Functional Mapping before and after Low-Grade Glioma Surgery: A New Way to Decipher Various Spatiotemporal Patterns of Individual Neuroplastic Potential in Brain Tumor Patients
Abstract
:Simple Summary
Abstract
1. Introduction
2. The Concept of Neuroplasticity in a Meta-Networking Framework of Brain Processes
3. The Contribution of FNI and TMS Mapping to Investigate Individual Mechanisms of Neuroplasticity before LGG Surgery
3.1. Preoperative Structural Changes in Glioma Patients
3.2. Functional Redeployment and Correlation with Degree of Neurological Compensation
3.2.1. Preoperative Reorganization of the Motor Network
3.2.2. Preoperative Reorganization of the Language Network
3.2.3. Preoperative Reorganization of Other Networks
3.3. Preoperative Prediction of Postoperative Functional Outcomes Based on FNI/TMS
4. Functional Compensation after LGG Surgery: Postoperative and Longitudinal FNI/TMS Mapping Studies
4.1. Postoperative Reorganization of Motor Network
4.2. Postoperative Reorganization of the Language Network
5. Conclusions and Perspectives
Funding
Conflicts of Interest
Abbreviations
| fMRI | functional MRI |
| rsfMRI | resting-state functional MRI |
| FNI | functional neuroimaging |
| DTI | diffusion tensor imaging |
| MEG | magnetoencephalography |
| TMS | transcranial magnetic stimulation |
| rTMS | repetitive TMS |
| DEM | direct electrostimulation mapping |
| LGG | low-grade glioma |
| HGG | high-grade glioma |
| WM | white matter |
| FC | functional connectivity |
| ALFF | amplitude of low-frequency fluctuation |
| EOR | extent of resection |
| SPC | superior parietal cortex |
| ECN | executive control network |
| DMN | default-mode network |
| SN | salience network |
| HC | healthy control |
| SMA | supplementary motor area |
| QoL | quality of life |
| SVZ | subventricular zone |
References
- Duffau, H. Long-term outcomes after supratotal resection of diffuse low-grade gliomas: A consecutive series with 11-year follow-up. Acta Neurochir. (Wien.) 2016, 158, 51–58. [Google Scholar] [CrossRef]
- Rossi, M.; Ambrogi, F.; Gay, L.; Gallucci, M.; Conti Nibali, M.; Leonetti, A.; Puglisi, G.; Sciortino, T.; Howells, H.; Riva, M.; et al. Is supratotal resection achievable in low-grade gliomas? Feasibility, putative factors, safety, and functional outcome. J. Neurosurg. 2019, 1–14. [Google Scholar] [CrossRef] [Green Version]
- Zigiotto, L.; Annicchiarico, L.; Corsini, F.; Vitali, L.; Falchi, R.; Dalpiaz, C.; Rozzanigo, U.; Barbareschi, M.; Avesani, P.; Papagno, C.; et al. Effects of supra-total resection in neurocognitive and oncological outcome of high-grade gliomas comparing asleep and awake surgery. J. Neurooncol. 2020. [Google Scholar] [CrossRef]
- Conti Nibali, M.; Rossi, M.; Sciortino, T.; Riva, M.; Gay, L.G.; Pessina, F.; Bello, L. Preoperative surgical planning of glioma: Limitations and reliability of fMRI and DTI tractography. J. Neurosurg. Sci. 2019, 6, 127–134. [Google Scholar] [CrossRef] [PubMed]
- Krieg, S.M.; Shiban, E.; Buchmann, N.; Gempt, J.; Foerschler, A.; Meyer, B.; Ringel, F. Utility of presurgical navigated transcranial magnetic brain stimulation for the resection of tumors in eloquent motor areas. J. Neurosurg. 2012, 116, 994–1001. [Google Scholar] [CrossRef]
- Weng, H.H.; Noll, K.R.; Johnson, J.M.; Prabhu, S.S.; Tsai, Y.H.; Chang, S.W.; Huang, Y.C.; Lee, J.D.; Yang, J.T.; Yang, C.T.; et al. Accuracy of presurgical functional MR imaging for language mapping of brain tumors: A systematic review and meta-analysis. Radiology 2018, 286, 512–523. [Google Scholar] [CrossRef] [Green Version]
- Southwell, D.G.; Birk, H.S.; Han, S.J.; Li, J.; Sall, J.W.; Berger, M.S. Resection of gliomas deemed inoperable by neurosurgeons based on preoperative imaging studies. J. Neurosurg. 2018, 129, 567–575. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Maier-Hein, K.H.; Neher, P.F.; Houde, J.C.; Côté, M.A.; Garyfallidis, E.; Zhong, J.; Chamberland, M.; Yeh, F.C.; Lin, Y.C.; Ji, Q.; et al. The challenge of mapping the human connectome based on diffusion tractography. Nat. Commun. 2017, 8, 1349. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Pujol, S.; Wells, W.; Pierpaoli, C.; Brun, C.; Gee, J.; Cheng, G.; Vemuri, B.; Commowick, O.; Prima, S.; Stamm, A.; et al. The DTI Challenge: Toward standardized evaluation of diffusion tensor imaging tractography for neurosurgery. J. Neuroimaging 2015, 25, 875–882. [Google Scholar] [CrossRef] [PubMed]
- Motomura, K.; Takeuchi, H.; Nojima, I.; Aoki, K.; Chalise, L.; Iijima, K.; Wakabayashi, T.; Natsume, A. Navigated repetitive transcranial magnetic stimulation as preoperative assessment in patients with brain tumors. Sci. Rep. 2020, 10, 9044. [Google Scholar] [CrossRef]
- Azad, T.D.; Duffau, H. Limitations of functional neuroimaging for patient selection and surgical planning in glioma surgery. Neurosurg. Focus. 2020, 48, E12. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Duffau, H. Mapping the connectome in awake surgery for gliomas: An update. J. Neurosurg. Sci. 2017, 61, 612–630. [Google Scholar] [PubMed]
- Duffau, H. Stimulation mapping of white matter tracts to study brain functional connectivity. Nat. Rev. Neurol. 2015, 11, 255–265. [Google Scholar] [CrossRef]
- Duffau, H. Diffuse low-grade glioma, oncological outcome and quality of life: A surgical perspective. Curr. Opin. Oncol. 2018, 30, 383–389. [Google Scholar] [CrossRef] [PubMed]
- Duffau, H. Lessons from brain mapping in surgery for low-grade glioma: Insights into associations between tumour and brain plasticity. Lancet Neurol. 2005, 4, 476–486. [Google Scholar] [CrossRef]
- Herbet, G.; Duffau, H. Revisiting the functional anatomy of the human brain: Toward a meta-networking theory of cerebral functions. Physiol. Rev. 2020, 100, 1181–1228. [Google Scholar] [CrossRef]
- Desmurget, M.; Bonnetblanc, F.; Duffau, H. Contrasting acute and slow-growing lesions: A new door to brain plasticity. Brain 2007, 130, 898–914. [Google Scholar] [CrossRef] [Green Version]
- Duffau, H. Diffuse Low-Grade Gliomas in Adults, 2nd ed.; Duffau, H., Ed.; Springer: London, UK, 2017. [Google Scholar]
- Duffau, H. The huge plastic potential of adult brain and the role of connectomics: New insights provided by serial mappings in glioma surgery. Cortex 2014, 58, 325–337. [Google Scholar] [CrossRef]
- Duffau, H. Higher-Order Surgical Questions for Diffuse Low-Grade Gliomas: Supramaximal Resection, Neuroplasticity, and Screening. Neurosurg. Clin. N. Am. 2019, 30, 119–128. [Google Scholar] [CrossRef]
- Noorani, I.; Sanai, N. Surgical Management of Incidental Gliomas. Neurosurg. Clin. N. Am. 2017, 28, 397–406. [Google Scholar] [CrossRef]
- Duffau, H. New philosophy, clinical pearls and methods for intraoperative cognition mapping and monitoring “à la carte” in brain tumor patients. Neurosurgery 2020, in press. [Google Scholar]
- Vanacor, C.; Duffau, H. Analysis of Legal, Cultural, and Socioeconomic Parameters in Low-Grade Glioma Management: Variability Across Countries and Implications for Awake Surgery. World Neurosurg. 2018, 120, 47–53. [Google Scholar] [CrossRef] [PubMed]
- Rech, F.; Herbet, G.; Gaudeau, Y.; Mézières, S.; Moureau, J.M.; Moritz-Gasser, S.; Duffau, H. A Probabilistic Map of Negative Motor Areas of the Upper Limb and Face: A Brain Stimulation Study. Brain 2019, 142, 952–965. [Google Scholar] [CrossRef] [PubMed]
- Herbet, G.; Moritz-Gasser, S.; Duffau, H. Electrical stimulation of the dorsolateral prefrontal cortex impairs semantic cognition. Neurology 2018, 90, e1077–e1084. [Google Scholar] [CrossRef] [PubMed]
- Fernández Coello, A.; Moritz-Gasser, S.; Martino, J.; Martinoni, M.; Matsuda, R.; Duffau, H. Selection of intraoperative tasks for awake mapping based on relationships between tumor location and functional networks. J. Neurosurg. 2013, 119, 1380–1394. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Herbet, G.; Moritz-Gasser, S.; Duffau, H. Direct evidence for the contributive role of the right inferior fronto-occipital fasciculus in non-verbal semantic cognition. Brain Struct. Funct. 2017, 222, 1597–1610. [Google Scholar] [CrossRef]
- Moritz-Gasser, S.; Herbet, G.; Duffau, H. Mapping the connectivity underlying multimodal (verbal and non-verbal) semantic processing: A brain electrostimulation study. Neuropsychologia 2013, 51, 1814–1822. [Google Scholar] [CrossRef]
- Herbet, G.; Maheu, M.; Costi, E.; Lafargue, G.; Duffau, H. Mapping neuroplastic potential in brain-damaged patients. Brain 2016, 139, 829–844. [Google Scholar] [CrossRef]
- Sarubbo, S.; Tate, M.; De Benedictis, A.; Merler, S.; Moritz-Gasser, S.; Herbet, G.; Duffau, H. Mapping critical cortical hubs and white matter pathways by direct electrical stimulation: An original functional atlas of the human brain. Neuroimage 2020, 205, 116237. [Google Scholar] [CrossRef]
- Almairac, F.; Duffau, H.; Herbet, G. Contralesional macrostructural plasticity of the insular cortex in patients with glioma: A VBM study. Neurology 2018, 91, e1902–e1908. [Google Scholar] [CrossRef]
- Celeghin, A.; Diano, M.; de Gelder, B.; Weiskrantz, L.; Marzi, C.A.; Tamietto, M. Intact hemisphere and corpus callosum compensate for visuomotor functions after early visual cortex damage. Proc. Natl. Acad. Sci. USA 2017, 114, E10475–E10483. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Liu, Y.; Hu, G.; Yu, Y.; Jiang, Z.; Yang, K.; Hu, X.; Li, Z.; Liu, D.; Zou, Y.; Liu, H.; et al. Structural and Functional Reorganization Within Cognitive Control Network Associated With Protection of Executive Function in Patients With Unilateral Frontal Gliomas. Front. Oncol. 2020, 10, 794. [Google Scholar] [CrossRef] [PubMed]
- Zhang, N.; Xia, M.; Qiu, T.; Wang, X.; Lin, C.P.; Guo, Q.; Lu, J.; Wu, Q.; Zhuang, D.; Yu, Z.; et al. Reorganization of cerebro-cerebellar circuit in patients with left hemispheric gliomas involving language network: A combined structural and resting-state functional MRI study. Hum. Brain Mapp. 2018, 39, 4802–4819. [Google Scholar] [CrossRef] [PubMed]
- Ceko, M.; Seminowicz, D.A.; Bushnell, M.C.; Olausson, H.W. Anatomical and functional enhancements of the insula after loss of large primary somatosensory fibers. Cereb. Cortex. 2013, 23, 2017–2024. [Google Scholar] [CrossRef]
- May, A.; Hajak, G.; Ganssbauer, S.; Steffens, T.; Langguth, B.; Kleinjung, T.; Eichhammer, P. Structural brain alterations following 5 days of intervention: Dynamic aspects of neuroplasticity. Cereb. Cortex. 2007, 17, 205–210. [Google Scholar] [CrossRef] [Green Version]
- Liu, D.; Liu, Y.; Hu, X.; Hu, G.; Yang, K.; Xiao, C.; Hu, J.; Li, Z.; Zou, Y.; Chen, J.; et al. Alterations of white matter integrity associated with cognitive deficits in patients with glioma. Brain Behav. 2020, 10, e01639. [Google Scholar] [CrossRef]
- Almairac, F.; Herbet, G.; Moritz-Gasser, S.; de Champfleur, N.M.; Duffau, H. The left inferior fronto-occipital fasciculus subserves language semantics: A multilevel lesion study. Brain Struct. Funct. 2015, 220, 1983–1995. [Google Scholar] [CrossRef]
- Herbet, G.; Lafargue, G.; Bonnetblanc, F.; Moritz-Gasser, S.; Menjot de Champfleur, N.; Duffau, H. Inferring a dual-stream model of mentalizing from associative white matter fibres disconnection. Brain 2014, 137, 944–959. [Google Scholar] [CrossRef]
- Duffau, H. Does post-lesional subcortical plasticity exist in the human brain? Neurosci. Res. 2009, 65, 131–135. [Google Scholar] [CrossRef]
- Fox, M.E.; King, Z.T. Functional connectivity in adult brain tumor patients: A systematic review. Brain Connect. 2018, 8, 381–397. [Google Scholar] [CrossRef]
- Biswal, B.; Yetkin, F.Z.; Haughton, V.M.; Hyde, J.S. Functional connectivity in the motor cortex of resting human brain using echo-planar MRI. Magn. Reson. Med. 1995, 34, 537–541. [Google Scholar] [CrossRef] [PubMed]
- Böttger, J.; Margulies, D.S.; Horn, P.; Thomale, U.W.; Podlipsky, I.; Shapira-Lichter, I.; Chaudhry, S.J.; Szkudlarek, C.; Mueller, K.; Lohmann, G.; et al. A software tool for interactive exploration of intrinsic functional connectivity opens new perspectives for brain surgery. Acta Neurochir. (Wien.) 2011, 153, 1561–1572. [Google Scholar] [CrossRef]
- Derks, J.; Dirkson, A.R.; de Witt Hamer, P.C.; van Geest, Q.; Hulst, H.E.; Barkhof, F.; Pouwels, P.J.; Geurts, J.J.; Reijneveld, J.C.; Douw, L. Connectomic profile and clinical phenotype in newly diagnosed glioma patients. Neuroimage Clin. 2017, 14, 87–96. [Google Scholar] [CrossRef]
- Greicius, M.D.; Krasnow, B.; Reiss, A.L.; Menon, V. Functional connectivity in the resting brain: A network analysis of the default mode hypothesis. Proc. Natl. Acad. Sci. USA 2003, 100, 253–258. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Cirillo, S.; Caulo, M.; Pieri, V.; Falini, A.; Castellano, A. Role of Functional Imaging Techniques to Assess Motor and Language Cortical Plasticity in Glioma Patients: A Systematic Review. Neural Plast. 2019, 2019, 4056436. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Barz, A.; Noack, A.; Baumgarten, P.; Seifert, V.; Forster, M.T. Motor cortex reorganization in patients with glioma assessed by repeated navigated transcranial magnetic stimulation-a longitudinal study. World Neurosurg. 2018, 112, e442–e453. [Google Scholar] [CrossRef]
- Conway, N.; Wildschuetz, N.; Moser, T.; Bulubas, L.; Sollmann, N.; Tanigawa, N.; Meyer, B.; Krieg, S.M. Cortical plasticity of motor-eloquent areas measured by navigated transcranial magnetic stimulation in patients with glioma. J. Neurosurg. 2017, 127, 981–991. [Google Scholar] [CrossRef]
- Viganò, L.; Fornia, L.; Rossi, M.; Howells, H.; Leonetti, A.; Puglisi, G.; Conti Nibali, M.; Bellacicca, A.; Grimaldi, M.; Bello, L.; et al. Anatomo-functional characterisation of the human “hand-knob”: A direct electrophysiological study. Cortex 2019, 113, 239–254. [Google Scholar] [CrossRef]
- Ius, T.; Angelini, E.; Thiebaut de Schotten, M.; Mandonnet, E.; Duffau, H. Evidence for potentials and limitations of brain plasticity using an atlas of functional resectability of WHO grade II gliomas: Towards a “minimal common brain”. Neuroimage 2011, 56, 992–1000. [Google Scholar] [CrossRef]
- Otten, M.L.; Mikell, C.B.; Youngerman, B.E.; Liston, C.; Sisti, M.B.; Bruce, J.N.; Small, S.A.; McKhann, G.M., 2nd. Motor deficits correlate with resting state motor network connectivity in patients with brain tumours. Brain 2012, 135, 1017–1026. [Google Scholar] [CrossRef] [PubMed]
- Majos, A.; Bryszewski, B.; Kośla, K.N.; Pfaifer, L.; Jaskólski, D.J.; Stefańczyk, L. Process of the functional reorganization of the cortical centers for movement in GBM patients: fMRI study. Clin. Neuroradiol. 2017, 27, 71–79. [Google Scholar] [CrossRef] [PubMed]
- Herbet, G.; Lemaitre, A.L.; Moritz-Gasser, S.; Cochereau, J.; Duffau, H. The antero-dorsal precuneal cortex supports specific aspects of bodily awareness. Brain 2019, 142, 2207–2214. [Google Scholar] [CrossRef] [PubMed]
- Howells, H.; Puglisi, G.; Leonetti, A.; Vigano, L.; Fornia, L.; Simone, L.; Forkel, S.J.; Rossi, M.; Riva, M.; Cerri, G.; et al. The role of left fronto-parietal tracts in hand selection: Evidence from neurosurgery. Cortex 2020, 128, 297–311. [Google Scholar] [CrossRef] [PubMed]
- Stefaniak, J.D.; Halai, A.D.; Lambon Ralph, M.A. The neural and neurocomputational bases of recovery from post-stroke aphasia. Nat. Rev. Neurol. 2020, 16, 43–55. [Google Scholar] [CrossRef] [PubMed]
- Murase, N.; Duque, J.; Mazzocchio, R.; Cohen, L.G. Influence of interhemispheric interactions on motor function in chronic stroke. Ann. Neurol. 2004, 55, 400–409. [Google Scholar] [CrossRef] [PubMed]
- Ferbert, A.; Priori, A.; Rothwell, J.C.; Day, B.L.; Colebatch, J.G.; Marsden, C.D. Interhemispheric inhibition of the human motor cortex. J. Physiol. (Lond.) 1992, 453, 525–546. [Google Scholar] [CrossRef]
- Fandino, J.; Kollias, S.S.; Wieser, H.G.; Valavanis, A.; Yonekawa, Y. Intraoperative validation of functional magnetic resonance imaging and cortical reorganization patterns in patients with brain tumors involving the primary motor cortex. J. Neurosurg. 1999, 91, 238–250. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Mallela, A.N.; Peck, K.K.; Petrovich-Brennan, N.M.; Zhang, Z.; Lou, W.; Holodny, A.I. Altered Resting-State Functional Connectivity in the Hand Motor Network in Glioma Patients. Brain Connect. 2016, 6, 587–595. [Google Scholar] [CrossRef]
- Niu, C.; Zhang, M.; Min, Z.; Rana, N.; Zhang, Q.; Liu, X.; Li, M.; Lin, P. Motor network plasticity and low-frequency oscillations abnormalities in patients with brain gliomas: A functional MRI study. PLoS ONE 2014, 9, e96850. [Google Scholar] [CrossRef]
- Agarwal, S.; Lu, H.; Pillai, J.J. Value of Frequency Domain Resting-State Functional Magnetic Resonance Imaging Metrics Amplitude of Low-Frequency Fluctuation and Fractional Amplitude of Low-Frequency Fluctuation in the Assessment of Brain Tumor-Induced Neurovascular Uncoupling. Brain Connect. 2017, 7, 382–389. [Google Scholar] [CrossRef]
- Marrelec, G.; Bellec, P.; Krainik, A.; Duffau, H.; Pélégrini-Issac, M.; Lehéricy, S.; Benali, H.; Doyon, J. Regions, systems, and the brain: Hierarchical measures of functional integration in fMRI. Med. Image Anal. 2008, 12, 484–496. [Google Scholar] [CrossRef] [PubMed]
- Zhang, D.; Johnston, J.M.; Fox, M.D.; Leuthardt, E.C.; Grubb, R.L.; Chicoine, M.R.; Smyth, M.D.; Snyder, A.Z.; Raichle, M.E.; Shimony, J.S. Preoperative sensorimotor mapping in brain tumor patients using spontaneous fluctuations in neuronal activity imaged with functional magnetic resonance imaging: Initial experience. Neurosurgery 2009, 65, 226–236. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kinno, R.; Ohta, S.; Muragaki, Y.; Maruyama, T.; Sakai, K.L. Differential reorganization of three syntax-related networks induced by a left frontal glioma. Brain 2014, 137, 1193–1212. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wang, L.; Chen, D.; Yang, X.; Olson, J.J.; Gopinath, K.; Fan, T.; Mao, H. Group independent component analysis and functional MRI examination of changes in language areas associated with brain tumors at different locations. PLoS ONE 2013, 8, e59657. [Google Scholar] [CrossRef] [PubMed]
- Li, Q.; Dong, J.W.; Del Ferraro, G.; Petrovich Brennan, N.; Peck, K.K.; Tabar, V.; Makse, H.A.; Holodny, A.I. Functional Translocation of Broca’s Area in a Low-Grade Left Frontal Glioma: Graph Theory Reveals the Novel, Adaptive Network Connectivity. Front. Neurol. 2019, 10, 702. [Google Scholar] [CrossRef]
- Kośla, K.; Pfajfer, L.; Bryszewski, B.; Jaskólski, D.; Stefańczyk, L.; Majos, A. Functional rearrangement of language areas in patients with tumors of the central nervous system using functional magnetic resonance imaging. Pol. J. Radiol. 2012, 77, 39–45. [Google Scholar] [CrossRef]
- Briganti, S.; Sestieri, C.; Mattei, P.A.; Esposito, R.; Galzio, R.J.; Tartaro, A.; Romani, G.L.; Caulo, M. Reorganization of functional connectivity of the language network in patients with brain gliomas. Am. J. Neuroradiol. 2012, 33, 1983–1990. [Google Scholar] [CrossRef] [Green Version]
- Raffa, G.; Quattropani, M.C.; Scibilia, A.; Conti, A.; Angileri, F.F.; Esposito, F.; Sindorio, C.; Cardali, S.M.; Germanò, A.; Tomasello, F. Surgery of language-eloquent tumors in patients not eligible for awake surgery: The impact of a protocol based on navigated transcranial magnetic stimulation on presurgical planning and language outcome, with evidence of tumor-induced intra-hemispheric plasticity. Clin. Neurol. Neurosurg. 2018, 168, 127–139. [Google Scholar]
- Thiel, A.; Habedank, B.; Winhuisen, L.; Herholz, K.; Kessler, J.; Haupt, W.F.; Heiss, W.D. Essential language function of the right hemisphere in brain tumor patients. Ann. Neurol. 2005, 57, 128–131. [Google Scholar] [CrossRef]
- Kinno, R.; Ohta, S.; Muragaki, Y.; Maruyama, T.; Sakai, K.L. Left frontal glioma induces functional connectivity changes in syntax-related networks. Springerplus 2015, 4, 317. [Google Scholar] [CrossRef] [Green Version]
- Yuan, B.; Zhang, N.; Yan, J.; Cheng, J.; Lu, J.; Wu, J. Resting-state functional connectivity predicts individual language impairment of patients with left hemispheric gliomas involving language network. Neuroimage Clin. 2019, 24, 102023. [Google Scholar] [CrossRef] [PubMed]
- Liu, D.; Chen, J.; Hu, X.; Hu, G.; Liu, Y.; Yang, K.; Xiao, C.; Zou, Y.; Liu, H. Contralesional homotopic functional plasticity in patients with temporal glioma. J. Neurosurg. 2020, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Alvarez, J.A.; Emory, E. Executive function and the frontal lobes: A meta-analytic review. Neuropsychol. Rev. 2006, 16, 17–42. [Google Scholar] [CrossRef]
- Lang, S.; Gaxiola-Valdez, I.; Opoku-Darko, M.; Partlo, L.A.; Goodyear, B.G.; Kelly, J.J.P.; Federico, P. Functional Connectivity in Frontoparietal Network: Indicator of Preoperative Cognitive Function and Cognitive Outcome Following Surgery in Patients with Glioma. World Neurosurg. 2017, 105, 913–922. [Google Scholar] [CrossRef] [PubMed]
- Maesawa, S.; Bagarinao, E.; Fujii, M.; Futamura, M.; Motomura, K.; Watanabe, H.; Mori, D.; Sobue, G.; Wakabayashi, T. Evaluation of resting state networks in patients with gliomas: Connectivity changes in the unaffected side and its relation to cognitive function. PLoS ONE 2015, 10, e0118072. [Google Scholar] [CrossRef] [Green Version]
- Liu, D.; Hu, X.; Liu, Y.; Yang, K.; Xiao, C.; Hu, J.; Li, Z.; Zou, Y.; Chen, J.; Liu, H. Potential Intra- or Cross-Network Functional Reorganization of the Triple Unifying Networks in Patients with Frontal Glioma. World Neurosurg. 2019, 128, e732–e743. [Google Scholar] [CrossRef]
- Alves, P.N.; Foulon, C.; Karolis, V.; Bzdok, D.; Margulies, D.S.; Volle, E.; Thiebaut de Schotten, M. An improved neuroanatomical model of the default-mode network reconciles previous neuroimaging and neuropathological findings. Commun Biol. 2019, 2, 370. [Google Scholar] [CrossRef]
- Ghumman, S.; Fortin, D.; Noel-Lamy, M.; Cunnane, S.C.; Whittingstall, K. Exploratory study of the effect of brain tumors on the default mode network. J. Neurooncol. 2016, 128, 437–444. [Google Scholar] [CrossRef]
- Hart, M.G.; Price, S.J.; Suckling, J. Functional connectivity networks for preoperative brain mapping in neurosurgery. J. Neurosurg. 2017, 126, 1941–1950. [Google Scholar] [CrossRef] [PubMed]
- Zhang, H.; Shi, Y.; Yao, C.; Tang, W.; Yao, D.; Zhang, C.; Wang, M.; Wu, J.; Song, Z. Alteration of the Intra- and Cross- Hemisphere Posterior Default Mode Network in Frontal Lobe Glioma. Sci. Rep. 2016, 6, 26972. [Google Scholar] [CrossRef]
- Harris, R.J.; Bookheimer, S.Y.; Cloughesy, T.F.; Kim, H.J.; Pope, W.B.; Lai, A.; Nghiemphu, P.L.; Liau, L.M.; Ellingson, B.M. Altered functional connectivity of the default mode network in diffuse gliomas measured with pseudo-resting state fMRI. J. Neurooncol. 2014, 116, 373–379. [Google Scholar] [CrossRef] [PubMed]
- Esposito, R.; Mattei, P.A.; Briganti, C.; Romani, G.L.; Tartaro, A.; Caulo, M. Modifications of default-mode network connectivity in patients with cerebral glioma. PLoS ONE 2012, 7, e40231. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Xu, H.; Ding, S.; Hu, X.; Yang, K.; Xiao, C.; Zou, Y.; Chen, Y.; Tao, L.; Liu, H.; Qian, Z. Reduced efficiency of functional brain network underlying intellectual decline in patients with low-grade glioma. Neurosci. Lett. 2013, 543, 27–31. [Google Scholar] [CrossRef] [PubMed]
- Huang, Q.; Zhang, R.; Hu, X.; Ding, S.; Qian, J.; Lei, T.; Cao, X.; Tao, L.; Qian, Z.; Liu, H. Disturbed small-world networks and neurocognitive function in frontal lobe low-grade glioma patients. PLoS ONE 2014, 9, e94095. [Google Scholar] [CrossRef] [PubMed]
- Bosma, I.; Stam, C.J.; Douw, L.; Bartolomei, F.; Heimans, J.J.; van Dijk, B.W.; Postma, T.J.; Klein, M.; Reijneveld, J.C. The influence of low-grade glioma on resting state oscillatory brain activity: A magnetoencephalography study. J. Neurooncol. 2008, 88, 77–85. [Google Scholar] [CrossRef] [Green Version]
- Bosma, I.; Douw, L.; Bartolomei, F.; Heimans, J.J.; van Dijk, B.W.; Postma, T.J.; Stam, C.J.; Reijneveld, J.C.; Klein, M. Synchronized brain activity and neurocognitive function in patients with low-grade glioma: A magnetoencephalography study. Neuro Oncol. 2008, 10, 734–744. [Google Scholar] [CrossRef]
- Menon, V.; Uddin, L.Q. Saliency, switching, attention and control: A network model of insula function. Brain Struct. Funct. 2010, 214, 655–667. [Google Scholar] [CrossRef] [Green Version]
- Heiss, W.D.; Thiel, A.; Kessler, J.; Herholz, K. Disturbance and recovery of language function: Correlates in PET activation studies. Neuroimage 2003, 20 (Suppl. 1), S42–S49. [Google Scholar] [CrossRef]
- Park, J.E.; Kim, H.S.; Kim, S.J.; Kim, J.H.; Shim, W.H. Alteration of long-distance functional connectivity and network topology in patients with supratentorial gliomas. Neuroradiology 2016, 58, 311–320. [Google Scholar] [CrossRef]
- Krainik, A.; Lehéricy, S.; Duffau, H.; Vlaicu, M.; Poupon, F.; Capelle, L.; Cornu, P.; Clemenceau, S.; Sahel, M.; Valery, C.A.; et al. Role of the supplementary motor area in motor deficit following medial frontal lobe surgery. Neurology 2001, 57, 871–878. [Google Scholar] [CrossRef] [Green Version]
- Krainik, A.; Lehéricy, S.; Duffau, H.; Capelle, L.; Chainay, H.; Cornu, P.; Cohen, L.; Boch, A.L.; Mangin, J.F.; Le Bihan, D.; et al. Postoperative speech disorder after medial frontal surgery: Role of the supplementary motor area. Neurology 2003, 60, 587–594. [Google Scholar] [CrossRef]
- Tarapore, P.E.; Martino, J.; Guggisberg, A.G.; Owen, J.; Honma, S.M.; Findlay, A.; Berger, M.S.; Kirsch, H.E.; Nagarajan, S.S. Magnetoencephalographic imaging of resting-state functional connectivity predicts postsurgical neurological outcome in brain gliomas. Neurosurgery 2012, 71, 1012–1022. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lee, A.T.; Faltermeier, C.; Morshed, R.A.; Young, J.S.; Kakaizada, S.; Valdivia, C.; Findlay, A.M.; Tarapore, P.E.; Nagarajan, S.S.; Hervey-Jumper, S.L.; et al. The impact of high functional connectivity network hub resection on language task performance in adult low- and high-grade glioma. J. Neurosurg. 2020, 1–11. [Google Scholar] [CrossRef]
- Caverzasi, E.; Hervey-Jumper, S.L.; Jordan, K.M.; Lobach, I.V.; Li, J.; Panara, V.; Racine, C.A.; Sankaranarayanan, V.; Amirbekian, B.; Papinutto, N.; et al. Identifying preoperative language tracts and predicting postoperative functional recovery using HARDI q-ball fiber tractography in patients with gliomas. J. Neurosurg. 2016, 125, 33–45. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Herbet, G.; Lafargue, G.; Moritz-Gasser, S.; Menjot de Champfleur, N.; Costi, E.; Bonnetblanc, F.; Duffau, H. A disconnection account of subjective empathy impairments in diffuse low-grade glioma patients. Neuropsychologia 2015, 70, 165–176. [Google Scholar] [CrossRef]
- Nakajima, R.; Yordanova, Y.N.; Duffau, H.; Herbet, G. Neuropsychological evidence for the crucial role of the right arcuate fasciculus in the face-based mentalizing network: A disconnection analysis. Neuropsychologia 2018, 115, 179–187. [Google Scholar] [CrossRef]
- Mandonnet, E.; Jbabdi, S.; Taillandier, L.; Galanaud, D.; Benali, H.; Capelle, L.; Duffau, H. Preoperative estimation of residual volume for WHO grade II glioma resected with intraoperative functional mapping. Neuro Oncol. 2007, 9, 63–69. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- van Dellen, E.; Douw, L.; Hillebrand, A.; Ris-Hilgersom, I.H.; Schoonheim, M.M.; Baayen, J.C.; De Witt Hamer, P.C.; Velis, D.N.; Klein, M.; Heimans, J.J.; et al. MEG network differences between low- and high-grade glioma related to epilepsy and cognition. PLoS ONE 2012, 7, e50122. [Google Scholar] [CrossRef]
- Lavrador, J.P.; Gioti, I.; Hoppe, S.; Jung, J.; Patel, S.; Gullan, R.; Ashkan, K.; Bhangoo, R.; Vergani, F. Altered Motor Excitability in Patients With Diffuse Gliomas Involving Motor Eloquent Areas: The Impact of Tumor Grading. Neurosurgery 2020, nyaa354. [Google Scholar] [CrossRef]
- Vergani, F.; Martino, J.; Gozé, C.; Rigau, V.; Duffau, H. World Health Organization grade II gliomas and subventricular zone: Anatomic, genetic and clinical considerations. Neurosurgery 2011, 68, 1293–1299. [Google Scholar] [CrossRef]
- Rofes, A.; Mandonnet, E.; Godden, J.; Baron, M.H.; Colle, H.; Darlix, A.; de Aguiar, V.; Duffau, H.; Herbet, G.; Klein, M.; et al. Survey on current cognitive practices within the European Low-Grade Glioma Network: Towards a European assessment protocol. Acta Neurochir. (Wien.) 2017, 159, 1167–1178. [Google Scholar] [CrossRef] [PubMed]
- Bulubas, L.; Sardesh, N.; Traut, T.; Findlay, A.; Mizuiri, D.; Honma, S.M.; Krieg, S.M.; Berger, M.S.; Nagarajan, S.S.; Tarapore, P.E. Motor Cortical Network Plasticity in Patients with Recurrent Brain Tumors. Front. Hum. Neurosci. 2020, 14, 118. [Google Scholar] [CrossRef] [PubMed]
- Krainik, A.; Duffau, H.; Capelle, L.; Cornu, P.; Boch, A.L.; Mangin, J.F.; Le Bihan, D.; Marsault, C.; Chiras, J.; Lehéricy, S. Role of the healthy hemisphere in recovery after resection of the supplementary motor area. Neurology 2004, 62, 1323–1332. [Google Scholar] [CrossRef] [Green Version]
- Bryszewski, B.; Tybor, K.; Ormezowska, E.A.; Jaskólski, D.J.; Majos, A. Rearrangement of motor centers and its relationship to the neurological status of low-grade glioma examined on pre- and postoperative fMRI. Clin. Neurol. Neurosurg. 2013, 115, 2464–2470. [Google Scholar] [CrossRef] [PubMed]
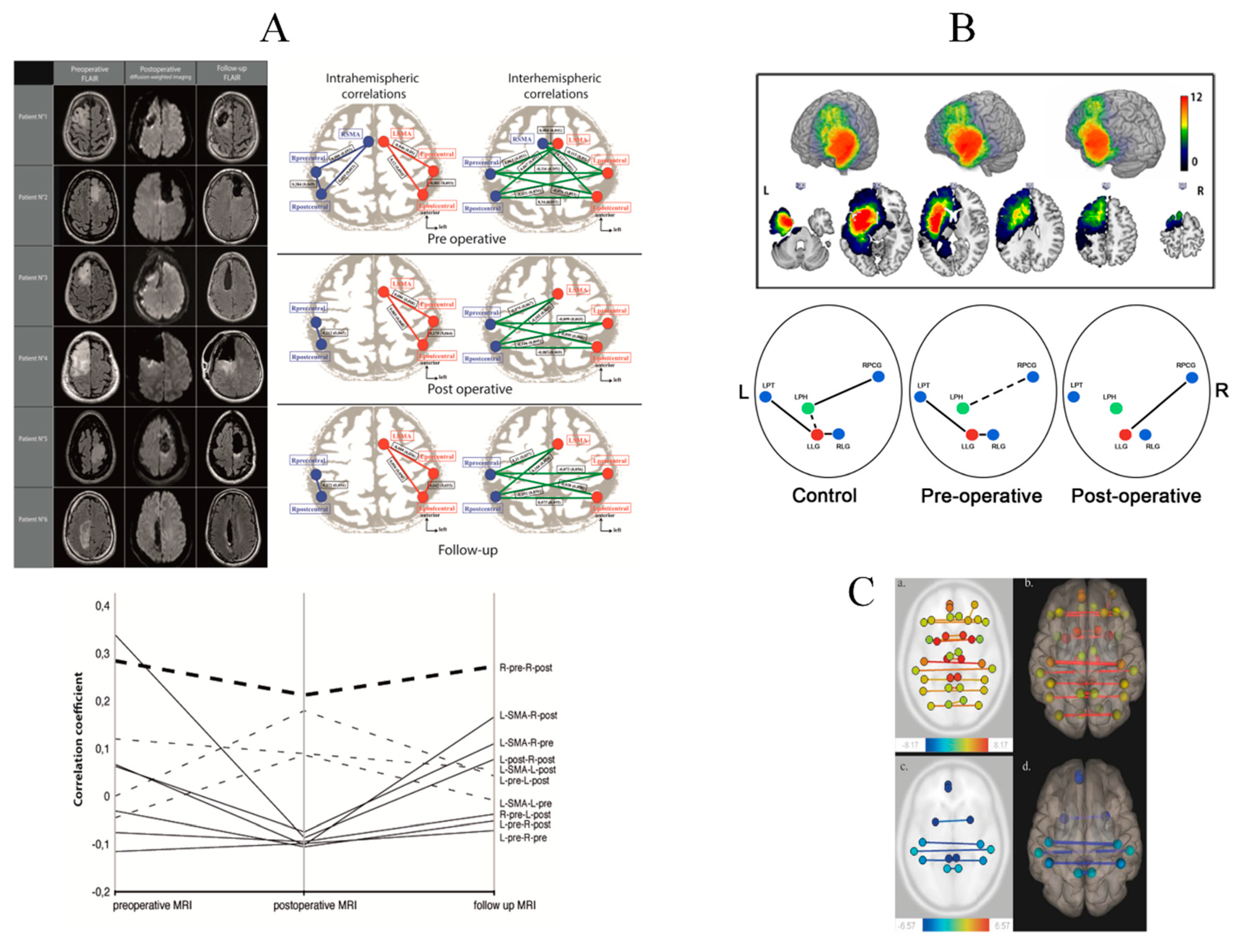
- Vassal, M.; Charroud, C.; Deverdun, J.; Le Bars, E.; Molino, F.; Bonnetblanc, F.; Boyer, A.; Dutta, A.; Herbet, G.; Moritz-Gasser, S.; et al. Recovery of functional connectivity of the sensorimotor network after surgery for diffuse low-grade gliomas involving the supplementary motor area. J. Neurosurg. 2017, 126, 1181–1190. [Google Scholar] [CrossRef] [Green Version]
- Deverdun, J.; van Dokkum, L.E.H.; Le Bars, E.; Herbet, G.; Mura, T.; D’agata, B.; Picot, M.C.; Menjot, N.; Molino, F.; Duffau, H.; et al. Language reorganization after resection of low-grade gliomas: An fMRI task based connectivity study. Brain Imaging Behav. 2019. [Google Scholar] [CrossRef]
- Coget, A.; Deverdun, J.; Bonafé, A.; van Dokkum, L.; Duffau, H.; Molino, F.; Le Bars, E.; de Champfleur, N.M. Transient immediate postoperative homotopic functional disconnectivity in low-grade glioma patients. Neuroimage Clin. 2018, 18, 656–662. [Google Scholar] [CrossRef]
- Boyer, A.; Deverdun, J.; Duffau, H.; Le Bars, E.; Molino, F.; Menjot de Champfleur, N.; Bonnetblanc, F. Longitudinal changes in cerebellar and thalamic spontaneous neuronal activity after wide-awake surgery of brain tumors: A resting-state fMRI study. Cerebellum 2016, 15, 451–465. [Google Scholar] [CrossRef]
- Kristo, G.; Raemaekers, M.; Rutten, G.J.; de Gelder, B.; Ramsey, N.F. Inter-hemispheric language functional reorganization in low-grade glioma patients after tumour surgery. Cortex 2015, 64, 235–248. [Google Scholar] [CrossRef]
- Avramescu-Murphy, M.; Hattingen, E.; Forster, M.T.; Oszvald, A.; Anti, S.; Frisch, S.; Russ, M.O.; Jurcoane, A. Post-Surgical Language Reorganization Occurs in Tumors of the Dominant and Non-Dominant Hemisphere. Clin. Neuroradiol. 2017, 27, 299–309. [Google Scholar] [CrossRef]
- Lizarazu, M.; Gil-Robles, S.; Pomposo, I.; Nara, S.; Amoruso, L.; Quiñones, I.; Carreiras, M. Spatiotemporal dynamics of postoperative functional plasticity in patients with brain tumors in language areas. Brain Lang. 2020, 202, 104741. [Google Scholar] [CrossRef] [PubMed]
- Gębska-Kośla, K.; Bryszewski, B.; Jaskólski, D.J.; Fortuniak, J.; Niewodniczy, M.; Stefańczyk, L.; Majos, A. Reorganization of language centers in patients with brain tumors located in eloquent speech areas—A pre- and postoperative preliminary fMRI study. Neurol. Neurochir. Pol. 2017, 51, 403–410. [Google Scholar] [CrossRef] [PubMed]
- Kośla, K.; Bryszewski, B.; Jaskólski, D.; Błasiak-Kołacińska, N.; Stefańczyk, L.; Majos, A. Reorganization of Language Areas in Patient with a Frontal Lobe Low Grade Glioma-fMRI Case Study. Pol. J. Radiol. 2015, 80, 290–295. [Google Scholar] [PubMed] [Green Version]
- Sarubbo, S.; Le Bars, E.; Moritz-Gasser, S.; Duffau, H. Complete recovery after surgical resection of left Wernicke’s area in awake patient: A brain stimulation and functional MRI study. Neurosurg. Rev. 2012, 35, 287–292. [Google Scholar] [CrossRef]
- van Dokkum, L.E.H.; Moritz Gasser, S.; Deverdun, J.; Herbet, G.; Mura, T.; D’Agata, B.; Picot, M.C.; Menjot de Champfleur, N.; Duffau, H.; Molino, F.; et al. Resting state network plasticity related to picture naming in low-grade glioma patients before and after resection. Neuroimage Clin. 2019, 24, 102010. [Google Scholar] [CrossRef]
- Herbet, G.; Moritz-Gasser, S.; Lemaitre, A.L.; Almairac, F.; Duffau, H. Functional compensation of the left inferior longitudinal fasciculus for picture naming. Cogn. Neuropsychol. 2019, 36, 140–157. [Google Scholar] [CrossRef]
- Mandonnet, E.; Cerliani, L.; Siuda-Krzywicka, K.; Poisson, I.; Zhi, N.; Volle, E.; de Schotten, M.T. A network-level approach of cognitive flexibility impairment after surgery of a right temporo-parietal glioma. Neurochirurgie 2017, 63, 308–313. [Google Scholar] [CrossRef]
- Cochereau, J.; Lemaitre, A.L.; Wager, M.; Moritz-Gasser, S.; Duffau, H.; Herbet, G. Network-behavior mapping of lasting executive impairments after low-grade glioma surgery. Brain Struct. Funct. in press. 2020. [CrossRef]
- Ding, Z.; Huang, Y.; Bailey, S.K.; Gao, Y.; Cutting, L.E.; Rogers, B.P.; Newton, A.T.; Gore, J.C. Detection of synchronous brain activity in white matter tracts at rest and under functional loading. Proc. Natl. Acad. Sci. USA 2018, 115, 595–600. [Google Scholar] [CrossRef] [Green Version]
- Mosca, C.; Zoubrinetzy, R.; Baciu, M.; Aguilar, L.; Minotti, L.; Kahane, P.; Perrone-Bertolotti, M. Rehabilitation of verbal memory by means of preserved nonverbal memory abilities after epilepsy surgery. Epilepsy Behav. Case Rep. 2014, 2, 167–173. [Google Scholar] [CrossRef]
- Hebb, D.O. The Organization of Behavior: A Neuropsychological Theory; Wiley: New York, NY, USA, 1949. [Google Scholar]
- Turrigiano, G.G.; Nelson, S.B. Homeostatic plasticity in the developing nervous system. Nat. Rev. Neurosci. 2004, 5, 97–107. [Google Scholar] [CrossRef] [PubMed]
- Yger, P.; Gilson, M. Models of Metaplasticity: A Review of Concepts. Front. Comput. Neurosci. 2015, 9, 138. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Duffau, H. Why brain radiation therapy should take account of the individual structural and functional connectivity: Toward an irradiation “à la carte”. Crit. Rev. Oncol. Hematol. 2020, 154, 103073. [Google Scholar] [CrossRef] [PubMed]
- Robles, S.G.; Gatignol, P.; Lehéricy, S.; Duffau, H. Longterm brain plasticity allowing a multistage surgical approach to World Health Organization grade II gliomas in eloquent areas. J. Neurosurg. 2008, 109, 615–624. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Duffau, H.; Denvil, D.; Capelle, L. Long term reshaping of language, sensory, and motor maps after glioma resection: A new parameter to integrate in the surgical strategy. J. Neurol. Neurosurg. Psychiatry 2002, 72, 511–516. [Google Scholar] [PubMed]
- Southwell, D.G.; Hervey-Jumper, S.L.; Perry, D.W.; Berger, M.S. Intraoperative mapping during repeat awake craniotomy reveals the functional plasticity of adult cortex. J. Neurosurg. 2016, 124, 1460–1469. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Picart, T.; Herbet, G.; Moritz-Gasser, S.; Duffau, H. Iterative surgical resections of diffuse glioma with awake mapping: How to deal with cortical plasticity and connectomal constraints? Neurosurgery 2019, 85, 105–116. [Google Scholar] [CrossRef] [PubMed]
- Voets, N.L.; Parker Jones, O.; Mars, R.B.; Adcock, J.E.; Stacey, R.; Apostolopoulos, V.; Plaha, P. Characterising neural plasticity at the single patient level using connectivity fingerprints. Neuroimage Clin. 2019, 24, 101952. [Google Scholar] [CrossRef]
- Blonski, M.; Taillandier, L.; Herbet, G.; Maldonado, I.L.; Beauchesne, P.; Fabbro, M.; Campello, C.; Gozé, C.; Rigau, V.; Moritz-Gasser, S.; et al. Combination of neoadjuvant chemotherapy followed by surgical resection as a new strategy for WHO grade II gliomas: A study of cognitive status and quality of life. J. Neurooncol. 2012, 106, 353–366. [Google Scholar] [CrossRef]
- Blonski, M.; Pallud, J.; Gozé, C.; Mandonnet, E.; Rigau, V.; Bauchet, L.; Fabbro, M.; Beauchesne, P.; Baron, M.H.; Fontaine, D.; et al. Neoadjuvant chemotherapy may optimize the extent of resection of World Health Organization grade II gliomas: A case series of 17 patients. J. Neurooncol. 2013, 113, 267–275. [Google Scholar] [CrossRef]
- Keidel, J.L.; Welbourne, S.R.; Lambon Ralph, M.A. Solving the paradox of the equipotential and modular brain: A neurocomputational model of stroke vs. slow-growing glioma. Neuropsychologia 2010, 48, 1716–1724. [Google Scholar] [CrossRef] [PubMed]

© 2020 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Duffau, H. Functional Mapping before and after Low-Grade Glioma Surgery: A New Way to Decipher Various Spatiotemporal Patterns of Individual Neuroplastic Potential in Brain Tumor Patients. Cancers 2020, 12, 2611. https://doi.org/10.3390/cancers12092611
Duffau H. Functional Mapping before and after Low-Grade Glioma Surgery: A New Way to Decipher Various Spatiotemporal Patterns of Individual Neuroplastic Potential in Brain Tumor Patients. Cancers. 2020; 12(9):2611. https://doi.org/10.3390/cancers12092611
Chicago/Turabian StyleDuffau, Hugues. 2020. "Functional Mapping before and after Low-Grade Glioma Surgery: A New Way to Decipher Various Spatiotemporal Patterns of Individual Neuroplastic Potential in Brain Tumor Patients" Cancers 12, no. 9: 2611. https://doi.org/10.3390/cancers12092611




