Cyclin E2 Promotes Whole Genome Doubling in Breast Cancer
Abstract
1. Background
2. Results
2.1. Cyclin E2 Expression and Amplification Is Higher in Breast Cancers that Have Undergone Genome Doubling
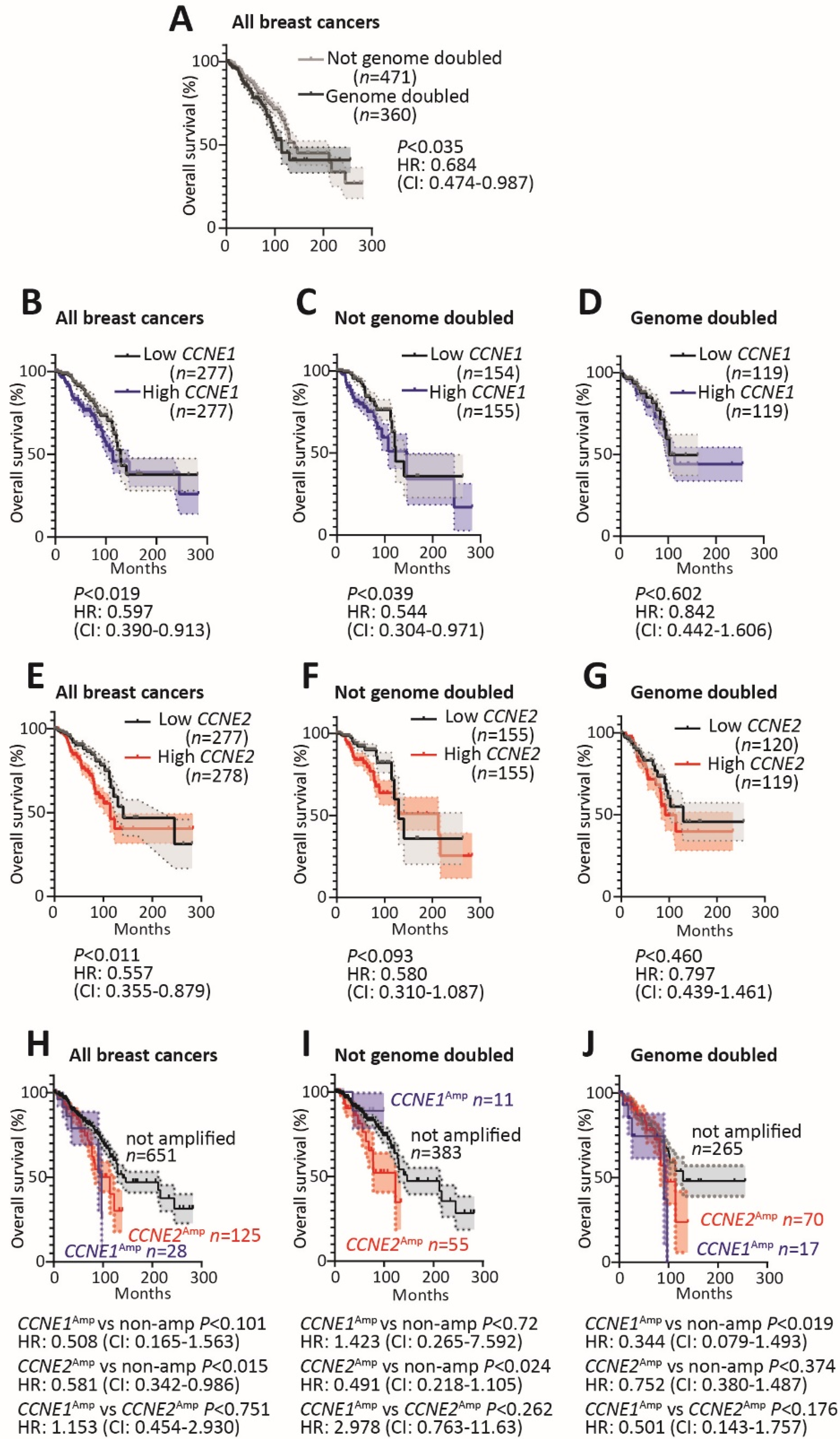
2.2. Survival Analysis in Genome Doubled and Non Genome Doubled Cancers Based on CCNE1 and CCNE2 Expression and Amplification
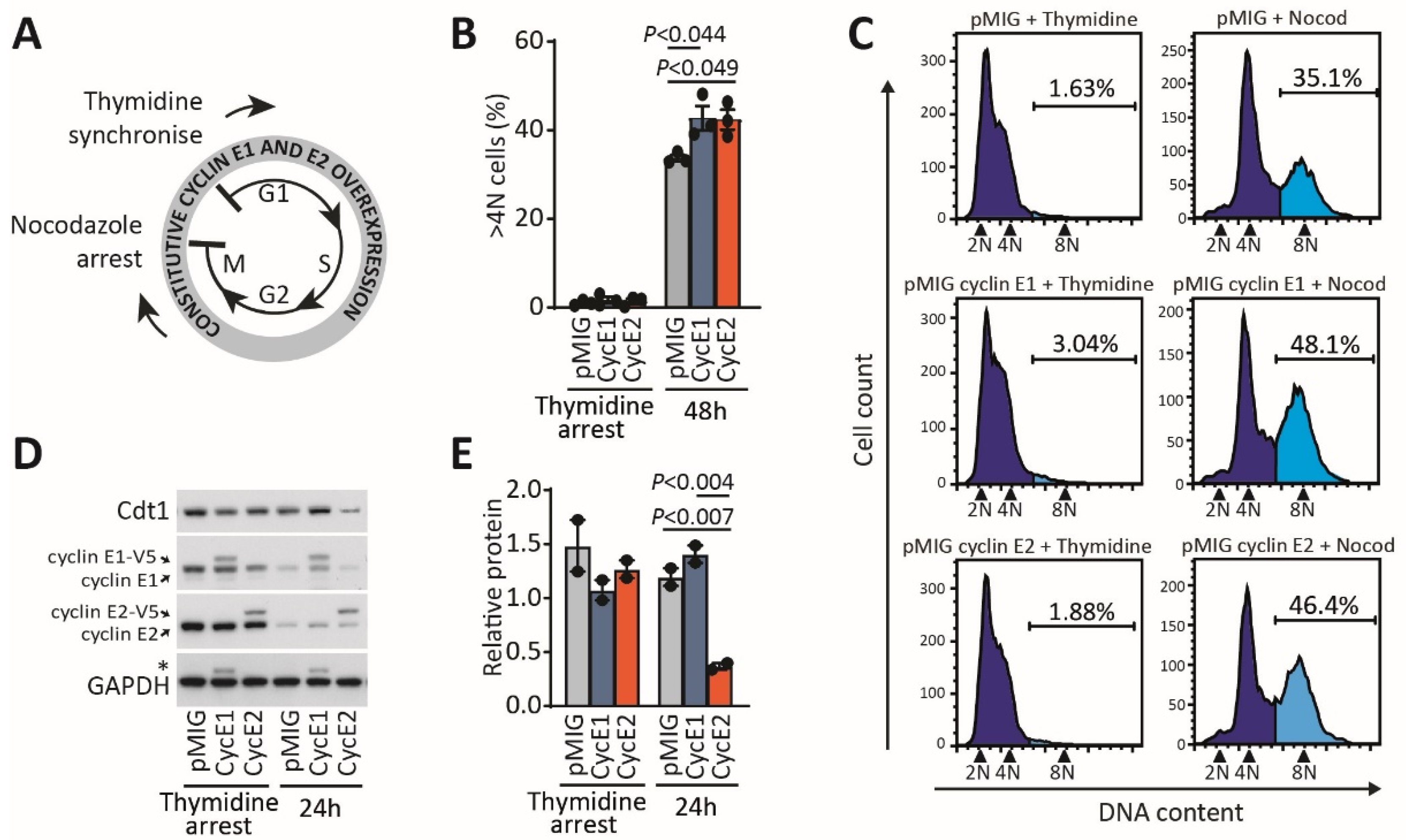
2.3. Excess Cyclin E2 Increases Mitotic Aberrations without Increasing the Length of Mitosis
2.4. Cyclin E2 Preferentially Localises to Chromatin during Endoreduplication
2.5. Depletion of Cyclin E2, But Not Cyclin E1, Ablates Rereplication
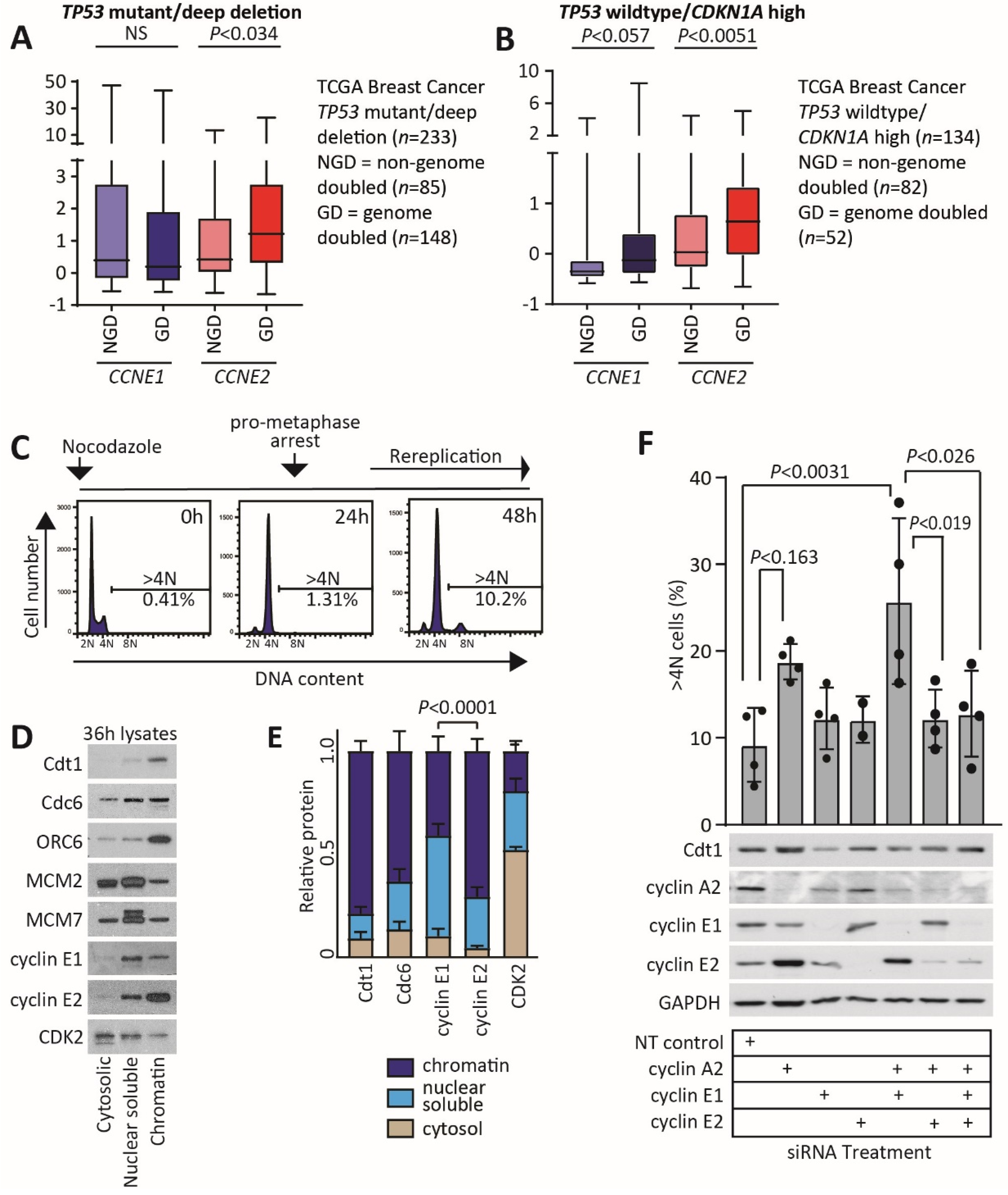
2.6. Cyclin E2 Is Associated with Genome Doubling in p53 Wildtype Breast Cancers, and Its Depletion in p53 Wildtype Breast Cancer Cells Reduces DNA Rereplication
3. Discussion
4. Methods
4.1. Cell Culture
4.2. Synchronisation/Drug Treatments
4.3. Immunoblotting and Immunoprecipitation
4.4. Flow Cytometry
4.5. Microscopy
4.6. siRNA Transfection
4.7. TCGA (The Cancer Genome Atlas) Dataset and Statistical Analysis
4.8. Survival Analyses
4.9. Statistics
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| CDK | cyclin-dependent kinase |
| CIN25 | 25-gene index of chromosomal instability |
| GD | genome doubled |
| GFP | green fluorescent protein |
| K-M | Kaplan–Meier |
| pMIG | pMSCV-IRES-GFP retroviral expression vector |
| preRC | pre-replication complex |
| MCM | minichromosome maintenance proteins |
| NGD | non-genome doubled |
| siRNA | small interfering RNA |
| TCGA | The Cancer Genome Atlas |
| 2N | two sets of chromosomes |
| 4N | four sets of chromosomes |
| 8N | eight sets of chromosomes |
References
- Bielski, C.M.; Zehir, A.; Penson, A.V.; Donoghue, M.T.A.; Chatila, W.; Armenia, J.; Chang, M.T.; Schram, A.M.; Jonsson, P.; Bandlamudi, C.; et al. Genome doubling shapes the evolution and prognosis of advanced cancers. Nat. Genet. 2018, 50, 1189–1195. [Google Scholar] [CrossRef] [PubMed]
- Zack, T.I.; Schumacher, S.E.; Carter, S.L.; Cherniack, A.D.; Saksena, G.; Tabak, B.; Lawrence, M.S.; Zhang, C.-Z.; Wala, J.; Mermel, C.H.; et al. Pan-cancer patterns of somatic copy number alteration. Nat. Genet. 2013, 45, 1134–1140. [Google Scholar] [CrossRef] [PubMed]
- Priestley, P.; Baber, J.; Lolkema, M.P.; Steeghs, N.; de Bruijn, E.; Shale, C.; Duyvesteyn, K.; Haidari, S.; Van Hoeck, A.; Onstenk, W.; et al. Pan-cancer whole-genome analyses of metastatic solid tumours. Nature 2019, 575, 210–216. [Google Scholar] [CrossRef] [PubMed]
- Dewhurst, S.M.; McGranahan, N.; Burrell, R.A.; Rowan, A.J.; Grönroos, E.; Endesfelder, D.; Joshi, T.; Mouradov, D.; Gibbs, P.; Ward, R.L.; et al. Tolerance of Whole-Genome Doubling Propagates Chromosomal Instability and Accelerates Cancer Genome Evolution. Cancer Discov. 2014, 4, 175–185. [Google Scholar] [CrossRef]
- Kuznetsova, A.Y.; Seget, K.; Moeller, G.K.; De Pagter, M.S.; De Roos, J.A.D.M.; Dürrbaum, M.; Kuffer, C.; Müller, S.; Zaman, G.J.R.; Kloosterman, W.P.; et al. Chromosomal instability, tolerance of mitotic errors and multidrug resistance are promoted by tetraploidization in human cells. Cell Cycle 2015, 14, 2810–2820. [Google Scholar] [CrossRef]
- Vargas-Rondón, N.; Villegas, V.E.; Rondón-Lagos, M. The Role of Chromosomal Instability in Cancer and Therapeutic Responses. Cancers 2017, 10, 4. [Google Scholar] [CrossRef]
- Etemadmoghadam, D.; Au-Yeung, G.; Wall, M.; Mitchell, C.; Kansara, M.; Loehrer, E.; Batzios, C.; George, J.; Ftouni, S.; Weir, B.A.; et al. Resistance to CDK2 Inhibitors Is Associated with Selection of Polyploid Cells in CCNE1-Amplified Ovarian Cancer. Clin. Cancer Res. 2013, 19, 5960–5971. [Google Scholar] [CrossRef]
- Geng, Y.; Yu, Q.; Sicinska, E.; Das, M.; Schneider, J.E.; Bhattacharya, S.; Rideout, W.M.; Bronson, R.T.; Gardner, H.; Sicinski, P. Cyclin E ablation in the mouse. Cell 2003, 114, 431–443. [Google Scholar] [CrossRef]
- Parisi, T.; Beck, A.R.; Rougier, N.; McNeil, T.; Lucian, L.; Werb, Z.; Amati, B. Cyclins E1 and E2 are required for endoreplication in placental trophoblast giant cells. EMBO J. 2003, 22, 4794–4803. [Google Scholar] [CrossRef]
- Gao, S.; Ma, J.J.; Lu, C. Prognostic value of cyclin E expression in breast cancer: A meta-analysis. Tumour Biol. 2013, 34, 3423–3430. [Google Scholar] [CrossRef]
- Bortner, D.M.; Rosenberg, M.P. Induction of mammary gland hyperplasia and carcinomas in transgenic mice expressing human cyclin E. Mol. Cell. Biol. 1997, 17, 453–459. [Google Scholar] [CrossRef]
- Teixeira Leonardo, K.; Wang, X.; Li, Y.; Ekholm-Reed, S.; Wu, X.; Wang, P.; Reed Steven, I. Cyclin E Deregulation Promotes Loss of Specific Genomic Regions. Curr. Biol. 2015, 25, 1327–1333. [Google Scholar] [CrossRef] [PubMed]
- Keck, J.M.; Summers, M.K.; Tedesco, D.; Ekholm-Reed, S.; Chuang, L.-C.; Jackson, P.K.; Reed, S.I. Cyclin E overexpression impairs progression through mitosis by inhibiting APCCdh1. J. Cell Biol. 2007, 178, 371–385. [Google Scholar] [CrossRef] [PubMed]
- Caldon, C.E.; Musgrove, E.A. Distinct and redundant functions of cyclin E1 and cyclin E2 in development and cancer. Cell Div. 2010, 5, 2. [Google Scholar] [PubMed]
- Wang, Y.; Klijn, J.G.; Zhang, Y.; Sieuwerts, A.M.; Look, M.P.; Yang, F.; Talantov, D.; Timmermans, M.; Meijer-Van Gelder, M.E.; Yu, J.; et al. Gene-expression profiles to predict distant metastasis of lymph-node-negative primary breast cancer. Lancet 2005, 365, 671–679. [Google Scholar] [CrossRef]
- Van’t Veer, L.J.; Dai, H.; Van De Vijver, M.J.; He, Y.D.; Hart, A.A.; Mao, M.; Peterse, H.L.; Van Der Kooy, K.; Marton, M.J.; Witteveen, A.T.; et al. Gene expression profiling predicts clinical outcome of breast cancer. Nature 2002, 415, 530–536. [Google Scholar] [CrossRef]
- Caldon, C.E.; Sergio, C.M.; Sutherland, R.L.; Musgrove, E.A. Differences in degradation lead to asynchronous expression of cyclin E1 and cyclin E2 in cancer cells. Cell Cycle 2013, 12, 596–605. [Google Scholar] [CrossRef]
- Caldon, C.E.; Sergio, C.M.; Schutte, J.; Boersma, M.N.; Sutherland, R.L.; Carroll, J.S.; Musgrove, E.A. Estrogen regulation of cyclin E2 requires cyclin D1, but not c-Myc. Mol. Cell. Biol. 2009, 29, 4623–4639. [Google Scholar] [CrossRef]
- Caldon, C.E.; Sergio, C.M.; Burgess, A.; Deans, A.J.; Sutherland, R.L.; Musgrove, E.A. Cyclin E2 induces genomic instability by mechanisms distinct from cyclin E1. Cell Cycle 2013, 12, 606–617. [Google Scholar] [CrossRef]
- Carter, S.L.; Eklund, A.C.; Kohane, I.S.; Harris, L.N.; Szallasi, Z. A signature of chromosomal instability inferred from gene expression profiles predicts clinical outcome in multiple human cancers. Nat. Genet. 2006, 38, 1043. [Google Scholar] [CrossRef]
- Cortés, F.; Mateos, S.; Pastor, N.; Domínguez, I. Toward a comprehensive model for induced endoreduplication. Life Sci. 2004, 76, 121–135. [Google Scholar] [CrossRef] [PubMed]
- Margolis, R.L. Tetraploidy and tumor development. Cancer Cell 2005, 8, 353–354. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Hall, J.R.; Lee, H.O.; Bunker, B.D.; Dorn, E.S.; Rogers, G.C.; Duronio, R.J.; Cook, J.G. Cdt1 and Cdc6 Are Destabilized by Rereplication-induced DNA Damage. J. Biol. Chem. 2008, 283, 25356–25363. [Google Scholar] [CrossRef] [PubMed]
- Geng, Y.; Lee, Y.-M.; Welcker, M.; Swanger, J.; Zagozdzon, A.; Winer, J.D.; Roberts, J.M.; Kaldis, P.; Clurman, B.E.; Sicinski, P. Kinase-independent function of cyclin E. Mol. Cell 2007, 25, 127–139. [Google Scholar] [CrossRef]
- Ekholm-Reed, S.; Mendez, J.; Tedesco, D.; Zetterberg, A.; Stillman, B.; Reed, S.I. Deregulation of cyclin E in human cells interferes with prereplication complex assembly. J. Cell Biol. 2004, 165, 789–800. [Google Scholar] [CrossRef]
- Ma, H.T.; Tsang, Y.H.; Marxer, M.; Poon, R.Y. Cyclin A2-cyclin-dependent kinase 2 cooperates with the PLK1-SCFbeta-TrCP1-EMI1-anaphase-promoting complex/cyclosome axis to promote genome reduplication in the absence of mitosis. Mol. Cell. Biol. 2009, 29, 6500–6514. [Google Scholar] [CrossRef]
- Chen, S.; Bell, S.P. CDK prevents Mcm2–7 helicase loading by inhibiting Cdt1 interaction with Orc6. Genes Dev. 2011, 25, 363–372. [Google Scholar] [CrossRef]
- Lovejoy, C.A.; Lock, K.; Yenamandra, A.; Cortez, D. DDB1 Maintains Genome Integrity through Regulation of Cdt1. Mol. Cell. Biol. 2006, 26, 7977–7990. [Google Scholar] [CrossRef]
- Ganem, N.J.; Pellman, D. Limiting the Proliferation of Polyploid Cells. Cell 2007, 131, 437–440. [Google Scholar] [CrossRef]
- Duffy, M.J.; Synnott, N.C.; Crown, J. Mutant p53 in breast cancer: Potential as a therapeutic target and biomarker. Breast Cancer Res. Treatment 2018, 170, 213–219. [Google Scholar] [CrossRef]
- Dimitrova, D.S.; Prokhorova, T.A.; Blow, J.J.; Todorov, I.T.; Gilbert, D.M. Mammalian nuclei become licensed for DNA replication during late telophase. J. Cell Sci. 2002, 115, 51–59. [Google Scholar] [PubMed]
- Fujita, M. Cdt1 revisited: Complex and tight regulation during the cell cycle and consequences of deregulation in mammalian cells. Cell Div. 2006, 1, 22. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Sansregret, L.; Vanhaesebroeck, B.; Swanton, C. Determinants and clinical implications of chromosomal instability in cancer. Nat. Rev. Clin. Oncol. 2018, 15, 139–150. [Google Scholar] [CrossRef] [PubMed]
- Blow, J.J.; Gillespie, P.J. Replication licensing and cancer—a fatal entanglement? Nat. Rev. Cancer 2008, 8, 799–806. [Google Scholar] [CrossRef] [PubMed]
- Lee, H.O.; Davidson, J.M.; Duronio, R.J. Endoreplication: Polyploidy with purpose. Genes Dev. 2009, 23, 2461–2477. [Google Scholar] [CrossRef]
- Sinha, D.; Duijf, P.H.G.; Khanna, K.K. Mitotic slippage: An old tale with a new twist. Cell Cycle 2019, 18, 7–15. [Google Scholar] [CrossRef]
- Caldon, C.E.; Sergio, C.M.; Kang, J.; Muthukaruppan, A.; Boersma, M.N.; Stone, A.; Barraclough, J.; Lee, C.S.; Black, M.A.; Miller, L.D.; et al. Cyclin E2 overexpression is associated with endocrine resistance but not insensitivity to CDK2 inhibition in human breast cancer cells. Mol. Cancer Ther. 2012, 11, 1488–1499. [Google Scholar] [CrossRef]
- Dai, H.; Van’t Veer, L.; Lamb, J.; He, Y.D.; Mao, M.; Fine, B.M.; Bernards, R.; Van De Vijver, M.; Deutsch, P.; Sachs, A.; et al. A Cell Proliferation Signature Is a Marker of Extremely Poor Outcome in a Subpopulation of Breast Cancer Patients. Cancer Res. 2005, 65, 4059–4066. [Google Scholar] [CrossRef]
- Zhang, Y.; Martens, J.W.M.; Yu, J.X.; Jiang, J.; Sieuwerts, A.M.; Smid, M.; Klijn, J.G.M.; Wang, Y.; Foekens, J.A. Copy Number alterations that predict metastatic capability of human breast cancer. Cancer Res. 2009, 69, 3795–3801. [Google Scholar] [CrossRef]
- Duffy, S.; Fam, H.K.; Wang, Y.K.; Styles, E.B.; Kim, J.-H.; Ang, J.S.; Singh, T.; Larionov, V.; Shah, S.P.; Andrews, B.; et al. Overexpression screens identify conserved dosage chromosome instability genes in yeast and human cancer. Proc. Natl. Acad. Sci. USA 2016, 113, 9967–9976. [Google Scholar] [CrossRef]
- Crockford, A.; Zalmas, L.P.; Grönroos, E.; Dewhurst, S.M.; McGranahan, N.; Cuomo, M.E.; Encheva, V.; Snijders, A.P.; Begum, J.; Purewal, S.; et al. Cyclin D mediates tolerance of genome-doubling in cancers with functional p53. Ann. Oncol. 2017, 28, 149–156. [Google Scholar] [CrossRef]
- Bagheri-Yarmand, R.; Biernacka, A.; Hunt, K.K.; Keyomarsi, K. Low molecular weight cyclin E overexpression shortens mitosis, leading to chromosome missegregation and centrosome amplification. Cancer Res. 2010, 70, 5074–5084. [Google Scholar] [CrossRef]
- Rogers, S.; Gloss, B.; Lee, C.; Sergio, C.; Dinger, M.; Musgrove, E.; Burgess, A.; Caldon, C. Cyclin E2 is the predominant E-cyclin associated with NPAT in breast cancer cells. Cell Div. 2015, 10, 1. [Google Scholar] [CrossRef] [PubMed]
- Caldon, C.E.; Swarbrick, A.; Lee, C.S.; Sutherland, R.L.; Musgrove, E.A. The helix-loop-helix protein Id1 requires cyclin D1 to promote the proliferation of mammary epithelial cell acini. Cancer Res. 2008, 68, 3026–3036. [Google Scholar] [CrossRef] [PubMed]
- Méndez, J.; Stillman, B. Chromatin association of human origin recognition complex, Cdc6, and minichromosome maintenance proteins during the cell cycle: Assembly of prereplication complexes in late mitosis. Mol. Cell. Biol. 2000, 20, 8602–8612. [Google Scholar] [CrossRef] [PubMed]
- Prall, O.W.J.; Rogan, E.M.; Musgrove, E.A.; Watts, C.K.W.; Sutherland, R.L. c-Myc or cyclin D1 mimics estrogen effects on cyclin E-Cdk2 activation and cell cycle reentry. Mol. Cell. Biol. 1998, 18, 4499–4508. [Google Scholar] [CrossRef] [PubMed]
- Rasband, W.S.; Image, J. In. Bethesda, Maryland, USA: U. S. National Institutes of Health, 1997–2007. Available online: http://rsb.info.nih.gov/ij/ (accessed on 13 August 2020).
- Caldon, C.E.; Burgess, A. Label free, quantitative single-cell fate tracking of time-lapse movies. MethodsX 2019, 6, 2468–2475. [Google Scholar] [CrossRef]
- Cerami, E.; Gao, J.; Dogrusoz, U.; Gross, B.E.; Sumer, S.O.; Aksoy, B.A.; Jacobsen, A.; Byrne, C.J.; Heuer, M.L.; Larsson, E.; et al. The cBio cancer genomics portal: An open platform for exploring multidimensional cancer genomics data. Cancer Discov. 2012, 2, 401–404. [Google Scholar] [CrossRef]
- Gao, J.; Aksoy, B.A.; Dogrusoz, U.; Dresdner, G.; Gross, B.; Sumer, S.O.; Sun, Y.; Jacobsen, A.; Sinha, R.; Larsson, E.; et al. Integrative Analysis of Complex Cancer Genomics and Clinical Profiles Using the cBioPortal. Sci. Signal. 2013, 6, pl1. [Google Scholar] [CrossRef]
- Chen, W.; Robertson, A.J.; Ganesamoorthy, D.; Coin, L.J.M. sCNAphase: Using haplotype resolved read depth to genotype somatic copy number alterations from low cellularity aneuploid tumors. Nucleic Acids Res. 2017, 45, e34. [Google Scholar] [CrossRef]
- Van Loo, P.; Nordgard, S.H.; Lingjærde, O.C.; Russnes, H.G.; Rye, I.H.; Sun, W.; Weigman, V.J.; Marynen, P.; Zetterberg, A.; Naume, B.; et al. Allele-specific copy number analysis of tumors. Proc. Natl. Acad. Sci. USA 2010, 107, 16910–16915. [Google Scholar] [CrossRef] [PubMed]




© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lee, C.; Fernandez, K.J.; Alexandrou, S.; Sergio, C.M.; Deng, N.; Rogers, S.; Burgess, A.; Caldon, C.E. Cyclin E2 Promotes Whole Genome Doubling in Breast Cancer. Cancers 2020, 12, 2268. https://doi.org/10.3390/cancers12082268
Lee C, Fernandez KJ, Alexandrou S, Sergio CM, Deng N, Rogers S, Burgess A, Caldon CE. Cyclin E2 Promotes Whole Genome Doubling in Breast Cancer. Cancers. 2020; 12(8):2268. https://doi.org/10.3390/cancers12082268
Chicago/Turabian StyleLee, Christine, Kristine J. Fernandez, Sarah Alexandrou, C. Marcelo Sergio, Niantao Deng, Samuel Rogers, Andrew Burgess, and C. Elizabeth Caldon. 2020. "Cyclin E2 Promotes Whole Genome Doubling in Breast Cancer" Cancers 12, no. 8: 2268. https://doi.org/10.3390/cancers12082268
APA StyleLee, C., Fernandez, K. J., Alexandrou, S., Sergio, C. M., Deng, N., Rogers, S., Burgess, A., & Caldon, C. E. (2020). Cyclin E2 Promotes Whole Genome Doubling in Breast Cancer. Cancers, 12(8), 2268. https://doi.org/10.3390/cancers12082268




