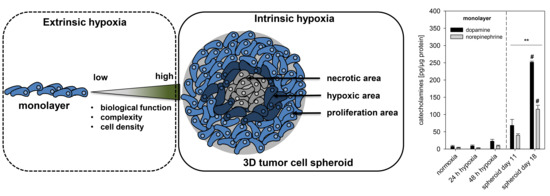
2.1. Spheroid Growth Pattern and Characteristics
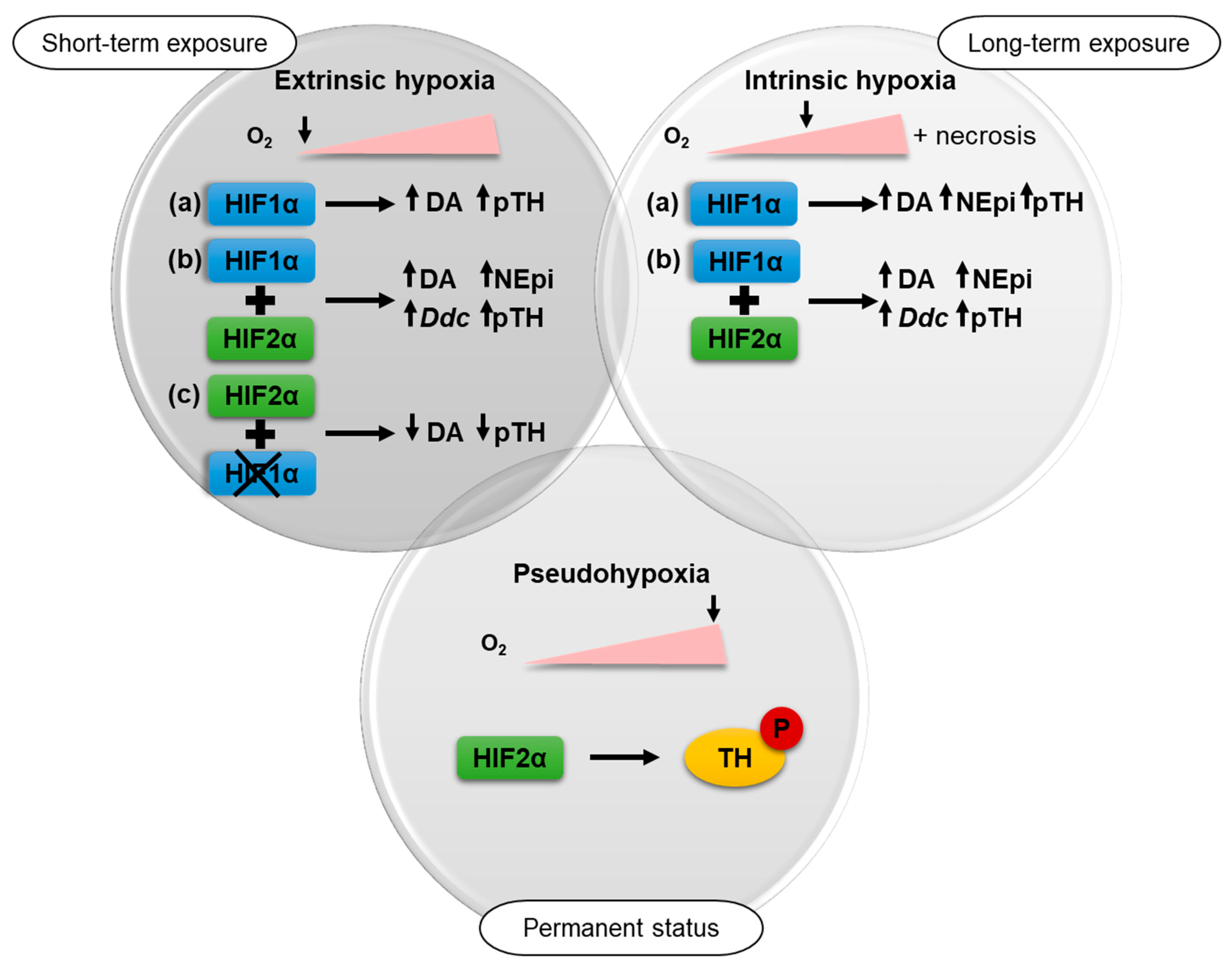
2.2. Impact of Extrinsic and Intrinsic Hypoxia on Catecholamine Biosynthesis
2.3. HIF1α- and HIF2α-Mediated Effects on Catecholamine Biosynthesis
2.4. TH Expression under Pseudohypoxic Conditions In Vitro and In Vivo
3. Discussion
4. Materials and Methods
4.1. Cell Culture
4.2. Hif2a Gene Knockin in MPC-mCherry and MTT Cells
4.3. HIF1α Gene Knockdown in PC12 Cells
4.4. Spheroid Generation
4.5. Protein Measurement
4.6. Catecholamine Measurements
4.7. Hematoxylin and Eosin Staining
4.8. Immunohistochemistry
4.9. Pimonidazole Staining
4.10. SDS-PAGE and Western Blot Analysis
4.11. Tumor Procurement and Genetic Testing
4.12. RNA Isolation and qRT-PCR
4.13. Statistical Analysis
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Fishbein, L.; Leshchiner, I.; Walter, V.; Danilova, L.; Robertson, A.G.; Johnson, A.R.; Lichtenberg, T.M.; Murray, B.A.; Ghayee, H.K.; Else, T. Comprehensive molecular characterization of pheochromocytoma and paraganglioma. Cancer Cell 2017, 31, 181–193. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Matro, J.; Giubellino, A.; Pacak, K. Current and future therapeutic approaches for metastatic pheochromocytoma and paraganglioma: Focus on sdhb tumors. Horm. Metab. Res. 2013, 45, 147–153. [Google Scholar] [CrossRef]
- Eisenhofer, G.; Pacak, K.; Huynh, T.-T.; Qin, N.; Bratslavsky, G.; Linehan, W.M.; Mannelli, M.; Friberg, P.; Grebe, S.K.; Timmers, H.J. Catecholamine metabolomic and secretory phenotypes in phaeochromocytoma. Endocr. Relat. Cancer 2011, 18, 97–111. [Google Scholar] [CrossRef] [PubMed]
- Eisenhofer, G.; Huynh, T.; Pacak, K.; Brouwers, F.; Walther, M.; Linehan, W.; Munson, P.; Mannelli, M.; Goldstein, D.; Elkahloun, A. Distinct gene expression profiles in norepinephrine-and epinephrine-producing hereditary and sporadic pheochromocytomas: Activation of hypoxia-driven angiogenic pathways in von hippel–lindau syndrome. Endocr. Relat. Cancer 2004, 11, 897–911. [Google Scholar] [CrossRef]
- Jochmanová, I.; Yang, C.; Zhuang, Z.; Pacak, K. Hypoxia-inducible factor signaling in pheochromocytoma: Turning the rudder in the right direction. J. Natl. Cancer Inst. 2013, 105, 1270–1283. [Google Scholar] [CrossRef] [PubMed]
- Kaelin Jr, W.G.; Ratcliffe, P.J. Oxygen sensing by metazoans: The central role of the hif hydroxylase pathway. Mol. Cell 2008, 30, 393–402. [Google Scholar] [CrossRef]
- Burnichon, N.; Vescovo, L.; Amar, L.; Libé, R.; de Reynies, A.; Venisse, A.; Jouanno, E.; Laurendeau, I.; Parfait, B.; Bertherat, J. Integrative genomic analysis reveals somatic mutations in pheochromocytoma and paraganglioma. Hum. Mol. Genet. 2011, 20, 3974–3985. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- López-Jiménez, E.; Gómez-López, G.; Leandro-García, L.J.; Muñoz, I.; Schiavi, F.; Montero-Conde, C.; De Cubas, A.A.; Ramires, R.; Landa, I.; Leskelä, S. Research resource: Transcriptional profiling reveals different pseudohypoxic signatures in sdhb and vhl-related pheochromocytomas. Mol. Endocrinol. 2010, 24, 2382–2391. [Google Scholar] [CrossRef]
- Favier, J.; Brière, J.-J.; Burnichon, N.; Rivière, J.; Vescovo, L.; Benit, P.; Giscos-Douriez, I.; De Reyniès, A.; Bertherat, J.; Badoual, C. The warburg effect is genetically determined in inherited pheochromocytomas. PloS ONE 2009, 4, e7094. [Google Scholar] [CrossRef] [PubMed]
- Richter, S.; Qin, N.; Pacak, K.; Eisenhofer, G. Role of hypoxia and hif2α in development of the sympathoadrenal cell lineage and chromaffin cell tumours with distinct catecholamine phenotypic features. Adv. Pharmacol. 2013, 68, 285–317. [Google Scholar] [CrossRef] [PubMed]
- Schnell, P.O.; Ignacak, M.L.; Bauer, A.L.; Striet, J.B.; Paulding, W.R.; Czyzyk-Krzeska, M.F. Regulation of tyrosine hydroxylase promoter activity by the von hippel–lindau tumor suppressor protein and hypoxia-inducible transcription factors. J. Neurochem. 2003, 85, 483–491. [Google Scholar] [CrossRef] [PubMed]
- Fukuda, T.; Ishii, K.; Nanmoku, T.; Isobe, K.; Kawakami, Y.; Takekoshi, K. 5-aminoimidazole-4-carboxamide-1-β-4-ribofuranoside stimulates tyrosine hydroxylase activity and catecholamine secretion by activation of amp-activated protein kinase in pc12 cells. J. Neuroendocrinol. 2007, 19, 621–631. [Google Scholar] [CrossRef] [PubMed]
- Dunkley, P.R.; Bobrovskaya, L.; Graham, M.E.; Von Nagy-Felsobuki, E.I.; Dickson, P.W. Tyrosine hydroxylase phosphorylation: Regulation and consequences. J Neurochem 2004, 91, 1025–1043. [Google Scholar] [CrossRef]
- Brown, S.T.; Kelly, K.F.; Daniel, J.M.; Nurse, C.A. Hypoxia inducible factor (hif)-2α is required for the development of the catecholaminergic phenotype of sympathoadrenal cells. J. Neurochem. 2009, 110, 622–630. [Google Scholar] [CrossRef] [PubMed]
- Park, S.Y.; Kang, J.H.; Jeong, K.J.; Lee, J.; Han, J.W.; Choi, W.S.; Kim, Y.K.; Kang, J.; Park, C.G.; Lee, H.Y. Norepinephrine induces vegf expression and angiogenesis by a hypoxia-inducible factor-1α protein-dependent mechanism. Int. J. Cancer 2011, 128, 2306–2316. [Google Scholar] [CrossRef] [PubMed]
- Evinger, M.J.; Cikos, S.; NWAFOR-ANENE, V.; Powers, J.F.; Tischler, A.S. Hypoxia activates multiple transcriptional pathways in mouse pheochromocytoma cells. Ann. N. Y. Acad. Sci. 2002, 971, 61–65. [Google Scholar] [CrossRef] [PubMed]
- Wong, D.L.; Tai, T.; Wong-Faull, D.C.; Claycomb, R.; Siddall, B.J.; Bell, R.A.; Kvetnansky, R. Stress and adrenergic function: Hif1α, a potential regulatory switch. Cell. Mol. Neurobiol. 2010, 30, 1451–1457. [Google Scholar] [CrossRef]
- Qin, N.; De Cubas, A.A.; Garcia-Martin, R.; Richter, S.; Peitzsch, M.; Menschikowski, M.; Lenders, J.W.; Timmers, H.J.; Mannelli, M.; Opocher, G. Opposing effects of hif1α and hif2α on chromaffin cell phenotypic features and tumor cell proliferation: Insights from myc-associated factor x. Int. J. Cancer 2014, 135, 2054–2064. [Google Scholar] [CrossRef] [PubMed]
- Pietras, A.; Johnsson, A.S.; Påhlman, S. The hif-2α-driven pseudo-hypoxic phenotype in tumor aggressiveness, differentiation, and vascularization. Curr. Top. Microbiol. Immunol. 2010, 345, 1–20. [Google Scholar] [CrossRef] [PubMed]
- Holmquist-Mengelbier, L.; Fredlund, E.; Löfstedt, T.; Noguera, R.; Navarro, S.; Nilsson, H.; Pietras, A.; Vallon-Christersson, J.; Borg, Å.; Gradin, K.; et al. Recruitment of hif-1α and hif-2α to common target genes is differentially regulated in neuroblastoma: Hif-2α promotes an aggressive phenotype. Cancer Cell 2006, 10, 413–423. [Google Scholar] [CrossRef]
- Chatzinikolaidou, M. Cell spheroids: The new frontiers in in vitro models for cancer drug validation. Drug Discov. Today 2016, 21, 1553–1560. [Google Scholar] [CrossRef] [PubMed]
- Seifert, V.; Liers, J.; Kniess, T.; Richter, S.; Bechmann, N.; Feldmann, A.; Bachmann, M.; Eisenhofer, G.; Pietzsch, J.; Ullrich, M. Fluorescent mouse pheochromocytoma spheroids expressing hypoxia-inducible factor 2 alpha: Morphologic and radiopharmacologic characterization. J. Cell. Biotechnol. 2019, in press. [Google Scholar]
- D’Antongiovanni, V.; Martinelli, S.; Richter, S.; Canu, L.; Guasti, D.; Mello, T.; Romagnoli, P.; Pacak, K.; Eisenhofer, G.; Mannelli, M. The microenvironment induces collective migration in sdhb-silenced mouse pheochromocytoma spheroids. Endocr. Relat. Cancer 2017, 24, 555–564. [Google Scholar] [CrossRef] [PubMed]
- Powers, J.; Evinger, M.; Tsokas, P.; Bedri, S.; Alroy, J.; Shahsavari, M.; Tischler, A. Pheochromocytoma cell lines from heterozygous neurofibromatosis knockout mice. Cell Tissue Res. 2000, 302, 309–320. [Google Scholar] [CrossRef]
- Martiniova, L.; Lai, E.W.; Elkahloun, A.G.; Abu-Asab, M.; Wickremasinghe, A.; Solis, D.C.; Perera, S.M.; Huynh, T.-T.; Lubensky, I.A.; Tischler, A.S. Characterization of an animal model of aggressive metastatic pheochromocytoma linked to a specific gene signature. Clin. Exp. Metastasis 2009, 26, 239–250. [Google Scholar] [CrossRef] [Green Version]
- Axelson, H.; Fredlund, E.; Ovenberger, M.; Landberg, G.; Påhlman, S. Hypoxia-induced dedifferentiation of tumor cells--a mechanism behind heterogeneity and aggressiveness of solid tumors. Semin. Cell Dev. Biol. 2005, 16, 554–563. [Google Scholar] [CrossRef] [PubMed]
- Edsjö, A.; Holmquist, L.; Påhlman, S. Neuroblastoma as an experimental model for neuronal differentiation and hypoxia-induced tumor cell dedifferentiation. Semin. Cancer Biol. 2007, 17, 248–256. [Google Scholar] [CrossRef] [PubMed]
- Bechmann, N.; Ehrlich, H.; Eisenhofer, G.; Ehrlich, A.; Meschke, S.; Ziegler, C.G.; Bornstein, S.R. Anti-tumorigenic and anti-metastatic activity of the sponge-derived marine drugs aeroplysinin-1 and isofistularin-3 against pheochromocytoma in vitro. Mar. Drugs 2018, 16, 172. [Google Scholar] [CrossRef]
- Verduzco, D.; Lloyd, M.; Xu, L.; Ibrahim-Hashim, A.; Balagurunathan, Y.; Gatenby, R.A.; Gillies, R.J. Intermittent hypoxia selects for genotypes and phenotypes that increase survival, invasion, and therapy resistance. PloS ONE 2015, 10, e0120958. [Google Scholar] [CrossRef]
- Muz, B.; de la Puente, P.; Azab, F.; Azab, A.K. The role of hypoxia in cancer progression, angiogenesis, metastasis, and resistance to therapy. Hypoxia 2015, 3, 83. [Google Scholar] [CrossRef]
- Carlsson, J.; Yuhas, J. Liquid-overlay culture of cellular spheroids. Recent Results Cancer Res. 1984, 95, 1–23. [Google Scholar] [PubMed]
- Friedrich, J.; Seidel, C.; Ebner, R.; Kunz-Schughart, L.A. Spheroid-based drug screen: Considerations and practical approach. Nat Protoc 2009, 4, 309. [Google Scholar] [CrossRef] [PubMed]
- Kunz-Schughart, L.A.; Freyer, J.P.; Hofstaedter, F.; Ebner, R. The use of 3-d cultures for high-throughput screening: The multicellular spheroid model. J. Biomol. Screen. 2004, 9, 273–285. [Google Scholar] [CrossRef] [PubMed]
- Augustin, H.G. Methods in Endothelial Cell Biology. Springer Science & Business Media. 2004. Available online: https://www.springer.com/gb/book/9783540213970 (accessed on 12 January 2019).
- Goldberg, M.; Zhang, H.L.; Steinberg, S.F. Hypoxia alters the subcellular distribution of protein kinase c isoforms in neonatal rat ventricular myocytes. J. Clin. Invest. 1997, 99, 55–61. [Google Scholar] [CrossRef] [PubMed]
- Kuri, B.A.; Khan, S.A.; Chan, S.A.; Prabhakar, N.R.; Smith, C.B. Increased secretory capacity of mouse adrenal chromaffin cells by chronic intermittent hypoxia: Involvement of protein kinase c. J. Physiol. 2007, 584, 313–319. [Google Scholar] [CrossRef]
- Kumar, G.K.; Kim, D.-K.; Lee, M.-S.; Ramachandran, R.; Prabhakar, N.R. Activation of tyrosine hydroxylase by intermittent hypoxia: Involvement of serine phosphorylation. J. Appl. Physiol. 2003. [Google Scholar] [CrossRef]
- Lee, J.W.; Park, J.A.; Kim, S.H.; Seo, J.H.; Lim, K.J.; Jeong, J.W.; Jeong, C.H.; Chun, K.H.; Lee, S.K.; Kwon, Y.G. Protein kinase c-δ regulates the stability of hypoxia-inducible factor-1α under hypoxia. Cancer Sci. 2007, 98, 1476–1481. [Google Scholar] [CrossRef] [PubMed]
- Torii, S.; Okamura, N.; Suzuki, Y.; Ishizawa, T.; Yasumoto, K.-i.; Sogawa, K. Cyclic amp represses the hypoxic induction of hypoxia-inducible factors in pc12 cells. J. Biochem. 2009, 146, 839–844. [Google Scholar] [CrossRef]
- Compernolle, V.; Brusselmans, K.; Franco, D.; Moorman, A.; Dewerchin, M.; Collen, D.; Carmeliet, P. Cardia bifida, defective heart development and abnormal neural crest migration in embryos lacking hypoxia-inducible factor-1α. Cardiovasc. Res. 2003, 60, 569–579. [Google Scholar] [CrossRef] [Green Version]
- Hu, C.-J.; Wang, L.-Y.; Chodosh, L.A.; Keith, B.; Simon, M.C. Differential roles of hypoxia-inducible factor 1α (hif-1α) and hif-2α in hypoxic gene regulation. Mol. Cell. Biol. 2003, 23, 9361–9374. [Google Scholar] [CrossRef]
- Toledo, R.A. New hif2α inhibitors: Potential implications as therapeutics for advanced pheochromocytomas and paragangliomas. Endoc. Relat. Cancer 2017, 24, C9–C19. [Google Scholar] [CrossRef] [PubMed]
- Tischler, A.S.; Greene, L.A.; Kwan, P.W.; Slayton, V.W. Ultrastructural effects of nerve growth factor on pc 12 pheochromocytoma cells in spinner culture. Cell Tissue Res. 1983, 228, 641–648. [Google Scholar] [CrossRef] [PubMed]
- Ullrich, M.; Bergmann, R.; Peitzsch, M.; Zenker, E.F.; Cartellieri, M.; Bachmann, M.; Ehrhart-Bornstein, M.; Block, N.L.; Schally, A.V.; Eisenhofer, G.; et al. Multimodal somatostatin receptor theranostics using [(64)cu]cu-/[(177)lu]lu-dota-(tyr(3))octreotate and an-238 in a mouse pheochromocytoma model. Theranostics 2016, 6, 650–665. [Google Scholar] [CrossRef] [PubMed]
- Ullrich, M.; Bergmann, R.; Pietzsch, J.; Ehrhart-Bornstein, M.; Bornstein, S.R.; Ziegler, C.G.; Eisenhofer, G.; Qin, N.; Peitzsch, M.; Cartellieri, M.; et al. In vivo fluorescence imaging and urinary monoamines as surrogate biomarkers of disease progression in a mouse model of pheochromocytoma. Endocrinology 2014, 155, 4149–4156. [Google Scholar] [CrossRef] [PubMed]
- Richter, S.; D’Antongiovanni, V.; Martinelli, S.; Bechmann, N.; Riverso, M.; Poitz, D.M.; Pacak, K.; Eisenhofer, G.; Mannelli, M.; Rapizzi, E. Primary fibroblast co-culture stimulates growth and metabolism in sdhb-impaired mouse pheochromocytoma mtt cells. Cell Tissue Res. 2018, 374, 473–485. [Google Scholar] [CrossRef] [PubMed]
- Eisenhofer, G.; Goldstein, D.S.; Stull, R.; Keiser, H.R.; Sunderland, T.; Murphy, D.L.; Kopin, I.J. Simultaneous liquid-chromatographic determination of 3,4-dihydroxyphenylglycol, catecholamines, and 3,4-dihydroxyphenylalanine in plasma, and their responses to inhibition of monoamine oxidase. Clin. Chem. 1986, 32, 2030–2033. [Google Scholar] [PubMed]
- Richter, S.; Gieldon, L.; Pang, Y.; Peitzsch, M.; Huynh, T.; Leton, R.; Viana, B.; Ercolino, T.; Mangelis, A.; Rapizzi, E.; et al. Metabolome-guided genomics to identify pathogenic variants in isocitrate dehydrogenase, fumarate hydratase, and succinate dehydrogenase genes in pheochromocytoma and paraganglioma. Genet. Med. 2019, 21, 705–717. [Google Scholar] [CrossRef] [PubMed]






© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bechmann, N.; Poser, I.; Seifert, V.; Greunke, C.; Ullrich, M.; Qin, N.; Walch, A.; Peitzsch, M.; Robledo, M.; Pacak, K.; et al. Impact of Extrinsic and Intrinsic Hypoxia on Catecholamine Biosynthesis in Absence or Presence of Hif2α in Pheochromocytoma Cells. Cancers 2019, 11, 594. https://doi.org/10.3390/cancers11050594
Bechmann N, Poser I, Seifert V, Greunke C, Ullrich M, Qin N, Walch A, Peitzsch M, Robledo M, Pacak K, et al. Impact of Extrinsic and Intrinsic Hypoxia on Catecholamine Biosynthesis in Absence or Presence of Hif2α in Pheochromocytoma Cells. Cancers. 2019; 11(5):594. https://doi.org/10.3390/cancers11050594
Chicago/Turabian StyleBechmann, Nicole, Isabel Poser, Verena Seifert, Christian Greunke, Martin Ullrich, Nan Qin, Axel Walch, Mirko Peitzsch, Mercedes Robledo, Karel Pacak, and et al. 2019. "Impact of Extrinsic and Intrinsic Hypoxia on Catecholamine Biosynthesis in Absence or Presence of Hif2α in Pheochromocytoma Cells" Cancers 11, no. 5: 594. https://doi.org/10.3390/cancers11050594