Molecular Comparison of Imatinib-Naïve and Resistant Gastrointestinal Stromal Tumors: Differentially Expressed microRNAs and mRNAs
Abstract
:1. Introduction
2. Results
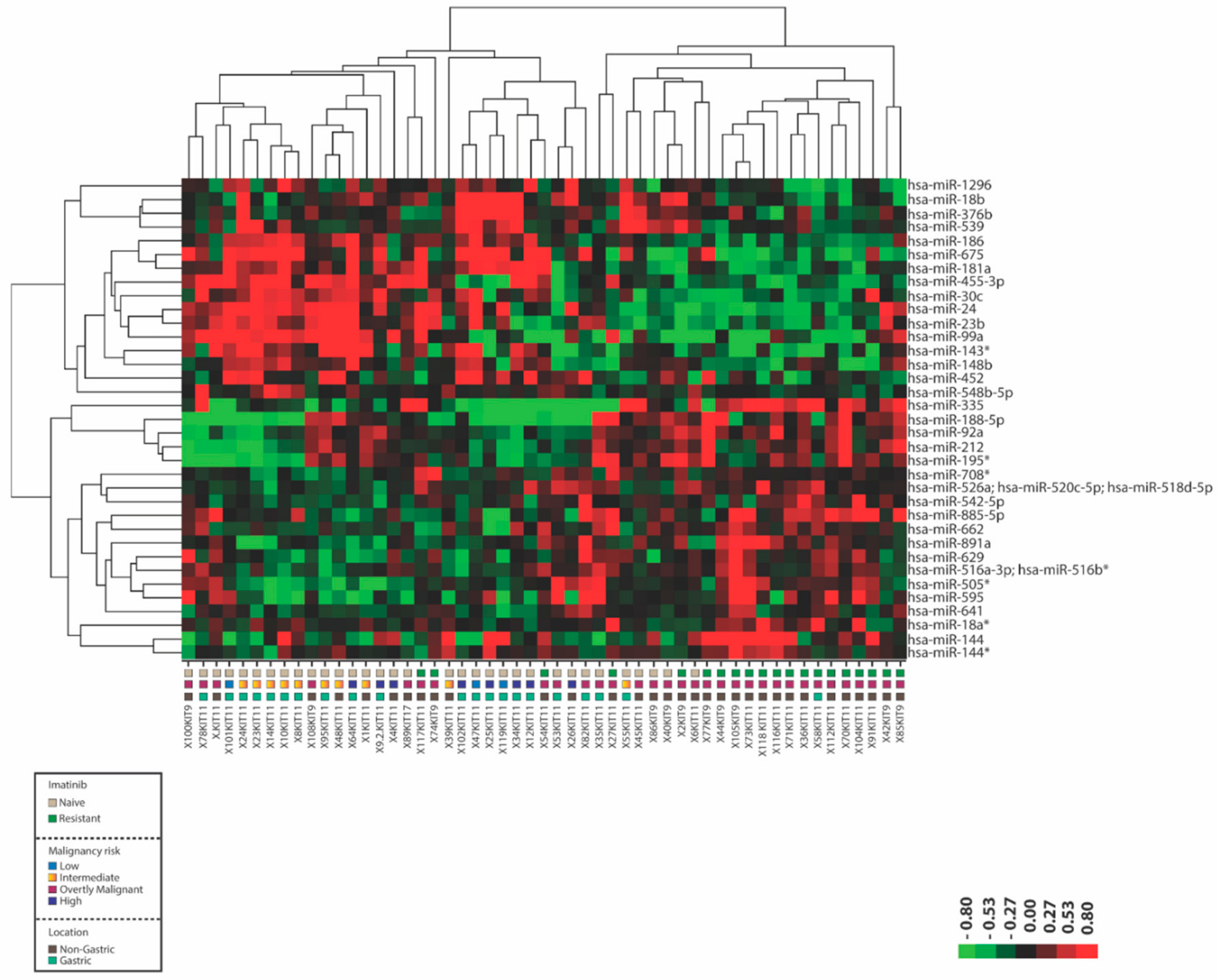
2.1. Differentially Expressed microRNAs between Imatinib-Naïve and Imatinib-Resistant GIST Samples
2.2. mRNA Expression Profiling and Ingenuity Pathway Analyses Reveal Differentially Expressed Genes and Pathways in Imatinib-Naïve and Imatinib-Resistant GIST Samples
2.3. Integration of Differentially Expressed microRNAs and mRNAs into Networks
3. Discussion
4. Materials and Methods
4.1. Patient Samples
4.2. RNA Isolation and microRNA Profiling
4.3. mRNA Expression Analysis
4.4. Quantitative RT-PCR
4.5. Pathway Analysis
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Kindblom, L.G.; Remotti, H.E.; Aldenborg, F.; Meis-Kindblom, J.M. Gastrointestinal pacemaker cell tumor (gipact): Gastrointestinal stromal tumors show phenotypic characteristics of the interstitial cells of cajal. Am. J. Pathol. 1998, 152, 1259–1269. [Google Scholar] [PubMed]
- Heinrich, M.C.; Corless, C.L.; Demetri, G.D.; Blanke, C.D.; von Mehren, M.; Joensuu, H.; McGreevey, L.S.; Chen, C.J.; Van den Abbeele, A.D.; Druker, B.J.; et al. Kinase mutations and imatinib response in patients with metastatic gastrointestinal stromal tumor. J. Clin. Oncol. 2003, 21, 4342–4349. [Google Scholar] [CrossRef]
- Hirota, S.; Isozaki, K.; Moriyama, Y.; Hashimoto, K.; Nishida, T.; Ishiguro, S.; Kawano, K.; Hanada, M.; Kurata, A.; Takeda, M.; et al. Gain-of-function mutations of c-kit in human gastrointestinal stromal tumors. Science 1998, 279, 577–580. [Google Scholar] [CrossRef]
- Ricci, R.; Dei Tos, A.P.; Rindi, G. Gistogram: A graphic presentation of the growing gist complexity. Virchows Arch. 2013, 463, 481–487. [Google Scholar] [CrossRef] [PubMed]
- Corless, C.L.; Barnett, C.M.; Heinrich, M.C. Gastrointestinal stromal tumours: Origin and molecular oncology. Nat. Rev. Cancer 2011, 11, 865–878. [Google Scholar] [CrossRef] [PubMed]
- Blanke, C.D.; Demetri, G.D.; von Mehren, M.; Heinrich, M.C.; Eisenberg, B.; Fletcher, J.A.; Corless, C.L.; Fletcher, C.D.; Roberts, P.J.; Heinz, D.; et al. Long-term results from a randomized phase ii trial of standard- versus higher-dose imatinib mesylate for patients with unresectable or metastatic gastrointestinal stromal tumors expressing kit. J. Clin. Oncol. 2008, 26, 620–625. [Google Scholar] [CrossRef] [PubMed]
- Verweij, J.; Casali, P.G.; Zalcberg, J.; LeCesne, A.; Reichardt, P.; Blay, J.Y.; Issels, R.; van Oosterom, A.; Hogendoorn, P.C.; Van Glabbeke, M.; et al. Progression-free survival in gastrointestinal stromal tumours with high-dose imatinib: Randomised trial. Lancet 2004, 364, 1127–1134. [Google Scholar] [CrossRef]
- Casali, P.G.; Zalcberg, J.; Le Cesne, A.; Reichardt, P.; Blay, J.Y.; Lindner, L.H.; Judson, I.R.; Schoffski, P.; Leyvraz, S.; Italiano, A.; et al. Ten-year progression-free and overall survival in patients with unresectable or metastatic gi stromal tumors: Long-term analysis of the european organisation for research and treatment of cancer, italian sarcoma group, and australasian gastrointestinal trials group intergroup phase iii randomized trial on imatinib at two dose levels. J. Clin. Oncol. 2017, 35, 1713–1720. [Google Scholar]
- Debiec-Rychter, M.; Sciot, R.; Le Cesne, A.; Schlemmer, M.; Hohenberger, P.; van Oosterom, A.T.; Blay, J.Y.; Leyvraz, S.; Stul, M.; Casali, P.G.; et al. Kit mutations and dose selection for imatinib in patients with advanced gastrointestinal stromal tumours. Eur. J. Cancer 2006, 42, 1093–1103. [Google Scholar] [CrossRef]
- Antonescu, C.R.; Besmer, P.; Guo, T.; Arkun, K.; Hom, G.; Koryotowski, B.; Leversha, M.A.; Jeffrey, P.D.; Desantis, D.; Singer, S.; et al. Acquired resistance to imatinib in gastrointestinal stromal tumor occurs through secondary gene mutation. Clin. Cancer Res. 2005, 11, 4182–4190. [Google Scholar] [CrossRef]
- Debiec-Rychter, M.; Cools, J.; Dumez, H.; Sciot, R.; Stul, M.; Mentens, N.; Vranckx, H.; Wasag, B.; Prenen, H.; Roesel, J.; et al. Mechanisms of resistance to imatinib mesylate in gastrointestinal stromal tumors and activity of the pkc412 inhibitor against imatinib-resistant mutants. Gastroenterology 2005, 128, 270–279. [Google Scholar] [CrossRef] [PubMed]
- Heinrich, M.C.; Corless, C.L.; Blanke, C.D.; Demetri, G.D.; Joensuu, H.; Roberts, P.J.; Eisenberg, B.L.; von Mehren, M.; Fletcher, C.D.; Sandau, K.; et al. Molecular correlates of imatinib resistance in gastrointestinal stromal tumors. J. Clin. Oncol. 2006, 24, 4764–4774. [Google Scholar] [CrossRef]
- Liegl, B.; Kepten, I.; Le, C.; Zhu, M.; Demetri, G.D.; Heinrich, M.C.; Fletcher, C.D.; Corless, C.L.; Fletcher, J.A. Heterogeneity of kinase inhibitor resistance mechanisms in gist. J. Pathol. 2008, 216, 64–74. [Google Scholar] [CrossRef] [PubMed]
- Wardelmann, E.; Merkelbach-Bruse, S.; Pauls, K.; Thomas, N.; Schildhaus, H.U.; Heinicke, T.; Speidel, N.; Pietsch, T.; Buettner, R.; Pink, D.; et al. Polyclonal evolution of multiple secondary kit mutations in gastrointestinal stromal tumors under treatment with imatinib mesylate. Clin. Cancer Res. 2006, 12, 1743–1749. [Google Scholar] [CrossRef]
- Mahadevan, D.; Cooke, L.; Riley, C.; Swart, R.; Simons, B.; Della Croce, K.; Wisner, L.; Iorio, M.; Shakalya, K.; Garewal, H. A novel tyrosine kinase switch is a mechanism of imatinib resistance in gastrointestinal stromal tumors. Oncogene 2007, 26, 3909–3919. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sakurama, K.; Noma, K.; Takaoka, M.; Tomono, Y.; Watanabe, N.; Hatakeyama, S.; Ohmori, O.; Hirota, S.; Motoki, T.; Shirakawa, Y.; et al. Inhibition of focal adhesion kinase as a potential therapeutic strategy for imatinib-resistant gastrointestinal stromal tumor. Mol. Cancer Ther. 2009, 8, 127–134. [Google Scholar] [CrossRef]
- Takahashi, T.; Serada, S.; Ako, M.; Fujimoto, M.; Miyazaki, Y.; Nakatsuka, R.; Ikezoe, T.; Yokoyama, A.; Taguchi, T.; Shimada, K.; et al. New findings of kinase switching in gastrointestinal stromal tumor under imatinib using phosphoproteomic analysis. Int. J. Cancer 2013, 133, 2737–2743. [Google Scholar] [CrossRef]
- Gits, C.M.; van Kuijk, P.F.; Jonkers, M.B.; Boersma, A.W.; van IJcken, W.F.; Wozniak, A.; Sciot, R.; Rutkowski, P.; Schoffski, P.; Taguchi, T.; et al. Mir-17-92 and mir-221/222 cluster members target kit and etv1 in human gastrointestinal stromal tumours. Br. J. Cancer 2013, 109, 1625–1635. [Google Scholar] [CrossRef]
- Yun, S.; Kim, W.K.; Kwon, Y.; Jang, M.; Bauer, S.; Kim, H. Survivin is a novel transcription regulator of kit and is downregulated by mirna-494 in gastrointestinal stromal tumors. Int. J. Cancer 2018, 142, 2080–2093. [Google Scholar] [CrossRef]
- Fanini, F.; Fabbri, M. Micrornas and cancer resistance: A new molecular plot. Clin. Pharmacol. Ther. 2016, 99, 485–493. [Google Scholar] [CrossRef]
- Calin, G.A.; Croce, C.M. Microrna-cancer connection: The beginning of a new tale. Cancer Res. 2006, 66, 7390–7394. [Google Scholar] [CrossRef]
- Xu, J.; Li, C.X.; Li, Y.S.; Lv, J.Y.; Ma, Y.; Shao, T.T.; Xu, L.D.; Wang, Y.Y.; Du, L.; Zhang, Y.P.; et al. Mirna-mirna synergistic network: Construction via co-regulating functional modules and disease mirna topological features. Nucleic Acids Res. 2011, 39, 825–836. [Google Scholar] [CrossRef] [PubMed]
- Akcakaya, P.; Caramuta, S.; Ahlen, J.; Ghaderi, M.; Berglund, E.; Ostman, A.; Branstrom, R.; Larsson, C.; Lui, W.O. Microrna expression signatures of gastrointestinal stromal tumours: Associations with imatinib resistance and patient outcome. Br. J. Cancer 2014, 111, 2091–2102. [Google Scholar] [CrossRef] [PubMed]
- Shi, Y.; Gao, X.; Hu, Q.; Li, X.; Xu, J.; Lu, S.; Liu, Y.; Xu, C.; Jiang, D.; Lin, J.; et al. Pik3c2a is a gene-specific target of microrna-518a-5p in imatinib mesylate-resistant gastrointestinal stromal tumor. Lab. Investig. 2016, 96, 652–660. [Google Scholar] [CrossRef]
- Huang, W.K.; Akcakaya, P.; Gangaev, A.; Lee, L.; Zeljic, K.; Hajeri, P.; Berglund, E.; Ghaderi, M.; Ahlen, J.; Branstrom, R.; et al. Mir-125a-5p regulation increases phosphorylation of fak that contributes to imatinib resistance in gastrointestinal stromal tumors. Exp. Cell. Res. 2018, 371, 287–296. [Google Scholar] [CrossRef]
- Farhadi, E.; Zaker, F.; Safa, M.; Rezvani, M.R. Mir-101 sensitizes k562 cell line to imatinib through jak2 downregulation and inhibition of nf-kappab target genes. Tumour Biol. 2016, 37, 14117–14128. [Google Scholar] [CrossRef] [PubMed]
- San Jose-Eneriz, E.; Roman-Gomez, J.; Jimenez-Velasco, A.; Garate, L.; Martin, V.; Cordeu, L.; Vilas-Zornoza, A.; Rodriguez-Otero, P.; Calasanz, M.J.; Prosper, F.; et al. Microrna expression profiling in imatinib-resistant chronic myeloid leukemia patients without clinically significant abl1-mutations. Mol. Cancer 2009, 8, 69. [Google Scholar] [CrossRef]
- Zimmerman, E.I.; Dollins, C.M.; Crawford, M.; Grant, S.; Nana-Sinkam, S.P.; Richards, K.L.; Hammond, S.M.; Graves, L.M. Lyn kinase-dependent regulation of mir181 and myeloid cell leukemia-1 expression: Implications for drug resistance in myelogenous leukemia. Mol. Pharmacol. 2010, 78, 811–817. [Google Scholar] [CrossRef]
- Gao, T.; Han, Y.; Yu, L.; Ao, S.; Li, Z.; Ji, J. Ccna2 is a prognostic biomarker for er+ breast cancer and tamoxifen resistance. PLoS ONE 2014, 9, e91771. [Google Scholar] [CrossRef]
- Bae, T.; Weon, K.Y.; Lee, J.W.; Eum, K.H.; Kim, S.; Choi, J.W. Restoration of paclitaxel resistance by cdk1 intervention in drug-resistant ovarian cancer. Carcinogenesis 2015, 36, 1561–1571. [Google Scholar] [CrossRef]
- Coley, H.M.; Safuwan, N.A.; Chivers, P.; Papacharalbous, E.; Giannopoulos, T.; Butler-Manuel, S.; Madhuri, K.; Lovell, D.P.; Crook, T. The cyclin-dependent kinase inhibitor p57 (kip2) is epigenetically regulated in carboplatin resistance and results in collateral sensitivity to the cdk inhibitor seliciclib in ovarian cancer. Br. J. Cancer 2012, 106, 482–489. [Google Scholar] [CrossRef] [PubMed]
- Chu, J.; Zhu, Y.; Liu, Y.; Sun, L.; Lv, X.; Wu, Y.; Hu, P.; Su, F.; Gong, C.; Song, E.; et al. E2f7 overexpression leads to tamoxifen resistance in breast cancer cells by competing with e2f1 at mir-15a/16 promoter. Oncotarget 2015, 6, 31944–31957. [Google Scholar] [CrossRef] [PubMed]
- Saenz-Ponce, N.; Pillay, R.; de Long, L.M.; Kashyap, T.; Argueta, C.; Landesman, Y.; Hazar-Rethinam, M.; Boros, S.; Panizza, B.; Jacquemyn, M.; et al. Targeting the xpo1-dependent nuclear export of e2f7 reverses anthracycline resistance in head and neck squamous cell carcinomas. Sci. Transl. Med. 2018, 10. [Google Scholar] [CrossRef] [PubMed]
- Ye, L.; Guo, L.; He, Z.; Wang, X.; Lin, C.; Zhang, X.; Wu, S.; Bao, Y.; Yang, Q.; Song, L.; et al. Upregulation of e2f8 promotes cell proliferation and tumorigenicity in breast cancer by modulating g1/s phase transition. Oncotarget 2016, 7, 23757–23771. [Google Scholar] [CrossRef] [PubMed]
- Tian, J.; Lin, Y.; Yu, J. E2f8 confers cisplatin resistance to er+ breast cancer cells via transcriptionally activating mastl. Biomed. Pharmacother. 2017, 92, 919–926. [Google Scholar] [CrossRef] [PubMed]
- Carvajal, R.D.; Tse, A.; Schwartz, G.K. Aurora kinases: New targets for cancer therapy. Clin. Cancer Res. 2006, 12, 6869–6875. [Google Scholar] [CrossRef] [PubMed]
- Lagarde, P.; Perot, G.; Kauffmann, A.; Brulard, C.; Dapremont, V.; Hostein, I.; Neuville, A.; Wozniak, A.; Sciot, R.; Schoffski, P.; et al. Mitotic checkpoints and chromosome instability are strong predictors of clinical outcome in gastrointestinal stromal tumors. Clin. Cancer Res. 2012, 18, 826–838. [Google Scholar] [CrossRef]
- Yeh, C.N.; Yen, C.C.; Chen, Y.Y.; Cheng, C.T.; Huang, S.C.; Chang, T.W.; Yao, F.Y.; Lin, Y.C.; Wen, Y.S.; Chiang, K.C.; et al. Identification of aurora kinase a as an unfavorable prognostic factor and potential treatment target for metastatic gastrointestinal stromal tumors. Oncotarget 2014, 5, 4071–4086. [Google Scholar] [CrossRef]
- Tsai, H.J.; Jiaang, W.T.; Shih, N.Y.; Fletcher, J.A.; Lin, M.J.; Yang, M.Y.; Chen, C.T.; Hsu, T.J.; Wu, C.C.; Lin, H.Y.; et al. Bpr1j373, a novel multitargeted kinase inhibitor, effectively suppresses the growth of gastrointestinal stromal tumor. Cancer Sci. 2018, 109, 3591–3601. [Google Scholar] [CrossRef]
- Bai, C.; Liu, X.; Qiu, C.; Zheng, J. Foxm1 is regulated by both hif-1alpha and hif-2alpha and contributes to gastrointestinal stromal tumor progression. Gastric Cancer 2019, 22, 91–103. [Google Scholar] [CrossRef]
- Nestal de Moraes, G.; Bella, L.; Zona, S.; Burton, M.J.; Lam, E.W. Insights into a critical role of the foxo3a-foxm1 axis in DNA damage response and genotoxic drug resistance. Curr. Drug. Targets 2016, 17, 164–177. [Google Scholar] [CrossRef] [PubMed]
- Li, D.; Liu, X.; Lin, L.; Hou, J.; Li, N.; Wang, C.; Wang, P.; Zhang, Q.; Zhang, P.; Zhou, W.; et al. Microrna-99a inhibits hepatocellular carcinoma growth and correlates with prognosis of patients with hepatocellular carcinoma. J. Biol. Chem. 2011, 286, 36677–36685. [Google Scholar] [CrossRef] [PubMed]
- Oneyama, C.; Ikeda, J.; Okuzaki, D.; Suzuki, K.; Kanou, T.; Shintani, Y.; Morii, E.; Okumura, M.; Aozasa, K.; Okada, M. Microrna-mediated downregulation of mtor/fgfr3 controls tumor growth induced by src-related oncogenic pathways. Oncogene 2011, 30, 3489–3501. [Google Scholar] [CrossRef] [PubMed]
- Fingar, D.C.; Richardson, C.J.; Tee, A.R.; Cheatham, L.; Tsou, C.; Blenis, J. Mtor controls cell cycle progression through its cell growth effectors s6k1 and 4e-bp1/eukaryotic translation initiation factor 4e. Mol. Cell Biol. 2004, 24, 200–216. [Google Scholar] [CrossRef] [PubMed]
- Sachdeva, M.; Wu, H.; Ru, P.; Hwang, L.; Trieu, V.; Mo, Y.Y. Microrna-101-mediated akt activation and estrogen-independent growth. Oncogene 2011, 30, 822–831. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.; He, X.; Liu, Y.; Zhang, H.; Chen, H.; Guo, S.; Liang, Y. Mir-101-3p inhibits the growth and metastasis of non-small cell lung cancer through blocking pi3k/akt signal pathway by targeting malat-1. Biomed. Pharmacother. 2017, 93, 1065–1073. [Google Scholar] [CrossRef] [PubMed]
- Zhao, R.; Yang, H.Y.; Shin, J.; Phan, L.; Fang, L.; Che, T.F.; Su, C.H.; Yeung, S.C.; Lee, M.H. Cdk inhibitor p57 (kip2) is downregulated by akt during her2-mediated tumorigenicity. Cell Cycle 2013, 12, 935–943. [Google Scholar] [CrossRef]
- Pothof, J.; Verkaik, N.S.; van IJcken, W.; Wiemer, E.A.; Ta, V.T.; van der Horst, G.T.; Jaspers, N.G.; van Gent, D.C.; Hoeijmakers, J.H.; Persengiev, S.P. Microrna-mediated gene silencing modulates the uv-induced DNA-damage response. EMBO J. 2009, 28, 2090–2099. [Google Scholar] [CrossRef]
- De Hoon, M.J.; Imoto, S.; Nolan, J.; Miyano, S. Open source clustering software. Bioinformatics 2004, 20, 1453–1454. [Google Scholar] [CrossRef] [Green Version]
- Saldanha, A.J. Java Treeview—extensible visualization of microarray data. Bioinformatics 2004, 20, 3246–3248. [Google Scholar] [CrossRef]
- Johnson, W.E.; Li, C.; Rabinovic, A. Adjusting batch effects in microarray expression data using empirical bayes methods. Biostatistics 2007, 8, 118–127. [Google Scholar] [CrossRef] [PubMed]
- McCall, M.N.; Bolstad, B.M.; Irizarry, R.A. Frozen robust multiarray analysis (frma). Biostatistics 2010, 11, 242–253. [Google Scholar] [CrossRef] [PubMed]





| Gastrointestinal Stromal Tumors Imatinib-Naïve (IM-n) | ||||||
| Male | n = 23 | |||||
| Female | n = 10 | |||||
| Median age (range) | 65 (41–85) | |||||
| Sample code | KIT mutation status | Location | Risk of malignancy * | miRNA | mRNA | |
| X1KIT11 | p.V560D/KIT11 | Small intestine | intermediate | ✓ | ✓ | |
| X4KIT11 | p.W557_V559delinsF/KIT11 | Small intestine | high | ✓ | ✓ | |
| X6KIT11 | p.W557R/KIT11 | Small intestine | overtly malignant ** | ✓ | ||
| X8KIT11 | p.L576_R588dup/KIT11 | Stomach | intermediate | ✓ | ✓ | |
| X9.2.KIT11 | p.W557_V559delinsF/KIT11 | Stomach | high | ✓ | ||
| X10KIT11 | p.W557R/KIT11 | Stomach | intermediate | ✓ | ||
| X12KIT11 | p.K550_V555del/KIT11 | Stomach | high | ✓ | ||
| X14KIT11 | p.581_590insKWEFPRNRLS/KIT11 | Stomach | intermediate | ✓ | ✓ | |
| X23KIT11 | p.W557_K558del/KIT11 | Stomach | intermediate | ✓ | ||
| X24KIT11 | p.V554D/KIT11 | Stomach | intermediate | ✓ | ✓ | |
| X25KIT11 | p.W557_G592dup (c.1669_1774 + 2dup)/KIT11 | Stomach | high | ✓ | ||
| X26KIT11 | p.K558_V559delinsN (AAT) homo/KIT11 | Mediastinum | high | ✓ | ||
| X34KIT11 | p.W557_V560delinsF/KIT11 | Stomach | high | ✓ | ||
| X35KIT11 | p.V560D/KIT11 | Stomach | overtly malignant | ✓ | ✓ | |
| X39KIT11 | p.L576P/KIT11 | Duodenum | intermediate | ✓ | ✓ | |
| X40KIT9 | p.A502_Y503dup/KIT9 | Colon | overtly malignant | ✓ | ||
| X45KIT11 | p.K550_K558delinsG/KIT11 | Small intestine | overtly malignant | ✓ | ||
| X47KIT11 | p.V559A/KIT11 | Stomach | low | ✓ | ||
| X48KIT11 | p.V560A/KIT11 | Duodenum | intermediate | ✓ | ✓ | |
| X53KIT11 | p.Q556_V559delinsH; c.1668_1676del9/KIT11 | Stomach | overtly malignant | ✓ | ✓ | |
| X55KIT11 | p.W557_K558del/KIT11 | Stomach | intermediate | ✓ | ||
| X64KIT11 | p.V560D/KIT11 | Stomach | high | ✓ | ✓ | |
| X78KIT11 | p.W557_K558del; c.1669_1674del/KIT11 | Stomach | overtly malignant | ✓ | ✓ | |
| X82KIT11 | p.W557_P573delinsFQ/KIT11 | Stomach | overtly malignant | ✓ | ||
| X86KIT9 | p.A502_Y503dup/KIT9 | Small intestine | overtly malignant | ✓ | ||
| X89KIT17 | p.N822K/KIT17 | Small intestine | overtly malignant | ✓ | ✓ | |
| X95KIT11 | p.T574_R586insK/KIT11 | Stomach | intermediate | ✓ | ||
| X100KIT9 | p.A502_Y503dup/KIT9 | Small intestine | overtly malignant | ✓ | ||
| X101KIT11 | p.E554_K558del/KIT11 | Stomach | low | ✓ | ||
| X102KIT11 | p.W557R/KIT11 | Stomach | high | ✓ | ✓ | |
| X108KIT9 | p.A502_Y503dup/KIT9 | Small intestine | overtly malignant | ✓ | ✓ | |
| X.KIT11 | p.M552_E554delinsK/KIT11 | Small intestine | overtly malignant | ✓ | ||
| X119KIT11 | p.Q556_I563del/KIT11 | Stomach | low | ✓ | ||
| Gastrointestinal Stromal Tumors Imatinib-Resistant (IM-r) *** | ||||||
| Male | n = 14 | |||||
| Female | n = 6 | |||||
| Median age (range) | 49.5 (22–67) | |||||
| Sample code | KIT mutation status | KIT secondary mutation | Location | Risk of malignancy * | miRNA | mRNA |
| X2KIT9 | p.A502_Y503dup/KIT9 | Not detected | Colon | overtly malignant | ✓ | ✓ |
| X27KIT11 | p.L576P; c.1727 T > C/27KIT11 | p.D820Y; c.2458G > T | Small intestine | overtly malignant | ✓ | ✓ |
| X36KIT11 | p.Q556_E561delinsQ/KIT11 | Not detected | Small intestine | overtly malignant | ✓ | ✓ |
| X42KIT9 | p.A502_Y503dup/KIT9 | KIT: p.V654A | Small intestine | overtly malignant | ✓ | ✓ |
| X44KIT9 | p.A502_Y503dup/KIT9 | Not detected | Small intestine | overtly malignant | ✓ | ✓ |
| X54KIT11 | p.K550_K558delinsQ/KIT11 | KIT: p.D820Y | Small intestine | overtly malignant | ✓ | ✓ |
| X58KIT11 | p.I563_Q575del/KIT11 | KIT: p.D820Y | Stomach | overtly malignant | ✓ | ✓ |
| X70KIT11 | p.E554_D572del/KIT11 | KIT: p.V654A | Small intestine | overtly malignant | ✓ | ✓ |
| X71KIT11 | p.V559D/KIT11 | KIT: p.D820G | Small intestine | overtly malignant | ✓ | ✓ |
| X73KIT11 | p.N567_L576delinsI/KIT11 | Not detected | Small intestine | overtly malignant | ✓ | ✓ |
| X74KIT9 | p.A502_Y503dup/KIT9 | Not detected | Small intestine | overtly malignant | ✓ | |
| X77KIT9 | p.A502_Y503dup/KIT9 | Not detected | Small intestine | overtly malignant | ✓ | ✓ |
| X85KIT9 | p.A502_Y503dup/KIT9 | Not detected | Small intestine | overtly malignant | ✓ | ✓ |
| X91KIT11 | p.K550_K558del/KIT11 | KIT: p.D820Y | Small intestine | overtly malignant | ✓ | |
| X104KIT11 | p.W557_K558del/KIT11 | Not detected | Small intestine | overtly malignant | ✓ | ✓ |
| X105KIT9 | p.A502_Y503dup/KIT9 | Not detected | Small intestine | overtly malignant | ✓ | ✓ |
| X112KIT11 | c.1654_1671del18 (p.M552_W557del)/KIT11 | Not detected | Small intestine | overtly malignant | ✓ | ✓ |
| X116KIT11 | p.557_558del homo/KIT11 | Not detected | Small intestine | overtly malignant | ✓ | |
| X117KIT11 | p.K550_V555delinsL; c.1648_1663delinsT/KIT11 | p.D820Y; c.2458 G > T | Small intestine | overtly malignant | ✓ | |
| X118KIT11 | p.V559D; c.1676 T > A/KIT11 | KIT p.V654A; c.1961 T > C | Small intestine | overtly malignant | ✓ | |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Amirnasr, A.; Gits, C.M.M.; van Kuijk, P.F.; Smid, M.; Vriends, A.L.M.; Rutkowski, P.; Sciot, R.; Schöffski, P.; Debiec-Rychter, M.; Sleijfer, S.; et al. Molecular Comparison of Imatinib-Naïve and Resistant Gastrointestinal Stromal Tumors: Differentially Expressed microRNAs and mRNAs. Cancers 2019, 11, 882. https://doi.org/10.3390/cancers11060882
Amirnasr A, Gits CMM, van Kuijk PF, Smid M, Vriends ALM, Rutkowski P, Sciot R, Schöffski P, Debiec-Rychter M, Sleijfer S, et al. Molecular Comparison of Imatinib-Naïve and Resistant Gastrointestinal Stromal Tumors: Differentially Expressed microRNAs and mRNAs. Cancers. 2019; 11(6):882. https://doi.org/10.3390/cancers11060882
Chicago/Turabian StyleAmirnasr, Azadeh, Caroline M.M. Gits, Patricia F. van Kuijk, Marcel Smid, Anne L.M. Vriends, Piotr Rutkowski, Raf Sciot, Patrick Schöffski, Maria Debiec-Rychter, Stefan Sleijfer, and et al. 2019. "Molecular Comparison of Imatinib-Naïve and Resistant Gastrointestinal Stromal Tumors: Differentially Expressed microRNAs and mRNAs" Cancers 11, no. 6: 882. https://doi.org/10.3390/cancers11060882






