Real-World, Long-Term Outcomes of Nivolumab Therapy for Recurrent or Metastatic Squamous Cell Carcinoma of the Head and Neck and Impact of the Magnitude of Best Overall Response: A Retrospective Multicenter Study of 88 Patients
Abstract
:Simple Summary
Abstract
1. Introduction
2. Results
2.1. Patient Characteristics
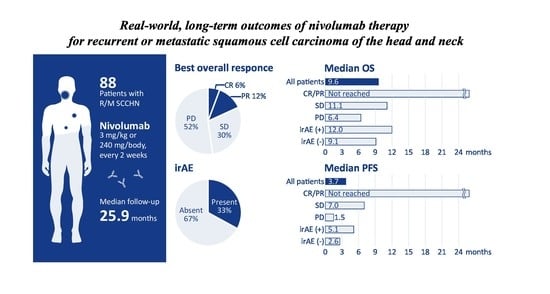
2.2. Treatment Outcomes
2.3. Immune-Related Adverse Events
2.4. Impact of Background Factors on Patient Survival
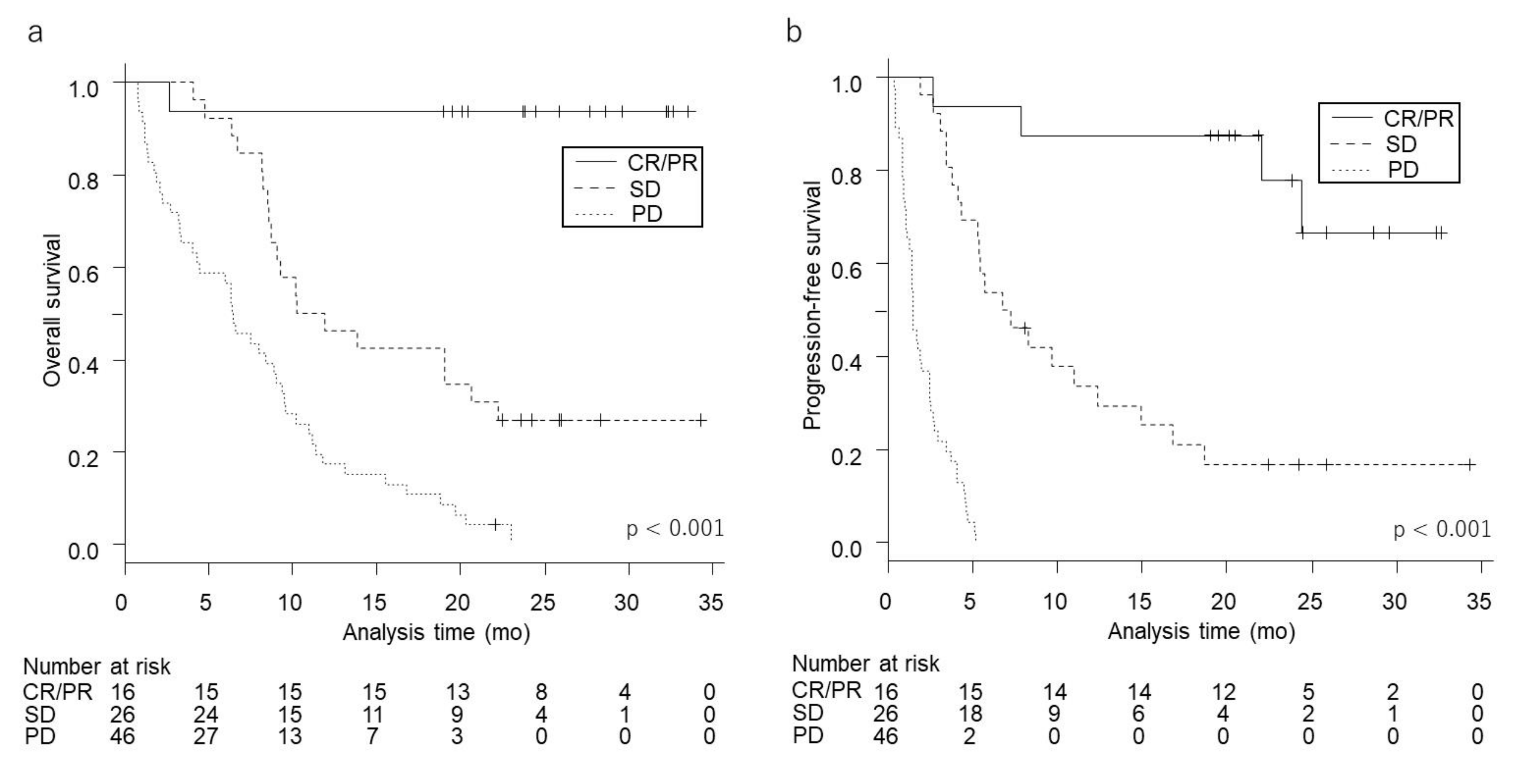
2.5. Survival According to Best Overall Response
3. Discussion
4. Materials and Methods
4.1. Patients
4.2. Treatment and Follow-up
4.3. Outcomes
4.4. Immune-Relate Adverse Events
4.5. Statistical Analysis
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Ferris, R.L.; Blumenschein, G., Jr.; Fayette, J.; Guigay, J.; Colevas, A.D.; Licitra, L.; Harrington, K.; Kasper, S.; Vokes, E.E.; Even, C.; et al. Nivolumab for Recurrent Squamous-Cell Carcinoma of the Head and Neck. N. Engl. J. Med. 2016, 375, 1856–1867. [Google Scholar] [CrossRef] [PubMed]
- Ferris, R.L.; Blumenschein, G., Jr.; Fayette, J.; Guigay, J.; Colevas, A.D.; Licitra, L.; Harrington, K.J.; Kasper, S.; Vokes, E.E.; Even, C.; et al. Nivolumab vs investigator’s choice in recurrent or metastatic squamous cell carcinoma of the head and neck: 2-year long-term survival update of CheckMate 141 with analyses by tumor PD-L1 expression. Oral Oncol. 2018, 81, 45–51. [Google Scholar] [CrossRef] [PubMed]
- Kiyota, N.; Hasegawa, Y.; Takahashi, S.; Yokota, T.; Yen, C.J.; Iwae, S.; Shimizu, Y.; Hong, R.L.; Goto, M.; Kang, J.H.; et al. A randomized, open-label, Phase III clinical trial of nivolumab vs. therapy of investigator’s choice in recurrent squamous cell carcinoma of the head and neck: A subanalysis of Asian patients versus the global population in checkmate 141. Oral Oncol. 2017, 73, 138–146. [Google Scholar] [CrossRef]
- Hori, R.; Shinohara, S.; Kojima, T.; Kagoshima, H.; Kitamura, M.; Tateya, I.; Tamaki, H.; Kumabe, Y.; Asato, R.; Harada, H.; et al. Real-World Outcomes and Prognostic Factors in Patients Receiving Nivolumab Therapy for Recurrent or Metastatic Head and Neck Carcinoma. Cancers 2019, 11, 1317. [Google Scholar] [CrossRef] [Green Version]
- Ueki, Y.; Takahashi, T.; Ota, H.; Shodo, R.; Yamazaki, K.; Horii, A. Predicting the treatment outcome of nivolumab in recurrent or metastatic head and neck squamous cell carcinoma: Prognostic value of combined performance status and modified Glasgow prognostic score. Eur. Arch. Otorhinolaryngol. 2020, 277, 2341–2347. [Google Scholar] [CrossRef] [PubMed]
- Inoue, H.; Yokota, T.; Hamauchi, S.; Onozawa, Y.; Kawakami, T.; Shirasu, H.; Notsu, A.; Yasui, H.; Onitsuka, T. Pre-treatment tumor size impacts on response to nivolumab in head and neck squamous cell carcinoma. Auris Nasus Larynx 2020, 47, 650–657. [Google Scholar] [CrossRef] [PubMed]
- Suzuki, C.; Kiyota, N.; Imamura, Y.; Rikitake, J.; Sai, S.; Koyama, T.; Hyogo, Y.; Nagatani, Y.; Funakoshi, Y.; Toyoda, M.; et al. Effect of tumor burden and growth rate on treatment outcomes of nivolumab in head and neck cancer. Int. J. Clin. Oncol. 2020, 25, 1270–1277. [Google Scholar] [CrossRef] [PubMed]
- Niwa, K.; Kawakita, D.; Nagao, T.; Takahashi, H.; Saotome, T.; Okazaki, M.; Yamazaki, K.; Okamoto, I.; Hirai, H.; Saigusa, N.; et al. Multicentre, retrospective study of the efficacy and safety of nivolumab for recurrent and metastatic salivary gland carcinoma. Sci. Rep. 2020, 10, 16988. [Google Scholar] [CrossRef] [PubMed]
- Fushimi, C.; Okamoto, I.; Matsuki, T.; Masubuchi, T.; Okada, T.; Sato, H.; Tsukahara, K.; Kondo, T.; Yamashita, T.; Hanyu, K.; et al. Salvage Chemotherapy After Nivolumab for Recurrent or Metastatic Head and Neck Carcinoma. Anticancer Res. 2020, 40, 5277–5283. [Google Scholar] [CrossRef]
- Matsuo, M.; Yasumatsu, R.; Masuda, M.; Toh, S.; Wakasaki, T.; Hashimoto, K.; Taura, M.; Uchi, R.; Nakagawa, T. Relationship between immune-related adverse events and the long-term outcomes in recurrent/metastatic head and neck squamous cell carcinoma treated with nivolumab. Oral Oncol. 2020, 101, 104525. [Google Scholar] [CrossRef]
- Okamoto, I.; Sato, H.; Kondo, T.; Koyama, N.; Fushimi, C.; Okada, T.; Miura, K.; Matsuki, T.; Yamashita, T.; Omura, G.; et al. Efficacy and safety of nivolumab in 100 patients with recurrent or metastatic head and neck cancer—A retrospective multicentre study. Acta Otolaryngol 2019, 139, 918–925. [Google Scholar] [CrossRef] [PubMed]
- Matsuki, T.; Okamoto, I.; Fushimi, C.; Sawabe, M.; Kawakita, D.; Sato, H.; Tsukahara, K.; Kondo, T.; Okada, T.; Tada, Y.; et al. Hematological predictive markers for recurrent or metastatic squamous cell carcinomas of the head and neck treated with nivolumab: A multicenter study of 88 patients. Cancer Med. 2020, 9, 5015–5024. [Google Scholar] [CrossRef] [PubMed]
- Kondo, T.; Okamoto, I.; Sato, H.; Koyama, N.; Fushimi, C.; Okada, T.; Masubuchi, T.; Miura, K.; Matsuki, T.; Yamashita, T.; et al. Age-based efficacy and safety of nivolumab for recurrent or metastatic head and neck squamous cell carcinoma: A multicenter retrospective study. Asia Pac. J. Clin. Oncol. 2020. [Google Scholar] [CrossRef]
- Nishikawa, D.; Suzuki, H.; Koide, Y.; Beppu, S.; Kadowaki, S.; Sone, M.; Hanai, N. Prognostic Markers in Head and Neck Cancer Patients Treated with Nivolumab. Cancers 2018, 10, 466. [Google Scholar] [CrossRef] [Green Version]
- Antonia, S.J.; Borghaei, H.; Ramalingam, S.S.; Horn, L.; De Castro Carpeño, J.; Pluzanski, A.; Burgio, M.A.; Garassino, M.; Chow, L.Q.M.; Gettinger, S.; et al. Four-year survival with nivolumab in patients with previously treated advanced non-small-cell lung cancer: A pooled analysis. Lancet Oncol. 2019, 20, 1395–1408. [Google Scholar] [CrossRef]
- Ishihara, H.; Takagi, T.; Kondo, T.; Tachibana, H.; Fukuda, H.; Yoshida, K.; Iizuka, J.; Kobayashi, H.; Okumi, M.; Ishida, H.; et al. Correlation between the magnitude of best tumor response and patient survival in nivolumab therapy for metastatic renal cell carcinoma. Med. Oncol. 2019, 36, 35. [Google Scholar] [CrossRef] [PubMed]
- Rogado, J.; Sánchez-Torres, J.M.; Romero-Laorden, N.; Ballesteros, A.I.; Pacheco-Barcia, V.; Ramos-Leví, A.; Arranz, R.; Lorenzo, A.; Gullón, P.; Donnay, O.; et al. Immune-related adverse events predict the therapeutic efficacy of anti-PD-1 antibodies in cancer patients. Eur. J. Cancer 2019, 109, 21–27. [Google Scholar] [CrossRef]
- Teraoka, S.; Fujimoto, D.; Morimoto, T.; Kawachi, H.; Ito, M.; Sato, Y.; Nagata, K.; Nakagawa, A.; Otsuka, K.; Uehara, K.; et al. Early Immune-Related Adverse Events and Association with Outcome in Advanced Non-Small Cell Lung Cancer Patients Treated with Nivolumab: A Prospective Cohort Study. J. Thorac. Oncol. 2017, 12, 1798–1805. [Google Scholar] [CrossRef]
- Haratani, K.; Hayashi, H.; Chiba, Y.; Kudo, K.; Yonesaka, K.; Kato, R.; Kaneda, H.; Hasegawa, Y.; Tanaka, K.; Takeda, M.; et al. Association of Immune-Related Adverse Events With Nivolumab Efficacy in Non-Small-Cell Lung Cancer. JAMA Oncol. 2018, 4, 374–378. [Google Scholar] [CrossRef]
- Ma, G.; Deng, Y.; Jiang, H.; Li, W.; Wu, Q.; Zhou, Q. The prognostic role of programmed cell death-ligand 1 expression in non-small cell lung cancer patients: An updated meta-analysis. Clin. Chim. Acta 2018, 482, 101–107. [Google Scholar] [CrossRef]
- Wang, Z.; Peng, S.; Xie, H.; Guo, L.; Cai, Q.; Shang, Z.; Jiang, N.; Niu, Y. Prognostic and clinicopathological significance of PD-L1 in patients with renal cell carcinoma: A meta-analysis based on 1863 individuals. Clin. Exp. Med. 2018, 18, 165–175. [Google Scholar] [CrossRef] [PubMed]
- Zhang, M.; Sun, H.; Zhao, S.; Wang, Y.; Pu, H.; Wang, Y.; Zhang, Q. Expression of PD-L1 and prognosis in breast cancer: A meta-analysis. Oncotarget 2017, 8, 31347–31354. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Troiano, G.; Caponio, V.C.A.; Zhurakivska, K.; Arena, C.; Pannone, G.; Mascitti, M.; Santarelli, A.; Lo Muzio, L. High PD-L1 expression in the tumour cells did not correlate with poor prognosis of patients suffering for oral squamous cells carcinoma: A meta-analysis of the literature. Cell Prolif. 2019, 52, e12537. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Jia, Y.Q.; Yang, B.; Wen, L.L.; Mu, W.X.; Wang, Z.; Cheng, B. Prognostic value of immune checkpoint molecules in head and neck cancer: A meta-analysis. Aging 2019, 11, 501–522. [Google Scholar] [CrossRef] [PubMed]
- Queirolo, P.; Spagnolo, F. Atypical responses in patients with advanced melanoma, lung cancer, renal-cell carcinoma and other solid tumors treated with anti-PD-1 drugs: A systematic review. Cancer Treat. Rev. 2017, 59, 71–78. [Google Scholar] [CrossRef]
- Lauber, K.; Dunn, L. Immunotherapy Mythbusters in Head and Neck Cancer: The Abscopal Effect and Pseudoprogression. Am. Soc. Clin. Oncol. Educ. Book 2019, 39, 352–363. [Google Scholar] [CrossRef]
- Ferrara, R.; Pilotto, S.; Caccese, M.; Grizzi, G.; Sperduti, I.; Giannarelli, D.; Milella, M.; Besse, B.; Tortora, G.; Bria, E. Do immune checkpoint inhibitors need new studies methodology? J. Thorac. Dis. 2018, 10, S1564–S1580. [Google Scholar] [CrossRef]
- Vivot, A.; Créquit, P.; Porcher, R. Use of Late-Life Expectancy for Assessing the Long-Term Benefit of Immune Checkpoint Inhibitors. J. Natl. Cancer Inst. 2019, 111, 519–521. [Google Scholar] [CrossRef]
- Burtness, B.; Harrington, K.J.; Greil, R.; Soulières, D.; Tahara, M.; de Castro, G., Jr.; Psyrri, A.; Basté, N.; Neupane, P.; Bratland, Å.; et al. Pembrolizumab alone or with chemotherapy versus cetuximab with chemotherapy for recurrent or metastatic squamous cell carcinoma of the head and neck (KEYNOTE-048): A randomised, open-label, phase 3 study. Lancet 2019, 394, 1915–1928. [Google Scholar] [CrossRef]
- Kanda, Y. Investigation of the freely available easy-to-use software ‘EZR’ for medical statistics. Bone Marrow Transplant. 2013, 48, 452–458. [Google Scholar] [CrossRef] [Green Version]

| Characteristic | N = 88 | (%) |
|---|---|---|
| Sex | ||
| Male | 71 | (81) |
| Female | 17 | (19) |
| Age | ||
| <65 years | 39 | (44) |
| ≥65 years | 49 | (56) |
| ECOG performance status | ||
| 0 | 59 | (67) |
| 1 | 24 | (27) |
| ≥2 | 5 | (6) |
| Primary site | ||
| Nasopharynx | 10 | (11) |
| Oropharynx | 19 | (22) |
| p16-positive | 8 | (9) |
| Hypopharynx | 21 | (24) |
| Larynx | 10 | (11) |
| Oral cavity | 17 | (19) |
| Paranasal sinus | 6 | (7) |
| Salivary gland | 2 | (2) |
| External auditory canal | 3 | (3) |
| Histology | ||
| Squamous cell carcinoma | 88 | (100) |
| Smoking | ||
| Ever | 61 | (69) |
| Never | 26 | (30) |
| Unknown | 1 | (1) |
| Alcohol | ||
| Ever | 69 | (78) |
| Never | 17 | (19) |
| Unknown | 2 | (2) |
| Distant metastasis | ||
| Present | 26 | (30) |
| Absent | 52 | (59) |
| Previous exposure to cetuximab | ||
| Yes | 30 | (34) |
| No | 58 | (66) |
| Platinum refractoriness a | ||
| Yes | 28 | (32) |
| No | 60 | (68) |
| Previous radiotherapy | ||
| Yes | 17 | (19) |
| No | 71 | (81) |
| Previous number of chemotherapy regimens b | ||
| 1 | 37 | (42) |
| 2 | 35 | (40) |
| ≥3 | 16 | (18) |
| PD-L1 expression | (0) | |
| Positive (≥1%) | 37 | (42) |
| Negative (<1%) | 7 | (8) |
| Unknown | 44 | (50) |
| Institution | (0) | |
| TMU | 41 | (47) |
| IUHW | 24 | (27) |
| KU | 15 | (17) |
| TMU/HMC | 8 | (9) |
| Best Overall Response | N = 88 | (%) |
|---|---|---|
| Complete response (CR) | 5 | (6) |
| Partial response (PR) | 11 | (13) |
| Stable disease (SD) | 26 | (30) |
| Progressive disease (PD) | 46 | (52) |
| Response rate (CR + PR) | 16 | (18) |
| Disease control rate (CR + PR + SD) | 42 | (48) |
| Immune-Related Adverse Events | Any Grade | Grade 3–5 |
|---|---|---|
| Interstitial lung disease | 13 | 5 |
| Thyroid dysfunction | 8 | 0 |
| Liver dysfunction | 5 | 3 |
| Skin disorders | 4 | 1 |
| Enteritis | 1 | 1 |
| Adrenal insufficiency | 1 | 0 |
| Infusion reactions | 1 | 0 |
| Myositis | 1 | 0 |
| Parathyroid dysfunction | 1 | 0 |
| Pituitary dysfunction | 1 | 0 |
| Renal dysfunction | 1 | 0 |
| Rheumatoid arthritis | 1 | 0 |
| Total | 38 | 10 |
| (29 patients) | (9 patients) |
| Overall Survival | Progression-Free Survival | |||||||
|---|---|---|---|---|---|---|---|---|
| Background Factors | Crude | Adjusted a | Crude | Adjusted a | ||||
| HR (95% CI) | p-Value | HR (95% CI) | p-Value | HR (95% CI) | p-Value | HR (95% CI) | p-Value | |
| Sex | ||||||||
| Male (Ref. Female) | 1.03 (0.54–1.98) | 0.922 | 0.54 (0.22–1.34) | 0.185 | 1.19 (0.64–2.23) | 0.581 | 0.64 (0.28–1.45) | 0.281 |
| Age | ||||||||
| ≥65 years (Ref. < 65 years) | 1.37 (0.83–2.24) | 0.216 | 1.27 (0.76–2.14) | 0.362 | 0.97 (0.61–1.56) | 0.914 | 0.79 (0.48–1.30) | 0.354 |
| ECOG performance status | ||||||||
| 2–3 (Ref. 0–1) | 16.91 (5.69–50.32) | <0.001 | 20.07 (6.35–63.39) | <0.001 | 6.00 (2.31–15.53) | <0.001 | 7.87 (2.77–22.35) | <0.001 |
| Smoking | ||||||||
| Ever (Ref. Never) | 1.17 (0.67–2.04) | 0.583 | 0.86 (0.42–1.75) | 0.669 | 1.16 (0.69–1.94) | 0.587 | 0.86 (0.45–1.66) | 0.660 |
| Alcohol consumption | ||||||||
| Ever (Ref. Never) | 1.51 (0.77–2.98) | 0.230 | 1.64 (0.67–4.03) | 0.278 | 1.61 (0.84–3.08) | 0.149 | 1.60 (0.71–3.60) | 0.257 |
| Distant metastasis | ||||||||
| Present (Ref. Absent) | 0.94 (0.57–1.55) | 0.818 | 0.95 (0.59–1.53) | 0.844 | ||||
| Previous exposure to cetuximab | ||||||||
| Yes (Ref. No) | 1.43 (0.84–2.45) | 0.188 | 1.71 (1.02–2.88) | 0.042 | ||||
| Platinum refractoriness | ||||||||
| Yes (Ref. No) | 1.69 (0.98–2.91) | 0.060 | 1.55 (0.80–3.01) | 0.196 | 1.80 (1.07–3.05) | 0.028 | 1.78 (0.96–3.30) | 0.067 |
| Previous radiotherapy | ||||||||
| Yes (Ref. No) | 1.05 (0.56–1.96) | 0.883 | 0.83 (0.46–1.50) | 0.539 | ||||
| Previous number of chemotherapy regimens | ||||||||
| two or more (Ref. 1) | 1.05 (0.63–1.73) | 0.860 | 1.12 (0.70–1.80) | 0.640 | ||||
| PD-L1 expression | ||||||||
| Positive (Ref. Negative) | 0.58 (0.25–1.35) | 0.203 | 0.50 (0.21–1.19) | 0.117 | ||||
| p16 expression | ||||||||
| Positive (Ref. Negative or Unknown) | 0.66 (0.28–1.53) | 0.332 | 0.76 (0.35–1.66) | 0.490 | ||||
| Immune-related adverse events | ||||||||
| Yes (Ref. No) | 0.60 (0.35–1.03) | 0.065 | 0.54 (0.31–0.96) | 0.036 | 0.59 (0.35–0.98) | 0.040 | 0.53 (0.31–0.92) | 0.023 |
| Overall Survival | |||||||||||
| Crude | |||||||||||
| N = 88 | (%) | median (months) | (95% CI) | 1-year OS (%) | (95% CI) | 2-year OS (%) | (95% CI) | HR | (95% CI) | p-value | |
| CR/PR | 16 | (18) | NR | 94 | (63–99) | 94 | (63–99) | 0.02 | (0.003–0.15) | <0.001 | |
| SD | 26 | (30) | 11.1 | (8.6–20.7) | 46 | (27–64) | 27 | (12–45) | 0.37 | (0.21–0.65) | 0.001 |
| PD | 46 | (52) | 6.4 | (3.3–9.0) | 17 | (8–30) | 0 | (NA-NA) | 1 (reference) | ||
| Progression-Free Survival | |||||||||||
| Crude | |||||||||||
| N = 88 | (%) | median (months) | (95% CI) | 1-year PFS (%) | (95% CI) | 2-year PFS (%) | (95% CI) | HR | (95% CI) | p-value | |
| CR/PR | 16 | (18) | NR | (22.1-NA) | 88 | (59–97) | 78 | (44–93) | 0.02 | (0.005–0.06) | <0.001 |
| SD | 26 | (30) | 7.0 | (4.3–12.4) | 34 | (16–52) | 17 | (5–34) | 0.10 | (0.05–0.22) | <0.001 |
| PD | 46 | (52) | 1.5 | (1.1–2.5) | NA | (NA-NA) | 0 | (NA-NA) | 1 (reference) | ||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Matsuki, T.; Okamoto, I.; Fushimi, C.; Takahashi, H.; Okada, T.; Kondo, T.; Sato, H.; Ito, T.; Tokashiki, K.; Tsukahara, K.; et al. Real-World, Long-Term Outcomes of Nivolumab Therapy for Recurrent or Metastatic Squamous Cell Carcinoma of the Head and Neck and Impact of the Magnitude of Best Overall Response: A Retrospective Multicenter Study of 88 Patients. Cancers 2020, 12, 3427. https://doi.org/10.3390/cancers12113427
Matsuki T, Okamoto I, Fushimi C, Takahashi H, Okada T, Kondo T, Sato H, Ito T, Tokashiki K, Tsukahara K, et al. Real-World, Long-Term Outcomes of Nivolumab Therapy for Recurrent or Metastatic Squamous Cell Carcinoma of the Head and Neck and Impact of the Magnitude of Best Overall Response: A Retrospective Multicenter Study of 88 Patients. Cancers. 2020; 12(11):3427. https://doi.org/10.3390/cancers12113427
Chicago/Turabian StyleMatsuki, Takashi, Isaku Okamoto, Chihiro Fushimi, Hideaki Takahashi, Takuro Okada, Takahito Kondo, Hiroki Sato, Tatsuya Ito, Kunihiko Tokashiki, Kiyoaki Tsukahara, and et al. 2020. "Real-World, Long-Term Outcomes of Nivolumab Therapy for Recurrent or Metastatic Squamous Cell Carcinoma of the Head and Neck and Impact of the Magnitude of Best Overall Response: A Retrospective Multicenter Study of 88 Patients" Cancers 12, no. 11: 3427. https://doi.org/10.3390/cancers12113427