Discoidin Domain Receptor-1 (DDR1) is Involved in Angiolymphatic Invasion in Oral Cancer
Abstract
1. Introduction
2. Results
2.1. Clinical Relevance of DDR1 Overexpression and ALI
2.2. Expression Levels of DDR1 and Basement Membrane Type IV Collagen are Positively Correlated
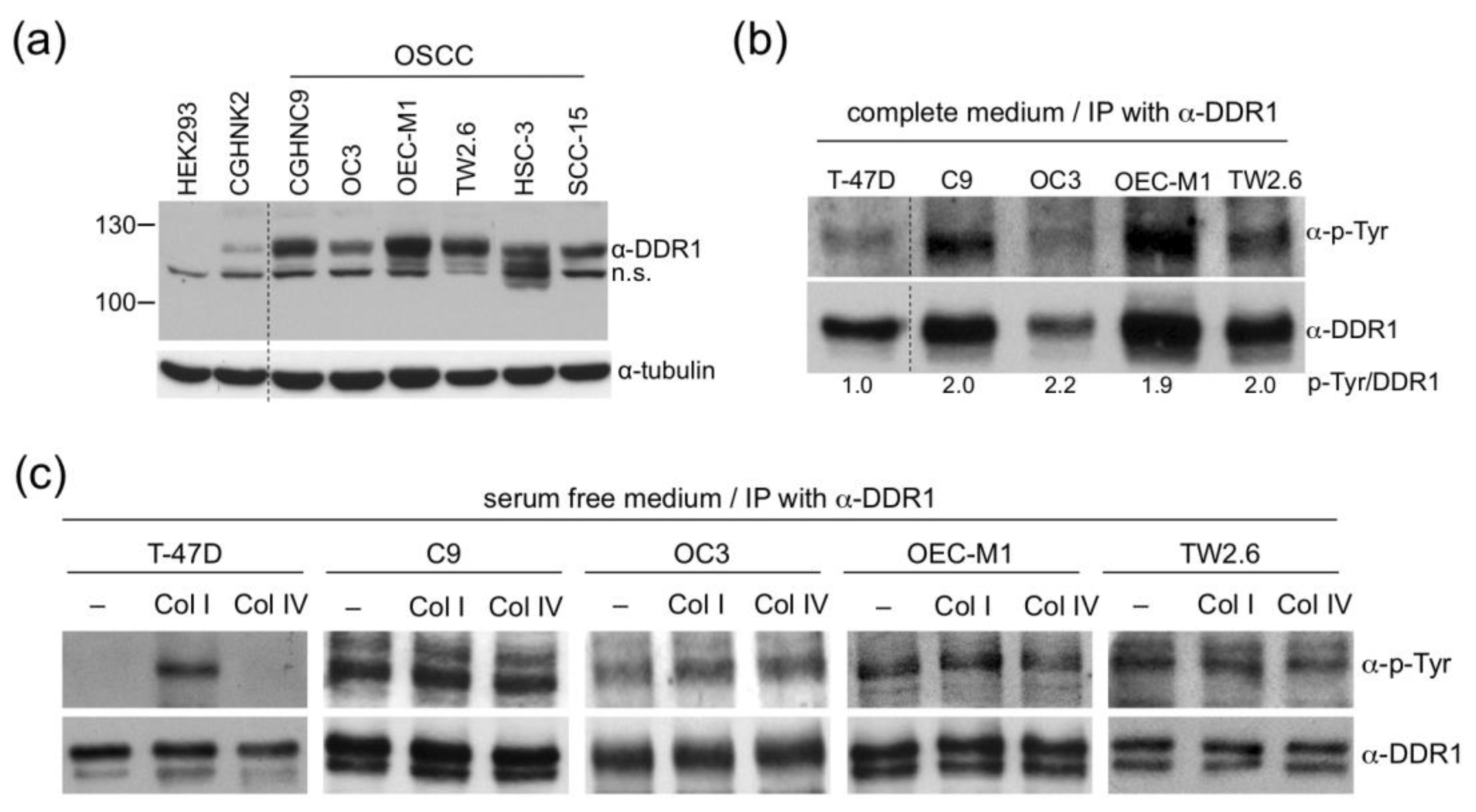
2.3. DDR1 in OSCC Cells is Ligand-Independent and Constitutively Active
2.4. Constitutively Active DDR1 Contributes to Cell Growth and Clonogenicity
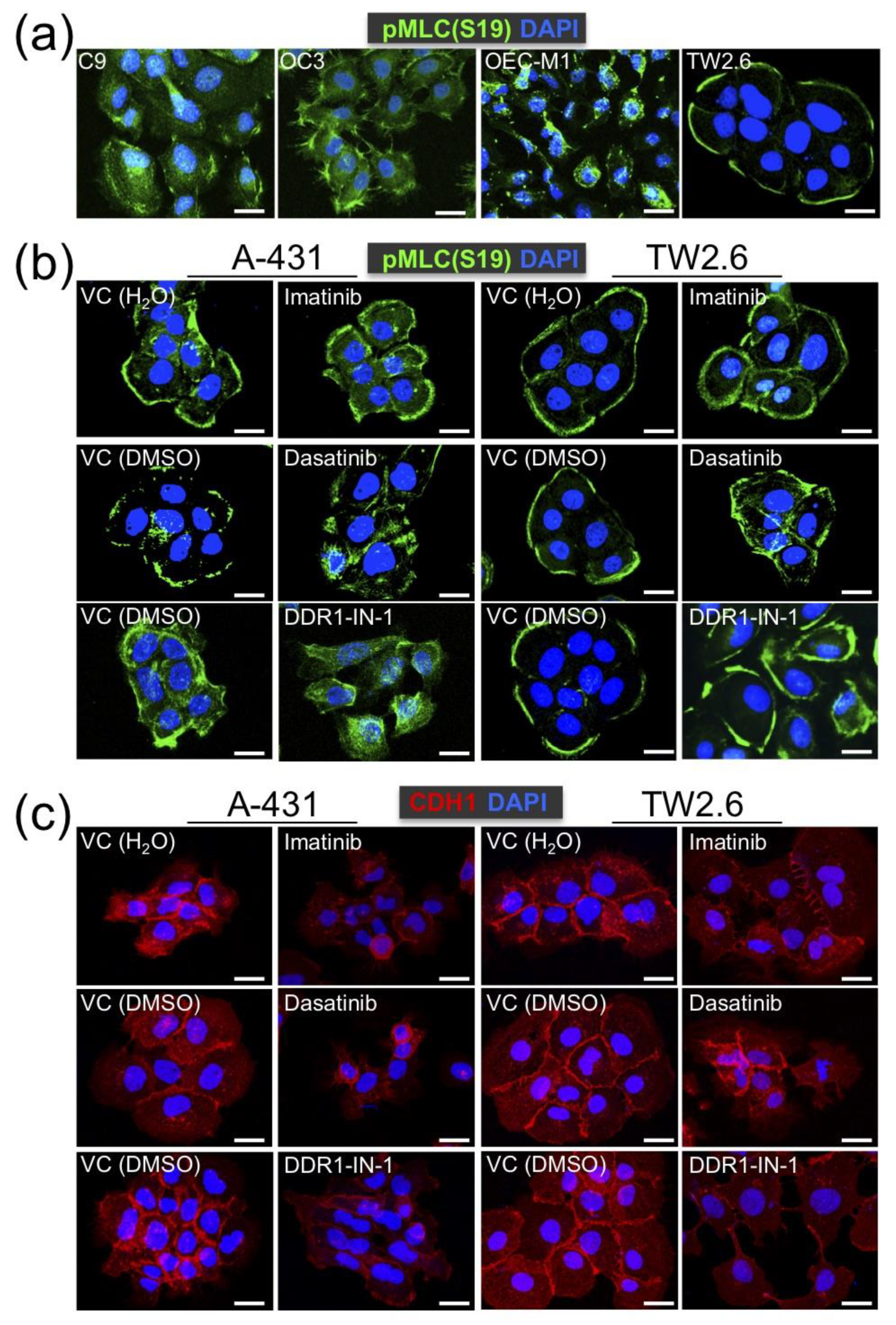
2.5. DDR1 is Involved in Collective Cell Invasion of OSCC TW2.6 Cells
2.6. DDR1 Specific Inhibitor, DDR1-IN-1, Suppressed ALI and PNI in TW2.6 Xenograft Tissues
3. Discussion
4. Materials and Methods
4.1. Cell Culture and Drug Sensitivity Assay
4.2. Antibodies and Reagents
4.3. Reverse Transcription-Quantitative Real-Time PCR (RT-qPCR)
4.4. Western Blot Analysis and Immunoprecipitation
4.5. Clonogenic Survival Assay
4.6. Immunofluorescence Assay
4.7. Spheroid Invasion Assays
4.8. Dual Immunohistochemical Staining
4.9. Animal Experiment
4.10. Bioinformatics and Statistical Analysis
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Appendix A
| Gene SYMBOL | Fold 1 (Yes/No) | Probability 2 | Gene Symbol | Fold (Yes/No) | Probability | Gene Symbol | Fold (Yes/No) | Probability |
|---|---|---|---|---|---|---|---|---|
| LDHD | 0.351 | 65.7 | MMP3 | 1.346 | 19.0 | GRHL3 | 0.481 | 11.9 |
| SORCS2 | 2.233 | 62.9 | TRIM56 | 1.241 | 19.0 | LRRC73 | 1.438 | 11.8 |
| NELL2 | 0.390 | 60.7 | AAMDC | 1.420 | 18.9 | FBXO2 | 1.878 | 11.7 |
| ACSF2 | 1.778 | 52.5 | TMPRSS7 | 2.135 | 18.9 | CYB561A3 | 1.388 | 11.2 |
| MMP1 | 1.457 | 45.7 | ITGA3 | 1.701 | 18.6 | ITGA2 | 1.221 | 11.1 |
| SERPINE1 | 2.103 | 45.6 | TNK1 | 1.717 | 18.2 | IQCE | 1.415 | 11.1 |
| DDR1 | 1.569 | 45.4 | CHST3 | 1.232 | 17.8 | ZNF114 | 1.927 | 11.0 |
| CHST15 | 1.304 | 44.3 | MCOLN3 | 2.266 | 17.0 | PRODH | 0.414 | 11.0 |
| GRINA | 1.257 | 38.5 | HECTD1 | 0.877 | 16.5 | ME1 | 0.758 | 10.8 |
| CCDC30 | 1.313 | 38.3 | PTGER1 | 0.680 | 15.8 | NBPF3 | 1.539 | 10.8 |
| ZNF704 | 2.084 | 33.4 | PPP2R2B | 2.034 | 15.6 | ASB4 | 0.806 | 10.6 |
| RNF5P1 | 1.425 | 33.3 | GPR107 | 1.662 | 15.5 | LYRM1 | 0.910 | 10.6 |
| APCDD1L | 2.075 | 32.3 | SLC16A9 | 0.595 | 15.4 | CYP4F22 | 0.276 | 10.6 |
| CYP24A1 | 1.540 | 31.3 | MCRIP1 | 1.522 | 15.1 | XYLT2 | 1.176 | 10.4 |
| REPS2 | 1.951 | 29.1 | DLL1 | 1.428 | 14.8 | KLC4 | 1.482 | 10.2 |
| CAMK2N1 | 1.613 | 27.1 | CDCA7L | 1.440 | 14.8 | CGB5 | 2.487 | 10.2 |
| TINAGL1 | 1.751 | 27.1 | FOXC2 | 1.850 | 14.6 | EVA1A | 1.926 | 10.1 |
| KLK14 | 0.269 | 26.1 | RTN4RL2 | 1.204 | 14.4 | MDFIC | 0.780 | 10.0 |
| TGFBI | 1.192 | 23.2 | SPECC1 | 1.479 | 14.0 | PRKCH | 0.813 | 9.9 |
| NOS3 | 0.606 | 21.3 | NCS1 | 1.581 | 13.6 | SLC2A6 | 1.359 | 9.8 |
| ZDHHC21 | 0.625 | 21.1 | CCM2 | 1.197 | 13.5 | EFHB | 0.487 | 9.8 |
| FSTL3 | 1.753 | 20.8 | UPK1A | 0.349 | 13.3 | EPHB2 | 2.235 | 9.8 |
| AUH | 1.562 | 20.7 | GLB1 | 1.318 | 13.1 | FREM1 | 0.681 | 9.6 |
| ITGB4 | 1.320 | 20.3 | SYK | 0.633 | 13.0 | MFSD6 | 0.789 | 9.5 |
| C4orf26 | 2.344 | 20.2 | GLTP | 0.795 | 12.8 | GUCA2B | 0.728 | 9.5 |
| GPAT3 | 0.680 | 20.1 | COL4A6 | 1.842 | 12.7 | ZBTB18 | 0.889 | 9.4 |
| RRAS | 1.308 | 19.6 | BMP8B | 1.412 | 12.5 | NELFB | 1.364 | 9.2 |
| GPRC5D | 0.672 | 19.4 | PDPN | 1.585 | 12.5 | LYN | 1.207 | 9.1 |
| SEMA3G | 0.731 | 19.3 | HPSE | 0.636 | 12.1 | ABHD12 | 0.812 | 9.0 |
| PRRC2A | 1.329 | 19.2 | SLC43A2 | 1.763 | 12.0 | TMEM63C | 0.565 | 8.9 |

References
- Leemans, C.R.; Braakhuis, B.J.; Brakenhoff, R.H. The molecular biology of head and neck cancer. Nat. Rev. Cancer 2011, 11, 9–22. [Google Scholar] [CrossRef] [PubMed]
- Taiwan MOHW. Cancer Registry Annual Cancer Report, Ministry of Health and Welfare, Taiwan. Available online: http://tcr.cph.ntu.edu.tw (accessed on 1 February 2020).
- Umar, A.; Dunn, B.K.; Greenwald, P. Future directions in cancer prevention. Nat. Rev. Cancer 2012, 12, 835–848. [Google Scholar] [CrossRef] [PubMed]
- International Agency for Research on Cancer. Betel-Quid and Areca-Nut Chewing and Some Areca-Nut-Derived Nitrosamines; IARC Monographs on the Evaluation of Carcinogenic Risks to Humans; IARC: Lyon, France, 2004; Volume 85, p. 349. [Google Scholar]
- Sharan, R.N.; Mehrotra, R.; Choudhury, Y.; Asotra, K. Association of betel nut with carcinogenesis: Revisit with a clinical perspective. PLoS ONE 2012, 7, e42759. [Google Scholar] [CrossRef] [PubMed]
- Chang, M.C.; Chiang, C.P.; Lin, C.L.; Lee, J.J.; Hahn, L.J.; Jeng, J.H. Cell-mediated immunity and head and neck cancer: With special emphasis on betel quid chewing habit. Oral Oncol. 2005, 41, 757–775. [Google Scholar] [CrossRef]
- Tsai, W.C.; Tsai, S.T.; Ko, J.Y.; Jin, Y.T.; Li, C.; Huang, W.; Young, K.C.; Lai, M.D.; Liu, H.S.; Wu, L.W. The mRNA profile of genes in betel quid chewing oral cancer patients. Oral Oncol. 2004, 40, 418–426. [Google Scholar] [CrossRef]
- Shieh, D.H.; Chiang, L.C.; Shieh, T.Y. Augmented mRNA expression of tissue inhibitor of metalloproteinase-1 in buccal mucosal fibroblasts by arecoline and safrole as a possible pathogenesis for oral submucous fibrosis. Oral Oncol. 2003, 39, 728–735. [Google Scholar] [CrossRef]
- Prime, S.S.; Davies, M.; Pring, M.; Paterson, I.C. The role of TGF-beta in epithelial malignancy and its relevance to the pathogenesis of oral cancer (part II). Crit. Rev. Oral. Biol. Med. 2004, 15, 337–347. [Google Scholar] [CrossRef]
- Rajalalitha, P.; Vali, S. Molecular pathogenesis of oral submucous fibrosis-a collagen metabolic disorder. J. Oral Pathol. Med. 2005, 34, 321–328. [Google Scholar] [CrossRef]
- Khan, I.; Kumar, N.; Pant, I.; Narra, S.; Kondaiah, P. Activation of TGF-beta pathway by areca nut constituents: A possible cause of oral submucous fibrosis. PLoS ONE 2012, 7, e51806. [Google Scholar] [CrossRef]
- Eswaramoorthy, R.; Wang, C.K.; Chen, W.C.; Tang, M.J.; Ho, M.L.; Hwang, C.C.; Wang, H.M.; Wang, C.Z. DDR1 regulates the stabilization of cell surface E-cadherin and E-cadherin-mediated cell aggregation. J. Cell Physiol. 2010, 224, 387–397. [Google Scholar] [CrossRef]
- Huang, Y.; Arora, P.; McCulloch, C.A.; Vogel, W.F. The collagen receptor DDR1 regulates cell spreading and motility by associating with myosin IIA. J. Cell Sci. 2009, 122, 1637–1646. [Google Scholar] [CrossRef]
- Valiathan, R.R.; Marco, M.; Leitinger, B.; Kleer, C.G.; Fridman, R. Discoidin domain receptor tyrosine kinases: New players in cancer progression. Cancer Metastasis Rev. 2012, 31, 295–321. [Google Scholar] [CrossRef]
- Lemeer, S.; Bluwstein, A.; Wu, Z.; Leberfinger, J.; Muller, K.; Kramer, K.; Kuster, B. Phosphotyrosine mediated protein interactions of the discoidin domain receptor 1. J. Proteom. 2012, 75, 3465–3477. [Google Scholar] [CrossRef]
- Vogel, W.F.; Abdulhussein, R.; Ford, C.E. Sensing extracellular matrix: An update on discoidin domain receptor function. Cell. Signal. 2006, 18, 1108–1116. [Google Scholar] [CrossRef] [PubMed]
- Hidalgo-Carcedo, C.; Hooper, S.; Chaudhry, S.I.; Williamson, P.; Harrington, K.; Leitinger, B.; Sahai, E. Collective cell migration requires suppression of actomyosin at cell-cell contacts mediated by DDR1 and the cell polarity regulators Par3 and Par6. Nat. Cell Biol. 2011, 13, 49–58. [Google Scholar] [CrossRef] [PubMed]
- Shimada, K.; Nakamura, M.; Ishida, E.; Higuchi, T.; Yamamoto, H.; Tsujikawa, K.; Konishi, N. Prostate cancer antigen-1 contributes to cell survival and invasion though discoidin receptor 1 in human prostate cancer. Cancer Sci. 2008, 99, 39–45. [Google Scholar] [CrossRef] [PubMed]
- Yamamoto, E.; Kohama, G.; Sunakawa, H.; Iwai, M.; Hiratsuka, H. Mode of invasion, bleomycin sensitivity, and clinical course in squamous cell carcinoma of the oral cavity. Cancer 1983, 51, 2175–2180. [Google Scholar] [CrossRef]
- Cheung, K.J.; Gabrielson, E.; Werb, Z.; Ewald, A.J. Collective invasion in breast cancer requires a conserved Basal epithelial program. Cell 2013, 155, 1639–1651. [Google Scholar] [CrossRef]
- Peglion, F.; Llense, F.; Etienne-Manneville, S. Adherens junction treadmilling during collective migration. Nat. Cell Biol. 2014, 16, 639–651. [Google Scholar] [CrossRef]
- Hirata, E.; Park, D.; Sahai, E. Retrograde flow of cadherins in collective cell migration. Nat. Cell Biol. 2014, 16, 621–623. [Google Scholar] [CrossRef]
- Li, C.F.; Chen, J.Y.; Ho, Y.H.; Hsu, W.H.; Wu, L.C.; Lan, H.Y.; Hsu, D.S.; Tai, S.K.; Chang, Y.C.; Yang, M.H. Snail-induced claudin-11 prompts collective migration for tumour progression. Nat. Cell Biol. 2019, 21, 251–262. [Google Scholar] [CrossRef] [PubMed]
- Retzbach, E.P.; Sheehan, S.A.; Nevel, E.M.; Batra, A.; Phi, T.; Nguyen, A.T.P.; Kato, Y.; Baredes, S.; Fatahzadeh, M.; Shienbaum, A.J.; et al. Podoplanin emerges as a functionally relevant oral cancer biomarker and therapeutic target. Oral Oncol. 2018, 78, 126–136. [Google Scholar] [CrossRef] [PubMed]
- Wicki, A.; Lehembre, F.; Wick, N.; Hantusch, B.; Kerjaschki, D.; Christofori, G. Tumor invasion in the absence of epithelial-mesenchymal transition: Podoplanin-mediated remodeling of the actin cytoskeleton. Cancer Cell 2006, 9, 261–272. [Google Scholar] [CrossRef] [PubMed]
- Dos Santos Almeida, A.; Oliveira, D.T.; Pereira, M.C.; Faustino, S.E.; Nonogaki, S.; Carvalho, A.L.; Kowalski, L.P. Podoplanin and VEGF-C immunoexpression in oral squamous cell carcinomas: Prognostic significance. Anticancer Res. 2013, 33, 3969–3976. [Google Scholar] [PubMed]
- Friedl, P.; Gilmour, D. Collective cell migration in morphogenesis, regeneration and cancer. Nat. Rev. Mol. Cell Biol. 2009, 10, 445–457. [Google Scholar] [CrossRef] [PubMed]
- Slattum, G.M.; Rosenblatt, J. Tumour cell invasion: An emerging role for basal epithelial cell extrusion. Nat. Rev. Cancer 2014, 14, 495–501. [Google Scholar] [CrossRef]
- Tsai, W.-H.; Chen, Y.-L.; Chen, H.-C.; Cheng, A.J.; Chang, K.-Y.; Chu, P.-Y.; Hsiao, J.-R.; Chang, J.-Y.; Lin, S.-F. Overexpression of Discoidin Domain Receptor 1 (DDR1) in oral squamous cell carcinoma. Eur. J. Cancer 2012, 48, 167. [Google Scholar] [CrossRef]
- Chou, S.T.; Peng, H.Y.; Mo, K.C.; Hsu, Y.M.; Wu, G.H.; Hsiao, J.R.; Lin, S.F.; Wang, H.D.; Shiah, S.G. MicroRNA-486-3p functions as a tumor suppressor in oral cancer by targeting DDR1. J. Exp. Clin. Cancer Res. 2019, 38, 281. [Google Scholar] [CrossRef]
- Lee, C.H.; Wong, T.S.; Chan, J.Y.; Lu, S.C.; Lin, P.; Cheng, A.J.; Chen, Y.J.; Chang, J.S.; Hsiao, S.H.; Leu, Y.W.; et al. Epigenetic regulation of the X-linked tumour suppressors BEX1 and LDOC1 in oral squamous cell carcinoma. J. Pathol. 2013, 230, 298–309. [Google Scholar] [CrossRef]
- Subramanian, A.; Tamayo, P.; Mootha, V.K.; Mukherjee, S.; Ebert, B.L.; Gillette, M.A.; Paulovich, A.; Pomeroy, S.L.; Golub, T.R.; Lander, E.S.; et al. Gene set enrichment analysis: A knowledge-based approach for interpreting genome-wide expression profiles. Proc. Natl. Acad. Sci. USA 2005, 102, 15545–15550. [Google Scholar] [CrossRef]
- Ritchie, M.E.; Phipson, B.; Wu, D.; Hu, Y.; Law, C.W.; Shi, W.; Smyth, G.K. limma powers differential expression analyses for RNA-sequencing and microarray studies. Nucleic Acids Res. 2015, 43, e47. [Google Scholar] [CrossRef] [PubMed]
- Huang da, W.; Sherman, B.T.; Lempicki, R.A. Systematic and integrative analysis of large gene lists using DAVID bioinformatics resources. Nat. Protoc. 2009, 4, 44–57. [Google Scholar] [CrossRef] [PubMed]
- Kawaguchi, H.; El-Naggar, A.K.; Papadimitrakopoulou, V.; Ren, H.; Fan, Y.H.; Feng, L.; Lee, J.J.; Kim, E.; Hong, W.K.; Lippman, S.M.; et al. Podoplanin: A novel marker for oral cancer risk in patients with oral premalignancy. J. Clin. Oncol. 2008, 26, 354–360. [Google Scholar] [CrossRef] [PubMed]
- Chen, J.H.; Lim, J.S.; Shyu, K.W.; Meng, C.L. Direct cytotoxicity of garlic on human oral cancer cells. Zhonghua Ya Yi Xue Hui Za Zhi 1988, 7, 13–18. [Google Scholar]
- Kok, S.H.; Hong, C.Y.; Lin, S.K.; Lee, J.J.; Chiang, C.P.; Kuo, M.Y. Establishment and characterization of a tumorigenic cell line from areca quid and tobacco smoke-associated buccal carcinoma. Oral Oncol. 2007, 43, 639–647. [Google Scholar] [CrossRef]
- Lin, S.C.; Liu, C.J.; Chiu, C.P.; Chang, S.M.; Lu, S.Y.; Chen, Y.J. Establishment of OC3 oral carcinoma cell line and identification of NF-kappa B activation responses to areca nut extract. J. Oral Pathol. Med. 2004, 33, 79–86. [Google Scholar] [CrossRef]
- Lu, Y.C.; Chen, Y.J.; Wang, H.M.; Tsai, C.Y.; Chen, W.H.; Huang, Y.C.; Fan, K.H.; Tsai, C.N.; Huang, S.F.; Kang, C.J.; et al. Oncogenic function and early detection potential of miRNA-10b in oral cancer as identified by microRNA profiling. Cancer Prev. Res. 2012, 5, 665–674. [Google Scholar] [CrossRef]
- Vogel, W.; Gish, G.D.; Alves, F.; Pawson, T. The discoidin domain receptor tyrosine kinases are activated by collagen. Mol. Cell 1997, 1, 13–23. [Google Scholar] [CrossRef]
- Bantscheff, M.; Eberhard, D.; Abraham, Y.; Bastuck, S.; Boesche, M.; Hobson, S.; Mathieson, T.; Perrin, J.; Raida, M.; Rau, C.; et al. Quantitative chemical proteomics reveals mechanisms of action of clinical ABL kinase inhibitors. Nat. Biotechnol. 2007, 25, 1035–1044. [Google Scholar] [CrossRef]
- Day, E.; Waters, B.; Spiegel, K.; Alnadaf, T.; Manley, P.W.; Buchdunger, E.; Walker, C.; Jarai, G. Inhibition of collagen-induced discoidin domain receptor 1 and 2 activation by imatinib, nilotinib and dasatinib. Eur. J. Pharmacol. 2008, 599, 44–53. [Google Scholar] [CrossRef]
- Rix, U.; Hantschel, O.; Durnberger, G.; Remsing Rix, L.L.; Planyavsky, M.; Fernbach, N.V.; Kaupe, I.; Bennett, K.L.; Valent, P.; Colinge, J.; et al. Chemical proteomic profiles of the BCR-ABL inhibitors imatinib, nilotinib, and dasatinib reveal novel kinase and nonkinase targets. Blood 2007, 110, 4055–4063. [Google Scholar] [CrossRef] [PubMed]
- Canning, P.; Tan, L.; Chu, K.; Lee, S.W.; Gray, N.S.; Bullock, A.N. Structural mechanisms determining inhibition of the collagen receptor DDR1 by selective and multi-targeted type II kinase inhibitors. J. Mol. Biol. 2014, 426, 2457–2470. [Google Scholar] [CrossRef] [PubMed]
- Kim, H.G.; Tan, L.; Weisberg, E.L.; Liu, F.; Canning, P.; Choi, H.G.; Ezell, S.A.; Wu, H.; Zhao, Z.; Wang, J.; et al. Discovery of a potent and selective DDR1 receptor tyrosine kinase inhibitor. ACS Chem. Biol. 2013, 8, 2145–2150. [Google Scholar] [CrossRef] [PubMed]
- Justus, C.R.; Leffler, N.; Ruiz-Echevarria, M.; Yang, L.V. In vitro cell migration and invasion assays. J. Vis. Exp. 2014. [Google Scholar] [CrossRef] [PubMed]
- Andersen, K.; Mori, H.; Fata, J.; Bascom, J.; Oyjord, T.; Maelandsmo, G.M.; Bissell, M. The metastasis-promoting protein S100A4 regulates mammary branching morphogenesis. Dev. Biol. 2011, 352, 181–190. [Google Scholar] [CrossRef] [PubMed]
- Lu, P.; Takai, K.; Weaver, V.M.; Werb, Z. Extracellular matrix degradation and remodeling in development and disease. Cold Spring Harbor Perspect. Biol. 2011, 3. [Google Scholar] [CrossRef] [PubMed]
- Lu, S.L.; Reh, D.; Li, A.G.; Woods, J.; Corless, C.L.; Kulesz-Martin, M.; Wang, X.J. Overexpression of transforming growth factor beta1 in head and neck epithelia results in inflammation, angiogenesis, and epithelial hyperproliferation. Cancer Res. 2004, 64, 4405–4410. [Google Scholar] [CrossRef]
- Gross, O.; Girgert, R.; Beirowski, B.; Kretzler, M.; Kang, H.G.; Kruegel, J.; Miosge, N.; Busse, A.C.; Segerer, S.; Vogel, W.F.; et al. Loss of collagen-receptor DDR1 delays renal fibrosis in hereditary type IV collagen disease. Matrix Biol. 2010, 29, 346–356. [Google Scholar] [CrossRef]
- Marcotte, R.; Brown, K.R.; Suarez, F.; Sayad, A.; Karamboulas, K.; Krzyzanowski, P.M.; Sircoulomb, F.; Medrano, M.; Fedyshyn, Y.; Koh, J.L.; et al. Essential gene profiles in breast, pancreatic, and ovarian cancer cells. Cancer Discov. 2012, 2, 172–189. [Google Scholar] [CrossRef]
- Ambrogio, C.; Gomez-Lopez, G.; Falcone, M.; Vidal, A.; Nadal, E.; Crosetto, N.; Blasco, R.B.; Fernandez-Marcos, P.J.; Sanchez-Cespedes, M.; Ren, X.; et al. Combined inhibition of DDR1 and Notch signaling is a therapeutic strategy for KRAS-driven lung adenocarcinoma. Nat. Med. 2016, 22, 270–277. [Google Scholar] [CrossRef]
- Jeitany, M.; Leroy, C.; Tosti, P.; Lafitte, M.; Le Guet, J.; Simon, V.; Bonenfant, D.; Robert, B.; Grillet, F.; Mollevi, C.; et al. Inhibition of DDR1-BCR signalling by nilotinib as a new therapeutic strategy for metastatic colorectal cancer. EMBO Mol. Med. 2018, 10. [Google Scholar] [CrossRef] [PubMed]
- Jing, H.; Song, J.; Zheng, J. Discoidin domain receptor 1: New star in cancer-targeted therapy and its complex role in breast carcinoma. Oncol. Lett. 2018, 15, 3403–3408. [Google Scholar] [CrossRef] [PubMed]
- Cheung, K.J.; Ewald, A.J. A collective route to metastasis: Seeding by tumor cell clusters. Science 2016, 352, 167–169. [Google Scholar] [CrossRef] [PubMed]
- Puram, S.V.; Tirosh, I.; Parikh, A.S.; Patel, A.P.; Yizhak, K.; Gillespie, S.; Rodman, C.; Luo, C.L.; Mroz, E.A.; Emerick, K.S.; et al. Single-Cell Transcriptomic Analysis of Primary and Metastatic Tumor Ecosystems in Head and Neck Cancer. Cell 2017, 171, 1611–1624. [Google Scholar] [CrossRef] [PubMed]
- Lindberg, K.; Rheinwald, J.G. Three distinct keratinocyte subtypes identified in human oral epithelium by their patterns of keratin expression in culture and in xenografts. Differentiation 1990, 45, 230–241. [Google Scholar] [CrossRef] [PubMed]
- Tsai, W.H.; Wang, P.W.; Lin, S.Y.; Wu, I.L.; Ko, Y.C.; Chen, Y.L.; Li, M.; Lin, S.F. Ser-634 and Ser-636 of Kaposi’s Sarcoma-Associated Herpesvirus RTA are Involved in Transactivation and are Potential Cdk9 Phosphorylation Sites. Front. Microbiol. 2012, 3, 60. [Google Scholar] [CrossRef] [PubMed]
- Goldman, M.; Craft, B.; Hastie, M.; Repečka, K.; McDade, F.; Kamath, A.; Banerjee, A.; Luo, Y.; Rogers, D.; Brooks, A.N.; et al. The UCSC Xena platform for public and private cancer genomics data visualization and interpretation. bioRxiv 2019. [Google Scholar] [CrossRef]





© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chen, Y.-L.; Tsai, W.-H.; Ko, Y.-C.; Lai, T.-Y.; Cheng, A.-J.; Shiah, S.-G.; Hsiao, J.-R.; Chang, J.-Y.; Lin, S.-F. Discoidin Domain Receptor-1 (DDR1) is Involved in Angiolymphatic Invasion in Oral Cancer. Cancers 2020, 12, 841. https://doi.org/10.3390/cancers12040841
Chen Y-L, Tsai W-H, Ko Y-C, Lai T-Y, Cheng A-J, Shiah S-G, Hsiao J-R, Chang J-Y, Lin S-F. Discoidin Domain Receptor-1 (DDR1) is Involved in Angiolymphatic Invasion in Oral Cancer. Cancers. 2020; 12(4):841. https://doi.org/10.3390/cancers12040841
Chicago/Turabian StyleChen, Yu-Lian, Wan-Hua Tsai, Ying-Chieh Ko, Ting-Yu Lai, Ann-Joy Cheng, Shine-Gwo Shiah, Jenn-Ren Hsiao, Jang-Yang Chang, and Su-Fang Lin. 2020. "Discoidin Domain Receptor-1 (DDR1) is Involved in Angiolymphatic Invasion in Oral Cancer" Cancers 12, no. 4: 841. https://doi.org/10.3390/cancers12040841
APA StyleChen, Y.-L., Tsai, W.-H., Ko, Y.-C., Lai, T.-Y., Cheng, A.-J., Shiah, S.-G., Hsiao, J.-R., Chang, J.-Y., & Lin, S.-F. (2020). Discoidin Domain Receptor-1 (DDR1) is Involved in Angiolymphatic Invasion in Oral Cancer. Cancers, 12(4), 841. https://doi.org/10.3390/cancers12040841






