Loss of CDKN1A mRNA and Protein Expression Are Independent Predictors of Poor Outcome in Chromophobe Renal Cell Carcinoma Patients
Abstract
:1. Introduction
2. Results
2.1. Chromosomal Loss and Patient Outcome
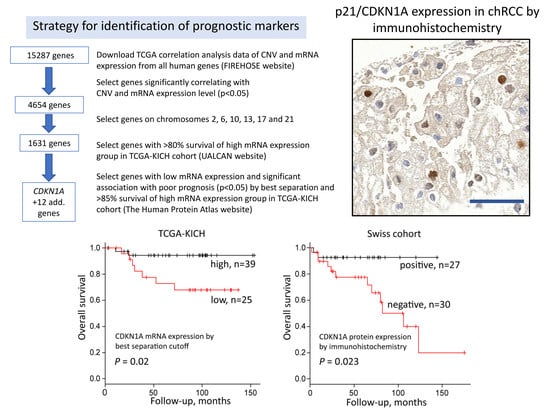
2.2. Identification of Genes Associated with Chromosomal Loss, Decreased Expression and Patient Survival
2.3. CDKN1A mRNA and Protein Expression
2.4. CDKN1A Expression, Tumor Stage, Grade and Outcome
3. Discussion
4. Materials and Methods
4.1. Data Acquisition and Processing Using the Cancer Genome Atlas Data Portal
4.2. Strategy for Gene Candidate Selection
4.3. Swiss Chromophobe Renal Cell Carcinomas
4.4. OncoScan Assay
4.5. Immunohistochemistry
4.6. Statistical Analysis
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Paner, G.; Amin, M.B.; Moch, H.; Störkel, S. Chromophobe renal cell carcinoma. In WHO Classification of Tumours of the Urinary System and Male Genital Organs, 4th ed.; Moch, H., Humphrey, P.A., Ulbright, T.M., Reuter, V.E., Eds.; International Agency for Research on Cancer: Lyon, France, 2016; pp. 27–28. [Google Scholar]
- Thoenes, W.; Störkel, S.; Rumpelt, H.J.; Moll, R.; Baum, H.P.; Werner, S. Chromophobe cell renal carcinoma and its variants—A report on 32 cases. J. Pathol. 1988, 155, 277–287. [Google Scholar] [CrossRef]
- Cheville, J.C.; Lohse, C.M.; Zincke, H.; Weaver, A.L.; Blute, M.L. Comparisons of outcome and prognostic features among histologic subtypes of renal cell carcinoma. Am. J. Surg. Pathol. 2003, 27, 612–624. [Google Scholar] [CrossRef] [PubMed]
- Volpe, A.; Novara, G.; Antonelli, A.; Bertini, R.; Billia, M.; Carmignani, G.; Cunico, S.C.; Longo, N.; Martignoni, G.; Minervini, A.; et al. Chromophobe renal cell carcinoma (RCC): Oncological outcomes and prognostic factors in a large multicentre series. BJU Int. 2012, 110, 76–83. [Google Scholar] [CrossRef] [PubMed]
- Yap, N.Y.; Rajandram, R.; Ng, K.L.; Pailoor, J.; Fadzli, A.; Gobe, G.C. Genetic and chromosomal aberrations and their clinical significance in renal neoplasms. Biomed. Res. Int. 2015, 2015, 476508. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Przybycin, C.G.; Cronin, A.M.; Darvishian, F.; Gopalan, A.; Al-Ahmadie, H.A.; Fine, S.W.; Chen, Y.B.; Bernstein, M.; Russo, P.; Reuter, V.E.; et al. Chromophobe renal cell carcinoma: A clinicopathologic study of 203 tumors in 200 patients with primary resection at a single institution. Am. J. Surg. Pathol. 2011, 35, 962–970. [Google Scholar] [CrossRef] [PubMed]
- Casuscelli, J.; Weinhold, N.; Gundem, G.; Wang, L.; Zabor, E.C.; Drill, E.; Wang, P.I.; Nanjangud, G.J.; Redzematovic, A.; Nargund, A.M.; et al. Genomic landscape and evolution of metastatic chromophobe renal cell carcinoma. JCI Insight 2017, 2. [Google Scholar] [CrossRef]
- Delahunt, B.; Sika-Paotonu, D.; Bethwaite, P.B.; McCredie, M.R.; Martignoni, G.; Eble, J.N.; Jordan, T.W. Fuhrman grading is not appropriate for chromophobe renal cell carcinoma. Am. J. Surg. Pathol. 2007, 31, 957–960. [Google Scholar] [CrossRef]
- Amin, M.B.; Paner, G.P.; Alvarado-Cabrero, I.; Young, A.N.; Stricker, H.J.; Lyles, R.H.; Moch, H. Chromophobe renal cell carcinoma: Histomorphologic characteristics and evaluation of conventional pathologic prognostic parameters in 145 cases. Am. J. Surg. Pathol. 2008, 32, 1822–1834. [Google Scholar] [CrossRef]
- Paner, G.P.; Amin, M.B.; Alvarado-Cabrero, I.; Young, A.N.; Stricker, H.J.; Moch, H.; Lyles, R.H. A novel tumor grading scheme for chromophobe renal cell carcinoma: Prognostic utility and comparison with Fuhrman nuclear grade. Am. J. Surg. Pathol. 2010, 34, 1233–1240. [Google Scholar] [CrossRef]
- Xie, Y.; Ma, X.; Li, H.; Gao, Y.; Gu, L.; Chen, L.; Zhang, X. Prognostic value of clinical and pathological features in chinese patients with chromophobe renal cell carcinoma: A 10-year single-center study. J. Cancer 2017, 8, 3474–3479. [Google Scholar] [CrossRef]
- Finley, D.S.; Shuch, B.; Said, J.W.; Galliano, G.; Jeffries, R.A.; Afifi, A.A.; Castor, B.; Magyar, C.; Sadaat, A.; Kabbinavar, F.F.; et al. The chromophobe tumor grading system is the preferred grading scheme for chromophobe renal cell carcinoma. J. Urol. 2011, 186, 2168–2174. [Google Scholar] [CrossRef] [PubMed]
- Leibovich, B.C.; Lohse, C.M.; Cheville, J.C.; Zaid, H.B.; Boorjian, S.A.; Frank, I.; Thompson, R.H.; Parker, W.P. Predicting oncologic outcomes in renal cell carcinoma after surgery. Eur. Urol. 2018, 73, 772–780. [Google Scholar] [CrossRef] [PubMed]
- Ohashi, R.; Schraml, P.; Angori, S.; Batavia, A.A.; Rupp, N.J.; Ohe, C.; Otsuki, Y.; Kawasaki, T.; Kobayashi, H.; Kobayashi, K.; et al. Classic chromophobe renal cell carcinoma incur a larger number of chromosomal losses than seen in the eosinophilic subtype. Cancers 2019, 11, 1492. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ohashi, R.; Schraml, P.; Batavia, A.; Angori, S.; Simmler, P.; Rupp, N.; Ajioka, Y.; Oliva, E.; Moch, H. Allele Loss and reduced expression of CYCLOPS genes is a characteristic feature of chromophobe renal cell carcinoma. Transl. Oncol. 2019, 12, 1131–1137. [Google Scholar] [CrossRef] [PubMed]
- Davis, C.F.; Ricketts, C.J.; Wang, M.; Yang, L.; Cherniack, A.D.; Shen, H.; Buhay, C.; Kang, H.; Kim, S.C.; Fahey, C.C.; et al. The somatic genomic landscape of chromophobe renal cell carcinoma. Cancer Cell 2014, 26, 319–330. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Durinck, S.; Stawiski, E.W.; Pavía-Jiménez, A.; Modrusan, Z.; Kapur, P.; Jaiswal, B.S.; Zhang, N.; Toffessi-Tcheuyap, V.; Nguyen, T.T.; Pahuja, K.B.; et al. Spectrum of diverse genomic alterations define non-clear cell renal carcinoma subtypes. Nat. Genet. 2015, 47, 13–21. [Google Scholar] [CrossRef] [Green Version]
- Brunelli, M.; Eble, J.N.; Zhang, S.; Martignoni, G.; Delahunt, B.; Cheng, L. Eosinophilic and classic chromophobe renal cell carcinomas have similar frequent losses of multiple chromosomes from among chromosomes 1, 2, 6, 10 and 17, and this pattern of genetic abnormality is not present in renal oncocytoma. Mod. Pathol. 2005, 18, 161–169. [Google Scholar] [CrossRef]
- Quddus, M.B.; Pratt, N.; Nabi, G. Chromosomal aberrations in renal cell carcinoma: An overview with implications for clinical practice. Urol. Ann. 2019, 11, 6–14. [Google Scholar]
- Chandrashekar, D.S.; Bashel, B.; Balasubramanya, S.A.H.; Creighton, C.J.; Ponce-Rodriguez, I.; Chakravarthi, B.V.S.K.; Varambally, S. UALCAN: A portal for facilitating tumor subgroup gene expression and survival analyses. Neoplasia 2017, 19, 649–658. [Google Scholar] [CrossRef]
- UALCAN. TCGA Analysis. Available online: http://ualcan.path.uab.edu/analysis.html (accessed on 25 March 2018).
- Thul, P.J.; Lindskog, C. The human protein atlas: A spatial map of the human proteome. Protein Sci. 2018, 27, 233–244. [Google Scholar] [CrossRef] [Green Version]
- The Human Protein Atlas. Available online: https://www.proteinatlas.org/ (accessed on 25 March 2018).
- Broad GDAC FIREHOSE-Broad Institute. Available online: http://gdac.broadinstitute.org/ (accessed on 23 March 2018).
- Abbas, T.; Dutta, A. p21 in cancer: Intricate networks and multiple activities. Nat. Rev. Cancer 2009, 9, 400–414. [Google Scholar] [CrossRef] [PubMed]
- NCBI > Genes & Expression > Gene. National Center for Biotechnology Information, U.S. National Library of Medicine. Available online: https://www.ncbi.nlm.nih.gov/gene/ (accessed on 16 April 2019).
- GeneCards. The Human Gene Database. Available online: https://www.genecards.org/ (accessed on 21 November 2019).
- Georgakilas, A.G.; Martin, O.A.; Bonner, W.M. p21: A two-faced genome guardian. Trends Mol. Med. 2017, 23, 310–319. [Google Scholar] [CrossRef] [PubMed]
- Dahinden, C.; Ingold, B.; Wild, P.; Boysen, G.; Luu, V.D.; Montani, M.; Kristiansen, G.; Sulser, T.; Bühlmann, P.; Moch, H.; et al. Mining tissue microarray data to uncover combinations of biomarker expression patterns that improve intermediate staging and grading of clear cell renal cell cancer. Clin. Cancer Res. 2010, 16, 88–98. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ohashi, R.; Martignoni, G.; Hartmann, A.; Caliò, A.; Segala, D.; Stöhr, C.; Wach, S.; Erlmeier, F.; Weichert, W.; Autenrieth, M.; et al. Multi-institutional re-evaluation of prognostic factors in chromophobe renal cell carcinoma: Proposal of a novel two-tiered grading scheme. Virchows Arch. 2019, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Kreis, N.N.; Friemel, A.; Zimmer, B.; Roth, S.; Rieger, M.A.; Rolle, U.; Louwen, F.; Yuan, J. Mitotic p21Cip1/CDKN1A is regulated by cyclin-dependent kinase 1 phosphorylation. Oncotarget 2016, 7, 50215–50228. [Google Scholar] [CrossRef] [Green Version]
- Willenbring, H.; Sharma, A.D.; Vogel, A.; Lee, A.Y.; Rothfuss, A.; Wang, Z.; Finegold, M.; Grompe, M. Loss of p21 permits carcinogenesis from chronically damaged liver and kidney epithelial cells despite unchecked apoptosis. Cancer Cell. 2008, 14, 59–67. [Google Scholar] [CrossRef] [Green Version]
- Roninson, I.B. Oncogenic functions of tumour suppressor p21Waf1/Cip1/Sdi1: Association with cell senescence and tumour-promoting activities of stromal fibroblasts. Cancer Lett. 2002, 179, 1–14. [Google Scholar] [CrossRef]
- Kreis, N.N.; Louwen, F.; Yuan, J. The multifaceted p21 (Cip1/Waf1/CDKN1A) in cell differentiation, migration and cancer therapy. Cancers 2019, 11, 1220. [Google Scholar] [CrossRef] [Green Version]
- Freeman, D.J.; Li, A.G.; Wei, G.; Li, H.H.; Kertesz, N.; Lesche, R.; Whale, A.D.; Martinez-Diaz, H.; Rozengurt, N.; Cardiff, R.D.; et al. PTEN tumor suppressor regulates p53 protein levels and activity through phosphatase-dependent and -independent mechanisms. Cancer Cell. 2003, 3, 117–130. [Google Scholar] [CrossRef] [Green Version]
- Tang, Y.; Eng, C. p53 down-regulates phosphatase and tensin homologue deleted on chromosome 10 protein stability partially through caspase-mediated degradation in cells with proteasome dysfunction. Cancer Res. 2006, 66, 6139–6148. [Google Scholar] [CrossRef] [Green Version]
- Li, A.G.; Piluso, L.G.; Cai, X.; Wei, G.; Sellers, W.R.; Liu, X. Mechanistic insights into maintenance of high p53 acetylation by PTEN. Mol. Cell 2006, 23, 575–587. [Google Scholar] [CrossRef] [PubMed]
- Sarbia, M.; Stahl, M.; zur Hausen, A.; Zimmermann, K.; Wang, L.; Fink, U.; Heep, H.; Dutkowski, P.; Willers, R.; Müller, W.; et al. Expression of p21WAF1 predicts outcome of esophageal cancer patients treated by surgery alone or by combined therapy modalities. Clin. Cancer Res. 1998, 4, 2615–2623. [Google Scholar] [PubMed]
- Lin, Y.; Shen, L.Y.; Fu, H.; Dong, B.; Yang, H.L.; Yan, W.P.; Kang, X.Z.; Dai, L.; Zhou, H.T.; Yang, Y.B.; et al. P21, COX-2, and E-cadherin are potential prognostic factors for esophageal squamous cell carcinoma. Dis. Esophagus 2017, 30, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Ferrandina, G.; Stoler, A.; Fagotti, A.; Fanfani, F.; Sacco, R.; De Pasqua, A.; Mancuso, S.; Scambia, G. p21WAF1/CIP1 protein expression in primary ovarian cancer. Int. J. Oncol. 2000, 17, 1231–1235. [Google Scholar] [CrossRef] [PubMed]
- Baretton, G.; Klenk, U.; Diebold, J.; Schmeller, N.; Löhrs, U. Proliferation-and apoptosis-associated factors in advanced prostatic carcinomas before and after androgen deprivation therapy: Prognostic significance of p21/WAF1/CIP1 expression. Br. J. Cancer 1999, 80, 546. [Google Scholar] [CrossRef]
- Aaltomaa, S.; Lipponen, P.; Eskelinen, M.; Ala-Opas, M.; Kosma, V. Prognostic value and expression of p21 (waf1/cip1) protein in prostate cancer. Prostate 1999, 39, 8–15. [Google Scholar] [CrossRef]
- Korkolopoulou, P.; Kouzelis, K.; Christodoulou, P.; Papanikolaou, A.; Thomas-Tsagli, E. Expression of retinoblastoma gene product and p21 (WAF1/Cip 1) protein in gliomas: Correlations with proliferation markers, p53 expression and survival. Acta Neuropathol. 1998, 95, 617–624. [Google Scholar] [CrossRef]
- Caffo, O.; Doglioni, C.; Veronese, S.; Bonzanini, M.; Marchetti, A.; Buttitta, F.; Fina, P.; Leek, R.; Morelli, L.; Palma, P.D.; et al. Prognostic value of p21(WAF1) and p53 expression in breast carcinoma: An immunohistochemical study in 261 patients with long-term follow-up. Clin. Cancer Res. 1996, 2, 1591–1599. [Google Scholar] [CrossRef]
- Lu, X.; Toki, T.; Konishi, I.; Nikaido, T.; Fujii, S. Expression of p21WAF1/CIP1 in adenocarcinoma of the uterine cervix: A possible immunohistochemical marker of a favorable prognosis. Cancer 1998, 82, 2409–2417. [Google Scholar] [CrossRef]
- Ogawa, M.; Onoda, N.; Maeda, K.; Kato, Y.; Nakata, B.; Kang, S.M.; Sowa, M.; Hirakawa, K. A combination analysis of p53 and p21 in gastric carcinoma as a strong indicator for prognosis. Int. J. Mol. Med. 2001, 7, 479–483. [Google Scholar] [CrossRef]
- Anttila, M.A.; Kosma, V.M.; Hongxiu, J.; Puolakka, J.; Juhola, M.; Saarikoski, S.; Syrjänen, K. p21/WAF1 expression as related to p53, cell proliferation and prognosis in epithelial ovarian cancer. Br. J. Cancer. 1999, 79, 1870. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Mukherjee, S.; Conrad, S.E. c-Myc suppresses p21WAF1/CIP1 expression during estrogen signaling and antiestrogen resistance in human breast cancer cells. J. Biol. Chem. 2005, 280, 17617–17625. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Alam, S.; Sen, E.; Brashear, H.; Meyers, C. Adeno-associated virus type 2 increases proteosome-dependent degradation of p21WAF1 in a human papillomavirus type 31b-positive cervical carcinoma line. J. Virol. 2006, 80, 4927–4939. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- El-Deiry, W.S. p21 (WAF1) mediates cell-cycle inhibition, relevant to cancer suppression and therapy. Cancer Res. 2016, 76, 5189–5191. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zhou, B.P.; Liao, Y.; Xia, W.; Spohn, B.; Lee, M.H.; Hung, M.C. Cytoplasmic localization of p21Cip1/WAF1 by Akt-induced phosphorylation in HER-2/neu-overexpressing cells. Nat. Cell Biol. 2001, 3, 245–252. [Google Scholar] [CrossRef] [PubMed]
- Winters, Z.E.; Hunt, N.C.; Bradburn, M.J.; Royds, J.A.; Turley, H.; Harris, A.L.; Norbury, C.J. Subcellular localisation of cyclin B, Cdc2 and p21(WAF1/CIP1) in breast cancer. association with prognosis. Eur. J. Cancer 2001, 37, 2405–2412. [Google Scholar] [CrossRef]
- Xia, W.; Chen, J.S.; Zhou, X.; Sun, P.R.; Lee, D.F.; Liao, Y.; Zhou, B.P.; Hung, M.C. Phosphorylation/cytoplasmic localization of p21Cip1/WAF1 is associated with HER2/neu overexpression and provides a novel combination predictor for poor prognosis in breast cancer patients. Clin. Cancer Res. 2004, 10, 3815–3824. [Google Scholar] [CrossRef] [Green Version]
- Ohata, M.; Nakamura, S.; Fujita, H.; Isemura, M. Prognostic implications of p21 (Waf1/Cip1) immunolocalization in multiple myeloma. Biomed. Res. 2005, 26, 91–98. [Google Scholar] [CrossRef] [Green Version]
- Voith von Voithenberg, L.; Fomitcheva Khartchenko, A.; Huber, D.; Schraml, P.; Kaigala, G.V. Spatially multiplexed RNA in situ hybridization to reveal tumor heterogeneity. Nucleic Acids Res. 2019. [Google Scholar] [CrossRef] [Green Version]
- Digital Slide Archive (DSA). Available online: https://cancer.digitalslidearchive.org/ (accessed on 9 May 2019).
- GDC Data Portal-National Cancer Institute. Available online: https://portal.gdc.cancer.gov/ (accessed on 23 March 2019).
- Sun, M.; Tong, P.; Kong, W.; Dong, B.; Huang, Y.; Park, I.Y.; Zhou, L.; Liu, X.D.; Ding, Z.; Zhang, X.; et al. HNF1B loss exacerbates the development of chromophobe renal cell carcinomas. Cancer Res. 2017, 77, 5313–5326. [Google Scholar] [CrossRef] [Green Version]
- Brierley, J.D.; Gospodarowicz, M.K.; Wittekind, C. TNM Classification of Malignant Tumours; John Wiley & Sons: Hoboken, NJ, USA, 2016. [Google Scholar]
- Deml, K.F.; Schildhaus, H.U.; Compérat, E.; von Teichman, A.; Storz, M.; Schraml, P.; Bonventre, J.V.; Fend, F.; Fleige, B.; Nerlich, A.; et al. Clear cell papillary renal cell carcinoma and renal angiomyoadenomatous tumor: Two variants of a morphologic, immunohistochemical, and genetic distinct entity of renal cell carcinoma. Am. J. Surg. Pathol. 2015, 39, 889–901. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Noske, A.; Brandt, S.; Valtcheva, N.; Wagner, U.; Zhong, Q.; Bellini, E.; Fink, D.; Obermann, E.C.; Moch, H.; Wild, P.J. Detection of CCNE1/URI (19q12) amplification by in situ hybridisation is common in high grade and type II endometrial cancer. Oncotarget 2017, 8, 14794–14805. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bihr, S.; Ohashi, R.; Moore, A.L.; Rüschoff, J.H.; Beisel, C.; Hermanns, T.; Mischo, A.; Corrò, C.; Beyer, J.; Beerenwinkel, N.; et al. Expression and mutation patterns of PBRM1, BAP1 and SETD2 mirror specific evolutionary subtypes in clear cell renal cell carcinoma. Neoplasia 2019, 21, 247–256. [Google Scholar] [CrossRef] [PubMed]
- McCarty, K.S., Jr.; Szabo, E.; Flowers, J.L.; Cox, E.B.; Leight, G.S.; Miller, L.; Konrath, J.; Soper, J.T.; Budwit, D.A.; Creasman, W.T.; et al. Use of a monoclonal anti-estrogen receptor antibody in the immunohistochemical evaluation of human tumors. Cancer Res. 1986, 46, 4244s–4248s. [Google Scholar] [PubMed]
- Kanda, Y. Investigation of the freely-available easy-to-use software “EZR” (Easy R) for medical statistics. Bone Marrow Transplant. 2013, 48, 452–458. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Firth, D. Bias reduction of maximum likelihood estimates. Biometrika 1993, 80, 27–38. [Google Scholar] [CrossRef]
- coxphf: Cox Regression with Firth’s Penalized Likelihood. Available online: https://CRAN.R-project.org/package=coxphf, https://cran.r-project.org/web/packages/coxphf/coxphf.pdf (accessed on 10 October 2018).




| Gene Name | Chromosomal Locus 1 | CNV vs mRNA Pearson’s Correlation Coefficient 2 | Protein Expression 3 | Protein Function (GeneCards 4) |
|---|---|---|---|---|
| CDKN1A | 6p21.2 | R = 0.4434, p = 0.0002 | nucleus | Cell cycle regulation |
| KLF6 | 10p15.2 | R = 0.5474, p < 0.0001 | nucleus | Transcriptional activator |
| FAM160B1 | 10q25.3 | R = 0.7632, p < 0.0001 | cytoplasm | unknown |
| PAOX | 10q26.3 | R = 0.6088, p < 0.0001 | cytoplasm | Polyamine oxidase |
| PWWP2B | 10q26.3 | R = 0.52, p < 0.0001 | cytoplasm | unknown |
| FBXW4 | 10q24.32 | R = 0.4296, p = 0.0003 | golgi | Ubiquitination |
| FBXL15 | 10q24.32 | R = 0.4048, p = 0.0007 | cytoplasm | Ubiquitination |
| CASKIN2 | 17q25.1 | R = 0.4364, p = 0.0002 | cytoplasm | unknown |
| RTN4RL1 | 17p13.3 | R = 0.4013, p = 0.0008 | secreted | Brain development |
| FMNL1 | 17q21.31 | R = 0.3974. p = 0.001 | cytoplasm | Regulation of cell morphology |
| RAB37 | 17q25.1 | R = 0.369, p = 0.002 | cytoplasm | GTPase |
| SOCS3 | 17q25.3 | R = 0.3611, p = 0.003 | cytoplasm | Cytokine signaling suppression |
| C21orf2 | 21q22.3 | R = 0.5435, p < 0.0001 | mitochondria | Regulation of cell morphology, DNA damage repair |
| Cohort | TCGA-KICH | Swiss Patients | ||||||
|---|---|---|---|---|---|---|---|---|
| Variables | Univariate | Multivariate2 | Univariate | Multivariate | ||||
| HR (95%CI) | p-value | HR (95%CI) | p-value | HR (95%CI) | p-value | HR (95%CI) | p-value | |
| Tumor stage (3–4 vs 1–2) 1 | 10.22 (2.12–49.29) | 0.004 | 6.442 (1.488–37.214) | 0.012 | 1.447 (0.398–5.264) | n.s. | 1.266 (0.343–4.678) | n.s. |
| Grade (High vs Low) | 18.03 (4.448–73.05) | <0.001 | 6.087 (1.374–32.266) | 0.017 | - | - | - | - |
| CDKN1A expression (Low vs High) 2, 3 | 22.528 (2.862–2904.443) | <0.001 | 12.527 (1.289–1675.059) | 0.026 | 4.812 (1.07–21.64) | <0.05 | 4.741 (1.051–21.390) | <0.05 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ohashi, R.; Angori, S.; Batavia, A.A.; Rupp, N.J.; Ajioka, Y.; Schraml, P.; Moch, H. Loss of CDKN1A mRNA and Protein Expression Are Independent Predictors of Poor Outcome in Chromophobe Renal Cell Carcinoma Patients. Cancers 2020, 12, 465. https://doi.org/10.3390/cancers12020465
Ohashi R, Angori S, Batavia AA, Rupp NJ, Ajioka Y, Schraml P, Moch H. Loss of CDKN1A mRNA and Protein Expression Are Independent Predictors of Poor Outcome in Chromophobe Renal Cell Carcinoma Patients. Cancers. 2020; 12(2):465. https://doi.org/10.3390/cancers12020465
Chicago/Turabian StyleOhashi, Riuko, Silvia Angori, Aashil A. Batavia, Niels J. Rupp, Yoichi Ajioka, Peter Schraml, and Holger Moch. 2020. "Loss of CDKN1A mRNA and Protein Expression Are Independent Predictors of Poor Outcome in Chromophobe Renal Cell Carcinoma Patients" Cancers 12, no. 2: 465. https://doi.org/10.3390/cancers12020465