Immunological Backbone of Uveal Melanoma: Is There a Rationale for Immunotherapy?
Abstract
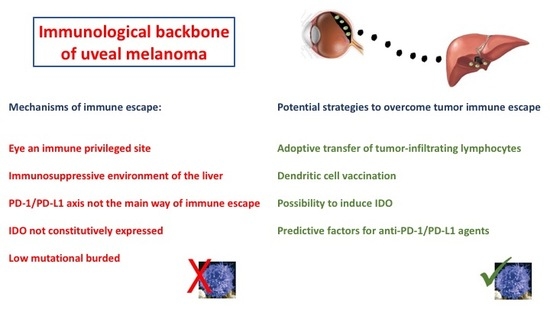
1. Introduction
2. Microenvironment of the Eye
3. Microenvironment of the Liver
4. Immune Infiltrate in Uveal Melanoma
4.1. The Role of PD-1/PD-L1 Interaction in UM
4.2. IDO and Immune Escape
5. Mutational Burden and Epitopes of UM
6. Conclusions
Funding
Conflicts of Interest
References
- Carvajal, R.D.; Piperno-Neumann, S.; Kapiteijn, E.; Chapman, P.B.; Frank, S.; Joshua, A.M.; Piulats, J.M.; Wolter, P.; Cocquyt, V.; Chmielowski, B.; et al. Selumetinib in combination with dacarbazine in patients with metastatic uveal melanoma: A phase III, multicenter, randomized trial (SUMIT). J. Clin. Oncol. 2018, 36, 1232–1239. [Google Scholar] [CrossRef] [PubMed]
- Schinzari, G.; Rossi, E.; Cassano, A.; Dadduzio, V.; Quirino, M.; Pagliara, M.; Blasi, M.A.; Barone, C. Cisplatin, dacarbazine and vinblastine as first line chemotherapy for liver metastatic uveal melanoma in the era of immunotherapy: A single institution phase II study. Melanoma Res. 2017, 27, 591–595. [Google Scholar] [CrossRef] [PubMed]
- Buder, K.; Gesierich, A.; Gelbrich, G.; Goebeler, M. Systemic treatment of metastatic uveal melanoma: Review of literature and future perspectives. Cancer Med. 2013, 2, 674–686. [Google Scholar] [CrossRef] [PubMed]
- Komatsubara, K.M.; Carvajal, R.D. Immunotherapy for the treatment of uveal melanoma: Current status and emerging therapies. Curr. Oncol. Rep. 2017, 19, 45. [Google Scholar] [CrossRef] [PubMed]
- Oliva, M.; Rullan, A.J.; Piulats, J.M. Uveal melanoma as a target for immune-therapy. Ann. Transl. Med. 2016, 4, 172. [Google Scholar] [CrossRef] [PubMed]
- Singh, A.D.; Turell, M.E.; Topham, A.K. Uveal melanoma: Trends in incidence, treatment, and survival. Ophthalmology 2011, 118, 1881–1885. [Google Scholar] [CrossRef] [PubMed]
- Spagnolo, F.; Picasso, V.; Spano, L.; Tanda, E.; Venzano, C.; Queirolo, P. Update on metastatic uveal melanoma: Progress and challenges. BioDrugs 2016, 30, 161–172. [Google Scholar] [CrossRef] [PubMed]
- Bender, C.; Enk, A.; Gutzmer, R.; Hasser, C. Anti-PD-1 antibodies in metastatic uveal melanoma: A treatment option? Cancer Med. 2017, 6, 1581–1586. [Google Scholar] [CrossRef] [PubMed]
- Algazi, A.P.; Tsai, K.K.; Shoushtari, A.N.; Munhoz, R.R.; Eroglu, Z.; Piulats, J.M.; Ott, P.A.; Johnson, D.B.; Hwang, J.; Daud, A.I.; et al. Clinical outcomes in metastatic uveal melanoma treated with PD-1 and PD-L1 antibodies. Cancer 2016, 122, 3344–3353. [Google Scholar] [CrossRef]
- Danielli, R.; Ridolfi, R.; Chiarion-Sileni, V.; Queirolo, P.; Testori, A.; Plummer, R.; Boitano, M.; Calabrò, L.; Rossi, C.D.; Giacomo, A.M.; et al. Ipilimumab in pretreated patients with metastatic uveal melanoma: Safety and clinical efficacy. Cancer Immunol. Immunother. 2012, 61, 41–48. [Google Scholar] [CrossRef] [PubMed]
- Joshua, A.M.; Monzon, J.G.; Mihalcioiu, C.; Hogg, D.; Smylie, M.; Cheng, T. A phase 2 study of tremelimumab in patients with advanced uveal melanoma. Melanoma Res. 2015, 25, 342–347. [Google Scholar] [CrossRef]
- Karydis, I.; Chan, P.Y.; Wheater, M.; Arriola, E.; Szlosarek, P.W.; Ottensmeier, C.H. Clinical activity and safety of pembrolizumab in ipilimumab pre-treated patients with uveal melanoma. Oncoimmunology 2016, 5, e1143997. [Google Scholar] [CrossRef]
- Khattak, M.A.; Fisher, R.; Hughes, P.; Gore, M.; Larkin, J. Ipilimumab activity in advanced uveal melanoma. Melanoma Res. 2013, 23, 79–81. [Google Scholar] [CrossRef]
- Kottschade, L.A.; McWilliams, R.R.; Markovic, S.N.; Block, M.S.; Villasboas Bisneto, J.; Pham, A.Q.; Esplin, B.L.; Dronca, R.S. The use of pembrolizumab for the treatment of metastatic uveal melanoma. Melanoma Res. 2016, 26, 300–303. [Google Scholar] [CrossRef] [PubMed]
- Luke, J.J.; Callahan, M.K.; Postow, M.A.; Romano, E.; Ramaiya, N.; Bluth, M.; Giobbie-Hurder, A.; Lawrence, D.P.; Ibrahim, N.; Ott, P.A.; et al. Clinical activity of ipilimumab for metastatic uveal melanoma: A retrospective review of the Dana-Farber Cancer Institute, Massachusetts General Hospital, Memorial Sloan-Kettering Cancer Center, and University Hospital of Lausanne experience. Cancer 2013, 119, 3687–3695. [Google Scholar] [CrossRef]
- Mignard, C.; Deschamps Huvier, A.; Gillibert, A.; Duval Modeste, A.B.; Dutriaux, C.; Khammari, A.; Avril, M.F.; Kramkimel, N.; Mortier, L.; Marcant, P.; et al. Efficacy of Immunotherapy in Patients with Metastatic Mucosal or Uveal Melanoma. J. Oncol. 2018, 2018, 1908065. [Google Scholar] [CrossRef]
- Maio, M.; Danielli, R.; Chiarion-Sileni, V.; Pigozzo, J.; Parmiani, G.; Ridolfi, R.; De Rosa, F.; Del Vecchio, M.; Di Guardo, L.; Queirolo, P.; et al. Efficacy and safety of ipilimumab in patients with pre-treated, uveal melanoma. Ann. Oncol. 2013, 24, 2911–2915. [Google Scholar] [CrossRef]
- Rossi, E.; Pagliara, M.M.; Orteschi, D.; Dosa, T.; Sammarco, M.G.; Caputo, C.G.; Petrone, G.; Rindi, G.; Zollino, M.; Blasi, M.A.; et al. Pembrolizumab as first-line treatment for metastatic uveal melanoma. Cancer Immunol. Immunother. 2019, 68, 1179–1185. [Google Scholar] [CrossRef] [PubMed]
- Zimmer, L.; Vaubel, J.; Mohr, P.; Hauschild, A.; Utikal, J.; Simon, J. Phase II DeCOG-study of ipilimumab in pretreated and treatment naive patients with metastatic uveal melanoma. PLoS ONE 2015, 10, e0118564. [Google Scholar] [CrossRef] [PubMed]
- Kelderman, S.; van der Kooij, M.K.; van den Eertwegh, A.J.; Soetekouw, P.M.; Jansen, R.L.; van den Brom, R.R.; Hospers, G.A.; Haanen, J.B.; Kapiteijn, E.; Blank, C.U. Ipilimumab in pretreated metastastic uveal melanoma patients. Results of the Dutch Working group on Immunotherapy of Oncology (WIN-O). Acta Oncol. 2013, 52, 1786–1788. [Google Scholar] [CrossRef]
- Piulats Rodriguez, J.M.; Ochoa de Olza, M.; Codes, M.; Lopez-Martin, J.A.; Berrocal, A.; García, M.; Gurpide, A.; Homet, B.; Martin-Algarra, S. Phase II study evaluating ipilimumab as a single agent in the first-line treatment of adult patients (Pts) with metastatic uveal melanoma (MUM): The GEM-1 trial. J. Clin. Oncol. 2014, 32, 9033. [Google Scholar] [CrossRef]
- Deo, M.A. Long-term survival benefit from ipilimumab treatment in metastatic uveal melanoma patients. J. Clin. Oncol. 2014, 32, 3060. [Google Scholar] [CrossRef]
- Shaw, H.; Larkin, J.; Corrie, P. Ipilimumab for advanced melanoma in an expanded access program (EAP): Ocular, mucosal and acral subtype UK experience. Ann. Oncol. 2012, 23, 374. [Google Scholar]
- Heppt, M.V.; Heinzerling, L.; Kähler, K.C.; Forschner, A.; Kirchberger, M.C.; Loquai, C.; Meissner, M.; Meier, F.; Terheyden, P.; Schell, B.; et al. Prognostic factors and outcomes in metastatic uveal melanoma treated with programmed cell death-1 or combined PD-1/cytotoxic T-lymphocyte antigen-4 inhibition. Eur. J. Cancer 2017, 82, 56–65. [Google Scholar] [CrossRef]
- Piperno-Neumann, S.; Servois, V.; Mariani, P.; Cassoux, N.; Barnhill, R.; Rodrigues, M.J. Activity of anti-PD1 drugs in uveal melanoma patients. J. Clin. Oncol. 2016, 34, 9588. [Google Scholar] [CrossRef]
- Van der Kooij, M.K.; Joosse, A.; Speetjens, F.M.; Hospers, G.A.; Bisschop, C.; de Groot, J.W.; Koornstra, R.; Blank, C.U.; Kapiteijn, E. Anti-PD1 treatment in metastatic uveal melanoma in the Netherlands. Acta Oncol. 2017, 56, 101–103. [Google Scholar] [CrossRef] [PubMed]
- Schadendorf, D.; Ascierto, P.A.; Haanen, J.; Espinosa, E.; Demidov, L.V.; Garbe, C.; Lorigan, P.; Gogas, H.; Hoeller, C.; Guren, T.K.; et al. Efficacy and safety of nivolumab (NIVO) in patients with advanced melanoma (MEL) and poor prognostic factors who progressed on or after ipilimumab (IPI): Results from a phase II study (CheckMate 172). J. Clin. Oncol. 2017, 35, 9524. [Google Scholar] [CrossRef]
- Jung, M.; Lee, J.; Kim, T.M.; Lee, D.H.; Kang, J.H.; Oh, S.Y.; Lee, S.J.; Shin, S.J. Ipilimumab real-world efficacy and safety in korean melanoma patients from the korean named patient program cohort. Cancer Res. Treat. 2017, 49, 44–53. [Google Scholar] [CrossRef]
- Shoushtari, A.N.; Navld-Azarbaljanl, P.; Friedman, C.F.; Panageas, K.; Postow, M.A.; Callahan, M.K.; Momtaz, P.; Campbell, S.C.; Shames, Y.; Prempeh-Keteku, N.A.; et al. Efficacy of nivolumab and ipilimumab (Nivo + Ipi) combination in melanoma patients (pts) treated at a single institution on an expanded-access program (EAP). J. Clin. Oncol. 2016, 34, 9554. [Google Scholar] [CrossRef]
- Robert, C.; Long, G.V.; Brady, B.; Dutriaux, C.; Maio, M.; Mortier, L.; Hassel, J.C.; Rutkowski, P.; McNeil, C.; Kalinka-Warzocha, E.; et al. Nivolumab in previously untreated melanoma without BRAF mutation. N. Engl. J. Med. 2015, 372, 320–330. [Google Scholar] [CrossRef]
- Robert, C.; Ribas, A.; Wolchok, J.D.; Hodi, F.S.; Hamid, O.; Kefford, R. Anti-programmed-death-receptor-1 treatment with pembrolizumab in ipilimumab-refractory advanced melanoma: A randomised dose-comparison cohort of a phase 1 trial. Lancet 2014, 384, 1109–1117. [Google Scholar] [CrossRef]
- Robert, C.; Schachter, J.; Long, G.V.; Arance, A.; Grob, J.J.; Mortier, L.; Daud, A.; Carlino, M.S.; McNeil, C.; Lotem, M.; et al. Pembrolizumab versus Ipilimumab in Advanced Melanoma. N. Engl. J. Med. 2015, 372, 2521–2532. [Google Scholar] [CrossRef] [PubMed]
- Weber, J.S.; D’Angelo, S.P.; Minor, D.; Hodi, F.S.; Gutzmer, R.; Neyns, B.; Hoeller, C.; Khushalani, N.I.; Miller, W.H., Jr.; Lao, C.D.; et al. Nivolumab versus chemotherapy in patients with advanced melanoma who progressed after anti-CTLA-4 treatment (CheckMate 037): A randomised, controlled, open-label, phase 3 trial. Lancet Oncol. 2015, 16, 375–384. [Google Scholar] [CrossRef]
- Niederkorn, J.Y. Immune escape mechanisms of intraocular tumors. Progr. Retin. Eye Res. 2009, 28, 329–347. [Google Scholar] [CrossRef] [PubMed]
- Terai, M.; Mastrangelo, M.J.; Sato, T. Immunological aspect of the liver and metastatic uveal melanoma. J. Cancer Metastasis Treat. 2017, 3, 231–243. [Google Scholar] [CrossRef]
- Furney, S.J.; Pedersen, M.; Gentien, D.; Dumont, A.G.; Rapinat, A.; Desjardins, L.; Turajlic, S.; Piperno-Neumann, S.; de la Grange, P.; Roman-Roman, S.; et al. SF3B1 mutations are associated with alternative splicing in uveal melanoma. Cancer Discov. 2013, 3, 1122–1129. [Google Scholar] [CrossRef] [PubMed]
- Bill, A. Ocular circulation. In Adler’s Physiology of the Eye, 5th ed.; Moses, R.A., Ed.; Mosby Co: St. Louis, MO, USA, 1970; pp. 278–296. [Google Scholar]
- Taylor, A.W. Ocular immunosuppressive microenvironment. Chem. Immunol. 2008, 92, 71–85. [Google Scholar] [CrossRef]
- Sugita, S.; Ng, T.F.; Lucas, P.J.; Gress, R.E.; Streilein, J.W. B7+ iris pigment epithelium induce CD8+ T regulatory cells; both suppress CTLA-4+T cells. J. Immunol. 2006, 176, 118–127. [Google Scholar] [CrossRef]
- Sohn, J.H.; Kaplan, H.J. Chronic low level complement activation within the eye is controlled by intraocular complement regulatory proteins. Investig. Ophthalmol. Vis. Sci. 2000, 41, 3492–3502. [Google Scholar]
- Stein-Streilein, J.; Streilein, J.W. Anterior chamber associated immune deviation (ACAID): Regulation, biological relevance, and implications for therapy. Int. Rev. Immunol. 2002, 21, 123–152. [Google Scholar] [CrossRef]
- Vendomèle, J.; Khebizi, Q.; Fisson, S. Cellular and molecular mechanisms of Anterior Chamber-Associated Immune Deviation (ACAID): What we have learned from knockout mice. Front. Immunol. 2017, 8, 1686. [Google Scholar] [CrossRef]
- Racanelli, V.; Rehermann, B. The liver as an immunological organ. Hepatology 2006, 43 (Suppl. 1), S54–S62. [Google Scholar] [CrossRef] [PubMed]
- Jenne, C.N.; Kubes, P. Immune surveillance by the liver. Nat. Immunol. 2013, 14, 996–1006. [Google Scholar] [CrossRef] [PubMed]
- Platten, M.; von Knebel Doeberitz, N.; Oezen, I.; Wick, W.; Ochs, K. Cancer immunotherapy by targeting IDO1/TDO and their downstreamveffectors. Front. Immunol. 2014, 5, 673. [Google Scholar] [CrossRef] [PubMed]
- Kimura, Y.; Inoue, A.; Hangai, S.; Saijo, S.; Negishi, H.; Nishio, J.; Yamasaki, S.; Iwakura, Y.; Yanai, H.; Taniguchi, T. The innate immune receptor Dectin-2 mediates the phagocytosis of cancer cells by Kupffer cells for the suppression of liver metastasis. Proc. Natl. Acad. Sci. USA 2016, 113, 14097–14102. [Google Scholar] [CrossRef] [PubMed]
- Nemeth, E.; Baird, A.W.; O’Farrelly, C. Microanatomy of the liver immune system. Semin. Immunopathol. 2009, 31, 333–343. [Google Scholar] [CrossRef] [PubMed]
- Bronkhorst, I.H.; Jager, M.J. Uveal melanoma: The inflammatory microenvironment. J. Innate Immun. 2012, 4, 454–462. [Google Scholar] [CrossRef]
- Jager, M.J.; Ly, L.V.; El Filali, M.; Madigan, M.C. Macrophages in uveal melanoma and in experimental ocular tumor models: Friends or foes? Prog. Retin. Eye Res. 2011, 30, 129–146. [Google Scholar] [CrossRef]
- Clarijs, R.; Schalkwijk, L.; Ruiter, D.J.; de Waal, R.M. EMAP-II expression is associated with macrophage accumulation in primary uveal melanoma. Investig. Ophthalmol. Vis. Sci. 2003, 44, 1801–1806. [Google Scholar] [CrossRef][Green Version]
- Callejo, S.A.; Marshall, J.C.; Cools-Lartigue, J.; Saraiva, V.S.; Burnier, M.N., Jr. Macrophage-derived soluble factor enhances melanoma inhibitory activity expression by uveal melanoma cells In Vitro. Melanoma Res. 2004, 14, 91–95. [Google Scholar] [CrossRef]
- Meecham, W.J.; Char, D.H.; Kaleta-Michaels, S. Infiltrating lymphocytes and antigen expression in uveal melanoma. Ophthalmic Res. 1992, 24, 20–26. [Google Scholar] [CrossRef] [PubMed]
- Lagouros, E.; Salomao, D.; Thorland, E.; Hodge, D.O.; Vile, R.; Pulido, J.S. Infiltrative T regulatory cells in enucleated uveal melanomas. Trans. Am. Ophthalmol. Soc. 2009, 107, 223–228. [Google Scholar] [PubMed]
- Mougiakakos, D.; Johansson, C.C.; Trocme, E.; All-Ericsson, C.; Economou, M.A.; Larsson, O.; Seregard, S.; Kiessling, R. Intratumoral forkhead box P3-positive regulatory T cells predict poor survival in cyclooxygenase-2-positive uveal melanoma. Cancer 2010, 116, 2224–2233. [Google Scholar] [CrossRef] [PubMed]
- Bronkhorst, I.H.; Ly, L.V.; Jordanova, E.S.; Vrolijk, J.; Versluis, M.; Luyten, G.P.; Jager, M.J. Detection of M2-macrophages in uveal melanoma and relation with survival. Investig. Ophthalmol. Vis. Sci. 2011, 52, 643–650. [Google Scholar] [CrossRef] [PubMed]
- Makitie, T.; Summanen, P.; Tarkkanen, A.; Kivela, T. Tumor-infiltrating macrophages (CD68(+) cells) and prognosis in malignant uveal melanoma. Investig. Ophthalmol. Vis. Sci. 2001, 42, 1414–1421. [Google Scholar]
- Ly, L.V.; Baghat, A.; Versluis, M.; Jordanova, E.S.; Luyten, G.P.M.; van Rooijen, N.; van Hall, T.; van der Velden, P.A.; Jager, M.J. In aged mice, outgrowth of intraocular melanoma depends on proangiogenic M2-type macrophages. J. Immunol. 2010, 185, 3481–3488. [Google Scholar] [CrossRef]
- Ericsson, C.; Seregard, S.; Bartolazzi, A.; Levitskaya, E.; Ferrone, S.; Kiessling, R.; Larssonet, O. Association of HLA class I and class II antigen expression and mortality in uveal melanoma. Investig. Ophthalmol. Vis. Sci. 2001, 42, 2153–2156. [Google Scholar]
- Vetter, C.S.; Lieb, W.; Brocker, E.B.; Becker, J.C. Loss of nonclassical MHC molecules MIC-A/B expression during progression of uveal melanoma. Br. J. Cancer 2004, 91, 1495–1499. [Google Scholar] [CrossRef][Green Version]
- Jager, M.J.; Hurks, H.M.; Levitskaya, J.; Kiessling, R. HLA expression in uveal melanoma: There is no rule without some exception. Hum. Immunol. 2002, 63, 444–451. [Google Scholar] [CrossRef]
- Blom, D.J.; Luyten, G.P.; Mooy, C.; Kerkvliet, S.; Zwinderman, A.H.; Jager, M.J. Human leukocyte antigen class I expression. Marker of poor prognosis in uveal melanoma. Investig. Ophthalmol. Vis. Sci. 1997, 38, 1865–1872. [Google Scholar]
- De Waard-Siebinga, I.; Hilders, C.G.; Hansen, B.E.; van Delft, J.L.; Jager, M.J. HLA expression and tumor-infiltrating immune cells in uveal melanoma. Graefes Arch. Clin. Exp. Ophthalmol. 1996, 234, 34–42. [Google Scholar] [CrossRef] [PubMed]
- Repp, A.C.; Mayhew, E.S.; Apte, S.; Niederkorn, J.Y. Human uveal melanoma cells produce macrophage migration-inhibitory factor to prevent lysis by NK cells. J. Immunol. 2000, 165, 710–715. [Google Scholar] [CrossRef]
- Apte, R.S.; Mayhew, E.; Niederkorn, J.Y. Local inhibition of natural killer cell activity promotes the progressive growth of intraocular tumors. Investig. Ophthalmol. Vis. Sci. 1997, 38, 1277–1282. [Google Scholar] [CrossRef]
- Apte, R.S.; Sinha, D.; Mayhew, E.; Wistow, G.J.; Niederkorn, J.Y. Cutting edge: Role of macrophage migration inhibitory factor in inhibiting NK cell activity and preserving immune privilege. J. Immunol. 1998, 160, 5693–5696. [Google Scholar]
- Anastassiou, G.; Coupland, S.E.; Stang, A.; Boeloeni, R.; Schilling, H.; Bornfeld, N. Expression of Fas and Fas ligand in uveal melanoma: Biological implication and prognostic value. J. Pathol. 2001, 194, 466–472. [Google Scholar] [CrossRef] [PubMed]
- He, Y.G.; Mayhew, E.; Mellon, J.; Niederkorn, J.Y. Expression and possible function of IL-2 and IL-15 receptors on human uveal melanoma cells. Investig. Ophthalmol. Vis. Sci. 2004, 45, 4240–4246. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Polak, M.E.; Borthwick, N.J.; Johnson, P.; Hungerford, J.L.; Higgins, B.; Di Palma, S.; Jager, M.J.; Cree, I.A. Presence and phenotype of dendritic cells in uveal melanoma. Br. J. Ophthalmol. 2007, 91, 971–976. [Google Scholar] [CrossRef] [PubMed]
- Bronkhorst, I.H.; Vu, T.H.; Jordanova, E.S.; Luyten, G.P.; Burg, S.H.; Jager, M.J. Different subsets of tumor-infiltrating lymphocytes correlate with macrophage influx and monosomy 3 in uveal melanoma. Investig. Ophthalmol. Vis. Sci. 2012, 53, 5370–5378. [Google Scholar] [CrossRef]
- Achberger, S.; Aldrich, W.; Tubbs, R.; Crabb, J.W.; Singh, A.D.; Triozzi, P.L. Circulating immune cell and microRNA in patients with uveal melanoma developing metastatic disease. Mol. Immunol. 2014, 58, 182–186. [Google Scholar] [CrossRef]
- Krishna, Y.; McCarthy, C.; Kalirai, H.; Coupland, S.E. Inflammatory cell infiltrates in advanced metastatic uveal melanoma. Hum. Pathol. 2017, 66, 159–166. [Google Scholar] [CrossRef]
- Whelchel, J.C.; Farah, S.E.; McLean, I.W.; Burnier, M.N. Immunohistochemistry of infiltrating lymphocytes in uveal malignant melanoma. Investig. Ophthalmol. Vis. Sci. 1993, 34, 2603–2606. [Google Scholar]
- De la Cruz, P.O., Jr.; Specht, C.S.; McLean, I.W. Lymphocytic infiltration in uveal malignant melanoma. Cancer 1990, 65, 112–115. [Google Scholar] [CrossRef]
- Durie, F.H.; Campbell, A.M.; Lee, W.R.; Damato, B.E. Analysis of lymphocytic infiltration in uveal melanoma. Investig. Ophthalmol. Vis. Sci. 1990, 31, 2106–2110. [Google Scholar]
- Rothermel, L.D.; Sabesan, A.C.; Stephens, D.J.; Chandran, S.S.; Paria, B.C.; Srivastava, A.K.; Somerville, R.; Wunderlich, J.R.; Lee, C.C.; Xi, L.; et al. Identification of an immunogenic subset of metastatic uveal melanoma. Clin. Cancer Res. 2016, 22, 2237–2249. [Google Scholar] [CrossRef]
- Jia, R.; Jiao, Z.; Xu, X. Functional significance of B7-H1 expressed by human uveal melanoma cells. Mol. Med. Rep. 2011, 4, 163–167. [Google Scholar] [CrossRef]
- Ma, J.; Usui, Y.; Kezuka, T.; Okunuki, Y.; Zhang, L.; An, X.; Mizota, A.; Goto, H. Costimulatory molecule expression on human uveal melanoma cells: Functional analysis of CD40 and B7-H1. Exp. Eye Res. 2012, 96, 98–110. [Google Scholar] [CrossRef]
- Qin, Y.; Petaccia de Macedo, M.; Reuben, A.; Forgeta, M.A.; Haymaker, C.; Bernatcheza, C.; Spencerd, C.N.; Gopalakrishnan, V.; Reddy, S.; Cooper, Z.A.; et al. Parallel profiling of immune infiltrate subsets in uveal melanoma versus cutaneous melanoma unveils similarities and differences: A pilot study. Oncoimmunology 2017, 6, e1321187. [Google Scholar] [CrossRef]
- Javed, A.; Arguello, D.; Johnston, C.; Gatalica, Z.; Terai, M.; Weight, R.M.; Orloff, M.; Mastrangelo, M.J.; Sato, T. PD-L1 expression in tumor metastasis is different between uveal melanoma and cutaneous melanoma. Immunotherapy 2017, 9, 1323–1330. [Google Scholar] [CrossRef]
- Zoroquiain, P.; Esposito, E.; Logan, P.; Aldrees, S.; Dias, A.B.; Mansure, J.J.; Santapau, D.; Garcia, C.; Saornil, M.A.; Belfort Neto, R.; et al. Programmed cell death ligand-1 expression in tumor and immune cells is associated with better patient outcome and decreased tumor-infiltrating lymphocytes in uveal melanoma. Mod. Pathol. 2018, 31, 1201–1210. [Google Scholar] [CrossRef]
- Yang, W.; Chen, P.W.; Li, H.; Alizadeh, H.; Niederkorn, J.Y. PD-L1: PD-1 interaction contributes to the functional suppression of T-Cell responses to human uveal melanoma cells In Vitro. Investig. Ophtalmol. Vis. Sci. 2008, 49, 2518–2525. [Google Scholar] [CrossRef]
- Chen, P.; Mellon, J.K.; Mayhew, E.; Wang, S.; He, Y.G.; Hogan, N.; Niederkorn, J.Y. Uveal melanoma expression of indoleamine 2,3-deoxygenase: Establishment of an immune privileged environment by tryptophan depletion. Exp. Eye Res. 2007, 85, 617–625. [Google Scholar] [CrossRef] [PubMed]
- Ryu, Y.H.; Kim, J.C. Expression of indoleamine 2,3-dioxygenase in human corneal cells as a local immunosuppressive factor. Investig. Ophthalmol. Vis. Sci. 2007, 48, 4148–4152. [Google Scholar] [CrossRef] [PubMed]
- Long, G.V.; Dummer, R.; Hamid, O.; Gajewski, T.F.; Caglevic, C.; Dalle, S.; Arance, A.; Carlino, M.S.; Grob, J.J.; Kim, T.M.; et al. Epacadostat plus pembrolizumab versus placebo plus pembrolizumab in patients with unresectable or metastatic melanoma (ECHO-301/KEYNOTE-252): A phase 3, randomised, double-blind study. Lancet Oncol. 2019. [Google Scholar] [CrossRef]
- Sisley, K.; Rennie, I.G.; Parsons, M.A.; Jacques, R.; Hammond, D.W.; Bell, S.M.; Potter, A.M.; Rees, R.C. Abnormalities of chromosomes 3 and 8 in posterior uveal melanoma correlate with prognosis. Genes Chromosomes Cancer 1997, 19, 22–28. [Google Scholar] [CrossRef]
- White, J.S.; Becker, R.L.; McLean, I.W.; Director-Myska, A.E.; Nath, J. Molecular cytogenetic evaluation of 10 uveal melanoma cell lines. Cancer Genet Cytogenet. 2006, 168, 11–21. [Google Scholar] [CrossRef] [PubMed]
- Cross, N.A.; Ganesh, A.; Parpia, M.; Murray, A.K.; Rennie, I.G.; Sisley, K. Multiple locations on chromosome 3 are the targets of specific deletions in uveal melanoma. Eye 2006, 20, 476–481. [Google Scholar] [CrossRef]
- Van Gils, W.; Lodder, E.M.; Mensink, H.W. Gene expression profiling in uveal melanoma: Two regions on 3p related to prognosis. Investig. Ophthalmol. Vis. Sci. 2008, 49, 4254–4262. [Google Scholar] [CrossRef]
- Abdel-Rahman, M.H.; Cebulla, C.M.; Verma, V.; Christopher, B.N.; Carson, W.E., 3rd; Olencki, T.; Davidorf, F.H. Monosomy 3 status of uveal melanoma metastases is associated with rapidly progressive tumors and short survival. Exp. Eye Res. 2012, 100, 26–31. [Google Scholar] [CrossRef]
- Damato, B.; Duke, C.; Coupland, S.E.; Hiscott, P.; Smith, P.A.; Campbell, I.; Douglas, A.; Howard, P. Cytogenetics of uveal melanoma: A 7-year clinical experience. Ophthalmology 2007, 114, 1925–1931. [Google Scholar] [CrossRef]
- Maat, W.; Ly, L.V.; Jordanova, E.S.; de Wolff-Rouendaal, D.; Schalij-Delfos, N.E.; Jager, M.J. Monosomy of chromosome 3 and an inflammatory phenotype occur together in uveal melanoma. Investig. Ophthalmol. Vis. Sci. 2008, 49, 505–510. [Google Scholar] [CrossRef]
- Harbour, J.W.; Onken, M.D.; Roberson, E.D.; Duan, S.; Cao, L.; Worley, L.A.; Council, M.L.; Matatall, K.A.; Helms, C.; Bowcock, A.M. Frequent mutation of BAP1 in metastasizing uveal melanomas. Science 2010, 330, 1410–1413. [Google Scholar] [CrossRef] [PubMed]
- Van Raamsdonk, C.D.; Bezrookove, V.; Green, G.; Bauer, J.; Gaugler, L.; O’Brien, J.M.; Simpson, E.M.; Barsh, G.S.; Bastian, B.C. Frequent somatic mutations of GNAQ in uveal melanoma and blue naevi. Nature 2009, 457, 599–602. [Google Scholar] [CrossRef] [PubMed]
- Van Raamsdonk, C.D.; Griewank, K.G.; Crosby, M.B.; Garrido, M.C.; Vemula, S.; Wiesner, T.; Obenauf, A.C.; Wackernagel, W.; Green, G.; Bouvier, N.; et al. Mutations in GNA11 in uveal melanoma. N. Engl. J. Med. 2010, 363, 2191–2199. [Google Scholar] [CrossRef] [PubMed]
- Rodrigues, M.; Mobuchon, L.; Houy, A.; Fiévet, A.; Gardrat, S.; Barnhill, R.L.; Popova, T.; Servois, V.; Rampanou, A.; Mouton, A.; et al. Outlier response to anti-PD1 in uveal melanoma reveals germline MBD4 mutations in hypermutated tumors. Nat. Commun. 2018, 9, 1866. [Google Scholar] [CrossRef] [PubMed]
- De Vries, T.J.; Trancikova, D.; Ruiter, D.J.; Muijen, G.N. High expression of immunotherapy candidate proteins gp100, MART-I, tyrosinase and TRP-I in uveal melanoma. Br. J. Cancer 1998, 78, 1156–1161. [Google Scholar] [CrossRef] [PubMed]
- De Vries, T.J.; Fourkour, A.; Wobbes, T.; Verkroost, G.; Ruiter, D.J.; van Muijen, G.N. Heterogeneous expression of immunotherapy candidate proteins gp100, MART-1, and tyrosinase in human melanoma cell lines and in human melanocytic lesions. Cancer Res. 1997, 57, 3223–3229. [Google Scholar] [PubMed]
- Luyten, G.P.; van der Spek, C.W.; Sintnicolaas, K.; de Waard-Siebinga, I.; Jager, M.J.; de Jong, P.T.; Schrier, P.I.; Luider, T.M. Expression of MAGE, gp100 and tyrosinase genes in uveal melanoma cell lines. Melanoma Res. 1998, 8, 11–16. [Google Scholar] [CrossRef]
- Bol, K.F.; Mensink, H.W.; Aarntzen, E.H.; Schreibelt, G.; Keunen, J.E.; Coulie, P.G.; de Klein, A.; Punt, C.J.; Paridaens, D.; Figdor, C.G.; et al. Long overall survival after dendritic cell vaccination in metastatic uveal melanoma patients. Am. J. Ophthalmol. 2014, 158, 939–947. [Google Scholar] [CrossRef]
- Jindal, V. Role of immune checkpoint inhibitors and novel immunotherapies in uveal melanoma. Chin. Clin. Oncol. 2018, 7, 8. [Google Scholar] [CrossRef]
- Ellerhorst, J.A.; Cooksley, C.D.; Grimm, E.A. Autoimmunity and hypothyroidism in patients with uveal melanoma. Melanoma Res. 2001, 11, 633–637. [Google Scholar] [CrossRef]
- Chandran, S.S.; Somerville, R.P.; Yang, J.C.; Sherry, R.M.; Klebanoff, C.A.; Goff, S.L.; Wunderlich, J.R.; Danforth, D.N.; Zlott, D.; Paria, B.C.; et al. Treatment of metastatic uveal melanoma with adoptive transfer of tumour-infiltrating lymphocytes: A single-centre, two-stage, single-arm, phase 2 study. Lancet Oncol. 2017, 18, 792–802. [Google Scholar] [CrossRef]
- Dudley, M.E.; Wunderlich, J.R.; Shelton, T.E.; Even, J.; Rosenberg, S.A. Generation of tumor-infiltrating lymphocyte cultures for use in adoptive transfer therapy for melanoma patients. J. Immunother. 2003, 26, 332–342. [Google Scholar] [CrossRef] [PubMed]
- Wculek, S.K.; Amores-Iniesta, J.; Conde-Garrosa, R.; Khouili, S.C.; Melero, I.; Sancho, D. Effective cancer immunotherapy by natural mouse conventional type-1 dendritic cells bearing dead tumor antigen. J. Immunother. Cancer 2019, 7, 100. [Google Scholar] [CrossRef] [PubMed]

| Authors | Treatment | Type of Study | No. of Enrolled Patients | Year |
|---|---|---|---|---|
| Zimmer et al. [19] | Ipilimumab | Phase II trial. Pre-treated and naïve patients. | 53 | 2015 |
| Maio et al. [17] | Ipilimumab | Retrospective analysis. Pre-treated patients. | 82 | 2013 |
| Kelderman et al. [20] | Ipilimumab | Retrospective analysis. Pre-treated patients. | 22 | 2013 |
| Luke et al. [15] | Ipilimumab | Retrospective, multi-center analysis. Pre-treated and naïve patients. | 39 | 2013 |
| Piulats Rodriguez et al. [21] | Ipilimumab | Phase II trial. Naïve patients | 32 | 2014 |
| Danielli et al. [10] | Ipilimumab | Retrospective analysis. Pre-treated patients. | 13 | 2012 |
| Khattak et al. [13] | Ipilimumab | Retrospective analysis, single center analysis. Pre-treated patients. | 5 | 2016 |
| Deo [22] | Ipilimumab | Retrospective, single center analysis. Pre-treated patients. | 24 | 2014 |
| Shaw et al. [23] | Ipilimumab | EAP. | 18 | 2012 |
| Joshua et al. [11] | Tremelimumab | Phase II trial. Naïve patients. | 11 | 2015 |
| Algazi et al. [9] | Pembrolizumab, Nivolumab, Atezolizumab | Retrospective, multi-center analysis. Pre-treated and naïve patients. | 56 | 2016 |
| Mignard et al [16] | Pembrolizumab, Nivolumab, Ipilimumab | Retrospective, multi-center analysis. | 100 | 2018 |
| Bender et al. [27] | Pembrolizumab, Nivolumab | Retrospective, multi-center analysis. Pre-treated patients. | 15 | 2017 |
| Heppt et al. [24] | Pembrolizumab, Nivolumab, Ipilimumab | Retrospective, multi-center analysis. Pre-treated and naïve patients. | 96 | 2017 |
| Piperno-Neumann et al. [25] | Pembrolizumab, Nivolumab | Retrospective, single center analysis. Naïve patients. | 21 | 2016 |
| Karydis et al. [12] | Pembrolizumab | Retrospective analysis. Pre-treated patients. | 25 | 2016 |
| Rossi et al. [18] | Pembrolizumab | Prospective. Naïve patients. | 17 | 2019 |
| Kottschade et al. [14] | Pembrolizumab | Retrospective, single-center analysis. Pre-treated patients. | 8 | 2016 |
| Van der Kooij et al. [26] | Pembrolizumab | Prospective. Pre-treated and naïve patients. | 17 | 2017 |
| Schadendorf et al. [27] | Nivolumab | Phase II. Pre-treated patients. | 75 | 2017 |
| Jung et al. [28] | Ipilimumab | Named patient use. Pre-treated patients. | 10 | 2017 |
| Shoushtari et al. [29] | Nivolumab, Ipilimumab | Expanded access program. | 6 | 2016 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Rossi, E.; Schinzari, G.; Zizzari, I.G.; Maiorano, B.A.; Pagliara, M.M.; Sammarco, M.G.; Fiorentino, V.; Petrone, G.; Cassano, A.; Rindi, G.; et al. Immunological Backbone of Uveal Melanoma: Is There a Rationale for Immunotherapy? Cancers 2019, 11, 1055. https://doi.org/10.3390/cancers11081055
Rossi E, Schinzari G, Zizzari IG, Maiorano BA, Pagliara MM, Sammarco MG, Fiorentino V, Petrone G, Cassano A, Rindi G, et al. Immunological Backbone of Uveal Melanoma: Is There a Rationale for Immunotherapy? Cancers. 2019; 11(8):1055. https://doi.org/10.3390/cancers11081055
Chicago/Turabian StyleRossi, Ernesto, Giovanni Schinzari, Ilaria Grazia Zizzari, Brigida Anna Maiorano, Monica Maria Pagliara, Maria Grazia Sammarco, Vincenzo Fiorentino, Gianluigi Petrone, Alessandra Cassano, Guido Rindi, and et al. 2019. "Immunological Backbone of Uveal Melanoma: Is There a Rationale for Immunotherapy?" Cancers 11, no. 8: 1055. https://doi.org/10.3390/cancers11081055
APA StyleRossi, E., Schinzari, G., Zizzari, I. G., Maiorano, B. A., Pagliara, M. M., Sammarco, M. G., Fiorentino, V., Petrone, G., Cassano, A., Rindi, G., Bria, E., Blasi, M. A., Nuti, M., & Tortora, G. (2019). Immunological Backbone of Uveal Melanoma: Is There a Rationale for Immunotherapy? Cancers, 11(8), 1055. https://doi.org/10.3390/cancers11081055