CENP-A Subnuclear Localization Pattern as Marker Predicting Curability by Chemoradiation Therapy for Locally Advanced Head and Neck Cancer Patients
Abstract
:Simple Summary
Abstract
1. Introduction
2. Results
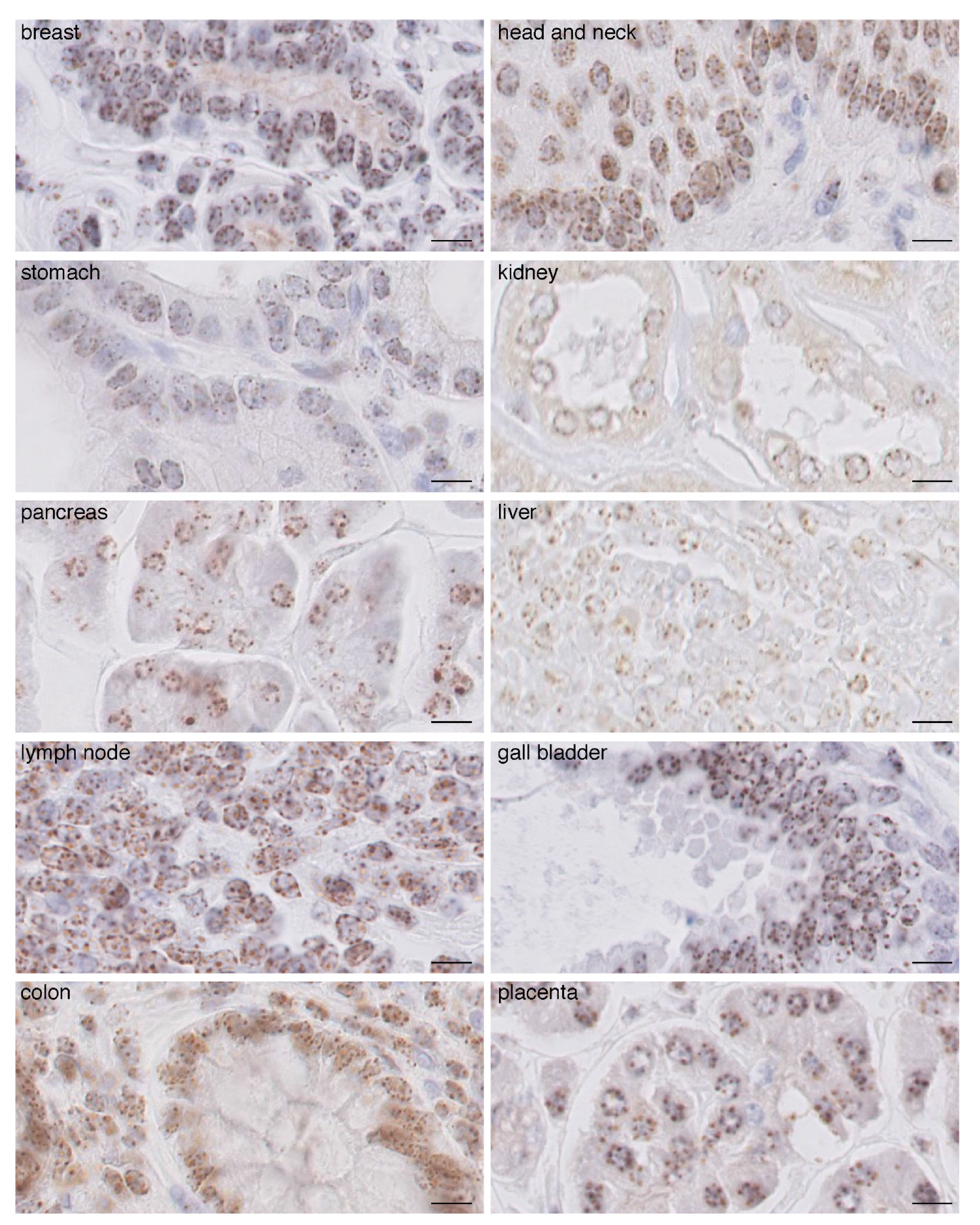
2.1. CENP-A Clusters in Foci at the Nuclear Periphery in Normal Human Tissue Fixed with AFA
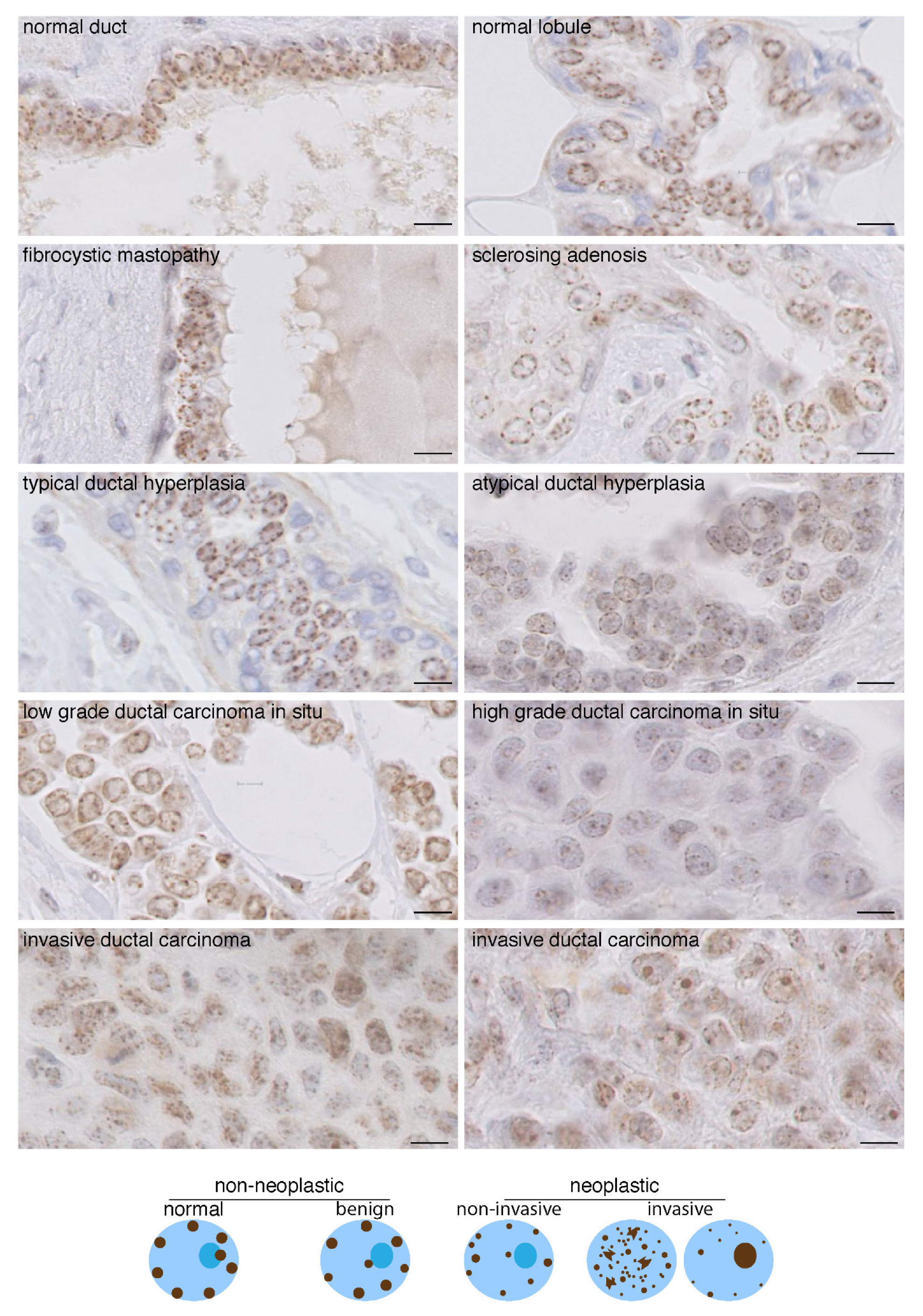
2.2. In Carcinomas CENP-A Clustering and Localization at the Nuclear Periphery Are Altered
2.3. Change in CENP-A Nuclear Localization Pattern as a Marker of Malignancy
2.4. CENP-A Nuclear Localization in Radioresistant and Radiosensitive HNSSC-Derived Cell Line Xenografts
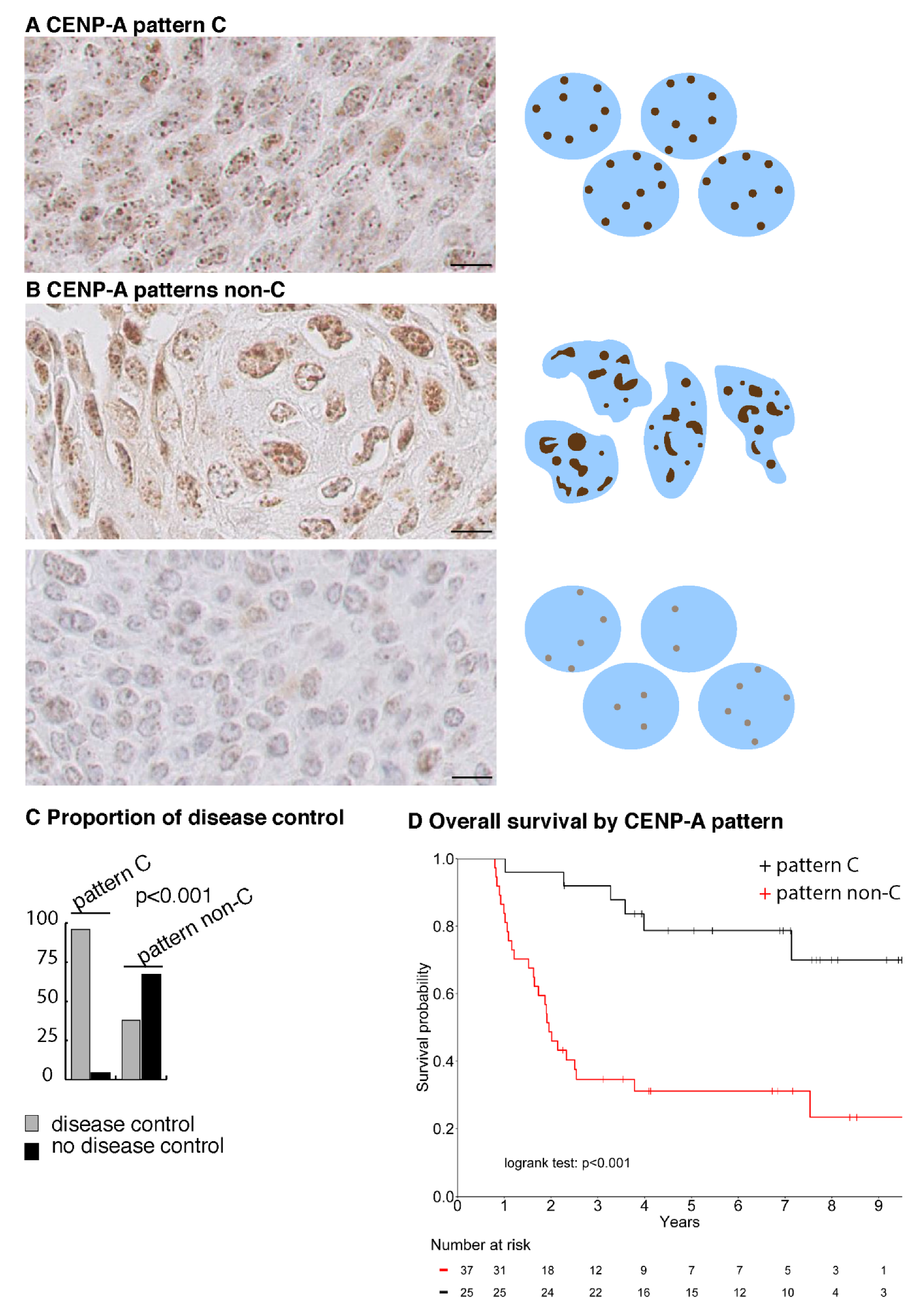
2.5. CENP-A Nuclear Localization Pattern Is Associated with Response to CCRT
2.6. A CENP-A Nuclear Localization Pattern Is a Marker of Predictive Value for Curability of HNSCC Patients by CCRT
3. Discussion
4. Methods
4.1. Tissue Samples
4.2. Immunohistochemistry Imaging
4.3. HNSCC Patients
4.4. Statistical Analysis
5. Conclusions
6. Patent
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- You, G.R.; Cheng, A.J.; Lee, L.Y.; Huang, Y.C.; Liu, H.; Chen, Y.J.; Chang, J.T. Prognostic signature associated with radioresistance in head and neck cancer via transcriptomic and bioinformatic analyses. BMC Cancer 2019, 19, 64. [Google Scholar] [CrossRef] [Green Version]
- Ahmad, P.; Sana, J.; Slavik, M.; Slampa, P.; Smilek, P.; Slaby, O. MicroRNAs Involvement in Radioresistance of Head and Neck Cancer. Dis. Markers 2017, 2017, 8245345. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Budach, V.; Tinhofer, I. Novel prognostic clinical factors and biomarkers for outcome prediction in head and neck cancer: A systematic review. Lancet Oncol. 2019, 20, e313–e326. [Google Scholar] [CrossRef]
- Fakhry, C.; Psyrri, A.; Chaturvedhi, A. HPV and head and neck cancers: State-of-the-science. Oral Oncol. 2014, 50, 353–355. [Google Scholar] [CrossRef] [PubMed]
- Lassen, P.; Eriksen, J.G.; Hamilton-Dutoit, S.; Tramm, T.; Alsner, J.; Overgaard, J. Effect of HPV-associated p16INK4A expression on response to radiotherapy and survival in squamous cell carcinoma of the head and neck. J. Clin. Oncol. 2009, 27, 1992–1998. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Witkiewicz, A.K.; Knudsen, K.E.; Dicker, A.P.; Knudsen, E.S. The meaning of p16(ink4a) expression in tumors: Functional significance, clinical associations and future developments. Cell Cycle 2011, 10, 2497–2503. [Google Scholar] [CrossRef] [Green Version]
- He, X.; Xu, H.; Zhao, W.; Zhan, M.; Li, Y.; Liu, H.; Tan, L.; Lu, L. POPDC3 is a potential biomarker for prognosis and radioresistance in patients with head and neck squamous cell carcinoma. Oncol. Lett. 2019, 18, 5468–5480. [Google Scholar] [CrossRef] [Green Version]
- Couture, C.; Raybaud-Diogene, H.; Tetu, B.; Bairati, I.; Murry, D.; Allard, J.; Fortin, A. p53 and Ki-67 as markers of radioresistance in head and neck carcinoma. Cancer 2002, 94, 713–722. [Google Scholar] [CrossRef] [Green Version]
- Zhou, G.; Liu, Z.; Myers, J.N. TP53 Mutations in Head and Neck Squamous Cell Carcinoma and Their Impact on Disease Progression and Treatment Response. J. Cell Biochem. 2016, 117, 2682–2692. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Alexandrov, L.B.; Kim, J.; Haradhvala, N.J.; Huang, M.N.; Tian Ng, A.W.; Wu, Y.; Boot, A.; Covington, K.R.; Gordenin, D.A.; Bergstrom, E.N.; et al. The repertoire of mutational signatures in human cancer. Nature 2020, 578, 94–101. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Krijger, P.H.; de Laat, W. Identical cells with different 3D genomes; cause and consequences? Curr. Opin. Genet. Dev. 2013, 23, 191–196. [Google Scholar] [CrossRef]
- Bonev, B.; Cavalli, G. Organization and function of the 3D genome. Nat. Rev. Genet. 2016, 17, 661–678. [Google Scholar] [CrossRef]
- Fritz, A.J.; Sehgal, N.; Pliss, A.; Xu, J.; Berezney, R. Chromosome territories and the global regulation of the genome. Genes Chromosomes Cancer 2019, 58, 407–426. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lever, E.; Sheer, D. The role of nuclear organization in cancer. J. Pathol. 2010, 220, 114–125. [Google Scholar] [CrossRef]
- Zaidi, S.K.; Fritz, A.J.; Tracy, K.M.; Gordon, J.A.; Tye, C.E.; Boyd, J.; Van Wijnen, A.J.; Nickerson, J.A.; Imbalzano, A.N.; Lian, J.B.; et al. Nuclear organization mediates cancer-compromised genetic and epigenetic control. Adv. Biol. Regul. 2018, 69, 1–10. [Google Scholar] [CrossRef]
- Mai, S. The three-dimensional cancer nucleus. Genes Chromosomes Cancer 2019, 58, 462–473. [Google Scholar] [CrossRef] [Green Version]
- Morgan, M.A.; Shilatifard, A. Chromatin signatures of cancer. Genes Dev. 2015, 29, 238–249. [Google Scholar] [CrossRef] [Green Version]
- Helin, K.; Minucci, S. The Role of Chromatin-Associated Proteins in Cancer. Annu. Rev. Cancer Biol. 2017, 1, 355–377. [Google Scholar] [CrossRef] [Green Version]
- Morel, D.; Jeffery, D.; Aspeslagh, S.; Almouzni, G.; Postel-Vinay, S. Combining epigenetic drugs with other therapies for solid tumours—Past lessons and future promise. Nat. Rev. Clin. Oncol. 2020, 17, 91–107. [Google Scholar] [CrossRef] [PubMed]
- Earnshaw, W.C.; Rothfield, N. Identification of a family of human centromere proteins using autoimmune sera from patients with scleroderma. Chromosoma 1985, 91, 313–321. [Google Scholar] [CrossRef] [PubMed]
- Palmer, D.K.; O’Day, K.; Trong, H.L.; Charbonneau, H.; Margolis, R.L. Purification of the centromere-specific protein CENP-A and demonstration that it is a distinctive histone. Proc. Natl. Acad. Sci. USA 1991, 88, 3734–3738. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Muller, S.; Almouzni, G. Chromatin dynamics during the cell cycle at centromeres. Nat. Rev. Genet. 2017, 18, 192–208. [Google Scholar] [CrossRef] [PubMed]
- Yan, K.; Yang, J.; Zhang, Z.; McLaughlin, S.H.; Chang, L.; Fasci, D.; Ehrenhofer-Murray, A.E.; Heck, A.J.R.; Barford, D. Structure of the inner kinetochore CCAN complex assembled onto a centromeric nucleosome. Nature 2019, 574, 278–282. [Google Scholar] [CrossRef]
- Muller, H.; Gil, J., Jr.; Drinnenberg, I.A. The Impact of Centromeres on Spatial Genome Architecture. Trends Genet. 2019, 35, 565–578. [Google Scholar] [CrossRef] [Green Version]
- Jansz, N.; Torres-Padilla, M.E. Genome activation and architecture in the early mammalian embryo. Curr. Opin. Genet. Dev. 2019, 55, 52–58. [Google Scholar] [CrossRef]
- Schlesinger, S.; Meshorer, E. Open Chromatin, Epigenetic Plasticity, and Nuclear Organization in Pluripotency. Dev. Cell 2019, 48, 135–150. [Google Scholar] [CrossRef] [Green Version]
- Feodorova, Y.; Falk, M.; Mirny, L.A.; Solovei, I. Viewing Nuclear Architecture through the Eyes of Nocturnal Mammals. Trends Cell Biol. 2020, 30, 276–289. [Google Scholar] [CrossRef] [PubMed]
- Ma, X.J.; Salunga, R.; Tuggle, J.T.; Gaudet, J.; Enright, E.; McQuary, P.; Payette, T.; Pistone, M.; Stecker, K.; Zhang, B.M.; et al. Gene expression profiles of human breast cancer progression. Proc. Natl. Acad. Sci. USA 2003, 100, 5974–5979. [Google Scholar] [CrossRef] [Green Version]
- Montes de Oca, R.; Gurard-Levin, Z.A.; Berger, F.; Rehman, H.; Martel, E.; Corpet, A.; de Koning, L.; Vassias, I.; Wilson, L.O.; Meseure, D.; et al. The histone chaperone HJURP is a new independent prognostic marker for luminal A breast carcinoma. Mol. Oncol. 2015, 9, 657–674. [Google Scholar] [CrossRef]
- Tomonaga, T.; Matsushita, K.; Yamaguchi, S.; Oohashi, T.; Shimada, H.; Ochiai, T.; Yoda, K.; Nomura, F. Overexpression and mistargeting of centromere protein-A in human primary colorectal cancer. Cancer Res. 2003, 63, 3511–3516. [Google Scholar] [PubMed]
- Li, Y.; Zhu, Z.; Zhang, S.; Yu, D.; Yu, H.; Liu, L.; Cao, X.; Wang, L.; Gao, H.; Zhu, M. ShRNA-targeted centromere protein A inhibits hepatocellular carcinoma growth. PLoS ONE 2011, 6, e17794. [Google Scholar] [CrossRef] [Green Version]
- Wu, Q.; Qian, Y.M.; Zhao, X.L.; Wang, S.M.; Feng, X.J.; Chen, X.F.; Zhang, S.H. Expression and prognostic significance of centromere protein A in human lung adenocarcinoma. Lung Cancer 2012, 77, 407–414. [Google Scholar] [CrossRef]
- Qiu, J.J.; Guo, J.J.; Lv, T.J.; Jin, H.Y.; Ding, J.X.; Feng, W.W.; Zhang, Y.; Hua, K.Q. Prognostic value of centromere protein-A expression in patients with epithelial ovarian cancer. Tumour Biol. J. Int. Soc. Oncodev. Biol. Med. 2013, 34, 2971–2975. [Google Scholar] [CrossRef] [PubMed]
- Gu, X.M.; Fu, J.; Feng, X.J.; Huang, X.; Wang, S.M.; Chen, X.F.; Zhu, M.H.; Zhang, S.H. Expression and prognostic relevance of centromere protein A in primary osteosarcoma. Pathol. Res. Pract. 2014, 210, 228–233. [Google Scholar] [CrossRef]
- Hu, Z.; Huang, G.; Sadanandam, A.; Gu, S.; Lenburg, M.E.; Pai, M.; Bayani, N.; Blakely, E.A.; Gray, J.W.; Mao, J.H. The expression level of HJURP has an independent prognostic impact and predicts the sensitivity to radiotherapy in breast cancer. Breast Cancer Res. 2010, 12, R18. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lacoste, N.; Woolfe, A.; Tachiwana, H.; Garea, A.V.; Barth, T.; Cantaloube, S.; Kurumizaka, H.; Imhof, A.; Almouzni, G. Mislocalization of the Centromeric Histone Variant CenH3/CENP-A in Human Cells Depends on the Chaperone DAXX. Mol. Cell 2014, 53, 631–644. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Jeffery, D.; Gatto, A.; Podsypanina, K.; Renaud-Pageot, C.; Ponce Landete, R.; Bonneville, L.; Dumont, M.; Fachinetti, D.; Almouzni, G. CENP-A overexpression promotes distinct fates in human cells, depending on p53 status. Commun. Biol. 2021, 4, 417. [Google Scholar] [CrossRef]
- Zhang, W.; Mao, J.H.; Zhu, W.; Jain, A.K.; Liu, K.; Brown, J.B.; Karpen, G.H. Centromere and kinetochore gene misexpression predicts cancer patient survival and response to radiotherapy and chemotherapy. Nat. Commun. 2016, 7, 12619. [Google Scholar] [CrossRef]
- Weichselbaum, R.R.; Dahlberg, W.; Beckett, M.; Karrison, T.; Miller, D.; Clark, J.; Ervin, T.J. Radiation-resistant and repair-proficient human tumor cells may be associated with radiotherapy failure in head- and neck-cancer patients. Proc. Natl. Acad. Sci. USA 1986, 83, 2684–2688. [Google Scholar] [CrossRef] [Green Version]
- Storch, K.; Eke, I.; Borgmann, K.; Krause, M.; Richter, C.; Becker, K.; Schrock, E.; Cordes, N. Three-dimensional cell growth confers radioresistance by chromatin density modification. Cancer Res. 2010, 70, 3925–3934. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Weimer, R.; Haaf, T.; Kruger, J.; Poot, M.; Schmid, M. Characterization of centromere arrangements and test for random distribution in G0, G1, S, G2, G1, and early S’ phase in human lymphocytes. Hum. Genet. 1992, 88, 673–682. [Google Scholar] [CrossRef]
- Alcobia, I.; Dilao, R.; Parreira, L. Spatial associations of centromeres in the nuclei of hematopoietic cells: Evidence for cell-type-specific organizational patterns. Blood 2000, 95, 1608–1615. [Google Scholar] [CrossRef]
- Weierich, C.; Brero, A.; Stein, S.; von Hase, J.; Cremer, C.; Cremer, T.; Solovei, I. Three-dimensional arrangements of centromeres and telomeres in nuclei of human and murine lymphocytes. Chromosome Res. 2003, 11, 485–502. [Google Scholar] [CrossRef]
- Skalnikova, M.; Kozubek, S.; Lukasova, E.; Bartova, E.; Jirsova, P.; Cafourkova, A.; Koutna, I.; Kozubek, M. Spatial arrangement of genes, centromeres and chromosomes in human blood cell nuclei and its changes during the cell cycle, differentiation and after irradiation. Chromosome Res. 2000, 8, 487–499. [Google Scholar] [CrossRef] [PubMed]
- Finn, E.H.; Misteli, T. Molecular basis and biological function of variability in spatial genome organization. Science 2019, 365, eaaw9498. [Google Scholar] [CrossRef]
- Filipescu, D.; Naughtin, M.; Podsypanina, K.; Lejour, V.; Wilson, L.; Gurard-Levin, Z.A.; Orsi, G.A.; Simeonova, I.; Toufektchan, E.; Attardi, L.D.; et al. Essential role for centromeric factors following p53 loss and oncogenic transformation. Genes Dev. 2017, 31, 463–480. [Google Scholar] [CrossRef] [Green Version]
- Benson, E.; Li, R.; Eisele, D.; Fakhry, C. The clinical impact of HPV tumor status upon head and neck squamous cell carcinomas. Oral Oncol. 2014, 50, 565–574. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Amin, M.B.; Edge, S.; Greene, F.; Byrd, D.R.; Brookland, R.K.; Washington, M.K.; Gershenwald, J.E.; Compton, C.C.; Hess, K.R.; Sullivan, D.C.; et al. AJCC Cancer Staging Manual, 8th ed.; Springer International Publishing: New York, NY, USA, 2017. [Google Scholar]
- Ang, K.K.; Harris, J.; Wheeler, R.; Weber, R.; Rosenthal, D.I.; Nguyen-Tân, P.F.; Westra, W.H.; Chung, C.H.; Jordan, R.C.; Lu, C.; et al. Human papillomavirus and survival of patients with oropharyngeal cancer. N. Engl. J. Med. 2010, 363, 24–35. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Courtiol, P.; Maussion, C.; Moarii, M.; Pronier, E.; Pilcer, S.; Sefta, M.; Manceron, P.; Toldo, S.; Zaslavskiy, M.; Le Stang, N.; et al. Deep learning-based classification of mesothelioma improves prediction of patient outcome. Nat. Med. 2019, 25, 1519–1525. [Google Scholar] [CrossRef] [PubMed]
- International Union Against Cancer (UICC). TNM Classification of Malignant Tumours, 7th ed.; Sobin, L.H., Gospodarowicz, M.K., Wittekind, C., Eds.; Wiley-Blackwell: Hoboken, NJ, USA, 2011. [Google Scholar]

| Characteristic | Total (N = 62) | CENP-A Pattern | |||
|---|---|---|---|---|---|
| C (N = 25) | Non-C (N = 37) | Test | |||
| Age (year) | NS | ||||
| Median (IQR) | 62 (56–68) | 61 (56–64) | 63 (56–69) | ||
| Ki67 (%) | NS | ||||
| Median (IQR) | 60 (35–75) | 60 (35–75) | 60 (30–75) | ||
| Gender N (%) | NS | ||||
| F | 10 (16.1%) | 6 (24%) | 4 (10.8%) | ||
| M | 52 (83.9%) | 19 (76%) | 33 (89.2%) | ||
| T (TNM) N (%) | 0.06 | ||||
| T1 | 4 (6.5%) | 1 (4%) | 3 (8.1%) | ||
| T2 | 10 (16.1%) | 4 (16%) | 6 (16.2%) | ||
| T3 | 23 (37.1%) | 14 (56%) | 9 (24.3%) | ||
| T4 | 25 (40.3%) | 6 (24%) | 19 (51.4%) | ||
| N (TNM) N (%) | NS | ||||
| N0 | 17 (27.4%) | 8 (32%) | 9 (24.3%) | ||
| N1 | 7 (11.3%) | 2 (8%) | 5 (13.5%) | ||
| N2a | 3 (4.8%) | 2 (8%) | 1 (2.7%) | ||
| N2b | 9 (14.5%) | 5 (20%) | 4 (10.8%) | ||
| N2c | 19 (30.6%) | 6 (24%) | 13 (35.1%) | ||
| N3 | 7 (11.3%) | 2 (8%) | 5 (13.5%) | ||
| Stage | NS | ||||
| I | 1 (1.6%) | 1 (4%) | 0 (0%) | ||
| II | 3 (4.8%) | 0 (0%) | 3 (8.1%) | ||
| III | 18 (29%) | 8 (32%) | 10 (27%) | ||
| IV | 40 (64.5%) | 16 (64%) | 24 (64.9%) | ||
| Tumor Site N (%) | NS | ||||
| Oral cavity | 5 (8.1%) | 3 (12%) | 2 (5.4%) | ||
| Oropharynx | 42 (67.7%) | 17 (68%) | 25 (67.6%) | ||
| Hypopharynx | 4 (6.5%) | 0 (0%) | 4 (10.8%) | ||
| Larynx | 11 (17.7%) | 5 (20%) | 6 (16.2%) | ||
| Metastatic Relapse N (%) | 0.025 | ||||
| No | 49 (80.3%) | 24 (96%) | 25 (69.4%) | ||
| Yes | 12 (19.7%) | 1 (4%) | 11 (30.6%) | ||
| NA | 1 | 0 | 1 | ||
| HPV N (%) | 0.002 | ||||
| HPV- | 27 (43.5%) | 5 (20%) | 22 (59.5%) | ||
| HPV+ | 35 (56.5%) | 20 (80%) | 15 (40.5%) | ||
| Anisokaryosis | <0.001 | ||||
| Mild | 28 (45.2%) | 20 (80%) | 8 (21.6%) | ||
| Moderate | 19 (30.6%) | 5 (20%) | 14 (37.8%) | ||
| Marked | 15 (24.2%) | 0 (0%) | 15 (40.5%) | ||
| CENP-A H-score | 0.003 | ||||
| Median (IQR) | 1.6 (1.2–2.7) | 1.8 (1.6–2.7) | 1.5 (0.7–1.8) | ||
| Local Disease Control at 2 years | <0.001 | ||||
| Yes | 38 (61.3%) | 24 (96%) | 14 (37.8%) | ||
| No | 24 (38.7%) | 1 (4%) | 23 (62.2%) | ||
| Univariate Analysis | Multivariate Analysis | ||||||
|---|---|---|---|---|---|---|---|
| Factors | Category | OR | 95% IC | p -Value | OR | 95% IC | p -Value |
| T (TNM) | NS | ||||||
| T1/T2 | 1 | ||||||
| T3/T4 | 0.56 | [0.1; 1.9] | |||||
| N (TNM) | NS | ||||||
| N0 | 1 | ||||||
| N+ | 0.57 | [0.2; 1.8] | |||||
| Stage | |||||||
| I/II/III | 1 | NS | |||||
| IVa or IVb | 0.46 | [0.1; 1.4] | |||||
| HPV status | 0.02 | ||||||
| HPV- | 1 | ||||||
| HPV+ | 3.61 | [1.3; 11.0] | |||||
| Anisokaryosis | |||||||
| Mild | 1 | <0.001 | 1 | 0.03 | |||
| Moderate | 0.29 | [0.06; 1.1] | 0.74 | [0.14; 3.9] | |||
| Marked | 0.03 | [0; 0.1] | 0.11 | [0.01; 0.7] | |||
| CENP-A H-score | |||||||
| 3.1 | [1.5; 7.2] | 0.005 | |||||
| CENP-A pattern | 0.001 | 0.002 | |||||
| non-C | 1 | 1 | |||||
| C | 39.4 | [7.1; 744.0] | 17.6 | [2.6; 362.8] | |||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Verrelle, P.; Meseure, D.; Berger, F.; Forest, A.; Leclère, R.; Nicolas, A.; Fortas, E.; Sastre-Garau, X.; Lae, M.; Boudjemaa, S.; et al. CENP-A Subnuclear Localization Pattern as Marker Predicting Curability by Chemoradiation Therapy for Locally Advanced Head and Neck Cancer Patients. Cancers 2021, 13, 3928. https://doi.org/10.3390/cancers13163928
Verrelle P, Meseure D, Berger F, Forest A, Leclère R, Nicolas A, Fortas E, Sastre-Garau X, Lae M, Boudjemaa S, et al. CENP-A Subnuclear Localization Pattern as Marker Predicting Curability by Chemoradiation Therapy for Locally Advanced Head and Neck Cancer Patients. Cancers. 2021; 13(16):3928. https://doi.org/10.3390/cancers13163928
Chicago/Turabian StyleVerrelle, Pierre, Didier Meseure, Frédérique Berger, Audrey Forest, Renaud Leclère, André Nicolas, Emilie Fortas, Xavier Sastre-Garau, Marick Lae, Sabah Boudjemaa, and et al. 2021. "CENP-A Subnuclear Localization Pattern as Marker Predicting Curability by Chemoradiation Therapy for Locally Advanced Head and Neck Cancer Patients" Cancers 13, no. 16: 3928. https://doi.org/10.3390/cancers13163928








