In-Vitro and In-Vivo Establishment and Characterization of Bioluminescent Orthotopic Chemotherapy-Resistant Human Osteosarcoma Models in NSG Mice
Abstract
:1. Introduction
2. Results
2.1. Development of In-Vitro Osteosarcoma Cell Lines Resistant to Chemotherapy
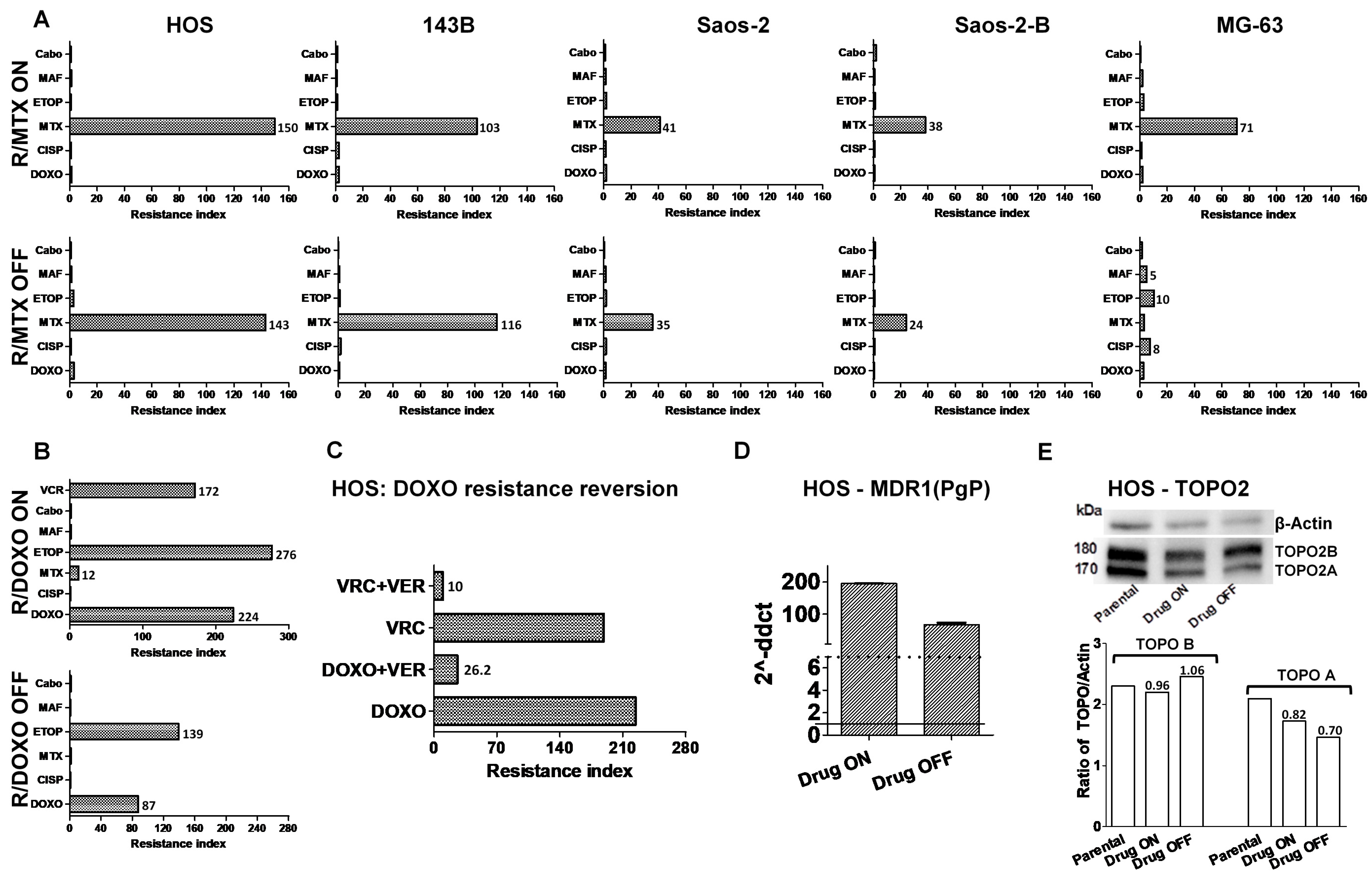
2.2. Cross-Resistance to Other Drugs
2.3. Copy Number and Gene Expression Differential Analysis between Resistant and Parental Lines
2.4. In-Vivo Primary Tumor Characteristics of HOS and Saos-2-B Bioluminescent Orthotopic Parental and MTX and DOXO Resistant-CDX Models
2.5. In-Vivo Metastatic Behavior of the Resistant Orthotopic Bioluminescent-CDX Models
2.6. In-Vitro Secondary Cultures Issued from CDX Models
3. Discussion
4. Materials and Methods
4.1. Cells Culture
4.2. Compounds
4.3. In-Vitro Development of Chemo-Resistant Osteosarcoma Cell Lines
4.4. In-Vitro Cell Proliferation and Cell Viability Assays
4.5. Wound-Healing Assay
4.6. Transfection and Cell Transduction With Luc/Mkate2 (Transgene) In-Vitro
4.7. Orthotopic Bioluminescent CDX Models
4.8. In-Vivo and Ex-Vivo Computed Tomography (CT) Scan and Bioluminescence (BLI) Imaging
4.9. Histological Analysis and Immunohistochemistry (IHC)
4.10. Nucleic Acid Extraction
4.11. Oligonucleotide Comparative Genetic Hybridization Array (aCGH) Assay
4.12. RNA Sequencing (Rnaseq)
4.13. Reverse Transcription-Quantitative PCR (RT-qPCR)
4.14. Western-Blot (WB)
4.15. Statistical Analysis
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Yang, X.; Yang, P.; Shen, J.; Osaka, E.; Choy, E.; Cote, G.; Harmon, D.; Zhang, Z.; Mankin, H.; Hornicek, F.J.; et al. Prevention of multidrug resistance (MDR) in osteosarcoma by NSC23925. Br. J. Cancer 2014, 110, 2896–2904. [Google Scholar] [CrossRef]
- Wang, J.; Li, G. Mechanisms of methotrexate resistance in osteosarcoma cell lines and strategies for overcoming this resistance. Oncol. Lett. 2015, 9, 940–944. [Google Scholar] [CrossRef]
- PosthumaDeBoer, J.; van Royen, B.J.; Helder, M.N. Mechanisms of therapy resistance in osteosarcoma: A review. Oncol. Discov. 2013, 1, 8. [Google Scholar] [CrossRef]
- Duan, Z.; Gao, Y.; Shen, J.; Choy, E.; Cote, G.; Harmon, D.; Bernstein, K.; Lozano-Calderon, S.; Mankin, H.; Hornicek, F.J. miR-15b modulates multidrug resistance in human osteosarcoma in vitro and in vivo. Mol. Oncol. 2017, 11, 151–166. [Google Scholar] [CrossRef]
- Bielack, S.S.; Smeland, S.; Whelan, J.S.; Marina, N.; Jovic, G.; Hook, J.M.; Krailo, M.D.; Gebhardt, M.; Pápai, Z.; Meyer, J.; et al. Methotrexate, doxorubicin, and cisplatin (MAP) plus maintenance pegylated interferon Alfa-2b versus MAP alone in patients with resectable high-grade osteosarcoma and good histologic response to preoperative MAP: First results of the EURAMOS-1 good response randomized controlled trial. J. Clin. Oncol. 2015, 33, 2279–2287. [Google Scholar]
- He, H.; Ni, J.; Huang, J. Molecular mechanisms of chemoresistance in osteosarcoma (Review). Oncol. Lett. 2014, 7, 1352–1362. [Google Scholar] [CrossRef] [Green Version]
- Both, J.; Krijgsman, O.; Bras, J.; Schaap, G.R.; Baas, F.; Ylstra, B.; Hulsebos, T.J.M. Focal chromosomal copy number aberrations identify CMTM8 and GPR177 as new candidate driver genes in osteosarcoma. PLoS ONE 2014, 9, 115835. [Google Scholar] [CrossRef]
- Gordon, N.; Kleinerman, E.S. The role of Fas/FasL in the metastatic potential of osteosarcoma and targeting this pathway for the treatment of osteosarcoma lung metastases. Hematop. Growth Factors Oncol. 2009, 152, 497–508. [Google Scholar]
- Choi, Y.J.; Lee, Y.S.; Lee, H.W.; Shim, D.M.; Seo, S.W. Silencing of translation initiation factor eIF3b promotes apoptosis in osteosarcoma cells. Bone Jt. Res. 2017, 6, 186–193. [Google Scholar] [CrossRef]
- Capobianco, E.; Mora, A.; La Sala, D.; Roberti, A.; Zaki, N.; Badidi, E.; Taranta, M.; Cinti, C. Separate and Combined Effects of DNMT and HDAC Inhibitors in Treating Human Multi-Drug Resistant Osteosarcoma HosDXR150 Cell Line. PLoS ONE 2014, 9, e95596. [Google Scholar] [CrossRef]
- Gu, P.; Deng, W.; Lei, M.; Chang, S. Single strand DNA binding proteins 1 and 2 protect newly replicated telomeres. Cell Res. 2013, 23, 705–719. [Google Scholar] [CrossRef] [Green Version]
- Di Fiore, R.; Fanale, D.; Drago-Ferrante, R.; Chiaradonna, F.; Giuliano, M.; De Blasio, A.; Amodeo, V.; Corsini, L.R.; Bazan, V.; Tesoriere, G.; et al. Genetic and molecular characterization of the human Osteosarcoma 3AB-OS cancer stem cell line: A possible model for studying osteosarcoma origin and stemness. J. Cell. Physiol. 2013, 228, 1189–1201. [Google Scholar] [CrossRef]
- Li, Y.; Meng, G.; Guo, Q.-N. Changes in genomic imprinting and gene expression associated with transformation in a model of human osteosarcoma. Exp. Mol. Pathol. 2008, 84, 234–239. [Google Scholar] [CrossRef]
- Cavalcanti, A.D.S.; Meohas, W.; Ribeiro, G.D.O.; Lopes, A.C.D.S.; Gholamin, S.; Razavi, M.; Brunswick, T.H.K.; Avan, A.; Guimarães, J.A.M.; Duarte, M.E.L.; et al. Patient-derived osteosarcoma cells are resistant to methotrexate. PLoS ONE 2017, 12, e0184891. [Google Scholar]
- Serra, M.; Reverter-Branchat, G.; Maurici, D.; Benini, S.; Shen, J.-N.; Chano, T.; Hattinger, C.; Manara, M.-C.; Pasello, M.; Scotlandi, K.; et al. Analysis of dihydrofolate reductase and reduced folate carrier gene status in relation to methotrexate resistance in osteosarcoma cells. Ann. Oncol. 2004, 15, 151–160. [Google Scholar] [CrossRef]
- Hattinger, C.M.; Stoico, G.; Michelacci, F.; Pasello, M.; Scionti, I.; Remondini, D.; Castellani, G.C.; Fanelli, M.; Scotlandi, K.; Picci, P.; et al. Mechanisms of gene amplification and evidence of coamplification in drug-resistant human osteosarcoma cell lines. Genes Chromosom. Cancer 2009, 48, 289–309. [Google Scholar] [CrossRef]
- Selga, E.; Oleaga, C.; Ramírez, S.; De Almagro, M.C.; Noé, V.; Ciudad, C.J. Networking of differentially expressed genes in human cancer cells resistant to methotrexate. Genome Med. 2009, 1, 83. [Google Scholar] [CrossRef]
- Yang, J.; Guo, W.; Wang, L.; Yu, L.; Mei, H.; Fang, S.; Ji, P.; Liu, Y.; Liu, G.; Song, Q. Cisplatin-resistant osteosarcoma cells possess cancer stem cell properties in a mouse model. Oncol. Lett. 2016, 12, 2599–2605. [Google Scholar] [CrossRef] [Green Version]
- Wagner, F.; Holzapfel, B.M.; McGovern, J.A.; Shafiee, A.; Baldwin, J.G.; Martine, L.C.; Lahr, C.A.; Wunner, F.M.; Friis, T.; Bas, O.; et al. Humanization of bone and bone marrow in an orthotopic site reveals new potential therapeutic targets in osteosarcoma. Biomaterials 2018, 171, 230–246. [Google Scholar] [CrossRef] [Green Version]
- Wagner, F.; Holzapfel, B.M.; Martine, L.C.; McGovern, J.; Lahr, C.A.; Boxberg, M.; Prodinger, P.M.; Grässel, S.; Loessner, D.; Hutmacher, D.W. A humanized bone microenvironment uncovers HIF2 alpha as a latent marker for osteosarcoma. Acta Biomater. 2019, 89, 372–381. [Google Scholar] [CrossRef]
- Zahreddine, H.; Borden, K.L.B. Mechanisms and insights into drug resistance in cancer. Front. Pharmacol. 2013, 4, 28. [Google Scholar] [CrossRef] [Green Version]
- Lee, Y.; Yang, H.; Lu, M.; Tsai, L.; Huang, Y.; Chou, M.; Yu, C.; Hu, F.; Lee, Y.-H.; Yang, H.-W.; et al. DHFR and MDR1 upregulation is associated with chemoresistance in osteosarcoma stem-like cells. Oncol. Lett. 2017, 14, 171–179. [Google Scholar] [CrossRef] [Green Version]
- Susa, M.; Iyer, A.K.; Ryu, K.; Choy, E.; Hornicek, F.J.; Mankin, H.; Milane, L.; Amiji, M.M.; Duan, Z. Inhibition of ABCB1 (MDR1) expression by an siRNA nanoparticulate delivery system to overcome drug resistance in osteosarcoma. PLoS ONE 2010, 5, e10764. [Google Scholar] [CrossRef]
- Liu, T.; Li, Z.; Zhang, Q.; Bernstein, K.D.A.; Lozano-Calderon, S.; Choy, E.; Hornicek, F.J.; Duan, Z. Targeting ABCB1 (MDR1) in multi-drug resistant osteosarcoma cells using the CRISPR-Cas9 system to reverse drug resistance. Oncotarget 2016, 7, 83502–83513. [Google Scholar] [CrossRef]
- Perez, J.; Bardin, C.; Rigal, C.; Anthony, B.; Rousseau, R.; Dutour, A. Anti-MDR1 siRNA restores chemosensitivity in chemoresistant breast carcinoma and osteosarcoma cell lines. Anticancer Res. 2011, 31, 2813–2820. [Google Scholar]
- Gao, Y.; Liao, Y.; Shen, J.K.; Feng, Y.; Choy, E.; Cote, G.; Harmon, D.; Mankin, H.J.; Hornicek, F.J.; Duan, Z. Evaluation of P-glycoprotein (Pgp) expression in human osteosarcoma by high-throughput tissue microarray. J. Orthop. Res. 2016, 34, 1606–1612. [Google Scholar] [CrossRef]
- Fanelli, M.; Hattinger, C.M.; Vella, S.; Tavanti, E.; Michelacci, F.; Gudeman, B.; Barnett, D.; Picci, P.; Serra, M. Targeting ABCB1 and ABCC1 with their Specific Inhibitor CBT-1(®) can overcome drug resistance in osteosarcoma. Curr. Cancer Drug Targets 2016, 16, 261–274. [Google Scholar] [CrossRef]
- Cagliero, E.; Ferracini, R.; Morello, E.; Scotlandi, K.; Manara, M.C.; Buracco, P.; Comandone, A.; Parisi, R.B.; Baldini, N. Reversal of multidrug-resistance using Valspodar® (PSC 833) and doxorubicin in osteosarcoma. Oncol. Rep. 2004, 12, 1023–1031. [Google Scholar] [CrossRef]
- Italiano, A.; Penel, N.; Toulmonde, M.; Bompas, E.; Piperno-Neumann, S.; Pulido, M.; Entz-Werle, N.; Le Cesne, A.; Chevreau, C.M.; Duffaud, F.; et al. Cabozantinib in patients with advanced osteosarcomas and ewing sarcomas: A French Sarcoma Group (FSG)/US National Cancer Institute phase II Collab. |OncologyPRO. In Proceedings of the ESMO 2018 Congress, Munich, Germany, 19–23 October 2018. [Google Scholar]
- Xiang, Q.; Chen, W.; Ren, M.; Wang, J.; Zhang, H.; Deng, D.Y.B.; Zhang, L.; Shang, C.; Chen, Y. Cabozantinib suppresses tumor growth and metastasis in hepatocellular carcinoma by a dual blockade of VEGFR2 and MET. Clin. Cancer Res. 2014, 20, 2959–2970. [Google Scholar] [CrossRef]
- Šrámek, M.; Neradil, J.; Sterba, J.; Veselska, R. Non-DHFR-mediated effects of methotrexate in osteosarcoma cell lines: Epigenetic alterations and enhanced cell differentiation. Cancer Cell Int. 2016, 16, 495. [Google Scholar] [CrossRef]
- Neradil, J.; Pavlasova, G.; Sramek, M.; Kyr, M.; Veselska, R.; Sterba, J. DHFR-mediated effects of methotrexate in medulloblastoma and osteosarcoma cells: The same outcome of treatment with different doses in sensitive cell lines. Oncol. Rep. 2015, 33, 2169–2175. [Google Scholar] [CrossRef]
- Wang, J.; Li, G. Relationship between RFC gene expression and intracellular drug concentration in methotrexate-resistant osteosarcoma cells. Genet. Mol. Res. 2014, 13, 5313–5321. [Google Scholar] [CrossRef]
- Fan, H.; Lu, S.; Wang, S.; Zhang, S. Identification of critical genes associated with human osteosarcoma metastasis based on integrated gene expression profiling. Mol. Med. Rep. 2019, 20, 915–930. [Google Scholar] [CrossRef]
- Deng, B.; Qiu, B. Shikonin inhibits invasiveness of osteosarcoma through MMP13 suppression. Tumor Boil. 2015, 36, 9311–9317. [Google Scholar] [CrossRef]
- Smida, J.; Baumhoer, D.; Rosemann, M.; Walch, A.; Bielack, S.; Poremba, C.; Remberger, K.; Korsching, E.; Scheurlen, W.; Dierkes, C.; et al. Genomic alterations and allelic imbalances are strong prognostic predictors in osteosarcoma. Clin. Cancer Res. 2010, 16, 4256–4267. [Google Scholar] [CrossRef]
- Wang, D.; Niu, X.; Wang, Z.; Song, C.-L.; Huang, Z.; Chen, K.-N.; Duan, J.; Bai, H.; Xu, J.; Wang, Y.; et al. Multiregion sequencing reveals the genetic heterogeneity and evolutionary history of osteosarcoma and matched pulmonary metastases. Cancer Res. 2018, 79, 7–20. [Google Scholar] [CrossRef]
- Lou, K.-J. Screening for immunogenic cell death. Sci. Bus. Exch. 2012, 5, 808. [Google Scholar] [CrossRef]
- Martin, J.W.; Squire, J.A.; Zielenska, M. The Genetics of Osteosarcoma. Sarcoma 2012, 2012, 627254. [Google Scholar] [CrossRef]
- Ohata, N.; Ito, S.; Yoshida, A.; Kunisada, T.; Numoto, K.; Jitsumori, Y.; Kanzaki, H.; Ozaki, T.; Shimizu, K.; Ouchida, M. Highly frequent allelic loss of chromosome 6q16-23 in osteosarcoma: Involvement of cyclin C in osteosarcoma. Int. J. Mol. Med. 2006, 18, 1153–1158. [Google Scholar] [CrossRef] [Green Version]
- Zhang, Y.; Mai, Q.; Xie, C. Microenvironment Signals and Mechanisms in the Regulation of Osteosarcoma. In Osteosarcoma—Biology, Behavior and Mechanisms; IntechOpen: Rijeka, Croatia, 2017. [Google Scholar]
- Crenn, V.; Biteau, K.; Amiaud, J.; Dumars, C.; Guiho, R.; Vidal, L.; Le Nail, L.-R.; Heymann, D.; Moreau, A.; Gouin, F.; et al. Bone microenvironment has an influence on the histological response of osteosarcoma to chemotherapy: Retrospective analysis and preclinical modeling. Am. J. Cancer Res. 2017, 7, 2333–2349. [Google Scholar]
- Da Costa, M.E.M.; Daudigeos-Dubus, E.; Gomez-Brouchet, A.; Bawa, O.; Rouffiac, V.; Serra, M.; Scotlandi, K.; Santos, C.; Geoerger, B.; Gaspar, N. Establishment and characterization of in vivo orthotopic bioluminescent xenograft models from human osteosarcoma cell lines in Swiss nude and NSG mice. Cancer Med. 2018, 7, 665–676. [Google Scholar] [CrossRef]
- Serra, M.; Scotlandi, K.; Manara, M.C.; Maurici, D.; Lollini, P.L.; De Giovanni, C.; Toffoli, G.; Baldini, N. Establishment and characterization of multidrug-resistant human osteosarcoma cell lines. Anticancer Res. 1993, 13, 323–329. [Google Scholar]
- Mohseny, A.B.; Machado, I.; Cai, Y.; Schaefer, K.-L.; Serra, M.; Hogendoorn, P.C.W.; Llombart-Bosch, A.; Cleton-Jansen, A.-M. Functional characterization of osteosarcoma cell lines provides representative models to study the human disease. Lab. Investig. 2011, 91, 1195–1205. [Google Scholar] [CrossRef]
- Ottaviano, L.; Schaefer, K.-L.; Gajewski, M.; Huckenbeck, W.; Baldus, S.; Rogel, U.; Mackintosh, C.; De Alava, E.; Myklebost, O.; Kresse, S.H.; et al. Molecular characterization of commonly used cell lines for bone tumor research: A trans-European EuroBoNet effort. Genes Chromosom. Cancer 2010, 49, 40–51. [Google Scholar] [CrossRef]
- Lauvrak, S.U.; Munthe, E.; Kresse, S.H.; Stratford, E.W.; Namløs, H.M.; Meza-Zepeda, L.A.; Myklebost, O. Functional characterisation of osteosarcoma cell lines and identification of mRNAs and miRNAs associated with aggressive cancer phenotypes. Br. J. Cancer 2013, 109, 2228–2236. [Google Scholar] [CrossRef]
- Uluçkan, Ö.; Bakiri, L.; Wagner, E.F. Characterization of Mouse Model-Derived Osteosarcoma (OS) Cells In Vitro and In Vivo; Humana Press: New York, NY, USA, 2015; pp. 297–305. [Google Scholar]
- Daudigeos-Dubus, E.; LE Dret, L.; Rouffiac, V.; Bawa, O.; Leguerney, I.; Opolon, P.; Vassal, G.; Geoerger, B. Establishment and characterization of new orthotopic and metastatic neuroblastoma models. Vivo 2014, 28, 425–434. [Google Scholar]
- Venkatraman, E.S.; Olshen, A.B.; Lucito, R.; Wigler, M. Circular binary segmentation for the analysis of array-based DNA copy number data. Biostatistics 2004, 5, 557–572. [Google Scholar]
- Harttrampf, A.C.; Lacroix, L.; Deloger, M.; Deschamps, F.; Puget, S.; Auger, N.; Vielh, P.; Varlet, P.; Balogh, Z.; Abbou, S.; et al. Molecular Screening for Cancer Treatment Optimization (MOSCATO-01) in pediatric patients: A single-institutional prospective molecular stratification trial. Clin. Cancer Res. 2017, 23, 6101–6112. [Google Scholar] [CrossRef]
- Patro, R.; Duggal, G.; Love, M.I.; Irizarry, R.A.; Kingsford, C. Salmon provides fast and bias-aware quantification of transcript expression. Nat. Methods 2017, 14, 417–419. [Google Scholar] [CrossRef] [Green Version]
- Love, M.I.; Huber, W.; Anders, S. Moderated estimation of fold change and dispersion for RNA-seq data with DESeq2. Genome Boil. 2014, 15, 31. [Google Scholar] [CrossRef]
- Tsubaki, M.; Satou, T.; Itoh, T.; Imano, M.; Komai, M.; Nishinobo, M.; Yamashita, M.; Yanae, M.; Yamazoe, Y.; Nishida, S. Overexpression of MDR1 and survivin, and decreased Bim expression mediate multidrug-resistance in multiple myeloma cells. Leuk. Res. 2012, 36, 1315–1322. [Google Scholar] [CrossRef] [PubMed]




| Luc/mKate2 Cell Line | Parental | R/MTX | R/DOXO | |||
|---|---|---|---|---|---|---|
| Primary Tumor | Metastases | Primary Tumor | Metastases | Primary Tumor | Metastases | |
| HOS | 5/5 | 5/5 | 8/8 | 4/8 | 4/6 | 4/6 |
| Saos-2-B | 5/6 | 5/6 | 4/4 | 4/4 | - | - |
| Cell Lines | IC50 (µM) | RI | IC50 (µM) | RI | ||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Parental | R/MTX | R/MTX | Parental | R/DOXO | R/DOXO | |||||
| Before * | Before * | After * | Before * | After * | Before * | Before * | After * | Before * | After * | |
| HOS | 0.04 | 6.24 | >100 | 156 | >2000 | 0.06 | 11.3 | 2.09 | 212 | 41.8 |
| Saos-2-B | 0.05 | 1.93 | 2.05 | 37 | 34.2 | NA | NA | NA | NA | NA |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Marques da Costa, M.E.; Marchais, A.; Gomez-Brouchet, A.; Job, B.; Assoun, N.; Daudigeos-Dubus, E.; Fromigué, O.; Santos, C.; Geoerger, B.; Gaspar, N. In-Vitro and In-Vivo Establishment and Characterization of Bioluminescent Orthotopic Chemotherapy-Resistant Human Osteosarcoma Models in NSG Mice. Cancers 2019, 11, 997. https://doi.org/10.3390/cancers11070997
Marques da Costa ME, Marchais A, Gomez-Brouchet A, Job B, Assoun N, Daudigeos-Dubus E, Fromigué O, Santos C, Geoerger B, Gaspar N. In-Vitro and In-Vivo Establishment and Characterization of Bioluminescent Orthotopic Chemotherapy-Resistant Human Osteosarcoma Models in NSG Mice. Cancers. 2019; 11(7):997. https://doi.org/10.3390/cancers11070997
Chicago/Turabian StyleMarques da Costa, Maria Eugénia, Antonin Marchais, Anne Gomez-Brouchet, Bastien Job, Noémie Assoun, Estelle Daudigeos-Dubus, Olivia Fromigué, Conceição Santos, Birgit Geoerger, and Nathalie Gaspar. 2019. "In-Vitro and In-Vivo Establishment and Characterization of Bioluminescent Orthotopic Chemotherapy-Resistant Human Osteosarcoma Models in NSG Mice" Cancers 11, no. 7: 997. https://doi.org/10.3390/cancers11070997





