Progress towards Patient-Specific, Spatially-Continuous Radiobiological Dose Prescription and Planning in Prostate Cancer IMRT: An Overview
Abstract
:1. Introduction
2. Image-Guided Focal Dose Escalation in PCa IMRT
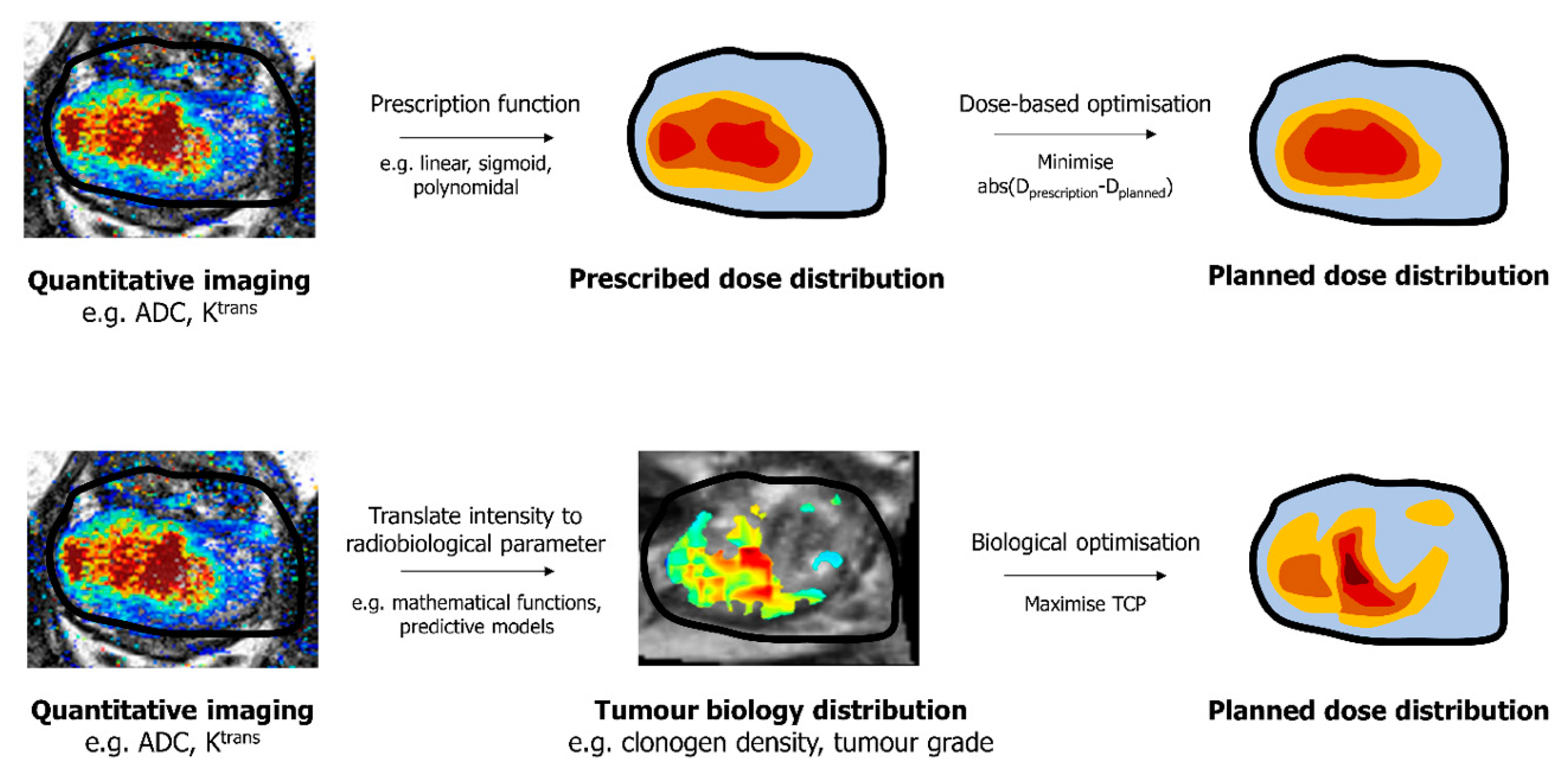
3. Deriving the Desired Dose Prescription from Voxel-Level Information
4. Deriving a Dose Distribution with the Desired Endpoint from Voxel-Level Information
5. Biological Optimisation of Prostate IMRT Using Population-Based Parameters
6. Biological Optimisation of Prostate IMRT Using Patient-Specific, Voxel-Level Parameters
7. Ongoing and Future Considerations
7.1. More Complete TCP Models
7.2. Sensitivity and Specificity of Quantitative Imaging
7.3. Robustness to Uncertainties
7.4. Hypofractionation
7.5. Adaptive Therapy
8. Conclusions
Funding
Conflicts of Interest
References
- Meiers, I.; Waters, D.J.; Bostwick, D.G. Preoperative prediction of multifocal prostate cancer and application of focal therapy: Review 2007. Urology 2007, 70, 3–8. [Google Scholar] [CrossRef] [PubMed]
- Løvf, M.; Zhao, S.; Axcrona, U.; Johannessen, B.; Bakken, A.C.; Carm, K.T.; Hoff, A.M.; Myklebost, O.; Meza-Zepeda, L.A.; Lie, A.K.; et al. Multifocal Primary Prostate Cancer Exhibits High Degree of Genomic Heterogeneity. Eur. Urol. 2019, 75, 498–505. [Google Scholar] [CrossRef] [PubMed]
- Wei, L.; Wang, J.; Lampert, E.; Schlanger, S.; DePriest, A.D.; Hu, Q.; Gomez, E.C.; Murakam, M.; Glenn, S.T.; Conroy, J.; et al. Intratumoral and Intertumoral Genomic Heterogeneity of Multifocal Localized Prostate Cancer Impacts Molecular Classifications and Genomic Prognosticators. Eur. Urol. 2017, 71, 183–192. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ling, C.C.; Humm, J.; Larson, S.; Amols, H.; Fuks, Z.; Leibel, S.; Koutcher, J.A. Towards multidimensional radiotherapy (MD-CRT): Biological imaging and biological conformality. Int. J. Radiat. Oncol. Biol. Phys. 2000, 47, 551–560. [Google Scholar] [CrossRef]
- Bentzen, S.M. Theragnostic imaging for radiation oncology: Dose-painting by numbers. Lancet Oncol. 2005, 6, 112–117. [Google Scholar] [CrossRef]
- Bentzen, S.M.; Gregoire, V. Molecular Imaging–Based Dose Painting: A Novel Paradigm for Radiation Therapy Prescription. Semin. Radiat. Oncol. 2011, 21, 101–110. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Thorwarth, D. Functional imaging for radiotherapy treatment planning: Current status and future directions—A review. Br. J. Radiol. 2015, 88. [Google Scholar] [CrossRef] [PubMed]
- Chopra, S.; Toi, A.; Taback, N.; Evans, A.; Haider, M.A.; Milosevic, M.; Bristow, R.G.; Chung, P.; Bayley, A.; Morton, G.; et al. Pathological predictors for site of local recurrence after radiotherapy for prostate cancer. Int. J. Radiat. Oncol. Biol. Phys. 2012, 82, e441–e448. [Google Scholar] [CrossRef] [PubMed]
- Arrayeh, E.; Westphalen, A.C.; Kurhanewicz, J.; Roach, M.; Jung, A.J.; Carroll, P.R.; Coakley, F.V. Does local recurrence of prostate cancer after radiation therapy occur at the site of primary tumor? Results of a longitudinal MRI and MRSI study. Int. J. Radiat. Oncol. Biol. Phys. 2012, 82, e787–e793. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Pucar, D.; Hricak, H.; Shukla-Dave, A.; Kuroiwa, K.; Drobnjak, M.; Eastham, J.; Scardino, P.T.; Zelefsky, M.J. Clinically significant prostate cancer local recurrence after radiation therapy occurs at the site of primary tumor: Magnetic resonance imaging and step-section pathology evidence. Int. J. Radiat. Oncol. Biol. Phys. 2007, 69, 62–69. [Google Scholar] [CrossRef] [PubMed]
- Cellini, N.; Morganti, A.G.; Mattiucci, G.C.; Valentini, V.; Leone, M.; Luzi, S.; Manfredi, R.; Dinapoli, N.; Digesu’, C.; Smaniotto, D. Analysis of intraprostatic failures in patients treated with hormonal therapy and radiotherapy: Implications for conformal therapy planning. Int. J. Radiat. Oncol. Biol. Phys. 2002, 53, 595–599. [Google Scholar] [CrossRef]
- Mouraviev, V.; Villers, A.; Bostwick, D.G.; Wheeler, T.M.; Montironi, R.; Polascik, T.J. Understanding the pathological features of focality, grade and tumour volume of early-stage prostate cancer as a foundation for parenchyma-sparing prostate cancer therapies: Active surveillance and focal targeted therapy. BJU Int. 2011, 108, 1074–1085. [Google Scholar] [CrossRef] [PubMed]
- Turkbey, B.; Mani, H.; Shah, V.; Rastinehad, A.R.; Bernardo, M.; Pohida, T.; Pang, Y.; Daar, D.; Benjamin, C.; McKinney, Y.L.; et al. Multiparametric 3T prostate magnetic resonance imaging to detect cancer: Histopathological correlation using prostatectomy specimens processed in customized magnetic resonance imaging based molds. J. Urol. 2011, 186, 1818–1824. [Google Scholar] [CrossRef] [Green Version]
- Lips, I.M.; van der Heide, U.A.; Haustermans, K.; van Lin, E.N.; Pos, F.; Franken, S.P.; Kotte, A.N.; van Gils, C.H.; van Vulpen, M. Single blind randomized Phase III trial to investigate the benefit of a focal lesion ablative microboost in prostate cancer (FLAME-trial): Study protocol for a randomized controlled trial. Trials 2011, 12, 255. [Google Scholar] [CrossRef] [Green Version]
- Monninkhof, E.M.; van Loon, J.W.L.; van Vulpen, M.; Kerkmeijer, L.G.W.; Pos, F.J.; Haustermans, K.; van den Bergh, L.; Isebaert, S.; McColl, G.M.; Smeenk, R.J.; et al. Standard whole prostate gland radiotherapy with and without lesion boost in prostate cancer: Toxicity in the FLAME randomized controlled trial. Radiother. Oncol. 2018, 127, 74–80. [Google Scholar] [CrossRef] [Green Version]
- Murray, J.R.; Tree, A.C.; Alexander, E.J.; Sohaib, A.; Hazell, S.; Thomas, K.; Gunapala, R.; Parker, C.C.; Huddart, R.A.; Gao, A.; et al. Standard and Hypofractionated Dose Escalation to Intraprostatic Tumor Nodules in Localized Prostate Cancer: Efficacy and Toxicity in the DELINEATE Trial. Int. J. Radiat. Oncol. Biol. Phys. 2020, 106, 715–724. [Google Scholar] [CrossRef] [Green Version]
- Blumenfeld, P.; Chowdhary, M.; Deane, L.A.; Pfanzelter, N.; Shors, S.; Braun, R.; White, G.M.; Coogan, C.; Hoeksema, J.; Khare, N.; et al. Multi-parametric MRI guided dose escalated radiotherapy for treatment of localized prostate cancer (PCa): Initial toxicity results of a prospective phase II trial. JCO 2018, 36, 25. [Google Scholar] [CrossRef]
- Meshman, J.; Farnia, B.; Stoyanova, R.; Reis, I.; Abramowitz, M.; Dal Pra, A.; Horwitz, E.M.; Pollack, A. Biopsy positivity in prostate cancer patients undergoing mpMRI-targeted radiation dose escalation. JCO 2020, 38, 336. [Google Scholar] [CrossRef]
- van Schie, M.A.; Dinh, C.V.; van Houdt, P.J.; Pos, F.J.; Heijmink, S.W.T.J.P.; Kerkmeijer, L.G.W.; Kotte, A.N.T.J.; Oyen, R.; Haustermans, K.; van der Heide, U.A. Contouring of prostate tumors on multiparametric MRI: Evaluation of clinical delineations in a multicenter radiotherapy trial. Radiother. Oncol. 2018, 128, 321–326. [Google Scholar] [CrossRef]
- Steenbergen, P.; Haustermans, K.; Lerut, E.; Oyen, R.; De Wever, L.; Van den Bergh, L.; Kerkmeijer, L.G.W.; Pameijer, F.A.; Veldhuis, W.B.; van der Voort van Zyp, J.R.N.; et al. Prostate tumor delineation using multiparametric magnetic resonance imaging: Inter-observer variability and pathology validation. Radiother. Oncol. 2015, 115, 186–190. [Google Scholar] [CrossRef] [Green Version]
- Anwar, M.; Westphalen, A.C.; Jung, A.J.; Noworolski, S.M.; Simko, J.P.; Kurhanewicz, J.; Roach, M.; Carroll, P.R.; Coakley, F.V. Role of endorectal MR imaging and MR spectroscopic imaging in defining treatable intraprostatic tumor foci in prostate cancer: Quantitative analysis of imaging contour compared to whole-mount histopathology. Radiother. Oncol. 2014, 110, 303–308. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bratan, F.; Niaf, E.; Melodelima, C.; Chesnais, A.L.; Souchon, R.; Mège-Lechevallier, F.; Colombel, M.; Rouvière, O. Influence of imaging and histological factors on prostate cancer detection and localisation on multiparametric MRI: A prospective study. Eur. Radiol. 2013, 23, 2019–2029. [Google Scholar] [CrossRef]
- Rischke, H.C.; Nestle, U.; Fechter, T.; Doll, C.; Volegova-Neher, N.; Henne, K.; Scholber, J.; Knippen, S.; Kirste, S.; Grosu, A.L.; et al. 3 Tesla multiparametric MRI for GTV-definition of Dominant Intraprostatic Lesions in patients with Prostate Cancer--an interobserver variability study. Radiat. Oncol. 2013, 8, 183. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Alber, M.; Paulsen, F.; Eschmann, S.M.; Machulla, H.J. On biologically conformal boost dose optimization. Phys. Med. Biol. 2003, 48, N31–N35. [Google Scholar] [CrossRef] [PubMed]
- Arnesen, M.R.; Knudtsen, I.S.; Rekstad, B.L.; Eilertsen, K.; Dale, E.; Bruheim, K.; Helland, Å.; Løndalen, A.M.; Hellebust, T.P.; Malinen, E. Dose painting by numbers in a standard treatment planning system using inverted dose prescription maps. Acta Oncol. 2015, 54, 1607–1613. [Google Scholar] [CrossRef]
- Berwouts, D.; Olteanu, L.A.M.; Speleers, B.; Duprez, F.; Madani, I.; Vercauteren, T.; De Neve, W.; De Gersem, W. Intensity modulated arc therapy implementation in a three phase adaptive 18F-FDG-PET voxel intensity-based planning strategy for head-and-neck cancer. Radiat. Oncol. 2016, 11, 52. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Das, S.K.; Miften, M.M.; Zhou, S.; Bell, M.; Munley, M.T.; Whiddon, C.S.; Craciunescu, O.; Baydush, A.H.; Wong, T.; Rosenman, J.G.; et al. Feasibility of optimizing the dose distribution in lung tumors using fluorine-18-fluorodeoxyglucose positron emission tomography and single photon emission computed tomography guided dose prescriptions. Med. Phys. 2004, 31, 1452–1461. [Google Scholar] [CrossRef]
- Vanderstraeten, B.; Duthoy, W.; Gersem, W.D.; Neve, W.D.; Thierens, H. [18F]fluoro-deoxy-glucose positron emission tomography ([18F]FDG-PET) voxel intensity-based intensity-modulated radiation therapy (IMRT) for head and neck cancer. Radiother. Oncol. 2006, 79, 249–258. [Google Scholar] [CrossRef]
- Bowen, S.R.; Flynn, R.T.; Bentzen, S.M.; Jeraj, R. On the sensitivity of IMRT dose optimization to the mathematical form of a biological imaging-based prescription function. Phys. Med. Biol. 2009, 54, 1483–1501. [Google Scholar] [CrossRef]
- van Schie, M.A.; Steenbergen, P.; Dinh, C.V.; Ghobadi, G.; van Houdt, P.J.; Pos, F.J.; Heijmink, S.W.T.J.P.; van der Poel, H.G.; Renisch, S.; Vik, T.; et al. Repeatability of dose painting by numbers treatment planning in prostate cancer radiotherapy based on multiparametric magnetic resonance imaging. Phys. Med. Biol. 2017, 62, 5575–5588. [Google Scholar] [CrossRef]
- Thorwarth, D.; Eschmann, S.-M.; Paulsen, F.; Alber, M. Hypoxia Dose Painting by Numbers: A Planning Study. Int. J. Radiat. Oncol. Biol. Phys. 2007, 68, 291–300. [Google Scholar] [CrossRef] [PubMed]
- Vanderstraeten, B.; Gersem, W.D.; Duthoy, W.; Neve, W.D.; Thierens, H. Implementation of biologically conformal radiation therapy (BCRT) in an algorithmic segmentation-based inverse planning approach. Phys. Med. Biol. 2006, 51, N277–N286. [Google Scholar] [CrossRef] [PubMed]
- Yan, D.; Chen, S.; Krauss, D.J.; Chen, P.Y.; Chinnaiyan, P.; Wilson, G.D. Tumor Voxel Dose-Response Matrix and Dose Prescription Function Derived Using 18F-FDG PET/CT Images for Adaptive Dose Painting by Number. Int. J. Radiat. Oncol. Biol. Phys. 2019, 104, 207–218. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zamboglou, C.; Sachpazidis, I.; Koubar, K.; Drendel, V.; Wiehle, R.; Kirste, S.; Mix, M.; Schiller, F.; Mavroidis, P.; Meyer, P.T.; et al. Evaluation of intensity modulated radiation therapy dose painting for localized prostate cancer using 68 Ga-HBED-CC PSMA-PET/CT: A planning study based on histopathology reference. Radiother. Oncol. 2017, 123, 472–477. [Google Scholar] [CrossRef] [Green Version]
- Zamboglou, C.; Thomann, B.; Koubar, K.; Bronsert, P.; Krauss, T.; Rischke, H.C.; Sachpazidis, I.; Drendel, V.; Salman, N.; Reichel, K.; et al. Focal dose escalation for prostate cancer using 68Ga-HBED-CC PSMA PET/CT and MRI: A planning study based on histology reference. Radiat. Oncol. 2018, 13, 81. [Google Scholar] [CrossRef] [Green Version]
- Wong, W.W.; Schild, S.E.; Vora, S.A.; Ezzell, G.A.; Nguyen, B.D.; Ram, P.C.; Roarke, M.C. Image-guided radiotherapy for prostate cancer: A prospective trial of concomitant boost using indium-111-capromab pendetide (ProstaScint) imaging. Int. J. Radiat. Oncol. Biol. Phys. 2011, 81, e423–e429. [Google Scholar] [CrossRef]
- Lea, D.E. Actions of Radiations on Living Cells; Cambridge University Press: London, UK, 1946. [Google Scholar]
- Lea, D.E.; Catcheside, D.G. The mechanism of the induction by radiation of chromosome aberrations inTradescantia. J. Genetics 1942, 44, 216–245. [Google Scholar] [CrossRef]
- Barendsen, G.W. Dose fractionation, dose rate and iso-effect relationships for normal tissue responses. Int. J. Radiat. Oncol. Biol. Phys. 1982, 8, 1981–1997. [Google Scholar] [CrossRef]
- Curtis, S.B. Lethal and potentially lethal lesions induced by radiation—A unified repair model. Radiat. Res. 1986, 106, 252–270. [Google Scholar] [CrossRef]
- Dale, R.G. The application of the linear-quadratic dose-effect equation to fractionated and protracted radiotherapy. Br. J. Radiol. 1985, 58, 515–528. [Google Scholar] [CrossRef]
- Douglas, B.G.; Fowler, J.F. Letter: Fractionation schedules and a quadratic dose-effect relationship. Br. J. Radiol. 1975, 48, 502–504. [Google Scholar] [CrossRef] [PubMed]
- Fowler, J.F. The linear-quadratic formula and progress in fractionated radiotherapy. BJR 1989, 62, 679–694. [Google Scholar] [CrossRef] [PubMed]
- Schultheiss, T.E.; Zagars, G.K.; Peters, L.J. An explanatory hypothesis for early- and late-effect parameter values in the LQ model. Radiother. Oncol. 1987, 9, 241–248. [Google Scholar] [CrossRef]
- Thames, H.D. An “incomplete-repair” model for survival after fractionated and continuous irradiations. Int. J. Radiat. Biol. Relat. Stud. Phys. Chem. Med. 1985, 47, 319–339. [Google Scholar] [CrossRef] [PubMed]
- Thames, H.D.; Ang, K.K.; Stewart, F.A.; van der Schueren, E. Does incomplete repair explain the apparent failure of the basic LQ model to predict spinal cord and kidney responses to low doses per fraction? Int. J. Radiat. Biol. 1988, 54, 13–19. [Google Scholar] [CrossRef]
- Sinclair, W.K. The shape of radiation survival curves of mammalian cells cultured in vitro. In Biophysical Aspects of Radiation Quality; Techn Rep Series 58 IAEA; International Atomic Energy Agency (IAEA): Vienna, Austria, 1966; pp. 21–43. [Google Scholar]
- Chadwick, K.H.; Leenhouts, H.P. A molecular theory of cell survival. Phys. Med. Biol. 1973, 18, 78–87. [Google Scholar] [CrossRef]
- Kellerer, A.M.; Rossi, H.H. A Generalized Formulation of Dual Radiation Action. Radiat. Res. 1978, 75, 471–488. [Google Scholar] [CrossRef] [Green Version]
- Dasu, A.; Toma-Dasu, I. Will intrafraction repair have negative consequences on extreme hypofractionation in prostate radiation therapy? Br. J. Radiol. 2015, 88. [Google Scholar] [CrossRef]
- Kim, Y.; Tomé, W.A. Risk-adaptive optimization: Selective boosting of high-risk tumor subvolumes. Int. J. Radiat. Oncol. Biol. Phy. 2006, 66, 1528–1542. [Google Scholar] [CrossRef] [Green Version]
- Levegrün, S.; Jackson, A.; Zelefsky, M.J.; Venkatraman, E.S.; Skwarchuk, M.W.; Schlegel, W.; Fuks, Z.; Leibel, S.A.; Ling, C.C. Risk group dependence of dose–response for biopsy outcome after three-dimensional conformal radiation therapy of prostate cancer. Radiother. Oncol. 2002, 63, 11–26. [Google Scholar] [CrossRef]
- Källman, P.; Agren, A.; Brahme, A. Tumour and normal tissue responses to fractionated non-uniform dose delivery. Int. J. Radiat. Biol. 1992, 62, 249–262. [Google Scholar] [CrossRef] [PubMed]
- Uzan, J.; Nahum, A.E.; Syndikus, I. Prostate Dose-painting Radiotherapy and Radiobiological Guided Optimisation Enhances the Therapeutic Ratio. Clin. Oncol. 2016, 28, 165–170. [Google Scholar] [CrossRef] [Green Version]
- Betts, J.M.; Mears, C.; Reynolds, H.M.; Tack, G.; Leo, K.; Ebert, M.A.; Haworth, A. Optimised Robust Treatment Plans for Prostate Cancer Focal Brachytherapy. Procedia Comput. Sci. 2015, 51, 914–923. [Google Scholar] [CrossRef] [Green Version]
- Betts, J.M.; Mears, C.; Reynolds, H.M.; Ebert, M.A.; Haworth, A. Prostate cancer focal brachytherapy: Improving treatment plan robustness using a convolved dose rate model. Procedia Comput. Sci. 2017, 108, 1522–1531. [Google Scholar] [CrossRef]
- Haworth, A.; Mears, C.; Betts, J.M.; Reynolds, H.M.; Tack, G.; Leo, K.; Williams, S.; Ebert, M.A. A radiobiology-based inverse treatment planning method for optimisation of permanent l-125 prostate implants in focal brachytherapy. Phys. Med. Biol. 2016, 61, 430–444. [Google Scholar] [CrossRef] [PubMed]
- Zeng, J.; Bauer, J.J.; Mun, S.K. Modeling and mapping of prostate cancer. Comput. Graph. 2000, 24, 683–694. [Google Scholar] [CrossRef]
- Dirscherl, T.; Rickhey, M.; Bogner, L. Feasibility of TCP-based dose painting by numbers applied to a prostate case with 18F-Choline PET imaging. Zeitschrift für Medizinische Physik 2012, 22, 48–57. [Google Scholar] [CrossRef]
- Ebert, M.A.; Hoban, P.W. Some characteristics of tumour control probability for heterogeneous tumours. Phys. Med. Biol. 1996, 41, 2125–2133. [Google Scholar] [CrossRef]
- Grönlund, E.; Johansson, S.; Nyholm, T.; Thellenberg, C.; Ahnesjö, A. Dose painting of prostate cancer based on Gleason score correlations with apparent diffusion coefficients. Acta Oncol. 2018, 57, 574–581. [Google Scholar] [CrossRef] [Green Version]
- Chaddad, A.; Kucharczyk, M.J.; Niazi, T. Multimodal Radiomic Features for the Predicting Gleason Score of Prostate Cancer. Cancers 2018, 10, 249. [Google Scholar] [CrossRef] [Green Version]
- Hambrock, T.; Somford, D.M.; Huisman, H.J.; van Oort, I.M.; Witjes, J.A.; Hulsbergen-van de Kaa, C.A.; Scheenen, T.; Barentsz, J.O. Relationship between apparent diffusion coefficients at 3.0-T MR imaging and Gleason grade in peripheral zone prostate cancer. Radiology 2011, 259, 453–461. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.; Carver, E.; Feldman, A.; Pantelic, M.V.; Elshaikh, M.; Wen, N. Volumetric and Voxel-Wise Analysis of Dominant Intraprostatic Lesions on Multiparametric MRI. Front. Oncol. 2019, 9, 616. [Google Scholar] [CrossRef] [PubMed]
- Sun, Y.; Reynolds, H.; Wraith, D.; Williams, S.; Finnegan, M.E.; Mitchell, C.; Murphy, D.; Ebert, M.A.; Haworth, A. Predicting prostate tumour location from multiparametric MRI using Gaussian kernel support vector machines: A preliminary study. Australas. Phys. Eng. 2017. [Google Scholar] [CrossRef] [PubMed]
- Chen, Q.; Hu, S.; Long, P.; Lu, F.; Shi, Y.; Li, Y. A Transfer Learning Approach for Malignant Prostate Lesion Detection on Multiparametric MRI. Technol. Cancer Res. Treat. 2019, 18. [Google Scholar] [CrossRef]
- Wang, Z.; Liu, C.; Cheng, D.; Wang, L.; Yang, X.; Cheng, K.-T. Automated Detection of Clinically Significant Prostate Cancer in mp-MRI Images Based on an End-to-End Deep Neural Network. IEEE Trans. Med. Imaging 2018, 37, 1127–1139. [Google Scholar] [CrossRef]
- Algohary, A.; Viswanath, S.; Shiradkar, R.; Ghose, S.; Pahwa, S.; Moses, D.; Jambor, I.; Shnier, R.; Böhm, M.; Haynes, A.-M.; et al. Radiomic features on MRI enable risk categorization of prostate cancer patients on active surveillance: Preliminary findings. J. Magn. Reson. Imaging 2018. [Google Scholar] [CrossRef]
- Toivonen, J.; Montoya Perez, I.; Movahedi, P.; Merisaari, H.; Pesola, M.; Taimen, P.; Boström, P.J.; Pohjankukka, J.; Kiviniemi, A.; Pahikkala, T.; et al. Radiomics and machine learning of multisequence multiparametric prostate MRI: Towards improved non-invasive prostate cancer characterization. PLoS ONE 2019, 14, e0217702. [Google Scholar] [CrossRef] [Green Version]
- Hectors, S.J.; Cherny, M.; Yadav, K.K.; Beksaç, A.T.; Thulasidass, H.; Lewis, S.; Davicioni, E.; Wang, P.; Tewari, A.K.; Taouli, B. Radiomics Features Measured with Multiparametric Magnetic Resonance Imaging Predict Prostate Cancer Aggressiveness. J. Urol. 2019, 202, 498–505. [Google Scholar] [CrossRef]
- Sun, Y.; Reynolds, H.M.; Wraith, D.; Williams, S.; Finnegan, M.E.; Mitchell, C.; Murphy, D.; Haworth, A. Automatic stratification of prostate tumour aggressiveness using multiparametric MRI: A horizontal comparison of texture features. Acta Oncol. 2019, 58, 1–9. [Google Scholar] [CrossRef]
- Li, J.; Weng, Z.; Xu, H.; Zhang, Z.; Miao, H.; Chen, W.; Liu, Z.; Zhang, X.; Wang, M.; Xu, X.; et al. Support Vector Machines (SVM) classification of prostate cancer Gleason score in central gland using multiparametric magnetic resonance images: A cross-validated study. Eur. J. Radiol. 2018, 98, 61–67. [Google Scholar] [CrossRef]
- Varghese, B.; Chen, F.; Hwang, D.; Palmer, S.L.; De Castro Abreu, A.L.; Ukimura, O.; Aron, M.; Aron, M.; Gill, I.; Duddalwar, V.; et al. Objective risk stratification of prostate cancer using machine learning and radiomics applied to multiparametric magnetic resonance images. Scientific Rep. 2019, 9, 1–10. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Rastinehad, A.R.; Baccala, A.A.; Chung, P.H.; Proano, J.M.; Kruecker, J.; Xu, S.; Locklin, J.K.; Turkbey, B.; Shih, J.; Bratslavsky, G.; et al. D’Amico Risk Stratification Correlates with Degree of Suspicion of Prostate Cancer on Multi-Parametric Magnetic Resonance Imaging (MRI). J. Urol 2011, 185, 815–820. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Haworth, A.; Sun, Y.; Ebert, M.; Reynolds, H.; Betts, J.; Wraith, D.; Mitchell, C. Incorporating a Novel Radiomics Framework for Biologically Optimised Prostate RadioTherapy (BiRT). Med. Phys. 2018, 45, E446. [Google Scholar]
- Sun, Y.; Reynolds, H.M.; Wraith, D.; Williams, S.; Finnegan, M.E.; Mitchell, C.; Murphy, D.; Haworth, A. Voxel-wise prostate cell density prediction using multiparametric magnetic resonance imaging and machine learning. Acta Oncol. 2018, 57, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Reynolds, H.M.; Williams, S.; Jackson, P.; Mitchell, C.; Hofman, M.S.; Hicks, R.J.; Murphy, D.G.; Haworth, A. Voxel-wise correlation of positron emission tomography/computed tomography with multiparametric magnetic resonance imaging and histology of the prostate using a sophisticated registration framework. BJU Int. 2018, 123, 1020–1030. [Google Scholar] [CrossRef]
- Her, E.; Sun, Y.; Kennedy, A.; Reynolds, H.M.; Williams, S.; Ebert, M.A.; Haworth, A. Biological optimization of prostate IMRT with patient-specific image-derived cancer biology. J. Med. Imaging Radiat. Oncol. 2017, 61, 103. [Google Scholar]
- Her, E.; Sun, Y.; Ebert, M.A.; Reynolds, H.M.; Kennedy, A.; Haworth, A.; Williams, S. O99 Biofocused approach to prostate cancer IMRT planning with voxel-level cancer biology derived from multiparametric MRI. Australas. Phys. Eng. Sci. Med. 2018, 41, 314. [Google Scholar]
- Fuks, Z.; Kolesnick, R. Engaging the vascular component of the tumor response. Cancer Cell 2005, 8, 89–91. [Google Scholar] [CrossRef] [Green Version]
- Garcia-Barros, M.; Paris, F.; Cordon-Cardo, C.; Lyden, D.; Rafii, S.; Haimovitz-Friedman, A.; Fuks, Z.; Kolesnick, R. Tumor response to radiotherapy regulated by endothelial cell apoptosis. Science 2003, 300, 1155–1159. [Google Scholar] [CrossRef] [Green Version]
- Park, H.J.; Griffin, R.J.; Hui, S.; Levitt, S.H.; Song, C.W. Radiation-Induced Vascular Damage in Tumors: Implications of Vascular Damage in Ablative Hypofractionated Radiotherapy (SBRT and SRS). Radiat. Res. 2012, 177, 311–327. [Google Scholar] [CrossRef] [Green Version]
- Schneider, B.F.; Eberhard, D.A.; Steiner, L.E. Histopathology of arteriovenous malformations after gamma knife radiosurgery. J. Neurosurg. 1997, 87, 352–357. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lee, Y.; Auh, S.L.; Wang, Y.; Burnette, B.; Wang, Y.; Meng, Y.; Beckett, M.; Sharma, R.; Chin, R.; Tu, T.; et al. Therapeutic effects of ablative radiation on local tumor require CD8+ T cells: Changing strategies for cancer treatment. Blood 2009, 114, 589–595. [Google Scholar] [CrossRef] [PubMed]
- Demaria, S.; Ng, B.; Devitt, M.L.; Babb, J.S.; Kawashima, N.; Liebes, L.; Formenti, S.C. Ionizing radiation inhibition of distant untreated tumors (abscopal effect) is immune mediated. Int. J. Radiat. Oncol. Biol. Phys. 2004, 58, 862–870. [Google Scholar] [CrossRef] [PubMed]
- Her, E.J.; Reynolds, H.M.; Mears, C.; Williams, S.; Moorehouse, C.; Millar, J.L.; Ebert, M.A.; Haworth, A. Radiobiological parameters in a tumour control probability model for prostate cancer LDR brachytherapy. Phys. Med. Biol. 2018, 63, 135011. [Google Scholar] [CrossRef]
- Lemaître, G.; Martí, R.; Freixenet, J.; Vilanova, J.C.; Walker, P.M.; Meriaudeau, F. Computer-Aided Detection and diagnosis for prostate cancer based on mono and multi-parametric MRI: A review. Comput. Biol. Med. 2015, 60, 8–31. [Google Scholar] [CrossRef] [Green Version]
- de Rooij, M.; Hamoen, E.H.J.; Fütterer, J.J.; Barentsz, J.O.; Rovers, M.M. Accuracy of Multiparametric MRI for Prostate Cancer Detection: A Meta-Analysis. Am. J. Roentgenol. 2014, 202, 343–351. [Google Scholar] [CrossRef]
- Sun, Y.; Reynolds, H.M.; Parameswaran, B.; Wraith, D.; Finnegan, M.E.; Williams, S.; Haworth, A. Multiparametric MRI and radiomics in prostate cancer: A review. Australas. Phys. Eng. Sci. Med. 2019, 42, 3–25. [Google Scholar] [CrossRef]
- Kim, Y.; Tomé, W.A. Dose-painting IMRT optimization using biological parameters. Acta Oncol. 2010, 49, 1374–1384. [Google Scholar] [CrossRef]
- Landberg, T.; Chavaudra, J.; Dobbs, J.; Gerard, J.-P.; Hanks, G.; Horiot, J.-C.; Johansson, K.-A.; Möller, T.; Purdy, J.; Suntharalingam, N.; et al. Report 62. J. Int. Commission Radiat. Units Measurements 1999, os32. [Google Scholar] [CrossRef]
- Unkelbach, J.; Alber, M.; Bangert, M.; Bokrantz, R.; Chan, T.C.Y.; Deasy, J.O.; Fredriksson, A.; Gorissen, B.L.; van Herk, M.; Liu, W.; et al. Robust radiotherapy planning. Phys. Med. Biol. 2018, 63, 22TR02. [Google Scholar] [CrossRef] [Green Version]
- Bangert, M.; Hennig, P.; Oelfke, U. Analytical probabilistic modeling for radiation therapy treatment planning. Phys. Med. Biol. 2013, 58, 5401–5419. [Google Scholar] [CrossRef] [PubMed]
- Baum, C.; Alber, M.; Birkner, M.; Nüsslin, F. Robust treatment planning for intensity modulated radiotherapy of prostate cancer based on coverage probabilities. Radiother. Oncol. 2006, 78, 27–35. [Google Scholar] [CrossRef] [PubMed]
- Bohoslavsky, R.; Witte, M.G.; Janssen, T.M.; van Herk, M. Probabilistic objective functions for margin-less IMRT planning. Phys. Med. Biol. 2013, 58, 3563–3580. [Google Scholar] [CrossRef] [PubMed]
- Chan, T.C.Y.; Bortfeld, T.; Tsitsiklis, J.N. A robust approach to IMRT optimization. Phys. Med. Biol. 2006, 51, 2567–2583. [Google Scholar] [CrossRef]
- Chan, T.C.Y.; Mišić, V.V. Adaptive and robust radiation therapy optimization for lung cancer. Eur. J. Operat. Res. 2013, 231, 745–756. [Google Scholar] [CrossRef] [Green Version]
- Lowe, M.; Aitkenhead, A.; Albertini, F.; Lomax, A.J.; MacKay, R.I. A robust optimisation approach accounting for the effect of fractionation on setup uncertainties. Phys. Med. Biol. 2017, 62, 8178–8196. [Google Scholar] [CrossRef]
- Unkelbach, J.; Oelfke, U. Inclusion of organ movements in IMRT treatment planning via inverse planning based on probability distributions. Phys. Med. Biol. 2004, 49, 4005–4029. [Google Scholar] [CrossRef]
- Witte, M.G.; van der Geer, J.; Schneider, C.; Lebesque, J.V.; Alber, M.; van Herk, M. IMRT optimization including random and systematic geometric errors based on the expectation of TCP and NTCP: Optimization of expected TCP and NTCP. Med. Phys. 2007, 34, 3544–3555. [Google Scholar] [CrossRef]
- Brenner, D.J.; Hall, E.J. Fractionation and protraction for radiotherapy of prostate carcinoma. Int. J. Radiat. Oncol. Biol. Phys. 1999, 43, 1095–1101. [Google Scholar] [CrossRef]
- Fowler, J.; Chappell, R.; Ritter, M. Is alpha/beta for prostate tumors really low? Int. J. Radiat. Oncol. Biol. Phys. 2001, 50, 1021–1031. [Google Scholar] [CrossRef]
- Wang, J.Z.; Guerrero, M.; Li, X.A. How low is the alpha/beta ratio for prostate cancer? Int. J. Radiat. Oncol. Biol. Phys. 2003, 55, 194–203. [Google Scholar] [CrossRef]
- Brenner, D.J. Fractionation and late rectal toxicity. Int. J. Radiat. Oncol. Biol. Phys. 2004, 60, 1013–1015. [Google Scholar] [CrossRef] [PubMed]
- Rana, S.; Rogers, K. Radiobiological Impact of Acuros XB Dose Calculation Algorithm on Low-Risk Prostate Cancer Treatment Plans Created by RapidArc Technique. Austral-Asian J. Cancer 2012, 11, 261–269. [Google Scholar]
- Abu-Gheida, I.; Reddy, C.A.; Kotecha, R.; Weller, M.A.; Shah, C.; Kupelian, P.A.; Mian, O.; Ciezki, J.P.; Stephans, K.L.; Tendulkar, R.D. Ten-year outcomes of moderately hypofractionated (70 Gy in 28 fractions) intensity modulated radiotherapy for localized prostate cancer. Int. J. Radiat. Oncol. Biol. Phys. 2019. [Google Scholar] [CrossRef]
- Catton, C.N.; Lukka, H.; Julian, J.A.; Gu, C.-S.; Martin, J.; Supiot, S.; Chung, P.W.M.; Bauman, G.; Bahary, J.-P.; Ahmed, S.; et al. A randomized trial of a shorter radiation fractionation schedule for the treatment of localized prostate cancer. JCO 2016, 34, 5003. [Google Scholar] [CrossRef]
- Dearnaley, D.; Syndikus, I.; Mossop, H.; Khoo, V.; Birtle, A.; Bloomfield, D.; Graham, J.; Kirkbride, P.; Logue, J.; Malik, Z.; et al. Conventional versus hypofractionated high-dose intensity-modulated radiotherapy for prostate cancer: 5-year outcomes of the randomised, non-inferiority, phase 3 CHHiP trial. Lancet Oncol. 2016, 17, 1047–1060. [Google Scholar] [CrossRef] [Green Version]
- Katz, A.J.; Kang, J. Quality of Life and Toxicity after SBRT for Organ-Confined Prostate Cancer, a 7-Year Study. Front. Oncol. 2014, 4, 301. [Google Scholar] [CrossRef] [Green Version]
- Loblaw, A.; Cheung, P.; D’Alimonte, L.; Deabreu, A.; Mamedov, A.; Zhang, L.; Tang, C.; Quon, H.; Jain, S.; Pang, G.; et al. Prostate stereotactic ablative body radiotherapy using a standard linear accelerator: Toxicity, biochemical, and pathological outcomes. Radiother. Oncol. 2013, 107, 153–158. [Google Scholar] [CrossRef]
- Tiberi, D.; Vavassis, P.; Nguyen, D.; Yassa, M. Hypofractionation for prostate cancer: An update. Expert Rev. Endocrinol. Metab. 2017, 12, 199–205. [Google Scholar] [CrossRef]
- Widmark, A.; Gunnlaugsson, A.; Beckman, L.; Thellenberg-Karlsson, C.; Hoyer, M.; Lagerlund, M.; Fransson, P.; Kindblom, J.; Ginman, C.; Johansson, B.; et al. Extreme Hypofractionation versus Conventionally Fractionated Radiotherapy for Intermediate Risk Prostate Cancer: Early Toxicity Results from the Scandinavian Randomized Phase III Trial “HYPO-RT-PC”. Int. J. Radiat. Oncol. Biol. Phys. 2016, 96, 938–939. [Google Scholar] [CrossRef]
- Widmark, A.; Gunnlaugsson, A.; Beckman, L.; Thellenberg-Karlsson, C.; Hoyer, M.; Lagerlund, M.; Kindblom, J.; Ginman, C.; Johansson, B.; Björnlinger, K.; et al. Ultra-hypofractionated versus conventionally fractionated radiotherapy for prostate cancer: 5-year outcomes of the HYPO-RT-PC randomised, non-inferiority, phase 3 trial. Lancet 2019, 394, 385–395. [Google Scholar] [CrossRef]
- Koontz, B.F.; Bossi, A.; Cozzarini, C.; Wiegel, T.; D’Amico, A. A systematic review of hypofractionation for primary management of prostate cancer. Eur. Urol. 2015, 68, 683–691. [Google Scholar] [CrossRef]
- Wortel, R.C.; Hoop, E.O.; Heemsbergen, W.D.; Pos, F.J.; Incrocci, L. Moderate Hypofractionation in Intermediate- and High-Risk, Localized Prostate Cancer: Health-Related Quality of Life From the Randomized, Phase 3 HYPRO Trial. Int. J. Radiat. Oncol. Biol. Phys. 2019, 103, 823–833. [Google Scholar] [CrossRef] [PubMed]
- Onjukka, E.; Uzan, J.; Baker, C.; Howard, L.; Nahum, A.; Syndikus, I. Twenty Fraction Prostate Radiotherapy with Intra-prostatic Boost: Results of a Pilot Study. Clin. Oncol. 2017, 29, 6–14. [Google Scholar] [CrossRef] [PubMed]
- Gatenby, R.A.; Silva, A.S.; Gillies, R.J.; Frieden, B.R. Adaptive Therapy. Cancer Res. 2009, 69, 4894–4903. [Google Scholar] [CrossRef] [Green Version]
- Berwouts, D.; Olteanu, L.A.M.; Duprez, F.; Vercauteren, T.; De Gersem, W.; De Neve, W.; Van de Wiele, C.; Madani, I. Three-phase adaptive dose-painting-by-numbers for head-and-neck cancer: Initial results of the phase I clinical trial. Radiother. Oncol. 2013, 107, 310–316. [Google Scholar] [CrossRef]
- Differding, S.; Sterpin, E.; Janssens, G.; Hanin, F.-X.; Lee, J.A.; Grégoire, V. Methodology for adaptive and robust FDG-PET escalated dose painting by numbers in head and neck tumors. Acta Oncol. 2016, 55, 217–225. [Google Scholar] [CrossRef] [Green Version]
- Duprez, F.; De Neve, W.; De Gersem, W.; Coghe, M.; Madani, I. Adaptive Dose Painting by Numbers for Head-and-Neck Cancer. Int. J. Radiat. Oncol. Biol. Phys. 2011, 80, 1045–1055. [Google Scholar] [CrossRef]
- Servagi-Vernat, S.; Differding, S.; Sterpin, E.; Hanin, F.-X.; Labar, D.; Bol, A.; Lee, J.A.; Grégoire, V. Hypoxia-guided adaptive radiation dose escalation in head and neck carcinoma: A planning study. Acta Oncol. 2015, 54, 1008–1016. [Google Scholar] [CrossRef] [Green Version]




© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Her, E.J.; Haworth, A.; Rowshanfarzad, P.; Ebert, M.A. Progress towards Patient-Specific, Spatially-Continuous Radiobiological Dose Prescription and Planning in Prostate Cancer IMRT: An Overview. Cancers 2020, 12, 854. https://doi.org/10.3390/cancers12040854
Her EJ, Haworth A, Rowshanfarzad P, Ebert MA. Progress towards Patient-Specific, Spatially-Continuous Radiobiological Dose Prescription and Planning in Prostate Cancer IMRT: An Overview. Cancers. 2020; 12(4):854. https://doi.org/10.3390/cancers12040854
Chicago/Turabian StyleHer, Emily Jungmin, Annette Haworth, Pejman Rowshanfarzad, and Martin A. Ebert. 2020. "Progress towards Patient-Specific, Spatially-Continuous Radiobiological Dose Prescription and Planning in Prostate Cancer IMRT: An Overview" Cancers 12, no. 4: 854. https://doi.org/10.3390/cancers12040854




