The Diagnostic Accuracy of Mutant KRAS Detection from Pancreatic Secretions for the Diagnosis of Pancreatic Cancer: A Meta-Analysis
Abstract
:1. Introduction
2. Results
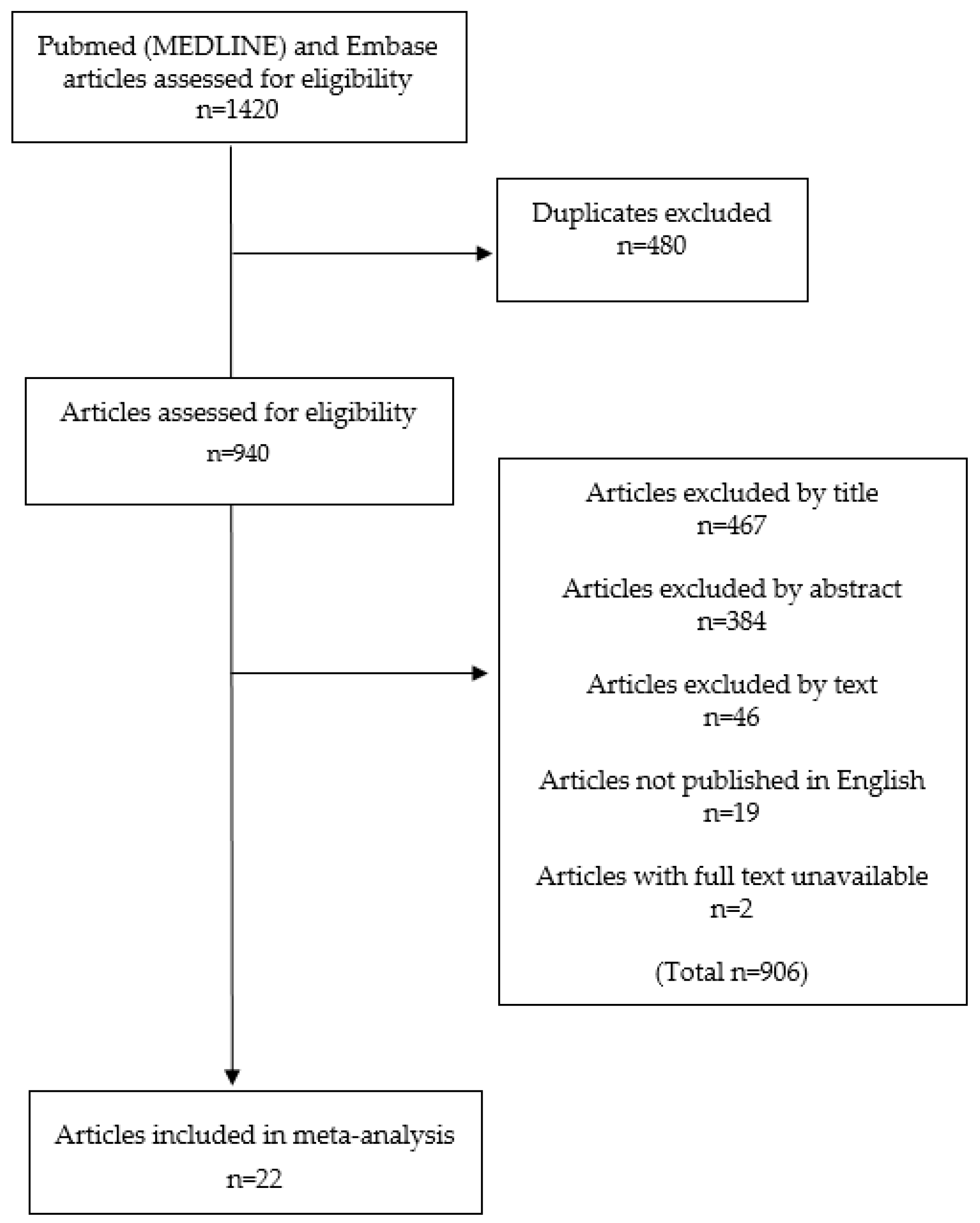
2.1. Study Inclusion
2.2. Study Demographics
2.3. Mutant KRAS Determination.
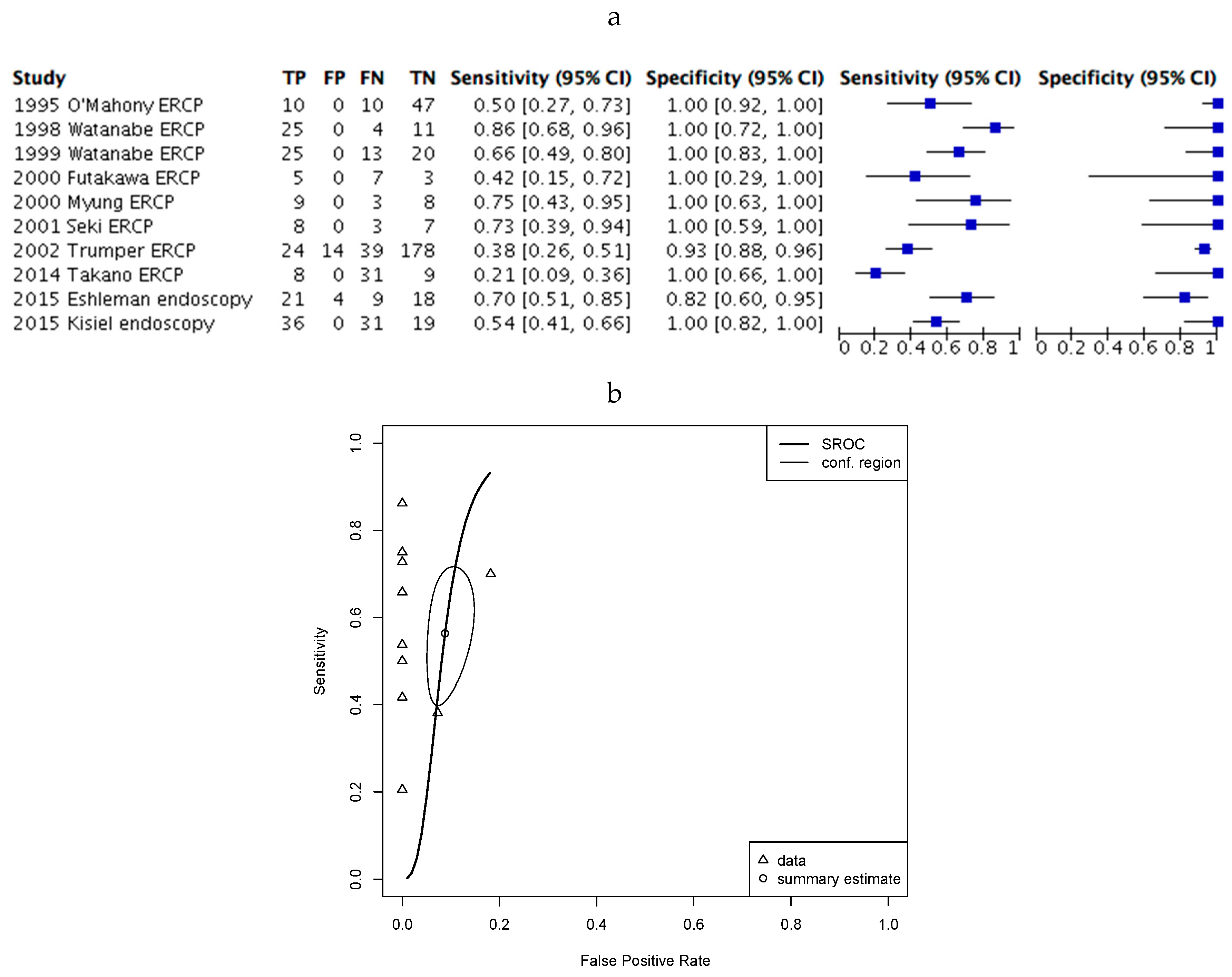
2.4. Assessment of Diagnostic Accuracy.
3. Discussion
4. Materials and Methods
4.1. Search Strategy
4.2. Inclusion/Exclusion Criteria
4.3. Data Extraction and Statistics
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Fokas, E.; O’Neill, E.; Gordon-Weeks, A.; Mukherjee, S.; McKenna, W.G.; Muschel, R.J. Pancreatic ductal adenocarcinoma: From genetics to biology to radiobiology to oncoimmunology and all the way back to the clinic. Biochim. Biophys. Acta 2015, 1855, 61–82. [Google Scholar] [CrossRef]
- Ghaneh, P.; Hanson, R.; Titman, A.; Lancaster, G.; Plumpton, C.; Lloyd-Williams, H.; Yeo, S.T.; Edwards, R.T.; Johnson, C.; Abu Hilal, M.; et al. PET-PANC: Multicentre prospective diagnostic accuracy and health economic analysis study of the impact of combined modality 18fluorine-2-fluoro-2-deoxy-d-glucose positron emission tomography with computed tomography scanning in the diagnosis and management of pancreatic cancer. Health Technol. Assess. 2018, 22, 1–114. [Google Scholar] [CrossRef] [PubMed]
- Klaiber, U.; Hackert, T.; Neoptolemos, J.P. Adjuvant treatment for pancreatic cancer. Transl. Gastroenterol. Hepatol. 2019, 4, 27. [Google Scholar] [CrossRef]
- Chawla, A.; Ferrone, C.R. Neoadjuvant Therapy for Resectable Pancreatic Cancer: An Evolving Paradigm Shift. Front. Oncol. 2019, 9, 1085. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Handgraaf, H.J.M.; Boonstra, M.C.; Van Erkel, A.R.; Bonsing, B.A.; Putter, H.; Van De Velde, C.J.; Vahrmeijer, A.L.; Mieog, J.S.D. Current and future intraoperative imaging strategies to increase radical resection rates in pancreatic cancer surgery. Biomed. Res. Int. 2014, 890230. [Google Scholar] [CrossRef] [PubMed]
- Saad, A.M.; Turk, T.; Al-Husseini, M.J.; Abdel-Rahman, O. Trends in pancreatic adenocarcinoma incidence and mortality in the United States in the last four decades; a SEER-based study. BMC Cancer 2018, 18, 688. [Google Scholar] [CrossRef]
- Ghaneh, P.; Kleeff, J.; Halloran, C.M.; Raraty, M.; Jackson, R.; Melling, J.; Jones, O.; Palmer, D.H.; Cox, T.F.; Smith, C.J.; et al. The Impact of Positive Resection Margins on Survival and Recurrence Following Resection and Adjuvant Chemotherapy for Pancreatic Ductal Adenocarcinoma. Ann. Surg. 2019, 269, 520–529. [Google Scholar] [CrossRef]
- Neoptolemos, J.P.; Stocken, D.D.; Dunn, J.A.; Almond, J.; Beger, H.G.; Pederzoli, P.; Bassi, C.; Dervenis, C.; Fernandez-Cruz, L.; Lacaine, F.; et al. Influence of resection margins on survival for patients with pancreatic cancer treated by adjuvant chemoradiation and/or chemotherapy in the ESPAC-1 randomized controlled trial. Ann. Surg. 2001, 234, 758–768. [Google Scholar] [CrossRef]
- Paniccia, A.; Hosokawa, P.; Henderson, W.; Schulick, R.D.; Edil, B.H.; McCarter, M.D.; Gajdos, C. Characteristics of 10-Year Survivors of Pancreatic Ductal Adenocarcinoma. JAMA Surg. 2015, 150, 701–710. [Google Scholar] [CrossRef] [Green Version]
- Schnelldorfer, T.; Ware, A.L.; Sarr, M.G.; Smyrk, T.C.; Zhang, L.; Qin, R.; Gullerud, R.E.; Donohue, J.H.; Nagorney, D.M.; Farnell, M.B. Long-Term Survival After Pancreatoduodenectomy for Pancreatic Adenocarcinoma: Is Cure Possible? Ann. Surg. 2008, 247, 456–462. [Google Scholar] [CrossRef]
- Ying, H.; Dey, P.; Yao, W.; Kimmelman, A.C.; Draetta, G.F.; Maitra, A.; DePinho, R.A. Genetics and biology of pancreatic ductal adenocarcinoma. Genes Dev. 2016, 30, 355–385. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Khan, M.A.; Azim, S.; Zubair, H.; Bhardwaj, A.; Patel, G.K.; Khushman, M.; Singh, S.; Singh, A.P. Molecular Drivers of Pancreatic Cancer Pathogenesis: Looking Inward to Move Forward. Int. J. Mol. Sci. 2017, 18, 779. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hruban, R.H.; Takaori, K.; Klimstra, D.S.; Adsay, N.V.; Albores-Saavedra, J.; Biankin, A.V.; Biankin, S.A.; Compton, C.; Fukushima, N.; Furukawa, T.; et al. An Illustrated Consensus on the Classification of Pancreatic Intraepithelial Neoplasia and Intraductal Papillary Mucinous Neoplasms. Am. J. Surg. Pathol. 2004, 28, 977–987. [Google Scholar] [CrossRef]
- Waters, A.M.; Der, C.J. KRAS: The Critical Driver and Therapeutic Target for Pancreatic Cancer. Cold Spring Harb. Perspect. Med. 2018, 8, a031435. [Google Scholar] [CrossRef]
- The Cancer Genome Atlas Research Network. Integrated Genomic Characterization of Pancreatic Ductal Adenocarcinoma. Cancer Cell 2017, 32, 185–203.e13. [Google Scholar] [CrossRef] [PubMed]
- Yachida, S.; Jones, S.; Bozic, I.; Antal, T.; Leary, R.; Fu, B.; Kamiyama, M.; Hruban, R.H.; Eshleman, J.R.; Nowak, M.A.; et al. Distant metastasis occurs late during the genetic evolution of pancreatic cancer. Nature 2010, 467, 1114–1117. [Google Scholar] [CrossRef] [Green Version]
- Urban, T.; Ricci, S.; Grange, J.-D.; Lacave, R.; Boudghene, F.; Breittmayer, F.; Languille, O.; Roland, J.; Bernaudin, J.-F. Detection of c-Ki-ras Mutation by PCR/RFLP Analysis and Diagnosis of Pancreatic Adenocarcinomas. J. Natl. Cancer Inst. 1993, 85, 2008–2012. [Google Scholar] [CrossRef]
- Bournet, B.; Buscail, C.; Muscari, F.; Cordelier, P.; Buscail, L. Targeting KRAS for diagnosis, prognosis, and treatment of pancreatic cancer: Hopes and realities. Eur. J. Cancer 2016, 54, 75–83. [Google Scholar] [CrossRef]
- Narayanan, V.; Konstantinov, S.R.; Peppelenbosch, M.P. Mutations in KRAS: Are they a valid biomarker for pancreatic ductal adenocarcinomas diagnosis? Transl. Cancer Res. 2017, 6, S72–S77. [Google Scholar] [CrossRef]
- Lang, A.H.; Drexel, H.; Geller-Rhomberg, S.; Stark, N.; Winder, T.; Geiger, K.; Muendlein, A. Optimized Allele-Specific Real-Time PCR Assays for the Detection of Common Mutations in KRAS and BRAF. J. Mol. Diagn. 2011, 13, 23–28. [Google Scholar] [CrossRef]
- Earl, J.; Garcia-Nieto, S.; Martinez-Avila, J.C.; Montans, J.; Sanjuanbenito, A.; Rodríguez-Garrote, M.; Lisa, E.; Mendía, E.; Lobo, E.; Malats, N.; et al. Circulating tumor cells (Ctc) and kras mutant circulating free Dna (cfdna) detection in peripheral blood as biomarkers in patients diagnosed with exocrine pancreatic cancer. BMC Cancer 2015, 15, 797. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Le Calvez-Kelm, F.; Foll, M.; Wozniak, M.B.; Delhomme, T.M.; Durand, G.; Chopard, P.; Pertesi, M.; Fabianova, E.; Adamcakova, Z.; Holcatova, I.; et al. KRAS mutations in blood circulating cell-free DNA: A pancreatic cancer case-control. Oncotarget 2016, 7, 78827–78840. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wang, Z.-Y.; Ding, X.-Q.; Zhu, H.; Wang, R.-X.; Pan, X.-R.; Tong, J.-H. KRAS Mutant Allele Fraction in Circulating Cell-Free DNA Correlates with Clinical Stage in Pancreatic Cancer Patients. Front. Oncol. 2019, 9, 1295. [Google Scholar] [CrossRef] [PubMed]
- Chan, T.L.; Zhao, W.; Project, C.G.; Leung, S.Y.; Yuen, S.T. BRAF and KRAS Mutations in Colorectal Hyperplastic Polyps and Serrated Adenomas. Cancer Res. 2003, 63, 4878–4881. [Google Scholar] [PubMed]
- Tímár, J. The clinical relevance of KRAS gene mutation in non-small-cell lung cancer. Curr. Opin. Oncol. 2014, 26, 138–144. [Google Scholar] [CrossRef]
- O’mahony, S.; Longfellow, M.; McMahon, M.J.; Axon, A.T.; Quirke, P. Detection of c-Ki-ras mutations in bile samples from patients with pancreatic and biliary cancers. Clin. Mol. Pathol. 1995, 48, M316–M318. [Google Scholar] [CrossRef]
- Iguchi, H.; Sugano, K.; Fukayama, N.; Ohkura, H.; Sadamoto, K.; Ohkoshi, K.; Seo, Y.; Tomoda, H.; Funakoshi, A.; Wakasugi, H. Analysis of Ki-ras codon 12 mutations in the duodenal juice of patients with pancreatic cancer. Gastroenterology 1996, 110, 221–226. [Google Scholar] [CrossRef]
- Uehara, H.; Nakaizumi, A.; Baba, M.; Iishi, H.; Tatsuta, M.; Kitamura, T.; Ohigashi, H.; Ishikawa, O.; Takenaka, A.; Ishiguro, S. Diagnosis of pancreatic cancer by K-ras point mutation and cytology of pancreatic juice. Am. J. Gastroenterol. 1996, 91, 1616–1621. [Google Scholar]
- Furuya, N.; Kawa, S.; Akamatsu, T.; Furihata, K. Long-term follow-up of patients with chronic pancreatitis and K-ras gene mutation detected in pancreatic juice. Gastroenterology 1997, 113, 593–598. [Google Scholar] [CrossRef]
- Fukushima, N.; Suzuki, M.; Fukayama, M. Analysis of K-ras oncogene mutation directly applied to atypical cell clusters on cytologic smear slides of bile and pancreatic juice. Pathol. Int. 1998, 48, 33–40. [Google Scholar] [CrossRef] [PubMed]
- Watanabe, H.; Yamaguchi, Y.; Ha, A.; Hu, Y.X.; Motoo, Y.; Okai, T.; Yoshimura, T.; Sawabu, N. Quantitative determination of K-ras mutations in pancreatic juice for diagnosis of pancreatic cancer using hybridization protection assay. Pancreas 1998, 17, 341–347. [Google Scholar] [CrossRef] [PubMed]
- Watanabe, H.; Ha, A.; Hu, Y.X.; Ohtsubo, K.; Yamaguchi, Y.; Motoo, Y.; Okai, T.; Toya, D.; Tanaka, N.; Sawabu, N. K-ras mutations in duodenal aspirate without secretin stimulation for screening of pancreatic and biliary tract carcinoma. Cancer 1999, 86, 1441–1448. [Google Scholar] [CrossRef]
- Yamaguchi, Y.; Watanabe, H.; Yrdiran, S.; Ohtsubo, K.; Motoo, Y.; Okai, T.; Sawabu, N. Detection of mutations of p53 tumor suppressor gene in pancreatic juice and its application to diagnosis of patients with pancreatic cancer: Comparison with K-ras mutation. Clin. Cancer Res. 1999, 5, 1147–1153. [Google Scholar] [PubMed]
- Futakawa, N.; Kimura, W.; Yamagata, S.; Zhao, B.; Ilsoo, H.; Inoue, T.; Sata, N.; Kawaguchi, Y.; Kubota, Y.; Muto, T. Significance of K-ras mutation and CEA level in pancreatic juice in the diagnosis of pancreatic cancer. J. Hepato-Biliary-Pancreat. Surg. 2000, 7, 63–71. [Google Scholar] [CrossRef]
- Myung, S.J.; Kim, M.H.; Kim, Y.S.; Kim, H.J.; Park, E.T.; Yoo, K.S.; Lim, B.C.; Wan Seo, D.; Lee, S.K.; Min, Y.I.; et al. Telomerase activity in pure pancreatic juice for the diagnosis of pancreatic cancer may be complementary to K-ras mutation. Gastrointest. Endosc. 2000, 51, 708–713. [Google Scholar] [CrossRef]
- Boadas, J.; Mora, J.; Urgell, E.; Puig, P.; Roca, M.; Cussó, X.; Capellà, G.; Lluís, F.; Farré, A. Clinical usefulness of K-ras gene mutation detection and cytology in pancreatic juice in the diagnosis and screening of pancreatic cancer. Eur. J. Gastroenterol. Hepatol. 2001, 13, 1153–1159. [Google Scholar] [CrossRef]
- Ha, A.; Watanabe, H.; Yamaguchi, Y.; Ohtsubo, K.; Wang, Y.; Motoo, Y.; Okai, T.; Wakabayahi, T.; Sawabu, N. Usefulness of supernatant of pancreatic juice for genetic analysis of K-ras in diagnosis of pancreatic carcinoma. Pancreas 2001, 23, 356–363. [Google Scholar] [CrossRef]
- Pugliese, V.; Pujic, N.; Saccomanno, S.; Gatteschi, B.; Pera, C.; Aste, H.; Ferrara, G.B.; Nicolò, G. Pancreatic intraductal sampling during ERCP in patients with chronic pancreatitis and pancreatic cancer: Cytologic studies and k-ras-2 codon 12 molecular analysis in 47 cases. Gastrointest. Endosc. 2001, 54, 595–599. [Google Scholar] [CrossRef]
- Seki, K.; Suda, T.; Aoyagi, Y.; Sugawara, S.; Natsui, M.; Motoyama, H.; Shirai, Y.; Sekine, T.; Kawai, H.; Mita, Y.; et al. Diagnosis of pancreatic adenocarcinoma by detection of human telomerase reverse transcriptase messenger RNA in pancreatic juice with sample qualification. Clin. Cancer Res. 2001, 7, 1976–1981. [Google Scholar]
- Costentin, L.; Pagès, P.; Bouisson, M.; Berthelémy, P.; Buscail, L.; Escourrou, J.; Pradayrol, L.; Vaysse, N. Frequent deletions of tumor suppressor genes in pure pancreatic juice from patients with tumoral or nontumoral pancreatic diseases. Pancreatology 2002, 2, 17–25. [Google Scholar] [CrossRef]
- Wang, Y.; Yamaguchi, Y.; Watanabe, H.; Ohtsubo, K.; Motoo, Y.; Sawabu, N. Detection of p53 gene mutations in the supernatant of pancreatic juice and plasma from patients with pancreatic carcinomas. Pancreas 2004, 28, 13–19. [Google Scholar] [CrossRef] [PubMed]
- Trümper, L.; Menges, M.; Daus, H.; Köhler, D.; Reinhard, J.-O.; Sackmann, M.; Moser, C.; Sek, A.; Jacobs, G.; Zeitz, M.; et al. Low sensitivity of the ki-ras polymerase chain reaction for diagnosing pancreatic cancer from pancreatic juice and bile: A multicenter prospective trial. J. Clin. Oncol. 2002, 20, 4331–4337. [Google Scholar] [CrossRef] [PubMed]
- Shi, C.; Fukushima, N.; Abe, T.; Bian, Y.; Hua, L.; Wendelburg, B.J.; Yeo, C.J.; Hruban, R.H.; Goggins, M.G.; Eshleman, J.R. Sensitive and quantitative detection of KRAS2 gene mutations in pancreatic duct juice differentiates patients with pancreatic cancer from chronic pancreatitis, potential for early detection. Cancer Biol. 2008, 7, 353–360. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Takano, S.; Fukasawa, M.; Maekawa, S.; Kadokura, M.; Miura, M.; Shindo, H.; Takahashi, E.; Sato, T.; Enomoto, N. Deep sequencing of cancer-related genes revealed GNAS mutations to be associated with intraductal papillary mucinous neoplasms and its main pancreatic duct dilation. PLoS ONE 2014, 9, e98718. [Google Scholar] [CrossRef]
- Eshleman, J.R.; Norris, A.L.; Sadakari, Y.; Debeljak, M.; Borges, M.; Harrington, C.; Lin, E.; Brant, A.; Barkley, T.; Almario, J.A.; et al. KRAS and guanine nucleotide-binding protein mutations in pancreatic juice collected from the duodenum of patients at high risk for neoplasia undergoing endoscopic ultrasound. Clin. Gastroenterol. Hepatol. 2015, 13, 963–969.e4. [Google Scholar] [CrossRef] [Green Version]
- Kisiel, J.B.; Raimondo, M.; Taylor, W.R.; Yab, T.C.; Mahoney, D.W.; Sun, Z.; Middha, S.; Baheti, S.; Zou, H.; Smyrk, T.C.; et al. New DNA Methylation Markers for Pancreatic Cancer: Discovery, Tissue Validation, and Pilot Testing in Pancreatic Juice. Clin. Cancer Res. 2015, 21, 4473–4481. [Google Scholar] [CrossRef] [Green Version]
- Ginesta, M.M.; Diaz-Riascos, Z.V.; Busquets, J.; Pelaez, N.; Serrano, T.; Peinado, M.À.; Jorba, R.; García-Borobia, F.J.; Capella, G.; Fabregat, J. APC promoter is frequently methylated in pancreatic juice of patients with pancreatic carcinomas or periampullary tumors. Oncol. Lett. 2016, 12, 2210–2216. [Google Scholar] [CrossRef] [Green Version]
- Canto, M.I.; Harinck, F.; Hruban, R.H.; Offerhaus, G.J.; Poley, J.-W.; Kamel, I.; Nio, Y.; Schulick, R.S.; Bassi, C.; Kluijt, I.; et al. International Cancer of the Pancreas Screening (CAPS) Consortium summit on the management of patients with increased risk for familial pancreatic cancer. Gut 2013, 62, 339–347. [Google Scholar] [CrossRef]
- Li, W.-M.; Hu, T.-T.; Zhou, L.-L.; Feng, Y.-M.; Wang, Y.-Y.; Fang, J. Highly sensitive detection of the PIK3CAH1047R mutation in colorectal cancer using a novel PCR-RFLP method. BMC Cancer 2016, 16, 454. [Google Scholar] [CrossRef] [Green Version]
- Rutjes, A.W.S.; Reitsma, J.B.; Di Nisio, M.; Smidt, N.; van Rijn, J.C.; Bossuyt, P.M.M. Evidence of bias and variation in diagnostic accuracy studies. CMAJ 2006, 174, 469–476. [Google Scholar] [CrossRef] [Green Version]
- Lijmer, J.G.; Mol, B.W.; Heisterkamp, S.; Bonsel, G.J.; Prins, M.H.; van der Meulen, J.H.; Bossuyt, P.M. Empirical evidence of design-related bias in studies of diagnostic tests. JAMA 1999, 282, 1061–1066. [Google Scholar] [CrossRef] [PubMed]
- Pourshams, A.; Sepanlou, S.G.; Ikuta, K.S.; Bisignano, C.; Safiri, S.; Roshandel, G.; Sharif, M.; Khatibian, M.; Fitzmaurice, C.; Nixon, M.R.; et al. The global, regional, and national burden of pancreatic cancer and its attributable risk factors in 195 countries and territories, 1990–2017: A systematic analysis for the Global Burden of Disease Study 2017. Lancet Gastroenterol. Hepatol. 2019, 4, 934–947. [Google Scholar] [CrossRef] [Green Version]
- Wang, X.; Wang, J.; Chen, F.; Zhong, Z.; Qi, L. Detection of K-ras gene mutations in feces by magnetic nanoprobe in patients with pancreatic cancer: A preliminary study. Exp. Med. 2018, 15, 527–531. [Google Scholar] [CrossRef] [PubMed]
- Kisiel, J.B.; Yab, T.C.; Taylor, W.R.; Chari, S.T.; Petersen, G.M.; Mahoney, D.W.; Ahlquist, D.A. Stool DNA testing for the detection of pancreatic cancer: Assessment of methylation marker candidates. Cancer 2012, 118, 2623–2631. [Google Scholar] [CrossRef] [Green Version]
- Choi, M.H.; Mejlænder-Andersen, E.; Manueldas, S.; El Jellas, K.; Steine, S.J.; Tjensvoll, K.; Sætran, H.A.; Knappskog, S.; Hoem, D.; Nordgård, O.; et al. Mutation analysis by deep sequencing of pancreatic juice from patients with pancreatic ductal adenocarcinoma. BMC Cancer 2019, 19, 11. [Google Scholar] [CrossRef]
- Taylor, S.C.; Laperriere, G.; Germain, H. Droplet Digital PCR versus qPCR for gene expression analysis with low abundant targets: From variable nonsense to publication quality data. Sci. Rep. 2017, 7, 1–8. [Google Scholar] [CrossRef] [Green Version]
- Reid, M.D.; Choi, H.; Balci, S.; Akkas, G.; Adsay, V. Serous cystic neoplasms of the pancreas: Clinicopathologic and molecular characteristics. Semin. Diagn. Pathol. 2014, 31, 475–483. [Google Scholar] [CrossRef]
- Nikiforova, M.N.; Khalid, A.; Fasanella, K.E.; McGrath, K.M.; Brand, R.E.; Chennat, J.S.; Slivka, A.; Zeh, H.J.; Zureikat, A.H.; Krasinskas, A.M.; et al. Integration of KRAS testing in the diagnosis of pancreatic cystic lesions: A clinical experience of 618 pancreatic cysts. Mod. Pathol. 2013, 26, 1478–1487. [Google Scholar] [CrossRef] [Green Version]
- Singhi, A.D.; Nikiforova, M.N.; Fasanella, K.E.; McGrath, K.M.; Pai, R.K.; Ohori, N.P.; Bartholow, T.L.; Brand, R.E.; Chennat, J.S.; Lu, X.; et al. Preoperative GNAS and KRAS testing in the diagnosis of pancreatic mucinous cysts. Clin. Cancer Res. 2014, 20, 4381–4389. [Google Scholar] [CrossRef] [Green Version]
- Singhi, A.D.; McGrath, K.; Brand, R.E.; Khalid, A.; Zeh, H.J.; Chennat, J.S.; Fasanella, K.E.; Papachristou, G.I.; Slivka, A.; Bartlett, D.L.; et al. Preoperative next-generation sequencing of pancreatic cyst fluid is highly accurate in cyst classification and detection of advanced neoplasia. Gut 2018, 67, 2131–2141. [Google Scholar] [CrossRef] [Green Version]
- van Enst, W.A.; Ochodo, E.; Scholten, R.J.; Hooft, L.; Leeflang, M.M. Investigation of publication bias in meta-analyses of diagnostic test accuracy: A meta-epidemiological study. BMC Med. Res. Methodol. 2014, 14, 70. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Stroup, D.F.; Berlin, J.A.; Morton, S.C.; Olkin, I.; Williamson, G.D.; Rennie, D.; Moher, D.; Becker, B.J.; Sipe, T.A.; Thacker, S.B.; et al. Meta-analysis of Observational Studies in Epidemiology: A Proposal for Reporting. JAMA 2000, 283, 2008–2012. [Google Scholar] [CrossRef] [PubMed]
- McInnes, M.D.F.; Moher, D.; Thombs, B.D.; McGrath, T.A.; Bossuyt, P.M.; Clifford, T.; Cohen, J.F.; Deeks, J.J.; Gatsonis, C.; Hooft, L.; et al. Preferred Reporting Items for a Systematic Review and Meta-analysis of Diagnostic Test Accuracy Studies: The PRISMA-DTA Statement. JAMA 2018, 319, 388–396. [Google Scholar] [CrossRef] [PubMed]
- R-Forge: Meta-Analysis of Diagnostic Accuracy: Project Home. Available online: http://r-forge.r-project.org/projects/mada/ (accessed on 13 August 2020).


| Study | Risk of Bias | Concerns Regarding Applicability | |||||
|---|---|---|---|---|---|---|---|
| Patient Selection | Index Tests | Reference Standard | Flow and Timing | Patient Selection | Index Tests | Reference Standard | |
| O’Mahony [26] | Low | Low | Unclear | Low | Low | High | Low |
| Iguchi [27] | Unclear | Low | Low | High | Low | Low | Low |
| Uehara [28] | Low | Unclear | High | High | Low | Unclear | Low |
| Furuya [29] | Low | Low | Low | Unclear | Low | Unclear | Low |
| Fukushima [30] | High | High | Low | Low | Low | High | Low |
| Watanabe (1998) [31] | Low | Unclear | Low | Unclear | Low | Unclear | Unclear |
| Watanabe (1999) [32] | Low | Unclear | High | Low | Low | Low | Low |
| Yamaguchi [33] | High | Low | Low | Unclear | Low | Unclear | Low |
| Futakawa [34] | Low | High | High | Unclear | Low | Low | Low |
| Myung [35] | Low | High | Unclear | Low | Low | High | Unclear |
| Boadas [36] | High | Unclear | Unclear | High | Low | Unclear | Unclear |
| Ha [37] | High | Unclear | Low | High | Low | Low | High |
| Pugliese [38] | High | High | High | High | Low | High | Unclear |
| Seki [39] | Unclear | Unclear | Unclear | Unclear | Low | Unclear | Low |
| Costentin [40] | High | High | Unclear | High | Low | Low | Unclear |
| Wang [41] | High | Low | High | High | Low | Unclear | Low |
| Trumper [42] | Unclear | Low | High | High | Low | Low | Low |
| Shi [43] | Unclear | Low | Low | Unclear | Low | Low | Low |
| Takano [44] | High | High | Unclear | Low | Low | Low | Low |
| Eshleman [45] | High | Unclear | Low | Low | Low | Unclear | Low |
| Kisiel [46] | High | Low | Low | Unclear | Low | Low | Low |
| Ginesta [47] | High | Low | Unclear | Unclear | Low | Low | Low |
| Author | Published | Country | Design | Number of Patients (% PDAC Prevalence) | Patient Population | Age | Male (%) |
|---|---|---|---|---|---|---|---|
| O’Mahony [26] | 1995 | UK | * | 67 (30) | PDAC, H | * | * |
| Iguchi [27] | 1996 | Japan | * | 60 (32) | PDAC, Pc, B | * | * |
| Uehara [28] | 1996 | Japan | Ret-Co | 47 (30) | PDAC, Pc, B, H | * | * |
| Furuya [29] | 1997 | Japan | Pro-Co | 70 (8.6) | PDAC, Pc, B | * | * |
| Fukushima [30] | 1998 | Japan | Ret-Co | 29 (72) | PDAC, Pc | * | * |
| Watanabe (1998) [31] | 1998 | Japan | Ret-Co | 66 (44) | PDAC, Pc, H | * (39–83) | 55 |
| Watanabe (1999) [32] | 1999 | Japan | Ret-Co | 140 (43) | PDAC, Pc, B, H | 61 (28–84) § | * |
| Yamaguchi [33] | 1999 | Japan | Ret-Co | 46(57) | PDAC, Pc, B | * | * |
| Futakawa [34] | 2000 | Japan | Pro-Co | 52 (23) | PDAC, Pc, B, H | * | * |
| Myung [35] | 2000 | Korea | Ret-Co | 31 (39) | PDAC, Pc, H | 63 (46–77) ± | 61 |
| Boadas [36] | 2001 | Spain | Pro-Co | 90 (20) | PDAC, Pc | * | * |
| Ha [37] | 2001 | Japan | Ret-Co | 44 (43) | PDAC, Pc | * (17–81) | * |
| Pugliese [38] | 2001 | Italy | Pro-Co | 45 (76) | PDAC, Pc | 66 (44–88) § | 56 |
| Seki [39] | 2001 | Japan | Ret-Co | 36 (47) | PDAC, Pc, H | * | 72 |
| Costentin [40] | 2002 | France | Ret-Co | 57 (32) | PDAC, Pc, B | * | * |
| Wang [41] | 2002 | Germany | Pro-Co | 358 (33) | PDAC, Pc, B, H | * | * |
| Trumper [42] | 2004 | Japan | Ret-Co | 46 (46) | PDAC, Pc | * | * |
| Shi [43] | 2008 | USA | Ret-Co | 36 (75) | PDAC, Pc | * | * |
| Takano [44] | 2014 | Japan | Ret-Co | 152(26) | PDAC, Pc, B, H | 65 (35–85) § | 58 |
| Eshleman [45] | 2015 | USA | Cas-Co | 272 (11) | PDAC, B, H | 57± | 48 |
| Kisiel [46] | 2015 | USA | Cas-Co | 102 (60) | PDAC, Pc, H | 64 (49–76) § | 52 |
| Ginesta [47] | 2016 | Spain | Cas-Co | 135 (82) | PDAC, Pc, B | 68 (40–79) ± | 60 |
| Author | Modality | Site | Secretin Stimulation | Mucus Volume (mls) | DNA Extraction Method | Amplification Method | Mut-KRAS Detection Method | Codon/Mutant Tested | Gold Standard Test |
|---|---|---|---|---|---|---|---|---|---|
| O’Mahony [26] | ERCP | Bile duct | n | * | Pheno-chlor | PCR | RFLP | 12 | Histology |
| Iguchi [27] | Endoscopy | Duodenum | y | (30–40) | Pheno-chlor | PCR | RFLP, sequencing | 12 | Histology |
| Uehara [28] | ERCP | Pancreas | n | * | * | PCR | Slot-blot | 12 | * |
| Furuya [29] | Endoscopy | Duodenum | Y | 0.5 ± | Acet-chlor | PCR | RFLP | 12 | Histology |
| Fukushima [30] | ERCP | Pancreas, bile duct | n | * | Pheno-chlor | PCR | RFLP | 12 | Histology |
| Watanabe (1998) [31] | ERCP | Pancreas | n | * | * | PCR | Hybridization probe | 12 | Histology |
| Watanabe (1999) [32] | ERCP | Pancreas | n | (2–3) | Pheno-chlor, Prot-K | PCR with A-sA | 12 | Histology | |
| Yamaguchi [33] | ERCP | Pancreas | y | * | Pheno-chlor | PCR | SSCP | 12 | Histology |
| Futakawa [34] | ERCP | Pancreas | n | 0.1 | Pheno-chlor | PCR | RFLP | 12 | * |
| Myung [35] | ERCP | Pancreas | y | 0.1 | * | PCR | RFLP | 12 | Histology |
| Boadas [36] | ERCP | Pancreas | y | 4.6 ± | * | PCR | RFLP | 12 | Histology |
| Ha [37] | Endoscopy | Duodenum | y | * | Pheno-chlor | PCR with A-sA | RFLP | 12 | Histology |
| Pugliese [38] | ERCP | Pancreas | n | * | * | PCR | RFLP, sequencing | 12 | Histology |
| Seki [39] | ERCP | Pancreas | y | 2 ± | * | PCR | SSCP | 12 | * |
| Costentin [40] | ERCP | Pancreas | n | * | * | PCR | RFLP | 12 | * |
| Wang [41] | ERCP | Pancreas | n | (1–2) | * | RFLP-Targeted enrichment | RFLP, sequencing | 12 | * |
| Trumper [42] | Endoscopy | Duodenum | y | (10–15) | Pheno-chlor | PCR with A-sA | RFLP | 12 | Histology |
| Shi [43] | Surgery | Pancreas | n | * | * | PCR | qPCR-primer based | G12V, G12D, G12R | Histology |
| Takano [44] | ERCP | Pancreas | n | * | Proprietary (QiAMP®Kit) | PCR | DNA sequencing | G12D, G12R, G12V, Q61H | Histology |
| Eshleman [45] | Endoscopy | Pancreas | y | (5–10) | * | * | HRMA | * | |
| Kisiel [46] | Endoscopy | Duodenum | y | 2 ± | * | * | QuARTS | G12D | * |
| Ginesta [47] | Surgery | Pancreas | n | * | Pheno-chlor | PCR | qPCR primer-based | G12C, G12V, G12D, G12A, G12s, G12R, G13D | Histology |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Patel, N.; Petrinic, T.; Silva, M.; Soonawalla, Z.; Reddy, S.; Gordon-Weeks, A. The Diagnostic Accuracy of Mutant KRAS Detection from Pancreatic Secretions for the Diagnosis of Pancreatic Cancer: A Meta-Analysis. Cancers 2020, 12, 2353. https://doi.org/10.3390/cancers12092353
Patel N, Petrinic T, Silva M, Soonawalla Z, Reddy S, Gordon-Weeks A. The Diagnostic Accuracy of Mutant KRAS Detection from Pancreatic Secretions for the Diagnosis of Pancreatic Cancer: A Meta-Analysis. Cancers. 2020; 12(9):2353. https://doi.org/10.3390/cancers12092353
Chicago/Turabian StylePatel, Nikhil, Tatjana Petrinic, Michael Silva, Zahir Soonawalla, Srikanth Reddy, and Alex Gordon-Weeks. 2020. "The Diagnostic Accuracy of Mutant KRAS Detection from Pancreatic Secretions for the Diagnosis of Pancreatic Cancer: A Meta-Analysis" Cancers 12, no. 9: 2353. https://doi.org/10.3390/cancers12092353





