Novel Prognostic Index of High-Risk Prostate Cancer Using Simple Summation of Very High-Risk Factors
Abstract
:Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Patients
2.2. Treatment Planning
2.2.1. Brachytherapy with or without External Beam Radiotherapy (BT ± EBRT)
2.2.2. External Beam Radiotherapy (EBRT)
2.3. Statistical Analysis
3. Results
3.1. Patient and Tumor Characteristics
3.2. Biochemical Disease-Free Survival Rate (bDFS)
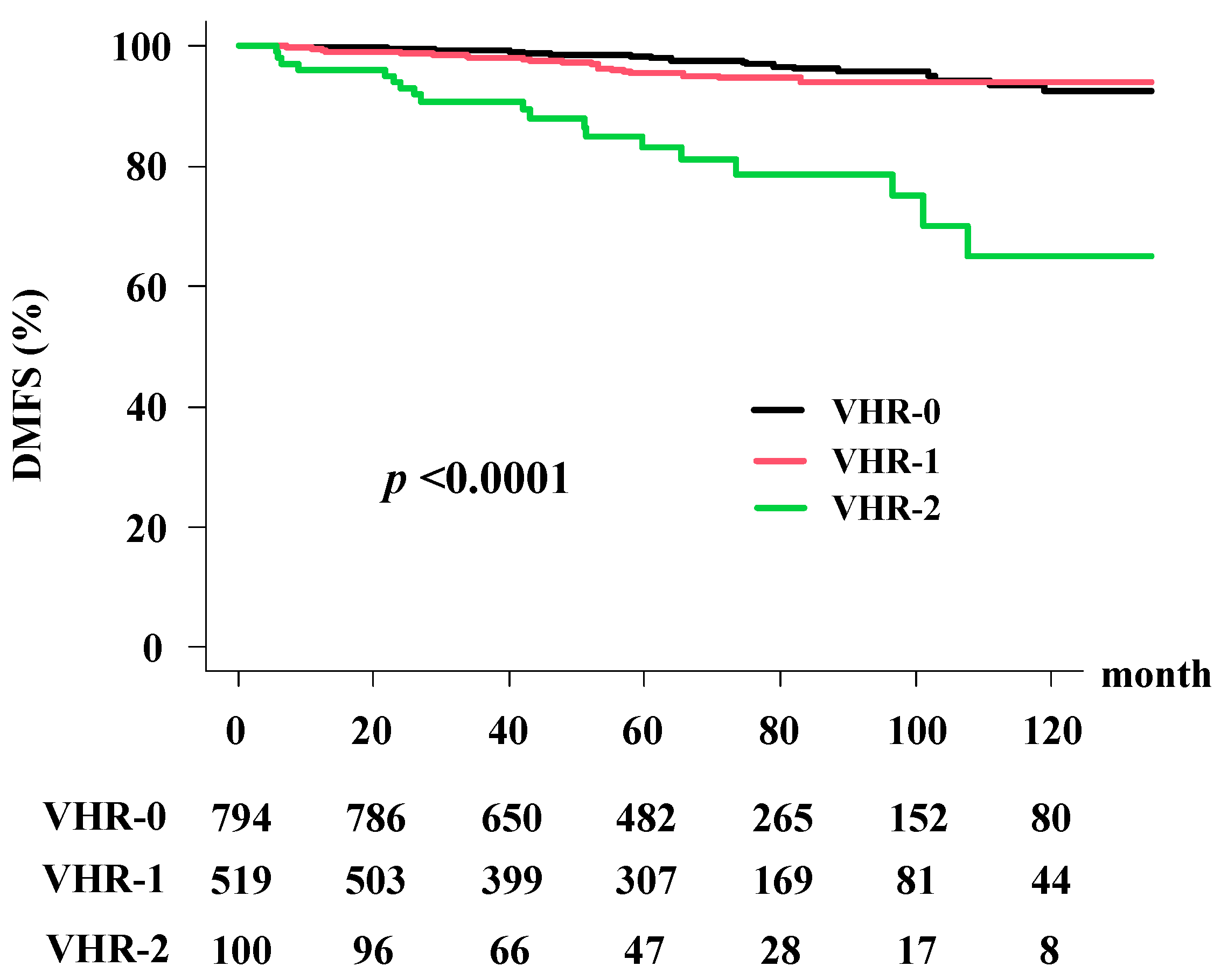
3.3. Distant Metastasis-Free Survival Rate (DMFS)
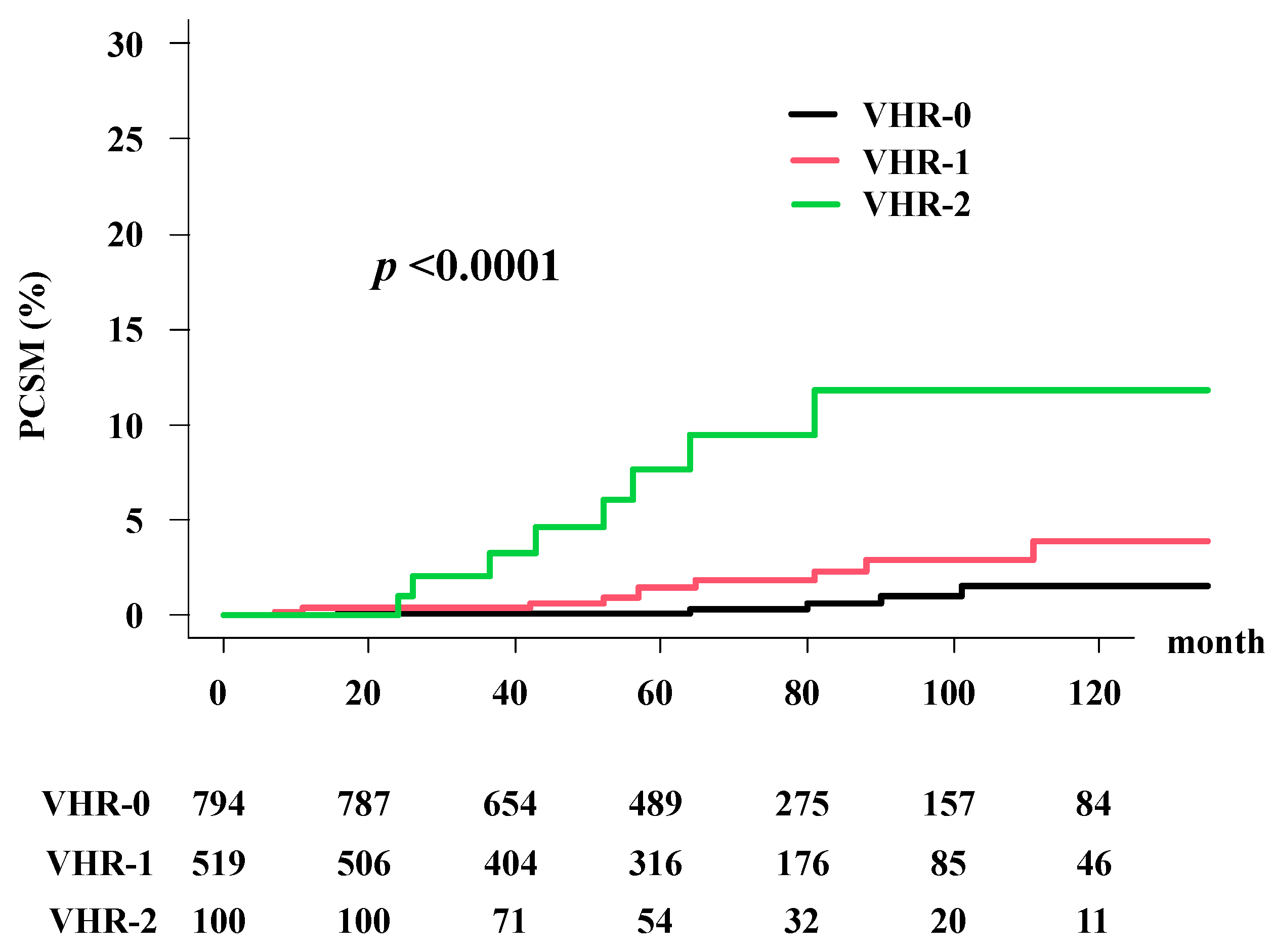
3.4. Prostate Cancer-Specific Morality (PCSM)
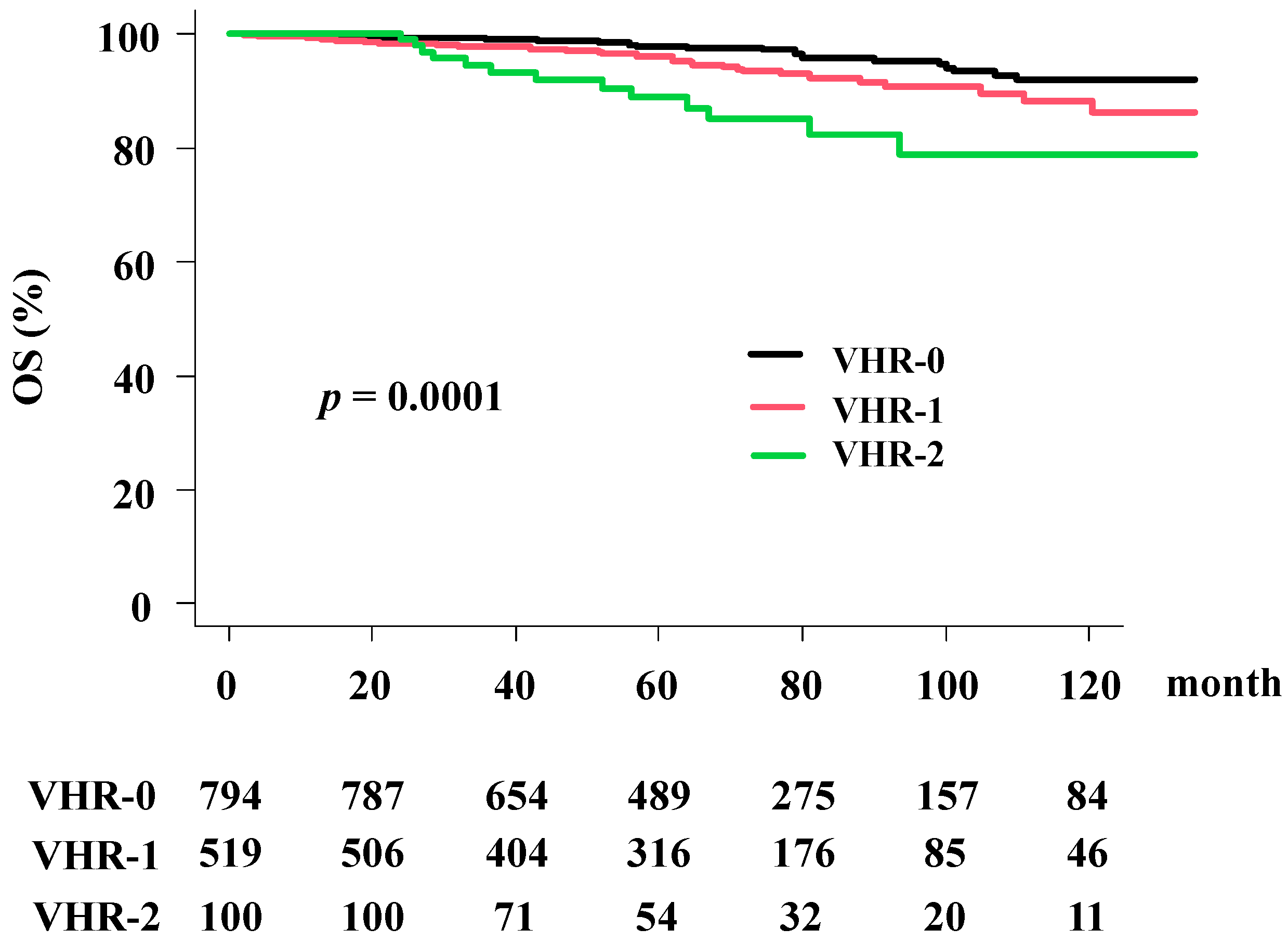
3.5. Overall Survival Rate (OS)
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- The National Comprehensive Cancer Network Clinical Practice Guidelines in Oncology. Prostate Cancer-Version 4. 2019. Available online: http://www.nccn.org/professionals/physician_gls/pdf/prostate.pdf (accessed on 2 February 2020).
- Sundi, D.; Wang, V.M.; Pierorazio, P.M.; Han, M.; Bivalacqua, T.J.; Ball, M.W.; Antonarakis, E.S.; Partin, A.W.; Schaeffer, E.M.; Ross, A.E. Very-high-risk localized prostate cancer: Definition and outcomes. Prostate Cancer Prostatic Dis. 2014, 17, 57–63. [Google Scholar] [CrossRef] [Green Version]
- Kasahara, T.; Ishizaki, F.; Kazama, A.; Yuki, E.; Yamana, K.; Maruyama, R.; Oshikane, T.; Kaidu, M.; Aoyama, H.; Bilim, V.; et al. High-dose-rate brachytherapy and hypofractionated external beam radiotherapy combined with long-term androgen deprivation therapy for very high-risk prostate cancer. Int. J. Urol. 2020, 27, 800–806. [Google Scholar] [CrossRef]
- Saad, A.; Goldstein, J.; Lawrence, Y.R.; Spieler, B.; Leibowitz-Amit, R.; Berger, R.; Davidson, T.; Urban, D.; Tsang, L.; Alezra, D.; et al. Classifying high-risk versus very high-risk prostate cancer: Is it relevant to outcomes of conformal radiotherapy and androgen deprivation? Radiat. Oncol. 2017, 12, 5. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Tomita, N.; Soga, N.; Ogura, Y.; Kageyama, T.; Kodaira, T. Very high-risk prostate cancer: Stratification by outcomes of radiotherapy and long-term androgen deprivation therapy. Asia-Pac. J. Clin. Oncol. 2017, 13, 145–151. [Google Scholar] [CrossRef]
- Ishiyama, H.; Satoh, T.; Kitano, M.; Tabata, K.-I.; Komori, S.; Ikeda, M.; Soda, I.; Kurosaka, S.; Sekiguchi, A.; Kimura, M.; et al. High-dose-rate brachytherapy and hypofractionated external beam radiotherapy combined with long-term hormonal therapy for high-risk and very high-risk prostate cancer: Outcomes after 5-year follow-up. J. Radiat. Res. 2014, 55, 509–517. [Google Scholar] [CrossRef] [PubMed]
- An Open Data of Multicenter Data Collection: Outcome of Radiation Therapy for Prostate Cancer to Establish a Prognostic Prediction System by Machine Learning (B17-278). Available online: https://www.khp.kitasato-u.ac.jp/ska/radiotherapy/arcivements/#results (accessed on 2 February 2020).
- Okihara, K.; Kobayashi, K.; Iwata, T.; Naitoh, Y.; Kamoi, K.; Kawauchi, A.; Yamada, K.; Miki, T. Assessment of permanent brachytherapy combined with androgen deprivation therapy in an intermediate-risk prostate cancer group without a Gleason score of 4 + 3: A single Japanese institutional experience. Int. J. Urol. 2014, 21, 271–276. [Google Scholar] [CrossRef] [PubMed]
- Sasaki, N.; Yamazaki, H.; Shimizu, D.; Suzuki, G.; Masui, K.; Nakamura, S.; Okabe, H.; Nishikawa, T.; Yoshida, K. Long-term Outcomes of a Dose–reduction Trial to Decrease Late Gastrointestinal Toxicity in Patients with Prostate Cancer Receiving Soft Tissue-matched Image-guided Intensity-modulated Radiotherapy. Anticancer Res. 2018, 38, 385–391. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kuban, D.A.; Levy, L.B.; Cheung, M.R.; Lee, A.K.; Choi, S.; Frank, S.; Pollack, A. Long-Term Failure Patterns and Survival in a Randomized Dose-Escalation Trial for Prostate Cancer. Who Dies of Disease? Int. J. Radiat. Oncol. Biol. Phys. 2011, 79, 1310–1317. [Google Scholar] [CrossRef]
- Epstein, J.I.; Amin, M.B.; Reuter, V.E.; Humphrey, P.A. Contemporary Gleason Grading of Prostatic Carcinoma: An Update with Discussion on Practical Issues to Implement the 2014 International Society of Urological Pathology (ISUP) Consensus Conference on Gleason Grading of Prostatic Carcinoma. Am. J. Surg. Pathol. 2017, 41, e1–e7. [Google Scholar] [CrossRef]
- Kanda, Y. Investigation of the freely available easy-to-use software ‘EZR’ for medical statistics. Bone Marrow Transplant. 2013, 48, 452–458. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sundi, D.; Tosoian, J.J.; Nyame, Y.A.; Alam, R.; Achim, M.; Reichard, C.A.; Li, J.; Wilkins, L.; Schwen, Z.; Han, M.; et al. Outcomes of very high-risk prostate cancer after radical prostatectomy: Validation study from 3 centers. Cancer 2019, 125, 391–397. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Narang, A.K.; Gergis, C.; Robertson, S.P.; He, P.; Ram, A.N.; McNutt, T.R.; Griffith, E.; DeWeese, T.A.; Honig, S.; Singh, H.; et al. Very High-Risk Localized Prostate Cancer: Outcomes Following Definitive Radiation. Int. J. Radiat. Oncol. 2016, 94, 254–262. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Umezawa, R.; Inaba, K.; Nakamura, S.; Wakita, A.; Okamoto, H.; Tsuchida, K.; Kashihara, T.; Kobayashi, K.; Harada, K.; Takahashi, K.; et al. Dose escalation of external beam radiotherapy for high-risk prostate cancer—Impact of multiple high-risk factor. Asian J. Urol. 2019, 6, 192–199. [Google Scholar] [CrossRef]
- Wattson, D.A.; Chen, M.H.; Moran, B.J.; Dosoretz, D.E.; Braccioforte, M.H.; Salenius, S.A.; D’Amico, A.V. The number of high-risk factors and the risk of prostate cancer-specific mortality after brachytherapy: Implications for treatment selection. Int. J. Radiat. Oncol. Biol. Phys. 2012, 82, e773–e779. [Google Scholar] [CrossRef]
- Rodrigues, G.; Lukka, H.; Warde, P.; Brundage, M.; Souhami, L.; Crook, J.; Cury, F.; Catton, C.; Mok, G.; Martin, A.-G.; et al. The prostate cancer risk stratification (ProCaRS) project: Recursive partitioning risk stratification analysis. Radiother. Oncol. 2013, 109, 204–210. [Google Scholar] [CrossRef]
- Fukagai, T.; Namiki, T.S.; Carlile, R.G.; Yoshida, H.; Namiki, M. Comparison of the clinical outcome after hormonal therapy for prostate cancer between Japanese and Caucasian men. BJU Int. 2006, 97, 1190–1193. [Google Scholar] [CrossRef]
- Sabolch, A.; Feng, F.Y.; Daignault-Newton, S.; Halverson, S.; Blas, K.; Phelps, L.; Olson, K.B.; Sandler, H.M.; Hamstra, D.A. Gleason Pattern 5 Is the Greatest Risk Factor for Clinical Failure and Death from Prostate Cancer after Dose-Escalated Radiation Therapy and Hormonal Ablation. Int. J. Radiat. Oncol. Biol. Phys. 2011, 81, e351–e360. [Google Scholar] [CrossRef]
- Tsumura, H.; Ishiyama, H.; Tabata, K.-I.; Katsumata, H.; Kobayashi, M.; Ikeda, M.; Kurosaka, S.; Fujita, T.; Kitano, M.; Satoh, T.; et al. Impact of five-tiered Gleason grade groups on prognostic prediction in clinical stage T3 prostate cancer undergoing high-dose-rate brachytherapy. Prostate 2017, 77, 1520–1527. [Google Scholar] [CrossRef]
- Chin, J.; Rumble, R.B.; Kollmeier, M.; Heath, E.; Efstathiou, J.; Dorff, T.; Berman, B.; Feifer, A.; Jacques, A.; Loblaw, D.A. Brachytherapy for Patients with Prostate Cancer: American Society of Clinical Oncology/Cancer Care Ontario Joint Guideline Update. J. Clin. Oncol. 2017, 35, 1737–1743. [Google Scholar] [CrossRef] [Green Version]
- Viani, G.A.; Stefano, E.J.; Afonso, S.L. Higher-Than-Conventional Radiation Doses in Localized Prostate Cancer Treatment: A Meta-analysis of Randomized, Controlled Trials. Int. J. Radiat. Oncol. 2009, 74, 1405–1418. [Google Scholar] [CrossRef] [PubMed]
- Spratt, D.E.; Zumsteg, Z.S.; Ghadjar, P.; Kollmeier, M.A.; Pei, X.; Cohen, G.; Polkinghorn, W.; Yamada, Y.; Zelefsky, M.J. Comparison of high-dose (86.4 Gy) IMRT vs combined brachytherapy plus IMRT for intermediate-risk prostate cancer. BJU Int. 2014, 114, 360–367. [Google Scholar] [CrossRef] [PubMed]
- Kent, A.R.; Matheson, B.; Millar, J.L. Improved survival for patients with prostate cancer receiving high-dose-rate brachytherapy boost to EBRT compared with EBRT alone. Brachytherapy 2019, 18, 313–321. [Google Scholar] [CrossRef] [PubMed]
- Wedde, T.B.; Småstuen, M.C.; Brabrand, S.; Fosså, S.D.; Kaasa, S.; Tafjord, G.; Russnes, K.M.; Hellebust, T.P.; Lilleby, W. Ten-year survival after High-Dose-Rate Brachytherapy combined with External Beam Radiation Therapy in high-risk prostate cancer: A comparison with the Norwegian SPCG-7 cohort. Radiother. Oncol. 2019, 132, 211–217. [Google Scholar] [CrossRef]
- Kishan, A.U.; Cook, R.R.; Ciezki, J.P.; Ross, A.E.; Pomerantz, M.M.; Nguyen, P.L.; Shaikh, T.; Tran, P.T.; Sandler, K.A.; Stock, R.G.; et al. Radical Prostatectomy, External Beam Radiotherapy, or External Beam Radiotherapy with Brachytherapy Boost and Disease Progression and Mortality in Patients with Gleason Score 9–10 Prostate Cancer. JAMA 2018, 319, 896–905. [Google Scholar] [CrossRef]
- Perera, M.; Papa, N.; Roberts, M.; Williams, M.; Udovicich, C.; Vela, I.; Christidis, D.; Bolton, D.; Hofman, M.S.; Lawrentschuk, N.; et al. Gallium-68 Prostate-specific Membrane Antigen Positron Emission Tomography in Advanced Prostate Cancer—Updated Diagnostic Utility, Sensitivity, Specificity, and Distribution of Prostate-specific Membrane Antigen-avid Lesions: A Systematic Review and Meta-analysis. Eur. Urol. 2020, 77, 403–417. [Google Scholar] [CrossRef] [PubMed]
- Komura, K.; Sweeney, C.J.; Inamoto, T.; Ibuki, N.; Azuma, H.; Kantoff, P.W. Current treatment strategies for advanced prostate cancer. Int. J. Urol. 2018, 25, 220–231. [Google Scholar] [CrossRef] [Green Version]

| Variables | Strata | Total | |
|---|---|---|---|
| (n = 1413) | |||
| No. or Median (range) | (%) | ||
| Age | 71 (60–89) | ||
| T Category | ≤2 3a 3b 4 | 583 587 215 28 | (41%) (41%) (15%) (2%) |
| iPSA (ng/mL) | ≤20 20< 50< 100< | 684 474 151 104 | (48%) (33%) (11%) (7%) |
| Gleason score | ≤7 8≤ 9≤ 10≤ | 591 346 436 40 | (42%) (24%) (31%) (3%) |
| Modality | EBRT BT ± EBRT | 558 855 | (39%) (60%) |
| Hormonal therapy | Yes | 1348 | (95%) |
| Duration | (Months) | 40 (1–140) | |
| No | 65 | (5%) | |
| Neoadjuvant | Yes | 1200 | (85%) |
| Duration | (Month) | 9 (1–92) | |
| Adjuvant | Yes | 921 | (65%) |
| Duration | (Month) | 36 (1–134) | |
| Follow-up | (Months) | 69.6 (2–177) | |
| Variables | Strata | VHR-0 (n = 794) | VHR-1 (n = 519) | VHR-2 (n = 100) | p-Value | |||
|---|---|---|---|---|---|---|---|---|
| No. or Median (Range) | (%) | No. or Median (Range) | (%) | No. or Median (Range) | (%) | |||
| Age | 70 (60–86) | 71 (60–86) | 70 (60–89) | 0.02202 | ||||
| T Category | ≤2 | 386 | (49%) | 197 | (38%) | 0 | (0%) | <0.0001 |
| 3a | 408 | (51%) | 179 | (34%) | 0 | (0%) | ||
| 3b | 0 | (0%) | 130 | (25%) | 85 | (85%) | ||
| 4 | 0 | (0%) | 13 | (3%) | 15 | (15%) | ||
| iPSA (ng/mL) | ≤20 | 373 | (47%) | 281 | (54%) | 30 | (30%) | <0.0001 |
| 20< | 306 | (39%) | 135 | (26%) | 33 | (33%) | ||
| 50< | 74 | (9%) | 51 | (10%) | 26 | (26%) | ||
| 100< | 41 | (5%) | 52 | (10%) | 11 | (11%) | ||
| Gleason score | −7 | 480 | (60%) | 111 | (21%) | 0 | (0%) | <0.0001 |
| 8 | 314 | (40%) | 32 | (6%) | 0 | (0%) | ||
| 9 | 0 | (0%) | 346 | (67%) | 90 | (90%) | ||
| 10 | 0 | (0%) | 30 | (6%) | 10 | (10%) | ||
| Modality | EBRT | 273 | (34%) | 224 | (43%) | 61 | (61%) | <0.0001 |
| BT ± EBRT | 521 | (66%) | 295 | (57%) | 39 | (39%) | ||
| Hormonal Therapy | Yes | 746 | (94%) | 503 | (97%) | 99 | (99%) | 0.0088 |
| (Months) | 41 (1–112) | 33 (2–140) | 25 (4–128) | 0.0777 | ||||
| No | 48 | (6%) | 16 | (3%) | 1 | (1%) | ||
| Neoadjuvant | Yes | 669 | 438 | 93 | ||||
| Duration | (Month) | 10 (1–89) | 9 (1–92) | 8 (4–24) | 0.0696 | |||
| Adjuvant | Yes | 526 | 334 | 61 | ||||
| Duration | (Month) | 36 (1–114) | 36 (1–134) | 36 (1–49) | 0.0171 | |||
| Follow-up | (Months) | 68.2 (9–177) | 67 (2–158) | 62.9 (20.4–153) | 0.2253 | |||
| Prostate cancer-specific mortality(PCSM) | No Yes | 787 7 | (99%) (1%) | 508 11 | (98%) (2%) | 92 8 | (92%) (8%) | <0.0001 |
| PSA failure | No | 720 | (91%) | 435 | (84%) | 60 | (60%) | <0.0001 |
| Yes | 74 | (9%) | 84 | (16%) | 40 | (40%) | ||
| Overall survival | Alive | 765 | (96%) | 486 | (94%) | 87 | (87%) | 0.0002 |
| Death | 29 | (4%) | 33 | (6%) | 13 | (13%) | ||
| Distant metastasis | No | 769 | (97%) | 497 | (96%) | 81 | (81%) | <0.0001 |
| Yes | 25 | (3%) | 22 | (4%) | 19 | (19%) | ||
| Variable | Strata | bDFS | PCSM | OS | MFS | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| HRa | 95% CI | p | HRa | 95% CI | p | HRa | 95% CI | p | HRa | 95% CI | p | ||
| Age | −70 vs. 71- | 1.106 | 0.836–1.464 | 0.4817 | 0.6257 | 0.281–1.39 | 0.25 | 1.493 | 0.9374–2.377 | 0.091 | 0.7179 | 0.4392–1.173 | 0.1862 |
| T Classification | ≤2 vs. 3a≤ | 2.234 | 1.625–3.071 | <0.0001 | 4.013 | 1385–11.62 | 0.01 | 1.95 | 1.169–3.254 | 0.01057 | 4.645 | 2.299–9.382 | <0.0001 |
| ≤3a vs. 3b≤ | 2.819 | 2.101–3.783 | <0.0001 | 3.1887 | 1.454–6.988 | 0.0038 | 2.074 | 1.261–3.411 | 0.004 | 3.144 | 1.911–5.173 | <0.0001 | |
| ≤3b vs. 4≤ | 4.837 | 2.855–8.195 | <0.0001 | 5.699 | 1.632–19.9 | 0.0064 | 1.747 | 0.5496–5.552 | 0.3444 | 7.153 | 3.41–15 | <0.0001 | |
| Gleason Score | ≤7 vs. 8≤ | 1.652 | 1.227–2.224 | 0.0009 | 2.227 | 0.934–5.308 | 0.0708 | 1.794 | 1.097–2.933 | 0.01989 | 1.848 | 1.141–3.319 | 0.0145 |
| ≤8 vs. 9≤ | 2.219 | 1.679–2.934 | <0.0001 | 3.833 | 1.591–8.295 | 0.0022 | 1.978 | 1.257–3.114 | 0.0032 | 2.376 | 1.464–3.855 | 0.0005 | |
| ≤9 vs. 10≤ | 1.316 | 0.619–2.799 | 0.475 | 4.94 | 1.435–17.0 | 0.011 | 2.769 | 1.115–6.874 | 0.0281 | 1.82 | 0.571–5.801 | 0.3111 | |
| Pretreatment PSA (ng/mL) | ≤20 vs. 20< | 1.635 | 1.223–2.187 | 0.0009 | 1.393 | 0.6331–3.065 | 0.41 | 0.7676 | 0.4818–1.223 | 0.2655 | 1.508 | 0.9127–2.492 | 0.1088 |
| ≤50 vs. 50< | 2.001 | 1.482–2.701 | <0.0001 | 1.774 | 0.7739–4.068 | 0.18 | 1.758 | 1.075–2.875 | 0.02454 | 2.074 | 1.242–3.461 | 0.005 | |
| ≤100 vs. 100< | 1.621 | 1.097–2.576 | 0.0171 | 2.316 | 0.3038–17.65 | 0.42 | 1.135 | 0.520–2.475 | 0.7498 | 1.371 | 0.6254–3.006 | 0.4304 | |
| ADT duration (months) | ≤40 vs. 41- | 0.5067 | 0.372–0.689 | <0.0001 | 0.9922 | 0.9746–1.01 | 0.39 | 1.193 | 0.7545–1.888 | 0.4497 | 0.8925 | 0.5434–1.466 | 0.6531 |
| Modality | EBRT vs. BT ± EBRT | 0.355 | 0.266–0.474 | <0.0001 | 1.128 | 0.5181–2.467 | 0.76 | 0.9053 | 0.566–1.447 | 0.6775 | 0.9109 | 0.5537–1.498 | 0.7133 |
| No. of very-high risk factors | VHR-0 | 1 | (referent) | - | 1 | (referent) | - | 1 | (referent) | - | 1 | (referent) | - |
| VHR-1 | 1.93 | 1.412–2.639 | <0.0001 | 2.408 | 0.9297–6.244 | 0.07 | 1.8 | 1.092–2.966 | 0.021 | 1.41 | 0.795–2.502 | 0.24 | |
| VHR-2 | 5.989 | 4.072–8.808 | <0.0001 | 10.07 | 3.659–27.74 | <0.0001 | 3.756 | 1.952–7.228 | <0.0001 | 7.045 | 3.878–12.798 | <0.0001 | |
| Variable | Strata | bDFS | PCSM | OS | MFS | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| HRa | 95% CI | p | HRa | 95% CI | p | HRa | 95% CI | p | HRa | 95% CI | p | ||
| Age | ≤70 vs. 71- | 0.798 | 0.601–1.059 | 0.1182 | 0.6676 | 0.2844–1.567 | 0.35 | 1.572 | 0.982–2.518 | 0.0597 | 0.772 | 0.469–1.270 | 0.308 |
| ADT Duration (months) | ≤40 vs. 41- | 0.87 | 0.606–1.250 | 0.4516 | 0.4535 | 0.1528–1.346 | 0.15 | 1.212 | 0.709–2.073 | 0.4823 | 0.825 | 0.466–1.463 | 0.5113 |
| Modality | EBRT vs, BT ± EBRT | 0.447 | 0.315–0.633 | <0.0001 | 0.6297 | 0.2433–1.630 | 0.34 | 1.314 | 0.750–2.301 | 0.3401 | 1.53 | 0.876–2.888 | 0.1277 |
| No. of very high-risk factors | VHR-0 | 1 | (referent) | - | 1 | (referent) | - | 1 | (referent) | - | 1 | (referent) | - |
| VHR-1 | 1.723 | 1.256–2.362 | 0.0007 | 2.391 | 0.8974–6.373 | 0.081 | 1.88 | 1.137–3.109 | 0.0139 | 1.491 | 0.837–2.656 | 0.1756 | |
| VHR-2 | 4.55 | 3.065–6.755 | <0.0001 | 9.607 | 3.29–28.05 | <0.0001 | 4.327 | 2.206–8.487 | <0.0001 | 7.904 | 4.251–14.696 | <0.0001 | |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Yamazaki, H.; Suzuki, G.; Masui, K.; Aibe, N.; Shimizu, D.; Kimoto, T.; Yamada, K.; Shiraishi, T.; Fujihara, A.; Okihara, K.; et al. Novel Prognostic Index of High-Risk Prostate Cancer Using Simple Summation of Very High-Risk Factors. Cancers 2021, 13, 3486. https://doi.org/10.3390/cancers13143486
Yamazaki H, Suzuki G, Masui K, Aibe N, Shimizu D, Kimoto T, Yamada K, Shiraishi T, Fujihara A, Okihara K, et al. Novel Prognostic Index of High-Risk Prostate Cancer Using Simple Summation of Very High-Risk Factors. Cancers. 2021; 13(14):3486. https://doi.org/10.3390/cancers13143486
Chicago/Turabian StyleYamazaki, Hideya, Gen Suzuki, Koji Masui, Norihiro Aibe, Daisuke Shimizu, Takuya Kimoto, Kei Yamada, Takumi Shiraishi, Atsuko Fujihara, Koji Okihara, and et al. 2021. "Novel Prognostic Index of High-Risk Prostate Cancer Using Simple Summation of Very High-Risk Factors" Cancers 13, no. 14: 3486. https://doi.org/10.3390/cancers13143486
APA StyleYamazaki, H., Suzuki, G., Masui, K., Aibe, N., Shimizu, D., Kimoto, T., Yamada, K., Shiraishi, T., Fujihara, A., Okihara, K., Yoshida, K., Nakamura, S., & Okabe, H. (2021). Novel Prognostic Index of High-Risk Prostate Cancer Using Simple Summation of Very High-Risk Factors. Cancers, 13(14), 3486. https://doi.org/10.3390/cancers13143486








